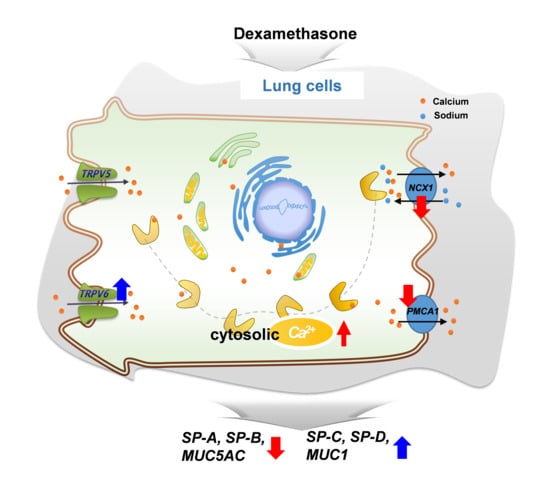
Dexamethasone Treatment Increases the Intracellular Calcium Level Through TRPV6 in A549 Cells
Abstract
:1. Introduction
2. Results
3. Discussion
4. Methods
4.1. Cell Culture and Treatments
4.2. Intracellular Calcium Response
4.2.1. Rhod-4 Calcium Response Assay
4.2.2. Calcium Response Assay with pGP-CMV-GCaMP6f and CMV-R-GECO1.2 Transfection
4.3. Western Blot Analysis
4.4. Preparation of RNA and Real-Time PCR
4.5. Data Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Yang, H.; Zhang, O.; He, J.; Lu, W. Regulation of calcium signaling in lung cancer. J. Thorac. Dis. 2010, 2, 52–56. [Google Scholar] [PubMed]
- Monteith, G.R.; Davis, F.M.; Roberts-Thomson, S.J. Calcium channels and pumps in cancer: Changes and consequences. J. Biol. Chem. 2012, 287, 31666–31673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, J.Y.; Ahn, C.; Kang, H.Y.; Jeung, E.B. Inhibition of mucin secretion via glucocorticoid-induced regulation of calcium-related proteins in mouse lung. Am. J. Physiol. Lung Cell. Mol. Physiol. 2018, 314, L956–L966. [Google Scholar] [CrossRef] [PubMed]
- Lehen’kyi, V.; Raphaël, M.; Prevarskaya, N. The role of the TRPV6 channel in cancer. J. Physiol. 2012, 590, 1369–1376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raphaël, M.; Lehen’kyi, V.; Vandenberghe, M.; Beck, B.; Khalimonchyk, S.; Vanden Abeele, F.; Farsetti, L.; Germain, E.; Bokhobza, A.; Mihalache, A.; et al. TRPV6 calcium channel translocates to the plasma membrane via Orai1-mediated mechanism and controls cancer cell survival. Proc. Natl. Acad. Sci. USA 2014, 111, E3870–E3879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rucka, Z.; Vanhara, P.; Koutna, I.; Tesarova, L.; Potesilova, M.; Stejskal, S.; Simara, P.; Dolezel, J.; Zvonicek, V.; Coufal, O.; et al. Differential effects of insulin and dexamethasone on pulmonary surfactant-associated genes and proteins in A549 and H441 cells and lung tissue. Int. J. Mol. Med. 2013, 32, 211–218. [Google Scholar] [CrossRef] [Green Version]
- Umeda, Y.; Hasegawa, Y.; Otsuka, M.; Ariki, S.; Takamiya, R.; Saito, A.; Uehara, Y.; Saijo, H.; Kuronuma, K.; Chiba, H.; et al. Surfactant protein D inhibits activation of non-small cell lung cancer-associated mutant EGFR and affects clinical outcomes of patients. Oncogene 2017, 36, 6432–6445. [Google Scholar] [CrossRef]
- Mitsuhashi, A.; Goto, H.; Kuramoto, T.; Tabata, S.; Yukishige, S.; Abe, S.; Hanibuchi, M.; Kakiuchi, S.; Saijo, A.; Aono, Y.; et al. Surfactant protein a suppresses lung cancer progression by regulating the polarization of tumor-associated macrophages. Am. J. Pathol. 2013, 182, 1843–1853. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, O.; Takahashi, H.; Hirasawa, M.; Chiba, H.; Shiratori, M.; Kuroki, Y.; Abe, S. Surfactant protein gene expressions for detection of lung carcinoma cells in peripheral blood. Respir. Med. 2005, 99, 1164–1174. [Google Scholar] [CrossRef] [Green Version]
- Creuwels, L.A.; Van Golde, L.M.; Haagsman, H.P. The pulmonary surfactant system: Biochemical and clinical aspects. Lung 1997, 175, 1–39. [Google Scholar] [CrossRef] [Green Version]
- Imai, M.; Hwang, H.Y.; Norris, J.S.; Tomlinson, S. The effect of dexamethasone on human mucin 1 expression and antibody-dependent complement sensitivity in a prostate cancer cell line in vitro and in vivo. Immunology 2004, 111, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Watson, A.M.; Williamson, C.D.; Rahimi, M.; Liang, C.; Colberg-Poley, A.M.; Rose, M.C. Glucocorticoid receptor and histone deacetylase—2 mediate dexamethasone-induced repression of MUC5AC gene expression. Am. J. Respir. Cell. Mol. Biol. 2012, 47, 637–644. [Google Scholar] [CrossRef] [Green Version]
- Forstner, J.F.; Forstner, G.G. Effects of calcium on intestinal mucin: Implications for cystic fibrosis. Pediatr. Res. 1976, 10, 609–613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Helline, B.P.; Tisdale, A.S.; Danjo, Y.; Spurr-Michaud, S.J.; Argüeso, P.; Gipson, I.K. The role of calcium in mucin packaging within goblet cells. Exp. Eye Res. 2003, 77, 69–75. [Google Scholar]
- Helassa, N.; Podor, B.; Fine, A.; Török, K. Design and mechanistic insight into ultrafast calcium indicators for monitoring intracellular calcium dynamics. Sci. Rep. 2016, 6, 38276. [Google Scholar] [CrossRef] [Green Version]
- Sadakane, O.; Masamizu, Y.; Watakabe, A.; Terada, S.; Ohtsuka, M.; Takaji, M.; Mizukami, H.; Ozawa, K.; Kawasaki, H.; Matsuzaki, M.; et al. Long-term two-photon calcium imaging of neuronal populations with subcellular resolution in adult non-human primates. Cell Rep. 2015, 13, 1989–1999. [Google Scholar] [CrossRef] [Green Version]
- Lock, J.T.; Parker, I.; Smith, I.F. A comparison of fluorescent Ca2+ indicators for imaging local Ca2+ signals in cultured cells. Cell Calcium 2015, 58, 638–648. [Google Scholar] [CrossRef] [Green Version]
- Gennari, C. Differential effect of glucocorticoids on calcium absorption and bone mass. Br. J. Rheumatol. 1993, 2, 11–14. [Google Scholar] [CrossRef]
- An, B.S.; Choi, K.C.; Hong, E.J.; Jung, Y.W.; Manabe, N.; Jeung, E.B. Differential transcriptional and translational regulations of calbindin-D9k by steroid hormones and their receptors in the uterus of immature mice. J. Reprod. Dev. 2004, 50, 445–453. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.; Lee, D.; Ahn, C.; Kang, H.Y.; An, B.S.; Seong, Y.H.; Jeung, E.B. Effects of estrogen on esophageal function through regulation of Ca2+-related proteins. J. Gastroenterol. 2017, 52, 929–939. [Google Scholar] [CrossRef]
- Kim, M.H.; Lee, G.S.; Jung, E.M.; Choi, K.C.; Jeung, E.B. The negative effect of dexamethasone on calcium-processing gene expressions is associated with a glucocorticoid-induced calcium-absorbing disorder. Life Sci. 2009, 85, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Lovell, P.V.; King, J.T.; McCobb, D.P. Acute modulation of adrenal chromaffin cell BK channel gating and cell excitability by glucocorticoids. J. Neurophysiol. 2004, 91, 561–570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, M.H.; So, E.C.; Liu, Y.C.; Wu, S.N. Glucocorticoids stimulate the activity of large-conductance Ca2+ -activated K+ channels in pituitary GH3 and AtT-20 cells via a non-genomic mechanism. Steroids 2006, 71, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Quesnell, R.R.; Han, X.; Schultz, B.D. Glucocorticoids stimulate ENaC upregulation in bovine mammary epithelium. Am. J. Physiol. Cell Physiol. 2007, 292, C1739–C1745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, H.T.; Lo, Y.C.; Huang, Y.M.; Tseng, Y.T.; Wu, S.N. Important modifications by sugammadex, a modified γ-cyclodextrin, of ion currents in differentiated NSC-34 neuronal cells. BMC Neurosci. 2017, 18, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariana, M.; Feiteiro, J.; Cairrao, E.; Verde, I. Mifepristone is a vasodilator due to the inhibition of smooth muscle cells L-Type Ca2+ channels. Reprod. Sci. 2016, 23, 723–730. [Google Scholar] [CrossRef]
- Lobaccaro-Henri, C.; Descomps, B.; Thaler-Dao, H. RU 38486 inhibits intracellular calcium mobilization and PGI2 release from human myometrium: Mechanisms of action. J. Steroid Biochem. Mol. Biol. 1996, 59, 63–73. [Google Scholar] [CrossRef]
- Serres, C.; Yang, J.; Jouannet, P. RU486 and calcium fluxes in human spermatozoa. Biochem. Biophys. Res. Commun. 1994, 204, 1009–1015. [Google Scholar] [CrossRef]
- Lee, B.M.; Lee, G.S.; Jung, E.M.; Choi, K.C.; Jeung, E.B. Uterine and placental expression of TRPV6 gene is regulated via progesterone receptor- or estrogen receptor-mediated pathways during pregnancy in rodents. Reprod. Biol. Endocrinol. 2009, 7, 49. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Liu, L.; Matsuda, T.; Zhao, Y.; Rebane, A.; Drobizhev, M.; Chang, Y.F.; Araki, S.; Arai, Y.; March, K.; et al. Improved orange and red Ca2+ indicators and photophysical considerations for optogenetic applications. ACS Chem. Neurosci. 2013, 4, 963–972. [Google Scholar] [CrossRef] [Green Version]
- Xie, W.; Santulli, G.; Guo, X.; Gao, M.; Chen, B.X.; Marks, A.R. Imaging atrial arrhythmic intracellular calcium in intact heart. J. Mol. Cell. Cardiol. 2013, 64, 120–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liley, H.G.; White, R.T.; Benson, B.J.; Ballard, P.L. Glucocorticoids both stimulate and inhibit production of pulmonary surfactant protein A in fetal human lung. Proc. Natl. Acad. Sci. USA 1988, 85, 9096–9100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariencheck, W.; Crouch, E. Modulation of surfactant protein D expression by glucocorticoids in fetal rat lung. Am. J. Respir. Cell Mol. Biol. 1994, 10, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, C.G.; Revak, S.D. Pulmonary surfactant protein B (SP-B): Structure-function relationships. Science 1991, 254, 566–568. [Google Scholar] [CrossRef]
- Milara, J.; Peiró, T.; Armengot, M.; Frias, S.; Morell, A.; Serrano, A.; Cortijo, J. Mucin 1 downregulation associates with corticosteroid resistance in chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2015, 135, 470–476. [Google Scholar] [CrossRef]
- Treon, S.P.; Mollick, J.A.; Urashima, M.; Teoh, G.; Chauhan, D.; Ogata, A.; Raje, N.; Hilgers, J.H.; Nadler, L.; Belch, A.R.; et al. Muc-1 core protein is expressed on multiple myeloma cells and is induced by dexamethasone. Blood 1999, 93, 1287–1298. [Google Scholar] [CrossRef]
- Kai, H.; Yoshitake, K.; Hisatsune, A.; Kido, T.; Isohama, Y.; Takahama, K.; Miyata, T. Dexamethasone suppresses mucus production and MUC-2 and MUC-5AC gene expression by NCI-H292 cells. Am. J. Physiol. 1996, 271, L484–L488. [Google Scholar] [CrossRef]
- Han, S.; Mallampalli, R.K. The role of surfactant in lung disease and host defense against pulmonary infections. Ann. Am. Thorac. Soc. 2015, 12, 765–774. [Google Scholar] [CrossRef] [Green Version]
- Peters, A.A.; Simpson, P.T.; Bassett, J.J.; Lee, J.M.; Da Silva, L.; Reid, L.E.; Song, S.; Parat, M.O.; Lakhani, S.R.; Kenny, P.A.; et al. Calcium channel TRPV6 as a potential therapeutic target in estrogen receptor-negative breast cancer. Mol. Cancer Ther. 2012, 11, 2158–2168. [Google Scholar] [CrossRef] [Green Version]
- Mitrovic, S.; Nogueira, C.; Cantero-Recasens, G.; Kiefer, K.; Fernández-Fernández, J.M.; Popoff, J.F.; Casano, L.; Bard, F.A.; Gomez, R.; Valverde, M.A.; et al. TRPM5-mediated calcium uptake regulates mucin secretion from human colon goblet cells. eLife 2013, 2, e00658. [Google Scholar] [CrossRef]







| Gene | Primer Sequence (5′→3′) | Accession Number |
|---|---|---|
| TRPV6 | F: GGAGCTAGGCCACTTCTACG | NM_018646.6 |
| R: ATGGCAATGAGGAGGTTGAG | ||
| NCX1 | F: GACCTCGGTCCTAGCACCAT | NM_001112800.2 |
| R: ACACCAGGAGATATGACAGACAA | ||
| PMCA1 | F: GCTGGAGGTGAAGAGGAA | NM_001001323.2 |
| R: GCACTGCGACCACTAAAA | ||
| SFTPA1 | F: TTTGATGCCATTCAGGAGGC | NM_001093770.3 |
| R: TCGGTACCAGTTGGTGTAGT | ||
| SFTPB | F: AAGTTCCTGGAGCAGGAGTG | NM_000542.5 |
| R: AGAGGAATGGGGAATTGCTG | ||
| SFTPC | F: GGCACCTGCTGCTACATCAT | NM_001172357.2 |
| R: CCAGCTTAGACGTAGGCACT | ||
| SFTPD | F: CTTCTCTGAGGCAGCAGGTT | NM_003019.5 |
| R: CTGTGCCTCCGTAAATGGTT | ||
| MUC1 | F: ATCTCATTGCCTTGGCTGTC | NM_001018016.3 |
| R: TAGGGGCTACGATCGGTACT | ||
| MUC5AC | F: AACCAGTCGACCTGTGCTGT | NM_001304359.2 |
| R: TCGAGCGAGTACATGGAAGA | ||
| GAPDH | F: AAGGTCATCCCTGAGCTGAA | NM_001256799.3 |
| R: GGGAGCCAAAAGGGTCATCA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeon, B.-H.; Yoo, Y.-M.; Jung, E.-M.; Jeung, E.-B. Dexamethasone Treatment Increases the Intracellular Calcium Level Through TRPV6 in A549 Cells. Int. J. Mol. Sci. 2020, 21, 1050. https://doi.org/10.3390/ijms21031050
Jeon B-H, Yoo Y-M, Jung E-M, Jeung E-B. Dexamethasone Treatment Increases the Intracellular Calcium Level Through TRPV6 in A549 Cells. International Journal of Molecular Sciences. 2020; 21(3):1050. https://doi.org/10.3390/ijms21031050
Chicago/Turabian StyleJeon, Bo-Hui, Yeong-Min Yoo, Eui-Man Jung, and Eui-Bae Jeung. 2020. "Dexamethasone Treatment Increases the Intracellular Calcium Level Through TRPV6 in A549 Cells" International Journal of Molecular Sciences 21, no. 3: 1050. https://doi.org/10.3390/ijms21031050