ABTS/PP Decolorization Assay of Antioxidant Capacity Reaction Pathways
Abstract
:1. Introduction
2. ABTS/PP Abundance Statistics
3. ABTS/PP Basic Chemistry
4. Reaction Stoichiometry
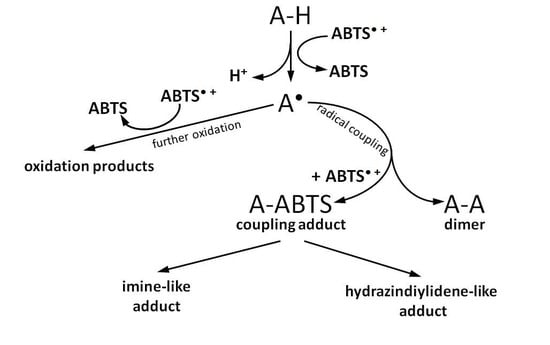
5. Reaction Pathways with Adducts Formation
- (1)
- As ABTS•+ was generated in the ABTS/laccase system, but not ABTS/PP, one could argue that it could hypothetically influence the observed ABTS•+-antioxidant reaction and subsequent ABTS•+ depletion. There are many precedents when certain antioxidants reveal different antioxidant capacities against the same model radical but a different radical-generating system, for example in the ABTS/metmyoglobin/H2O2 assay, the TEAC of quercetin and cyanidin was 4.72 and 4.4 [76,77], whereas in the ABTS/PP assay it was 3.03 and 2.48 [1], respectively. Additionally, laccase can act as an oxidizing agent alone, not to mention that the combination of laccase with ABTS or other mediators shows a higher oxidation ability than laccase or ABTS•+ separately [78,79,80,81,82]. Nevertheless, these concerns seem to be unfounded, as the same researchers observed these ABTS•+ degradation products again in their next report when applying the ABTS/PP system [83].
- (2)
- The time of reaction was 1–2 h, which is much more than the 4–6 min in the original TEAC assay, and thus there was much more time for ABTS•+ cleavage.
- (3)
- The fact that polyphenol was added “dropwise” to the concentrated ABTS/laccase mixture, which also differs from the original design of ABTS/PP assay, did not seem to matter either, as generally the same situation C(ABTS•+) > C(antioxidant) at any given time was reproduced.
- (1)
- The principal possibility of ABTS•+ degradation and ABTS-antioxidant adduct formation was demonstrated for the first time in the ABTS/PP assay.
- (2)
- The extent to which this degradation and adduct formation influenced the final TEAC was not obvious due to the modified experiment design and no quantitative estimations.
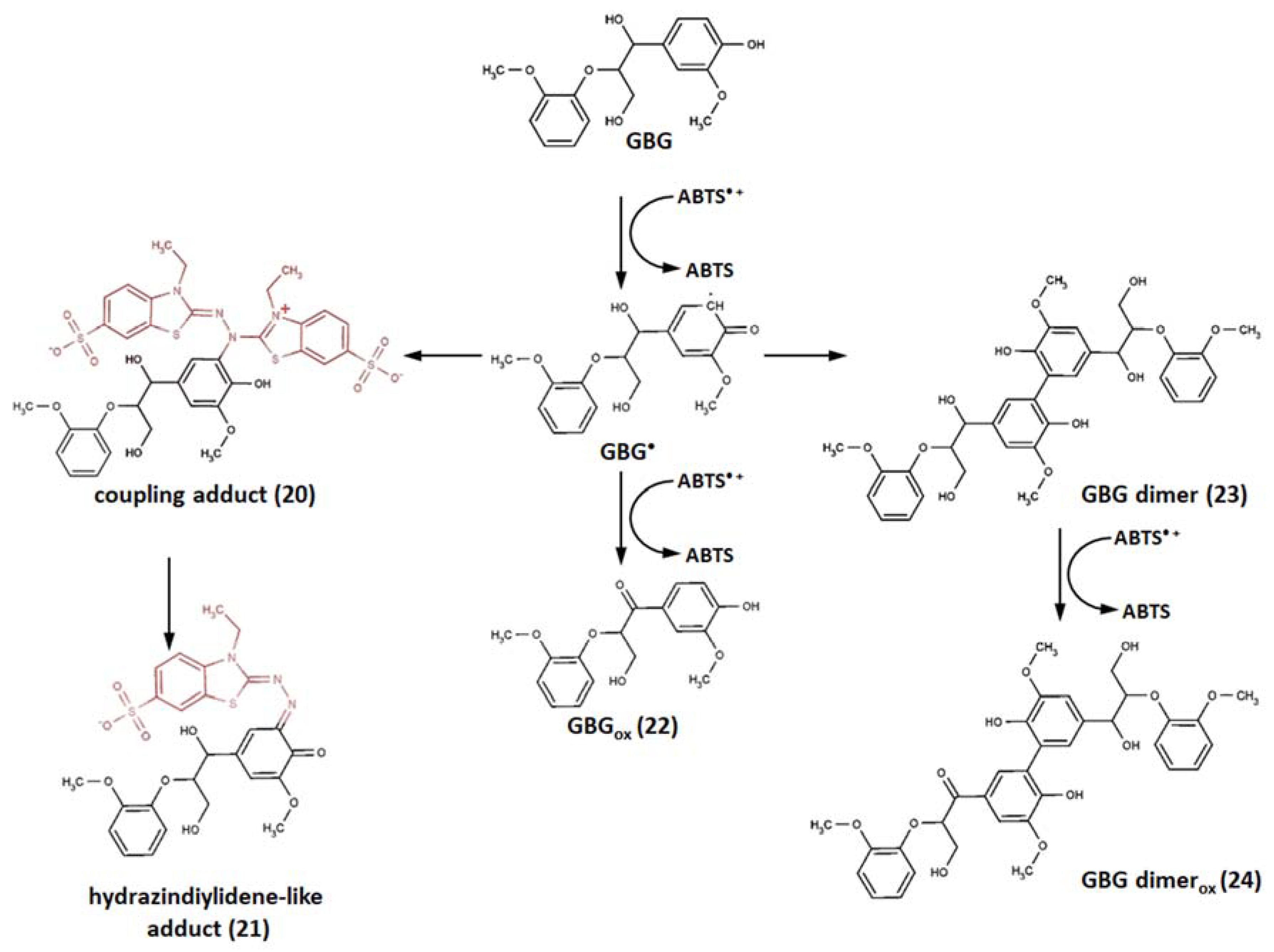
6. Reaction Pathways without Adduct Formation
7. Conclusions
- (1)
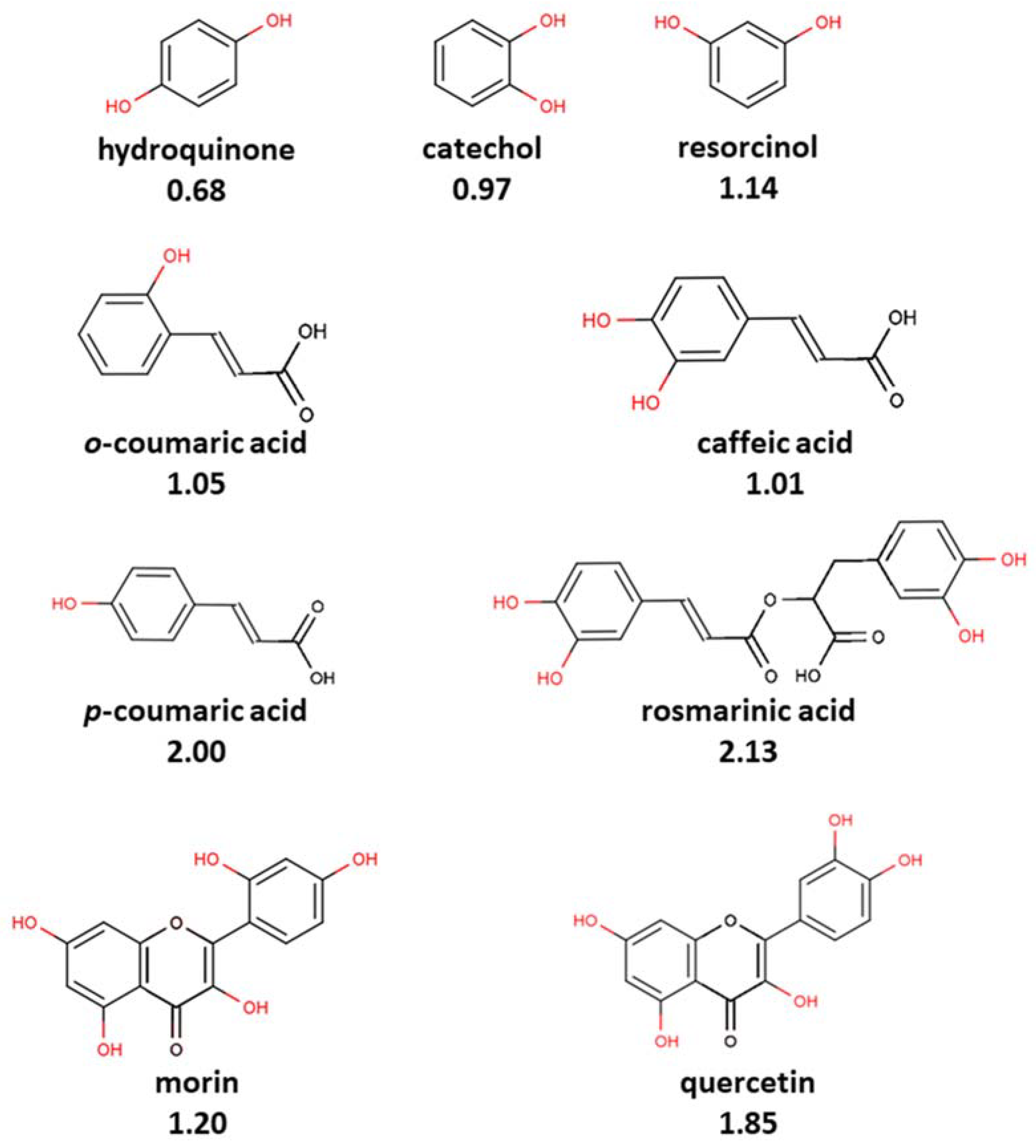
- Some antioxidants can form adducts with ABTS•+, whereas others can undergo oxidation without coupling with ABTS•+. Thus, coupling with ABTS•+ is a specific reaction for certain groups of antioxidants, apparently at least of phenolic nature. Establishing the structural features that determine the direction of antioxidant interaction with ABTS•+ is important for future understanding and interpretation of antioxidant capacity measurements.
- (2)
- Adduct-free oxidation pathways are substrate-specific and can be influenced both by ABTS•+ radical specific features or by the radical-initiator system. However, they seem to be reliably consistent with the results obtained when other oxidants are applied instead of ABTS•+.
- (3)
- The coupling reaction can occur with phenolic compounds, and the coupling adduct can be the principal product as well as undergoing further oxidative degradation, which might depend on the antioxidant/ABTS•+ ratio and ABTS•+ generation methodology.
- (4)
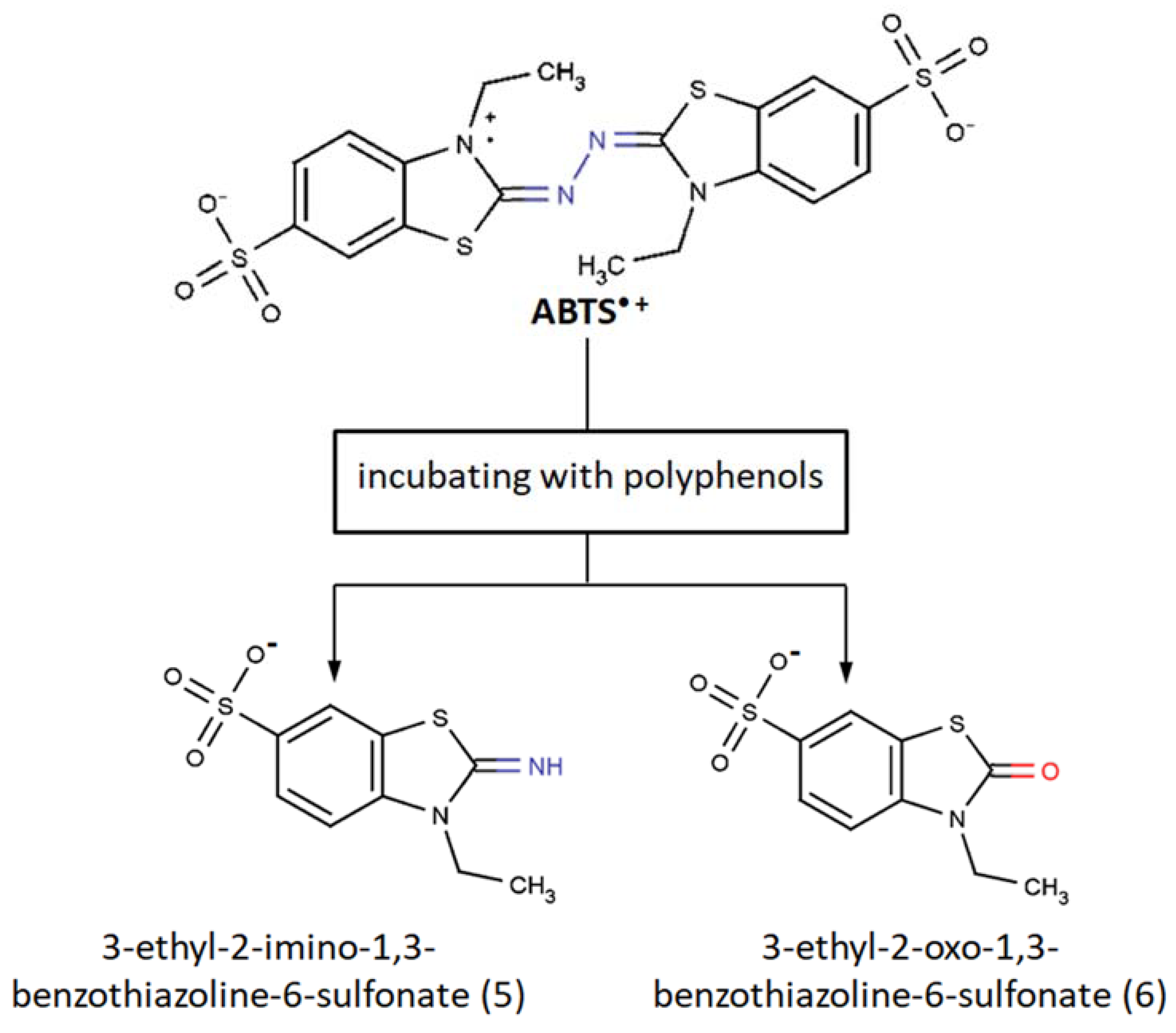
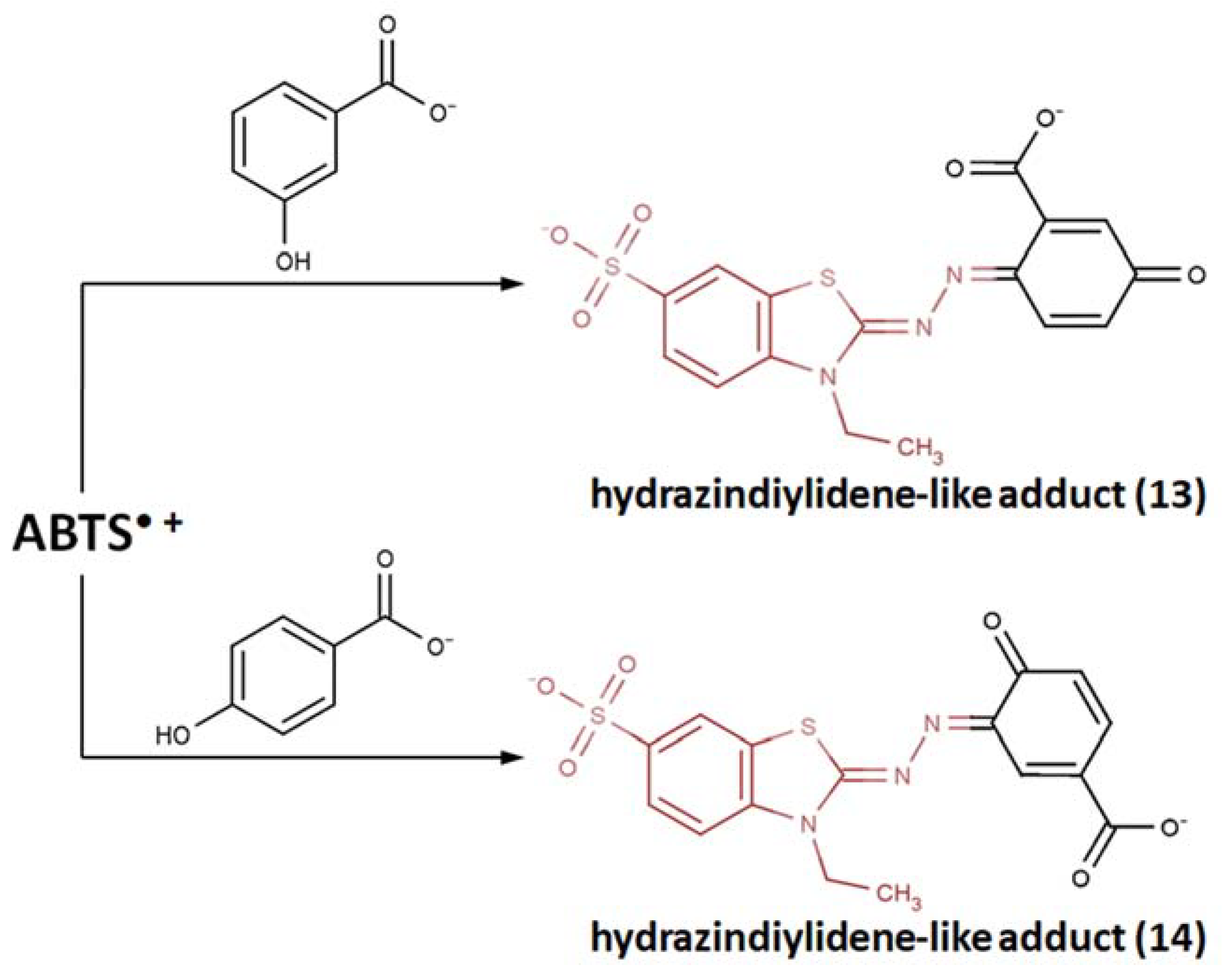
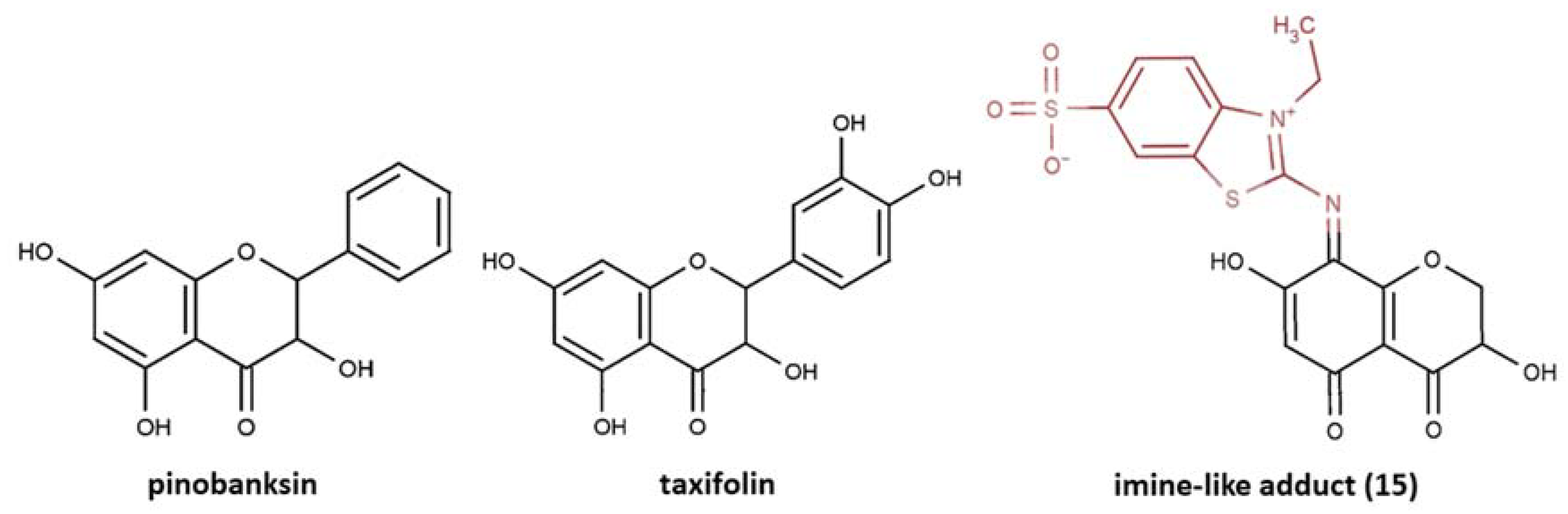
- Further oxidative degradation of the coupling product results from the oxidative cleavage between the two nitrogen-linked benzothiazole rings. This leads to hydrazindyilidene-like and imine-like adduct formation. 3-Ethyl-2-oxo-1,3-benzothiazoline-6-sulfonate can presumably witness a hydrazindyilidene-like adduct formation pathway, whereas 3-ethyl-2-imino-1,3-benzothiazoline-6-sulfonate testifies to the imine-like adduct formation pathway.
- (5)
- The extent that the coupling reaction contributes to the reaction between antioxidants and ABTS•+ (e.g., kinetics and stoichiometry) is unclear due to the lack of quantitative estimation of their formation, and sometimes this may be quite considerable.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt |
| AFMK | N1-acetyl-N2-formyl-5-methoxykynuramine |
| c3OHM | cyclic 3-hydroxymelatonin |
| CEP | concerted electron–proton transfer |
| COSY | correlation spectroscopy |
| CUPRAC | cupric reducing antioxidant capacity |
| DAD | diode-array detection |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| ESI-HRMS | high resolution electrospray ionization mass spectrometry |
| ESI-MS | electrospray ionization mass spectrometry |
| ET-PT | electron transfer–proton transfer |
| FRAP | ferric reducing antioxidant power |
| FKBP | fujimycin binding protein |
| GBG | guaiacylglycerol-β-guaiacyl ether |
| GBGox | oxidation products of guaiacylglycerol-β-guaiacyl ether |
| GSH | glutathione |
| GSSG | glutathione disulfide |
| HAT | hydrogen atom transfer |
| HMBC | heteronuclear multiple bond correlation |
| HSQC | heteronuclear single quantum coherence |
| HPLC-ECD | high performance liquid chromatography, electrochemical detection |
| HPLC-UV | high performance liquid chromatography, ultraviolet detection |
| IC50 | concentration which leads to 50% inhibition |
| IR | infrared spectroscopy |
| MS-MS | tandem mass spectrometry |
| NI | negative mode in electrospray ionization mass spectrometry |
| NMR | nuclear magnetic resonance |
| ORAC | oxygen radical absorbance capacity |
| PBS | phosphate buffer solution |
| PDA | photodiode array |
| PI | positive mode in electrospray ionization mass spectrometry |
| PP | potassium persulfate |
| QTOF-MS | quadrupole time of flight mass spectrometer |
| RP-UHPLC | reversed-phase ultrahigh-performance liquid chromatography |
| SET | single electron transfer |
| SIMS | secondary ion mass spectrometry |
| SPLET | sequential proton loss electron transfer |
| TEAC | trolox equivalent antioxidant capacity |
| TLC | thin layer chromatography |
| UV-VIS | ultraviolet–visible spectroscopy |
References
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Miller, N.J.; Rice-Evans, C.; Davies, M.J.; Gopinathan, V.; Milner, A. A Novel Method for Measuring Antioxidant Capacity and its Application to Monitoring the Antioxidant Status in Premature Neonates. Clin. Sci. 1993, 84, 407–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cano, A.; Arnao, M.B. ABTS/TEAC (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)/Trolox®-Equivalent Antioxidant Capacity) radical scavenging mixed-mode assay. In Measurement of Antioxidant Activity & Capacity; John Wiley & Sons, Ltd.: Chichester, UK, 2017; pp. 117–139. ISBN 9781119135388. [Google Scholar]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays. J. Funct. Foods 2015, 14, 111–125. [Google Scholar] [CrossRef]
- Apak, R. Current Issues in Antioxidant Measurement. J. Agric. Food Chem. 2019, 67, 9187–9202. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods 2015, 18, 757. [Google Scholar] [CrossRef]
- López-Alarcón, C.; Denicola, A. Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal. Chim. Acta 2013, 763, 1–10. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 2. Hydrogen Atom Transfer (HAT)-Based, Mixed-Mode (Electron Transfer (ET)/HAT), and Lipid Peroxidation Assays. J. Agric. Food Chem. 2016, 64, 1028–1045. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- Niki, E. Assessment of antioxidant capacity in vitro and in vivo. Free Radic. Biol. Med. 2010, 49, 503–515. [Google Scholar] [CrossRef]
- Pellegrini, N.; Vitaglione, P.; Granato, D.; Fogliano, V. Twenty-five years of total antioxidant capacity measurement of foods and biological fluids: Merits and limitations. J. Sci. Food Agric. 2019. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Pratt, D.A. Methods for determining the efficacy of radical-trapping antioxidants. Free Radic. Biol. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Ingold, K.U.; Pratt, D.A. Advances in Radical-Trapping Antioxidant Chemistry in the 21st Century: A Kinetics and Mechanisms Perspective. Chem. Rev. 2014, 114, 9022–9046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apak, R.; Capanoglu, E.; Shahidi, F. Measurement of Antioxidant Activity & Capacity. Recent Trends and Applications; Apak, R., Capanoglu, E., Shahidi, F., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2018; ISBN 9781119135388. [Google Scholar]
- Erel, O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin. Biochem. 2004, 37, 277. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Takamura, H.; Matoba, T.; Terao, J. Hplc method for evaluation of the free radical-scavenging activity of foods by using 1,1-diphenyl-2-picrylhydrazyl. Biosci. Biotechnol. Biochem. 1998, 62, 1201–1204. [Google Scholar] [CrossRef]
- Sanchez-Moreno, C.; Larrauri, J.A.; Saura-Calixto, F.A. A procedure to measure the antiradical efficiency of polyphenols. J. Sci. Food Agric. 1998, 76, 270. [Google Scholar] [CrossRef]
- Sharma, O.P.; Bhat, T.K. DPPH antioxidant assay revisited. Food Chem. 2009, 113, 1202–1205. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of ‘antioxidant power’: The FRAP assay. Anal. Biochem. 1996, 239, 70. [Google Scholar] [CrossRef] [Green Version]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzym. 1999, 299, 15. [Google Scholar]
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J. Agric. Food Chem. 2000, 48, 3396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzym. 1999, 299, 152. [Google Scholar]
- Ainsworth, E.A.; Gillespie, K.M. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Georgé, S.; Brat, P.; Alter, P.; Amiot, M.J. Rapid determination of polyphenols and vitamin C in plant-derived products. J. Agric. Food Chem. 2005, 53, 1370–1373. [Google Scholar] [CrossRef] [PubMed]
- Medina-Remón, A.; Barrionuevo-González, A.; Zamora-Ros, R.; Andres-Lacueva, C.; Estruch, R.; Martínez-González, M.-Á.; Diez-Espino, J.; Lamuela-Raventos, R.M. Rapid Folin–Ciocalteu method using microtiter 96-well plate cartridges for solid phase extraction to assess urinary total phenolic compounds, as a biomarker of total polyphenols intake. Anal. Chim. Acta 2009, 634, 54–60. [Google Scholar] [CrossRef]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Radic. Biol. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef] [Green Version]
- Cao, G.; Verdon, C.P.; Wu, A.H.; Wang, H.; Prior, R.L. Automated oxygen radical absorbance capacity assay using the COBAS FARA II. Clin. Chem. 1995, 41, 1738. [Google Scholar] [CrossRef]
- Dávalos, A.; Gómez-Cordovés, C.; Bartolomé, B. Extending Applicability of the Oxygen Radical Absorbance Capacity (ORAC−Fluorescein) Assay. J. Agric. Food Chem. 2004, 52, 48–54. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Prior, R.L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J. Agric. Food Chem. 2001. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Deemer, E.K. Development and validation of oxygen radical absorbance capacity assay for lipophilic antioxidants using randomly methylated β-cyclodextrin as the solubility enhancer. J. Agric. Food Chem. 2002. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E. Novel Total Antioxidant Capacity Index for Dietary Polyphenols and Vitamins C and E, Using Their Cupric Ion Reducing Capability in the Presence of Neocuproine: CUPRAC Method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E.; Altun, M. Total antioxidant capacity assay of human serum using copper(II)-neocuproine as chromogenic oxidant: The CUPRAC method. Free Radic. Res. 2005, 39, 949–961. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Güçlü, K.; Demirata, B.; Özyürek, M.; Çelik, S.E.; Bektaşoǧlu, B.; Berker, K.I.; Özyurt, D. Comparative evaluation of various total antioxidant capacity assays applied to phenolic compounds with the CUPRAC assay. Molecules 2007, 12, 1496–1547. [Google Scholar] [CrossRef] [Green Version]
- Arnao, M.B. Some methodological problems in the determination of antioxidant activity using chromogen radicals: A practical case. Trends Food Sci. Technol. 2000, 11, 419. [Google Scholar] [CrossRef]
- Dong, J.-W.; Cai, L.; Xing, Y.; Yu, J.; Ding, Z.-T. Re-evaluation of ABTS•+ Assay for Total Antioxidant Capacity of Natural Products. Nat. Prod. Commun. 2015. [Google Scholar] [CrossRef] [Green Version]
- Dawidowicz, A.L.; Olszowy, M. The importance of solvent type in estimating antioxidant properties of phenolic compounds by ABTS assay. Eur. Food Res. Technol. 2013, 236, 1099–1105. [Google Scholar] [CrossRef] [Green Version]
- Solís-Oba, M.; Ugalde-Saldívar, V.M.; González, I.; Viniegra-González, G. An electrochemical-spectrophotometrical study of the oxidized forms of the mediator 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid) produced by immobilized laccase. J. Electroanal. Chem. 2005, 579, 59–66. [Google Scholar] [CrossRef]
- Venkatasubramanian, L.; Maruthamuthu, P. Kinetics and mechanism of formation and decay of 2,2′-azinobis-(3-ethylbenzothiazole-6-sulphonate) radical cation in aqueous solution by inorganic peroxides. Int. J. Chem. Kinet. 1989, 21, 399–421. [Google Scholar] [CrossRef]
- Scott, S.L.; Chen, W.J.; Bakac, A.; Espenson, J.H. Spectroscopic parameters, electrode potentials, acid ionization constants, and electron exchange rates of the 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonate) radicals and ions. J. Phys. Chem. 1993, 97, 6710–6714. [Google Scholar] [CrossRef]
- Childs, R.E.; Bardsley, W.G. The steady-state kinetics of peroxidase with 2,2′-azino-di-(3-ethyl-benzthiazoline-6-sulphonic acid) as chromogen. Biochem. J. 1975, 145, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Wolfenden, B.S.; Willson, R.L. Radical-cations as reference chromogens in kinetic studies of ono-electron transfer reactions: Pulse radiolysis studies of 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate). J. Chem. Soc. Perkin Trans. 2 1982, 805–812. [Google Scholar] [CrossRef]
- Cano, A.; Acosta, M.; Arnao, M.B. A method to measure antioxidant activity in organic media: Application to lipophilic vitamins. Redox Rep. 2000, 5, 365–370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henriquez, C.; Aliaga, C.; Lissi, E. Formation and decay of the ABTS derived radical cation: A comparison of different preparation procedures. Int. J. Chem. Kinet. 2002, 34, 659–665. [Google Scholar] [CrossRef]
- Maruthamuthu, P.; Venkatasubramanian, L.; Dharmalingam, P. A Fast Kinetic Study of Formation and Decay of 2,2′-Azinobis(3-ethylbenzothiazole-6-sulfonate) Radical Cation in Aqueous Solution. Bull. Chem. Soc. Jpn. 1987, 60, 1113–1117. [Google Scholar] [CrossRef]
- Gramss, G. Reappraising a controversy: Formation and role of the azodication (ABTS 2+ ) in the laccase-ABTS catalyzed breakdown of lignin. Fermentation 2017, 3, 27. [Google Scholar] [CrossRef]
- Mahuzier, G.; Kirkacharian, B.S.; Harfouche-Obeika, C. Microdosage colorimétrique de l’acide periodique par l’acide 2,2′-azino-di(3-ethylbenzothiazole-6-sulfonique). Anal. Chim. Acta 1975. [Google Scholar] [CrossRef]
- Majcherczyk, A.; Johannes, C.; Hüttermann, A. Oxidation of aromatic alcohols by laccase from Trametes versicolor mediated by the 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulphonic acid) cation radical and dication. Appl. Microbiol. Biotechnol. 1999, 51, 267–276. [Google Scholar] [CrossRef]
- Branchi, B.; Galli, C.; Gentili, P. Kinetics of oxidation of benzyl alcohols by the dication and radical cation of ABTS. Comparison with laccase-ABTS oxidations: An apparent paradox. Org. Biomol. Chem. 2005. [Google Scholar] [CrossRef]
- van den Berg, R.; Haenen, G.R.M.M.; van den Berg, H.; Bast, A. Applicability of an improved Trolox equivalent antioxidant capacity (TEAC) assay for evaluation of antioxidant capacity measurements of mixtures. Food Chem. 1999, 66, 511–517. [Google Scholar] [CrossRef]
- Aliaga, C.; Lissi, E.A. Reaction of 2,2′-azinobis 3-ethylbenzothiazoline-6-sulfonic acid (ABTS) derived radicals with hydroperoxides. Kinetics and mechanism. Int. J. Chem. Kinet. 1998, 30, 565–570. [Google Scholar] [CrossRef]
- Campos, A.M.; Lissi, E.A. Kinetics of the reaction between 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid (ABTS) derived radical cations and phenols. Int. J. Chem. Kinet. 1997, 29, 219. [Google Scholar] [CrossRef]
- Aliaga, C.; Lissi, E.A. Reactions of the radical cation derived from 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS ·+) with amino acids. Kinetics and mechanism. Can. J. Chem. 2000, 78, 1052–1059. [Google Scholar] [CrossRef]
- Klein, O.I.; Kulikova, N.A.; Filimonov, I.S.; Koroleva, O.V.; Konstantinov, A.I. Long-term kinetics study and quantitative characterization of the antioxidant capacities of humic and humic-like substances. J. Soils Sediments 2018, 18, 1355–1364. [Google Scholar] [CrossRef]
- Tian, X.; Schaich, K.M. Effects of molecular structure on kinetics and dynamics of Trolox Equivalent Antioxidant Capacity (TEAC) Assay with ABTS +•. J. Agric. Food Chem. 2013, 61, 5511–5519. [Google Scholar] [CrossRef] [PubMed]
- Ilyasov, I.R.; Beloborodov, V.L.; Selivanova, I.A. Three ABTS•+ radical cation-based approaches for the evaluation of antioxidant activity: Fast- and slow-reacting antioxidant behavior. Chem. Pap. 2018, 72, 1917–1925. [Google Scholar] [CrossRef]
- Rosen, J.; Than, N.N.; Koch, D.; Poeggeler, B.; Laatsch, H.; Hardeland, R. Interactions of melatonin and its metabolites with the ABTS cation radical: Extension of the radical scavenger cascade and formation of a novel class of oxidation products, C2-substituted 3-indolinones. J. Pineal Res. 2006, 41, 374–381. [Google Scholar] [CrossRef]
- Walker, R.B.; Everette, J.D. Comparative reaction rates of various antioxidants with ABTS radical cation. J. Agric. Food Chem. 2009, 57, 1156–1161. [Google Scholar] [CrossRef]
- Magalhaes, L.M.; Segundo, M.A.; Reis, S.; Lima, J.L.F.C. Methodological aspects about in vitro evaluation of antioxidant properties. Anal. Chim. Acta 2008, 613, 1. [Google Scholar] [CrossRef]
- Zheng, L.; Zhao, M.; Xiao, C.; Zhao, Q.; Su, G. Practical problems when using ABTS assay to assess the radical-scavenging activity of peptides: Importance of controlling reaction pH and time. Food Chem. 2016, 192, 288–294. [Google Scholar] [CrossRef]
- Takebayashi, J.; Tai, A.; Yamamoto, I. pH-dependent long-term radical scavenging activity of AA-2G and 6-octa-AA-2G against 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) radical cation. Biol. Pharm. Bull. 2003, 26, 1368–1370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arts, M.J.T.J.; Haenen, G.R.M.M.; Voss, H.P.; Bast, A. Antioxidant capacity of reaction products limits the applicability of the Trolox Equivalent Antioxidant Capacity (TEAC) assay. Food Chem. Toxicol. 2004, 42, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Arts, M.J.T.J.; Sebastiaan Dallinga, J.; Voss, H.P.; Haenen, G.R.M.M.; Bast, A. A new approach to assess the total antioxidant capacity using the TEAC assay. Food Chem. 2004, 88, 567–570. [Google Scholar] [CrossRef]
- Çelik, S.E.; Özyürek, M.; Güçlü, K.; Apak, R. Solvent effects on the antioxidant capacity of lipophilic and hydrophilic antioxidants measured by CUPRAC, ABTS/persulphate and FRAP methods. Talanta 2010, 81, 1300. [Google Scholar] [CrossRef] [PubMed]
- Błauz, A.; Pilaszek, T.; Grzelak, A.; Dragan, A.; Bartosz, G. Interaction between antioxidants in assays of total antioxidant capacity. Food Chem. Toxicol. 2008, 46, 2365–2368. [Google Scholar] [CrossRef] [PubMed]
- Giles, G.I.; Jacob, C. Reactive sulfur species: An emerging concept in oxidative stress. Biol. Chem. 2002, 383, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Van Den Berg, R.; Haenen, G.R.M.M.; Van Den Berg, H.; Van Der Vijgh, W.; Bast, A. The predictive value of the antioxidant capacity of structurally related flavonoids using the Trolox equivalent antioxidant capacity (TEAC) assay. Food Chem. 2000, 70, 391–395. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Nenadis, N.; Wang, L.F.; Tsimidou, M.; Zhang, H.Y. Estimation of scavenging activity of phenolic compounds using the ABTS.+ assay. J. Agric. Food Chem. 2004. [Google Scholar] [CrossRef]
- Henriquez, C.; Aliaga, C.; Lissi, E. Kinetics profiles in the reaction of ABTS derived radicals with simple phenols and polyphenols. J. Chil. Chem. Soc. 2004, 49, 65–67. [Google Scholar] [CrossRef]
- Mira, L.; Silva, M.; Rocha, R.; Manso, C.F. Measurement of relative antioxidant activity of compounds: A methodological note. Redox Rep. 1999, 4, 69–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sekher Pannala, A.; Chan, T.S.; O’Brien, P.J.; Rice-Evans, C.A. Flavonoid B-ring chemistry and antioxidant activity: Fast reaction kinetics. Biochem. Biophys. Res. Commun. 2001, 282, 1161–1168. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.M.; Wong, K.K.Y.; Hill, S.J.; Fernyhough, A. Isolation and the characterization of the degradation products of the mediator ABTS-derived radicals formed upon reaction with polyphenols. Biochem. Biophys. Res. Commun. 2006, 340, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Rice-evans, C.A.; Miller, N.J.; Bolwell, P.G.; Bramley, P.M.; Pridham, J.B. The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic. Res. 1995. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Munk, L.; Sitarz, A.K.; Kalyani, D.C.; Mikkelsen, J.D.; Meyer, A.S. Can laccases catalyze bond cleavage in lignin? Biotechnol. Adv. 2015, 33, 13–24. [Google Scholar] [CrossRef]
- Rodríguez Couto, S.; Toca Herrera, J.L. Industrial and biotechnological applications of laccases: A review. Biotechnol. Adv. 2006, 24, 500–513. [Google Scholar] [CrossRef]
- Ramalingam, B.; Sana, B.; Seayad, J.; Ghadessy, F.J.; Sullivan, M.B. Towards understanding of laccase-catalysed oxidative oligomerisation of dimeric lignin model compounds. RSC Adv. 2017, 7, 11951–11958. [Google Scholar] [CrossRef] [Green Version]
- Rittstieg, K.; Suurnakki, A.; Suortti, T.; Kruus, K.; Guebitz, G.M.; Buchert, J. Polymerization of Guaiacol and a Phenolic β-O-4-Substructure by Trametes hirsuta Laccase in the Presence of ABTS. Biotechnol. Prog. 2003, 19, 1505–1509. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M.G. Oxidation of non-phenolic substrates. FEBS Lett. 1990, 267, 99–102. [Google Scholar] [CrossRef] [Green Version]
- Osman, A.M.; Wong, K.K.Y.; Fernyhough, A. ABTS radical-driven oxidation of polyphenols: Isolation and structural elucidation of covalent adducts. Biochem. Biophys. Res. Commun. 2006, 346, 321–329. [Google Scholar] [CrossRef]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Mukai, K.; Oka, W.; Watanabe, K.; Egawa, Y.; Nagaoka, S. Kinetic study of free-radical-scavenging action of flavonoids in homogeneous and aqueous triton X-100 micellar solutions. J. Phys. Chem. A 1997, 101, 3746. [Google Scholar] [CrossRef]
- Mitani, S.; Ouchi, A.; Watanabe, E.; Kanesaki, Y.; Nagaoka, S.I.; Mukai, K. Stopped-flow kinetic study of the aroxyl radical-scavenging action of catechins and vitamin C in ethanol and micellar solutions. J. Agric. Food Chem. 2008. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Vervoort, L.; Moalin, M.; Mommers, A.; Douny, C.; den Hartog, G.J.M.; Haenen, G.R.M.M. The chemical reactivity of (-)-epicatechin quinone mainly resides in its B-ring. Free Radic. Biol. Med. 2018, 124, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Ruijters, E.J.B.; Weseler, A.R.; Kicken, C.; Haenen, G.R.M.M.; Bast, A. The flavanol (-)-epicatechin and its metabolites protect against oxidative stress in primary endothelial cells via a direct antioxidant effect. Eur. J. Pharmacol. 2013, 715, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005. [Google Scholar] [CrossRef]
- Apak, R.; Gorinstein, S.; Böhm, V.; Schaich, K.M.; Özyürek, M.; Güçlü, K. Methods of measurement and evaluation of natural antioxidant capacity/activity (IUPAC Technical Report)*. Pure Appl. Chem 2013, 85, 957–998. [Google Scholar] [CrossRef] [Green Version]
- Fabbrini, M.; Galli, C.; Gentili, P. Radical or electron-transfer mechanism of oxidation with some laccase/mediator systems. J. Mol. Catal. B Enzym. 2002, 18, 169–171. [Google Scholar] [CrossRef]
- Litwinienko, G.; Ingold, K.U. Solvent effects on the rates and mechanisms of reaction of phenols with free radicals. Acc. Chem. Res. 2007. [Google Scholar] [CrossRef] [PubMed]
- Litwinienko, G.; Ingold, K.U. Abnormal solvent effects on hydrogen atom abstraction. 2. Resolution of the curcumin antioxidant controversy. The role of sequential proton loss electron transfer. J. Org. Chem. 2004, 69, 5888–5896. [Google Scholar] [CrossRef] [PubMed]
- Foti, M.C.; Daquino, C.; Geraci, C. Electron-Transfer Reaction of Cinnamic Acids and Their Methyl Esters with the DPPH • Radical in Alcoholic Solutions. J. Org. Chem. 2004, 69, 2309–2314. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, F.; Lemaur, V.; Cornil, J.; Lazzaroni, R.; Duroux, J.-L.; Olivier, Y.; Trouillas, P. Free radical scavenging by natural polyphenols: Atom versus electron transfer. J. Phys. Chem. A 2013, 117, 2082. [Google Scholar] [CrossRef]
- Cren-Olivé, C.; Wieruszeski, J.M.; Maes, E.; Rolando, C. Catechin and epicatechin deprotonation followed by 13C NMR. Tetrahedron Lett. 2002. [Google Scholar] [CrossRef]
- Cren-Olivé, C.; Lebrun, S.; Hapiot, P.; Pinson, J.; Rolando, C. Selective protection of catechin gives access to the intrinsic reactivity of the two phenol rings during H-abstraction and photo-oxidation. Tetrahedron Lett. 2000. [Google Scholar] [CrossRef]
- Dangles, O.; Fargeix, G.; Dufour, C. One-electron oxidation of quercetin and quercetin derivatives in protic and non protic media. J. Chem. Soc. Perkin Trans. 2 1999, 1387–1396. [Google Scholar] [CrossRef]
- Lluís Torres, J.; Carreras, A.; Jiménez, A.; Brillas, E.; Torrelles, X.; Rius, J.; Juliá, L. Reducing Power of Simple Polyphenols by Electron-Transfer Reactions Using a New Stable Radical of the PTM Series, Tris(2,3,5,6-tetrachloro-4-nitrophenyl)methyl Radical. J. Org. Chem. 2007, 72, 3750–3756. [Google Scholar] [CrossRef]
- Lemańska, K.; Szymusiak, H.; Tyrakowska, B.; Zieliński, R.; Soffers, A.E.M.F.; Rietjens, I.M.C.M. The influence of pH on antioxidant properties and the mechanism of antioxidant action of hydroxyflavones. Free Radic. Biol. Med. 2001, 31, 869–881. [Google Scholar] [CrossRef]
- Jovanovic, S.V.; Steenken, S.; Tosic, M.; Marjanovic, B.; Simic, M.G. Flavonoids as Antioxidants. J. Am. Chem. Soc. 1994, 116, 4846–4851. [Google Scholar] [CrossRef]
- Romero, R.; Salgado, P.R.; Soto, C.; Contreras, D.; Melin, V. An Experimental Validated Computational Method for pKa Determination of Substituted 1,2-Dihydroxybenzenes. Front. Chem. 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slabbert, N.P. Ionisation of some flavanols and dihydroflavonols. Tetrahedron 1977, 33, 821–824. [Google Scholar] [CrossRef]
- Kennedy, J.A.; Munro, M.H.G.; Powell, H.K.J.; Porter, L.J.; Foo, L.Y. The protonation reactions of catechin, epicatechin and related compounds. Aust. J. Chem. 1984, 37, 885. [Google Scholar] [CrossRef]
- Pratt, D.A.; Dilabio, G.A.; Mulder, P.; Ingold, K.U. Bond strengths of toluenes, anilines, and phenols: To hammett or not. Acc. Chem. Res. 2004. [Google Scholar] [CrossRef] [PubMed]
- Marín, F.R.; Hernández-Ruiz, J.; Arnao, M.B. A colorimetric method for the determination of different functional flavonoids using 2,2′-azino-bis-(3-ethylbenzthiazoline-6-sulphonic acid) (ABTS) and peroxidase. Prep. Biochem. Biotechnol. 2019, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Casas, J.L.; del Río, J.A.; Acosta, M.; García-Cánovas, F. An enzymatic colorimetric method for measuring naringin using 2,2′-azino-bis-(3-ethylbenzthiazoline-6-sulfonic acid) (ABTS) in the presence of peroxidase. Anal. Biochem. 1990, 185, 335–338. [Google Scholar] [CrossRef]
- Hotta, H.; Nagano, S.; Ueda, M.; Tsujino, Y.; Koyama, J.; Osakai, T. Higher radical scavenging activities of polyphenolic antioxidants can be ascribed to chemical reactions following their oxidation. Biochim. Biophys. Acta-Gen. Subj. 2002, 1572, 123–132. [Google Scholar] [CrossRef]
- Hotta, H.; Sakamoto, H.; Nagano, S.; Osakai, T.; Tsujino, Y. Unusually large numbers of electrons for the oxidation of polyphenolic antioxidants. Biochim. Biophys. Acta Gen. Subj. 2001, 1526, 159. [Google Scholar] [CrossRef]
- Osman, A.M.; Wong, K.K.Y.; Fernyhough, A. The laccase/ABTS system oxidizes (+)-catechin to oligomeric products. Enzyme Microb. Technol. 2007, 40, 1272–1279. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M.G.; Reid, I.D.; Lanthier, P.; Yaguchi, M. Lignin oxidation by laccase isozymes from Trametes versicolor and role of the mediator 2,2′-azinobis(3-ethylbenzthiazoline-6-sulfonate) in kraft lignin depolymerization. Appl. Environ. Microbiol. 1995, 61, 1876–1880. [Google Scholar] [CrossRef] [Green Version]
- Murao, S.; Matsumura, E.; Yamamoto, E.; Tanaka, N.; Shin, T. Substrate Specificity of Bilirubin Oxidase and Phenol Oxidase: Coloration Reaction against Benzoic Acid Derivatives. Yakugaku Zasshi 1985, 105, 86–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnao, M.B.; Sanchez-Bravo, J.; Acosta, M. Indole-3-carbinol as a scavenger of free radicals. Biochem. Mol. Biol. Int. 1996, 39, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, E.; Yamamoto, E.; Numata, A.; Kawano, T.; Shin, T.; Murao, S. Structures of the Laccase-catalyzed Oxidation Products of Hydroxy-benzoic Acids in the Presence of ABTS [2,2′-Azino-di-(3-ethylbenzothiazoline-6-sulfonic Acid)]. Agric. Biol. Chem. 1986, 50, 1355–1357. [Google Scholar] [CrossRef]
- Shin, T.; Murao, S.; Matsumura, E. A chromogenic oxidative coupling reaction of laccase: Applications for laccase and angiotensin I converting enzyme assay. Anal. Biochem. 1987, 166, 380–388. [Google Scholar] [CrossRef]
- Liu, Y.R.; Li, W.G.; Chen, L.F.; Xiao, B.K.; Yang, J.Y.; Yang, L.; Zhang, C.G.; Huang, R.Q.; Dong, J.X. ABTS+ scavenging potency of selected flavonols from Hypericum perforatum L. by HPLC-ESI/MS QQQ: Reaction observation, adduct characterization and scavenging activity determination. Food Res. Int. 2014. [Google Scholar] [CrossRef]
- Sokolová, R.; Ramešová, Š.; Degano, I.; Hromadová, M.; Gál, M.; Abka, J. The oxidation of natural flavonoid quercetin. Chem. Commun. 2012, 48, 3433–3435. [Google Scholar] [CrossRef]
- Khasanov, V.V.; Dychko, K.A.; Labutin, A.V.; Kravtsova, S.S.; Kuryaeva, T.T. Products of the Intermediate Oxidation of Flavonoids in Aqueous Solutions and the Determination of Their Composition by High-Performance Liquid Chromatography–Mass Spectrometry. J. Anal. Chem. 2018, 73, 1248–1252. [Google Scholar] [CrossRef]
- Krishnamachari, V.; Levine, L.H.; Paré, P.W. Flavonoid oxidation by the radical generator AIBN: A unified mechanism for quercetin radical scavenging. J. Agric. Food Chem. 2002, 50, 4357–4363. [Google Scholar] [CrossRef]
- Zenkevich, I.G.; Eshchenko, A.Y.; Makarova, S.V.; Vitenberg, A.G.; Dobryakov, Y.G.; Utsal, V.A. Identification of the products of oxidation of quercetin by air oxygen at ambient temperature. Molecules 2007, 12, 654–672. [Google Scholar] [CrossRef] [Green Version]
- Zhou, A.; Sadik, O.A. Comparative Analysis of Quercetin Oxidation by Electrochemical, Enzymatic, Autoxidation, and Free Radical Generation Techniques: A Mechanistic Study. J. Agric. Food Chem. 2008, 56, 12081–12091. [Google Scholar] [CrossRef]
- Foti, M.C.; Daquino, C.; Dilabio, G.A.; Ingold, K.U. Kinetics of the oxidation of quercetin by 2,2-diphenyl-1-picrylhydrazyl (dpph •). Org. Lett. 2011, 13, 4826–4829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hvattum, E.; Stenstroøm, Y.; Ekeberg, D. Study of the reaction products of flavonols with 2,2-diphenyl-1- picrylhydrazyl using liquid chromatography coupled with negative electrospray ionization tandem spectrometry. J. Mass Spectrom. 2004, 39, 1570–1581. [Google Scholar] [CrossRef] [PubMed]
- Rice-Evans, C.; Miller, N.J. Total antioxidant status in plasma and body fluids. Methods Enzymol. 1994, 234, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Cano, A. An end-point method for estimation of the total antioxidant activity in plant material. Phytochem. Anal. 1998. [Google Scholar] [CrossRef]
- Arnao, M.B.; Cano, A.; Acosta, M. The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem. 2001, 73, 239–244. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Y.; Liu, Y.; Yue, F.; Lu, Y.; Qiu, H.; Gao, D.; Gao, Y.; Wu, Y.; Wang, Z.; et al. In vitro kinetic evaluation of the free radical scavenging ability of propofol. Anesthesiology 2012, 116, 1258–1266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tai, A.; Ohno, A.; Ito, H. Isolation and Characterization of the 2,2′-Azinobis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) Radical Cation-Scavenging Reaction Products of Arbutin. J. Agric. Food Chem. 2016, 64, 7285–7290. [Google Scholar] [CrossRef]
- Hilgers, R.; Vincken, J.P.; Gruppen, H.; Kabel, M.A. Laccase/Mediator Systems: Their Reactivity toward Phenolic Lignin Structures. ACS Sustain. Chem. Eng. 2018, 6, 2037–2046. [Google Scholar] [CrossRef] [Green Version]
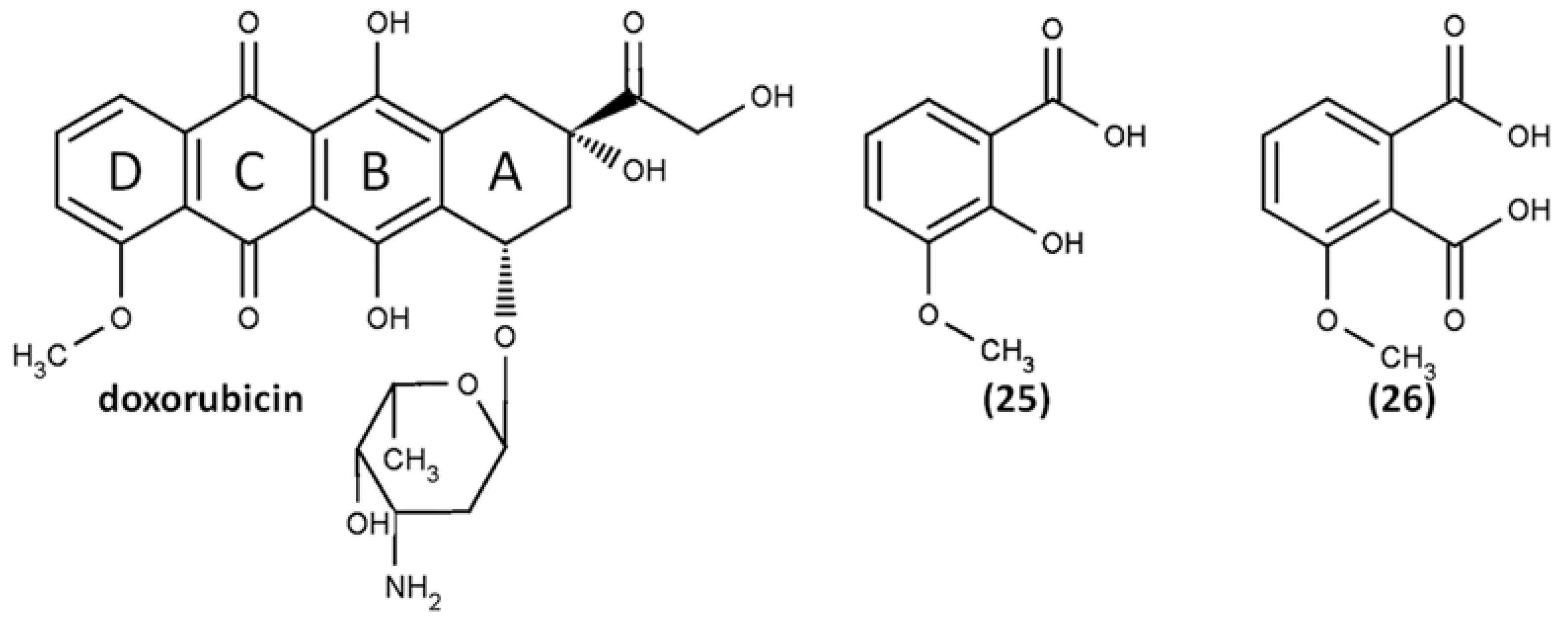
- Reszka, K.J.; Britigan, B.E. Doxorubicin inhibits oxidation of 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonate) (ABTS) by a lactoperoxidase/H2O2 system by reacting with ABTS-derived radical. Arch. Biochem. Biophys. 2007, 466, 164–171. [Google Scholar] [CrossRef] [Green Version]
- Nawara, K.; Krysinski, P.; Blanchard, G.J. Photoinduced Reactivity of Doxorubicin: Catalysis and Degradation. J. Phys. Chem. A 2012, 116, 4330–4337. [Google Scholar] [CrossRef]
- Reszka, K.J.; Wagner, B.A.; Teesch, L.M.; Britigan, B.E.; Spitz, D.R.; Burns, C.P. Inactivation of Anthracyclines by Cellular Peroxidase. Cancer Res. 2005, 65, 6346–6353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cartoni, A.; Menna, P.; Salvatorelli, E.; Braghiroli, D.; Giampietro, R.; Animati, F.; Urbani, A.; Del Boccio, P.; Minotti, G. Oxidative degradation of cardiotoxic anticancer anthracyclines to phthalic acids: Novel function for ferrylmyoglobin. J. Biol. Chem. 2004, 279, 5088–5099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramu, A.; Mehta, M.M.; Liu, J.; Turyan, I.; Aleksic, A. The riboflavin-mediated photooxidation of doxorubicin. Cancer Chemother. Pharmacol. 2000, 46, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Poeggeler, B.; Lopez-Burillo, S.; Mayo, J.C.; Sainz, R.M.; Reiter, R.J. Mechanistic and comparative studies of melatonin and classic antioxidants in terms of their interactions with the ABTS cation radical. J. Pineal Res. 2003, 34, 249–259. [Google Scholar] [CrossRef]
- Tesoriere, L.; Avellone, G.; Ceraulo, L.; D’Arpa, D.; Allegra, M.; Livrea, M.A. Oxidation of melatonin by oxoferryl hemoglobin: A mechanistic study. Free Radic. Res. 2001, 35, 633–642. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Plummer, B.F.; Hardies, L.J.; Weintraub, S.T.; Vijayalaxmi; Shepherd, A.M.M. A novel melatonin metabolite, cyclic 3-hydroxymelatonin: A biomarker of in vivo hydroxyl radical generation. Biochem. Biophys. Res. Commun. 1998, 253, 614–620. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin and reactive oxygen and nitrogen species: A model for the plant redox network. Melatonin Res. 2019, 2, 152–168. [Google Scholar] [CrossRef]
- Than, N.N.; Heer, C.; Laatsch, H.; Hardeland, R. Reactions of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK) with the ABTS cation radical: Identification of new oxidation products. Redox Rep. 2006, 11, 15–24. [Google Scholar] [CrossRef]
- Wybraniec, S.; Starzak, K.; Skopińska, A.; Nemzer, B.; Pietrzkowski, Z.; Michałowski, T. Studies on nonenzymatic oxidation mechanisms in neobetanin, betanin, and decarboxylated betanins. J. Agric. Food Chem. 2013, 61, 6465–6476. [Google Scholar] [CrossRef]
- Valent, I.; Topoľská, D.; Valachová, K.; Bujdák, J.; Šoltés, L. Kinetics of ABTS derived radical cation scavenging by bucillamine, cysteine, and glutathione. Catalytic effect of Cu2+ ions. Biophys. Chem. 2016, 212, 9–16. [Google Scholar] [CrossRef]








| Method | References of Basic Publications | Citation Rate 1 | Frequency of Abbreviation Use (Keywords Used) |
|---|---|---|---|
| TEAC | [1,2,16] | 13,220 | 3772 2 (“TEAC” or “Trolox equivalent antioxidant capacity”) since 1993 |
| ABTS/PP decolorization assay | [1] | 9845 | 10,109 (“ABTS antioxidant” or “ABTS antiradical” or “ABTS radical scavenging”) since 1988 |
| DPPH | [17,18,19,20] | 11,177 | 35,136 (“DPPH antioxidant” or “DPPH antiradical” or “DPPH radical scavenging”) since 1955 |
| FRAP | [21,22,23] | 11,040 | 9492 (“FRAP” or “Ferric reducing antioxidant power”) 3 since 1994 |
| Folin–Ciocalteu | [24,25,26,27] | 7630 | 2803 (“Folin–Ciocalteu antioxidant”) since 1976 |
| ORAC | [28,29,30,31,32,33] | 3478 | 3619 (“ORAC” or “Oxygen radical absorbance capacity”) since 1993 |
| CUPRAC | [34,35,36] | 1260 | 685 (“CUPRAC” or “Cupric ion reducing antioxidant capacity”) since 2004 |
| λ(ABTS•+), nm | Extinction Coefficient, ε | Reference |
|---|---|---|
| 415 nm | 36,000 L∙mol−1∙cm−1 in water | [43,44] |
| 414 nm | 31,100 l mol−1 cm−1 in water (sodium phosphate buffer, pH 7.5) 33,630 L∙mol−1∙cm−1 in ethanol | [3,45] |
| 734 nm | 15,000 L∙mol−1∙cm−1 in water 16,000 L∙mol−1∙cm−1 in ethanol | [1] |
| 730 nm | 12,947 l mol−1 cm−1 in water (sodium phosphate buffer, pH 7.5) 14,750 L∙mol−1∙cm−1 in ethanol | [3,45] |
| Antioxidant | The Calculated Number of ABTS•+ Molecules Reduced by One Molecule of Antioxidant 1 | ||
|---|---|---|---|
| Name | Formula | Decolorization Assay | Lag-Time Assay |
| Trolox |  | 2.4 | 1.7 |
| Quercetin |  | 12.0 | 4.7 |
| Morin |  | 6.9 | 3.3 |
| Rutin |  | 6.6 | 2.8 |
| Taxifolin |  | 5.9 | 2.7 |
| Apigenin |  | 5.3 | ND |
| Naringenin |  | 4.6 | ND |
| Glutathione |  | 2.7 | 0.8 |
| α-Tocopherol |  | 1.9 | 1.9 |
| Antioxidant | Antioxidant Concentration Needed to Inhibit 50% of ABTS•+, µM | The Ratio Antioxidant/ABTS•+ |
|---|---|---|
| Trolox | 11.0 | 1:4 |
| Glutathion | 8.4 | 1:6 |
| Taxifolin | 4.1 | 1:11 |
| Quercetin | 1.8 | 1:26 |
| α-Tocopherol | 12.4 | 1:4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ilyasov, I.R.; Beloborodov, V.L.; Selivanova, I.A.; Terekhov, R.P. ABTS/PP Decolorization Assay of Antioxidant Capacity Reaction Pathways. Int. J. Mol. Sci. 2020, 21, 1131. https://doi.org/10.3390/ijms21031131
Ilyasov IR, Beloborodov VL, Selivanova IA, Terekhov RP. ABTS/PP Decolorization Assay of Antioxidant Capacity Reaction Pathways. International Journal of Molecular Sciences. 2020; 21(3):1131. https://doi.org/10.3390/ijms21031131
Chicago/Turabian StyleIlyasov, Igor R., Vladimir L. Beloborodov, Irina A. Selivanova, and Roman P. Terekhov. 2020. "ABTS/PP Decolorization Assay of Antioxidant Capacity Reaction Pathways" International Journal of Molecular Sciences 21, no. 3: 1131. https://doi.org/10.3390/ijms21031131