Immunologic and Non-Immunologic Mechanisms Leading to Airway Remodeling in Asthma
Abstract
:1. Introduction
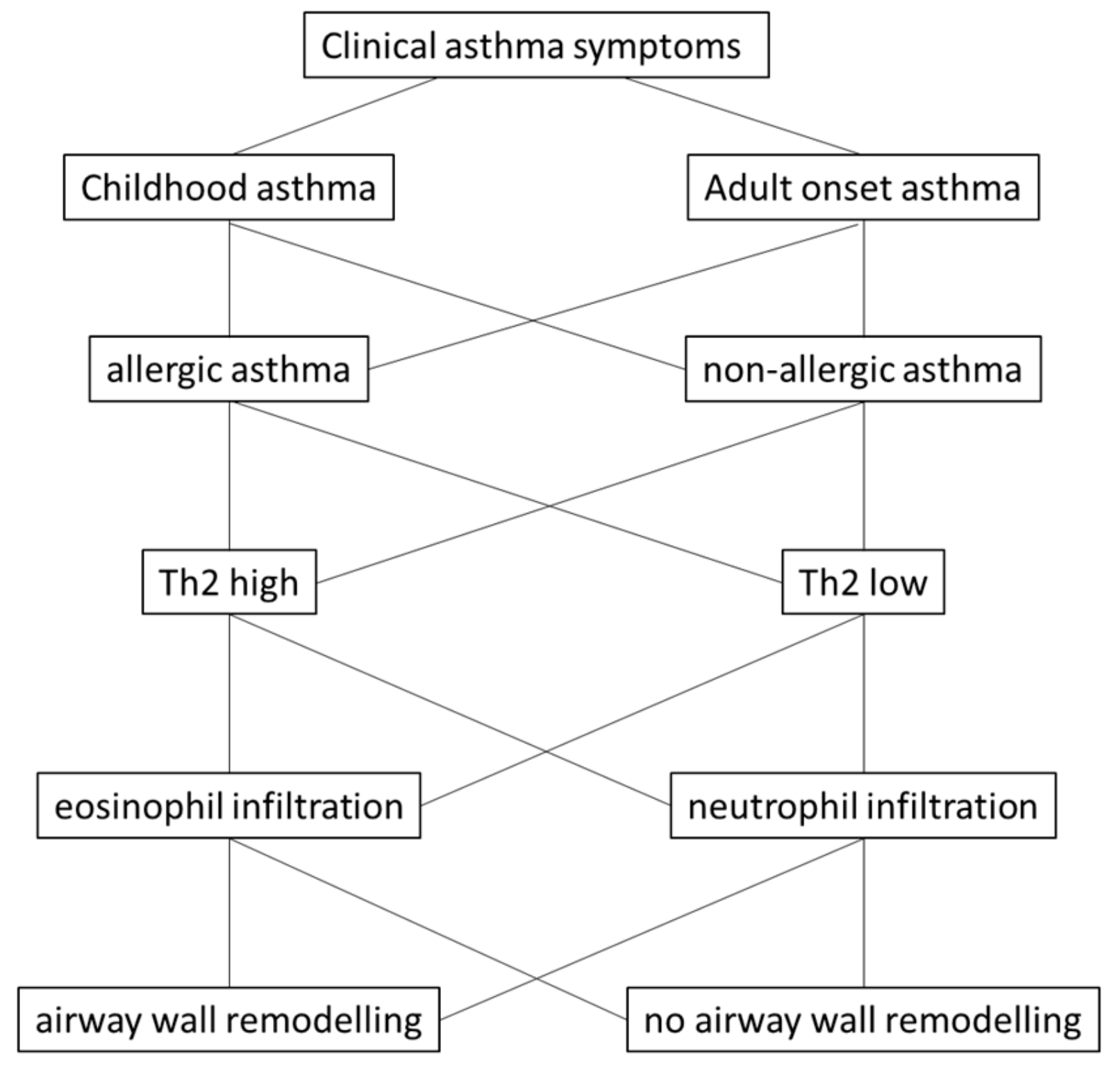
2. Asthma
3. Immunologic and Non-Immunologic Stimulation of Airway Wall Remodeling in Asthma
4. Controlling the Epithelial-Mesenchymal Interaction by Immunologic and Non-Immunologic Factors
5. Mechanisms to Explain How Air Pollution Triggers Remodeling in Asthma
6. Mechanisms that Control Remodeling in Non-Allergic Asthma
7. Early in Life Epigenetic events and the Predisposition of the Lung to Develop Asthma
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Akt | activated protein kinase B |
| ATP | adenosin-tri-phosphate |
| bFGF | basic fibroblast growth factor |
| COPD | chronic obstructive pulmonary disease |
| CD | cluster of differentiation |
| Erk | extracellular signal-regulated kinase |
| HSP | heat shock protein |
| IgE | immune-globulin E |
| IL | interleukin |
| IP3K | Inositol-trisphosphate 3-kinase |
| MAPK | mitogen activated protein kinase |
| miR | microRNA |
| mTOR | mammalian target of rapamycin |
| p70S6K | ribosomal protein S6 kinase |
| PGC | peroxisome proliferator-activated receptor gamma coactivator |
| PGE | prostaglandin E |
| PM | particulate matter |
| PPAR | peroxisome proliferator-activated receptor |
| PRMT | protein arginine methyltransferase |
| PTEN | phosphatase and tensin homolog |
| STAT | signal transducers and activators of transcription |
| TGF | transforming growth factor |
| Th | T-helper cell |
| TSLP | thymic stromal lymphopoietin |
| TLR | toll-like receptor |
| WHO | world health organization |
| Wnt | wingless-related integration site |
References
- Abramson, M.J.; Guo, Y. Indoor Endotoxin Exposure and Ambient Air Pollutants Interact on Asthma Outcomes. Am. J. Respir. Crit. Care Med. 2019, 200, 652–654. [Google Scholar] [CrossRef]
- Mutlu, G.M.; Peker, Y. Air Pollution, Asthma, and Sleep Apnea: New Epidemiological Links? Ann. Am. Thorac. Soc. 2019, 16, 307–308. [Google Scholar] [CrossRef] [PubMed]
- Garcia, E.; Berhane, K.T.; Islam, T.; McConnell, R.; Urman, R.; Chen, Z.; Gilliland, F.D. Association of Changes in Air Quality with Incident Asthma in Children in California, 1993–2014. JAMA 2019, 321, 1906–1915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thurston, G.D.; Rice, M.B. Air Pollution Exposure and Asthma Incidence in Children: Demonstrating the Value of Air Quality Standards. JAMA 2019, 321, 1875–1877. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.F.S.; Hashizume, M.; Obase, Y.; Doi, M.; Tamura, K.; Tomari, S.; Kawano, T.; Fukushima, C.; Matsuse, H.; Chung, Y.; et al. Associations of chemical composition and sources of PM(2.5) with lung function of severe asthmatic adults in a low air pollution environment of urban Nagasaki, Japan. Environ. Pollut. 2019, 252, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Muraro, A.; Steelant, B.; Pietikainen, S.; Borrelli, D.; Childers, N.; Callebaut, I.; Kortekaas Krohn, I.; Martens, K.; Pugin, B.; Popescu, F.D.; et al. European symposium on the awareness of allergy: Report of the promotional campaign in the European Parliament (26–28 April 2016). Allergy 2017, 72, 173–176. [Google Scholar] [CrossRef]
- Vercelli, D. Does epigenetics play a role in human asthma? Allergol. Int. 2016, 65, 123–126. [Google Scholar] [CrossRef]
- Matthews, N.C.; Pfeffer, P.E.; Mann, E.H.; Kelly, F.J.; Corrigan, C.J.; Hawrylowicz, C.M.; Lee, T.H. Urban Particulate Matter-Activated Human Dendritic Cells Induce the Expansion of Potent Inflammatory Th1, Th2, and Th17 Effector Cells. Am. J. Respir. Cell Mol. Biol. 2016, 54, 250–262. [Google Scholar] [CrossRef] [Green Version]
- Brandt, E.B.; Kovacic, M.B.; Lee, G.B.; Gibson, A.M.; Acciani, T.H.; Le Cras, T.D.; Ryan, P.H.; Budelsky, A.L.; Khurana Hershey, G.K. Diesel exhaust particle induction of IL-17A contributes to severe asthma. J. Allergy Clin. Immunol. 2013, 132, 1194–1204. [Google Scholar] [CrossRef] [Green Version]
- Brandt, E.B.; Biagini Myers, J.M.; Acciani, T.H.; Ryan, P.H.; Sivaprasad, U.; Ruff, B.; LeMasters, G.K.; Bernstein, D.I.; Lockey, J.E.; LeCras, T.D.; et al. Exposure to allergen and diesel exhaust particles potentiates secondary allergen-specific memory responses, promoting asthma susceptibility. J. Allergy Clin. Immunol. 2015, 136, 295–303. [Google Scholar] [CrossRef] [Green Version]
- Jung, K.H.; Lovinsky-Desir, S.; Yan, B.; Torrone, D.; Lawrence, J.; Jezioro, J.R.; Perzanowski, M.; Perera, F.P.; Chillrud, S.N.; Miller, R.L. Effect of personal exposure to black carbon on changes in allergic asthma gene methylation measured 5 days later in urban children: Importance of allergic sensitization. Clin. Epigenet. 2017, 9, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuda, M.; Bonatti, R.; Marquezini, M.V.; Garcia, M.L.; Santos, U.P.; Braga, A.L.; Alves, M.R.; Saldiva, P.H.; Monteiro, M.L. Lacrimal Cytokines Assessment in Subjects Exposed to Different Levels of Ambient Air Pollution in a Large Metropolitan Area. PLoS ONE 2015, 10, e0143131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, I.V.; Lozupone, C.A.; Schwartz, D.A. The environment, epigenome, and asthma. J. Allergy Clin. Immunol. 2017, 140, 14–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tyler, S.R.; Bunyavanich, S. Leveraging-omics for asthma endotyping. J. Allergy Clin. Immunol. 2019, 144, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, A.M.; Moore, W.C. Severe Asthma Phenotypes—How Should They Guide Evaluation and Treatment? J. Allergy Clin. Immunol. Pract. 2017, 5, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Papi, A.; Brightling, C.; Pedersen, S.E.; Reddel, H.K. Asthma. Lancet 2018, 391, 783–800. [Google Scholar] [CrossRef]
- Castro-Rodriguez, J.A.; Saglani, S.; Rodriguez-Martinez, C.E.; Oyarzun, M.A.; Fleming, L.; Bush, A. The relationship between inflammation and remodeling in childhood asthma: A systematic review. Pediatr. Pulmonol. 2018, 53, 824–835. [Google Scholar] [CrossRef]
- Peters, M.C.; Ringel, L.; Dyjack, N.; Herrin, R.; Woodruff, P.G.; Rios, C.; O’Connor, B.; Fahy, J.V.; Seibold, M.A. A Transcriptomic Method to Determine Airway Immune Dysfunction in T2-High and T2-Low Asthma. Am. J. Respir. Crit. Care Med. 2019, 199, 465–477. [Google Scholar] [CrossRef]
- Bigler, J.; Boedigheimer, M.; Schofield, J.P.R.; Skipp, P.J.; Corfield, J.; Rowe, A.; Sousa, A.R.; Timour, M.; Twehues, L.; Hu, X.; et al. A Severe Asthma Disease Signature from Gene Expression Profiling of Peripheral Blood from U-BIOPRED Cohorts. Am. J. Respir. Crit. Care Med. 2017, 195, 1311–1320. [Google Scholar] [CrossRef]
- Hekking, P.P.; Loza, M.J.; Pavlidis, S.; De Meulder, B.; Lefaudeux, D.; Baribaud, F.; Auffray, C.; Wagener, A.H.; Brinkman, P.; Lutter, R.; et al. Transcriptomic gene signatures associated with persistent airflow limitation in patients with severe asthma. Eur. Respir. J. 2017, 50, 1602298. [Google Scholar] [CrossRef] [Green Version]
- Barnes, P.J. Targeting cytokines to treat asthma and chronic obstructive pulmonary disease. Nat. Rev. Immunol. 2018, 18, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Bush, A. Cytokines and Chemokines as Biomarkers of Future Asthma. Front. Pediatr. 2019, 7, 72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elliot, J.G.; Noble, P.B.; Mauad, T.; Bai, T.R.; Abramson, M.J.; McKay, K.O.; Green, F.H.Y.; James, A.L. Inflammation-dependent and independent airway remodelling in asthma. Respirology 2018, 23, 1138–1145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bossley, C.J.; Fleming, L.; Gupta, A.; Regamey, N.; Frith, J.; Oates, T.; Tsartsali, L.; Lloyd, C.M.; Bush, A.; Saglani, S. Pediatric severe asthma is characterized by eosinophilia and remodeling without T(H)2 cytokines. J. Allergy Clin. Immunol. 2012, 129, 974–982. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grainge, C.L.; Lau, L.C.; Ward, J.A.; Dulay, V.; Lahiff, G.; Wilson, S.; Holgate, S.; Davies, D.E.; Howarth, P.H. Effect of bronchoconstriction on airway remodeling in asthma. N. Engl. J. Med. 2011, 364, 2006–2015. [Google Scholar] [CrossRef] [Green Version]
- Seibold, M.A. Interleukin-13 Stimulation Reveals the Cellular and Functional Plasticity of the Airway Epithelium. Ann. Am. Thorac. Soc. 2018, 15, S98–S102. [Google Scholar] [CrossRef]
- Russell, R.J.; Chachi, L.; FitzGerald, J.M.; Backer, V.; Olivenstein, R.; Titlestad, I.L.; Ulrik, C.S.; Harrison, T.; Singh, D.; Chaudhuri, R.; et al. MESOS study investigators. Effect of tralokinumab, an interleukin-13 neutralising monoclonal antibody, on eosinophilic airway inflammation in uncontrolled moderate-to-severe asthma (MESOS): A multicentre, double-blind, randomised, placebo-controlled phase 2 trial. Lancet Respir. Med. 2018, 6, 499–510. [Google Scholar]
- Yang, S.J.; Allahverdian, S.; Saunders, A.D.R.; Liu, E.; Dorscheid, D.R. IL-13 signaling through IL-13 receptor α2 mediates airway epithelial wound repair. FASEB J. 2019, 33, 3746–3757. [Google Scholar] [CrossRef] [Green Version]
- Siddiqui, S.; Shikotra, A.; Richardson, M.; Doran, E.; Choy, D.; Bell, A.; Austin, C.D.; Eastham-Anderson, J.; Hargadon, B.; Arron, J.R.; et al. Airway pathological heterogeneity in asthma: Visualization of disease microclusters using topological data analysis. J. Allergy Clin. Immunol. 2018, 142, 1457–1468. [Google Scholar] [CrossRef]
- Qin, W.; Deng, T.; Cui, H.; Zhang, Q.; Liu, X.; Yang, X.; Chen, M. Exposure to diisodecyl phthalate exacerbated Th2 and Th17-mediated asthma through aggravating oxidative stress and the activation of p38 MAPK. Food Chem. Toxicol. 2018, 114, 78–87. [Google Scholar] [CrossRef]
- Liu, G.; Cooley, M.A.; Nair, P.M.; Donovan, C.; Hsu, A.C.; Jarnicki, A.G.; Haw, T.J.; Hansbro, N.G.; Ge, Q.; Brown, A.C.; et al. Airway remodelling and inflammation in asthma are dependent on the extracellular matrix protein fibulin-1c. J. Pathol. 2017, 243, 510–523. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, A.I.; Jackson, D.J.; Edwards, M.R.; Johnston, S.L. Airway Epithelial Orchestration of Innate Immune Function in Response to Virus Infection. A Focus on Asthma. Ann. Am. Thorac. Soc. 2016, 13, S55–S63. [Google Scholar] [PubMed]
- Prakash, Y.S. Emerging concepts in smooth muscle contributions to airway structure and function: Implications for health and disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 311, L1113–L1140. [Google Scholar] [CrossRef] [PubMed]
- Noble, P.B.; Pascoe, C.D.; Lan, B.; Ito, S.; Kistemaker, L.E.; Tatler, A.L.; Pera, T.; Brook, B.S.; Gosens, R.; West, A.R. Airway smooth muscle in asthma: Linking contraction and mechanotransduction to disease pathogenesis and remodelling. Pulm. Pharmacol. Ther. 2014, 29, 96–107. [Google Scholar] [CrossRef] [PubMed]
- King, G.G.; Noble, P.B. Airway remodelling in asthma: It’s not going away. Respirology 2016, 21, 203–204. [Google Scholar] [CrossRef]
- Girodet, P.O.; Allard, B.; Thumerel, M.; Begueret, H.; Dupin, I.; Ousova, O.; Lassalle, R.; Maurat, E.; Ozier, A.; Trian, T.; et al. Bronchial Smooth Muscle Remodeling in Nonsevere Asthma. Am. J. Respir. Crit. Care Med. 2016, 193, 627–633. [Google Scholar] [CrossRef]
- Halwani, R.; Sultana, A.; Al-Kufaidy, R.; Jamhawi, A.; Vazquez-Tello, A.; Al-Muhsen, S. Th-17 regulatory cytokines inhibit corticosteroid induced airway structural cells apoptosis. Respir. Res. 2016, 17, 6. [Google Scholar] [CrossRef] [Green Version]
- Pillai, P.; Corrigan, C.J.; Ying, S. Airway epithelium in atopic and nonatopic asthma: Similarities and differences. ISRN Allergy 2011, 2011, 195846. [Google Scholar] [CrossRef] [Green Version]
- Shahana, S.; Björnsson, E.; Lúdvíksdóttir, D.; Janson, C.; Nettelbladt, O.; Venge, P.; Roomans, G.M.; BHR-group. Ultrastructure of bronchial biopsies from patients with allergic and non-allergic asthma. Respir. Med. 2005, 99, 429–443. [Google Scholar] [CrossRef] [Green Version]
- Huber, H.L.; Koessler, K.K. The pathology of bronchial asthma. Arch. Intern. Med. 1922, 30, 689–760. [Google Scholar] [CrossRef] [Green Version]
- McAlinden, K.D.; Deshpande, D.A.; Ghavami, S.; Xenaki, D.; Sohal, S.S.; Oliver, B.G.; Haghi, M.; Sharma, P. Autophagy Activation in Asthma Airways Remodeling. Am. J. Respir. Cell Mol. Biol. 2019, 60, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Lezmi, G.; Deschildre, A.; Abou Taam, R.; Fayon, M.; Blanchon, S.; Troussier, F.; Mallinger, P.; Mahut, B.; Gosset, P.; de Blic, J. Remodelling and inflammation in preschoolers with severe recurrent wheeze and asthma outcome at school age. Clin. Exp. Allergy 2018, 48, 806–813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaczmarek, K.A.; Clifford, R.L.; Knox, A.J. Epigenetic Changes in Airway Smooth Muscle as a Driver of Airway Inflammation and Remodeling in Asthma. Chest 2019, 155, 816–824. [Google Scholar] [CrossRef]
- Pan, S.; Conaway, S., Jr.; Deshpande, D.A. Mitochondrial regulation of airway smooth muscle functions in health and pulmonary diseases. Arch. Biochem. Biophys. 2019, 663, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Redhu, N.S.; Gounni, A.S. The high affinity IgE receptor (FcεRI) expression and function in airway smooth muscle. Pulm. Pharmacol. Ther. 2013, 26, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Roth, M.; Zhong, J.; Zumkeller, C.; S’ng, C.T.; Goulet, S.; Tamm, M. The role of IgE-receptors in IgE-dependent airway smooth muscle cell remodelling. PLoS ONE 2013, 8, e56015. [Google Scholar] [CrossRef] [PubMed]
- Redhu, N.S.; Shan, L.; Al-Subait, D.; Ashdown, H.L.; Movassagh, H.; Lamkhioued, B.; Gounni, A.S. IgE induces proliferation in human airway smooth muscle cells: Role of MAPK and STAT3 pathways. Allergy Asthma Clin. Immunol. 2013, 9, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roth, M.; Tamm, M. The effects of omalizumab on IgE-induced cytokine synthesis by asthmatic airway smooth muscle cells. Ann. Allergy Asthma Immunol. 2010, 104, 152–160. [Google Scholar] [CrossRef]
- Roth, M.; Zhao, F.; Zhong, J.; Lardinois, D.; Tamm, M. Serum IgE Induced Airway Smooth Muscle Cell Remodeling Is Independent of Allergens and Is Prevented by Omalizumab. PLoS ONE 2015, 10, e0136549. [Google Scholar] [CrossRef]
- Balhara, J.; Shan, L.; Zhang, J.; Muhuri, A.; Halayko, A.J.; Almiski, M.S.; Doeing, D.; McConville, J.; Matzuk, M.M.; Gounni, A.S. Pentraxin 3 deletion aggravates allergic inflammation through a T(H)17-dominant phenotype and enhanced CD4 T-cell survival. J. Allergy Clin. Immunol. 2017, 139, 950–963. [Google Scholar] [CrossRef] [Green Version]
- Fang, L.; Wang, X.; Sun, Q.; Papakonstantinou, E.; S’ng, C.; Tamm, M.; Stolz, D.; Roth, M. IgE Downregulates PTEN through MicroRNA-21-5p and Stimulates Airway Smooth Muscle Cell Remodeling. Int. J. Mol. Sci. 2019, 20, E875. [Google Scholar] [CrossRef] [Green Version]
- Movassagh, H.; Shan, L.; Chakir, J.; McConville, J.F.; Halayko, A.J.; Koussih, L.; Gounni, A.S. Expression of semaphorin 3E is suppressed in severe asthma. J. Allergy Clin. Immunol. 2017, 140, 1176–1179. [Google Scholar] [CrossRef] [Green Version]
- Movassagh, H.; Shan, L.; Duke-Cohan, J.S.; Halayko, A.J.; Uzonna, J.E.; Gounni, A.S. Semaphorin 3E Alleviates Hallmarks of House Dust Mite-Induced Allergic Airway Disease. Am. J. Pathol. 2017, 187, 1566–1576. [Google Scholar] [CrossRef] [Green Version]
- Tatari, N.; Movassagh, H.; Shan, L.; Koussih, L.; Gounni, A.S. Semaphorin 3E Inhibits House Dust Mite-Induced Angiogenesis in a Mouse Model of Allergic Asthma. Am. J. Pathol. 2019, 189, 762–772. [Google Scholar] [CrossRef]
- Trian, T.; Allard, B.; Ozier, A.; Maurat, E.; Dupin, I.; Thumerel, M.; Ousova, O.; Gillibert-Duplantier, J.; Le Morvan, V.; Begueret, H.; et al. Selective dysfunction of p53 for mitochondrial biogenesis induces cellular proliferation in bronchial smooth muscle from asthmatic patients. J. Allergy Clin. Immunol. 2016, 137, 1717–1726. [Google Scholar] [CrossRef]
- Defnet, A.E.; Huang, W.; Polischak, S.; Yadav, S.K.; Kane, M.A.; Shapiro, P.; Deshpande, D.A. Effects of ATP-competitive and function-selective ERK inhibitors on airway smooth muscle cell proliferation. FASEB J. 2019, 33, 10833–10843. [Google Scholar] [CrossRef]
- Johnson, P.R.; Burgess, J.K.; Underwood, P.A.; Au, W.; Poniris, M.H.; Tamm, M.; Ge, Q.; Roth, M.; Black, J.L. Extracellular matrix proteins modulate asthmatic airway smooth muscle cell proliferation via an autocrine mechanism. J. Allergy Clin. Immunol. 2004, 113, 690–696. [Google Scholar] [CrossRef]
- Roth, M.; Johnson, P.R.; Borger, P.; Bihl, M.P.; Rüdiger, J.J.; King, G.G.; Ge, Q.; Hostettler, K.; Burgess, J.K.; Black, J.L.; et al. Dysfunctional interaction of C/EBPalpha and the glucocorticoid receptor in asthmatic bronchial smooth-muscle cells. N. Engl. J. Med. 2004, 351, 560–574. [Google Scholar] [CrossRef] [Green Version]
- Chernyavsky, I.L.; Croisier, H.; Chapman, L.A.; Kimpton, L.S.; Hiorns, J.E.; Brook, B.S.; Jensen, O.E.; Billington, C.K.; Hall, I.P.; Johnson, S.R. The role of inflammation resolution speed in airway smooth muscle mass accumulation in asthma: Insight from a theoretical model. PLoS ONE 2014, 9, e90162. [Google Scholar] [CrossRef] [Green Version]
- Boulet, L.P. Airway remodeling in asthma: Update on mechanisms and therapeutic approaches. Curr. Opin. Pulm. Med. 2018, 24, 56–62. [Google Scholar] [CrossRef]
- Garantziotis, S.; Brezina, M.; Castelnuovo, P.; Drago, L. The role of hyaluronan in the pathobiology and treatment of respiratory disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 310, L785–L795. [Google Scholar] [CrossRef] [Green Version]
- Mehta, A.K.; Doherty, T.; Broide, D.; Croft, M. Tumor necrosis factor family member LIGHT acts with IL-1β and TGF-β to promote airway remodeling during rhinovirus infection. Allergy 2018, 73, 1415–1424. [Google Scholar] [CrossRef]
- Toffoli da Silva, G.; Espíndola, M.S.; Fontanari, C.; Rosada, R.S.; Faccioli, L.H.; Ramos, S.G.; Rodrigues, V.; Frantz, F.G. 5-lipoxygenase pathway is essential for the control of granuloma extension induced by Schistosoma mansoni eggs in lung. Exp. Parasitol. 2016, 167, 124–129. [Google Scholar] [CrossRef]
- Yang, R.; Tan, M.; Xu, J.; Zhao, X. Investigating the regulatory role of ORMDL3 in airway barrier dysfunction using in vivo and in vitro models. Int. J. Mol. Med. 2019, 44, 535–548. [Google Scholar] [CrossRef]
- Wu, X.; Qi, H.; Yang, Y.; Yin, Y.; Ma, D.; Li, H.; Qu, Y. Downregulation of matrix metalloproteinase-19 induced by respiratory syncytial viral infection affects the interaction between epithelial cells and fibroblasts. Mol. Med. Rep. 2016, 13, 167–173. [Google Scholar] [CrossRef] [Green Version]
- Feng, S.; Zeng, D.; Zheng, J.; Zhao, D. MicroRNAs: Mediators and Therapeutic Targets to Airway Hyper Reactivity After Respiratory Syncytial Virus Infection. Front. Microbiol. 2018, 9, 2177. [Google Scholar] [CrossRef]
- Saunders, R.; Kaul, H.; Berair, R.; Gonem, S.; Singapuri, A.; Sutcliffe, A.J.; Chachi, L.; Biddle, M.S.; Kaur, D.; Bourne, M.; et al. DP2 antagonism reduces airway smooth muscle mass in asthma by decreasing eosinophilia and myofibroblast recruitment. Sci. Transl. Med. 2019, 11, eaao6451. [Google Scholar] [CrossRef] [Green Version]
- Bel, E.H.; Ten Brinke, A. New Anti-Eosinophil Drugs for Asthma and COPD: Targeting the Trait! Chest 2017, 152, 1276–1282. [Google Scholar] [CrossRef]
- Evans, M.J.; Van Winkle, L.S.; Fanucchi, M.V.; Plopper, C.G. The attenuated fibroblast sheath of the respiratory tract epithelial-mesenchymal trophic unit. Am. J. Respir. Cell Mol. Biol. 1999, 21, 655–657. [Google Scholar] [CrossRef] [Green Version]
- Holgate, S.T.; Wenzel, S.; Postma, D.S.; Weiss, S.T.; Renz, H.; Sly, P.D. Asthma. Nat. Rev. Dis. Primers. 2015, 1, 15025. [Google Scholar] [CrossRef]
- Abs, V.; Bonicelli, J.; Kacza, J.; Zizzadoro, C.; Abraham, G. Equine bronchial fibroblasts enhance proliferation and differentiation of primary equine bronchial epithelial cells co-cultured under air-liquid interface. PLoS ONE 2019, 14, e0225025. [Google Scholar] [CrossRef]
- Stefanowicz, D.; Ullah, J.; Lee, K.; Shaheen, F.; Olumese, E.; Fishbane, N.; Koo, H.K.; Hallstrand, T.S.; Knight, D.A.; Hackett, T.L. Epigenetic modifying enzyme expression in asthmatic airway epithelial cells and fibroblasts. BMC Pulm. Med. 2017, 17, 24. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Li, Y.; Lv, Z.; Chen, Y.; Li, Y.; Huang, K.; Corrigan, C.J.; Ying, S. Bronchial Allergen Challenge of Patients with Atopic Asthma Triggers an Alarmin (IL-33, TSLP, and IL-25) Response in the Airways Epithelium and Submucosa. J. Immunol. 2018, 201, 2221–2231. [Google Scholar] [CrossRef] [Green Version]
- James, R.G.; Reeves, S.R.; Barrow, K.A.; White, M.P.; Glukhova, V.A.; Haghighi, C.; Seyoum, D.; Debley, J.S. Deficient Follistatin-like 3 Secretion by Asthmatic Airway Epithelium Impairs Fibroblast Regulation and Fibroblast-to-Myofibroblast Transition. Am. J. Respir. Cell Mol. Biol. 2018, 59, 104–113. [Google Scholar] [CrossRef]
- Skevaki, C.L.; Psarras, S.; Volonaki, E.; Pratsinis, H.; Spyridaki, I.S.; Gaga, M.; Georgiou, V.; Vittorakis, S.; Telcian, A.G.; Maggina, P.; et al. Rhinovirus-induced basic fibroblast growth factor release mediates airway remodeling features. Clin. Transl. Allergy 2012, 2, 14. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Tan, M.; Ouyang, H.; Deng, L. Effects of ozone stimulation of bronchial epithelial cells on proliferation and collagen synthesis of co-cultured lung fibroblasts. Exp. Ther. Med. 2018, 15, 5314–5322. [Google Scholar] [CrossRef] [Green Version]
- Avdalovic, M.V.; Tyler, N.K.; Putney, L.; Nishio, S.J.; Quesenberry, S.; Singh, P.J.; Miller, L.A.; Schelegle, E.S.; Plopper, C.G.; Vu, T.; et al. Ozone exposure during the early postnatal period alters the timing and pattern of alveolar growth and development in nonhuman primates. Anat. Rec. 2012, 295, 1707–1716. [Google Scholar] [CrossRef] [Green Version]
- Miller, L.A.; Gerriets, J.E.; Tyler, N.K.; Abel, K.; Schelegle, E.S.; Plopper, C.G.; Hyde, D.M. Ozone and allergen exposure during postnatal development alters the frequency and airway distribution of CD25+ cells in infant rhesus monkeys. Toxicol. Appl. Pharmacol. 2009, 236, 39–48. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.; Fang, L.; Roth, M.; Tang, X.; Papakonstantinou, E.; Zhai, W.; Louis, R.; Heinen, V.; Schleich, F.; Lu, S.; et al. Bronchial thermoplasty decreases airway remodelling by blocking epithelium-derived heat shock protein 60 (HSP60) secretion and protein arginine methyltransferase 1 (PRMT1) in fibroblasts. Eur. Respir. J. 2019, 54, 1900300. [Google Scholar] [CrossRef]
- Haj-Salem, I.; Plante, S.; Gounni, A.S.; Rouabhia, M.; Chakir, J. Fibroblast-derived exosomes promote epithelial cell proliferation through TGF-β2 signalling pathway in severe asthma. Allergy 2018, 73, 178–186. [Google Scholar] [CrossRef]
- Wu, X.; van Dijk, E.M.; Ng-Blichfeldt, J.P.; Bos, I.S.T.; Ciminieri, C.; Königshoff, M.; Kistemaker, L.E.M.; Gosens, R. Mesenchymal WNT-5A/5B Signaling Represses Lung Alveolar Epithelial Progenitors. Cells 2019, 8, E1147. [Google Scholar] [CrossRef] [Green Version]
- He, M.Z.; Zeng, X.; Zhang, K.; Kinney, P.L. Fine Particulate Matter Concentrations in Urban Chinese Cities, 2005–2016: A Systematic Review. Int. J. Environ. Res. Public Health 2017, 14, E191. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, A.; Agrawal, M. A Global Perspective of Fine Particulate Matter Pollution and Its Health Effects. Rev. Environ. Contam. Toxicol. 2018, 244, 5–51. [Google Scholar]
- Anenberg, S.C.; Henze, D.K.; Tinney, V.; Kinney, P.L.; Raich, W.; Fann, N.; Malley, C.S.; Roman, H.; Lamsal, L.; Duncan, B.; et al. Estimates of the Global Burden of Ambient Ozone, and on Asthma Incidence and Emergency Room Visits. Environ. Health Perspect. 2018, 126, 107004. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Dorans, K.S.; Wilker, E.H.; Rice, M.B.; Ljungman, P.L.; Schwartz, J.D.; Coull, B.A.; Koutrakis, P.; Gold, D.R.; Keaney, J.F., Jr.; et al. Short-Term Exposure to Ambient Air Pollution and Biomarkers of Systemic Inflammation: The Framingham Heart Study. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1793–1800. [Google Scholar] [CrossRef] [Green Version]
- Qin, F.; Yang, Y.; Wang, S.T.; Dong, Y.N.; Xu, M.X.; Wang, Z.W.; Zhao, J.X. Exercise and air pollutants exposure: A systematic review and meta-analysis. Life Sci. 2019, 218, 153–164. [Google Scholar] [CrossRef]
- Salimi, F.; Morgan, G.; Rolfe, M.; Samoli, E.; Cowie, C.T.; Hanigan, I.; Knibbs, L.; Cope, M.; Johnston, F.H.; Guo, Y.; et al. Long-term exposure to low concentrations of air pollutants and hospitalisation for respiratory diseases: A prospective cohort study in Australia. Environ. Int. 2018, 121, 415–420. [Google Scholar] [CrossRef]
- Finke, I.; de Jongste, J.C.; Smit, H.A.; Wijga, A.H.; Koppelman, G.H.; Vonk, J.; Brunekreef, B.; Gehring, U. Air pollution and airway resistance at age 8 years—The PIAMA birth cohort study. Environ. Health 2018, 17, 61. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Georas, S.; Alexis, N.; Fritz, P.; Xia, T.; Williams, M.A.; Horner, E.; Nel, A. A work group report on ultrafine particles (American Academy of Allergy, Asthma & Immunology): Why ambient ultrafine and engineered nanoparticles should receive special attention for possible adverse health outcomes in human subjects. J. Allergy Clin. Immunol. 2016, 138, 386–396. [Google Scholar]
- Veras, M.M.; de Oliveira Alves, N.; Fajersztajn, L.; Saldiva, P. Before the first breath: Prenatal exposures to air pollution and lung development. Cell Tissue Res. 2017, 367, 445–455. [Google Scholar] [CrossRef]
- Dysart, M.M.; Galvis, B.R.; Russell, A.G.; Barker, T.H. Environmental particulate (PM2.5) augments stiffness-induced alveolar epithelial cell mechanoactivation of transforming growth factor beta. PLoS ONE 2014, 9, e106821. [Google Scholar] [CrossRef] [Green Version]
- Song, Y.; Li, R.; Zhang, Y.; Wei, J.; Chen, W.; Chung, C.K.A.; Cai, Z. Mass spectrometry-based metabolomics reveals the mechanism of ambient fine particulate matter and its components on energy metabolic reprogramming in BEAS-2B cells. Sci. Total Environ. 2019, 651, 3139–3150. [Google Scholar] [CrossRef]
- Guo, Z.Q.; Dong, W.Y.; Xu, J.; Hong, Z.C.; Zhao, R.W.; Deng, C.R.; Zhuang, G.S.; Zhang, R.X. T-helper type 1-T-helper type 2 shift and nasal remodeling after fine particulate matter exposure in a rat model of allergic rhinitis. Am. J. Rhinol. Allergy 2017, 31, 148–155. [Google Scholar] [CrossRef]
- Li, R.; Kou, X.; Geng, H.; Xie, J.; Yang, Z.; Zhang, Y.; Cai, Z.; Dong, C. Effect of ambient PM(2.5) on lung mitochondrial damage and fusion/fission gene expression in rats. Chem. Res. Toxicol. 2015, 28, 408–418. [Google Scholar] [CrossRef]
- Soberanes, S.; Urich, D.; Baker, C.M.; Burgess, Z.; Chiarella, S.E.; Bell, E.L.; Ghio, A.J.; De Vizcaya-Ruiz, A.; Liu, J.; Ridge, K.M.; et al. Mitochondrial complex III-generated oxidants activate ASK1 and JNK to induce alveolar epithelial cell death following exposure to particulate matter air pollution. J. Biol. Chem. 2009, 284, 2176–2186. [Google Scholar] [CrossRef] [Green Version]
- Zhou-Suckow, Z.; Duerr, J.; Hagner, M.; Agrawal, R.; Mall, M.A. Airway mucus, inflammation and remodeling: Emerging links in the pathogenesis of chronic lung diseases. Cell Tissue Res. 2017, 367, 537–550. [Google Scholar] [CrossRef]
- Upadhyay, D.; Panduri, V.; Ghio, A.; Kamp, D.W. Particulate matter induces alveolar epithelial cell DNA damage and apoptosis: Role of free radicals and the mitochondria. Am. J. Respir. Cell Mol. Biol. 2003, 29, 180–187. [Google Scholar] [CrossRef] [Green Version]
- Benmerzoug, S.; Rose, S.; Bounab, B.; Gosset, D.; Duneau, L.; Chenuet, P.; Mollet, L.; Le Bert, M.; Lambers, C.; Geleff, S.; et al. STING-dependent sensing of self-DNA drives silica-induced lung inflammation. Nat. Commun. 2018, 9, 5226. [Google Scholar] [CrossRef]
- He, M.; Ichinose, T.; Yoshida, Y.; Arashidani, K.; Yoshida, S.; Takano, H.; Sun, G.; Shibamoto, T. Urban PM2.5 exacerbates allergic inflammation in the murine lung via a TLR2/TLR4/MyD88-signaling pathway. Sci. Rep. 2017, 7, 11027. [Google Scholar] [CrossRef] [Green Version]
- Ogino, K.; Nagaoka, K.; Ito, T.; Takemoto, K.; Okuda, T.; Nakayama, S.F.; Ogino, N.; Seki, Y.; Hamada, H.; Takashiba, S.; et al. Involvement of PM2.5-bound protein and metals in PM2.5-induced allergic airway inflammation in mice. Inhal. Toxicol. 2018, 30, 498–508. [Google Scholar] [CrossRef]
- Dai, P.; Shen, D.; Shen, J.; Tang, Q.; Xi, M.; Li, Y.; Li, C. The roles of Nrf2 and autophagy in modulating inflammation mediated by TLR4-NFκB in A549 cell exposed to layer house particulate matter 2.5 (PM(2.5)). Chemosphere 2019, 235, 1134–1145. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, C.; Wei, X.; Li, P.; Cui, Y.; Qin, Y.; Wei, X.; Jin, M.; Kohama, K.; Gao, Y. Heat shock protein 60 stimulates the migration of vascular smooth muscle cells via Toll-like receptor 4 and ERK MAPK activation. Sci. Rep. 2015, 5, 15352. [Google Scholar] [CrossRef]
- Gref, A.; Merid, S.K.; Gruzieva, O.; Ballereau, S.; Becker, A.; Bellander, T.; Bergström, A.; Bossé, Y.; Bottai, M.; Chan-Yeung, M.; et al. Genome-wide Interaction Analysis of Air Pollution Exposure and Childhood Asthma with Functional Follow-up. Am. J. Respir. Crit. Care Med. 2017, 195, 1373–1383. [Google Scholar] [CrossRef]
- Korten, I.; Ramsey, K.; Latzin, P. Air pollution during pregnancy and lung development in the child. Paediatr. Respir. Rev. 2017, 21, 38–46. [Google Scholar] [CrossRef]
- Janssen, B.G.; Byun, H.M.; Gyselaers, W.; Lefebvre, W.; Baccarelli, A.A.; Nawrot, T.S. Placental mitochondrial methylation and exposure to airborne particulate matter in the early life environment: An ENVIRONAGE birth cohort study. Epigenetics 2015, 10, 536–544. [Google Scholar] [CrossRef] [Green Version]
- Mei, M.; Song, H.; Chen, L.; Hu, B.; Bai, R.; Xu, D.; Liu, Y.; Zhao, Y.; Chen, C. Early-life exposure to three size-fractionated ultrafine and fine atmospheric particulates in Beijing exacerbates asthma development in mature mice. Part. Fibre Toxicol. 2018, 15, 13. [Google Scholar] [CrossRef] [Green Version]
- Arshad, S.H.; Karmaus, W.; Zhang, H.; Holloway, J.W. Multigenerational cohorts in patients with asthma and allergy. J. Allergy Clin. Immunol. 2017, 139, 415–421. [Google Scholar] [CrossRef] [Green Version]
- Lund, R.J.; Osmala, M.; Malonzo, M.; Lukkarinen, M.; Leino, A.; Salmi, J.; Vuorikoski, S.; Turunen, R.; Vuorinen, T.; Akdis, C.; et al. Atopic asthma after rhinovirus-induced wheezing is associated with DNA methylation change in the SMAD3 gene promoter. Allergy 2018, 73, 1735–1740. [Google Scholar] [CrossRef] [Green Version]
- Spalluto, C.M.; Singhania, A.; Cellura, D.; Woelk, C.H.; Sanchez-Elsner, T.; Staples, K.J.; Wilkinson, T.M.A. IFN-γ Influences Epithelial Antiviral Responses via Histone Methylation of the RIG-I Promoter. Am. J. Respir. Cell Mol. Biol. 2017, 57, 428–438. [Google Scholar] [CrossRef] [Green Version]
- Tantoh, D.M.; Lee, K.J.; Nfor, O.N.; Liaw, Y.C.; Lin, C.; Chu, H.W.; Chen, P.H.; Hsu, S.Y.; Liu, W.H.; Ho, C.C.; et al. Methylation at cg05575921 of a smoking-related gene (AHRR) in non-smoking Taiwanese adults residing in areas with different PM(2.5) concentrations. Clin. Epigenet. 2019, 11, 69. [Google Scholar] [CrossRef]
- Leclercq, B.; Kluza, J.; Antherieu, S.; Sotty, J.; Alleman, L.Y.; Perdrix, E.; Loyens, A.; Coddeville, P.; Lo Guidice, J.M.; Marchetti, P.; et al. Air pollution-derived PM(2.5) impairs mitochondrial function in healthy and chronic obstructive pulmonary diseased human bronchial epithelial cells. Environ. Pollut. 2018, 243, 1434–1449. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, L.; Mandal, J.; Molino, A.; Stolz, D.; Tamm, M.; Lu, S.; Roth, M. PDGF-BB induces PRMT1 expression through ERK1/2 dependent STAT1 activation and regulates remodeling in primary human lung fibroblasts. Cell Signal. 2016, 28, 307–315. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, L.; Wang, H.; Mandal, J.; Khan, P.; Hostettler, K.E.; Stolz, D.; Tamm, M.; Molino, A.; Lardinois, D.; et al. Constitutive high expression of protein arginine methyltransferase 1 in asthmatic airway smooth muscle cells is caused by reduced microRNA-19a expression and leads to enhanced remodeling. J. Allergy Clin. Immunol. 2017, 140, 510–524. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.; Fang, L.; Tang, X.; Lu, S.; Tamm, M.; Stolz, D.; Roth, M. TGF-β Upregulated Mitochondria Mass through the SMAD2/3→C/EBPβ→PRMT1 Signal Pathway in Primary Human Lung Fibroblasts. J. Immunol. 2019, 202, 37–47. [Google Scholar] [CrossRef]
- Liu, L.; Sun, Q.; Bao, R.; Roth, M.; Zhong, B.; Lan, X.; Tian, J.; He, Q.; Li, D.; Sun, J.; et al. Specific regulation of PRMT1 expression by PIAS1 and RKIP in BEAS-2B epithelia cells and HFL-1 fibroblasts in lung inflammation. Sci. Rep. 2016, 6, 21810. [Google Scholar] [CrossRef] [Green Version]
- Zou, W.; He, F.; Liu, S.; Pu, J.; Hu, J.; Sheng, Q.; Zhu, T.; Zhu, T.; Li, B.; Ran, P. PM2.5 Induced the Expression of Fibrogenic Mediators via HMGB1-RAGE Signaling in Human Airway Epithelial Cells. Can. Respir. J. 2018, 2018, 1817398. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Mittelstein, D.; Kam, W.; Pakbin, P.; Du, Y.; Tintut, Y.; Navab, M.; Sioutas, C.; Hsiai, T. Atmospheric ultrafine particles promote vascular calcification via the NF-κB signaling pathway. Am. J. Physiol. Cell Physiol. 2013, 304, C362–C369. [Google Scholar] [CrossRef] [Green Version]
- Wan, Q.; Liu, Z.; Yang, Y. Puerarin inhibits vascular smooth muscle cells proliferation induced by fine particulate matter via suppressing of the p38 MAPK signaling pathway. BMC Complement. Altern. Med. 2018, 18, 146. [Google Scholar] [CrossRef] [Green Version]
- Ye, X.; Hong, W.; Hao, B.; Peng, G.; Huang, L.; Zhao, Z.; Zhou, Y.; Zheng, M.; Li, C.; Liang, C.; et al. PM2.5 promotes human bronchial smooth muscle cell migration via the sonic hedgehog signaling pathway. Respir. Res. 2018, 19, 37. [Google Scholar] [CrossRef] [Green Version]
- Weng, C.M.; Lee, M.J.; He, J.R.; Chao, M.W.; Wang, C.H.; Kuo, H.P. Diesel exhaust particles up-regulate interleukin-17A expression via ROS/NF-κB in airway epithelium. Biochem. Pharmacol. 2018, 151, 1–8. [Google Scholar] [CrossRef]
- Ogawa, H.; Azuma, M.; Tsunematsu, T.; Morimoto, Y.; Kondo, M.; Tezuka, T.; Nishioka, Y.; Tsuneyama, K. Neutrophils induce smooth muscle hyperplasia via neutrophil elastase-induced FGF-2 in a mouse model of asthma with mixed inflammation. Clin. Exp. Allergy 2018, 48, 1715–1725. [Google Scholar] [CrossRef]
- Kang, Y.; Wang, F.; Lu, Z.; Ying, H.; Zhang, H.; Ding, W.; Wang, C.; Shi, L. MAPK kinase 3 potentiates Chlamydia HSP60-induced inflammatory response through distinct activation of NF-κB. J. Immunol. 2013, 191, 386–394. [Google Scholar] [CrossRef] [Green Version]
- Huittinen, T.; Hahn, D.; Anttila, T.; Wahlström, E.; Saikku, P.; Leinonen, M. Host immune response to Chlamydia pneumoniae heat shock protein 60 is associated with asthma. Eur. Respir. J. 2001, 17, 1078–1082. [Google Scholar] [CrossRef] [Green Version]
- Gencay, M.M.; Tamm, M.; Glanville, A.; Perruchoud, A.P.; Roth, M. Chlamydia pneumoniae activates epithelial cell proliferation via NF-kappaB and the glucocorticoid receptor. Infect. Immun. 2003, 71, 5814–5822. [Google Scholar] [CrossRef] [Green Version]
- Morishima, Y.; Mehta, R.K.; Yoshimura, M.; Lau, M.; Southworth, D.R.; Lawrence, T.S.; Pratt, W.B.; Nyati, M.K.; Osawa, Y. Chaperone Activity and Dimerization Properties of Hsp90α and Hsp90β in Glucocorticoid Receptor Activation by the Multiprotein Hsp90/Hsp70-Dependent Chaperone Machinery. Mol. Pharmacol. 2018, 94, 984–991. [Google Scholar] [CrossRef] [Green Version]
- Kooti, W.; Abdi, M.; Malik, Y.S.; Nouri, B.; Jalili, A.; Rezaee, M.A.; Rahmani, M.R.; Kalmarzi, R.N. Association of CYP2C19 and HSP70 genes polymorphism with aspirin-exacerbated respiratory disease in Kurdistan province. Endocr. Metab. Immune Disord. Drug Targets 2019. [Google Scholar] [CrossRef]
- Liu, L.; Huang, Y.; Feng, X.; Chen, J.; Duan, Y. Overexpressed Hsp70 alleviated formaldehyde-induced apoptosis partly via PI3K/Akt signaling pathway in human bronchial epithelial cells. Environ. Toxicol. 2019, 34, 495–504. [Google Scholar] [CrossRef]
- Shevchenko, M.A.; Troyanova, N.I.; Servuli, E.A.; Bolkhovitina, E.L.; Fedorina, A.S.; Sapozhnikov, A.M. Study of Immunomodulatory Effects of Extracellular HSP70 in a Mouse Model of Allergic Airway Inflammation. Biochemistry 2016, 81, 1384–1395. [Google Scholar] [CrossRef]
- Vercelli, D. Are we what our mothers made us? Lessons from epigenetics. J. Allergy Clin. Immunol. 2018, 141, 525–526. [Google Scholar] [CrossRef]
- Song, J.H.; Shim, A.; Kim, Y.J.; Ahn, J.H.; Kwon, B.E.; Pham, T.T.; Lee, J.; Chang, S.Y.; Ko, H.J. Antiviral and Anti-Inflammatory Activities of Pochonin D, a Heat Shock Protein 90 Inhibitor, against Rhinovirus Infection. Biomol. Ther. 2018, 26, 576–583. [Google Scholar] [CrossRef]
- Ye, C.; Huang, C.; Zou, M.; Hu, Y.; Luo, L.; Wei, Y.; Wan, X.; Zhao, H.; Li, W.; Cai, S.; et al. The role of secreted Hsp90α in HDM-induced asthmatic airway epithelial barrier dysfunction. BMC Pulm. Med. 2019, 19, 218. [Google Scholar] [CrossRef]
- Dong, H.M.; Le, Y.Q.; Wang, Y.H.; Zhao, H.J.; Huang, C.W.; Hu, Y.H.; Luo, L.S.; Wan, X.; Wei, Y.L.; Chu, Z.Q.; et al. Extracellular heat shock protein 90α mediates HDM-induced bronchial epithelial barrier dysfunction by activating RhoA/MLC signaling. Respir. Res. 2017, 18, 111. [Google Scholar] [CrossRef] [Green Version]
- Shende, P.; Bhandarkar, S.; Prabhakar, B. Heat Shock Proteins and their Protective Roles in Stem Cell Biology. Stem Cell Rev. Rep. 2019, 15, 637–651. [Google Scholar] [CrossRef]
- Dietz, K.; de Los Reyes Jiménez, M.; Gollwitzer, E.S.; Chaker, A.M.; Zissler, U.M.; Rådmark, O.P.; Baarsma, H.A.; Königshoff, M.; Schmidt-Weber, C.B.; Marsland, B.J.; et al. Age dictates a steroid-resistant cascade of Wnt5a, transglutaminase 2, and leukotrienes in inflamed airways. J. Allergy Clin. Immunol. 2017, 139, 1343–1354. [Google Scholar] [CrossRef] [Green Version]
- Brandsma, C.A.; de Vries, M.; Costa, R.; Woldhuis, R.R.; Königshoff, M.; Timens, W. Lung ageing and COPD: Is there a role for ageing in abnormal tissue repair? Eur. Respir. Rev. 2017, 26, 170073. [Google Scholar] [CrossRef] [Green Version]
- Ilmarinen, P.; Tuomisto, L.E.; Kankaanranta, H. Phenotypes, Risk Factors, and Mechanisms of Adult-Onset Asthma. Mediat. Inflamm. 2015, 2015, 514868. [Google Scholar] [CrossRef] [Green Version]
- Becerril-Ángeles, M.; Vargas, M.H.; Medina-Reyes, I.S.; Rascón-Pacheco, R.A. Factors related to changes in the asthma male: Female ratio by age in 403 Mexican counties. Ann. Allergy Asthma Immunol. 2019, 123, 288–292. [Google Scholar] [CrossRef]
- Zein, J.G.; Denson, J.L.; Wechsler, M.E. Asthma over the Adult Life Course: Gender and Hormonal Influences. Clin. Chest Med. 2019, 40, 149–161. [Google Scholar] [CrossRef]
- Carpaij, O.A.; van den Berge, M. The asthma-obesity relationship: Underlying mechanisms and treatment implications. Curr. Opin. Pulm. Med. 2018, 24, 42–49. [Google Scholar] [CrossRef]
- Gomez-Llorente, M.A.; Romero, R.; Chueca, N.; Martinez-Cañavate, A.; Gomez-Llorente, C. Obesity and Asthma: A Missing Link. Int. J. Mol. Sci. 2017, 18, 1490. [Google Scholar] [CrossRef] [Green Version]
- Karmaus, W.; Ziyab, A.H.; Everson, T.; Holloway, J.W. Epigenetic mechanisms and models in the origins of asthma. Curr. Opin. Allergy Clin. Immunol. 2013, 13, 63–69. [Google Scholar] [CrossRef] [Green Version]
- DeVries, A.; Wlasiuk, G.; Miller, S.J.; Bosco, A.; Stern, D.A.; Lohman, I.C.; Rothers, J.; Jones, A.C.; Nicodemus-Johnson, J.; Vasquez, M.M.; et al. Epigenome-wide analysis links SMAD3 methylation at birth to asthma in children of asthmatic mothers. J. Allergy Clin. Immunol. 2017, 140, 534–542. [Google Scholar] [CrossRef] [Green Version]
- Ji, H.; Biagini Myers, J.M.; Brandt, E.B.; Brokamp, C.; Ryan, P.H.; Khurana Hershey, G.K. Air pollution, epigenetics, and asthma. Allergy Asthma Clin. Immunol. 2016, 12, 51. [Google Scholar] [CrossRef] [Green Version]
- Kabesch, M. Early origins of asthma (and allergy). Mol. Cell. Pediatr. 2016, 3, 31. [Google Scholar] [CrossRef] [Green Version]
- Rider, C.F.; Carlsten, C. Air pollution and DNA methylation: Effects of exposure in humans. Clin. Epigenet. 2019, 11, 131. [Google Scholar] [CrossRef] [Green Version]
- Holgate, S.T. Genetic and environmental interaction in allergy and asthma. J. Allergy Clin. Immunol. 1999, 104, 1139–1146. [Google Scholar] [CrossRef]
- den Dekker, H.T.; Burrows, K.; Felix, J.F.; Salas, L.A.; Nedeljkovic, I.; Yao, J.; Rifas-Shiman, S.L.; Ruiz-Arenas, C.; Amin, N.; Bustamante, M.; et al. Newborn DNA-methylation, childhood lung function, and the risks of asthma and COPD across the life course. Eur. Respir. J. 2019, 53, 1801795. [Google Scholar] [CrossRef] [Green Version]
- Xu, C.J.; Söderhäll, C.; Bustamante, M.; Baïz, N.; Gruzieva, O.; Gehring, U.; Mason, D.; Chatzi, L.; Basterrechea, M.; Llop, S.; et al. DNA methylation in childhood asthma: An epigenome-wide meta-analysis. Lancet Respir. Med. 2018, 6, 379–388. [Google Scholar] [CrossRef] [Green Version]
- DeVries, A.; Vercelli, D. Of pleiotropy and trajectories: Does the TGF-β pathway link childhood asthma and chronic obstructive pulmonary disease? J. Allergy Clin. Immunol. 2018, 141, 1992–1996. [Google Scholar] [CrossRef] [Green Version]
- Wu, C.C.; Hsu, T.Y.; Chang, J.C.; Ou, C.Y.; Kuo, H.C.; Liu, C.A.; Wang, C.L.; Chuang, H.; Chen, C.P.; Yang, K.D. Paternal Tobacco Smoke Correlated to Offspring Asthma and Prenatal Epigenetic Programming. Front. Genet. 2019, 10, 471. [Google Scholar] [CrossRef] [Green Version]
- Peng, C.; Van Meel, E.R.; Cardenas, A.; Rifas-Shiman, S.L.; Sonawane, S.L.; Sonawane, A.R.; Glasse, K.R.; Gold, D.R.; Platts-Mills, T.A.; Platts-Mills, T.A.; et al. Epigenome-wide association study reveals methylation pathways associated with childhood allergic sensitization. Epigenetics 2019, 14, 445–466. [Google Scholar] [CrossRef] [PubMed]
- Reese, S.E.; Xu, C.J.; den Dekker, H.T.; Lee, M.K.; Sikdar, S.; Ruiz-Arenas, C.; Merid, S.K.; Rezwan, F.I.; Page, C.M.; Ullemar, V.; et al. Epigenome-wide meta-analysis of DNA methylation and childhood asthma. J. Allergy Clin. Immunol. 2019, 143, 2062–2074. [Google Scholar] [CrossRef] [PubMed] [Green Version]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, L.; Sun, Q.; Roth, M. Immunologic and Non-Immunologic Mechanisms Leading to Airway Remodeling in Asthma. Int. J. Mol. Sci. 2020, 21, 757. https://doi.org/10.3390/ijms21030757
Fang L, Sun Q, Roth M. Immunologic and Non-Immunologic Mechanisms Leading to Airway Remodeling in Asthma. International Journal of Molecular Sciences. 2020; 21(3):757. https://doi.org/10.3390/ijms21030757
Chicago/Turabian StyleFang, Lei, Qinzhu Sun, and Michael Roth. 2020. "Immunologic and Non-Immunologic Mechanisms Leading to Airway Remodeling in Asthma" International Journal of Molecular Sciences 21, no. 3: 757. https://doi.org/10.3390/ijms21030757





