The Rho/Rac Guanine Nucleotide Exchange Factor Vav1 Regulates Hif-1α and Glut-1 Expression and Glucose Uptake in the Brain
Abstract
:1. Introduction
2. Results
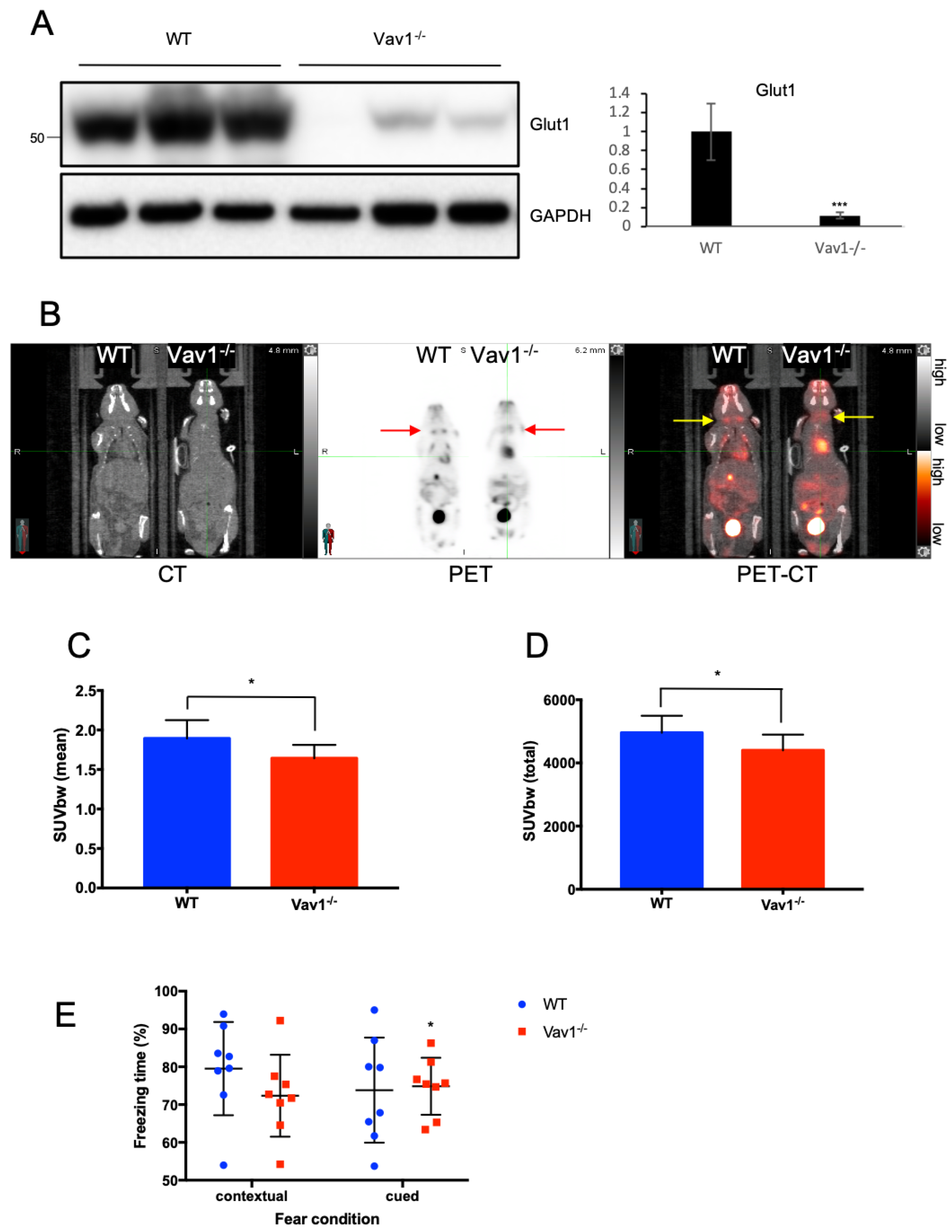
2.1. Vav1 Regulates HIF-1α and GLUT-1 in Endothelial Cells
2.2. Vav1 Regulates Glucose Uptake in Endothelial Cells
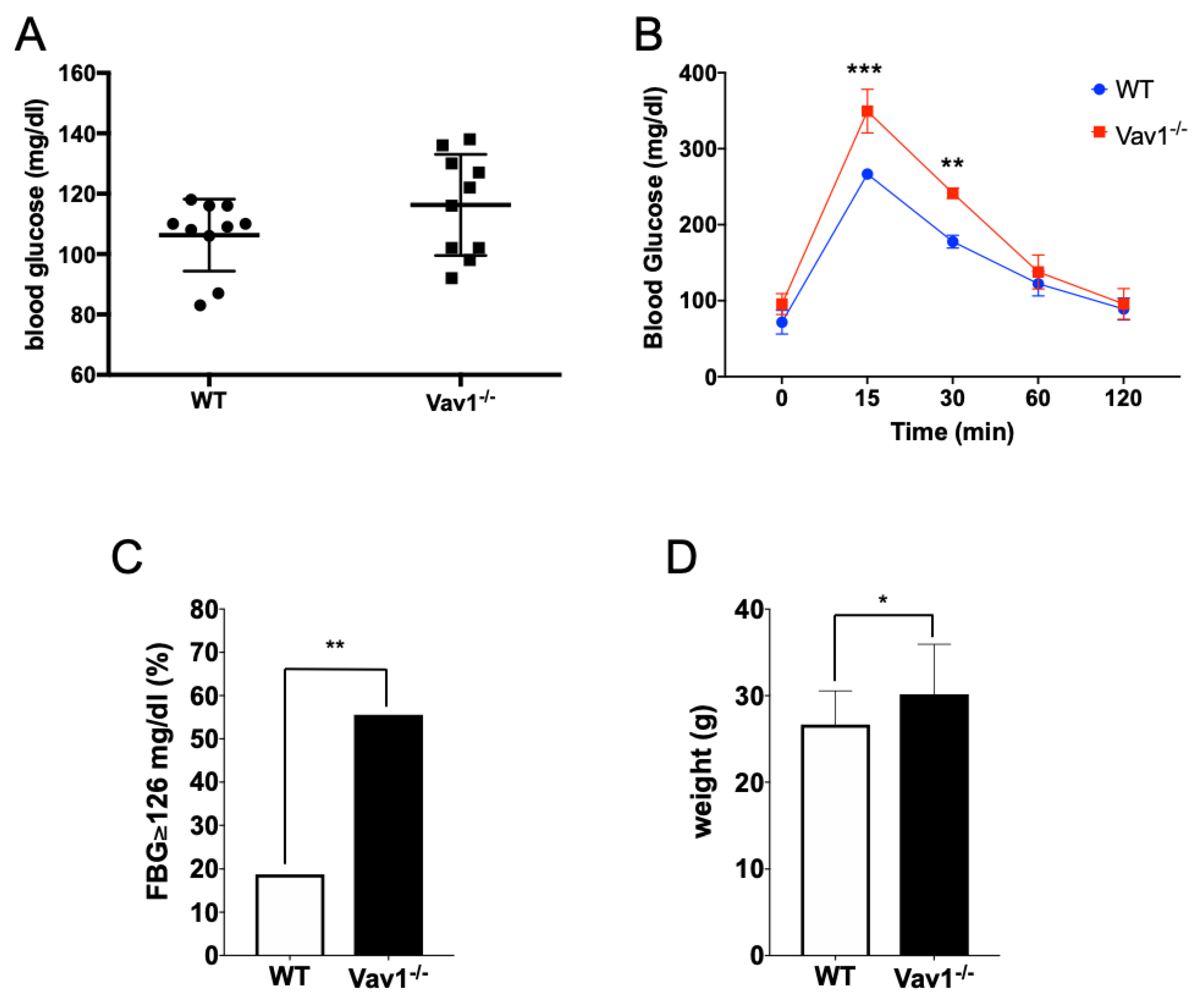
2.3. Vav1 Deficiency Delays Glucose Uptake In Vivo
2.4. Vav1 Deficiency Downregulates Glucose Distribution in the Brain
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Transfection and Lentiviral Transduction
4.3. Glucose Uptake Assay
4.4. Western Blot
4.5. RT-qPCR
4.6. Experimental Animals
4.7. Fear Conditioning
4.8. PET/CT Scanning
4.9. Histology
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Carruthers, A.; DeZutter, J.; Ganguly, A.; Devaskar, S.U. Will the original glucose transporter isoform please stand up! Am. J. Physiol. Endocrinol. Metab. 2009, 297, E836–E848. [Google Scholar] [CrossRef] [Green Version]
- Seidner, G.; Alvarez, M.G.; Yeh, J.I.; O’Driscoll, K.R.; Klepper, J.; Stump, T.S.; Wang, D.; Spinner, N.B.; Birnbaum, M.J.; De Vivo, D.C. GLUT-1 deficiency syndrome caused by haploinsufficiency of the blood-brain barrier hexose carrier. Nat. Genet. 1998, 18, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Thorens, B.; Mueckler, M. Glucose transporters in the 21st Century. Am. J. Physiol. Endocrinol. Metab. 2010, 298, E141–E145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westermeier, F.; Puebla, C.; Vega, J.L.; Farias, M.; Escudero, C.; Casanello, P.; Sobrevia, L. Equilibrative nucleoside transporters in fetal endothelial dysfunction in diabetes mellitus and hyperglycaemia. Curr. Vasc. Pharmacol. 2009, 7, 435–449. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lei, L.; Liu, D.; Jovin, I.; Russell, R.; Johnson, R.S.; Di Lorenzo, A.; Giordano, F.J. Normal glucose uptake in the brain and heart requires an endothelial cell-specific HIF-1alpha-dependent function. Proc. Natl. Acad. Sci. USA 2012, 109, 17478–17483. [Google Scholar] [CrossRef] [Green Version]
- Loike, J.D.; Cao, L.; Brett, J.; Ogawa, S.; Silverstein, S.C.; Stern, D. Hypoxia induces glucose transporter expression in endothelial cells. Am. J. Physiol. 1992, 263, C326–C333. [Google Scholar] [CrossRef] [PubMed]
- Olson, A.L.; Pessin, J.E. Structure, function, and regulation of the mammalian facilitative glucose transporter gene family. Annu. Rev. Nutr. 1996, 16, 235–256. [Google Scholar] [CrossRef]
- Wood, I.S.; Trayhurn, P. Glucose transporters (GLUT and SGLT): Expanded families of sugar transport proteins. Br. J. Nutr. 2003, 89, 3–9. [Google Scholar] [CrossRef]
- Guo, X.; Geng, M.; Du, G. Glucose transporter 1, distribution in the brain and in neural disorders: Its relationship with transport of neuroactive drugs through the blood-brain barrier. Biochem. Genet. 2005, 43, 175–187. [Google Scholar]
- Maher, F.; Vannucci, S.J.; Simpson, I.A. Glucose transporter isoforms in brain: Absence of GLUT3 from the blood-brain barrier. J. Cereb. Blood Flow Metab. 1993, 13, 342–345. [Google Scholar] [CrossRef] [Green Version]
- Pardridge, W.M.; Boado, R.J.; Farrell, C.R. Brain-type glucose transporter (GLUT-1) is selectively localized to the blood-brain barrier. Studies with quantitative western blotting and in situ hybridization. J. Biol. Chem. 1990, 265, 18035–18040. [Google Scholar] [PubMed]
- Wang, D.; Pascual, J.M.; Yang, H.; Engelstad, K.; Jhung, S.; Sun, R.P.; De Vivo, D.C. Glut-1 deficiency syndrome: Clinical, genetic, and therapeutic aspects. Ann. Neurol. 2005, 57, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.; Billadeau, D.D. VAV proteins as signal integrators for multi-subunit immune-recognition receptors. Nat. Rev. Immunol. 2002, 2, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Tybulewicz, V.L.; Ardouin, L.; Prisco, A.; Reynolds, L.F. Vav1: A key signal transducer downstream of the TCR. Immunol. Rev. 2003, 192, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Fischer, K.D.; Zmuldzinas, A.; Gardner, S.; Barbacid, M.; Bernstein, A.; Guidos, C. Defective T-cell receptor signalling and positive selection of Vav-deficient CD4+ CD8+ thymocytes. Nature 1995, 374, 474–477. [Google Scholar] [CrossRef]
- Turner, M.; Mee, P.J.; Walters, A.E.; Quinn, M.E.; Mellor, A.L.; Zamoyska, R.; Tybulewicz, V.L. A requirement for the Rho-family GTP exchange factor Vav in positive and negative selection of thymocytes. Immunity 1997, 7, 451–460. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Alt, F.W.; Davidson, L.; Orkin, S.H.; Swat, W. Defective signalling through the T- and B-cell antigen receptors in lymphoid cells lacking the vav proto-oncogene. Nature 1995, 374, 470–473. [Google Scholar] [CrossRef]
- DeBusk, L.M.; Boelte, K.; Min, Y.; Lin, P.C. Heterozygous deficiency of delta-catenin impairs pathological angiogenesis. J. Exp. Med. 2010, 207, 77–84. [Google Scholar] [CrossRef] [Green Version]
- Min, Y.; Hong, J.; Wuest, T.R.; Lin, P.C. Vav1 is essential for HIF-1α activation in vascular response to ischemic stress. bioRxiv 2019. [Google Scholar] [CrossRef] [Green Version]
- Roh, E.; Song, D.K.; Kim, M.S. Emerging role of the brain in the homeostatic regulation of energy and glucose metabolism. Exp. Mol. Med. 2016, 48, e216. [Google Scholar] [CrossRef] [Green Version]
- Roh, E.; Kim, M.S. Brain Regulation of Energy Metabolism. Endocrinol. Metab. 2016, 31, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhong, C. Decoding Alzheimer’s disease from perturbed cerebral glucose metabolism: Implications for diagnostic and therapeutic strategies. Prog. Neurobiol. 2013, 108, 21–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cunnane, S.; Nugent, S.; Roy, M.; Courchesne-Loyer, A.; Croteau, E.; Tremblay, S.; Castellano, A.; Pifferi, F.; Bocti, C.; Paquet, N.; et al. Brain fuel metabolism, aging, and Alzheimer’s disease. Nutrition 2011, 27, 3–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nugent, S.; Croteau, E.; Pifferi, F.; Fortier, M.; Tremblay, S.; Turcotte, E.; Cunnane, S.C. Brain and systemic glucose metabolism in the healthy elderly following fish oil supplementation. Prostaglandins Leukot Essent. Fatty Acids 2011, 85, 287–291. [Google Scholar] [CrossRef]
- Craft, S.; Baker, L.D.; Montine, T.J.; Minoshima, S.; Watson, G.S.; Claxton, A.; Arbuckle, M.; Callaghan, M.; Tsai, E.; Plymate, S.R.; et al. Intranasal insulin therapy for Alzheimer disease and amnestic mild cognitive impairment: A pilot clinical trial. Arch. Neurol. 2012, 69, 29–38. [Google Scholar] [CrossRef] [Green Version]
- Holscher, C. Central effects of GLP-1: New opportunities for treatments of neurodegenerative diseases. J. Endocrinol. 2014, 221, T31–T41. [Google Scholar] [CrossRef] [Green Version]
- Morris, J.K.; Burns, J.M. Insulin: An emerging treatment for Alzheimer’s disease dementia? Curr. Neurol. Neurosci. Rep. 2012, 12, 520–527. [Google Scholar] [CrossRef] [Green Version]
- Parthsarathy, V.; Holscher, C. Chronic treatment with the GLP1 analogue liraglutide increases cell proliferation and differentiation into neurons in an AD mouse model. PLoS ONE 2013, 8, e58784. [Google Scholar] [CrossRef] [Green Version]
- Reger, M.A.; Watson, G.S.; Green, P.S.; Wilkinson, C.W.; Baker, L.D.; Cholerton, B.; Fishel, M.A.; Plymate, S.R.; Breitner, J.C.; DeGroodt, W.; et al. Intranasal insulin improves cognition and modulates beta-amyloid in early AD. Neurology 2008, 70, 440–448. [Google Scholar] [CrossRef]
- Shingo, A.S.; Kanabayashi, T.; Kito, S.; Murase, T. Intracerebroventricular administration of an insulin analogue recovers STZ-induced cognitive decline in rats. Behav. Brain Res. 2013, 241, 105–111. [Google Scholar] [CrossRef]
- Talbot, K.; Wang, H.Y. The nature, significance, and glucagon-like peptide-1 analog treatment of brain insulin resistance in Alzheimer’s disease. Alzheimers Dement. 2014, 10, S12–S25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.S.; Kim, J.H.; Sun, B.K.; Song, S.U.; Suh, W.; Sung, J.H. Hypoxia induces glucose uptake and metabolism of adiposederived stem cells. Mol. Med. Rep. 2016, 14, 4706–4714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, O.; Li, X.; Qu, Y.; Liu, S.; An, J.; Wang, M.; Sun, Q.; Zhang, W.; Lu, X.; Pi, L.; et al. Regulation of glucose transporter protein-1 and vascular endothelial growth factor by hypoxia inducible factor 1alpha under hypoxic conditions in Hep-2 human cells. Mol. Med. Rep. 2012, 6, 1418–1422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carim-Todd, L.; Bath, K.G.; Fulgenzi, G.; Yanpallewar, S.; Jing, D.; Barrick, C.A.; Becker, J.; Buckley, H.; Dorsey, S.G.; Lee, F.S.; et al. Endogenous truncated TrkB.T1 receptor regulates neuronal complexity and TrkB kinase receptor function in vivo. J. Neurosci. 2009, 29, 678–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gras, D.; Roze, E.; Caillet, S.; Meneret, A.; Doummar, D.; Billette de Villemeur, T.; Vidailhet, M.; Mochel, F. GLUT1 deficiency syndrome: An update. Rev. Neurol. 2014, 170, 91–99. [Google Scholar] [CrossRef]
- Wijesekara, N.; Goncalves, R.A.; De Felice, F.G.; Fraser, P.E. Impaired peripheral glucose homeostasis and Alzheimer’s disease. Neuropharmacology 2018, 136, 172–181. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, J.; Kim, Y.; Yanpallewar, S.; Lin, P.C. The Rho/Rac Guanine Nucleotide Exchange Factor Vav1 Regulates Hif-1α and Glut-1 Expression and Glucose Uptake in the Brain. Int. J. Mol. Sci. 2020, 21, 1341. https://doi.org/10.3390/ijms21041341
Hong J, Kim Y, Yanpallewar S, Lin PC. The Rho/Rac Guanine Nucleotide Exchange Factor Vav1 Regulates Hif-1α and Glut-1 Expression and Glucose Uptake in the Brain. International Journal of Molecular Sciences. 2020; 21(4):1341. https://doi.org/10.3390/ijms21041341
Chicago/Turabian StyleHong, Jaewoo, Yurim Kim, Sudhirkumar Yanpallewar, and P. Charles Lin. 2020. "The Rho/Rac Guanine Nucleotide Exchange Factor Vav1 Regulates Hif-1α and Glut-1 Expression and Glucose Uptake in the Brain" International Journal of Molecular Sciences 21, no. 4: 1341. https://doi.org/10.3390/ijms21041341




