Metabolomics as an Approach to Characterise the Contrasting Roles of CCR5 in the Presence and Absence of Disease
Abstract
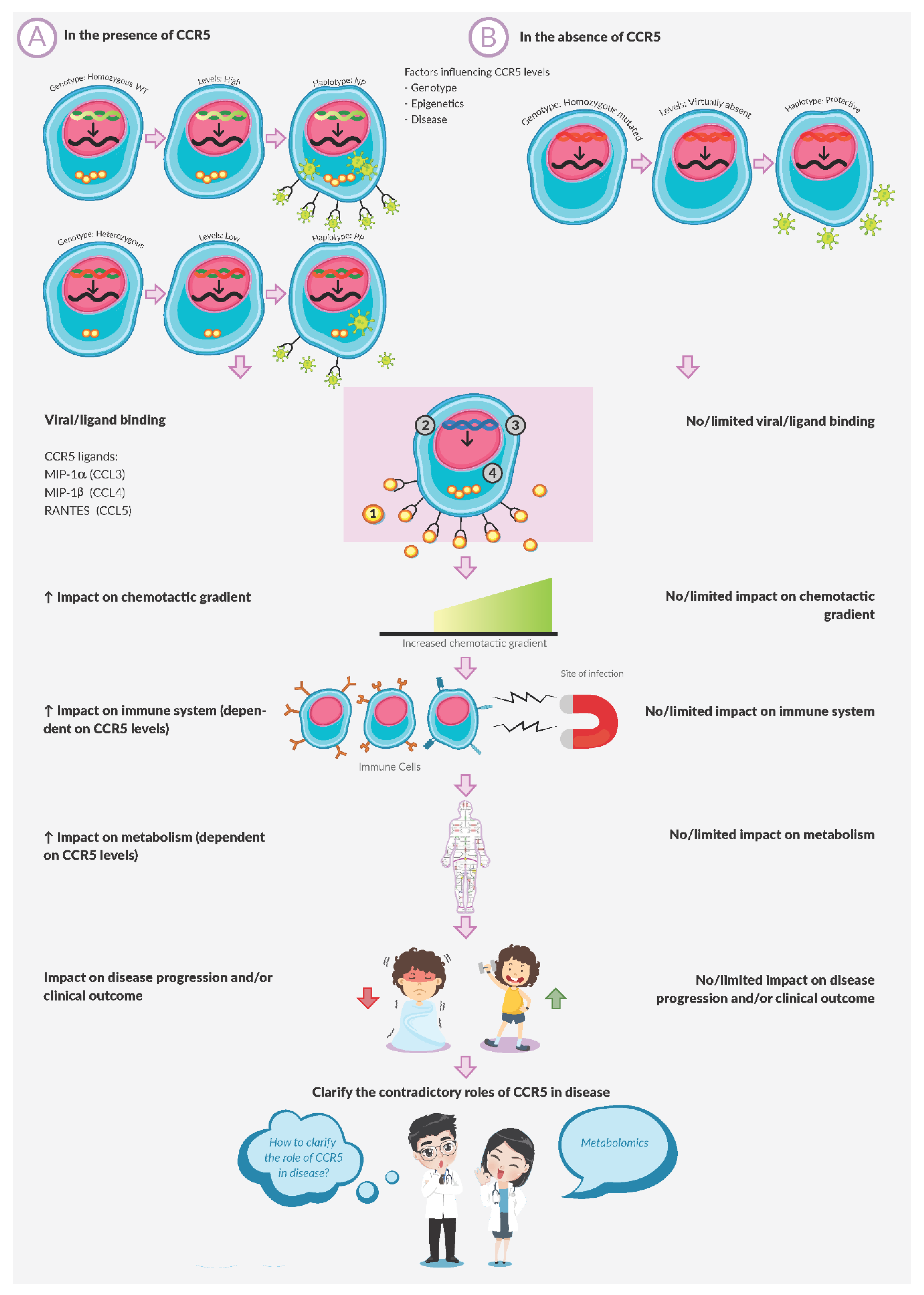
:1. Introduction
2. The Role of CCR5 in Disease
2.1. Cardiovascular Diseases
2.2. Diseases of the Nervous System
2.3. Immune System Diseases
2.4. Infectious Diseases
2.4.1. Acquired Immunodeficiency Syndrome
2.4.2. West Nile Fever
2.4.3. Hepatitis B Infection
2.4.4. Hepatitis C Infection
2.4.5. Chagas Disease
2.4.6. Influenza A
2.5. Cancer
3. Conclusions
Funding
Conflicts of Interest
Abbreviations
| CCR5 | C-C chemokine receptor 5 |
| kDa | kilodalton |
| CMI | Cell-mediated immunity |
| MIP1-α | Macrophage inflammatory protein one alpha |
| MIP1-β | Macrophage inflammatory protein one beta |
| RANTES | Regulated upon activation, normal T-cell expressed and secreted |
| CCL5 | C-C chemokine ligand 5 |
| MCP-2 | Monocyte chemotactic protein two |
| Th1 | T helper type 1 |
| Th2 | T helper type 2 |
| ATP | Adenosine triphosphate |
| LDL | Low-density lipoprotein |
| HDL | High-density lipoprotein |
| ∆32 | 32-base pair deletion |
| bp | Base pairs |
| WT | Wild type |
| HIV-1 | Human immunodeficiency virus |
| AIDS | Acquired immunodeficiency syndrome |
| CCR5+ | CCR5-positive |
| CRISPR | Clustered regularly interspaced short palindromic repeats |
| DNA | Deoxyribonucleic acid |
| UK | United Kingdom |
| CVD | Cardiovascular disease |
| MCP-1 | Monocyte chemoattractant protein-1 |
| CAD | Coronary artery disease |
| CRP | C-reactive protein |
| ELISA | Enzyme-linked immunosorbent assay |
| PCR | Polymerase chain reaction |
| HDL-C | High-density lipoprotein cholesterol |
| TG | Triglycerides |
| LDL-C | Low-density lipoprotein cholesterol |
| RFLP | Restriction fragment length polymorphism |
| MI | Myocardial infarction |
| ICAS | Ischemic carotid artery stenosis |
| AD | Alzheimer’s disease |
| PD | Parkinson’s disease |
| ms | Multiple sclerosis |
| RT-PCR | Reverse-transcription polymerase chain reaction |
| LOAD | Late-onset Alzheimer’s disease |
| mRNA | Messenger ribonucleic acid |
| IgE | Immunoglobulin E |
| T2DM | Type 2 diabetes mellitus |
| HbA1c | Haemoglobin A1c |
| T1DM | Type 1 diabetes mellitus |
| RA | Rheumatoid arthritis |
| RF | Rheumatoid factor |
| JIA | Juvenile idiopathic arthritis |
| SLE | Systemic lupus erythematosus |
| LN | Lupus nephritis |
| SDF-1 | Stromal cell-derived factor one |
| WNV | West Nile virus |
| WNF | West Nile fever |
| HBV | Hepatitis B virus |
| NK | Natural killer |
| HCV | Hepatitis C virus |
| CCR5- | CCR5-negative |
| HPV | Human papillomavirus |
| HPV+ | Human papillomavirus-positive |
| AML | Acute myeloid leukaemia |
| MS | Mass spectrometry |
| AS | Atherosclerosis |
| CHD | Coronary heart disease |
| CM | Cardiomyopathy |
| HT | Hypertension |
| NS | Nervous system |
| IS | Immune system |
| ID | Infectious disease |
| CD | Chagas disease |
| BC | Breast cancer |
| PanC | Pancreatic cancer |
| CC | Cervical cancer |
| GallC | Gallbladder cancer |
| GasC | Gastric cancer |
| PC | Prostate cancer |
| CCL3 | C-C chemokine ligand 3 |
| CCL4 | C-C chemokine ligand 4 |
| NP | Non-protective |
| PP | Partially protective |
References
- Groves, D.; Jiang, Y. Chemokines, a family of chemotactic cytokines. Crit. Rev. Oral Biol. Med. 1995, 6, 109–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachmann, M.F.; Kopf, M.; Marsland, B.J. Chemokines: More than just road signs. Nat. Rev. Immunol. 2006, 6, 159–164. [Google Scholar] [CrossRef]
- Gerard, C.; Rollins, B.J. Chemokines and disease. Nat. Immunol. 2001, 2, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Yoshie, O.; Imai, T.; Nomiyama, H. Chemokines in immunity. In Advances in Immunology; Elsevier: Amsterdam, The Netherlands, 2001; Volume 78, pp. 57–110. [Google Scholar]
- Guedes, J.R.; Lao, T.; Cardoso, A.L.; El Khoury, J. Roles of Microglial and Monocyte Chemokines and Their Receptors in Regulating Alzheimer’s Disease-Associated Amyloid-β and Tau Pathologies. Front. Neurol. 2018, 9, 549. [Google Scholar] [CrossRef]
- Ryu, H.; Baek, S.W.; Moon, J.Y.; Jo, I.S.; Kim, N.; Lee, H.J. C-C motif chemokine receptors in gastric cancer. Mol. Clin. Oncol. 2018, 8, 3–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raport, C.J.; Gosling, J.; Schweickart, V.L.; Gray, P.W.; Charo, I.F. Molecular cloning and functional characterization of a novel human CC chemokine receptor (CCR5) for RANTES, MIP-1β, and MIP-1α. J. Biol. Chem. 1996, 271, 17161–17166. [Google Scholar] [CrossRef] [Green Version]
- Samson, M.; Labbe, O.; Mollereau, C.; Vassart, G.; Parmentier, M. Molecular cloning and functional expression of a new human CC-chemokine receptor gene. Biochemistry 1996, 35, 3362–3367. [Google Scholar] [CrossRef]
- Oppermann, M. Chemokine receptor CCR5: Insights into structure, function, and regulation. Cell Signal 2004, 16, 1201–1210. [Google Scholar] [CrossRef]
- Rottman, J.B.; Ganley, K.P.; Williams, K.; Wu, L.; Mackay, C.R.; Ringler, D.J. Cellular localization of the chemokine receptor CCR5. Correlation to cellular targets of HIV-1 infection. Am. J. Pathol. 1997, 151, 1341. [Google Scholar]
- Nibbs, R.J.; Yang, J.; Landau, N.R.; Mao, J.-H.; Graham, G.J. LD78β, a non-allelic variant of human MIP-1α (LD78α), has enhanced receptor interactions and potent HIV suppressive activity. J. Biol. Chem. 1999, 274, 17478–17483. [Google Scholar] [CrossRef] [Green Version]
- Combadiere, C.; Ahuja, S.K.; Lee Tiffany, H.; Murphy, P.M. Cloning and functional expression of CC CKR5, a human monocyte CC chemokine receptor selective for MIP-1α, MIP-1β, and RANTES. J. Leukoc. Biol. 1996, 60, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Howard, O.Z.; Turpin, J.A.; Grimm, M.C.; Ueda, H.; Gray, P.W.; Raport, C.J.; Oppenheim, J.J.; Wang, J.M. Monocyte chemotactic protein-2 activates CCR5 and blocks CD4/CCR5-mediated HIV-1 entry/replication. J. Biol. Chem. 1998, 273, 4289–4292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachelerie, F.; Ben-Baruch, A.; Burkhardt, A.M.; Combadiere, C.; Farber, J.M.; Graham, G.J.; Horuk, R.; Sparre-Ulrich, A.H.; Locati, M.; Luster, A.D. International Union of Pharmacology. LXXXIX. Update on the extended family of chemokine receptors and introducing a new nomenclature for atypical chemokine receptors. Pharmacol. Rev. 2014, 66, 1–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lederman, M.M.; Penn-Nicholson, A.; Cho, M.; Mosier, D. Biology of CCR5 and its role in HIV infection and treatment. JAMA 2006, 296, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Stone, M. Regulation of Chemokine–Receptor Interactions and Functions. Int. J. Mol. Sci. 2017, 18, 2415. [Google Scholar] [CrossRef] [Green Version]
- Castellino, F.; Huang, A.Y.; Altan-Bonnet, G.; Stoll, S.; Scheinecker, C.; Germain, R.N. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell–dendritic cell interaction. Nature 2006, 440, 890–895. [Google Scholar] [CrossRef]
- Bonecchi, R.; Bianchi, G.; Bordignon, P.P.; D’Ambrosio, D.; Lang, R.; Borsatti, A.; Sozzani, S.; Allavena, P.; Gray, P.A.; Mantovani, A. Differential expression of chemokine receptors and chemotactic responsiveness of type 1 T helper cells (Th1s) and Th2s. J. Exp. Med. 1998, 187, 129–134. [Google Scholar] [CrossRef]
- Berger, A. Science commentary: Th1 and Th2 responses: What are they? Allergy 2000, 55, 2–10. [Google Scholar] [CrossRef] [Green Version]
- Loetscher, P.; Uguccioni, M.; Bordoli, L.; Baggiolini, M.; Moser, B.; Chizzolini, C.; Dayer, J.-M. CCR5 is characteristic of Th1 lymphocytes. Nature 1998, 391, 344–345. [Google Scholar] [CrossRef]
- Chan, O.; Burke, J.D.; Gao, D.F.; Fish, E.N. The chemokine CCL5 regulates glucose uptake and AMP kinase signaling in activated T cells to facilitate chemotaxis. J. Biol. Chem. 2012, 287, 29406–29416. [Google Scholar] [CrossRef] [Green Version]
- Gao, D.; Rahbar, R.; Fish, E.N. CCL5 activation of CCR5 regulates cell metabolism to enhance proliferation of breast cancer cells. Open Biol. 2016, 6, 160122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonfa, G.; Benevides, L.; Souza Mdo, C.; Fonseca, D.M.; Mineo, T.W.; Rossi, M.A.; Silva, N.M.; Silva, J.S.; de Barros Cardoso, C.R. CCR5 controls immune and metabolic functions during Toxoplasma gondii infection. PLoS ONE 2014, 9, e104736. [Google Scholar] [CrossRef] [Green Version]
- Bing, L.; Yu, F.; Yu, W.; Xie, Y. The expression and role of CC chemokine receptor type 5 in type II diabetes. Acta Med. Mediterr. 2018, 34, 669–675. [Google Scholar]
- Rosa, J.C.; Lira, F.S.; Festuccia, W.; Wessner, B.; Bishop, N.C. Immunometabolism: Molecular Mechanisms, Diseases, and Therapies 2018. Mediat. Inflamm. 2019, 2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghuraman, S.; Donkin, I.; Versteyhe, S.; Barres, R.; Simar, D. The Emerging Role of Epigenetics in Inflammation and Immunometabolism. Trends Endocrinol. Metab. 2016, 27, 782–795. [Google Scholar] [CrossRef]
- Ma, E.H.; Bantug, G.; Griss, T.; Condotta, S.; Johnson, R.M.; Samborska, B.; Mainolfi, N.; Suri, V.; Guak, H.; Balmer, M.L. Serine is an essential metabolite for effector T cell expansion. Cell Metab. 2017, 25, 345–357. [Google Scholar] [CrossRef]
- Pan, Y.; Tian, T.; Park, C.O.; Lofftus, S.Y.; Mei, S.; Liu, X.; Luo, C.; O’Malley, J.T.; Gehad, A.; Teague, J.E. Survival of tissue-resident memory T cells requires exogenous lipid uptake and metabolism. Nature 2017, 543, 252–256. [Google Scholar] [CrossRef] [Green Version]
- Martinson, J.J.; Chapman, N.H.; Rees, D.C.; Liu, Y.-T.; Clegg, J.B. Global distribution of the CCR5 gene 32-basepair deletion. Nat. Genet. 1997, 16, 100–103. [Google Scholar] [CrossRef]
- Dean, M.; Carrington, M.; Winkler, C.; Huttley, G.A.; Smith, M.W.; Allikmets, R.; Goedert, J.J.; Buchbinder, S.P.; Vittinghoff, E.; Gomperts, E. Genetic restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Science 1996, 273, 1856–1862. [Google Scholar] [CrossRef] [Green Version]
- Samson, M.; Libert, F.; Doranz, B.J.; Rucker, J.; Liesnard, C.; Farber, C.-M.; Saragosti, S.; Lapouméroulie, C.; Cognaux, J.; Forceille, C. Resistance to HIV-1 infection in caucasian individuals bearing mutant alleles of the CCR-5 chemokine receptor gene. Nature 1996, 382, 722–725. [Google Scholar] [CrossRef]
- Winkler, C.A.; Hendel, H.; Carrington, M.; Smith, M.W.; Nelson, G.W.; O’Brien, S.J.; Phair, J.; Vlahov, D.; Jacobson, L.P.; Rappaport, J. Dominant effects of CCR2-CCR5 haplotypes in HIV-1 disease progression. JAIDS J. Acquir. Immune Defic. Syndr. 2004, 37, 1534–1538. [Google Scholar] [CrossRef] [Green Version]
- Jaumdally, S.Z.; Picton, A.; Tiemessen, C.T.; Paximadis, M.; Jaspan, H.B.; Gamieldien, H.; Masson, L.; Coetzee, D.; Williamson, A.L.; Little, F.; et al. CCR5 expression, haplotype and immune activation in protection from infection in HIV-exposed uninfected individuals in HIV-serodiscordant relationships. Immunology 2017, 151, 464–473. [Google Scholar] [CrossRef]
- Reynes, J.; Portales, P.; Segondy, M.; Baillat, V.; André, P.; Réant, B.; Avinens, O.; Couderc, G.; Benkirane, M.; Clot, J. CD4+ T cell surface CCR5 density as a determining factor of virus load in persons infected with human immunodeficiency virus type 1. J. Infect. Dis. 2000, 181, 927–932. [Google Scholar] [CrossRef] [Green Version]
- Woitas, R.P.; Ahlenstiel, G.; Iwan, A.; Rockstroh, J.K.; Brackmann, H.H.; Kupfer§, B.; Matz§, B.; Offergeld, R.; Sauerbruch, T.; Spengler, U. Frequency of the HIV-protective CC chemokine receptor 5-Δ32/Δ32 genotype is increased in hepatitis C. Gastroenterology 2002, 122, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Paxton, W.A.; Wolinsky, S.M.; Neumann, A.U.; Zhang, L.; He, T.; Kang, S.; Ceradini, D.; Jin, Z.; Yazdanbakhsh, K. The role of a mutant CCR5 allele in HIV–1 transmission and disease progression. Nat. Med. 1996, 2, 1240–1243. [Google Scholar] [CrossRef]
- Marmor, M.; Sheppard, H.W.; Donnell, D.; Bozeman, S.; Celum, C. Homozygous and HeterozygousCCR5-Δ32Genotypes Are Associated With Resistance to HIV Infection. JAIDS J. Acquir. Immune Defic. Syndr. 2001, 27, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Munerato, P.; Azevedo, M.L.; Sucupira, M.C.A.; Pardini, R.; Pinto, G.H.N.; Catroxo, M.; Souza, I.E.; Diaz, R.S. Frequency of polymorphisms of genes coding for HIV-1 co-receptors CCR5 and CCR2 in a Brazilian population. Braz. J. Infect. Dis. 2003, 7, 236–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woollard, S.M.; Kanmogne, G.D. Maraviroc: A review of its use in HIV infection and beyond. Drug Des. Dev. Ther. 2015, 9, 5447. [Google Scholar]
- Vangelista, L.; Vento, S. The expanding therapeutic perspective of CCR5 blockade. Front. Immunol. 2018, 8, 1981. [Google Scholar] [CrossRef]
- Arendt, V.; Amand, M.; Iserentant, G.; Lemaire, M.; Masquelier, C.; Ndayisaba, G.F.; Verhofstede, C.; Karita, E.; Allen, S.; Chevigné, A. Predominance of the heterozygous CCR 5 delta-24 deletion in African individuals resistant to HIV infection might be related to a defect in CCR 5 addressing at the cell surface. J. Int. Aids Soc. 2019, 22, e25384. [Google Scholar] [CrossRef]
- An, P.; Martin, M.P.; Nelson, G.W.; Carrington, M.; Smith, M.W.; Gong, K.; Vlahov, D.; O’brien, S.J.; Winkler, C.A. Influence of CCR5 promoter haplotypes on AIDS progression in African–Americans. Aids 2000, 14, 2117–2122. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J.P.; Rosenberg, P.S.; Goedert, J.J.; Ashton, L.J.; Benfield, T.L.; Buchbinder, S.P.; Coutinho, R.A.; Eugen-Olsen, J.; Gallart, T.; Katzenstein, T.L. Effects of CCR5-Δ 32, CCR2-64I, and SDF-1 3′ A alleles on HIV-1 disease progression: An international meta-analysis of individual-patient data. Ann. Intern. Med. 2001, 135, 782–795. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, S.; Mimaya, J.-I.; Yamada, K.; Kurimura, T.; Shiraki, K. Polymorphism of CCR5 affecting HIV disease progression in the Japanese population. Aids Res. Hum. Retrovir. 2001, 17, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, T.; Gulden, F.O.; Sugaya, M.; McNamara, D.T.; Borris, D.L.; Lederman, M.M.; Orenstein, J.M.; Zimmerman, P.A.; Blauvelt, A. R5 HIV productively infects Langerhans cells, and infection levels are regulated by compound CCR5 polymorphisms. Proc. Natl. Acad. Sci. USA 2003, 100, 8401–8406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, D.; Choi, B.S.; Kim, S. The effects of RANTES/CCR5 promoter polymorphisms on HIV disease progression in HIV-infected Koreans. Int. J. Immunogenet. 2008, 35, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Hütter, G.; Nowak, D.; Mossner, M.; Ganepola, S.; Müßig, A.; Allers, K.; Schneider, T.; Hofmann, J.; Kücherer, C.; Blau, O. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. New Engl. J. Med. 2009, 360, 692–698. [Google Scholar] [CrossRef] [Green Version]
- Gupta, R.K.; Abdul-Jawad, S.; McCoy, L.E.; Mok, H.P.; Peppa, D.; Salgado, M.; Martinez-Picado, J.; Nijhuis, M.; Wensing, A.M.; Lee, H. HIV-1 remission following CCR5Δ32/Δ32 haematopoietic stem-cell transplantation. Nature 2019, 568, 244. [Google Scholar] [CrossRef]
- Cyranoski, D. First CRISPR babies: Six questions that remain. Nature 2018, 30. [Google Scholar] [CrossRef]
- Wei, X.; Nielsen, R. CCR5-∆ 32 is deleterious in the homozygous state in humans. Nat. Med. 2019, 25, 909–910. [Google Scholar] [CrossRef]
- Maier, R.; Akbari, A.; Wei, X.; Patterson, N.; Nielsen, R.; Reich, D. No statistical evidence for an effect of CCR5-∆ 32 on lifespan in the UK Biobank cohort. Nat. Med. 2019, 1–3. [Google Scholar] [CrossRef]
- Gornalusse, G.G.; Mummidi, S.; Gaitan, A.A.; Jimenez, F.; Ramsuran, V.; Picton, A.; Rogers, K.; Manoharan, M.S.; Avadhanam, N.; Murthy, K.K.; et al. Epigenetic mechanisms, T-cell activation, and CCR5 genetics interact to regulate T-cell expression of CCR5, the major HIV-1 coreceptor. Proc. Natl. Acad. Sci. USA 2015, 112, E4762–E4771. [Google Scholar] [CrossRef] [Green Version]
- Klein, R.S. A moving target: The multiple roles of CCR5 in infectious diseases. J. Infect. Dis. 2008, 197, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Dusi, V.; Ghidoni, A.; Ravera, A.; De Ferrari, G.M.; Calvillo, L. Chemokines and heart disease: A network connecting cardiovascular biology to immune and autonomic nervous systems. Mediat. Inflamm. 2016, 2016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hansson, G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Jonasson, L.; Holm, J.; Skalli, O.; Bondjers, G.; Hansson, G.K. Regional accumulations of T cells, macrophages, and smooth muscle cells in the human atherosclerotic plaque. Arterioscler. Off. J. Am. Heart Assoc. Inc. 1986, 6, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Libby, R. Cytokines and growth regulatory molecules. Atheroscler. Coron. Artery Dis. 1996, 585–594. [Google Scholar]
- Ross, R. Cell biology of atherosclerosis. Annu. Rev. Physiol. 1995, 57, 791–804. [Google Scholar] [CrossRef]
- Lin, J.; Kakkar, V.; Lu, X. Impact of MCP-1 in atherosclerosis. Curr. Pharm. Des. 2014, 20, 4580–4588. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, J.; Wang, H.; Wu, H.; Wu, X.; Dong, J.; Liao, L. Association between chemokine receptor 5 (CCR5) delta32 gene variant and atherosclerosis: A meta-analysis of 13 studies. Int. J. Clin. Exp. Med. 2015, 8, 658. [Google Scholar]
- Pai, J.K.; Kraft, P.; Cannuscio, C.C.; Manson, J.E.; Rexrode, K.M.; Albert, C.M.; Hunter, D.; Rimm, E.B. Polymorphisms in the CC-chemokine receptor-2 (CCR2) and-5 (CCR5) genes and risk of coronary heart disease among US women. Atherosclerosis 2006, 186, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Hyde, C.L.; MacInnes, A.; Sanders, F.A.; Thompson, J.F.; Mazzarella, R.A.; Faergeman, O.; van Wijk, D.F.; Wood, L.; Lira, M.; Paciga, S.A. Genetic association of the CCR5 region with lipid levels in at-risk cardiovascular patients. Circ. Cardiovasc. Genet. 2010, 3, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Apostolakis, S.; Baritaki, S.; Kochiadakis, G.E.; Igoumenidis, N.E.; Panutsopulos, D.; Spandidos, D.A. Effects of polymorphisms in chemokine ligands and receptors on susceptibility to coronary artery disease. Thromb. Res. 2007, 119, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Simeoni, E.; Winkelmann, B.R.; Hoffmann, M.M.; Fleury, S.; Ruiz, J.; Kappenberger, L.; März, W.; Vassalli, G. Association of RANTES G-403A gene polymorphism with increased risk of coronary arteriosclerosis. Eur. Heart J. 2004, 25, 1438–1446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharda, S.; Gilmour, A.; Harris, V.; Singh, V.; Sinha, N.; Tewari, S.; Ramesh, V.; Agrawal, S.; Mastana, S. Chemokine receptor 5 (CCR5) deletion polymorphism in North Indian patients with coronary artery disease. Int. J. Cardiol. 2008, 124, 254–258. [Google Scholar] [CrossRef]
- Afzal, A.R.; Kiechl, S.; Daryani, Y.P.; Weerasinghe, A.; Zhang, Y.; Reindl, M.; Mayr, A.; Weger, S.; Xu, Q.; Willeit, J. Common CCR5-del32 frameshift mutation associated with serum levels of inflammatory markers and cardiovascular disease risk in the Bruneck population. Stroke 2008, 39, 1972–1978. [Google Scholar] [CrossRef] [Green Version]
- Lassner, D.; Kühl, U.; Rohde, M.; Siegismund, C.S.; Schultheiss, H.-P. CCR5del32 polymorphism is a protective factor in non-ischemic cardiomyopathy. Int. J. Cardiol. 2014, 173, 561–562. [Google Scholar] [CrossRef]
- Szalai, C.; Duba, J.; Prohászka, Z.; Kalina, Á.; Szabó, T.; Nagy, B.; Horváth, L.; Császár, A. Involvement of polymorphisms in the chemokine system in the susceptibility for coronary artery disease (CAD). Coincidence of elevated Lp (a) and MCP-1−2518 G/G genotype in CAD patients. Atherosclerosis 2001, 158, 233–239. [Google Scholar] [CrossRef]
- Wang, L.; Hu, X.; Zhang, S.; Xu, X.; Wang, J. Association of the CCR5Δ32 polymorphism and its ligand RANTES-403 G/A polymorphism with coronary artery disease: A meta-analysis. Thromb. Res. 2013, 131, e77–e84. [Google Scholar] [CrossRef]
- Angeles-Martínez, J.; Posadas-Sánchez, R.; Álvarez-León, E.; Villarreal-Molina, T.; Cardoso-Saldana, G.; Fragoso, J.M.; Juárez-Rojas, J.G.; Medina-Urrutia, A.; Posadas-Romero, C.; Vargas-Alarcón, G. Monocyte chemoattractant protein-1 gene (MCP-1) polymorphisms are associated with risk of premature coronary artery disease in Mexican patients from the Genetics of Atherosclerotic Disease (GEA) study. Immunol. Lett. 2015, 167, 125–130. [Google Scholar] [CrossRef]
- Balistreri, C.R.; Candore, G.; Caruso, M.; Incalcaterra, E.; Franceschi, C.; Caruso, C. Role of polymorphisms of CC-chemokine receptor-5 gene in acute myocardial infarction and biological implications for longevity. Haematologica 2008, 93, 637–638. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, P.; Alvarez, R.; Batalla, A.; Reguero, J.R.; Alvarez, V.; Astudillo, A.; Cubero, G.I.; Cortina, A.; Coto, E. Genetic variation at the chemokine receptors CCR5/CCR2 in myocardial infarction. Genes Immun 2001, 2, 191–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karaali, Z.E.; Sozen, S.; Yurdum, M.; Cacina, C.; Toptas, B.; Gok, O.; Agachan, B. Effect of genetic variants of chemokine receptors on the development of myocardial infarction in Turkish population. Mol. Biol. Rep. 2010, 37, 3615–3619. [Google Scholar] [CrossRef] [PubMed]
- Ghilardi, G.; Biondi, M.L.; Turri, O.; Pateri, F.; d’Eril, G.M.; Scorza, R. Genetic control of chemokines in severe human internal carotid artery stenosis. Cytokine 2008, 41, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Mettimano, M.; Specchia, M.; Ianni, A.; Arzani, D.; Ricciardi, G.; Savi, L.; Romano-Spica, V. CCR5 and CCR2 gene polymorphisms in hypertensive patients. Br. J. Biomed. Sci. 2003, 60, 19–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Ardlie, K.; Wacholder, S.; Welch, R.; Chanock, S.; O’Brien, T.R. Genetic variations in CC chemokine receptors and hypertension. Am. J. Hypertens. 2006, 19, 67–72. [Google Scholar] [CrossRef]
- Marder, E.; Prinz, A.A. Modeling stability in neuron and network function: The role of activity in homeostasis. Bioessays 2002, 24, 1145–1154. [Google Scholar] [CrossRef]
- Bajetto, A.; Bonavia, R.; Barbero, S.; Florio, T.; Schettini, G. Chemokines and their receptors in the central nervous system. Front. Neuroendocrinol. 2001, 22, 147–184. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, A. A review on Alzheimer’s disease pathophysiology and its management: An update. Pharmacol. Rep. 2015, 67, 195–203. [Google Scholar] [CrossRef]
- Akiyama, H.; Barger, S.; Barnum, S.; Bradt, B.; Bauer, J.; Cole, G.M.; Cooper, N.R.; Eikelenboom, P.; Emmerling, M.; Fiebich, B.L. Inflammation and Alzheimer’s disease. Neurobiol. Aging 2000, 21, 383–421. [Google Scholar] [CrossRef]
- Xia, M.; Hyman, B.T. Chemokines/chemokine receptors in the central nervous system and Alzheimer’s disease. J. Neurovirol. 1999, 5, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.J.; Park, M.H.; Hwang, J.Y.; Kim, J.H.; Yun, N.Y.; Oh, S.Y.; Song, J.K.; Seo, H.O.; Kim, Y.-B.; Hwang, D.Y. CCR5 deficiency accelerates lipopolysaccharide-induced astrogliosis, amyloid-beta deposit and impaired memory function. Oncotarget 2016, 7, 11984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Combarros, O.; Infante, J.; Llorca, J.; Pena, N.; Fernandez-Viadero, C.; Berciano, J. The chemokine receptor CCR5-Delta32 gene mutation is not protective against Alzheimer’s disease. Neurosci. Lett. 2004, 366, 312–314. [Google Scholar] [CrossRef] [PubMed]
- Balistreri, C.R.; Grimaldi, M.P.; Vasto, S.; Listi, F.; Chiappelli, M.; Licastro, F.; Lio, D.; Caruso, C.; Candore, G. Association between the polymorphism of CCR5 and Alzheimer’s disease: Results of a study performed on male and female patients from Northern Italy. Ann. N. Y. Acad. Sci. 2006, 1089, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Galimberti, D.; Fenoglio, C.; Lovati, C.; Gatti, A.; Guidi, I.; Venturelli, E.; Cutter, G.R.; Mariani, C.; Forloni, G.; Pettenati, C.; et al. CCR2-64I polymorphism and CCR5Delta32 deletion in patients with Alzheimer’s disease. J. Neurol. Sci. 2004, 225, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Khorshid, H.R.K.; Manoochehri, M.; Nasehi, L.; Ohadi, M.; Rahgozar, M.; Kamali, K. Ccr2-64i and Ccr5 Δ32 Polymorphisms in Patients with Late-Onset Alzheimer’s disease; A Study from Iran (Ccr2-64i And Ccr5 Δ32 Polymorphisms in Alzheimer’s disease). Iran. J. Basic Med. Sci. 2012, 15, 937. [Google Scholar]
- Jorda, A.; Campos-Campos, J.; Iradi, A.; Aldasoro, M.; Aldasoro, C.; Vila, J.; Valles, S. The Role of Chemokines in Alzheimer’s Disease. Endocr. Metab. Immune Disord. Drug Targets 2020. [Google Scholar] [CrossRef]
- Li, T.; Zhu, J. Entanglement of CCR5 and Alzheimer’s Disease. Front. Aging Neurosci. 2019, 11, 209. [Google Scholar] [CrossRef]
- Huerta, C.; Alvarez, V.; Mata, I.F.; Coto, E.; Ribacoba, R.; Martinez, C.; Blazquez, M.; Guisasola, L.M.; Salvador, C.; Lahoz, C.H.; et al. Chemokines (RANTES and MCP-1) and chemokine-receptors (CCR2 and CCR5) gene polymorphisms in Alzheimer’s and Parkinson’s disease. Neurosci Lett 2004, 370, 151–154. [Google Scholar] [CrossRef]
- Gade-Andavolu, R.; Comings, D.E.; MacMurray, J.; Rostamkhani, M.; Cheng, L.S.-C.; Tourtellotte, W.W.; Cone, L.A. Association of CCR5 Δ32 deletion with early death in multiple sclerosis. Genet. Med. 2004, 6, 126. [Google Scholar] [CrossRef] [Green Version]
- Pulkkinen, K.; Luomala, M.; Kuusisto, H.; Lehtimäki, T.; Saarela, M.; Jalonen, T.; Elovaara, I. Increase in CCR5 Δ32/Δ32 genotype in multiple sclerosis. Acta Neurol. Scand. 2004, 109, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Kantor, R.; Bakhanashvili, M.; Achiron, A. A mutated CCR5 gene may have favorable prognostic implications in MS. Neurology 2003, 61, 238–240. [Google Scholar] [CrossRef] [PubMed]
- van Veen, T.; Nielsen, J.; Berkhof, J.; Barkhof, F.; Kamphorst, W.; Bo, L.; Ravid, R.; Verweij, C.L.; Huitinga, I.; Polman, C.H.; et al. CCL5 and CCR5 genotypes modify clinical, radiological and pathological features of multiple sclerosis. J. Neuroimmunol. 2007, 190, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Bennetts, B.H.; Teutsch, S.M.; Buhler, M.M.; Heard, R.N.; Stewart, G.J. The CCR5 deletion mutation fails to protect against multiple sclerosis. Hum. Immunol. 1997, 58, 52–59. [Google Scholar] [CrossRef]
- Kaimen-Maciel, D.R.; Vissoci Reiche, E.M.; Souza, B.; Guimarães, D.; Frota Comini, E.R.; Bobroff, F.; Morimoto, H.K.; Ehara Watanabe, M.A.; Carvalho De Oliveira, J.; Matsuo, T. CCR5-Δ32 genetic polymorphism associated with benign clinical course and magnetic resonance imaging findings in Brazilian patients with multiple sclerosis. Int. J. Mol. Med. 2007, 20, 337–344. [Google Scholar] [CrossRef] [Green Version]
- Silversides, J.; Heggarty, S.; McDonnell, G.; Hawkins, S.; Graham, C. Influence of CC R5 δ32 polymorphism on multiple sclerosis susceptibility and disease course. Mult. Scler. J. 2004, 10, 149–152. [Google Scholar] [CrossRef]
- Miotto, D.; Christodoulopoulos, P.; Olivenstein, R.; Taha, R.; Cameron, L.; Tsicopoulos, A.; Tonnel, A.-B.; Fahy, O.; Lafitte, J.; Luster, A.D. Expression of IFN-γ–inducible protein; monocyte chemotactic proteins 1, 3, and 4; and eotaxin in TH1-and TH2-mediated lung diseases. J. Allergy Clin. Immunol. 2001, 107, 664–670. [Google Scholar] [CrossRef]
- Ying, S.; Meng, Q.; Zeibecoglou, K.; Robinson, D.S.; Macfarlane, A.; Humbert, M.; Kay, A.B. Eosinophil chemotactic chemokines (eotaxin, eotaxin-2, RANTES, monocyte chemoattractant protein-3 (MCP-3), and MCP-4), and CC chemokine receptor 3 expression in bronchial biopsies from atopic and nonatopic (Intrinsic) asthmatics. J. Immunol. 1999, 163, 6321–6329. [Google Scholar]
- Hall, I.P.; Wheatley, A.; Christie, G.; McDougall, C.; Hubbard, R.; Helms, P.J. Association of CCR5∆ 32 with reduced risk of asthma. Lancet 1999, 354, 1264–1265. [Google Scholar] [CrossRef]
- Srivastava, P.; Helms, P.; Stewart, D.; Main, M.; Russell, G. Association of CCR5Δ32 with reduced risk of childhood but not adult asthma. Thorax 2003, 58, 222–226. [Google Scholar] [CrossRef] [Green Version]
- Gold, M.S.; Kemp, A.S. Atopic disease in childhood. Med. J. Aust. 2005, 182, 298–304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, T.J.; Walley, A.J.; Pease, J.E.; Venables, P.J.; Wiltshire, S.; Williams, T.J.; Cookson, W.O. Delta 32 deletion of CCR5 gene and association with asthma or atopy. Lancet 2000, 356, 1491–1492. [Google Scholar] [CrossRef]
- Abousaidi, H.; Vazirinejad, R.; Arababadi, M.K.; Rafatpanah, H.; Pourfathollah, A.A.; Derakhshan, R.; Daneshmandi, S.; Hassanshahi, G. Lack of association between chemokine receptor 5 (CCR5) δ32 mutation and pathogenesis of asthma in Iranian patients. South. Med. J. 2011, 104, 422–425. [Google Scholar] [CrossRef] [PubMed]
- Cracoviensia, F.M.; Gomulska, M.; Rusin, G.; Gwizdak, P. Prevalence of CCR5-delta32 mutation in asthmatic and non-asthmatic subjects from department of medicine, JUCM, Cracow. Pol. Acad. Sci. Krakow Branch 2014, 54, 5–13. [Google Scholar]
- Muntinghe, F.L.; Gross, S.; Bakker, S.J.; Landman, G.W.; van der Harst, P.; Bilo, H.J.; Navis, G.; Zuurman, M.W. CCR5Δ32 genotype is associated with outcome in type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2009, 86, 140–145. [Google Scholar] [CrossRef]
- Antonelli, A.; Ferrari, S.M.; Corrado, A.; Di Domenicantonio, A.; Fallahi, P. Autoimmune thyroid disorders. Autoimmun. Rev. 2015, 14, 174–180. [Google Scholar] [CrossRef]
- Karin, N.; Wildbaum, G. The role of chemokines in shaping the balance between CD4+ T cell subsets and its therapeutic implications in autoimmune and cancer diseases. Front. Immunol. 2015, 6, 609. [Google Scholar] [CrossRef] [Green Version]
- Xu, L.; Kitade, H.; Ni, Y.; Ota, T. Roles of chemokines and chemokine receptors in obesity-associated insulin resistance and nonalcoholic fatty liver disease. Biomolecules 2015, 5, 1563–1579. [Google Scholar] [CrossRef] [Green Version]
- Gambelunghe, G.; Ghaderi, M.; Brozzetti, A.; Del Sindaco, P.; Gharizadeh, B.; Nyren, P.; Hjelmström, P.; Nikitina-Zake, L.; Sanjeevi, C.B.; Falorni, A. Lack of association of CCR2–64I and CCR5-Δ32 with type 1 diabetes and latent autoimmune diabetes in adults. Hum. Immunol. 2003, 64, 629–632. [Google Scholar] [CrossRef]
- Szalai, C.; Császár, A.; Czinner, A.; Szabó, T.; Pánczél, P.; Madácsy, L.; Falus, A. Chemokine receptor CCR2 and CCR5 polymorphisms in children with insulin-dependent diabetes mellitus. Pediatric Res. 1999, 46, 82. [Google Scholar] [CrossRef] [Green Version]
- Yang, B.; Houlberg, K.; Millward, A.; Demaine, A. Polymorphisms of chemokine and chemokine receptor genes in Type 1 diabetes mellitus and its complications. Cytokine 2004, 26, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Kalev, I.; Oselin, K.; Pärlist, P.; Zilmer, M.; Rajasalu, T.; Podar, T.; Mikelsaar, A.-V. CC-chemokine receptor CCR5-del32 mutation as a modifying pathogenetic factor in type I diabetes. J. Diabetes Its Complicat. 2003, 17, 387–391. [Google Scholar] [CrossRef]
- Song, G.G.; Kim, J.-H.; Lee, Y.H. The chemokine receptor 5 delta32 polymorphism and type 1 diabetes, Behcet’s disease, and asthma: A meta-analysis. Immunol. Investig. 2014, 43, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Kohem, C.; Brenol, J.; Xavier, R.; Bredemeier, M.; Brenol, C.; Dedavid e Silva, T.; de Castilhos Mello, A.; Canedo, A.; Neves, A.; Chies, J. The chemokine receptor CCR5 genetic polymorphism and expression in rheumatoid arthritis patients. Scand. J. Rheumatol. 2007, 36, 359–364. [Google Scholar] [CrossRef]
- Lindner, E.; Nordang, G.B.; Melum, E.; Flatø, B.; Selvaag, A.M.; Thorsby, E.; Kvien, T.K.; Førre, Ø.T.; Lie, B.A. Lack of association between the chemokine receptor 5 polymorphism CCR5delta32 in rheumatoid arthritis and juvenile idiopathic arthritis. BMC Med. Genet. 2007, 8, 33. [Google Scholar] [CrossRef] [Green Version]
- Garred, P.; Madsen, H.; Petersen, J.; Marquart, H.; Hansen, T.; Freiesleben, S.S.; Volck, B.; Svejgaard, A.; Andersen, V. CC chemokine receptor 5 polymorphism in rheumatoid arthritis. J. Rheumatol. 1998, 25, 1462–1465. [Google Scholar]
- Gómez-Reino, J.J.; Pablos, J.L.; Carreira, P.E.; Santiago, B.; Serrano, L.; Vicario, J.L.; Balsa, A.; Figueroa, M.; De Juan, M.D. Association of rheumatoid arthritis with a functional chemokine receptor, CCR5. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 1999, 42, 989–992. [Google Scholar] [CrossRef]
- Pokorny, V.; McQueen, F.; Yeoman, S.; Merriman, M.; Merriman, A.; Harrison, A.; Highton, J.; McLean, L. Evidence for negative association of the chemokine receptor CCR5 d32 polymorphism with rheumatoid arthritis. Ann. Rheum. Dis. 2005, 64, 487–490. [Google Scholar] [CrossRef] [Green Version]
- Rossol, M.; Pierer, M.; Arnold, S.; Keyßer, G.; Burkhardt, H.; Baerwald, C.; Wagner, U. Negative association of the chemokine receptor CCR5 d32 polymorphism with systemic inflammatory response, extra-articular symptoms and joint erosion in rheumatoid arthritis. Arthritis Res. Ther. 2009, 11, R91. [Google Scholar] [CrossRef] [Green Version]
- Bauer, J.W.; Baechler, E.C.; Petri, M.; Batliwalla, F.M.; Crawford, D.; Ortmann, W.A.; Espe, K.J.; Li, W.; Patel, D.D.; Gregersen, P.K. Elevated serum levels of interferon-regulated chemokines are biomarkers for active human systemic lupus erythematosus. PLoS Med. 2006, 3, e491. [Google Scholar] [CrossRef]
- Carvalho, C.; Calvisi, S.; Leal, B.; Bettencourt, A.; Marinho, A.; Almeida, I.; Farinha, F.; Costa, P.; Silva, B.; Vasconcelos, C. CCR 5-D elta32: Implications in SLE development. Int. J. Immunogenet. 2014, 41, 236–241. [Google Scholar] [CrossRef]
- Schauren, J.; Marasca, J.; Veit, T.; Monticielo, O.; Xavier, R.; Brenol, J.; Chies, J. CCR5delta32 in systemic lupus erythematosus: Implications for disease susceptibility and outcome in a Brazilian population. Lupus 2013, 22, 802–809. [Google Scholar] [CrossRef]
- Baltus, T.H.L.; Kallaur, A.P.; Lozovoy, M.A.B.; Morimoto, H.K.; Delongui, F.; Alfieri, D.F.; Iriyoda, T.M.V.; Dichi, I.; Reiche, E.M.V. CCR5Δ32 (rs333) polymorphism is associated with the susceptibility to systemic lupus erythematosus in female Brazilian patients. Rheumatol. Int. 2016, 36, 7–15. [Google Scholar] [CrossRef]
- Segerer, S.; Mack, M.; Regele, H.; Kerjaschki, D.; Schlöndorff, D. Expression of the CC chemokine receptor 5 in human kidney diseases. Kidney Int. 1999, 56, 52–64. [Google Scholar] [CrossRef] [Green Version]
- Zhou, T.-B.; Jiang, Z.-P.; Zhou, J.-F.; Su, N. Relationship between chemokine receptor 5 Δ32/W gene polymorphism and lupus nephritis. Hum. Immunol. 2014, 75, 968–972. [Google Scholar] [CrossRef]
- Cocchi, F.; DeVico, A.L.; Garzino-Demo, A.; Arya, S.K.; Gallo, R.C.; Lusso, P. Identification of RANTES, MIP-1α, and MIP-1β as the major HIV-suppressive factors produced by CD8+ T cells. Science 1995, 270, 1811–1815. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.; Paxton, W.A.; Choe, S.; Ceradini, D.; Martin, S.R.; Horuk, R.; MacDonald, M.E.; Stuhlmann, H.; Koup, R.A.; Landau, N.R. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell 1996, 86, 367–377. [Google Scholar] [CrossRef] [Green Version]
- Rana, S.; Besson, G.; Cook, D.G.; Rucker, J.; Smyth, R.J.; Yi, Y.; Turner, J.D.; Guo, H.H.; Du, J.G.; Peiper, S.C. Role of CCR5 in infection of primary macrophages and lymphocytes by macrophage-tropic strains of human immunodeficiency virus: Resistance to patient-derived and prototype isolates resulting from the delta ccr5 mutation. J. Virol. 1997, 71, 3219–3227. [Google Scholar] [CrossRef] [Green Version]
- Balotta, C.; Bagnarelli, P.; Violin, M.; Ridolfo, A.L.; Zhou, D.; Berlusconi, A.; Corvasce, S.; Corbellino, M.; Clementi, M.; Clerici, M. Homozygous Δ32 deletion of the CCR-5 chemokine receptor gene in an HIV-1-infected patient. Aids 1997, 11, F67–F71. [Google Scholar] [CrossRef]
- Biti, R.; Ffrench, R.; Young, J.; Bennetts, B.; Stewart, G.; Liang, T. HIV-1 infection in an individual homozygous for the CCR5 deletion allele. Nat. Med. 1997, 3, 252. [Google Scholar] [CrossRef]
- Sheppard, H.W.; Celum, C.; Michael, N.L.; O’Brien, S.; Dean, M.; Carrington, M.; Dondero, D.; Buchbinder, S.P. HIV-1 infection in individuals with the CCR5-Delta32/Delta32 genotype: Acquisition of syncytium-inducing virus at seroconversion. J. Acquir. Immune Defic. Syndr. 2002, 29, 307–313. [Google Scholar] [CrossRef]
- Theodorou, I.; Meyer, L. HIV-1 infection in an individual homozygous for CCR5Delta32. Lancet 1997, 349, 1219–1220. [Google Scholar] [CrossRef]
- Michael, N.L.; Nelson, J.A.; KewalRamani, V.N.; Chang, G.; O’Brien, S.J.; Mascola, J.R.; Volsky, B.; Louder, M.; White, G.C.; Littman, D.R. Exclusive and persistent use of the entry coreceptor CXCR4 by human immunodeficiency virus type 1 from a subject homozygous for CCR5 Δ32. J. Virol. 1998, 72, 6040–6047. [Google Scholar] [CrossRef] [Green Version]
- Heiken, H.; Becker, S.; Bastisch, I.; Schmidt, R. HIV-1 infection in a heterosexual man homozygous for CCR-5Δ32. Aids 1999, 13, 529. [Google Scholar] [CrossRef]
- Kuipers, H.; Workman, C.; Dyer, W.; Geczy, A.; Sullivan, J.; Oelrichs, R. An HIV-1-infected individual homozygous for the CCR-5 D32 allele and the SDF-1 3¢ A allele. Aids 1999, 13, 433. [Google Scholar] [CrossRef]
- Glass, W.G.; Lim, J.K.; Cholera, R.; Pletnev, A.G.; Gao, J.L.; Murphy, P.M. Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in West Nile virus infection. J. Exp. Med. 2005, 202, 1087–1098. [Google Scholar] [CrossRef] [Green Version]
- Glass, W.G.; McDermott, D.H.; Lim, J.K.; Lekhong, S.; Yu, S.F.; Frank, W.A.; Pape, J.; Cheshier, R.C.; Murphy, P.M. CCR5 deficiency increases risk of symptomatic West Nile virus infection. J. Exp. Med. 2006, 203, 35–40. [Google Scholar] [CrossRef] [Green Version]
- Lim, J.K.; Louie, C.Y.; Glaser, C.; Jean, C.; Johnson, B.; Johnson, H.; McDermott, D.H.; Murphy, P.M. Genetic deficiency of chemokine receptor CCR5 is a strong risk factor for symptomatic West Nile virus infection: A meta-analysis of 4 cohorts in the US epidemic. J. Infect. Dis. 2008, 197, 262–265. [Google Scholar] [CrossRef] [Green Version]
- Fisicaro, P.; Valdatta, C.; Boni, C.; Massari, M.; Mori, C.; Zerbini, A.; Orlandini, A.; Sacchelli, L.; Missale, G.; Ferrari, C. Early kinetics of innate and adaptive immune responses during hepatitis B virus infection. Gut 2009, 58, 974–982. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.K.; Suh, J.H.; Cho, Y.S.; Han, K.H.; Chung, J.B.; Chon, C.Y.; Moon, Y.M. Chemokine receptor expression of hepatitis B virus-specific CD8+ lymphocyte in chronic B viral infection. Korean J. Hepatol. 2002, 8, 363–370. [Google Scholar]
- Shimada, N.; Yamamoto, K.; Kuroda, M.J.; Terada, R.; Hakoda, T.; Shimomura, H.; Hata, H.; Nakayama, E.; Shiratori, Y. HBcAg-specific CD8 T cells play an important role in virus suppression, and acute flare-up is associated with the expansion of activated memory T cells. J. Clin. Immunol. 2003, 23, 223–232. [Google Scholar] [CrossRef]
- Rahbar, R.; Murooka, T.T.; Fish, E.N. Role for CCR5 in dissemination of vaccinia virus in vivo. J. Virol. 2009, 83, 2226–2236. [Google Scholar] [CrossRef] [Green Version]
- TrehanPati, N.; Geffers, R.; Hissar, S.; Riese, P.; Toepfer, T.; Buer, J.; Kumar, M.; Guzman, C.A.; Sarin, S.K. Gene expression signatures of peripheral CD4+ T cells clearly discriminate between patients with acute and chronic hepatitis B infection. Hepatology 2009, 49, 781–790. [Google Scholar] [CrossRef]
- Arababadi, M.K.; Pourfathollah, A.A.; Jafarzadeh, A.; Hassanshahi, G.; Mohit, M.; Hajghani, M.; Ahmadabadi, B.N.; Kennedy, D. Evaluation of CCR5 expression on NK cells in Iranian patients with occult hepatitis B infection. Lab. Med. 2010, 41, 735–738. [Google Scholar] [CrossRef]
- Arababadi, M.K.; Pourfathollah, A.A.; Jafarzadeh, A.; Hassanshahi, G.; Mohit, M.; Hajghani, M.; Shamsizadeh, A. Peripheral blood CD8+ T cells CCR5 expression and its Δ32 mutation in Iranian patients with occult hepatitis B infections. Lab. Med. 2010, 41, 226–230. [Google Scholar] [CrossRef]
- Ahmadabadi, B.N.; Hassanshahi, G.; Khoramdelazad, H.; Mirzaei, V.; Sajadi, S.M.A.; Hajghani, M.; Khodadadi, H.; Pourali, R.; Arababadi, M.K.; Kennedy, D. Downregulation of CCR5 expression on the peripheral blood CD8+ T cells of southeastern Iranian patients with chronic hepatitis B infection. Inflammation 2013, 36, 136–140. [Google Scholar] [CrossRef]
- Thio, C.L.; Astemborski, J.; Bashirova, A.; Mosbruger, T.; Greer, S.; Witt, M.D.; Goedert, J.J.; Hilgartner, M.; Majeske, A.; O’Brien, S.J.; et al. Genetic protection against hepatitis B virus conferred by CCR5Delta32: Evidence that CCR5 contributes to viral persistence. J. Virol. 2007, 81, 441–445. [Google Scholar] [CrossRef] [Green Version]
- Ellwanger, J.H.; Kulmann-Leal, B.; Wolf, J.M.; Michita, R.T.; Simon, D.; Lunge, V.R.; Chies, J.A.B. Role of the genetic variant CCR5Δ32 in HBV infection and HBV/HIV co-infection. Virus Res. 2020, 277, 197838. [Google Scholar] [CrossRef]
- Soo, H.M.; Garzino-Demo, A.; Hong, W.; Tan, Y.H.; Tan, Y.J.; Goh, P.-Y.; Lim, S.G.; Lim, S.P. Expression of a full-length hepatitis C virus cDNA up-regulates the expression of CC chemokines MCP-1 and RANTES. Virology 2002, 303, 253–277. [Google Scholar]
- Nattermann, J.; Nischalke, H.; Feldmann, G.; Ahlenstiel, G.; Sauerbruch, T.; Spengler, U. Binding of HCV E2 to CD81 induces RANTES secretion and internalization of CC chemokine receptor 5. J. Viral Hepat. 2004, 11, 519–526. [Google Scholar] [CrossRef]
- Solari, R.; Offord, R.E.; Remy, S.; Aubry, J.-P.; Wells, T.N.; Whitehorn, E.; Oung, T.; Proudfoot, A.E. Receptor-mediated endocytosis of CC-chemokines. J. Biol. Chem. 1997, 272, 9617–9620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lichterfeld, M.; Leifeld, L.; Nischalke, H.D.; Rockstroh, J.K.; Heß, L.; Sauerbruch, T.; Spengler, U. Reduced CC chemokine receptor (CCR) 1 and CCR5 surface expression on peripheral blood T lymphocytes from patients with chronic hepatitis C infection. J. Infect. Dis. 2002, 185, 1803–1807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hellier, S.; Frodsham, A.J.; Hennig, B.J.; Klenerman, P.; Knapp, S.; Ramaley, P.; Satsangi, J.; Wright, M.; Zhang, L.; Thomas, H.C. Association of genetic variants of the chemokine receptor CCR5 and its ligands, RANTES and MCP-2, with outcome of HCV infection. Hepatology 2003, 38, 1468–1476. [Google Scholar] [CrossRef]
- Promrat, K.; McDermott, D.H.; Gonzalez, C.M.; Kleiner, D.E.; Koziol, D.E.; Lessie, M.; Merrell, M.; Soza, A.; Heller, T.; Ghany, M. Associations of chemokine system polymorphisms with clinical outcomes and treatment responses of chronic hepatitis C. Gastroenterology 2003, 124, 352–360. [Google Scholar] [CrossRef] [Green Version]
- Wald, O.; Pappo, O.; Ari, Z.B.; Azzaria, E.; Wiess, I.D.; Gafnovitch, I.; Wald, H.; Spengler, U.; Galun, E.; Peled, A. The CCR5Delta32 allele is associated with reduced liver inflammation in hepatitis C virus infection. Eur. J. Immunogenet. 2004, 31, 249–252. [Google Scholar] [CrossRef]
- Ruiz-Ferrer, M.; Barroso, N.; Antinolo, G.; Aguilar-Reina, J. Analysis of CCR5-Δ32 and CCR2-V64I polymorphisms in a cohort of Spanish HCV patients using Real-time Polymerase Chain Reaction and Fluorescence Resonance Energy Transfer technologies. J. Viral Hepat. 2004, 11, 319–323. [Google Scholar] [CrossRef]
- Goulding, C.; Murphy, A.; MacDonald, G.; Barrett, S.; Crowe, J.; Hegarty, J.; McKiernan, S.; Kelleher, D. The CCR5-Δ32 mutation: Impact on disease outcome in individuals with hepatitis C infection from a single source. Gut 2005, 54, 1157–1161. [Google Scholar] [CrossRef] [Green Version]
- Frade, A.F.; Pissetti, C.W.; Ianni, B.M.; Saba, B.; Lin-Wang, H.T.; Nogueira, L.G.; de Melo Borges, A.; Buck, P.; Dias, F.; Baron, M. Genetic susceptibility to Chagas disease cardiomyopathy: Involvement of several genes of the innate immunity and chemokine-dependent migration pathways. BMC Infect. Dis. 2013, 13, 587. [Google Scholar] [CrossRef]
- De Mattos, L.; Ayo, C.; Bestetti, R.; de Mattos, C.B. Host’s immunogenetic risk factors in patients with Chagas Disease. Int. J. Infect. Dis. 2019, 79, 82. [Google Scholar] [CrossRef] [Green Version]
- Talvani, A.; Rocha, M.O.; Ribeiro, A.L.; Correa-Oliveira, R.; Teixeira, M.M. Chemokine receptor expression on the surface of peripheral blood mononuclear cells in Chagas disease. J. Infect. Dis. 2004, 189, 214–220. [Google Scholar] [CrossRef]
- Fernandez-Mestre, M.; Montagnani, S.; Layrisse, Z. Is the CCR5-59029-G/G genotype a protective factor for cardiomyopathy in Chagas disease? Hum. Immunol. 2004, 65, 725–728. [Google Scholar] [CrossRef]
- Calzada, J.; Nieto, A.; Beraun, Y.; Martin, J. Chemokine receptor CCR5 polymorphisms and Chagas’ disease cardiomyopathy. Tissue Antigens 2001, 58, 154–158. [Google Scholar] [CrossRef]
- Flórez, O.; Martín, J.; González, C.I. Genetic variants in the chemokines and chemokine receptors in Chagas disease. Hum. Immunol. 2012, 73, 852–858. [Google Scholar] [CrossRef]
- Nogueira, L.G.; Santos, R.H.B.; Ianni, B.M.; Fiorelli, A.I.; Mairena, E.C.; Benvenuti, L.A.; Frade, A.; Donadi, E.; Dias, F.; Saba, B. Myocardial chemokine expression and intensity of myocarditis in Chagas cardiomyopathy are controlled by polymorphisms in CXCL9 and CXCL10. PLoS Negl. Trop. Dis. 2012, 6, e1867. [Google Scholar] [CrossRef]
- Tisoncik, J.R.; Korth, M.J.; Simmons, C.P.; Farrar, J.; Martin, T.R.; Katze, M.G. Into the eye of the cytokine storm. Microbiol. Mol. Biol. Rev. 2012, 76, 16–32. [Google Scholar] [CrossRef] [Green Version]
- Kohlmeier, J.E.; Miller, S.C.; Smith, J.; Lu, B.; Gerard, C.; Cookenham, T.; Roberts, A.D.; Woodland, D.L. The chemokine receptor CCR5 plays a key role in the early memory CD8+ T cell response to respiratory virus infections. Immunity 2008, 29, 101–113. [Google Scholar] [CrossRef] [Green Version]
- Tavares, L.P.; Teixeira, M.M.; Garcia, C.C. The inflammatory response triggered by Influenza virus: A two edged sword. Inflamm. Res. 2017, 66, 283–302. [Google Scholar] [CrossRef]
- Keynan, Y.; Juno, J.; Meyers, A.; Ball, T.B.; Kumar, A.; Rubinstein, E.; Fowke, K.R. Chemokine receptor 5 Δ32 allele in patients with severe pandemic (H1N1) 2009. Emerg. Infect. Dis. 2010, 16, 1621. [Google Scholar] [CrossRef]
- Sironi, M.; Cagliani, R.; Pontremoli, C.; Rossi, M.; Migliorino, G.; Clerici, M.; Gori, A. The CCR5Δ32 allele is not a major predisposing factor for severe H1N1pdm09 infection. BMC Res. Notes 2014, 7, 504. [Google Scholar] [CrossRef] [Green Version]
- Maestri, A.; Dos Santos, M.C.; Ribeiro-Rodrigues, E.M.; De Mello, W.A.; Sousa, R.C.M.; Dos Santos, S.E.; Sortica, V.A. The CCR5Δ32 (rs333) polymorphism is not a predisposing factor for severe pandemic influenza in the Brazilian admixed population. BMC Res. Notes 2015, 8, 326. [Google Scholar] [CrossRef] [Green Version]
- Matos, A.R.; Martins, J.S.; Maria de Lourdes, A.O.; Garcia, C.C.; Siqueira, M.M. Human CCR5Δ32 (rs333) polymorphism has no influence on severity and mortality of influenza A (H1N1) pdm09 infection in Brazilian patients from the post pandemic period. Infect. Genet. Evol. 2019, 67, 55–59. [Google Scholar] [CrossRef]
- Aldinucci, D.; Colombatti, A. The inflammatory chemokine CCL5 and cancer progression. Mediat. Inflamm 2014, 2014, 292376. [Google Scholar] [CrossRef] [Green Version]
- Vindrieux, D.; Escobar, P.; Lazennec, G. Emerging roles of chemokines in prostate cancer. Endocr. Relat. Cancer 2009, 16, 663. [Google Scholar] [CrossRef] [Green Version]
- Lavergne, E.; Combadière, C.; Iga, M.; Boissonnas, A.; Bonduelle, O.; Maho, M.; Debré, P.; Combadiere, B. Intratumoral CC chemokine ligand 5 overexpression delays tumor growth and increases tumor cell infiltration. J. Immunol. 2004, 173, 3755–3762. [Google Scholar] [CrossRef]
- Gao, D.; Cazares, L.H.; Fish, E.N. CCL5-CCR5 interactions modulate metabolic events during tumor onset to promote tumorigenesis. BMC Cancer 2017, 17, 834. [Google Scholar] [CrossRef] [Green Version]
- Eskandari-Nasab, E.; Hashemi, M.; Ebrahimi, M.; Amininia, S.; Bahari, G.; Mashhadi, M.-A.; Taheri, M. Evaluation of CCL5-403 G> A and CCR5 Δ32 gene polymorphisms in patients with breast cancer. Cancer Biomark. 2014, 14, 343–351. [Google Scholar] [CrossRef]
- Li, J.; Peng, Y.; Liu, H.; Wu, Q. The association between CCR5 Δ32 polymorphism and susceptibility to breast cancer. Oncotarget 2017, 8, 82796. [Google Scholar]
- Jiao, X.; Velasco-Velázquez, M.A.; Wang, M.; Li, Z.; Rui, H.; Peck, A.R.; Korkola, J.E.; Chen, X.; Xu, S.; DuHadaway, J.B. CCR5 governs DNA damage repair and breast cancer stem cell expansion. Cancer Res. 2018, 78, 1657–1671. [Google Scholar] [CrossRef] [Green Version]
- Degerli, N.; Yılmaz, E.; Bardakci, F. The Δ32 allele distribution of the CCR5 gene and its relationship with certain cancers in a Turkish population. Clin. Biochem. 2005, 38, 248–252. [Google Scholar] [CrossRef]
- Duell, E.J.; Casella, D.P.; Burk, R.D.; Kelsey, K.T.; Holly, E.A. Inflammation, genetic polymorphisms in proinflammatory genes TNF-A, RANTES, and CCR5, and risk of pancreatic adenocarcinoma. Cancer Epidemiol. Prev. Biomark. 2006, 15, 726–731. [Google Scholar] [CrossRef] [Green Version]
- Singh, H.; Sachan, R.; Jain, M.; Mittal, B. CCR5-Δ32 polymorphism and susceptibility to cervical cancer: Association with early stage of cervical cancer. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2008, 17, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Wiklund, F.; Gharizadeh, B.; Sadat, M.; Gambelunghe, G.; Hallmans, G.; Dillner, J.; Wallin, K.-L.; Ghaderi, M. Genetic polymorphism of chemokine receptors CCR2 and CCR5 in Swedish cervical cancer patients. Anticancer Res. 2006, 26, 3669–3674. [Google Scholar] [PubMed]
- Srivastava, A.; Pandey, S.; Choudhuri, G.; Mittal, B. CCR5 Δ32 Polymorphism: Associated with Gallbladder Cancer Susceptibility. Scand. J. Immunol. 2008, 67, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Sugasawa, H.; Ichikura, T.; Kinoshita, M.; Ono, S.; Majima, T.; Tsujimoto, H.; Chochi, K.; Hiroi, S.; Takayama, E.; Saitoh, D. Gastric cancer cells exploit CD4+ cell-derived CCL5 for their growth and prevention of CD8+ cell-involved tumor elimination. Int. J. Cancer 2008, 122, 2535–2541. [Google Scholar] [CrossRef]
- Gawron, A.J.; Fought, A.J.; Lissowska, J.; Ye, W.; Zhang, X.; Chow, W.-H.; Beane Freeman, L.E.; Hou, L. Polymorphisms in chemokine and receptor genes and gastric cancer risk and survival in a high risk Polish population. Scand. J. Gastroenterol. 2011, 46, 333–340. [Google Scholar] [CrossRef] [Green Version]
- Cao, Z.; Xu, X.; Luo, X.; Li, L.; Huang, B.; Li, X.; Tao, D.; Hu, J.; Gong, J. Role of RANTES and its receptor in gastric cancer metastasis. J. Huazhong Univ. Sci. Technol. 2011, 31, 342–347. [Google Scholar] [CrossRef]
- Kucukgergin, C.; Isman, F.K.; Dasdemir, S.; Cakmakoglu, B.; Sanli, O.; Gokkusu, C.; Seckin, S. The role of chemokine and chemokine receptor gene variants on the susceptibility and clinicopathological characteristics of bladder cancer. Gene 2012, 511, 7–11. [Google Scholar] [CrossRef]
- Kucukgergin, C.; Isman, F.K.; Cakmakoglu, B.; Sanli, O.; Seckin, S. Association of polymorphisms in MCP-1, CCR2, and CCR5 genes with the risk and clinicopathological characteristics of prostate cancer. DNA Cell Biol. 2012, 31, 1418–1424. [Google Scholar] [CrossRef]
- Zambra, F.M.B.; Biolchi, V.; Brum, I.S.; Chies, J.A.B. CCR2 and CCR5 genes polymorphisms in benign prostatic hyperplasia and prostate cancer. Hum. Immunol. 2013, 74, 1003–1008. [Google Scholar] [CrossRef]
- Petersen, D.C.; Severi, G.; Hoang, H.N.; Padilla, E.J.; Southey, M.C.; English, D.R.; Hopper, J.L.; Giles, G.G.; Hayes, V.M. No association between common chemokine and chemokine receptor gene variants and prostate cancer risk. Cancer Epidemiol. Prev. Biomark. 2008, 17, 3615–3617. [Google Scholar] [CrossRef] [Green Version]
- Khorramdelazad, H.; Mortazavi, Y.; Momeni, M.; Arababadi, M.K.; Khandany, B.K.; Moogooei, M.; Hassanshahi, G. Lack of correlation between the CCR5-Δ32 mutation and acute myeloid leukemia in Iranian patients. Indian J. Hematol. Blood Transfus. 2015, 31, 29–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramsden, J. Metabolomics and Metabonomics. In Bioinformatics; Springer: Berlin/Heidelberg, Germany, 2015; pp. 265–270. [Google Scholar]
- Fiehn, O. Metabolomics—The link between genotypes and phenotypes. In Functional Genomics; Springer: Berlin/Heidelberg, Germany, 2002; pp. 155–171. [Google Scholar]
- Dunn, W.B.; Bailey, N.J.; Johnson, H.E. Measuring the metabolome: Current analytical technologies. Analyst 2005, 130, 606–625. [Google Scholar] [CrossRef] [PubMed]
- Patti, G.J.; Yanes, O.; Siuzdak, G. Innovation: Metabolomics: The apogee of the omics trilogy. Nat. Rev. Mol. Cell Biol. 2012, 13, 263. [Google Scholar] [CrossRef] [PubMed]
- Esterhuizen, K.; Van der Westhuizen, F.H.; Louw, R. Metabolomics of mitochondrial disease. Mitochondrion 2017, 35, 97–110. [Google Scholar] [CrossRef] [PubMed]
| Reference | [61] | [62] | [63] | [64] | [65] | [66] | [67] | [68] | [69] | [70] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | CVD | CVD | CVD | CVD | CVD | CVD | CVD | CVD | CVD | CVD | |
| Disease | AS | CHD | CAD | CAD | CAD | CAD | CAD | CM | CAD | CAD | |
| Population | Mixed | Caucasian | Caucasian | Greek | German; Caucasian | Indian | Bruneck | German | Hungarian | Mixed | |
| Model | Meta-analysis | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Meta-analysis | |
| CCR5∆32 frequency | Patients | ↑ Asian | ↑ | ||||||||
| No difference | X Mixed | X | X | X | X | X | X | ||||
| Controls | |||||||||||
| CCR5 levels | Patients/test subject/sample | ||||||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X Asian | X | X | X | X | X | ||||
| Heterozygous CCR5∆32 | X Asian | X | X | X | X | X | X | ||||
| Absent CCR5∆32 | X Px | X a | X a | X Px,a | |||||||
| Homozygous WT | X | ||||||||||
| Association/effect of CCR5∆32 | Protective | X a | X | X M | X a | ||||||
| No association/effect | X S, Mixed | X b | X | X S | X b,S | X | |||||
| Risk factor/non-protective | X S,Asian | X O | X S | ||||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | ||
| RFLP | X | X | |||||||||
| Sequencing | |||||||||||
| Immune-based | X | X | X | X | |||||||
| Microscopy | |||||||||||
| Spectroscopy | X | X | |||||||||
| Flow Cytometry | |||||||||||
| Chromatography | |||||||||||
| MS | |||||||||||
| Other | Meta-analysis | Meta-analysis | |||||||||
| Parameters | Immune | X | X | X | |||||||
| Metabolic | X | X | X | X | X | X | |||||
| Reference | [72] | [73] | [74] | [75] | [76] | [77] | [84] | [85] | [86] | [87] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | CVD | CVD | CVD | CVD | CVD | CVD | NS | NS | NS | NS | |
| Disease | MI | MI | MI | ICAS | HT | HT | AD | AD | AD | AD | |
| Population | Sicilian | Caucasian | Turkish | Italian | Caucasian | Caucasian | Spanish | Italian | Italian | Iranian | |
| Model | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Blood | |
| CCR5∆32 frequency | Patients | ↑ >60 years | ↑ a,b | ↑ a,b | |||||||
| No difference | X | X | X | X | X | X | |||||
| Controls | ↑ | ↑ <55 years | |||||||||
| CCR5 levels | Patients/test subject/sample | ||||||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | X | X | X | |||||
| Heterozygous CCR5∆32 | X | X | X | X | X | X | |||||
| Absent CCR5∆32 | X a | ||||||||||
| Homozygous WT | |||||||||||
| Association/ effect of CCR5∆32 | Protective | X | X <55 years | ||||||||
| No association/effect | X | X | X | X P | X | X | |||||
| Risk factor/non-protective | X >60 years | X a,b | X a,b | ||||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | X |
| RFLP | X | X | X | X | |||||||
| Sequencing | |||||||||||
| Immune-based | |||||||||||
| Microscopy | |||||||||||
| Spectroscopy | |||||||||||
| Flow Cytometry | |||||||||||
| Chromatography | |||||||||||
| MS | |||||||||||
| Other | |||||||||||
| Parameters | Immune | ||||||||||
| Metabolic | X | ||||||||||
| Reference | [88] | [90] | [91] | [92] | [93] | [94] | [95] | [96] | [97] | [100] | [101] | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | NS | NS | NS | NS | NS | NS | NS | NS | NS | IS | IS | |
| Disease | AD | PD | ms | ms | ms | ms | ms | ms | ms | Asthma | Asthma | |
| Population | N/A | Spanish | Caucasian | Finland | Israeli | Dutch | Australian | Brazilian | Irish | Scottish | Scottish | |
| Model | Mice | Blood | Tissue; Blood | Blood | Blood | Blood; Brain Tissue | Blood | Blood | Blood | Cells | Blood | |
| CCR5∆32 frequency | Patients | ↑ a | ↑ | |||||||||
| No difference | X | X | X | X | X | |||||||
| Controls | ||||||||||||
| CCR5 levels | Patients/test subject/sample | ↓ | ↓ | |||||||||
| No difference | ||||||||||||
| Controls | ||||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | X | ||||||||
| Heterozygous CCR5∆32 | X | X | X | X | ||||||||
| Absent CCR5∆32 | X a | X Px | XPx,a,CA | |||||||||
| Homozygous WT | ||||||||||||
| Association/ effect of CCR5∆32 | Protective | X | X P,C | X | X O,b | X | X CA | |||||
| No association/effect | X S,C | X S | X S | X S,C | X S | X S,C | X S,C,P | |||||
| Risk factor/non-protective | X C,M | X S,C | X O | |||||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | X | X |
| RFLP | X | |||||||||||
| Sequencing | X | |||||||||||
| Immune-based | X | |||||||||||
| Microscopy | ||||||||||||
| Spectroscopy | ||||||||||||
| Flow Cytometry | X | |||||||||||
| Chromatography | ||||||||||||
| MS | ||||||||||||
| Other | ||||||||||||
| Parameters | Immune | X | ||||||||||
| Metabolic | ||||||||||||
| Reference | [103] | [104] | [105] | [106] | [24] | [110] | [111] | [112] | [113] | [114] | [115] | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | IS | IS | IS | IS | IS | IS | IS | IS | IS | IS | IS | |
| Disease | Asthma | Asthma | Asthma | T2DM | T2DM | T1DM | T1DM | T1DM | T1DM; T2DM | Asthma; T1DM | RA | |
| Population | Mixed | Iranian | Polish | Dutch; Swedish | N/A | Italian | Hungarian | British | Estonian | Mixed | Brazilian | |
| Model | Blood | Blood | Blood | Blood; Urine | Rats: Blood; Tissue | Blood | Blood | Blood | Blood | Meta-Analysis | Blood; Synovial fluid | |
| CCR5∆32 frequency | Patients | |||||||||||
| No difference | X | X | X | X | X | X | ||||||
| Controls | ↑ b | |||||||||||
| CCR5 levels | Patients/test subject/sample | ↓ | ↑ CCR5+cells | |||||||||
| No difference | ||||||||||||
| Controls | ||||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | X | ||||||||
| Heterozygous CCR5∆32 | X | X | X | X | X | X Px | ||||||
| Absent CCR5∆32 | XPx,a | Xa | ||||||||||
| Homozygous WT | ||||||||||||
| Association /effect of CCR5∆32 | Protective | X M | X C | X C,b | X T1DM | |||||||
| No association/effect | X | X | X | X S | X | X | X S | X Asthma | X | |||
| Risk factor/non-protective | X * | |||||||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | X | |
| RFLP | X | |||||||||||
| Sequencing | X | |||||||||||
| Immune-based | X | X | ||||||||||
| Microscopy | ||||||||||||
| Spectroscopy | ||||||||||||
| Flow Cytometry | X | |||||||||||
| Chromatography | X | |||||||||||
| MS | ||||||||||||
| Other | Meta-analysis | |||||||||||
| Parameters | Immune | X | X | |||||||||
| Metabolic | X | |||||||||||
| Reference | [116] | [117] | [118] | [119] | [120] | [122] | [123] | [124] | [126] | [30] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | IS | IS | IS | IS | IS | IS | IS | IS | IS | ID | |
| Disease | RA; JIA | RA | RA; SLE | RA | RA | SLE | SLE; LN | SLE | LN | AIDS | |
| Population | Norwegian | Danish | Caucasian | New Zealand | German | Portuguese | Brazilian | Brazilian | Mixed | Mixed | |
| Model | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Blood | Meta-analysis | Meta-analysis | |
| CCR5∆32 frequency | Patients | ↓ b | ↑ * | ↓ b | ↓ European; ↑ African | ↑ | ↑ a,b,EU | ||||
| No difference | X | X | X SLE | X | |||||||
| Controls | |||||||||||
| CCR5 levels | Patients/test subject/sample | ||||||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | X C | X | X | X | X | ||||
| Heterozygous CCR5∆32 | X | X C | X | X | X | X | X | X | X | ||
| Absent CCR5∆32 | X Px (RA),a | X Px,a | X Px,a | X Px,a (European) | |||||||
| Homozygous WT | X | ||||||||||
| Association /effect of CCR5∆32 | Protective | X C | X RA,a | Xa | X S,C | X | X SLE-European | X S,P | |||
| No association/effect | X | X SLE | X C | X C | X S,Caucasian,Asian | ||||||
| Risk factor/non-protective | X LN African | X S,O | X African | ||||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | ||
| RFLP | |||||||||||
| Sequencing | |||||||||||
| Immune-based | X | X | |||||||||
| Microscopy | |||||||||||
| Spectroscopy | |||||||||||
| Flow Cytometry | |||||||||||
| Chromatography | |||||||||||
| MS | |||||||||||
| Other | X | Meta-analysis | Meta-analysis | ||||||||
| Parameters | Immune | X | |||||||||
| Metabolic | |||||||||||
| Reference | [31] | [33] | [37] | [128] | [129] | [130] | [131] | [132] | [133] | [134] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | ID | ID | ID | ID | ID | ID | ID | ID | ID | ID | |
| Disease | AIDS | AIDS | AIDS | AIDS | AIDS | AIDS | AIDS | AIDS | AIDS | AIDS | |
| Population | Caucasian | Black South African | Caucasian | European descent | Human | Italian | European descent | American | Caucasian | N/A | |
| Model | Cells | Blood | Blood | Cells | Cells | Blood; cells | Blood | Blood | Blood | Blood | |
| CCR5∆32 frequency | Patients | ↓ b | |||||||||
| No difference | X | ||||||||||
| Controls | |||||||||||
| CCR5 levels | Patients/test subject/sample | ↓ | X NE | ||||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | X | X | X | X | X | X | X | |
| Heterozygous CCR5∆32 | X | X | |||||||||
| Absent CCR5∆32 | X Px,a | ||||||||||
| Homozygous WT | |||||||||||
| Association /effect of CCR5∆32 | Protective | X | X | X S,P | X | X | |||||
| No association/effect | |||||||||||
| Risk factor/non-protective | X | X | X | X | X | ||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | X |
| RFLP | |||||||||||
| Sequencing | X | X | X | X | X | X | |||||
| Immune-based | X | X | X | X | |||||||
| Microscopy | X | ||||||||||
| Spectroscopy | X | ||||||||||
| Flow Cytometry | X | X | |||||||||
| Chromatography | |||||||||||
| MS | |||||||||||
| Other | |||||||||||
| Parameters | Immune | X | |||||||||
| Metabolic | |||||||||||
| Reference | [135] | [136] | [137] | [138] | [139] | [144] | [145] | [146] | [148] | [149] | [153] | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | ID | ID | ID | ID | ID | ID | ID | ID | ID | ID | ID | |
| Disease | AIDS | AIDS | WNF | WNF | WNF | HBV | HBV | HBV | HBV | HBV/HIV-1 | HCV | |
| Population | Caucasian | Caucasian | N/A | Caucasians | American | N/A | Iranian | Iranian | Caucasian | Brazilian | Mixed | |
| Model | Blood | Blood | Mice | Blood; CSF | Meta-analysis | Blood; CD4+ cells | Blood; NK cells | Blood; CD8+ cells | Blood | Blood | Blood | |
| CCR5∆32 frequency | Patients | ↑ Recovery | ||||||||||
| No difference | X | |||||||||||
| Controls | ||||||||||||
| CCR5 levels | Patients/test subject/sample | ↑ | ↓ | ↑ A | ↓ | ↓ | ↓ CCR5+cells | |||||
| No difference | ||||||||||||
| Controls | ↑ | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | X | X | |||||||
| Heterozygous CCR5∆32 | ||||||||||||
| Absent CCR5∆32 | X Px | X Px | ||||||||||
| Homozygous WT | X | X Px | ||||||||||
| Association/effect of CCR5∆32 | Protective | X M | X | X C | X HBV/HIV | X | ||||||
| No association/effect | XS,HBV | |||||||||||
| Risk factor/ non-protective | X | X | X | X | X | X | ||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | X | |
| RFLP | X | |||||||||||
| Sequencing | X | X | ||||||||||
| Immune-based | X | X | X | X | X | X | ||||||
| Microscopy | ||||||||||||
| Spectroscopy | ||||||||||||
| Flow Cytometry | X | X | X | X | X | |||||||
| Chromatography | ||||||||||||
| MS | ||||||||||||
| Other | Meta-analysis | |||||||||||
| Parameters | Immune | X | X | X | ||||||||
| Metabolic | ||||||||||||
| Reference | [154] | [155] | [156] | [157] | [158] | [161] | [162] | [169] | [170] | [171] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | ID | ID | ID | ID | ID | ID | ID | ID | ID | ID | |
| Disease | HCV | HCV | HCV | HCV | HCV | CD | CD | H1N1 | H1N1 | H1N1 | |
| Population | European | Caucasian | Jewish Israeli | Spanish | British | Mixed | Venezuelan | Mixed | Southern Europe | Brazilian admixed | |
| Model | Tissue | Blood | Blood; Tissue | Blood; Tissue | Blood | Blood | Blood | Blood | Blood | Blood | |
| CCR5∆32 frequency | Patients | ↑ b | ↓ | ||||||||
| No difference | X | X | X | X | X | X | |||||
| Controls | |||||||||||
| CCR5 levels | Patients/test subject/sample | ↑ Mild disease; ↓ Severe disease | |||||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | ||||||||
| Heterozygous CCR5∆32 | X | X | X | X | X | ||||||
| Absent CCR5∆32 | X Px | X | |||||||||
| Homozygous WT | |||||||||||
| Association/effect of CCR5∆32 | Protective | X C | X b | X C | |||||||
| No association/effect | X S | X S,C | X S,P | X C | X | X S | X | X | |||
| Risk factor/non-protective | X C | X C | |||||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | |
| RFLP | X | ||||||||||
| Sequencing | |||||||||||
| Immune-based | X | X | |||||||||
| Microscopy | |||||||||||
| Spectroscopy | |||||||||||
| Flow Cytometry | X | ||||||||||
| Chromatography | |||||||||||
| MS | |||||||||||
| Other | |||||||||||
| Parameters | Immune | ||||||||||
| Metabolic | |||||||||||
| Reference | [172] | [175] | [22] | [176] | [177] | [178] | [179] | [180] | [181] | [182] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | ID | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | |
| Disease | H1N1 | General | General | General | BC | BC | BC | Carcinomas | PanC | CC | |
| Population | Brazilian | N/A | Breast; tumour | Breast; tumour | Iranian | Mixed | N/A | Turkish | Mixed | Indian | |
| Model | Tissue; cells | Mice | Cells | Cells | Blood | Meta-analysis | Human cells; Mice | Blood | Blood | Tissue | |
| CCR5∆32 frequency | Patients | ↑ b,BC | ↑ b | ||||||||
| No difference | X | ||||||||||
| Controls | |||||||||||
| CCR5 levels | Patients/ test subject/ sample | ↓ | ↑ | ↓ | ↑ | ||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | |||||||||
| Heterozygous CCR5∆32 | X | X | X | X | |||||||
| Absent CCR5∆32 | X KO | X Px,a | |||||||||
| Homozygous WT | X | ||||||||||
| Association/effect of CCR5∆32 | Protective | X O,P | X | ||||||||
| No association/effect | X S.M | X | X S | X Carcinoma | X P,C | ||||||
| Risk factor/ non-protective | X | X | X BC | X | X O | ||||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | |||
| RFLP | X | ||||||||||
| Sequencing | X | X | |||||||||
| Immune-based | X | X | X | ||||||||
| Microscopy | X | ||||||||||
| Spectroscopy | X | X | X | ||||||||
| Flow Cytometry | X | X | X | X | |||||||
| Chromatography | X | ||||||||||
| MS | X | X | X | ||||||||
| Other | Meta-analysis | ||||||||||
| Parameters | Immune | ||||||||||
| Metabolic | X | X | |||||||||
| Reference | [183] | [184] | [185] | [186] | [187] | [188] | [189] | [190] | [191] | [192] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Category of Disease | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | Cancer | |
| Disease | CC; HPV | GallC | GasC | GasC | GasC | BlC | PC | PC | PC | AML | |
| Population | Swedish | Indian | Japanese | Polish | Chinese | Turkish | Turkish | Brazilian | Australian | Iranian | |
| Model | Tissue | Blood | Blood | Blood | Tissue; Cell line | Tissue | Blood | Blood | Blood | Blood | |
| CCR5∆32 frequency | Patients | ↑ (HPV+) | ↑ b | ||||||||
| No difference | X | ||||||||||
| Controls | ↑ b | ||||||||||
| CCR5 levels | Patients/test subject/sample | ↑ | ↑ | ||||||||
| No difference | |||||||||||
| Controls | |||||||||||
| Genotype | Homozygous CCR5∆32 | X | X | X | |||||||
| Heterozygous CCR5∆32 | X | X | X | X | X | ||||||
| Absent CCR5∆32 | X | X Px | |||||||||
| Homozygous WT | |||||||||||
| Association/effect of CCR5∆32 | Protective | ||||||||||
| No association/effect | X S | X | X | X S | X S | ||||||
| Risk factor/non-protective | X | X O | X C | X | X | X | |||||
| Assay(s) used | PCR | X | X | X | X | X | X | X | X | X | |
| RFLP | X | X | X | X | |||||||
| Sequencing | X | ||||||||||
| Immune-based | X | X | X | ||||||||
| Microscopy | |||||||||||
| Spectroscopy | |||||||||||
| Flow Cytometry | X | ||||||||||
| Chromatography | |||||||||||
| MS | |||||||||||
| Other | |||||||||||
| Parameters | Immune | X | |||||||||
| Metabolic | |||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rautenbach, A.; Williams, A.A. Metabolomics as an Approach to Characterise the Contrasting Roles of CCR5 in the Presence and Absence of Disease. Int. J. Mol. Sci. 2020, 21, 1472. https://doi.org/10.3390/ijms21041472
Rautenbach A, Williams AA. Metabolomics as an Approach to Characterise the Contrasting Roles of CCR5 in the Presence and Absence of Disease. International Journal of Molecular Sciences. 2020; 21(4):1472. https://doi.org/10.3390/ijms21041472
Chicago/Turabian StyleRautenbach, Anandi, and Aurelia A. Williams. 2020. "Metabolomics as an Approach to Characterise the Contrasting Roles of CCR5 in the Presence and Absence of Disease" International Journal of Molecular Sciences 21, no. 4: 1472. https://doi.org/10.3390/ijms21041472





