4.1.3. Final Products and their Characterization
1-(2-fluoro-4-hydroxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4a)
Yield 66%; mp: 270 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.91 (s, 1H), 9.75 (s, 1H), 8.70 (s, 1H), 7.83 (dd, J = 8.6, 2.3 Hz, 1H), 7.71–7.62 (m, 2H), 7.23 (td, J = 9.1, 2.6 Hz, 1H), 6.67 (dd, J = 12.5, 2.3 Hz, 1H), 6.61 (d, J = 8.8 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.13, 158.30 (d, J = 239.2 Hz), 154.87 (d, J = 11.0 Hz), 154.21 (d, J = 242.5 Hz), 151.65, 145.76, 132.71 (d, J = 7.8 Hz), 124.16, 120.90, 116.87 (d, J = 11.4 Hz), 113.80 (d, J = 24.3 Hz), 111.11 (d, J = 2.8 Hz), 108.04 (d, J = 27.0 Hz), 102.74 (d, J = 21.6 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −118.9, −124.8; ESI-HRMS: m/z 322.0454 [M+H]+ (calc. for C14H10F2N3O2S: 322.0456 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(2-fluoro-4-hydroxyphenyl)urea (4b)
Yield 94%; mp: 261–262 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.98 (s, 1H), 9.76 (s, 1H), 8.71 (s, 1H), 8.05 (d, J = 2.0 Hz, 1H), 7.68 (d, J = 9.1 Hz, 1H), 7.65 (d, J = 8.8 Hz, 1H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H), 6.67 (dd, J = 12.5, 2.6 Hz, 1H), 6.61 (dd, J = 8.8, 2.4 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.02, 154.91 (d, J = 10.9 Hz), 154.23 (d, J = 243.1 Hz), 151.62, 147.92, 133.21, 126.91, 126.17, 124.17, 121.19, 121.06, 116.82 (d, J = 11.6 Hz), 111.11 (d, J = 2.8 Hz), 102.74 (d, J = 21.6 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −124.7; ESI-HRMS: m/z 338.0157 [M+H]+ (calc. for C14H10ClFN3O2S: 338.0161 [M+H]+).
1-(2-fluoro-4-hydroxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4c)
Yield 97%; mp: 241 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.75 (s, 1H), 9.73 (s, 1H), 8.72 (s, 1H), 7.69 (t, J = 9.1 Hz, 1H), 7.56 (d, J = 8.8 Hz, 1H), 7.51 (d, J = 2.6 Hz, 1H), 6.98 (dd, J = 8.8, 2.6 Hz, 1H), 6.67 (dd, J = 12.5, 2.6 Hz, 1H), 6.64–6.58 (m, 1H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.20, 155.70, 154.72 (d, J = 10.9 Hz), 154.12 (d, J = 242.3 Hz), 151.62, 143.11, 132.66, 124.04, 120.46, 117.05 (d, J = 11.7 Hz), 114.38, 111.09 (d, J = 2.8 Hz), 104.88, 102.72 (d, J = 21.6 Hz), 55.60; 19F NMR (471 MHz, DMSO-d6): δ (ppm) -125.0; ESI-HRMS: m/z 334.0663 [M+H]+ (calc. for C15H13FN3O3S: 334.0656 [M+H]+).
1-(3-fluoro-4-hydroxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4d)
Yield 72%; mp: 243–244 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 10.84 (br s, 1H), 9.62 (br s, 1H), 9.04 (s, 1H), 7.82 (dd, J = 8.7, 2.6 Hz, 1H), 7.64 (dd, J = 8.8, 4.8 Hz, 1H), 7.43 (dd, J = 13.2, 2.4 Hz, 1H), 7.22 (td, J = 9.1, 2.7 Hz, 1H), 7.07–6.97 (m, 1H), 6.96–6.85 (m, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 159.55, 158.30 (d, J = 239.4 Hz), 150.48 (d, J = 239.5 Hz), 145.11, 140.59 (d, J = 12.2 Hz), 132.49 (d, J = 10.6 Hz), 130.26 (d, J = 9.2 Hz), 120.49 (d, J = 11.6 Hz), 117.76 (d, J = 4.0 Hz), 113.80 (d, J = 24.4 Hz), 108.22 (d, J = 11.8 Hz), 107.89 (d, J = 7.7 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) -119.0, -134.1; ESI-HRMS: m/z 322.0455 [M+H]+ (calc. for C14H10F2N3O2S: 322.0456 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3-fluoro-4-hydroxyphenyl)urea (4e)
Yield 29%; mp: 281–282 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.89 (br s, 1H), 9.61 (br s, 1H), 9.04 (s, 1H), 8.04 (d, J = 2.2 Hz, 1H), 7.63 (d, J = 8.6 Hz, 1H), 7.43 (dd, J = 13.2, 2.6 Hz, 1H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H), 7.06–6.99 (m, 1H), 6.91 (dd, J = 9.8, 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.23, 150.46 (d, J = 239.2 Hz), 140.62 (d, J = 12.0 Hz), 133.01, 130.16, 126.87, 126.20, 121.23, 120.68, 117.74 (d, J = 3.9 Hz), 115.62, 107.99 (d, J = 22.5 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) -134.2; ESI-HRMS: m/z 338.0158 [M+H]+ (calc. for C14H10ClFN3O2S: 338.0161 [M+H]+).
1-(3-fluoro-4-hydroxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4f)
Yield 30%; mp: 250 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.68 (br s, 1H), 9.57 (s, 1H), 9.05 (s, 1H), 7.54 (d, J = 8.6 Hz, 1H), 7.50 (s, 1H), 7.43 (d, J = 13.0 Hz, 1H), 7.01 (d, J = 8.4 Hz, 1H), 6.98 (d, J = 8.5 Hz, 1H), 6.91 (t, J = 9.2 Hz, 1H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.49, 155.67, 151.91, 150.47 (d, J = 239.0 Hz), 140.43 (d, J = 12.0 Hz), 132.39, 130.39, 120.06, 117.73, 115.43, 114.35, 107.82 (d, J = 22.8 Hz), 104.96, 55.60; 19F NMR (471 MHz, DMSO-d6): δ (ppm) −134.2; ESI-HRMS: m/z 334.0653 [M+H]+ (calc. for C15H13FN3O3S: 334.0656 [M+H]+).
1-(3,5-difluoro-4-hydroxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4g)
Yield 62%; mp: 317–319 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.94 (br s, 1H), 9.83 (br s, 1H), 9.25 (s, 1H), 7.88–7.79 (m, 1H), 7.65 (s, 1H), 7.28–7.19 (m, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.68, 158.31 (d, J = 239.2 Hz), 152.45, 152.16 (dd, J = 239.6, 8.7 Hz), 144.41, 132.22, 129.88 (t, J = 11.2 Hz), 129.03 (t, J = 16.3 Hz), 120.14, 113.85 (d, J = 24.2 Hz), 108.17 (d, J = 26.9 Hz), 103.34–102.69 (m); 19F NMR (471 MHz, DMSO-d6): δ (ppm) -118.8 (s, 1F), −131.0 (s, 2F); ESI-HRMS: m/z 340.0371 [M+H]+ (calc. for C14H9F3N3O2S: 340.0362 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3,5-difluoro-4-hydroxyphenyl)urea (4h)
Yield 90%; mp: 304 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 9.82 (br s, 1H), 9.34 (s, 1H), 8.04 (d, J = 2.2 Hz, 1H), 7.62 (d, J = 8.6 Hz, 1H), 7.40 (dd, J = 8.6, 2.2 Hz, 1H), 7.26–7.18 (m, 2H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.52, 152.52, 152.15 (dd, J = 239.7, 8.7 Hz), 146.42, 132.68, 129.84 (t, J = 12.6 Hz), 129.07 (t, J = 16.3 Hz), 126.95, 126.26, 121.30, 120.19, 103.24–102.85 (m); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −131.0 (s, 2F); ESI-HRMS: m/z 356.0077 [M+H]+ (calc. for C14H9ClF2N3O2S: 356.0067 [M+H]+).
1-(3,5-difluoro-4-hydroxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4i)
Yield 93%; mp: 161–162 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 9.80 (s, 1H), 9.28 (s, 1H), 7.54 (d, J = 8.9 Hz, 1H), 7.51 (d, J = 2.6 Hz, 1H), 7.27–7.17 (m, 2H), 6.98 (dd, J = 8.8, 2.6 Hz, 1H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 155.94, 155.73, 152.17 (dd, J = 239.5, 8.8 Hz), 132.01, 130.27–129.85 (m), 128.89 (t, J = 17.3 Hz), 119.74, 114.68, 114.43, 105.22, 105.08, 103.22–102.56 (m), 55.63; 19F NMR (471 MHz, DMSO-d6): δ (ppm) −131.0 (s, 2F); ESI-HRMS: m/z 352.0558 [M+H]+ (calc. for C15H12F2N3O3S: 352.0562 [M+H]+).
1-(4-fluoro-2-hydroxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4j)
Yield 78%; mp: 226–228 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.16 (s, 1H), 10.63 (s, 1H), 8.76 (s, 1H), 7.99 (dd, J = 9.0, 6.3 Hz, 1H), 7.83 (dd, J = 8.7, 2.7 Hz, 1H), 7.66 (dd, J = 8.8, 4.8 Hz, 1H), 7.22 (td, J = 9.1, 2.8 Hz, 1H), 6.69 (dd, J = 10.0, 2.9 Hz, 1H), 6.63 (td, J = 8.8, 2.9 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.10, 158.28 (d, J = 238.9 Hz), 157.91 (d, J = 239.3 Hz), 151.48, 147.73 (d, J = 10.9 Hz), 145.77, 132.71 (d, J = 10.9 Hz), 122.98 (d, J = 2.8 Hz), 120.83 (d, J = 8.9 Hz), 120.18 (d, J = 9.6 Hz), 113.76 (d, J = 24.2 Hz), 107.99 (d, J = 26.9 Hz), 105.04 (d, J = 21.8 Hz), 102.11 (d, J = 25.1 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −118.1, −118.9; ESI-HRMS: m/z 322.0453 [M+H]+ (calc. for C14H10F2N3O2S: 322.0456 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(4-fluoro-2-hydroxyphenyl)urea (4k)
Yield 94%; mp: 216–218 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.74 (br s, 1H), 8.85 (s, 1H), 8.05 (d, J = 2.2 Hz, 1H), 7.96 (dd, J = 9.0, 6.3 Hz, 1H), 7.64 (d, J = 8.6 Hz, 1H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H), 6.74 (dd, J = 10.1, 2.9 Hz, 1H), 6.62 (td, J = 8.7, 2.9 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.03, 157.99 (d, J = 239.2 Hz), 151.55, 148.00 (d, J = 11.1 Hz), 147.84, 133.22, 126.87, 126.18, 122.90 (d, J = 2.7 Hz), 121.17, 120.97, 120.31 (d, J = 9.7 Hz), 104.95 (d, J = 21.6 Hz), 102.20 (d, J = 25.2 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −118.0; ESI-HRMS: m/z 338.0157 [M+H]+ (calc. for C14H10ClFN3O2S: 338.0161 [M+H]+).
1-(4-fluoro-2-hydroxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4l)
Yield 99%; mp: 207–208 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.72 (br s, 1H), 8.88 (s, 1H), 7.96 (dd, J = 9.0, 6.3 Hz, 1H), 7.56 (d, J = 8.8 Hz, 1H), 7.52 (d, J = 2.6 Hz, 1H), 6.99 (dd, J = 8.8, 2.7 Hz, 1H), 6.75 (dd, J = 10.1, 2.9 Hz, 1H), 6.61 (td, J = 8.8, 3.0 Hz, 1H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.91 (d, J = 239.0 Hz), 157.51, 155.76, 151.54, 147.97 (d, J = 11.0 Hz), 142.36, 132.42, 123.03 (d, J = 2.8 Hz), 120.28 (d, J = 10.1 Hz), 120.17, 114.48, 104.9, 104.90 (d, J = 21.6 Hz), 102.18 (d, J = 25.0 Hz), 55.64; 19F NMR (471 MHz, DMSO-d6): δ (ppm) −118.2; ESI-HRMS: m/z 334.0652 [M+H]+ (calc. for C15H13FN3O3S: 334.0656 [M+H]+).
1-(3,4-difluoro-2-hydroxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4m)
Yield 96%; mp: 209–210 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.23 (s, 1H), 10.88 (s, 1H), 8.91 (s, 1H), 7.84 (dd, J = 8.7, 2.7 Hz, 1H), 7.83–7.78 (m, 1H), 7.67 (dd, J = 8.8, 4.7 Hz, 1H), 7.24 (td, J = 9.1, 2.7 Hz, 1H), 6.91–6.82 (m, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 159.03 (d, J = 2.3 Hz), 158.32 (d, J = 239.0 Hz), 151.73, 146.29 (dd, J = 240.5, 10.7 Hz), 145.65, 140.11 (dd, J = 238.6, 15.0 Hz), 136.27 (dd, J = 13.5, 2.0 Hz), 132.70 (d, J = 11.1 Hz), 125.39–125.25 (m), 120.87 (d, J = 9.0 Hz), 114.15 (dd, J = 8.1, 3.6 Hz), 113.84 (d, J = 24.3 Hz), 108.05 (d, J = 27.0 Hz), 106.17 (d, J = 17.7 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) -118.8, -143.9 (3J(19F, 19F) = 22.6 Hz), −158.4 (3J(19F, 19F) = 22.6 Hz); ESI-HRMS: m/z 340.0361 [M+H]+ (calc. for C14H9F3N3O2S: 340.0362 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3,4-difluoro-2-hydroxyphenyl)urea (4n)
Yield 75%; mp: 212–213 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.29 (s, 1H), 10.89 (s, 1H), 8.92 (s, 1H), 8.07 (d, J = 2.2 Hz, 1H), 7.84–7.77 (m, 1H), 7.66 (d, J = 8.6 Hz, 1H), 7.41 (dd, J = 8.6, 2.2 Hz, 1H), 6.93–6.82 (m, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.87, 151.53, 147.85, 146.26 (dd, J = 240.8, 10.7 Hz), 140.07 (dd, J = 238.4, 15.1 Hz), 136.10 (d, J = 13.3 Hz), 133.20, 126.94, 126.20, 125.26 (t, J = 3.0 Hz), 121.18, 121.04, 113.96 (d, J = 6.4 Hz), 106.19 (d, J = 17.6 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) -143.9 (3J(19F, 19F) = 22.5 Hz), −158.5 (3J(19F, 19F) = 22.5 Hz); ESI-HRMS: m/z 356.0065 [M+H]+ (calc. for C14H9ClF2N3O2S: 356.0067 [M+H]+).
1-(3,4-difluoro-2-hydroxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4o)
Yield 99%; mp: 198–200 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.73 (br s, 2H), 9.05 (s, 1H), 7.83–7.75 (m, 1H), 7.57 (d, J = 8.8 Hz, 1H), 7.52 (d, J = 2.6 Hz, 1H), 6.99 (dd, J = 8.9, 2.6 Hz, 1H), 6.90–6.80 (m, 1H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.27, 155.77, 151.68, 146.22 (dd, J = 240.9, 11.1 Hz), 142.61, 140.11 (dd, J = 238.3, 14.9 Hz), 136.18 (d, J = 13.6 Hz), 132.52, 125.46 (d, J = 3.1 Hz), 120.32, 114.47, 114.07 (dd, J = 7.5, 3.0 Hz), 106.15 (d, J = 17.4 Hz), 104.92, 55.62; 19F NMR (471 MHz, DMSO-d6): δ (ppm) -144.1 (3J(19F, 19F) = 22.6 Hz), −158.4 (3J(19F, 19F) = 22.6 Hz); ESI-HRMS: m/z 352.0560 [M+H]+ (calc. for C15H12F2N3O3S: 352.0562 [M+H]+).
1-(3-fluoro-2-methoxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4p)
Yield 85%; mp: 366–368 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.34 (s, 1H), 9.16 (br s, 1H), 8.01 (d, J = 8.4 Hz, 1H), 7.84 (dd, J = 8.7, 2.5 Hz, 1H), 7.69 (dd, J = 8.8, 4.7 Hz, 1H), 7.24 (td, J = 9.0, 2.5 Hz, 1H), 7.09 (td, J = 8.3, 6.3 Hz, 1H), 6.99–6.92 (m, 1H), 3.92 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 158.96, 158.36 (d, J = 239.3 Hz), 154.79 (d, J = 243.4 Hz), 151.24, 145.75, 136.19 (d, J = 13.5 Hz), 133.02 (d, J = 4.6 Hz), 132.63 (d, J = 10.9 Hz), 124.09 (d, J = 8.9 Hz), 121.00 (d, J = 8.1 Hz), 114.59 (d, J = 2.5 Hz), 113.88 (d, J = 24.3 Hz), 110.50 (d, J = 18.5 Hz), 108.07 (d, J = 27.0 Hz), 61.53 (d, J = 4.8 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −119.7, −130.5; ESI-HRMS: m/z 336.0607 [M+H]+ (calc. for C15H12F2N3O2S: 336.0613 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3-fluoro-2-methoxyphenyl)urea (4q)
Yield 71%; mp: 341–343 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.41 (s, 1H), 9.16 (br s, 1H), 8.06 (s, 1H), 8.00 (d, J = 8.4 Hz, 1H), 7.67 (d, J = 8.7 Hz, 1H), 7.40 (dd, J = 8.7, 1.7 Hz, 1H), 7.13–7.05 (m, 1H), 6.99–6.92 (m, 1H), 3.92 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.79, 154.78 (d, J = 243.3 Hz), 151.17, 147.94, 136.19 (d, J = 13.5 Hz), 133.15, 132.96 (d, J = 4.5 Hz), 127.04, 126.23, 124.08 (d, J = 8.9 Hz), 121.21, 121.16, 114.58 (d, J = 3.2 Hz), 110.54 (d, J = 18.6 Hz), 61.53 (d, J = 4.9 Hz); 19F NMR (471 MHz, DMSO-d6): δ (ppm) −130.5; ESI-HRMS: m/z 352.0316 [M+H]+ (calc. for C15H12ClFN3O2S: 352.0317 [M+H]+).
1-(3-fluoro-2-methoxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4r)
Yield 54%; mp: 329–331 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.20 (s, 1H), 9.22 (br s, 1H), 8.02 (d, J = 8.2 Hz, 1H), 7.59 (d, J = 8.7 Hz, 1H), 7.53 (s, 1H), 7.14–7.04 (m, 1H), 7.00 (d, J = 8.6 Hz, 1H), 6.97–6.91 (m, 1H), 3.92 (s, 3H), 3.80 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.04, 155.79, 154.82 (d, J = 244.9 Hz), 151.20, 143.14, 136.13 (d, J = 13.5 Hz), 133.18 (d, J = 4.5 Hz), 132.60, 124.08 (d, J = 9.0 Hz), 120.59, 114.56, 114.47, 110.35 (d, J = 18.6 Hz), 104.90, 61.54, 55.60; 19F NMR (471 MHz, DMSO-d6): δ (ppm) −130.5; ESI-HRMS: m/z 348.0806 [M+H]+ (calc. for C16H15FN3O3S: 348.0813 [M+H]+).
1-(4-fluoro-2-methoxyphenyl)-3-(6-fluorobenzo[d]thiazol-2-yl)urea (4s)
Yield 97%; mp: 364–366 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.30 (br s, 1H), 8.96 (br s, 1H), 8.05 (dd, J = 8.9, 6.3 Hz, 1H), 7.83 (dd, J = 8.7, 2.6 Hz, 1H), 7.71–7.62 (m, 1H), 7.23 (td, J = 9.1, 2.6 Hz, 1H), 7.01 (dd, J = 10.6, 2.6 Hz, 1H), 6.77 (td, J = 8.7, 2.7 Hz, 1H), 3.90 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.13, 158.32 (d, J = 239.6 Hz), 158.30 (d, J = 238.9 Hz), 151.52, 149.63 (d, J = 10.3 Hz), 145.67, 132.64 (d, J = 11.1 Hz), 123.66 (d, J = 3.0 Hz), 120.85 (d, J = 9.0 Hz), 119.91 (d, J = 9.4 Hz), 113.81 (d, J = 24.3 Hz), 108.02 (d, J = 27.0 Hz), 106.16 (d, J = 21.8 Hz), 99.72 (d, J = 27.2 Hz), 56.38; 19F NMR (471 MHz, DMSO-d6): δ (ppm) -116.9, −118.8; ESI-HRMS: m/z 336.0610 [M+H]+ (calc. for C15H12F2N3O2S: 336.0613 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(4-fluoro-2-methoxyphenyl)urea (4t)
Yield 94%; mp: 356–358 °C decomp.; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 11.28 (s, 1H), 8.89 (s, 1H), 8.11–8.01 (m, 2H), 7.66 (d, J = 8.6 Hz, 1H), 7.40 (dd, J = 8.6, 2.2 Hz, 1H), 7.02 (dd, J = 10.7, 2.7 Hz, 1H), 6.78 (td, J = 8.7, 2.8 Hz, 1H), 3.91 (s, 3H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 159.92, 158.33 (d, J = 239.8 Hz), 151.35, 149.52 (d, J = 10.2 Hz), 147.99, 133.21, 126.95, 126.22, 123.61 (d, J = 3.1 Hz), 121.21, 121.12, 119.76 (d, J = 9.5 Hz), 106.21 (d, J = 21.9 Hz), 99.73 (d, J = 27.2 Hz), 56.42; 19F NMR (471 MHz, DMSO-d6): δ (ppm) −116.8; ESI-HRMS: m/z 352.0316 [M+H]+ (calc. for C15H12ClFN3O2S: 352.0317 [M+H]+).
1-(4-fluoro-2-methoxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4u)
Yield 88%; mp: 324 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.07 (s, 1H), 8.94 (br s, 1H), 8.08 (dd, J = 8.9, 6.3 Hz, 1H), 7.57 (d, J = 8.8 Hz, 1H), 7.52 (d, J = 2.6 Hz, 1H), 7.01 (dd, J = 10.7, 2.8 Hz, 1H), 6.98 (dd, J = 8.8, 2.6 Hz, 1H), 6.77 (td, J = 8.7, 2.8 Hz, 1H), 3.91 (s, 3H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 158.19 (d, J = 239.3 Hz), 157.16, 155.71, 149.45 (d, J = 10.3 Hz), 143.16, 132.61, 123.82 (d, J = 3.0 Hz), 120.49, 119.68 (d, J = 9.5 Hz), 114.40, 106.15 (d, J = 21.8 Hz), 104.86, 99.67 (d, J = 27.2 Hz), 56.38, 55.59; 19F NMR (471 MHz, DMSO-d6): δ (ppm) −117.1; ESI-HRMS: m/z 348.0812 [M+H]+ (calc. for C16H15FN3O3S: 348.0813 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3-chlorophenyl)urea (4v)
Yield 83%; mp: 351–353 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.07 (br s, 1H), 9.36 (br s, 1H), 8.04 (s, 1H), 7.73 (s, 1H), 7.63 (d, J = 8.4 Hz, 1H), 7.47–7.27 (m, 3H), 7.10 (d, J = 7.3 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.42, 152.17, 146.76, 139.99, 133.28, 132.77, 130.54, 127.04, 126.30, 122.69, 121.32, 120.54, 118.29, 117.40; ESI-HRMS: m/z 337.9914 [M+H+] (calc. for C14H9Cl2N3OS: 337.9916 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(4-chlorophenyl)urea (4w)
Yield 86%; mp: 335–337 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.98 (br s, 1H), 9.29 (br s, 1H), 8.04 (s, 1H), 7.63 (d, J = 8.3 Hz, 1H), 7.55 (d, J = 8.6 Hz, 2H), 7.47–7.27 (m, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.24, 152.01, 147.33, 137.40, 132.83, 128.79, 126.99, 126.68, 126.26, 121.28, 120.48; ESI-HRMS: m/z 337.9914 [M+H+] (calc. for C14H9Cl2N3OS: 337.9916 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3,4-dichlorophenyl)urea (4x)
Yield 85%; mp: 334–336 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 11.25 (br s, 1H), 9.47 (br s, 1H), 8.03 (s, 1H), 7.90 (s, 1H), 7.77–7.19 (m, 4H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 160.70, 152.89, 145.92, 138.79, 132.47, 131.13, 130.66, 127.05, 126.31, 124.36, 121.35, 120.01, 119.01; ESI-HRMS: m/z 371.9526 [M+H+] (calc. for C14H8Cl3N3OS: 371.9526 [M+H]+).
1-(2-chloro-4-hydroxyphenyl)-3-(6-chlorobenzo[d]thiazol-2-yl)urea (4y)
Yield 71%; mp: 283–285 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.29 (br s, 1H), 9.77 (br s, 1H), 8.74 (br s, 1H), 8.05 (d, J = 2.4 Hz, 1H), 7.73 (d, J = 8.9 Hz, 1H), 7.65 (d, J = 8.6 Hz, 1H), 7.40 (dd, J = 8.6, 2.4 Hz, 1H), 6.90 (d, J = 2.6 Hz, 1H), 6.77 (dd, J = 8.9, 2.5 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.16, 154.72, 151.80, 147.81, 133.14, 126.94, 126.19, 125.94, 125.42, 125.04, 121.19, 121.00, 115.61, 114.65; ESI-HRMS: m/z 353.9864 [M+H+] (calc. for C14H9Cl2N3O2S: 353.9865 [M+H]+).
1-(3-chloro-4-methoxyphenyl)-3-(6-chlorobenzo[d]thiazol-2-yl)urea (4z)
Yield 93%; mp: 307–309 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.00 (br s, 1H), 9.12 (br s, 1H), 8.03 (d, J = 2.2 Hz, 1H), 7.68 (d, J = 2.4 Hz, 1H), 7.62 (d, J = 8.5 Hz, 1H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H), 7.36 (dd, J = 8.9, 2.3 Hz, 1H), 7.11 (d, J = 9.0 Hz, 1H), 3.83 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.39, 152.14, 150.50, 147.12, 132.84, 131.96, 126.90, 126.20, 121.23, 120.86, 120.82, 119.16, 113.04, 56.20; ESI-HRMS: m/z 368.0021 [M+H+] (calc. for C15H11Cl2N3O2S: 368.0022 [M+H]+).
2-chloro-4-(3-(6-chlorobenzo[d]thiazol-2-yl)ureido)benzoic acid (4aa)
Yield 83%; mp: 324–326 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 12.27 (br s, 1H), 9.69 (br s, 1H), 8.05 (d, J = 1.9 Hz, 1H), 7.92–7.77 (m, 2H), 7.62 (d, J = 8.6 Hz, 1H), 7.50 (dd, J = 8.6, 1.6 Hz, 1H), 7.41 (dd, J = 8.6, 2.0 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 166.00, 160.99, 152.98, 145.64, 142.54, 133.30, 132.53, 132.40, 127.13, 126.40, 124.12, 121.43, 119.90, 119.74, 116.74; ESI-HRMS: m/z 381.9815 [M+H+] (calc. for C15H9Cl2N3O3S: 381.9814 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3,5-dichloro-4-hydroxyphenyl)urea (4ab)
Yield 69%; mp: 300 °C decomp.; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 10.61 (br s, 1H), 9.19 (br s, 1H), 8.03 (d, J = 2.1 Hz, 1H), 7.61 (d, J = 8.6 Hz, 1H), 7.55 (s, 2H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 160.83, 152.82, 146.07, 144.82, 132.58, 131.55, 126.97, 126.28, 122.42, 121.33, 120.06, 119.41; ESI-HRMS: m/z 387.9476 [M+H+] (calc. for C14H8Cl3N3O2S: 387.9476 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3,5-dichloro-4-methoxyphenyl)urea (4ac)
Yield 90%; mp: 290 °C decomp.; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.26 (br s, 1H), 9.39 (br s, 1H), 8.02 (d, J = 2.0 Hz, 1H), 7.65 (s, 2H), 7.60 (d, J = 8.6 Hz, 1H), 7.40 (dd, J = 8.7, 2.0 Hz, 1H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 146.74, 135.97, 132.25, 128.14, 127.03, 126.31, 121.36, 119.07, 60.64; ESI-HRMS: m/z 401.9632 [M+H+] (calc. for C15H10Cl3N3O2S: 401.9632 [M+H]+).
3-chloro-5-(3-(6-chlorobenzo[d]thiazol-2-yl)ureido)-2-hydroxybenzoic acid (4ad)
Yield 68%; mp: 268–270 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 11.34 (br s, 1H), 9.23 (br s, 1H), 8.03 (d, J = 2.1 Hz, 1H), 7.93 (d, J = 2.6 Hz, 1H), 7.87 (d, J = 2.7 Hz, 1H), 7.61 (d, J = 8.6 Hz, 1H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 171.37, 160.76, 152.64, 146.45, 132.67, 130.24, 126.97, 126.73, 126.28, 121.30, 120.47, 119.53, 114.20; ESI-HRMS: m/z 397.9764 [M+H+] (calc. for C15H9Cl2N3O4S: 397.9764 [M+H]+).
3-chloro-5-(3-(6-chlorobenzo[d]thiazol-2-yl)ureido)-2-methoxybenzoic acid (4ae)
Yield 60%; mp: 263.5–265 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 12.10 (br s, 1H), 9.41 (br s, 1H), 8.04 (d, J = 2.1 Hz, 1H), 7.89 (d, J = 2.7 Hz, 1H), 7.79 (d, J = 2.7 Hz, 1H), 7.61 (d, J = 8.6 Hz, 1H), 7.40 (dd, J = 8.6, 2.2 Hz, 1H), 3.80 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 166.11, 160.84, 152.95, 149.82, 145.83, 135.06, 132.50, 128.18, 128.02, 127.04, 126.31, 123.23, 121.34, 119.79, 61.67; ESI-HRMS: m/z 411.9919 [M+H+] (calc. for C16H11Cl2N3O4S: 411.9920 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(2,4-dihydroxyphenyl)urea (4af)
Yield 81%; mp: 241–243 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 8.60 (br s, 1H), 8.03 (d, J = 2.1 Hz, 1H), 7.72–7.53 (m, 2H), 7.38 (dd, J = 8.6, 2.1 Hz, 1H), 6.42 (d, J = 2.6 Hz, 1H), 6.21 (dd, J = 8.7, 2.5 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.33, 153.99, 151.53, 148.23, 147.76, 133.19, 126.78, 126.16, 121.37, 121.15, 120.81, 117.75, 105.63, 102.57; ESI-HRMS: m/z 336.0202 [M+H+] (calc. for C14H10ClN3O3S: 336.0204 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3,4-dihydroxyphenyl)urea (4ag)
Yield 28%; mp: 257–259 °C; 1H NMR (500 MHz DMSO-d6): δ (ppm) 9.18 (s, 1H), 8.03 (d, J = 2.2 Hz, 1H), 7.63 (d, J = 8.6 Hz, 1H), 7.38 (ddd, J = 8.6, 2.2, 0.5 Hz, 1H), 7.03 (d, J = 2.1 Hz, 1H), 6.73–6.64 (m, 2H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.17, 151.60, 147.56, 145.29, 141.39, 133.14, 130.10, 126.75, 126.13, 121.13, 120.70, 115.58, 110.12, 107.69; ESI-HRMS: m/z 336.0202 [M+H]+ (calc. for C14H10ClN3O3S: 336.0204 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(4-hydroxy-3-methoxyphenyl)urea (4ah)
Yield 98%; mp: 257–259 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 9.42 (s, 1H), 8.03 (d, J = 2.2 Hz, 1H), 7.63 (d, J = 8.6 Hz, 1H), 7.39 (dd, J = 8.6, 2.2 Hz, 1H), 7.17 (d, J = 2.4 Hz, 1H), 6.82 (dd, J = 8.5, 2.4 Hz, 1H), 6.73 (d, J = 8.4 Hz, 1H), 3.77 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.19, 151.85, 147.51, 147.47, 142.61, 133.11, 130.21, 126.78, 126.16, 121.15, 120.68, 115.45, 111.74, 104.76, 55.61; ESI-HRMS: m/z 350.0357 [M+H]+ (calc. for C15H12ClN3O3S: 350.0361 [M+H]+).
1-(6-chlorobenzo[d]thiazol-2-yl)-3-(3-hydroxy-4-methoxyphenyl)urea (4ai)
Yield 79%; mp: 281–283 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.77 (s, 1H), 9.12 (s, 1H), 8.88 (s, 1H), 8.04 (d, J = 2.1 Hz, 1H), 7.63 (d, J = 8.7 Hz, 1H), 7.39 (dd, J = 8.7, 2.2 Hz, 1H), 7.07 (d, J = 2.4 Hz, 1H), 6.86 (d, J = 8.8 Hz, 1H), 6.82 (dd, J = 8.7, 2.4 Hz, 1H), 3.73 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.23, 151.67, 147.38, 146.73, 143.85, 133.06, 131.68, 126.81, 126.15, 121.16, 120.65, 112.81, 109.67, 107.56, 55.96; ESI-HRMS: m/z 350.0359 [M+H]+ (calc. for C15H12ClN3O3S: 350.0361 [M+H]+).
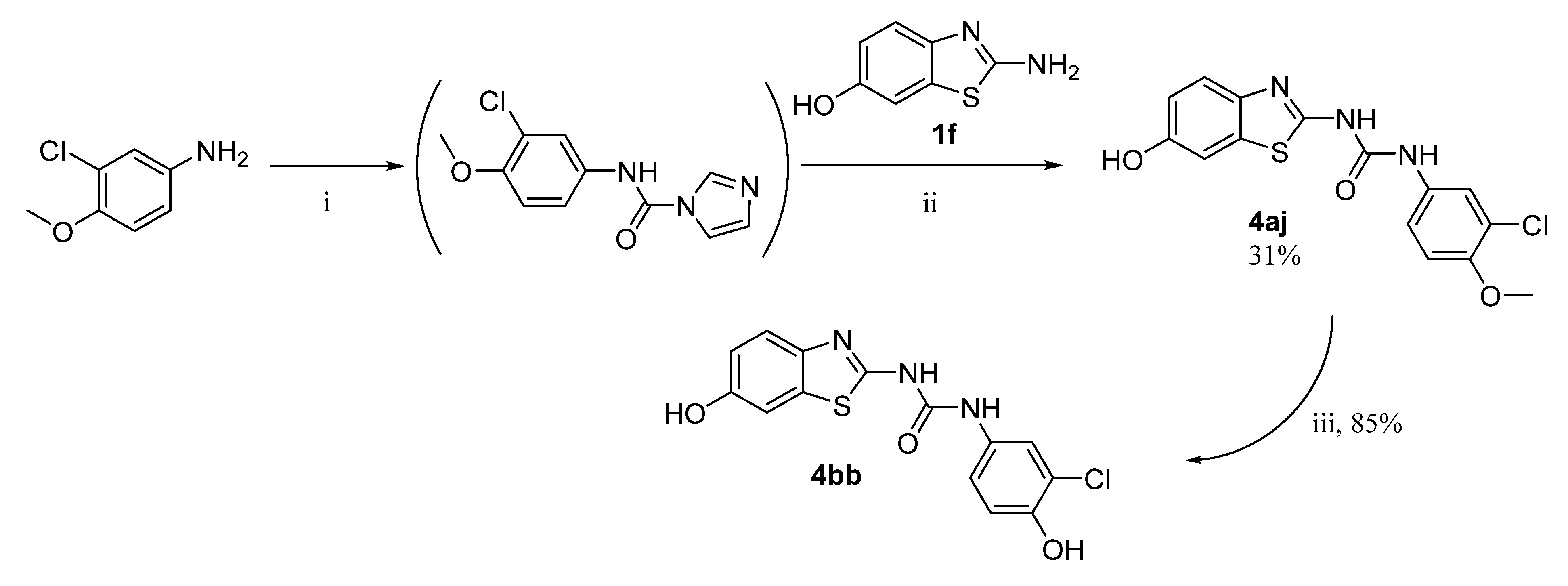
1-(3-chloro-4-methoxyphenyl)-3-(6-hydroxybenzo[d]thiazol-2-yl)urea (4aj)
Yield 31%; mp: 290–292 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.68 (s, 1H), 9.43 (s, 1H), 9.09 (s, 1H), 7.69 (d, J = 2.6 Hz, 1H), 7.45 (d, J = 8.6 Hz, 1H), 7.35 (dd, J = 8.9, 2.5 Hz, 1H), 7.22 (d, J = 2.4 Hz, 1H), 7.11 (d, J = 9.0 Hz, 1H), 6.84 (dd, J = 8.6, 2.4 Hz, 1H), 3.83 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.00, 153.67, 152.21, 150.31, 141.07, 132.26, 120.82, 120.67, 119.86, 118.95, 114.74, 113.09, 106.68, 56.20; ESI-HRMS: m/z 350.0354 [M+H]+ (calc. for C15H12ClN3O3S: 350.0361 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-chlorobenzo[d]thiazol-2-yl)thiourea (4ak)
Yield 80%; mp: 232.5–234 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 12.60 (s, 1H), 10.67 (s, 1H), 10.15 (s, 1H), 8.00 (s, 1H), 7.75–7.27 (m, 4H), 6.95 (d, J = 8.7 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 150.68, 131.10, 127.32, 126.75, 125.22, 124.04, 121.93, 118.86, 116.10, 114.96; ESI-HRMS: m/z 369.9637 [M+H]+ (calc. for C14H9Cl2N3OS2: 369.9637 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-chlorobenzo[d]thiazol-2-yl)guanidine (4al)
Yield 38%; mp: 274–275 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 10.58 (s, 1H), 8.68 (s, 1H), 8.09 (d, J = 2.0 Hz, 1H), 7.67 (d, J = 8.0 Hz, 1H), 7.52–7.40 (m, 2H), 7.18 (dd, J = 8.7, 2.5 Hz, 1H), 7.11 (d, J = 8.7 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 164.14, 155.06, 152.54, 130.55, 128.14, 127.02, 126.42, 125.50, 121.91, 119.91, 117.09; ESI-HRMS: m/z 353.0021 [M+H]+ (calc. for C14H10Cl2N4OS: 353.0025 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(4-chlorobenzo[d]thiazol-2-yl)urea (4am)
Yield 54%; mp: 237–239 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.32 (br s, 1H), 9.97 (br s, 1H), 8.83 (s, 1H), 7.89 (d, J = 7.2 Hz, 1H), 7.57 (d, J = 2.5 Hz, 1H), 7.47 (d, J = 7.2 Hz, 1H), 7.22 (t, J = 7.9 Hz, 1H), 7.17 (dd, J = 8.7, 2.6 Hz, 1H), 6.93 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.35, 151.59, 149.20, 145.83, 133.10, 130.33, 125.98, 123.70, 123.65, 121.18, 120.54, 119.83, 119.35, 116.66; ESI-HRMS: m/z 353.9861 [M+H]+ (calc. for C14H9Cl2N3O2S: 353.9865 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(5-chlorobenzo[d]thiazol-2-yl)urea (4an)
Yield 49%; mp: 317.5–319 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.97 (s, 1H), 9.98 (s, 1H), 9.01 (s, 1H), 7.93 (d, J = 8.4 Hz, 1H), 7.70 (s, 1H), 7.59 (d, J = 2.5 Hz, 1H), 7.27 (dd, J = 8.4, 2.0 Hz, 1H), 7.18 (dd, J = 8.7, 2.4 Hz, 1H), 6.93 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 161.45, 151.87, 149.93, 149.15, 130.68, 130.53, 130.17, 123.03, 122.84, 121.03, 119.70, 119.40, 119.09, 116.71; ESI-HRMS: m/z 353.9858 [M+H]+ (calc. for C14H9Cl2N3O2S: 353.9865 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(7-chlorobenzo[d]thiazol-2-yl)urea (4ao)
Yield 52%; mp: 272–274 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 9.54 (s, 1H), 7.69–7.53 (m, 2H), 7.41 (t, J = 7.9 Hz, 1H), 7.32 (d, J = 7.8 Hz, 1H), 7.19 (dd, J = 8.7, 2.2 Hz, 1H), 6.95 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.38, 152.20, 149.29, 149.05, 130.63, 130.58, 127.39, 125.23, 122.50, 120.65, 119.38, 119.30, 118.20, 116.72; ESI-HRMS: m/z 353.9857 [M+H]+ (calc. for C14H9Cl2N3O2S: 353.9865 [M+H]+).
1-(benzo[d]oxazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4ap)
Yield 59%; mp: 190.5–191.5 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.33 (s, 1H), 10.22 (s, 1H), 9.94 (s, 1H), 7.70 (d, J = 1.5 Hz, 1H), 7.60–7.47 (m, 2H), 7.33–7.18 (m, 3H), 6.95 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 156.78, 149.75, 149.39, 147.01, 140.03, 130.11, 124.62, 123.09, 121.47, 120.07, 119.34, 117.45, 116.63, 109.90; ESI-HRMS: m/z 304.0481 [M+H]+ (calc. for C14H10ClN3O3: 304.0484 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(5-chlorobenzo[d]oxazol-2-yl)urea (4aq)
Yield 89%; mp: 176–178 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.17 (s, 1H), 7.65 (d, J = 2.5 Hz, 1H), 7.62–7.57 (m, 2H), 7.27–7.23 (m, 2H), 6.97 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 158.08, 151.04, 149.25, 145.39, 140.22, 130.54, 128.71, 122.81, 121.11, 119.67, 119.35, 116.68, 116.33, 111.18; ESI-HRMS: m/z 338.0090 [M+H]+ (calc. for C14H9Cl2N3O3: 338.0094 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-chlorobenzo[d]oxazol-2-yl)urea (4ar)
Yield 16%; mp: 188.5–190.5 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.46 (s, 1H), 10.21 (s, 1H), 10.00 (s, 1H), 7.77 (d, J = 1.7 Hz, 1H), 7.67 (d, J = 2.4 Hz, 1H), 7.52 (d, J = 8.3 Hz, 1H), 7.34 (dd, J = 8.4, 2.0 Hz, 1H), 7.26 (dd, J = 8.8, 2.6 Hz, 1H), 6.94 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.70, 152.28, 149.23, 146.84, 138.05, 130.53, 126.96, 124.83, 121.26, 119.88, 119.34, 116.65, 116.62, 110.61; ESI-HRMS: m/z 338.0091 [M+H]+ (calc. for C14H9Cl2N3O3: 338.0094 [M+H]+).
1-(1H-benzo[d]imidazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4as)
Yield 90%; mp: 264–266 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.78 (br s, 3H), 9.43 (s, 1H), 7.73 (d, J = 2.6 Hz, 1H), 7.39–7.33 (m, 2H), 7.21 (dd, J = 8.7, 2.6 Hz, 1H), 7.08–7.02 (m, 2H), 6.91 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) δ 154.49, 149.23, 148.33, 134.62, 131.84, 120.96, 120.38, 119.27, 118.93, 116.61, 112.69; ESI-HRMS: m/z 303.0645 [M+H]+ (calc. for C14H11ClN4O2: 303.0643 [M+H]+).
1-(5-chloro-1H-benzo[d]imidazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4at)
Yield 37%; mp: 256–258 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 10.98 (s, 1H), 9.94 (s, 1H), 9.34 (s, 1H), 7.69 (d, J = 2.5 Hz, 1H), 7.40 (d, J = 1.9 Hz, 1H), 7.37 (d, J = 8.4 Hz, 1H), 7.18 (dd, J = 8.8, 2.5 Hz, 1H), 7.07 (dd, J = 8.4, 2.0 Hz, 1H), 6.93 (d, J = 8.7 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 152.91, 149.27, 148.69, 136.85, 134.35, 131.21, 125.03, 120.78, 120.64, 119.32, 119.19, 116.66, 114.15, 113.01; ESI-HRMS: m/z 337.0251 [M+H]+ (calc. for C14H10Cl2N4O2: 337.0254 [M+H]+).
1-(benzo[d]thiazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4au)
Yield 55%; mp: 115–117 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.90 (br s, 1H), 9.92 (s, 1H), 9.03 (s, 1H), 7.89 (d, J = 7.8 Hz, 1H), 7.63 (d, J = 8.0 Hz, 1H), 7.61 (d, J = 2.4 Hz, 1H), 7.38 (t, J = 7.6 Hz, 1H), 7.23 (t, J = 7.6 Hz, 1H), 7.20 (dd, J = 8.7, 2.3 Hz, 1H), 6.94 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.87, 152.35, 148.93, 148.17, 130.78, 125.95, 122.84, 121.52, 120.80, 119.46, 119.33, 116.65; ESI-HRMS: m/z 320.0252 [M+H]+ (calc. for C14H10ClN3O2S: 320.0255 [M+H]+).
1-(5-bromobenzo[d]thiazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4av)
Yield 50%; mp: 319–321 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.18 (br s, 1H), 9.98 (br s, 1H), 9.03 (s, 1H), 7.88 (d, J = 8.5 Hz, 1H), 7.84 (d, J = 1.2 Hz, 1H), 7.59 (d, J = 2.4 Hz, 1H), 7.39 (dd, J = 8.5, 1.2 Hz, 1H), 7.19 (dd, J = 8.9, 2.4 Hz, 1H), 6.94 (d, J = 8.9 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 161.26, 152.03, 149.78, 149.10, 130.49, 125.41, 123.32, 121.80, 120.98, 119.65, 119.34, 118.65, 116.65; ESI-HRMS: m/z 397.9359 [M+H]+ (calc. for C14H9BrClN3O2S: 397.9360 [M+H]+).
1-(6-bromobenzo[d]thiazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4aw)
Yield 38%; mp: 290–292 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.02 (br s, 1H), 9.94 (br s, 1H), 9.00 (s, 1H), 8.17 (s, 1H), 7.55 (dd, J = 30.6, 9.7 Hz, 3H), 7.18 (d, J = 8.2 Hz, 1H), 6.93 (d, J = 8.5 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.40, 152.20, 149.06, 133.39, 130.54, 128.88, 124.02, 120.92, 119.58, 119.33, 116.65, 114.66; ESI-HRMS: m/z 397.9360 [M+H]+ (calc. for C14H9BrClN3O2S: 397.9360 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-iodobenzo[d]thiazol-2-yl)urea (4ax)
Yield 58%; mp: 271–273 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.86 (br s, 1H), 9.94 (s, 1H), 9.01 (s, 1H), 8.30 (s, 1H), 7.66 (dd, J = 8.4, 1.6 Hz, 1H), 7.59 (d, J = 2.2 Hz, 1H), 7.44 (d, J = 8.2 Hz, 1H), 7.18 (dd, J = 8.6, 1.8 Hz, 1H), 6.93 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 160.16, 151.89, 149.04, 134.50, 133.90, 130.59, 129.67, 120.90, 119.56, 119.33, 116.65, 86.22; ESI-HRMS: m/z 445.9215 [M+H]+ (calc. for C14H9ClIN3O2S: 445.9221 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-methylbenzo[d]thiazol-2-yl)urea (4ay)
Yield 61%; mp: 281–283 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.87 (br s, 1H), 9.91 (br s, 1H), 9.03 (s, 1H), 7.67 (s, 1H), 7.61 (d, J = 2.1 Hz, 1H), 7.51 (d, J = 8.3 Hz, 1H), 7.19 (d, J = 8.8 Hz, 2H), 6.93 (d, J = 8.9 Hz, 1H), 2.38 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 159.13, 152.44, 148.89, 145.54, 132.20, 131.02, 130.83, 127.14, 121.22, 120.74, 119.39, 119.33, 118.70, 116.65, 20.88; ESI-HRMS: m/z 334.0407 [M+H]+ (calc. for C15H12ClN3O2S: 334.0412 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-(trifluoromethyl)benzo[d]thiazol-2-yl)urea (4az)
Yield 75%; mp: 223–225 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 9.78 (s, 1H), 8.40 (s, 1H), 7.80 (d, J = 8.5 Hz, 1H), 7.68 (dd, J = 8.5, 1.7 Hz, 1H), 7.59 (d, J = 2.5 Hz, 1H), 7.19 (dd, J = 8.6, 2.4 Hz, 1H), 6.96 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 162.41, 152.08, 151.18, 149.08, 131.92, 130.63, 124.67 (q, J = 271.8 Hz), 123.05 (q, J = 31.9 Hz), 122.83 (q, J = 3.5 Hz), 120.59, 119.73, 119.51 (q, J = 4.0 Hz), 119.40, 119.25, 116.77; 19F NMR (471 MHz, DMSO-d6): δ (ppm) -58.8; ESI-HRMS: m/z 388.0126 [M+H]+ (calc. for C15H9ClF3N3O2S: 388.0129 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-methoxybenzo[d]thiazol-2-yl)urea (4ba)
Yield 63%; mp: 281–283 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 9.33 (s, 1H), 7.69–7.40 (m, 3H), 7.17 (d, J = 7.5 Hz, 1H), 7.06–6.86 (m, 2H), 3.79 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 157.70, 155.70, 152.14, 148.88, 142.05, 132.34, 130.85, 120.57, 119.92, 119.35, 119.26, 116.71, 114.42, 104.95, 55.63; ESI-HRMS: m/z 350.0357 [M+H]+ (calc. for C15H12ClN3O3S: 350.0361 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-hydroxybenzo[d]thiazol-2-yl)urea (4bb)
Yield 85%; mp: 249–250 °C; 1H NMR (300 MHz, DMSO-d6): δ (ppm) 10.57 (s, 1H), 9.91 (s, 1H), 9.44 (s, 1H), 8.98 (s, 1H), 7.59 (d, J = 2.5 Hz, 1H), 7.44 (d, J = 8.6 Hz, 1H), 7.22 (d, J = 2.3 Hz, 1H), 7.17 (dd, J = 8.8, 2.5 Hz, 1H), 6.92 (d, J = 8.7 Hz, 1H), 6.83 (dd, J = 8.7, 2.5 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ (ppm) 156.96, 153.68, 152.29, 148.86, 141.49, 132.31, 130.88, 120.71, 120.02, 119.38, 119.33, 116.67, 114.75, 106.68; ESI-HRMS: m/z 336.0199 [M+H]+ (calc. for C14H10ClN3O3S: 336.0204 [M+H]+).
1-(6-acetylbenzo[d]thiazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4bc)
Yield 56%; mp: 242–244 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.20 (br s, 1H), 9.96 (br s, 1H), 9.06 (s, 1H), 8.57 (d, J = 1.4 Hz, 1H), 7.96 (dd, J = 8.5, 1.6 Hz, 1H), 7.68 (d, J = 8.5 Hz, 1H), 7.61 (d, J = 2.5 Hz, 1H), 7.20 (dd, J = 8.7, 2.5 Hz, 1H), 6.94 (d, J = 8.7 Hz, 1H), 2.61 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 196.69, 163.07, 152.33, 151.37, 149.12, 131.71, 131.28, 130.57, 126.24, 122.96, 120.98, 119.64, 119.38, 118.71, 116.68, 26.72; ESI-HRMS: m/z 362.0356 [M+H]+ (calc. for C16H12ClN3O3S: 362.0361 [M+H]+).
methyl 2-(3-(3-chloro-4-hydroxyphenyl)ureido)benzo[d]thiazole-6-carboxylate (4bd)
Yield 84%; mp: 278–280 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.14 (br s, 1H), 9.95 (s, 1H), 9.04 (s, 1H), 8.54 (s, 1H), 7.96 (dd, J = 8.5, 1.7 Hz, 1H), 7.69 (d, J = 8.4 Hz, 1H), 7.61 (d, J = 2.5 Hz, 1H), 7.20 (dd, J = 8.7, 2.4 Hz, 1H), 6.94 (d, J = 8.7 Hz, 1H), 3.86 (s, 3H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 166.00, 163.02, 152.18, 149.09, 131.35, 130.54, 127.11, 123.84, 123.49, 120.92, 119.58, 119.34, 116.65, 52.07; ESI-HRMS: m/z 378.0306 [M+H]+ (calc. for C16H12ClN3O4S: 378.0309 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-cyanobenzo[d]thiazol-2-yl)urea (4be)
Yield 79%; mp: 309–311 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.98 br (s, 1H), 9.74 (br s, 1H), 8.99 (s, 1H), 8.32 (s, 1H), 7.70 (d, J = 8.4 Hz, 1H), 7.68–7.64 (m, 1H), 7.55 (d, J = 2.5 Hz, 1H), 7.13 (dd, J = 8.7, 2.5 Hz, 1H), 6.90 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 162.89, 151.58, 149.07, 132.11, 130.10, 129.10, 125.95, 120.83, 119.83, 119.41, 119.18, 118.89, 116.37, 104.59; ESI-HRMS: m/z 345.0204 [M+H]+ (calc. for C15H9ClN4O2S: 345.0208 [M+H]+).
1-(3-chloro-4-hydroxyphenyl)-3-(6-nitrobenzo[d]thiazol-2-yl)urea (4bf)
Yield 52%; mp: 277–279 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 11.53 (br s, 1H), 9.99 (br s, 1H), 9.15 (s, 1H), 8.95 (d, J = 2.2 Hz, 1H), 8.22 (dd, J = 8.9, 2.3 Hz, 1H), 7.77 (d, J = 8.9 Hz, 1H), 7.59 (d, J = 2.4 Hz, 1H), 7.20 (dd, J = 8.7, 2.5 Hz, 1H), 6.94 (d, J = 8.7 Hz, 1H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 164.86, 153.25, 152.02, 149.25, 142.44, 132.01, 130.32, 121.78, 121.05, 119.71, 119.37, 119.22, 118.64, 116.67; ESI-HRMS: m/z 365.0106 [M+H]+ (calc. for C14H9ClN4O4S: 365.0106 [M+H]+).
1-(6-aminobenzo[d]thiazol-2-yl)-3-(3-chloro-4-hydroxyphenyl)urea (4bg)
Yield 77%; mp: 223–224 °C; 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.48 (s, 1H), 9.88 (s, 1H), 8.98 (s, 1H), 7.60 (d, J = 2.6 Hz, 1H), 7.32 (d, J = 8.5 Hz, 1H), 7.16 (dd, J = 8.7, 2.6 Hz, 1H), 6.97 (d, J = 2.2 Hz, 1H), 6.92 (d, J = 8.7 Hz, 1H), 6.66 (dd, J = 8.5, 2.2 Hz, 1H), 5.07 (br s, 2H); 13C NMR (126 MHz, DMSO-d6): δ (ppm) 155.24, 152.15, 148.75, 145.19, 139.03, 132.30, 130.98, 120.62, 119.61, 119.33, 119.28, 116.66, 114.00, 104.50; ESI-HRMS: m/z 335.0363 [M+H]+ (calc. for C14H11ClN4O2S: 335.0364 [M+H]+).