The Expression of Selected Factors Related to T Lymphocyte Activity in Canine Mammary Tumors
Abstract
:1. Introduction
2. Results
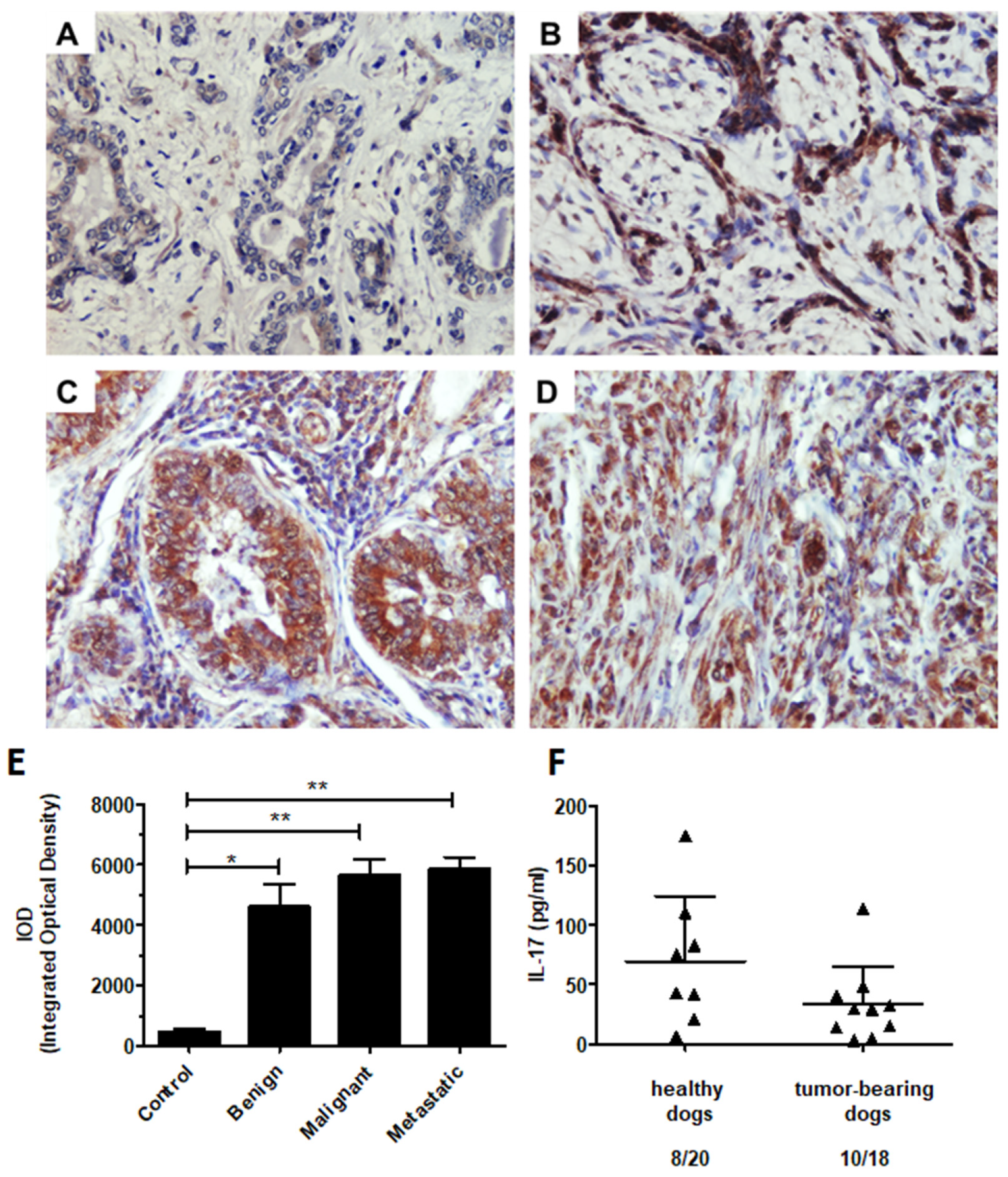
2.1. Expression of Ligands Regulating T Cell Activation
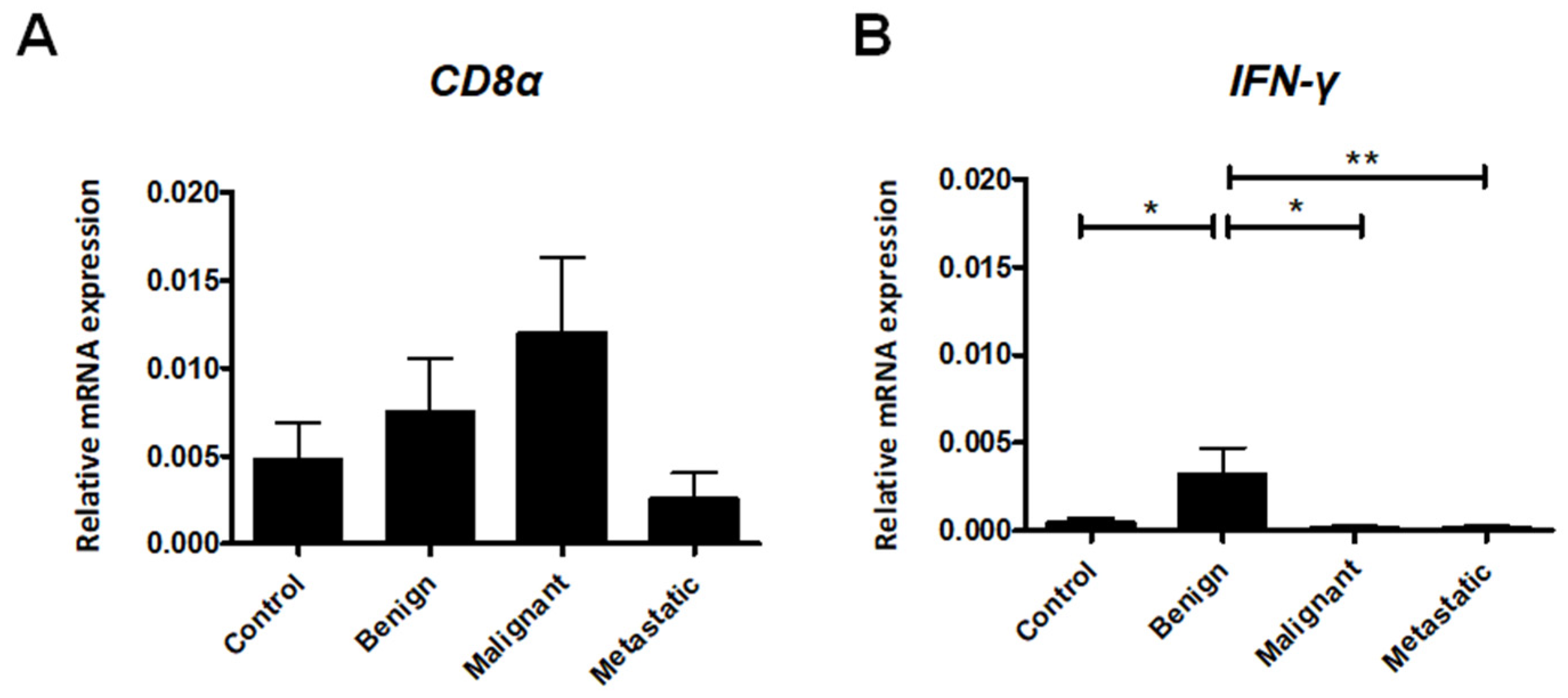
2.2. Expression of Cytotoxic T Lymphocyte Markers
2.3. Th1 Cell-Associated Gene Expression
2.4. Th2 Cell-Associated Gene Expression
2.5. Anti-Inflammatory and Pro-Inflammatory Cytokine Expression
2.6. Th17cell-Associated Gene Expression
2.7. IL-17 Protein Level in the Canine Mammary Tumors and Tumor-Bearing Dog Plasma
3. Discussion
4. Materials and Methods
4.1. Canine Mammary Tumor Samples
4.2. RNA Isolation and qPCR
4.3. Immunohistochemistry (IHC)
4.4. ELISA
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nunes, F.C.; Damasceno, K.A.; De Campos, C.B.; Bertagnolli, A.C.; Lavalle, G.E.; Cassali, G.D. Mixed tumors of the canine mammary glands: Evaluation of prognostic factors, treatment, and overall survival. Vet. Anim. Sci. 2019, 7, 100039. [Google Scholar] [CrossRef]
- Choi, J.-W.; Yoon, H.-Y.; Jeong, S.-W. Clinical Outcomes of Surgically Managed Spontaneous Tumors in 114 Client-owned Dogs. Immune Netw. 2016, 16, 116–125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philibert, J.C.; Snyder, P.W.; Glickman, N.; Glickman, L.T.; Knapp, D.W.; Waters, D.J. Influence of host factors on survival in dogs with malignant mammary gland tumors. J. Vet. Intern. Med. 2003, 17, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Abdelmegeed, S.M.; Mohammed, S. Canine mammary tumors as a model for human disease. Oncol. Lett. 2018, 15, 8195–8205. [Google Scholar] [CrossRef] [Green Version]
- Park, J.S.; Withers, S.S.; Modiano, J.F.; Kent, M.S.; Chen, M.; Luna, J.I.; Culp, W.T.N.; Sparger, E.E.; Rebhun, R.B.; Monjazeb, A.M.; et al. Canine cancer immunotherapy studies: Linking mouse and human. J. Immunother. Cancer 2016, 4, 97. [Google Scholar] [CrossRef] [Green Version]
- De Souza, T.A.; De Campos, C.B.; De Gonçalves, A.B.B.; Nunes, F.C.; Monteiro, L.N.; De Oliveira Vasconcelos, R.; Cassali, G.D. Relationship between the inflammatory tumor microenvironment and different histologic types of canine mammary tumors. Res. Vet. Sci. 2018, 119, 209–214. [Google Scholar] [CrossRef] [Green Version]
- Markkanen, E. Know Thy Model: Charting Molecular Homology in Stromal Reprogramming Between Canine and Human Mammary Tumors. Front. Cell Dev. Biol. 2019, 7. [Google Scholar] [CrossRef]
- Carvalho, M.I.; Silva-Carvalho, R.; Pires, I.; Prada, J.; Bianchini, R.; Jensen-Jarolim, E.; Queiroga, F.L. A Comparative Approach of Tumor-Associated Inflammation in Mammary Cancer between Humans and Dogs. Biomed. Res. Int. 2016, 2016. [Google Scholar] [CrossRef]
- Balkwill, F.R.; Capasso, M.; Hagemann, T. The tumor microenvironment at a glance. J. Cell Sci. 2012, 125, 5591–5596. [Google Scholar] [CrossRef] [Green Version]
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437. [Google Scholar] [CrossRef]
- Amini, P.; Nassiri, S.; Ettlin, J.; Malbon, A.; Markkanen, E. Next-generation RNA sequencing of FFPE subsections reveals highly conserved stromal reprogramming between canine and human mammary carcinoma. Dis. Models Mech. 2019, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ettlin, J.; Clementi, E.; Amini, P.; Malbon, A.; Markkanen, E. Analysis of Gene Expression Signatures in Cancer-Associated Stroma from Canine Mammary Tumours Reveals Molecular Homology to Human Breast Carcinomas. Int. J. Mol. Sci. 2017, 18, 1101. [Google Scholar] [CrossRef] [PubMed]
- Badalamenti, G.; Fanale, D.; Incorvaia, L.; Barraco, N.; Listì, A.; Maragliano, R.; Vincenzi, B.; Calò, V.; Iovanna, J.L.; Bazan, V.; et al. Role of tumor-infiltrating lymphocytes in patients with solid tumors: Can a drop dig a stone? Cell. Immunol. 2019, 343, 103753. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Chon, S.-K.; Im, K.-S.; Kim, N.-H.; Sur, J.-H. Correlation of tumor-infiltrating lymphocytes to histopathological features and molecular phenotypes in canine mammary carcinoma: A morphologic and immunohistochemical morphometric study. Can. J. Vet. Res. 2013, 77, 142–149. [Google Scholar] [PubMed]
- Carvalho, M.I.; Pires, I.; Prada, J.; Queiroga, F.L. A Role for T-Lymphocytes in Human Breast Cancer and in Canine Mammary Tumors. Available online: https://www.hindawi.com/journals/bmri/2014/130894/ (accessed on 18 January 2018).
- Farhood, B.; Najafi, M.; Mortezaee, K. CD8+ cytotoxic T lymphocytes in cancer immunotherapy: A review. J. Cell. Physiol. 2019, 234, 8509–8521. [Google Scholar] [CrossRef] [PubMed]
- Maimela, N.R.; Liu, S.; Zhang, Y. Fates of CD8+ T cells in Tumor Microenvironment. Comput. Struct. Biotechnol. J. 2019, 17, 1–13. [Google Scholar] [CrossRef]
- Estrela-Lima, A.; Araújo, M.S.; Costa-Neto, J.M.; Teixeira-Carvalho, A.; Barrouin-Melo, S.M.; Cardoso, S.V.; Martins-Filho, O.A.; Serakides, R.; Cassali, G.D. Immunophenotypic features of tumor infiltrating lymphocytes from mammary carcinomas in female dogs associated with prognostic factors and survival rates. BMC Cancer 2010, 10, 256. [Google Scholar] [CrossRef] [Green Version]
- Chraa, D.; Naim, A.; Olive, D.; Badou, A. T lymphocyte subsets in cancer immunity: Friends or foes. J. Leukoc. Biol. 2019, 105, 243–255. [Google Scholar] [CrossRef]
- Zhou, M.; Ouyang, W. The function role of GATA-3 in Th1 and Th2 differentiation. Immunol. Res. 2003, 28, 25–37. [Google Scholar] [CrossRef]
- Ellyard, J.I.; Simson, L.; Parish, C.R. Th2-mediated anti-tumour immunity: Friend or foe? Tissue Antigens 2007, 70, 1–11. [Google Scholar] [CrossRef]
- Kryczek, I.; Banerjee, M.; Cheng, P.; Vatan, L.; Szeliga, W.; Wei, S.; Huang, E.; Finlayson, E.; Simeone, D.; Welling, T.H.; et al. Phenotype, distribution, generation, and functional and clinical relevance of Th17 cells in the human tumor environments. Blood 2009, 114, 1141–1149. [Google Scholar] [CrossRef] [Green Version]
- Bailey, S.R.; Nelson, M.H.; Himes, R.A.; Li, Z.; Mehrotra, S.; Paulos, C.M. Th17 cells in cancer: The ultimate identity crisis. Front. Immunol. 2014, 5, 276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, W.; Huang, X.; Wang, J. Correlation between Th17 cells and tumor microenvironment. Cell. Immunol. 2013, 285, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Asadzadeh, Z.; Mohammadi, H.; Safarzadeh, E.; Hemmatzadeh, M.; Mahdian-shakib, A.; Jadidi-Niaragh, F.; Azizi, G.; Baradaran, B. The paradox of Th17 cell functions in tumor immunity. Cell. Immunol. 2017, 322, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Muranski, P.; Restifo, N.P. Essentials of Th17 cell commitment and plasticity. Blood 2013, 121, 2402–2414. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Farhood, B.; Mortezaee, K. Contribution of regulatory T cells to cancer: A review. J. Cell. Physiol. 2019, 234, 7983–7993. [Google Scholar] [CrossRef]
- Oh, S.Y.; Ryu, H.H.; Yoo, D.Y.; Hwang, I.K.; Kweon, O.K.; Kim, W.H. Evaluation of FOXP3 expression in canine mammary gland tumours. Vet. Comp. Oncol. 2014, 12, 20–28. [Google Scholar] [CrossRef]
- Mucha, J.; Rybicka, A.; Dolka, I.; Szymańska, J.; Manuali, E.; Parzeniecka-Jaworska, M.; Kluciński, W.; Król, M. Immunosuppression in Dogs During Mammary Cancer Development. Vet. Pathol. 2016, 53, 1147–1153. [Google Scholar] [CrossRef]
- Hartley, G.; Faulhaber, E.; Caldwell, A.; Coy, J.; Kurihara, J.; Guth, A.; Regan, D.; Dow, S. Immune regulation of canine tumour and macrophage PD-L1 expression. Vet. Comp. Oncol. 2017, 15, 534–549. [Google Scholar] [CrossRef]
- Vinogradov, S.; Warren, G.; Wei, X. Macrophages associated with tumors as potential targets and therapeutic intermediates. Nanomedicine 2014, 9, 695–707. [Google Scholar] [CrossRef] [Green Version]
- Qiu, S.-Q.; Waaijer, S.J.H.; Zwager, M.C.; De Vries, E.G.E.; Van der Vegt, B.; Schröder, C.P. Tumor-associated macrophages in breast cancer: Innocent bystander or important player? Cancer Treat. Rev. 2018, 70, 178–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, E.-S.; Uchida, K.; Nakayama, H. Th1-, Th2-, and Th17-Related Cytokine and Chemokine Receptor mRNA and Protein Expression in the Brain Tissues, T Cells, and Macrophages of Dogs with Necrotizing and Granulomatous Meningoencephalitis. Vet. Pathol. 2013, 50, 1127–1134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rozali, E.N.; Hato, S.V.; Robinson, B.W.; Lake, R.A.; Lesterhuis, W.J. Programmed Death Ligand 2 in Cancer-Induced Immune Suppression. Clin. Dev. Immunol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Sideras, K.; Biermann, K.; Verheij, J.; Takkenberg, B.R.; Mancham, S.; Hansen, B.E.; Schutz, H.M.; De Man, R.A.; Sprengers, D.; Buschow, S.I.; et al. PD-L1, Galectin-9 and CD8+tumor-infiltrating lymphocytes are associated with survival in hepatocellular carcinoma. OncoImmunology 2017, 6, e1273309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chou, F.-C.; Chen, H.-Y.; Kuo, C.-C.; Sytwu, H.-K. Role of Galectins in Tumors and in Clinical Immunotherapy. Int. J. Mol. Sci. 2018, 19, 430. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; Manzotti, C.N.; Liu, M.; Burke, F.; Mead, K.I.; Sansom, D.M. CD86 and CD80 Differentially Modulate the Suppressive Function of Human Regulatory T Cells. J. Immunol. 2004, 172, 2778–2784. [Google Scholar] [CrossRef]
- Kanhere, A.; Hertweck, A.; Bhatia, U.; Gökmen, M.R.; Perucha, E.; Jackson, I.; Lord, G.M.; Jenner, R.G. T-bet and GATA3 orchestrate Th1 and Th2 differentiation through lineage-specific targeting of distal regulatory elements. Nat. Commun. 2012, 3, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Tokunaga, R.; Zhang, W.; Naseem, M.; Puccini, A.; Berger, M.D.; Soni, S.; McSkane, M.; Baba, H.; Lenz, H.-J. CXCL9, CXCL10, CXCL11/CXCR3 axis for immune activation—A target for novel cancer therapy. Cancer Treat. Rev. 2018, 63, 40–47. [Google Scholar] [CrossRef]
- Gu, L.; Tseng, S.; Horner, R.M.; Tam, C.; Loda, M.; Rollins, B.J. Control of TH2 polarization by the chemokine monocyte chemoattractant protein-1. Nature 2000, 404, 407–411. [Google Scholar] [CrossRef]
- Landskron, G.; De la Fuente, M.; Thuwajit, P.; Thuwajit, C.; Hermoso, M.A. Chronic Inflammation and Cytokines in the Tumor Microenvironment. J. Immunol. Res. 2014, 2014. [Google Scholar] [CrossRef] [Green Version]
- Changkija, B.; Konwar, R. Role of interleukin-10 in breast cancer. Breast Cancer Res. Treat. 2012, 133, 11–21. [Google Scholar]
- Lasek, W.; Zagożdżon, R.; Jakobisiak, M. Interleukin 12: Still a promising candidate for tumor immunotherapy? Cancer Immunol. Immunother. 2014, 63, 419–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carvalho, M.I.; Pires, I.; Prada, J.; Queiroga, F.L. T-lymphocytic infiltrate in canine mammary tumours: Clinic and prognostic implications. In Vivo 2011, 25, 963–969. [Google Scholar]
- Carvalho, M.I.; Pires, I.; Dias, M.; Prada, J.; Gregório, H.; Lobo, L.; Queiroga, F. Intratumoral CD3+ T-lymphocytes immunoexpression and its association with c-Kit, angiogenesis, and overall survival in malignant canine mammary tumors. Anal. Cell. Pathol. Amst. 2015, 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shosu, K.; Sakurai, M.; Inoue, K.; Nakagawa, T.; Sakai, H.; Morimoto, M.; Okuda, M.; Noguchi, S.; Mizuno, T. Programmed Cell Death Ligand 1 Expression in Canine Cancer. In Vivo 2016, 30, 195–204. [Google Scholar] [PubMed]
- Raposo, T.; Gregório, H.; Pires, I.; Prada, J.; Queiroga, F.L. Prognostic value of tumour-associated macrophages in canine mammary tumours: Prognostic value of TAMs in CMT. Vet. Comp. Oncol. 2014, 12, 10–19. [Google Scholar] [CrossRef]
- Karnik, T.; Kimler, B.F.; Fan, F.; Tawfik, O. PD-L1 in breast cancer: Comparative analysis of 3 different antibodies. Hum. Pathol. 2018, 72, 28–34. [Google Scholar] [CrossRef]
- Baptista, M.Z.; Sarian, L.O.; Derchain, S.F.M.; Pinto, G.A.; Vassallo, J. Prognostic significance of PD-L1 and PD-L2 in breast cancer. Hum. Pathol. 2016, 47, 78–84. [Google Scholar] [CrossRef]
- Irie, A. Galectin-9 as a Prognostic Factor with Antimetastatic Potential in Breast Cancer. Clin. Cancer Res. 2005, 11, 2962–2968. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Sun, L.; Jing, D.; Xu, G.; Zhang, J.; Lin, L.; Zhao, J.; Yao, Z.; Lin, H. Galectin-9 Expression Predicts Favorable Clinical Outcome in Solid Tumors: A Systematic Review and Meta-Analysis. Front. Physiol. 2018, 9, 452. [Google Scholar] [CrossRef] [Green Version]
- Kageshita, T.; Kashio, Y.; Yamauchi, A.; Seki, M.; Abedin, M.J.; Nishi, N.; Shoji, H.; Nakamura, T.; Ono, T.; Hirashima, M. Possible role of galectin-9 in cell aggregation and apoptosis of human melanoma cell lines and its clinical significance. Int. J. Cancer 2002, 99, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.; Ueno, M.; Oomizu, S.; Arikawa, T.; Shinonaga, R.; Zhang, S.; Yamauchi, A.; Hirashima, M. Galectin-9 expression links to malignant potential of cervical squamous cell carcinoma. J. Cancer Res. Clin. Oncol. 2008, 134, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, J.; Ma, C.; Gao, W.; Song, B.; Xue, H.; Chen, W.; Chen, X.; Zhang, Y.; Shao, Q.; et al. Reduced Expression of Galectin-9 Contributes to a Poor Outcome in Colon Cancer by Inhibiting NK Cell Chemotaxis Partially through the Rho/ROCK1 Signaling Pathway. PLoS ONE 2016, 11, e0152599. [Google Scholar] [CrossRef] [Green Version]
- Mulligan, A.M.; Pinnaduwage, D.; Tchatchou, S.; Bull, S.B.; Andrulis, I.L. Validation of Intratumoral T-bet+ Lymphoid Cells as Predictors of Disease-Free Survival in Breast Cancer. Cancer Immunol. Res. 2016, 4, 41–48. [Google Scholar] [CrossRef] [Green Version]
- Kouros-Mehr, H.; Kim, J.; Bechis, S.K.; Werb, Z. GATA-3 and the regulation of the mammary luminal cell fate. Curr. Opin. Cell Biol. 2008, 20, 164–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gentile, L.B.; Nagamine, M.K.; Biondi, L.R.; Sanches, D.S.; Toyota, F.; Giovani, T.M.; De Jesus, I.P.; Da Fonseca, I.I.M.; Queiroz-Hazarbassanov, N.; Diaz, B.L.; et al. Establishment of primary mixed cell cultures from spontaneous canine mammary tumors: Characterization of classic and new cancer-associated molecules. PLoS ONE 2017, 12, e0184228. [Google Scholar] [CrossRef] [Green Version]
- Kouros-Mehr, H.; Bechis, S.K.; Slorach, E.M.; Littlepage, L.E.; Egeblad, M.; Ewald, A.J.; Pai, S.-Y.; Ho, I.-C.; Werb, Z. GATA-3 links tumor differentiation and dissemination in a luminal breast cancer model. Cancer Cell 2008, 13, 141–152. [Google Scholar] [CrossRef] [Green Version]
- Fang, S.H.; Chen, Y.; Weigel, R.J. GATA-3 as a marker of hormone response in breast cancer. J. Surg. Res. 2009, 157, 290–295. [Google Scholar] [CrossRef]
- McCune, K.; Bhat-Nakshatri, P.; Thorat, M.A.; Nephew, K.P.; Badve, S.; Nakshatri, H. Prognosis of Hormone-Dependent Breast Cancers: Implications of the Presence of Dysfunctional Transcriptional Networks Activated by Insulin via the Immune Transcription Factor T-bet. Cancer Res. 2010, 70, 685–696. [Google Scholar] [CrossRef] [Green Version]
- Nieto, A.; Peña, L.; Pérez-Alenza, M.D.; Sánchez, M.A.; Flores, J.M.; Castaño, M. Immunohistologic detection of estrogen receptor alpha in canine mammary tumors: Clinical and pathologic associations and prognostic significance. Vet. Pathol. 2000, 37, 239–247. [Google Scholar] [CrossRef] [Green Version]
- Toniti, W.; Buranasinsup, S.; Kongcharoen, A.; Charoonrut, P.; Puchadapirom, P.; Kasorndorkbua, C. Immunohistochemical determination of estrogen and progesterone receptors in canine mammary tumors. Asian Pac. J. Cancer Prev. 2009, 10, 907–911. [Google Scholar] [PubMed]
- Oh, T.G.; Wang, S.-C.M.; Acharya, B.R.; Goode, J.M.; Graham, J.D.; Clarke, C.L.; Yap, A.S.; Muscat, G.E.O. The Nuclear Receptor, RORγ, Regulates Pathways Necessary for Breast Cancer Metastasis. EBioMedicine 2016, 6, 59–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hembruff, S.L.; Cheng, N. Chemokine signaling in cancer: Implications on the tumor microenvironment and therapeutic targeting. Cancer Ther. 2009, 7, 254–267. [Google Scholar] [PubMed]
- Reynders, N.; Abboud, D.; Baragli, A.; Noman, M.Z.; Rogister, B.; Niclou, S.P.; Heveker, N.; Janji, B.; Hanson, J.; Szpakowska, M.; et al. The Distinct Roles of CXCR3 Variants and Their Ligands in the Tumor Microenvironment. Cells 2019, 8, 613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, G.; Yan, H.H.; Pang, Y.; Jian, J.; Achyut, B.R.; Liang, X.; Weiss, J.M.; Wiltrout, R.H.; Hollander, M.C.; Yang, L. CXCR3 as a molecular target in breast cancer metastasis: Inhibition of tumor cell migration and promotion of host anti-tumor immunity. Oncotarget 2015, 6, 43408. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Xu, L.; Peng, M. CXCR3 is a prognostic marker and a potential target for patients with solid tumors: A meta-analysis. Oncotargets Ther. 2018, 11, 1045–1054. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.Y.; Yuzhalin, A.E.; Gordon-Weeks, A.N.; Muschel, R.J. Targeting the CCL2-CCR2 signaling axis in cancer metastasis. Oncotarget 2016, 7, 28697. [Google Scholar] [CrossRef] [Green Version]
- Raposo, T.P.; Beirão, B.C.B.; Pires, I.; Prada, J.; Brilhante, P.; Argyle, D.J.; Queiroga, F.L. Immunohistochemical Expression of CCR2, CSF1R and MMP9 in Canine Inflammatory Mammary Carcinomas. Anticancer Res. 2016, 36, 1805–1813. [Google Scholar]
- Sheu, B.-C.; Chang, W.-C.; Cheng, C.-Y.; Lin, H.-H.; Chang, D.-Y.; Huang, S.-C. Cytokine regulation networks in the cancer microenvironment. Front. Biosci. 2008, 13, 6255–6268. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.-H.; Yu, C.-H.; Yhee, J.-Y.; Im, K.-S.; Sur, J.-H. Lymphocyte infiltration, expression of interleukin (IL) -1, IL-6 and expression of mutated breast cancer susceptibility gene-1 correlate with malignancy of canine mammary tumours. J. Comp. Pathol. 2010, 142, 177–186. [Google Scholar] [CrossRef]
- Machado, V.S.; Crivellenti, L.Z.; Bottari, N.B.; Tonin, A.A.; Pelinson, L.P.; Borin-Crivellenti, S.; Santana, A.E.; Torbitz, V.D.; Moresco, R.N.; Duarte, T.; et al. Oxidative stress and inflammatory response biomarkers in dogs with mammary carcinoma. Pathol. Res. Pract. 2015, 211, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Heckel, M.C.; Wolfson, A.; Slachta, C.A.; Schwarting, R.; Salgame, P.; Katsetos, C.D.; Platsoucas, C.D. Human breast tumor cells express IL-10 and IL-12p40 transcripts and proteins, but do not produce IL-12p70. Cell. Immunol. 2011, 266, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Lyon, D.E.; McCain, N.L.; Walter, J.; Schubert, C. Cytokine comparisons between women with breast cancer and women with a negative breast biopsy. Nurs. Res. 2008, 57, 51–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Andrés, P.J.; Illera, J.C.; Cáceres, S.; Díez, L.; Pérez-Alenza, M.D.; Peña, L. Increased levels of interleukins 8 and 10 as findings of canine inflammatory mammary cancer. Vet. Immunol. Immunopathol. 2013, 152, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Mulcahy, L.A.; Mohammed, R.A.; Lee, A.H.; Franks, H.A.; Kilpatrick, L.; Yilmazer, A.; Paish, E.C.; Ellis, I.O.; Patel, P.M.; et al. IL-17 expression by breast-cancer-associated macrophages: IL-17 promotes invasiveness of breast cancer cell lines. Breast Cancer Res. 2008, 10, R95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corrigan, C.J.; Wang, W.; Meng, Q.; Fang, C.; Eid, G.; Caballero, M.R.; Lv, Z.; An, Y.; Wang, Y.-H.; Liu, Y.-J.; et al. Allergen-induced expression of IL-25 and IL-25 receptor in atopic asthmatic airways and late-phase cutaneous responses. J. Allergy Clin. Immunol. 2011, 128, 116–124. [Google Scholar] [CrossRef]
- Li, H.; Chen, J.; Huang, A.; Stinson, J.; Heldens, S.; Foster, J.; Dowd, P.; Gurney, A.L.; Wood, W.I. Cloning and characterization of IL-17B and IL-17C, two new members of the IL-17 cytokine family. Proc. Natl. Acad. Sci. USA 2000, 97, 773–778. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.-C.; Lai, Y.-H.; Chen, H.-Y.; Guo, H.-R.; Su, I.-J.; Chen, H.H.W. Interleukin-17-producing cell infiltration in the breast cancer tumour microenvironment is a poor prognostic factor. Histopathology 2013, 63, 225–233. [Google Scholar] [CrossRef]
- Faucheux, L.; Grandclaudon, M.; Perrot-Dockès, M.; Sirven, P.; Berger, F.; Hamy, A.S.; Fourchotte, V.; Vincent-Salomon, A.; Mechta-Grigoriou, F.; Reyal, F.; et al. A multivariate Th17 metagene for prognostic stratification in T cell non-inflamed triple negative breast cancer. OncoImmunology 2019, 8, e1624130. [Google Scholar] [CrossRef]
- Lv, Z.; Liu, M.; Shen, J.; Xiang, D.; Ma, Y.; Ji, Y. Association of serum interleukin-10, interleukin-17A and transforming growth factor-α levels with human benign and malignant breast diseases. Exp. Ther. Med. 2018, 15, 5475–5480. [Google Scholar] [CrossRef]
- Wim, M. Armed Forces Institute of Pathology (U.S.), American Registry of Pathology, WHO Collaborating Center for Worldwide Reference on Comparative Oncology. International histological classification of tumors of domestic animals. Histol. Classif. Mammary Tumors Dog Cat 1999, 7, 1–59. [Google Scholar]
- Goldschmidt, M.; Peña, L.; Rasotto, R.; Zappulli, V. Classification and grading of canine mammary tumors. Vet. Pathol. 2011, 48, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Im, K.S.; Kim, N.H.; Lim, H.Y.; Kim, H.W.; Shin, J.I.; Sur, J.H. Analysis of a new histological and molecular-based classification of canine mammary neoplasia. Vet. Pathol. 2014, 51, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]





| Gene group | Gene | Accession no. | Sequence | Product Size (bp) |
|---|---|---|---|---|
| Ligands regulating T cell activation | PD-L2 | XM_847012.5 | F: ACTGGCTGCTTCATGTTGTC | 169 |
| R: CAGGTTCAAATAGCTCCGTCC | ||||
| Gal9 | NM_001003345.1 | F: GCTGCGATTTCAAGGTGACG | 134 | |
| R: GCCTGGAGACTGGAAGCTAA | ||||
| CD8 | NM_001003146.2 | F: GAAACCCACCCCTGATGGAG | 191 | |
| R: ACCGTACTCTTTCCTTGGTCTG | ||||
| T cytotoxic cell-associated genes | CD8α | NM_001002935.2 | F: GTGGGTTAGACTTCGCCTGT | 117 |
| R: CACGTCTTCTGTTCCTGTGGT | ||||
| IFN-γ | AF126247.1 | F: TCAAGGAAGACATGCTTGGCAAGTT | 74 | |
| R: GACCTGCAGATCGTTCACAGGAAT | ||||
| Th1 cell-associated genes | CXCR3 | NM 001011887.1 | F: TGGATGTGGCCAAGTCTGTC | 200 |
| R: TGAGGGGGTCTCGGACCAG | ||||
| TBX21 | XM_548164.4 | F: AAGCAGGGGCGGCGGATGTT | 139 | |
| R: ACTGCACCCACTTGCCGCTC | ||||
| Th2 cell-associated genes | IL-4 | AF187322.1 | F: TCACCAGCACCTTTGTCCACGG | 96 |
| R: TGCACGAGTCGTTTCTCGCTGT | ||||
| GATA3 | XM 844060.1 | F: CGAAGGCTGTCGGCAGCAAGAA | 98 | |
| R: ACGGGGTCTCCGTTGGCATT | ||||
| CCR2 | XM 541906.1 | F: ACATGCTGTCCACATCGCA | 91 | |
| R: GGCGCGCTGTAATCATAGTC | ||||
| Anti-inflammatory and pro-inflammatory cytokines | IL-10 | NM 0010030771 | F: CAGGTGAAGAGCGCATTTAGT | 107 |
| R: TCAAACTCACTCATGGCTTTGT | ||||
| IL-12A p35 | NM 001003293.1 | F: TGCCTGGCCTCTGGAAAG | 74 | |
| R: TACATCTTCAAGTCCTCAT | ||||
| IL-12B p40 | NM 001003292.1 | F: GCCAAGGTCGTGTGCCA | 81 | |
| R: CCAGTCGCTCCAGGATGAAC | ||||
| Th17 cell-associated genes | IL-17 | NM 001165878.1 | F: CACTCCTTCCGGCTAGAGAA | 71 |
| R: CACATGGCGAACAATAGGG | ||||
| RORγt | XM 540323.3 | F: TCAACGCCAACCGTCCGGG | 143 | |
| R: CCGAAGCTTCCCCTTGGGCG | ||||
| Housekeeping genes | HPRT | NG_042858.1 | F: TTATAGTCAAGGGCATATCC | 104 |
| R: AGCTTGCTGGTGAAAAGGAC | ||||
| RPS19 | XM_005616513.3 | F: GTTCTCATCGTAGGGAGCAAG | 95 | |
| R: CCTTCCTCAAAAAGTCTGGG |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bujak, J.K.; Szopa, I.M.; Pingwara, R.; Kruczyk, O.; Krzemińska, N.; Mucha, J.; Majchrzak-Kuligowska, K. The Expression of Selected Factors Related to T Lymphocyte Activity in Canine Mammary Tumors. Int. J. Mol. Sci. 2020, 21, 2292. https://doi.org/10.3390/ijms21072292
Bujak JK, Szopa IM, Pingwara R, Kruczyk O, Krzemińska N, Mucha J, Majchrzak-Kuligowska K. The Expression of Selected Factors Related to T Lymphocyte Activity in Canine Mammary Tumors. International Journal of Molecular Sciences. 2020; 21(7):2292. https://doi.org/10.3390/ijms21072292
Chicago/Turabian StyleBujak, Joanna K., Iwona M. Szopa, Rafał Pingwara, Olga Kruczyk, Natalia Krzemińska, Joanna Mucha, and Kinga Majchrzak-Kuligowska. 2020. "The Expression of Selected Factors Related to T Lymphocyte Activity in Canine Mammary Tumors" International Journal of Molecular Sciences 21, no. 7: 2292. https://doi.org/10.3390/ijms21072292






