Effects of Cholesterol in Stress-Related Neuronal Death—A Statistical Analysis Perspective
Abstract
:1. Introduction
2. Materials and Methods
2.1. Culture of Neurons/N27 Cells
2.2. Calcein AM Viability Assay
2.3. Neuron Death Behavior
2.4. Cholesterol Assay and MßCD-Cholesterol Modeling
2.5. MßCD-tBuOOH Cell Viability Assay and Modeling
3. Results
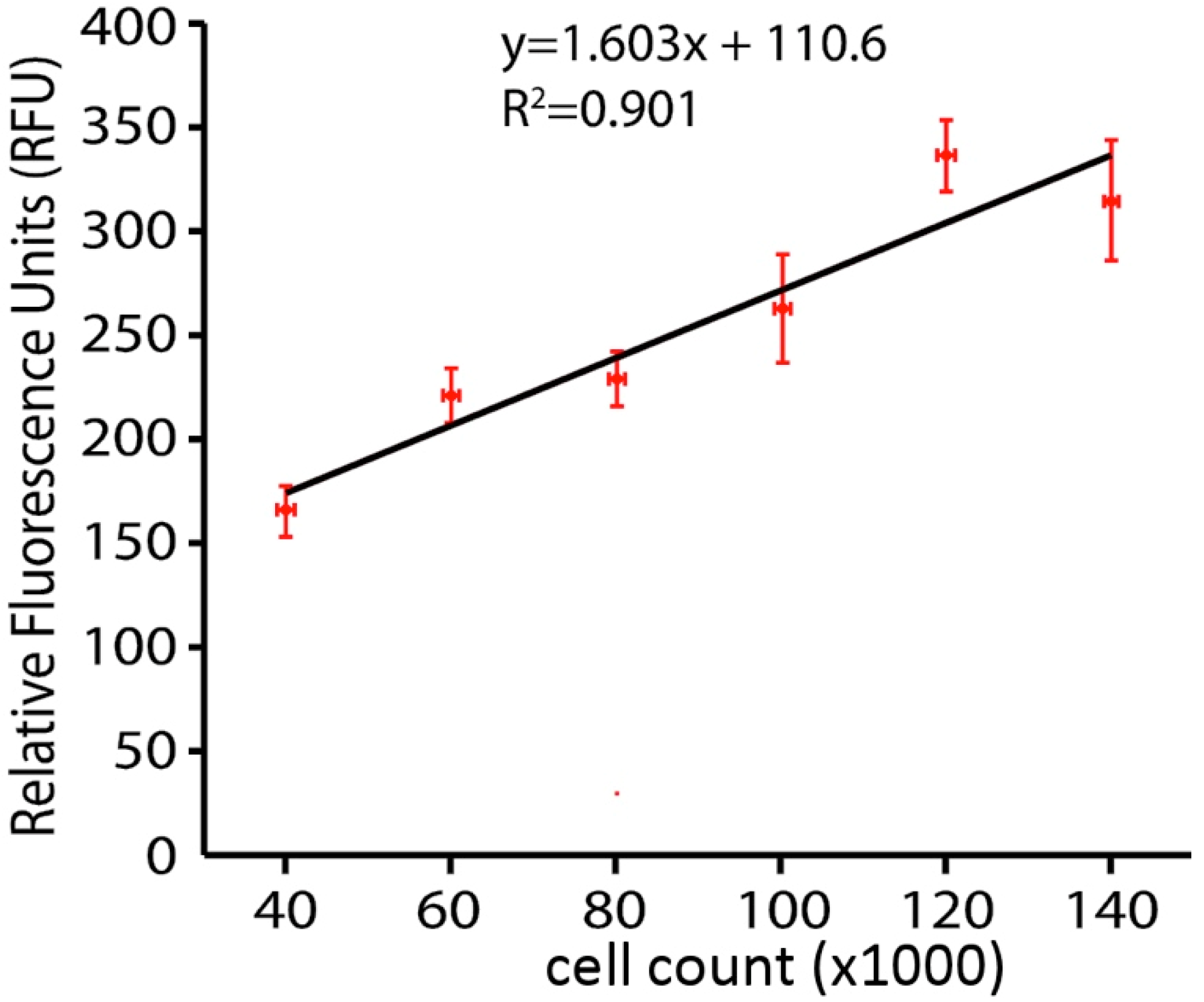
3.1. Methyl-ß-Cyclodextrin (MßCD) Depletion of Plasma Membrane Cholesterol
3.2. Effect of MßCD’s reduction of Plasma Membrane Cholesterol on the Sensitivity of N27 Cells
4. Statistical Analysis
5. Experimental Limitations
6. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pike, L.J. The challenge of lipid rafts. J. Lipid Res. 2009, 50, S323–S328. [Google Scholar] [CrossRef] [Green Version]
- Claas, C.; Stipp, C.S.; Hemler, M.E. Evaluation of Prototype Transmembrane 4 Superfamily Protein Complexes and Their Relation to Lipid Rafts. J. Biol. Chem. 2001, 276, 7974. [Google Scholar] [CrossRef] [Green Version]
- Frühbeck, G. Intracellular signaling pathways activated by leptin. Biochem. J. 2006, 393, 7–20. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.C.; Liu, C.C.; Kanekiyo, T.; Xu, H.; Bu, G. Apolipoprotein E and Alzheimer disease: Risk, mechanisms and therapy. Nat. Rev. Neurol. 2013, 9, 106–118. [Google Scholar] [CrossRef] [Green Version]
- Linay, K.J.L.; Dominguez, J.C.; Daroy, M.L.; Reandelar, M.; Casingal, C. Apolipoproteine polymorphism distribution among filipino elderly community dwellers. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2015, 11, P828. [Google Scholar] [CrossRef]
- Chen, X.; Guo, C.; Kong, J. Oxidative stress in neurodegenerative diseases. Neural Regen. Res. 2012, 7, 376–385. [Google Scholar] [PubMed]
- Aiken, C.T.; Kaake, R.M.; Wang, X.; Huang, L. Oxidative stress-mediated regulation of proteasome complexes. Mol. Cell Proteom. 2011, 10, R110.006924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ienco, E.C.; LoGerfo, A.; Carlesi, C.; Orsucci, D.; Ricci, G.; Mancuso, M.; Siciliano, G. Oxidative stress treatment for clinical trials in neurodegenerative diseases. J. Alzheimers Dis. 2011, 24, 111–126. [Google Scholar] [CrossRef]
- Axelsen, P.H.; Komatsu, H.; Murray, I.V.J. Oxidative Stress and Cell Membranes in the Pathogenesis of Alzheimer’s Disease. Physiology 2011, 26, 54–69. [Google Scholar] [CrossRef]
- Danning, S.; ur Rehman, A.; Tiebosch, M.H.; Hannivoort, R.A.; Haijer, F.W.; Woudenberg, J.; van den Heuvel, F.A.J.; Buist-Homan, M.; Faber, K.N.; Moshage, H. Glutathione and antioxidant enzymes serve complementary roles in protecting activated hepatic stellate cells against hydrogen peroxide-induced cell death. Biochimica et Biophysica Acta 2013, 1832, 2027–2034. [Google Scholar] [CrossRef] [Green Version]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense et al. World Allergy Organ J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bratic, A.; Larsson, N.-G. The role of mitochondria in aging. J. Clin. Investig. 2013, 123, 951–957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trifunovic, A.; Wredenberg, A.; Falkenberg, M.; Spelbrink, J.N.; Rovio, A.T.; Bruder, C.E.; Bohlooly, Y.M.; Gidlöf, S.; Oldfors, A.; Wibom, R.; et al. Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 2004, 429, 417–423. [Google Scholar] [CrossRef]
- Vance, J.E.; Hayashi, H.; Karen, B. Cholesterol homeostasis in neurons and glial cells. Semin. Cell Dev. Biol. 2005, 16, 193–212. [Google Scholar] [CrossRef] [PubMed]
- Romano, T.A.; Ridgway, S.H.; Felten, D.L.; Quaranta, V. Molecular Cloning and Characterization of CD4 in an Aquatic Mammal, the White Whale Delphinapterus leucas. Immunogenetics 1999, 49, 376–383. [Google Scholar] [CrossRef]
- Jarvik, G.P. Complex Segregation Analyses: Uses and Limitations. Am. J. Hum. Genet. 1998, 63, 942–946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evans, R.M.; Emsley, C.L.; Gao, S.; Sahota, A.; Hall, K.S.; Farlow, M.R.; Hendrie, H. Serum Cholesterol, APOE Genotype, and the Risk of Alzheimer’s Disease: A Population-Based Study of African Americans. Neurology 2000, 54, 240–242. [Google Scholar] [CrossRef]
- Kivipelto, M.; Laasko, M.P.; Tuomilehto, J.; Nissinen, A.; Soininen, H. Hypertension and Hypercholesterolaemia as Risk Factors for Alzheimer’s Disease: Potential for Pharmacological Intervention. CNS Drugs 2002, 16, 435–444. [Google Scholar] [CrossRef]
- Livadiotis, G.; Assas, L.; Dayeh, M.; Elaydi, S.; Phea, C.; Roberts, J.; Samman, Y.; Tchen, R. Experimental Analysis of Interacting HT22 Plasma Membrane cholesterol and beta Amyloid. Adv. Alzheimer’s Dis. 2017, 6, 75–96. [Google Scholar] [CrossRef] [Green Version]
- Pike, C.J.; Burdick, D.; Walencewicz, A.J.; Glabe, C.G.; Cotman, C.W. Neurodegeneration induced by β-amyloid peptides in vitro: The role of peptide assembly state. J. Neurosci. 1993, 13, 1676–1687. [Google Scholar] [CrossRef]
- Pryor, N.E.; Moss, M.A.; Hestekin, C.N. Unraveling the Early Events of Amyloid-β Protein (Aβ) Aggregation: Techniques for the Determination of Aβ Aggregate Size. Int. J. Mol. Sci. 2012, 13, 3038–3072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, F.S.; La Rosa, F.G.; Kumar, S.; Edwards-Prasad, J.; Kentrotti, S.; Vernadakis, A.; Freed, C.F.; Prasad, K.N. Characterization and Transplantation of Two Neuronal Cell Lines with Dopaminergic Properties. Neurochem. Res. 1996, 21, 619–627. [Google Scholar] [CrossRef]
- Cunningham, R.L.; Giuffrida, A.; Roberts, J.L. Androgens induce dopaminergic neurotoxicity via caspase-3-dependent activation of protein kinase Cdelta. Endocrinology 2009, 150, 5539–5548. [Google Scholar] [CrossRef] [Green Version]
- Kaul, S.; Kanthasamy, A.; Kitazawa, M.; Anantharam, V.; Kanthasamy, A.G. Caspase-3 dependent proteolytic activation of protein kinase Cδ mediates and regulates 1-methyl-4-phenylpyridinium (MPP+)-induced apoptotic cell death in dopaminergic cells: Relevance to oxidative stress in dopaminergic degeneration. Eur. J. Neurosci. 2003, 18, 1387–1401. [Google Scholar] [CrossRef]
- Kaul, S.; Anantharam, V.; Yang, Y.; Choi, C.J.; Kanthasamy, A.; Kanthasamy, A.G. Tyrosine phosphorylation regulates the proteolytic activation of protein kinase Cδ in dopaminergic neuronal cells. J. Biol. Chem. 2005, 280, 28721–28730. [Google Scholar] [CrossRef] [Green Version]
- Sun, F.; Anantharam, V.; Zhang, D.; Latchoumycandane, C.; Kanthasamy, A.; Kanthasamy, A.G. Proteasome inhibitor MG-132 induces dopaminergic degeneration in cell culture and animal models. Neurotoxicology 2006, 27, 807–815. [Google Scholar] [CrossRef]
- Lazic, S.E.; Clarke-Williams, C.J.; Munafò, M.R. What exactly is ‘N’in cell culture and animal experiments? PLoS Biol. 2018, 16, e2005282. [Google Scholar]
- Weyermann, J.; Lochmann, D.; Zimmer, A. A practical note on the use of cytotoxicity assays. Int. J. Pharm. 2005, 288, 369–376. [Google Scholar] [CrossRef]
- Lambert, R.J.W.; Pearson, J. Susceptibility testing: Accurate and reproducible minimum inhibitory concentration (MIC) and non-inhibiting concentration (NIC) values. J. Appl. Microbiol. 2000, 88, 784–790. [Google Scholar] [CrossRef]
- Mackey, B.M.; Derrick, C.M. The effect of sublethalinjury by heating, freezing, drying and gamma-radiation on theduration of the lag phase ofSalmonella typhimurium. J. Appl. Bacteriol. 1982, 53, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Livadiotis, G. Approach to general methods for fitting and their sensitivity. Physica A 2007, 375, 518–536. [Google Scholar] [CrossRef]
- Livadiotis, G. Superposition of polytropes in the inner heliosheath. Astrophys. J. Suppl. Ser. 2016, 223, 13. [Google Scholar] [CrossRef]
- Dayeh, M.A.; Zirnstein, E.J.; Desai, M.I.; Funsten, H.O.; Fuselier, S.A.; Heerikhuisen, J.; McComas, D.J.; Schwadron, N.A.; Szalay, J.R. Variability in the Position of the IBEX Ribbon over Nine Years: More Observational Evidence for a Secondary ENA Source. Astrophys. J. 2019, 87, 84. [Google Scholar] [CrossRef] [Green Version]
- Schwadron, N.A.; Möbius, E.; McComas, D.J.; Bochsler, P.; Bzowski, M.; Fuselier, S.A.; Livadiotis, G.; Frisch, P.; Müller, H.-R.; Heirtzler, D.; et al. Determination of interstellar O parameters using the first 2 years of data from IBEX. Astrophys. J. 2016, 828, 81. [Google Scholar] [CrossRef]
- Frisch, P.C.; Bzowski, M.; Livadiotis, G.; McComas, D.J.; Moebius, E.; Mueller, H.R.; Pryor, W.R.; Schwadron, N.A.; Sokół, J.M.; Vallerga, J.V.; et al. Decades-long changes of the interstellar wind through our solar system. Science 2013, 341, 1080. [Google Scholar] [CrossRef]
- Livadiotis, G. Chi-p distribution: Characterization of the goodness of the fitting using Lp norms. J. Stat. Distr. Appl. 2014, 1, 4. [Google Scholar] [CrossRef] [Green Version]
- Livadiotis, G.; McComas, D.J. Evidence of large scale phase space quantization in plasmas. Entropy 2013, 15, 1118–1132. [Google Scholar] [CrossRef] [Green Version]
- Fuselier, S.A.; Allegrini, F.; Bzowski, M. Low energy neutral atoms from the heliosheath. Astrophys. J. 2014, 784, 89. [Google Scholar] [CrossRef]
- Nicholson, A.M.; Methner, D.N.R.; Ferreira, A. Membrane Cholesterol Modulates β-Amyloid-Dependent Tau Cleavage by Inducing Changes in the Membrane Content and Localization of N-Methyl-d-Aspartic Acid Receptors. J. Biol. Chem. 2011, 286, 976–986. [Google Scholar] [CrossRef] [Green Version]
- Dayeh, M.A.; Livadiotis, G.; Elaydi, S. A discrete mathematical model for the aggregation of β-Amyloid. PLoS ONE 2018, 13, e0196402. [Google Scholar] [CrossRef]
- Elaydi, S. An Introduction to Difference Equations, 3rd ed.; Springer-Verlag: New York, NY, USA, 2005. [Google Scholar]
- Livadiotis, G.; Elaydi, S. General Allee effect in two-species population biology. J. Biol. Dyn. 2012, 6, 959–973. [Google Scholar] [CrossRef] [PubMed]







| MßCD [μM] | Cholesterol [%] |
|---|---|
| 0.0 | 100 |
| 0.5 | 79.97 |
| 1.0 | 69.25 |
| 1.5 | 57.02 |
| 2.0 | 52.50 |
| 2.5 | 43.66 |
| H2O2 [µM] | Exp. 1 | Exp. 2 | Exp. 3 | Exp. 1 | Exp. 2 | Exp. 3 | Exp. 1 | Exp. 2 | Exp. 3 |
| 0 | 473.02 | 328.33 | 362.08 | 461.07 | 431.04 | 410.68 | 182.08 | 177.97 | 121.52 |
| 10 | 368.59 | 361.37 | 332.60 | 541.67 | 416.46 | 408.28 | 134.25 | 113.8 | 135.96 |
| 15 | 11.62 * | 13.06 | 10.18 | 44.82 * | 81.23 | 8.41 | 8.40 * | 9.58 | 7.23 |
| 20 | 13.13 * | 12.98 | 13.28 | 13.60 | 23.54 | 21.59 | 4.25 | 14.39 | 4.127 |
| 25 | 9.92 * | 9.01 | 10.836 | 16.10 | 12.12 | 14.56 | 5.90 | 14.39 | 4.12 |
| 30 | 27.25 | 15.04 | 44.33 | 25.51 | 10.05 | 54.34 | 8.68 | 6.10 | 7.39 * |
| 40 | 11.23 * | 12.07 | 10.39 | 12.44 * | 14.31 | 10.58 | 4.64 * | 5.13 | 4.16 |
| 80 | 19.08 | 14.45 | 25.26 | 11.94 | 10.32 | 24.08 | 1.88 | 5.42 | 3.65 * |
| = 1.5 | = 2.0 | = 2.5 | |||||||
| H2O2 [µM] | Exp. 1 | Exp. 2 | Exp. 3 | Exp. 1 | Exp. 2 | Exp. 3 | Exp. 1 | Exp. 2 | Exp. 3 |
| 0 | 223.38 | 171.39 | 185.32 | 164.88 | 180.13 | 212.25 | 226.04 | 196.60 | 183.61 |
| 10 | 152.32 | 191.64 | 220.30 | 151.79 | 190.53 | 255.27 | 198.92 | 188.58 | 195.53 |
| 15 | 78.76 | 10.21 | 22.35 | 77.53 | 30.58 | 30.57 | 141.26 | 78.38 | 109.82 * |
| 20 | 13.89 | 14.50 | 37.59 | 16.64 | 13.35 | 41.04 | 9.07 | 5.90 | 7.82 * |
| 25 | 8.47 | 7.31 | 6.69 | 20.06 | 12.81 | 17.66 | 11.93 | 14.20 | 20.45 |
| 30 | 10.98 | 9.36 | 30.56 | 21.19 | 9.90 | 37.64 | 19.33 | 18.44 | 45.11 |
| 40 | 46.68 | 7.54 | 4.40 | 45.53 | 17.39 | 13.59 | 3.20 | 12.40 | 8.08 |
| 80 | 8.01 | 7.42 | 26.70 | 17.04 | 12.49 | 18.69 | 4.61 | 6.31 | 5.46 * |
| H2O2 [µM] | ||||||
|---|---|---|---|---|---|---|
| 0 | 387.81 ± 75.70 ** | 434.26 ± 25.35 | 160.52 ± 33.84 | 193.36 ± 26.91 | 185.76 ± 24.18 | 202.08 ± 21.74 |
| 10 | 354.19 ± 54.19 | 455.47 ± 74.77 | 128.00 ± 12.33 | 188.09 ± 34.13 | 199.20 ± 52.28 | 194.35 ± 5.28 |
| 15 | 11.62 ± 1.44 | 44.82 ± 36.41 | 8.41 ± 1.17 | 37.11 ± 36.58 | 46.23 ± 27.11 | 109.82 ± 31.44 |
| 20 | 13.13 ± 0.15 | 19.58 ± 5.27 | 7.59 ± 5.89 | 21.99 ± 13.51 | 23.68 ± 15.13 | 7.48 ± 1.58 |
| 25 | 9.92 ± 1.29 | 14.26 ± 2.01 | 8.14 ± 5.49 | 7.49 ± 0.91 | 16.84 ± 3.69 | 15.53 ± 4.41 |
| 30 | 28.87 ± 8.87 | 29.97 ± 22.48 | 7.39 ± 1.29 | 16.97 ± 11.80 | 22.91 ± 13.95 | 27.63 ± 15.15 |
| 40 | 11.23 ± 1.19 | 12.44 ± 2.64 | 4.64 ± 0.68 | 19.54 ± 23.56 | 25.50 ± 17.45 | 7.89 ± 4.60 |
| 80 | 19.60 ± 5.42 | 15.45 ± 7.52 | 3.65 ± 1.77 | 14.04 ± 10.96 | 16.07 ± 3.21 | 5.64 ± 0.85 |
| # | MßCD | α | σα | δα | β | σβ | δβ | γ | σγ | δγ | δ | σδ | δδ | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0 | 13.05 | 5.22 | 1.85 | 374.8 | 86.9 | 30.7 | 1.20 | 0.30 | 0.11 | 11.96 | 2.00 | 0.71 | 3.02 |
| 2 | 0.5 | 14.22 | 1.08 | 0.38 | 422.2 | 18.3 | 6.5 | 2.67 | 1.64 | 0.58 | 14.64 | 9.27 | 3.28 | 0.52 |
| 3 | 1 | 5.13 | 0.62 | 0.22 | 155.5 | 37.3 | 13.2 | 0.56 | 0.22 | 0.08 | 12.58 | 1.02 | 0.36 | 1.18 |
| 4 | 1.5 | 7.67 | 0.69 | 0.24 | 185.7 | 20.8 | 7.4 | 0.83 | 1.27 | 0.45 | 14.27 | 1.28 | 0.45 | 0.59 |
| 5 | 2 | 16.93 | 1.01 | 0.36 | 171.2 | 9.6 | 3.4 | 2.72 | 0.10 | 0.04 | 14.79 | 0.08 | 0.03 | 0.19 |
| 6 | 2.5 | 5.97 | 1.11 | 0.39 | 202.2 | 20.5 | 7.2 | 0.42 | 0.14 | 0.05 | 16.20 | 1.22 | 0.43 | 1.85 |
| Constant Model | Linear Model | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| # | Mean | Error | M.F. | r (%) | Intercept a | Error δa | Slope b | Error δb | b/δb | M.F. | r (%) |
| α | 9.2 | 2.8 | 5.8 | −18.6 | 10.8 | 3.8 | −1.29 | 2.8 | −0.46 | 6.5 | −32.7 |
| β | 266 | 70 | 8.2 | −90.8 | 401 | 57 | −109 | 33 | −3.34 | 3.8 | −85.6 |
| γ | 1.69 | 0.70 | 7.5 | −8.0 | 1.95 | 1.36 | −0.17 | 0.75 | −0.22 | 8.7 | −10.9 |
| δ | 14.78 | 0.53 | 1.3 | 95.6 | 11.14 | 0.48 | 1.87 | 0.27 | 7.04 | 0.36 | 96.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dayeh, M.A.; Livadiotis, G.; Aminian, F.; Cheng, K.H.; Roberts, J.L.; Viswasam, N.; Elaydi, S. Effects of Cholesterol in Stress-Related Neuronal Death—A Statistical Analysis Perspective. Int. J. Mol. Sci. 2020, 21, 2905. https://doi.org/10.3390/ijms21082905
Dayeh MA, Livadiotis G, Aminian F, Cheng KH, Roberts JL, Viswasam N, Elaydi S. Effects of Cholesterol in Stress-Related Neuronal Death—A Statistical Analysis Perspective. International Journal of Molecular Sciences. 2020; 21(8):2905. https://doi.org/10.3390/ijms21082905
Chicago/Turabian StyleDayeh, Maher A., George Livadiotis, Farzan Aminian, Kwan H. Cheng, James L. Roberts, Nikita Viswasam, and Saber Elaydi. 2020. "Effects of Cholesterol in Stress-Related Neuronal Death—A Statistical Analysis Perspective" International Journal of Molecular Sciences 21, no. 8: 2905. https://doi.org/10.3390/ijms21082905




