Quantitative Assessment of Arthritis Activity in Rheumatoid Arthritis Patients Using [11C]DPA-713 Positron Emission Tomography
Abstract
:1. Introduction
2. Results
2.1. Clinical Data
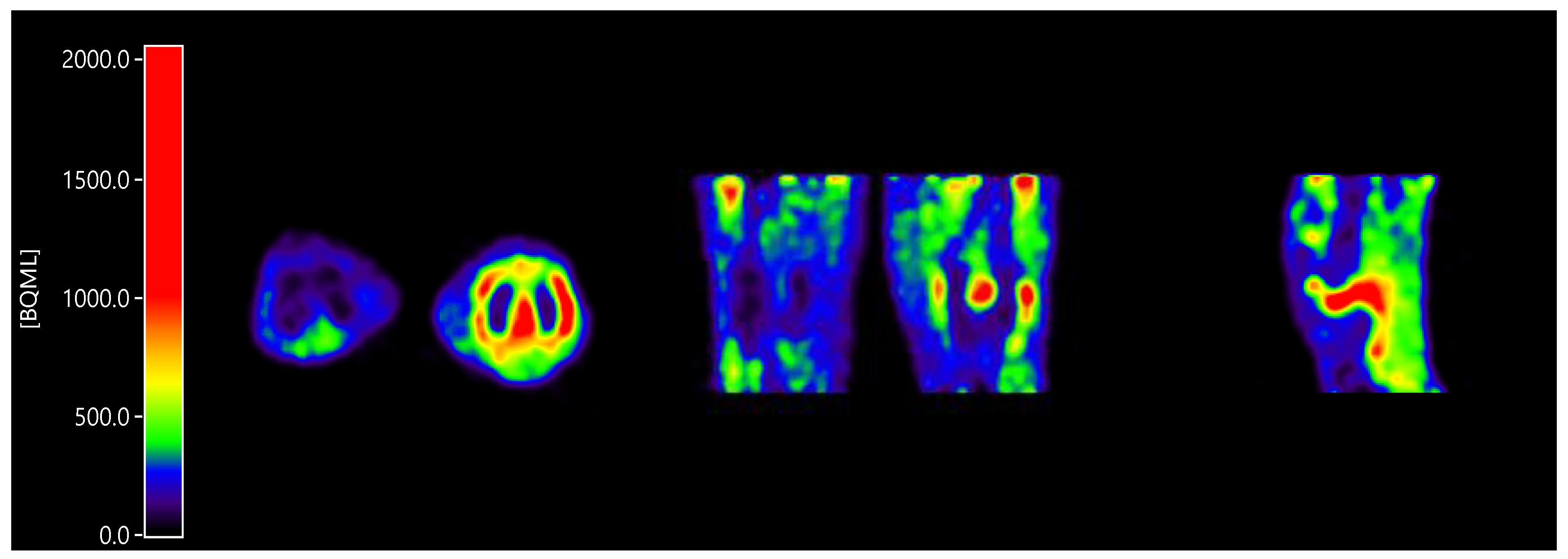
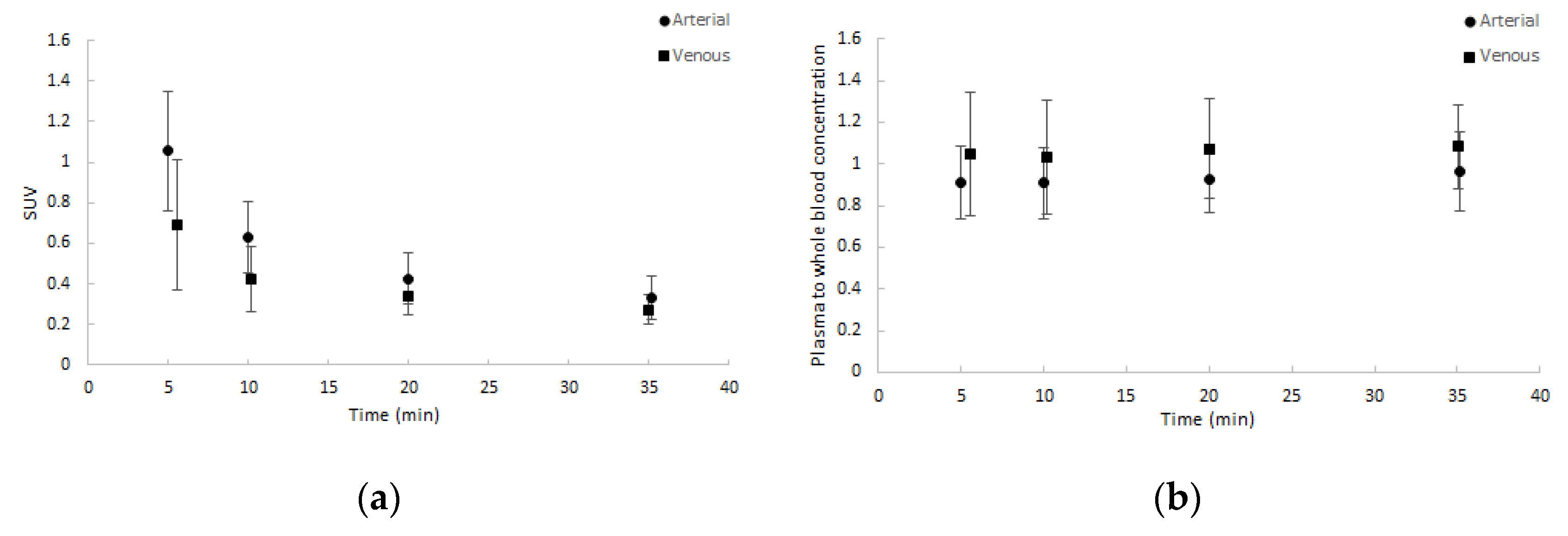
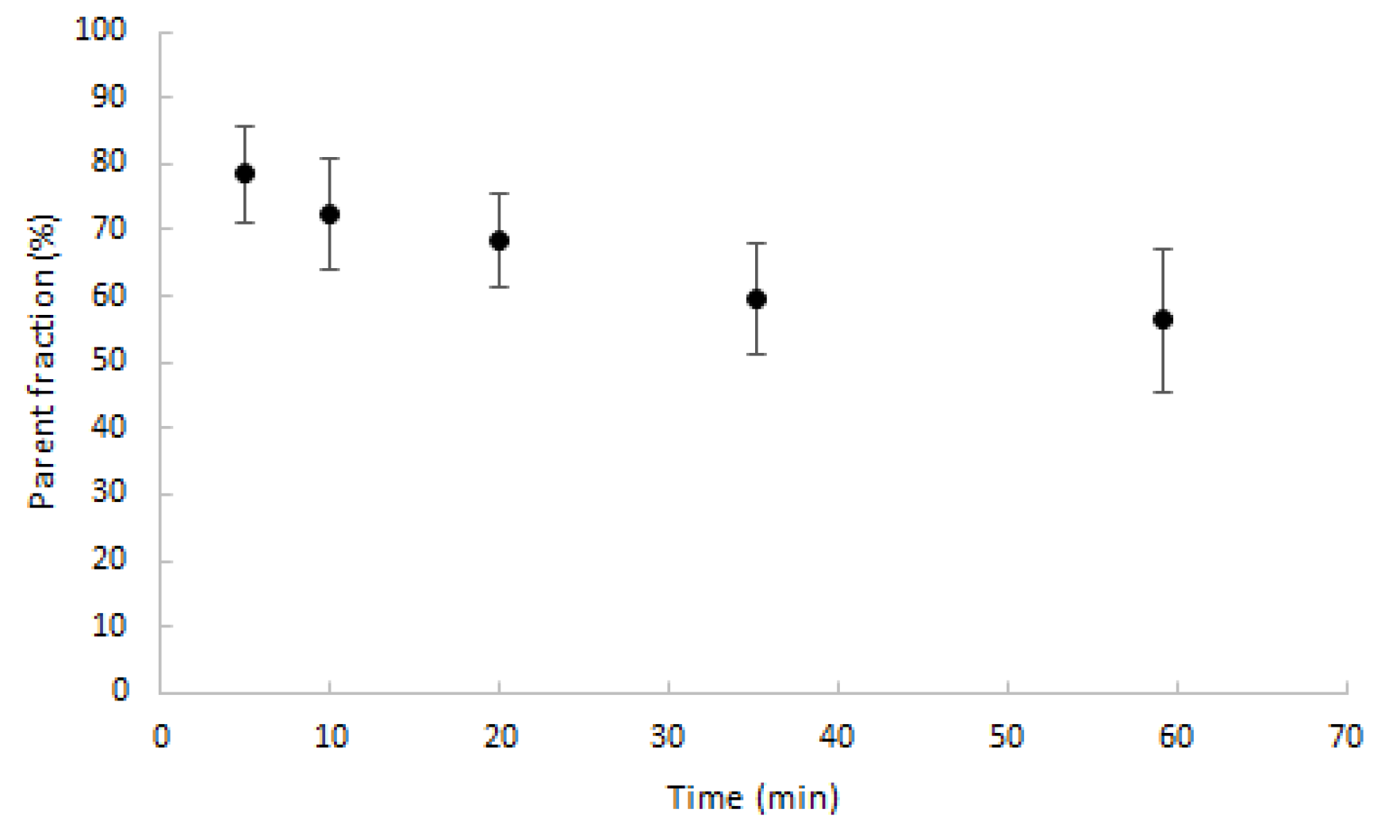
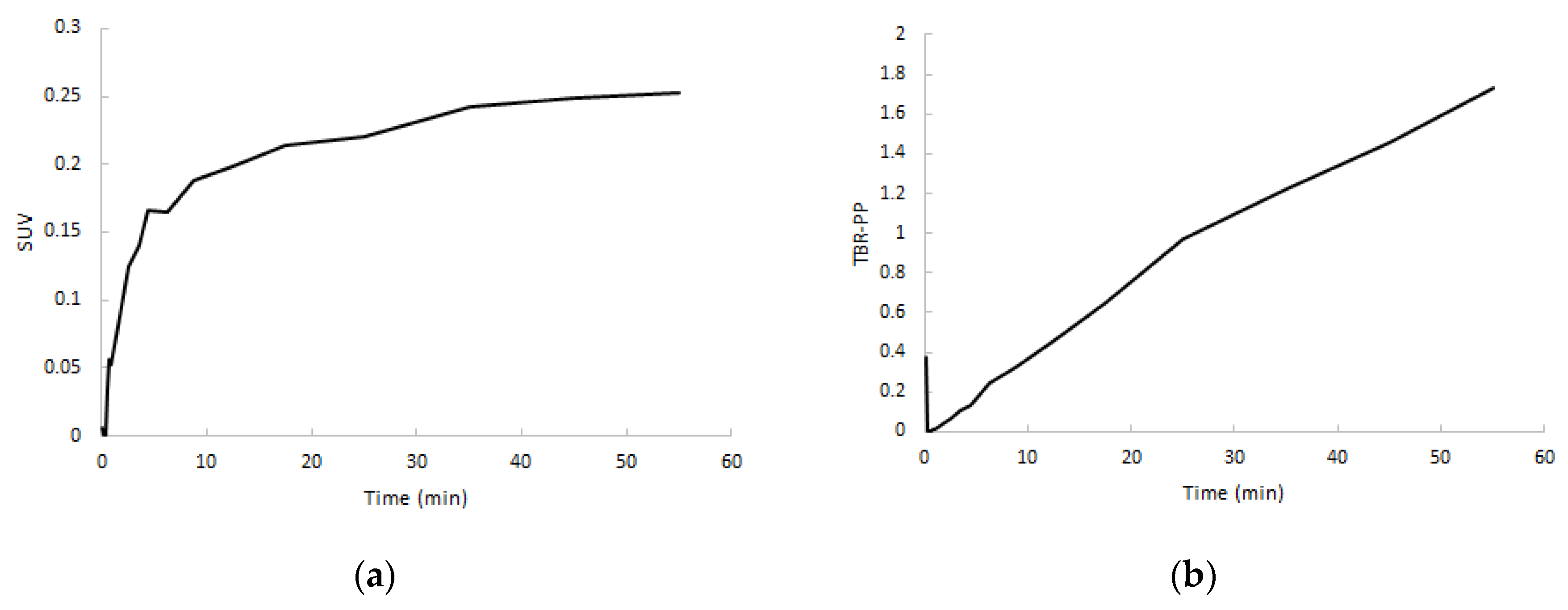
2.2. Input Functions for Dynamic PET Data Analyses
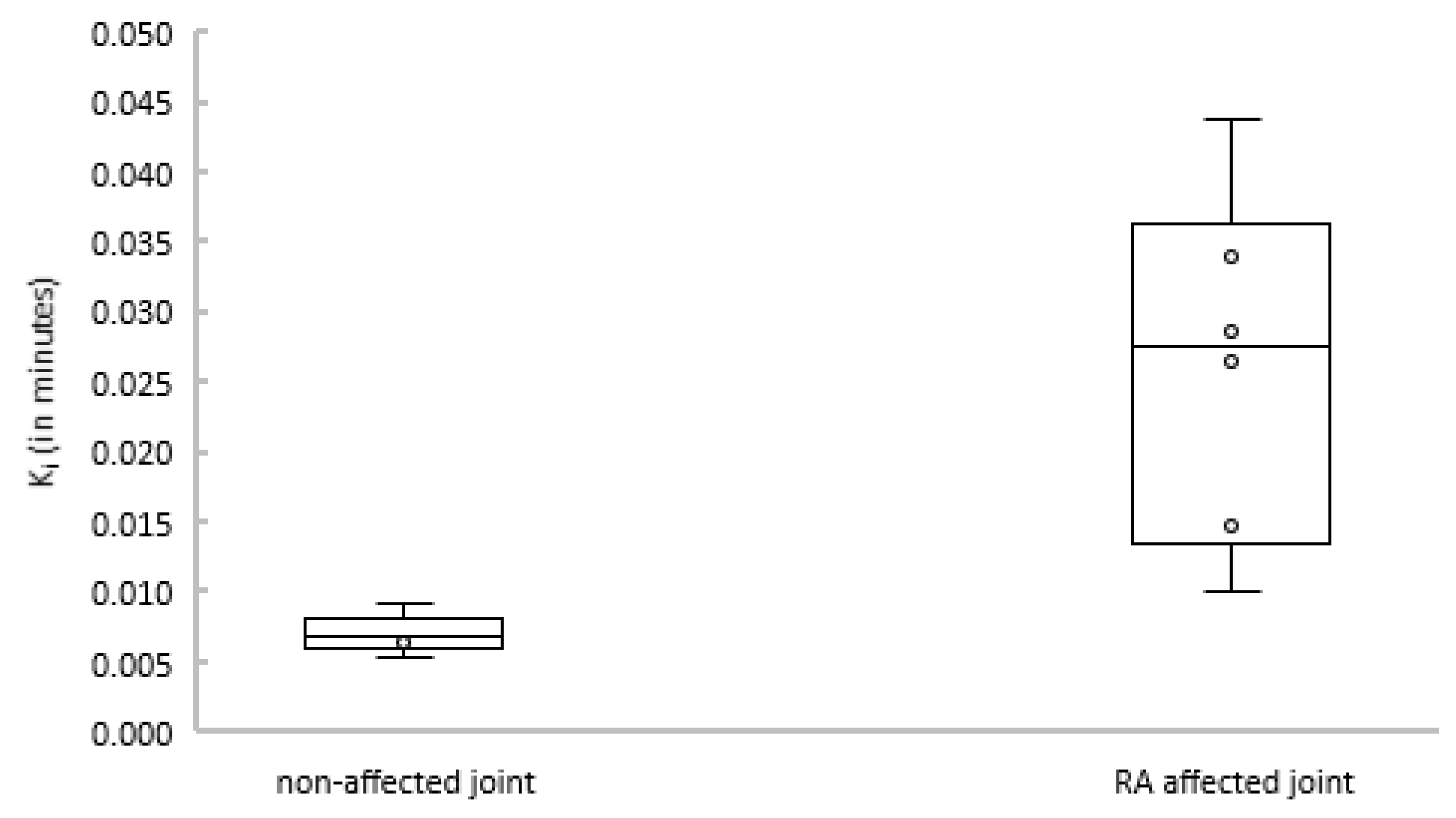
2.3. Kinetic Analyses Using Non-linear Plasma Input Models
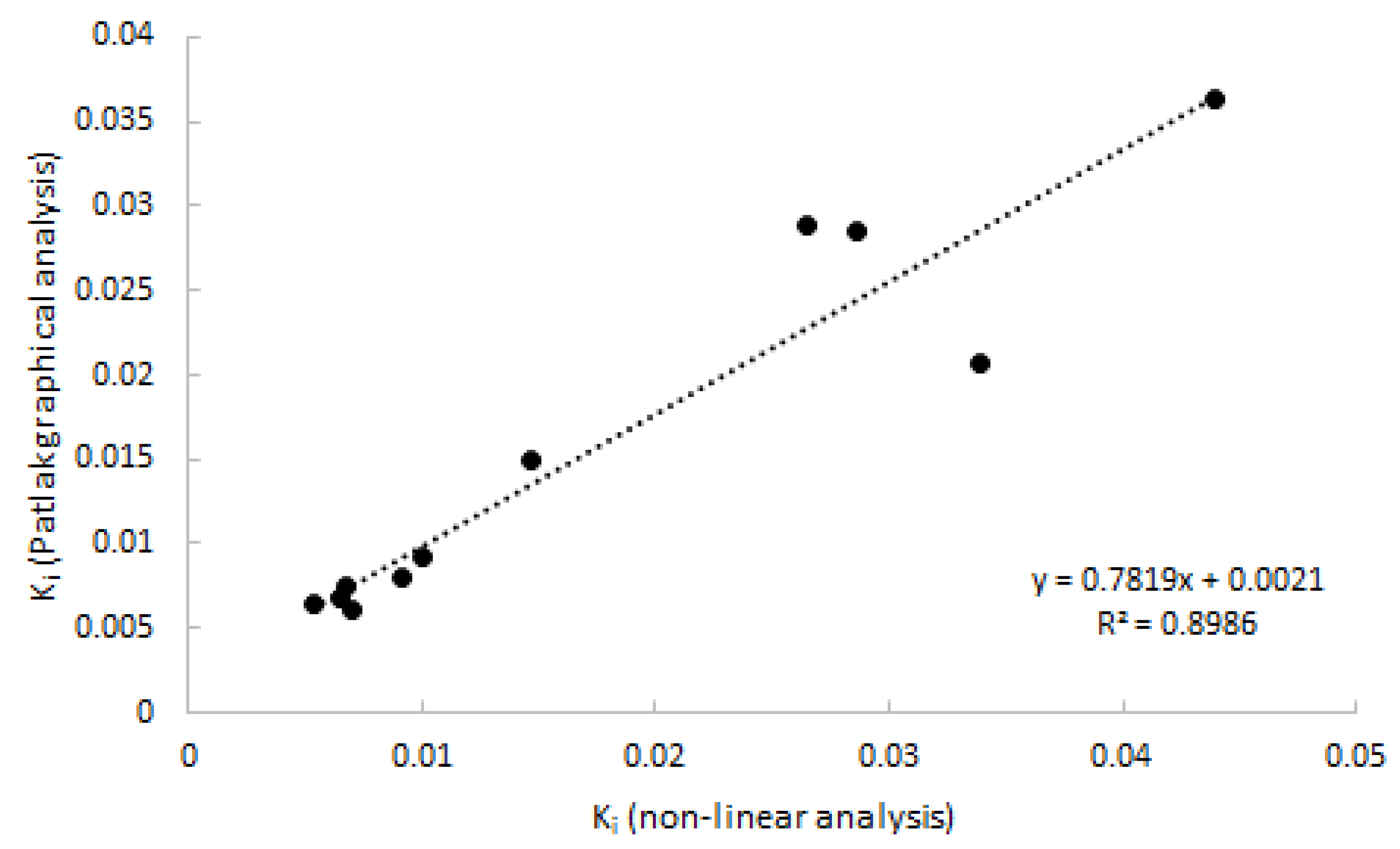
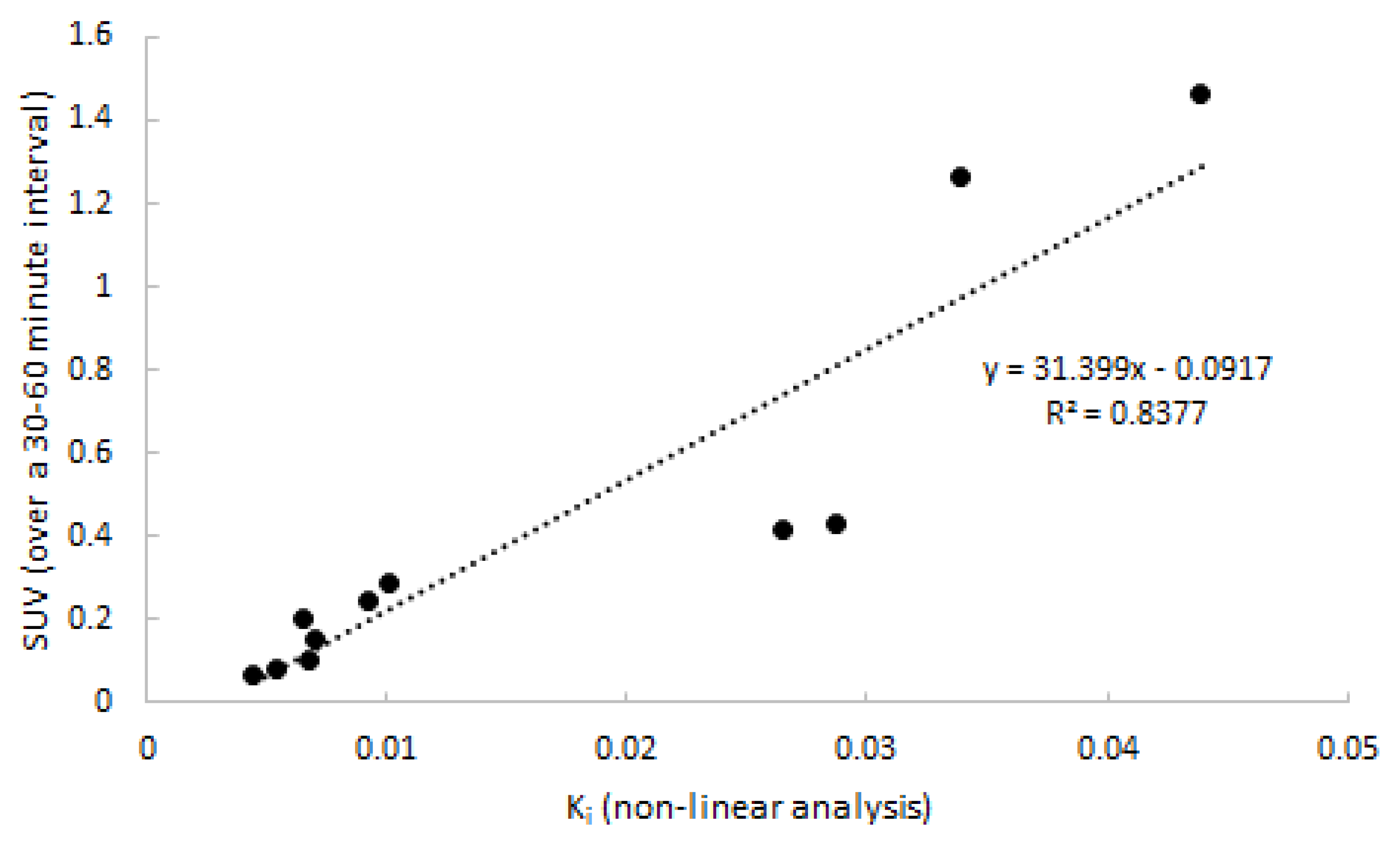
2.4. Linearized and Simplified Methods
2.5. SUV in Relation to Clinical Activity
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Clinical Assessment
4.3. Scanning Protocol
4.4. Image Segmentation
4.5. Kinetic Analysis Using Non-linear Plasma Input Models
4.6. Linearized and Simplified (Static) Analysis
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cross, M.; Smith, E.; Hoy, D.; Carmona, L.; Wolfe, F.; Vos, T.; Williams, B.; Gabriel, S.; Lassere, M.; Johns, N.; et al. The global burden of rheumatoid arthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038. [Google Scholar] [CrossRef]
- Smolen, J.S.; Landewe, R.; Bijlsma, J.; Burmester, G.; Chatzidionysiou, K.; Dougados, M.; Nam, J.; Ramiro, S.; Voshaar, M.; van Vollenhoven, R.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann. Rheum. Dis. 2017, 76, 960–977. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.; Townsend, D. History and future technical innovation in positron emission tomography. J. Med. Imaging 2017, 4, 011013. [Google Scholar] [CrossRef]
- Shukla, A.K.; Kumar, U. Positron emission tomography: An overview. J. Med. Phys. 2006, 31, 13–21. [Google Scholar] [CrossRef]
- Bruijnen, S.T.; Gent, Y.Y.; Voskuyl, A.E.; Hoekstra, O.S.; van der Laken, C.J. Present role of positron emission tomography in the diagnosis and monitoring of peripheral inflammatory arthritis: A systematic review. Arthritis Care Res. (Hoboken) 2014, 66, 120–130. [Google Scholar] [CrossRef] [Green Version]
- Udalova, I.A.; Mantovani, A.; Feldmann, M. Macrophage heterogeneity in the context of rheumatoid arthritis. Nat. Rev. Rheumatol. 2016, 12, 472–485. [Google Scholar] [CrossRef]
- Hamilton, J.A.; Tak, P.P. The dynamics of macrophage lineage populations in inflammatory and autoimmune diseases. Arthritis Rheum. 2009, 60, 1210–1221. [Google Scholar] [CrossRef]
- Kennedy, A.; Fearon, U.; Veale, D.J.; Godson, C. Macrophages in synovial inflammation. Front. Immunol. 2011, 2, 52. [Google Scholar] [CrossRef] [Green Version]
- Haringman, J.J.; Gerlag, D.M.; Zwinderman, A.H.; Smeets, T.J.; Kraan, M.C.; Baeten, D.; McInnes, I.B.; Bresnihan, B.; Tak, P.P. Synovial tissue macrophages: A sensitive biomarker for response to treatment in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2005, 64, 834–838. [Google Scholar] [CrossRef]
- Jahangier, Z.N.; Jacobs, J.W.; Kraan, M.C.; Wenting, M.J.; Smeets, T.J.; Bijlsma, J.W.; Lafeber, F.P.; Tak, P.P. Pretreatment macrophage infiltration of the synovium predicts the clinical effect of both radiation synovectomy and intra-articular glucocorticoids. Ann. Rheum. Dis. 2006, 65, 1286–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gent, Y.Y.; Weijers, K.; Molthoff, C.F.; Windhorst, A.D.; Huisman, M.C.; Kassiou, M.; Jansen, G.; Lammertsma, A.A.; van der Laken, C.J. Promising potential of new generation translocator protein tracers providing enhanced contrast of arthritis imaging by positron emission tomography in a rat model of arthritis. Arthritis Res. Ther. 2014, 16, R70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Laken, C.J.; Elzinga, E.H.; Kropholler, M.A.; Molthoff, C.F.; van der Heijden, J.W.; Maruyama, K.; Boellaard, R.; Dijkmans, B.A.; Lammertsma, A.A.; Voskuyl, A.E. Noninvasive imaging of macrophages in rheumatoid synovitis using 11C-(R)-PK11195 and positron emission tomography. Arthritis Rheum. 2008, 58, 3350–3355. [Google Scholar] [CrossRef] [PubMed]
- Narayan, N.; Owen, D.R.; Mandhair, H.; Smyth, E.; Carlucci, F.; Saleem, A.; Gunn, R.N.; Rabiner, E.A.; Wells, L.; Dakin, S.G.; et al. Translocator Protein as an Imaging Marker of Macrophage and Stromal Activation in Rheumatoid Arthritis Pannus. J. Nucl. Med. 2018, 59, 1125–1132. [Google Scholar] [CrossRef] [Green Version]
- Gent, Y.Y.; Voskuyl, A.E.; Kloet, R.W.; van Schaardenburg, D.; Hoekstra, O.S.; Dijkmans, B.A.; Lammertsma, A.A.; van der Laken, C.J. Macrophage positron emission tomography imaging as a biomarker for preclinical rheumatoid arthritis: Findings of a prospective pilot study. Arthritis Rheum. 2012, 64, 62–66. [Google Scholar] [CrossRef]
- Gent, Y.Y.; Ter Wee, M.M.; Voskuyl, A.E.; den Uyl, D.; Ahmadi, N.; Dowling, C.; van Kuijk, C.; Hoekstra, O.S.; Boers, M.; Lems, W.F.; et al. Subclinical synovitis detected by macrophage PET, but not MRI, is related to short-term flare of clinical disease activity in early RA patients: An exploratory study. Arthritis Res. Ther. 2015, 17, 266. [Google Scholar] [CrossRef] [Green Version]
- Yokokura, M.; Terada, T.; Bunai, T.; Nakaizumi, K.; Takebayashi, K.; Iwata, Y.; Yoshikawa, E.; Futatsubashi, M.; Suzuki, K.; Mori, N.; et al. Depiction of microglial activation in aging and dementia: Positron emission tomography with [(11)C]DPA713 versus [(11)C]( R)PK11195. J. Cereb. Blood Flow Metab. 2017, 37, 877–889. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, M.; Jiang, T.; Telu, S.; Zoghbi, S.S.; Gunn, R.N.; Rabiner, E.A.; Owen, D.R.; Guo, Q.; Pike, V.W.; Innis, R.B.; et al. (11)C-DPA-713 has much greater specific binding to translocator protein 18 kDa (TSPO) in human brain than (11)C-( R)-PK11195. J. Cereb. Blood Flow Metab. 2018, 38, 393–403. [Google Scholar] [CrossRef] [Green Version]
- Bruijnen, S.T.G.; Verweij, N.J.F.; Gent, Y.Y.J.; Huisman, M.C.; Windhorst, A.D.; Kassiou, M.; van de Ven, P.M.; Lammertsma, A.A.; Hoekstra, O.S.; Voskuyl, A.E.; et al. Imaging disease activity of rheumatoid arthritis by macrophage targeting using second generation translocator protein positron emission tomography tracers. PLoS ONE 2019, 14, e0222844. [Google Scholar] [CrossRef]
- Gent, Y.Y.; Ahmadi, N.; Voskuyl, A.E.; Hoetjes, N.; van Kuijk, C.; Britsemmer, K.; Turkstra, F.; Boers, M.; Hoekstra, O.S.; van der Laken, C.J. Detection of subclinical synovitis with macrophage targeting and positron emission tomography in patients with rheumatoid arthritis without clinical arthritis. J. Rheumatol. 2014, 41, 2145–2152. [Google Scholar] [CrossRef]
- Sewerin, P.; Schleich, C.; Brinks, R.; Muller-Lutz, A.; Fichter, F.; Eichner, M.; Schneider, M.; Ostendorf, B.; Vordenbaumen, S. Assessing Associations of Synovial Perfusion, Cartilage Quality, and Outcome in Rheumatoid Arthritis Using Dynamic Contrast-enhanced Magnetic Resonance Imaging. J. Rheumatol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kropholler, M.A.; Boellaard, R.; Elzinga, E.H.; van der Laken, C.J.; Maruyama, K.; Kloet, R.W.; Voskuyl, A.E.; Dijkmans, B.A.; Lammertsma, A.A. Quantification of (R)-[11C]PK11195 binding in rheumatoid arthritis. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 624–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 2010, 62, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Fransen, J.; van Riel, P.L. The Disease Activity Score and the EULAR response criteria. Clin. Exp. Rheumatol. 2005, 23, S93–S99. [Google Scholar] [CrossRef]
- Boellaard, R.; van Lingen, A.; van Balen, S.C.; Hoving, B.G.; Lammertsma, A.A. Characteristics of a new fully programmable blood sampling device for monitoring blood radioactivity during PET. Eur. J. Nucl. Med. 2001, 28, 81–89. [Google Scholar] [CrossRef]
- Greuter, H.N.; van Ophemert, P.L.; Luurtsema, G.; Franssen, E.J.; Boellaard, R.; Lammertsma, A.A. Validation of a multiwell gamma-counter for measuring high-pressure liquid chromatography metabolite profiles. J. Nucl. Med. Technol. 2004, 32, 28–32. [Google Scholar]
- Akaike, H. Fitting Autoregressive Models for Prediction. Ann. I Stat. Math. 1969, 21, 243. [Google Scholar] [CrossRef]
| Baseline Patient Demographics | |
|---|---|
| Male, number (%) | 1 (20) |
| Age, years (mean ± SD) | 58 ± 5 |
| Height, cm (mean ± SD) | 169 ± 15 |
| Weight, kg (mean ± SD) | 82 ± 16 |
| Disease duration, months (mean ± SD) | 314 ± 195 |
| Anti-CCP positivity, number (%) | 3 (60) |
| IgM RF positivity, number (%) | 3 (60) |
| DAS 44 (mean ± SD) | 2.78 ± 0.43 |
| 44-swollen joint count (median ± IQR) | 7 ± 5 |
| 44-tender joint count (median ± IQR) | 6 ± 7 |
| CRP, mg/mL (median ± IQR) | 3 ± 9 |
| ESR, mm/h (median ± IQR) | 9 ± 27 |
| VAS disease activity, 0 – 100 mm (mean ± SD) | 45 ± 19 |
| DMARD therapy, number (%) | 3 (60) |
| Oral prednisolone, number (%) | 4 (80) |
| NSAID therapy, number (%) | 2 (40) |
| Kinetic Parameter | Lower Bound | Upper Bound |
|---|---|---|
| K1 | 0.005 | 0.2 |
| k2 | 0.01 | 0.5 |
| k3 | 0.01 | 1.0 |
| VT | 0.01 | 50 |
| Vb | 0.001 | 1.0 |
| Volume of Interest | 1T2k | 2T4k | 2T3k |
|---|---|---|---|
| Bone marrow | 0 | 0 | 100% |
| Non-affected joint | 0 | 0 | 100% |
| Affected joint | 0 | 28.6% | 71.4% |
| Patient | Clinical Activity | Type of Joint | Arthritic Joint | Non-Affected, Contralateral Joint |
|---|---|---|---|---|
| 1 | + | Knee | 3.6 | 2.4 |
| 2 | ++ | MCP2 | 10.2 | 4.0 |
| 3 | +++ | Knee | 16.5 | 2.4 |
| 4 | +++ | Knee | 12.4 | 8.1 |
| 5 | +++ | MTP1 | 12.7 | 2.0 |
| Average | 11.1 | 3.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaqub, M.; Verweij, N.J.F.; Pieplenbosch, S.; Boellaard, R.; Lammertsma, A.A.; van der Laken, C.J. Quantitative Assessment of Arthritis Activity in Rheumatoid Arthritis Patients Using [11C]DPA-713 Positron Emission Tomography. Int. J. Mol. Sci. 2020, 21, 3137. https://doi.org/10.3390/ijms21093137
Yaqub M, Verweij NJF, Pieplenbosch S, Boellaard R, Lammertsma AA, van der Laken CJ. Quantitative Assessment of Arthritis Activity in Rheumatoid Arthritis Patients Using [11C]DPA-713 Positron Emission Tomography. International Journal of Molecular Sciences. 2020; 21(9):3137. https://doi.org/10.3390/ijms21093137
Chicago/Turabian StyleYaqub, Maqsood, Nicki J.F. Verweij, Simone Pieplenbosch, Ronald Boellaard, Adriaan A. Lammertsma, and Conny J. van der Laken. 2020. "Quantitative Assessment of Arthritis Activity in Rheumatoid Arthritis Patients Using [11C]DPA-713 Positron Emission Tomography" International Journal of Molecular Sciences 21, no. 9: 3137. https://doi.org/10.3390/ijms21093137





