The Expression and Function of CD300 Molecules in the Main Players of Allergic Responses: Mast Cells, Basophils and Eosinophils
Abstract
:1. Introduction
2. IgE-Mediated Allergic Responses
3. CD300 Receptor Family
4. CD300 Receptor Family in Mast Cells, Basophils and Eosinophils
4.1. CD300a
4.2. CD300f
4.3. Other CD300 Molecules
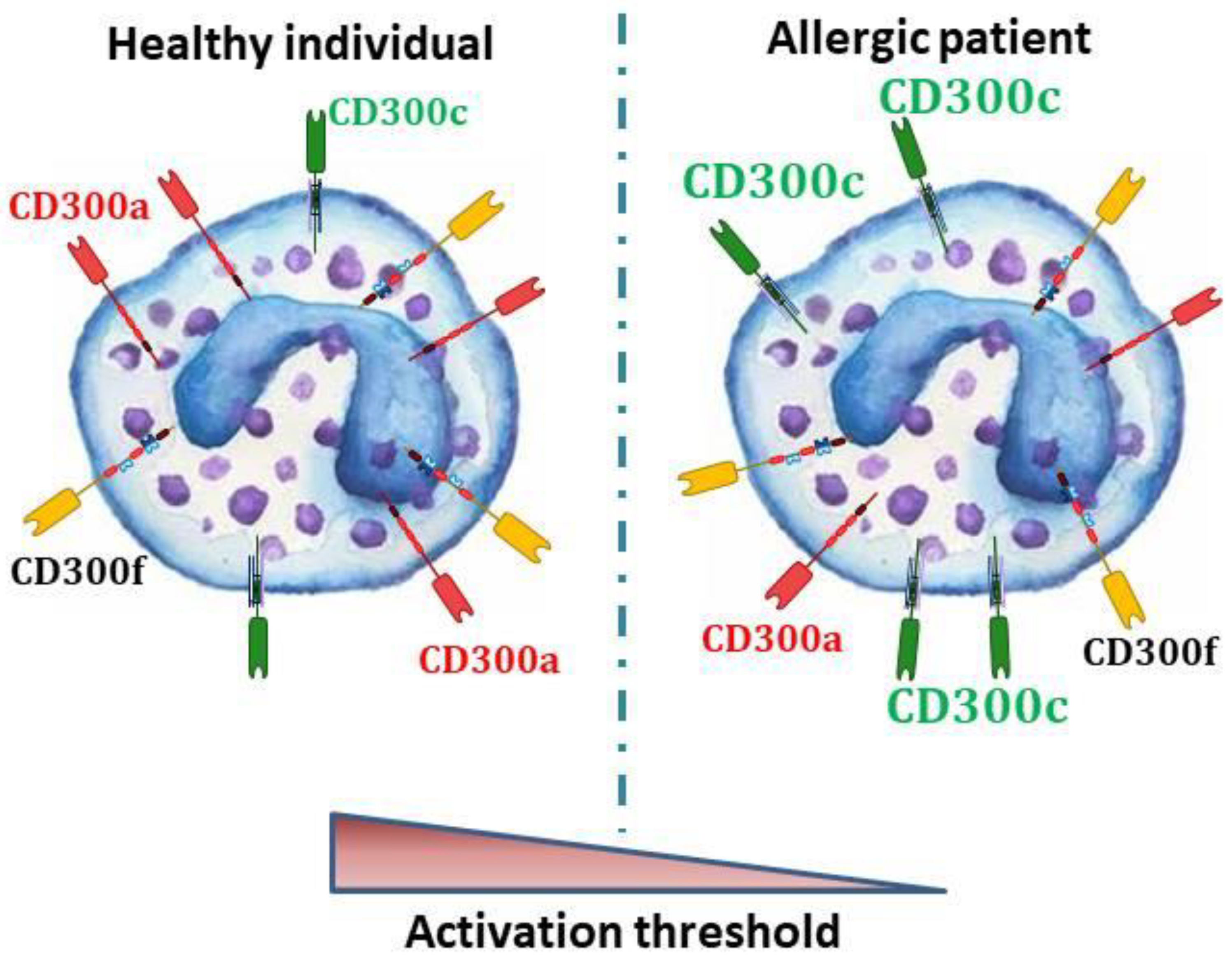
5. CD300 Molecules in Allergic Individuals
6. CD300a as Potential Therapeutic Target in Allergic Diseases
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AAAAI | American Academy of Allergy, Asthma and Immunology |
| Ab | antibody |
| AD | atopic dermatitis |
| BMMCs | bone marrow-derived mast cells |
| CBMCs | cord blood-derived mast cells |
| DAP | DNAX-activating protein |
| EDN | eosinophil-derived neurotoxin |
| ERK | extracellular signal-regulated kinase |
| FcR | Fc receptor |
| GM-CSF | granulocyte macrophage-colony stimulating factor |
| Grb2 | growth factor receptor-bound protein 2 |
| HIF | hypoxia inducible factor |
| Ig | immunoglobulin |
| IL | interleukin |
| ITAM | immunoreceptor tyrosine-based activating motif |
| ITIM | immunoreceptor tyrosine-based inhibitory motif |
| LPS | lipopolysaccharide |
| LT | leukotrienes |
| mAb | monoclonal antibody |
| MBP | major basic protein |
| NGF | nerve growth factor |
| OVA | ovalbumin |
| PBdMC | peripheral blood-derived mast cells |
| PCA | passive cutaneous anaphylaxis |
| PE | phosphatidylethanolamine |
| PI3K | phosphatidylinositol 3-kinase |
| PMA | phorbol 12-myristate 13-acetate |
| PS | phosphatidylserine |
| SCF | stem cell factor |
| TGF-β1 | transforming growth factor beta-1 |
| Th2 | T helper type 2 |
| TNF | tumor necrosis factor |
| TSLP | thymic stromal lymphopoietin |
| VEGFA | vascular endothelial growth factor |
| WT | wild type |
References
- Prescott, S.; Allen, K.J. Food allergy: Riding the second wave of the allergy epidemic. Pediatr. Allergy Immunol. 2011, 22, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.E.; Mehr, S. Fifty years of allergy: 1965–2015. J. Paediatr. Child Health 2015, 51, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Holgate, S.T. The epidemic of allergy and asthma. Nature 1999, 402, 2–4. [Google Scholar] [CrossRef]
- Kallinich, T.; Beier, K.C.; Wahn, U.; Stock, P.; Hamelmann, E. T-cell co-stimulatory molecules: Their role in allergic immune reactions. Eur. Respir. J. 2007, 29, 1246–1255. [Google Scholar] [CrossRef] [Green Version]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The development of allergic inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minai-Fleminger, Y.; Levi-Schaffer, F. Mast cells and eosinophils: The two key effector cells in allergic inflammation. Inflamm. Res. 2009, 58, 631–638. [Google Scholar] [CrossRef]
- Stone, K.D.; Prussin, C.; Metcalfe, D.D. IgE, mast cells, basophils, and eosinophils. J. Allergy Clin. Immunol. 2010, 125, S73–S80. [Google Scholar] [CrossRef]
- Bulfone-Paus, S.; Nilsson, G.; Draber, P.; Blank, U.; Levi-Schaffer, F. Positive and Negative Signals in Mast Cell Activation. Trends Immunol. 2017, 38, 657–667. [Google Scholar] [CrossRef] [Green Version]
- Levi-Schaffer, F.; Mandelboim, O. Inhibitory and Coactivating Receptors Recognising the Same Ligand: Immune Homeostasis Exploited by Pathogens and Tumours. Trends Immunol. 2018, 39, 112–122. [Google Scholar] [CrossRef]
- Odorizzi, P.M.; Wherry, E.J. Inhibitory Receptors on Lymphocytes: Insights from Infections. J. Immunol. 2012, 188, 2957–2965. [Google Scholar] [CrossRef] [Green Version]
- Geha, R.S.; Jabara, H.H.; Brodeur, S.R. The regulation of immunoglobulin E class-switch recombination. Nat. Rev. Immunol. 2003, 3, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Rigoni, A.; Colombo, M.P.; Pucillo, C. Mast cells, basophils and eosinophils: From allergy to cancer. Semin. Immunol. 2018, 35, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J. Mast cells and basophils. Curr. Opin. Hematol. 2000, 7, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.T.; MacGlashan, D.W.; Lichtenstein, L.M. Human basophils: Mediator release and cytokine production. Adv. Immunol. 2001, 77, 93–122. [Google Scholar] [PubMed]
- Galli, S.J.; Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 2012, 18, 693–704. [Google Scholar] [CrossRef] [Green Version]
- Korošec, P.; Gibbs, B.F.; Rijavec, M.; Custovic, A.; Turner, P.J. Important and specific role for basophils in acute allergic reactions. Clin. Exp. Allergy 2018, 48, 502–512. [Google Scholar] [CrossRef] [Green Version]
- Minai-Fleminger, Y.; Elishmereni, M.; Vita, F.; Rosa Soranzo, M.; Mankuta, D.; Zabucchi, G.; Levi-Schaffer, F. Ultrastructural evidence for human mast cell-eosinophil interactions in vitro. Cell Tissue Res. 2010, 341, 405–415. [Google Scholar] [CrossRef]
- Elishmereni, M.; Alenius, H.T.; Bradding, P.; Mizrahi, S.; Shikotra, A.; Minai-Fleminger, Y.; Mankuta, D.; Eliashar, R.; Zabucchi, G.; Levi-Schaffer, F. Physical interactions between mast cells and eosinophils: A novel mechanism enhancing eosinophil survival in vitro. Allergy 2011, 66, 376–385. [Google Scholar] [CrossRef]
- Nissim Ben Efraim, A.H.; Levi-Schaffer, F. Roles of Eosinophils in the Modulation of Angiogenesis. Chem. Immunol. Allergy 2013, 99, 138–154. [Google Scholar] [PubMed]
- Gasiorowski, R.E.; Ju, X.; Hart, D.N.J.; Clark, G.J. CD300 molecule regulation of human dendritic cell functions. Immunol. Lett. 2013, 149, 93–100. [Google Scholar] [CrossRef]
- Borrego, F. The CD300 molecules: An emerging family of regulators of the immune system. Blood 2013, 121, 1951–1960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niizuma, K.; Tahara-Hanaoka, S.; Noguchi, E.; Shibuya, A. Identification and Characterization of CD300H, a New Member of the Human CD300 Immunoreceptor Family. J. Biol. Chem. 2015, 290, 22298–22308. [Google Scholar] [CrossRef] [Green Version]
- Zenarruzabeitia, O.; Vitallé, J.; Eguizabal, C.; Simhadri, V.R.; Borrego, F. The Biology and Disease Relevance of CD300a, an Inhibitory Receptor for Phosphatidylserine and Phosphatidylethanolamine. J. Immunol. 2015, 194, 5053–5060. [Google Scholar] [CrossRef] [Green Version]
- Zenarruzabeitia, O.; Vitallé, J.; García-Obregón, S.; Astigarraga, I.; Eguizabal, C.; Santos, S.; Simhadri, V.R.; Borrego, F. The expression and function of human CD300 receptors on blood circulating mononuclear cells are distinct in neonates and adults. Sci. Rep. 2016, 6, 32693. [Google Scholar] [CrossRef] [Green Version]
- Vitallé, J.; Terrén, I.; Orrantia, A.; Zenarruzabeitia, O.; Borrego, F. CD300 receptor family in viral infections. Eur. J. Immunol. 2019, 49, 364–374. [Google Scholar] [CrossRef] [Green Version]
- Clark, G.J.; Ju, X.; Tate, C.; Hart, D.N.J. The CD300 family of molecules are evolutionarily significant regulators of leukocyte functions. Trends Immunol. 2009, 30, 209–217. [Google Scholar] [CrossRef]
- Rozenberg, P.; Reichman, H.; Zab-Bar, I.; Itan, M.; Pasmanik-Chor, M.; Bouffi, C.; Qimron, U.; Bachelet, I.; Fulkerson, P.C.; Rothenberg, M.E.; et al. CD300f:IL-5 cross-talk inhibits adipose tissue eosinophil homing and subsequent IL-4 production. Sci. Rep. 2017, 7, 5922. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, M.B.; Lanier, L.L.; Nakamura, M.C. Role of ITAM-containing adapter proteins and their receptors in the immune system and bone. Immunol. Rev. 2005, 208, 50–65. [Google Scholar] [CrossRef] [PubMed]
- Clark, G.J.; Ju, X.; Azlan, M.; Tate, C.; Ding, Y.; Hart, D.N.J. The CD300 molecules regulate monocyte and dendritic cell functions. Immunobiology 2009, 214, 730–736. [Google Scholar] [CrossRef] [PubMed]
- Izawa, K.; Kitaura, J.; Yamanishi, Y.; Matsuoka, T.; Kaitani, A.; Sugiuchi, M.; Takahashi, M.; Maehara, A.; Enomoto, Y.; Oki, T.; et al. An Activating and Inhibitory Signal from an Inhibitory Receptor LMIR3/CLM-1: LMIR3 Augments Lipopolysaccharide Response through Association with FcRγ in Mast Cells. J. Immunol. 2009, 183, 925–936. [Google Scholar] [CrossRef] [Green Version]
- DeBell, K.E.; Simhadri, V.R.; Mariano, J.L.; Borrego, F. Functional requirements for inhibitory signal transmission by the immunomodulatory receptor CD300a. BMC Immunol. 2012, 13, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakahashi-Oda, C.; Tahara-Hanaoka, S.; Honda, S.; Shibuya, K.; Shibuya, A. Identification of phosphatidylserine as a ligand for the CD300a immunoreceptor. Biochem. Biophys. Res. Commun. 2012, 417, 646–650. [Google Scholar] [CrossRef]
- Alvarez-Errico, D.; Aguilar, H.; Kitzig, F.; Brckalo, T.; Sayós, J.; López-Botet, M. IREM-1 is a novel inhibitory receptor expressed by myeloid cells. Eur. J. Immunol. 2004, 34, 3690–3701. [Google Scholar] [CrossRef]
- Álvarez-Errico, D.; Sayós, J.; López-Botet, M. The IREM-1 (CD300f) Inhibitory Receptor Associates with the p85α Subunit of Phosphoinositide 3-Kinase. J. Immunol. 2007, 178, 808–816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takatsu, H.; Hase, K.; Ohmae, M.; Ohshima, S.; Hashimoto, K.; Taniura, N.; Yamamoto, A.; Ohno, H. CD300 antigen like family member G: A novel Ig receptor like protein exclusively expressed on capillary endothelium. Biochem. Biophys. Res. Commun. 2006, 348, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Chung, D.-H.; Humphrey, M.B.; Nakamura, M.C.; Ginzinger, D.G.; Seaman, W.E.; Daws, M.R. CMRF-35-Like Molecule-1, a Novel Mouse Myeloid Receptor, Can Inhibit Osteoclast Formation. J. Immunol. 2003, 171, 6541–6548. [Google Scholar] [CrossRef] [Green Version]
- Tian, L.; Choi, S.-C.; Murakami, Y.; Allen, J.; Morse III, H.C.; Qi, C.-F.; Krzewski, K.; Coligan, J.E. p85α recruitment by the CD300f phosphatidylserine receptor mediates apoptotic cell clearance required for autoimmunity suppression. Nat. Commun. 2014, 5, 3146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ejarque-Ortiz, A.; Solà, C.; Martínez-Barriocanal, Á.; Schwartz, S.; Martín, M.; Peluffo, H.; Sayós, J. The Receptor CMRF35-Like Molecule-1 (CLM-1) Enhances the Production of LPS-Induced Pro-Inflammatory Mediators during Microglial Activation. PLoS ONE 2015, 10, e0123928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, S.-C.; Simhadri, V.R.; Tian, L.; Gil-Krzewska, A.; Krzewski, K.; Borrego, F.; Coligan, J.E. Cutting Edge: Mouse CD300f (CMRF-35–Like Molecule-1) Recognizes Outer Membrane-Exposed Phosphatidylserine and Can Promote Phagocytosis. J. Immunol. 2011, 187, 3483–3487. [Google Scholar] [CrossRef] [PubMed]
- Nakahashi-Oda, C.; Tahara-Hanaoka, S.; Shoji, M.; Okoshi, Y.; Nakano-Yokomizo, T.; Ohkohchi, N.; Yasui, T.; Kikutani, H.; Honda, S.; Shibuya, K.; et al. Apoptotic cells suppress mast cell inflammatory responses via the CD300a immunoreceptor. J. Exp. Med. 2012, 209, 1493–1503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simhadri, V.R.; Andersen, J.F.; Calvo, E.; Choi, S.C.; Coligan, J.E.; Borrego, F. Human CD300a binds to phosphatidylethanolamine and phosphatidylserine, and modulates the phagocytosis of dead cells. Blood 2012, 119, 2799–2809. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Izawa, K.; Kashiwakura, J.; Yamanishi, Y.; Enomoto, Y.; Kaitani, A.; Maehara, A.; Isobe, M.; Ito, S.; Matsukawa, T.; et al. Human CD300C Delivers an Fc Receptor-γ-dependent Activating Signal in Mast Cells and Monocytes and Differs from CD300A in Ligand Recognition. J. Biol. Chem. 2013, 288, 7662–7675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murakami, Y.; Tian, L.; Voss, O.H.; Margulies, D.H.; Krzewski, K.; Coligan, J.E. CD300b regulates the phagocytosis of apoptotic cells via phosphatidylserine recognition. Cell Death Differ. 2014, 21, 1746–1757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimitrova, M.; Zenarruzabeitia, O.; Borrego, F.; Simhadri, V.R. CD300c is uniquely expressed on CD56bright Natural Killer Cells and differs from CD300a upon ligand recognition. Sci. Rep. 2016, 6, 23942. [Google Scholar] [CrossRef] [Green Version]
- Birge, R.B.; Boeltz, S.; Kumar, S.; Carlson, J.; Wanderley, J.; Calianese, D.; Barcinski, M.; Brekken, R.A.; Huang, X.; Hutchins, J.T.; et al. Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer. Cell Death Differ. 2016, 23, 962–978. [Google Scholar] [CrossRef] [Green Version]
- Bevers, E.M.; Williamson, P.L. Getting to the Outer Leaflet: Physiology of Phosphatidylserine Exposure at the Plasma Membrane. Physiol. Rev. 2016, 96, 605–645. [Google Scholar] [CrossRef]
- Voss, O.H.; Murakami, Y.; Pena, M.Y.; Lee, H.-N.; Tian, L.; Margulies, D.H.; Street, J.M.; Yuen, P.S.T.; Qi, C.-F.; Krzewski, K.; et al. Lipopolysaccharide-Induced CD300b Receptor Binding to Toll-like Receptor 4 Alters Signaling to Drive Cytokine Responses that Enhance Septic Shock. Immunity 2016, 44, 1365–1378. [Google Scholar] [CrossRef] [Green Version]
- Izawa, K.; Yamanishi, Y.; Maehara, A.; Takahashi, M.; Isobe, M.; Ito, S.; Kaitani, A.; Matsukawa, T.; Matsuoka, T.; Nakahara, F.; et al. The Receptor LMIR3 Negatively Regulates Mast Cell Activation and Allergic Responses by Binding to Extracellular Ceramide. Immunity 2012, 37, 827–839. [Google Scholar] [CrossRef] [Green Version]
- Izawa, K.; Isobe, M.; Matsukawa, T.; Ito, S.; Maehara, A.; Takahashi, M.; Yamanishi, Y.; Kaitani, A.; Oki, T.; Okumura, K.; et al. Sphingomyelin and ceramide are physiological ligands for human LMIR3/CD300f, inhibiting FcεRI-mediated mast cell activation. J. Allergy Clin. Immunol. 2014, 133, 270–273.e7. [Google Scholar] [CrossRef]
- Izawa, K.; Kaitani, A.; Ando, T.; Maehara, A.; Nagamine, M.; Yamada, H.; Ando, T.; Ide, T.; Matsuzawa, M.; Okamoto, Y.; et al. Differential Lipid Recognition by Mouse versus Human CD300f, Inhibiting Passive Cutaneous Anaphylaxis, Depends on a Single Amino Acid Substitution in its Immunoglobulin-Like Domain. J. Investig. Dermatol. 2020, 140, 710–713.e3. [Google Scholar] [CrossRef]
- Isobe, M.; Izawa, K.; Sugiuchi, M.; Sakanishi, T.; Kaitani, A.; Takamori, A.; Maehara, A.; Matsukawa, T.; Takahashi, M.; Yamanishi, Y.; et al. The CD300e molecule in mice is an immune-activating receptor. J. Biol. Chem. 2018, 293, 3793–3805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izawa, K.; Maehara, A.; Isobe, M.; Yasuda, Y.; Urai, M.; Hoshino, Y.; Ueno, K.; Matsukawa, T.; Takahashi, M.; Kaitani, A.; et al. Disrupting ceramide-CD300f interaction prevents septic peritonitis by stimulating neutrophil recruitment. Sci. Rep. 2017, 7, 4298. [Google Scholar] [CrossRef] [PubMed]
- Vitallé, J.; Terrén, I.; Orrantia, A.; Segurola, A.; Seras, Y.; Gamboa, P.M.; Borrego, F.; Zenarruzabeitia, O. Increased expression levels of CD300c on basophils from allergic individuals. World Allergy Organ. J. 2019, 12, 100060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karra, L.; Gangwar, R.S.; Puzzovio, P.G.; Fyhrquist, N.; Minai-Fleminger, Y.; Landolina, N.; Simon, H.-U.; Alenius, H.; Leibovici, V.; Simon, D.; et al. CD300a expression is modulated in atopic dermatitis and could influence the inflammatory response. Allergy 2019, 74, 1377–1380. [Google Scholar] [CrossRef] [PubMed]
- Speckman, R.; Wright Daw, J.; Helms, C.; Duan, S.; Cao, L.; Taillon-Miller, P.; Kwok, P.-Y.; Menter, A.; Bowcock, A. Novel immunoglobulin superfamily gene cluster, mapping to a region of human chromosome 17q25, linked to psoriasis susceptibility. Hum. Genet. 2003, 112, 34–41. [Google Scholar] [CrossRef]
- Silva, R.; Moir, S.; Kardava, L.; Debell, K.; Simhadri, V.R.; Ferrando-Martínez, S.; Leal, M.; Peña, J.; Coligan, J.E.; Borrego, F. CD300a is expressed on human B cells, modulates BCR-mediated signaling, and its expression is down-regulated in HIV infection. Blood 2011, 117, 5870–5880. [Google Scholar] [CrossRef]
- Sabato, V.; Verweij, M.M.; Bridts, C.H.; Levi-Schaffer, F.; Gibbs, B.F.; De Clerck, L.S.; Schiavino, D.; Ebo, D.G. CD300a is expressed on human basophils and seems to inhibit IgE/FcεRI-dependent anaphylactic degranulation. Cytom. Part B Clin. Cytom. 2012, 82B, 132–138. [Google Scholar] [CrossRef]
- Zenarruzabeitia, O.; Vitallé, J.; Terrén, I.; Orrantia, A.; Astigarraga, I.; Dopazo, L.; Gonzalez, C.; Santos-Díez, L.; Tutau, C.; Gamboa, P.M.; et al. CD300c costimulates IgE-mediated basophil activation, and its expression is increased in patients with cow’s milk allergy. J. Allergy Clin. Immunol. 2019, 143, 700–711.e5. [Google Scholar] [CrossRef] [Green Version]
- Moshkovits, I.; Karo-Atar, D.; Itan, M.; Reichman, H.; Rozenberg, P.; Morgenstern-Ben-Baruch, N.; Shik, D.; Ejarque-Ortiz, A.; Hershko, A.Y.; Tian, L.; et al. CD300f associates with IL-4 receptor α and amplifies IL-4–induced immune cell responses. Proc. Natl. Acad. Sci. USA 2015, 112, 8708–8713. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Barriocanal, Á.; Arcas-García, A.; Magallon-Lorenz, M.; Ejarque-Ortíz, A.; Negro-Demontel, M.L.; Comas-Casellas, E.; Schwartz, S.; Malhotra, S.; Montalban, X.; Peluffo, H.; et al. Effect of Specific Mutations in Cd300 Complexes Formation; Potential Implication of Cd300f in Multiple Sclerosis. Sci. Rep. 2017, 7, 13544. [Google Scholar] [CrossRef]
- Moshkovits, I.; Reichman, H.; Karo-Atar, D.; Rozenberg, P.; Zigmond, E.; Haberman, Y.; Ben Baruch-Morgenstern, N.; Lampinen, M.; Carlson, M.; Itan, M.; et al. A key requirement for CD300f in innate immune responses of eosinophils in colitis. Mucosal Immunol. 2017, 10, 172–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachelet, I.; Munitz, A.; Moretta, A.; Moretta, L.; Levi-Schaffer, F. The Inhibitory Receptor IRp60 (CD300a) Is Expressed and Functional on Human Mast Cells. J. Immunol. 2005, 175, 7989–7995. [Google Scholar] [CrossRef] [Green Version]
- Munitz, A. The inhibitory receptor IRp60 (CD300a) suppresses the effects of IL-5, GM-CSF, and eotaxin on human peripheral blood eosinophils. Blood 2006, 107, 1996–2003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabato, V.; Boita, M.; Shubber, S.; Bridts, C.H.; Shibuya, A.; De Clerck, L.S.; Falcone, F.H.; Ebo, D.G. Mechanism of phosphatidylserine inhibition of IgE/FcεRI-dependent anaphylactic human basophil degranulation via CD300a. J. Allergy Clin. Immunol. 2014, 134, 734–737.e3. [Google Scholar] [CrossRef] [PubMed]
- Nissim Ben Efraim, A.H.; Karra, L.; Ben-Zimra, M.; Levi-Schaffer, F. The inhibitory receptor CD300a is up-regulated by hypoxia and GM-CSF in human peripheral blood eosinophils. Allergy 2013, 68, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Karra, L.; Singh Gangwar, R.; Shamri, R.; Puzzovio, P.G.; Cohen-Mor, S.; Levy, B.D.; Levi-Schaffer, F. Leukocyte CD300a Contributes to the Resolution of Murine Allergic Inflammation. J. Immunol. 2018, 201, 2998–3005. [Google Scholar] [CrossRef]
- Alvarez, Y.; Tang, X.; Coligan, J.E.; Borrego, F. The CD300a (IRp60) inhibitory receptor is rapidly up-regulated on human neutrophils in response to inflammatory stimuli and modulates CD32a (FcγRIIa) mediated signaling. Mol. Immunol. 2008, 45, 253–258. [Google Scholar] [CrossRef] [Green Version]
- Munitz, A.; Bachelet, I.; Levi-Schaffer, F. Reversal of airway inflammation and remodeling in asthma by a bispecific antibody fragment linking CCR3 to CD300a. J. Allergy Clin. Immunol. 2006, 118, 1082–1089. [Google Scholar] [CrossRef]
- Yotsumoto, K.; Okoshi, Y.; Shibuya, K.; Yamazaki, S.; Tahara-Hanaoka, S.; Honda, S.; Osawa, M.; Kuroiwa, A.; Matsuda, Y.; Tenen, D.G.; et al. Paired Activating and Inhibitory Immunoglobulin-like Receptors, MAIR-I and MAIR-II, Regulate Mast Cell and Macrophage Activation. J. Exp. Med. 2003, 198, 223–233. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Nakahashi-Oda, C.; Okayama, Y.; Shibuya, A. Autonomous regulation of IgE-mediated mast cell degranulation and immediate hypersensitivity reaction by an inhibitory receptor CD300a. J. Allergy Clin. Immunol. 2019, 144, 323–327.e7. [Google Scholar] [CrossRef] [Green Version]
- Juncadella, I.J.; Kadl, A.; Sharma, A.K.; Shim, Y.M.; Hochreiter-Hufford, A.; Borish, L.; Ravichandran, K.S. Apoptotic cell clearance by bronchial epithelial cells critically influences airway inflammation. Nature 2013, 493, 547–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jyonouchi, H. Airway epithelium and apoptosis. Apoptosis 1999, 4, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.-P.; Zhou, H.-B.; Xia, L.-X.; Shen, H.-H.; Ying, S. Balance of apoptotic cell death and survival in allergic diseases. Microbes Infect. 2014, 16, 811–821. [Google Scholar] [CrossRef]
- Izawa, K.; Kitaura, J.; Yamanishi, Y.; Matsuoka, T.; Oki, T.; Shibata, F.; Kumagai, H.; Nakajima, H.; Maeda-Yamamoto, M.; Hauchins, J.P.; et al. Functional analysis of activating receptor LMIR4 as a counterpart of inhibitory receptor LMIR3. J. Biol. Chem. 2007, 282, 17997–18008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moshkovits, I.; Shik, D.; Itan, M.; Karo-Atar, D.; Bernshtein, B.; Hershko, A.Y.; Van Lookeren Campagne, M.; Munitz, A. CMRF35-like molecule 1 (CLM-1) regulates eosinophil homeostasis by suppressing cellular chemotaxis. Mucosal Immunol. 2014, 7, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Shik, D.; Moshkovits, I.; Karo-Atar, D.; Reichman, H.; Munitz, A. Interleukin-33 requires CMRF35-like molecule-1 expression for induction of myeloid cell activation. Allergy 2014, 69, 719–729. [Google Scholar] [CrossRef]
- Matsukawa, T.; Izawa, K.; Isobe, M.; Takahashi, M.; Maehara, A.; Yamanishi, Y.; Kaitani, A.; Okumura, K.; Teshima, T.; Kitamura, T.; et al. Ceramide-CD300f binding suppresses experimental colitis by inhibiting ATP-mediated mast cell activation. Gut 2016, 65, 777–787. [Google Scholar] [CrossRef]
- Shiba, E.; Izawa, K.; Kaitani, A.; Isobe, M.; Maehara, A.; Uchida, K.; Maeda, K.; Nakano, N.; Ogawa, H.; Okumura, K.; et al. Ceramide-CD300f Binding Inhibits Lipopolysaccharide-induced Skin Inflammation. J. Biol. Chem. 2017, 292, 2924–2932. [Google Scholar] [CrossRef] [Green Version]
- Uchida, S.; Izawa, K.; Ando, T.; Yamada, H.; Uchida, K.; Negishi, N.; Kaitani, A.; Maehara, A.; Nagamine, M.; Kamei, A.; et al. CD300f is a potential therapeutic target for the treatment of food allergy. Allergy 2020, 75, 471–474. [Google Scholar] [CrossRef]
- Yamanishi, Y.; Kitaura, J.; Izawa, K.; Matsuoka, T.; Oki, T.; Lu, Y.; Shibata, F.; Yamazaki, S.; Kumagai, H.; Nakajima, H.; et al. Analysis of mouse LMIR5/CLM-7 as an activating receptor: Differential regulation of LMIR5/CLM-7 in mouse versus human cells. Blood 2008, 111, 688–698. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Barriocanal, Á.; Sayós, J. Molecular and Functional Characterization of CD300b, a New Activating Immunoglobulin Receptor Able to Transduce Signals through Two Different Pathways. J. Immunol. 2006, 177, 2819–2830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schroeder, J.T.; Chichester, K.L.; Bieneman, A.P. Human Basophils Secrete IL-3: Evidence of Autocrine Priming for Phenotypic and Functional Responses in Allergic Disease. J. Immunol. 2009, 182, 2432–2438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Enomoto, Y.; Yamanishi, Y.; Izawa, K.; Kaitani, A.; Takahashi, M.; Maehara, A.; Oki, T.; Takamatsu, R.; Kajikawa, M.; Takai, T.; et al. Characterization of Leukocyte Mono-immunoglobulin-like Receptor 7 (LMIR7)/CLM-3 as an Activating Receptor. J. Biol. Chem. 2010, 285, 35274–35283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larsen, L.F.; Juel-Berg, N.; Hansen, A.; Hansen, K.S.; Mills, E.N.C.; Van Ree, R.; Rådinger, M.; Poulsen, L.K.; Jensen, B.M. No difference in human mast cells derived from peanut allergic versus non-allergic subjects. Immun. Inflamm. Dis. 2018, 6, 416–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imai, Y.; Yasuda, K.; Sakaguchi, Y.; Haneda, T.; Mizutani, H.; Yoshimoto, T.; Nakanishi, K.; Yamanishi, K. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc. Natl. Acad. Sci. USA 2013, 110, 13921–13926. [Google Scholar] [CrossRef] [Green Version]
- Weidinger, S.; Novak, N. Atopic dermatitis. Lancet 2016, 387, 1109–1122. [Google Scholar] [CrossRef]
- Meyer zu Heringdorf, D. Sphingosylphosphorylcholine—Biological functions and mechanisms of action. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2002, 1582, 178–189. [Google Scholar] [CrossRef]
- Kang, J.S.; Youm, J.-K.; Jeong, S.K.; Park, B.D.; Yoon, W.K.; Han, M.H.; Lee, H.; Han, S.-B.; Lee, K.; Park, S.-K.; et al. Topical application of a novel ceramide derivative, K6PC-9, inhibits dust mite extract-induced atopic dermatitis-like skin lesions in NC/Nga mice. Int. Immunopharmacol. 2007, 7, 1589–1597. [Google Scholar] [CrossRef]
- Harvima, I.T.; Levi-Schaffer, F.; Draber, P.; Friedman, S.; Polakovicova, I.; Gibbs, B.F.; Blank, U.; Nilsson, G.; Maurer, M. Molecular targets on mast cells and basophils for novel therapies. J. Allergy Clin. Immunol. 2014, 134, 530–544. [Google Scholar] [CrossRef]
- Jo-Watanabe, A.; Okuno, T.; Yokomizo, T. The Role of Leukotrienes as Potential Therapeutic Targets in Allergic Disorders. Int. J. Mol. Sci. 2019, 20, 3580. [Google Scholar] [CrossRef] [Green Version]
- Evans, D.J.; Barnes, P.J.; Spaethe, S.M.; Van Alstyne, E.L.; Mitchell, M.I.; O’Connor, B.J. Effect of a leukotriene B4 receptor antagonist, LY293111, on allergen induced responses in asthma. Thorax 1996, 51, 1178–1184. [Google Scholar] [CrossRef] [Green Version]
- Asanuma, F.; Kuwabara, K.; Arimura, A.; Furue, Y.; Fleisch, J.H.; Hori, Y. Effects of leukotriene B 4 receptor antagonist, LY293111Na, on antigen-induced bronchial hyperresponsiveness and leukocyte infiltration in sensitized guinea pigs. Inflamm. Res. 2001, 50, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Gangwar, R.S.; Landolina, N.; Arpinati, L.; Levi-Schaffer, F. Mast cell and eosinophil surface receptors as targets for anti-allergic therapy. Pharmacol. Ther. 2017, 170, 37–63. [Google Scholar] [CrossRef] [PubMed]
- Kocatürk, E.; Zuberbier, T. New biologics in the treatment of urticaria. Curr. Opin. Allergy Clin. Immunol. 2018, 18, 425–431. [Google Scholar] [CrossRef]
- Holgate, S.T. New strategies with anti-IgE in allergic diseases. World Allergy Organ. J. 2014, 7, 17. [Google Scholar] [CrossRef] [Green Version]
- Holgate, S.; Buhl, R.; Bousquet, J.; Smith, N.; Panahloo, Z.; Jimenez, P. The use of omalizumab in the treatment of severe allergic asthma: A clinical experience update. Respir. Med. 2009, 103, 1098–1113. [Google Scholar] [CrossRef] [Green Version]
- Massanari, M.; Holgate, S.T.; Busse, W.W.; Jimenez, P.; Kianifard, F.; Zeldin, R. Effect of omalizumab on peripheral blood eosinophilia in allergic asthma. Respir. Med. 2010, 104, 188–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Humbert, M.; Busse, W.; Hanania, N.A.; Lowe, P.J.; Canvin, J.; Erpenbeck, V.J.; Holgate, S. Omalizumab in Asthma: An Update on Recent Developments. J. Allergy Clin. Immunol. Pract. 2014, 2, 525–536.e1. [Google Scholar] [CrossRef]
- D’Amato, G.; Stanziola, A.; Sanduzzi, A.; Liccardi, G.; Salzillo, A.; Vitale, C.; Molino, A.; Vatrella, A.; D’Amato, M. Treating severe allergic asthma with anti-IgE monoclonal antibody (omalizumab): A review. Multidiscip. Respir. Med. 2014, 9, 23. [Google Scholar] [CrossRef] [Green Version]
- Stokes, J.R.; Casale, T.B. The Use of Anti-IgE Therapy Beyond Allergic Asthma. J. Allergy Clin. Immunol. Pract. 2015, 3, 162–166. [Google Scholar] [CrossRef]
- Kawakami, T.; Blank, U. From IgE to Omalizumab. J. Immunol. 2016, 197, 4187–4192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beck, L.A.; Marcotte, G.V.; MacGlashan, D.; Togias, A.; Saini, S. Omalizumab-induced reductions in mast cell FcεRI expression and function. J. Allergy Clin. Immunol. 2004, 114, 527–530. [Google Scholar] [CrossRef] [PubMed]
- Djukanović, R.; Wilson, S.J.; Kraft, M.; Jarjour, N.N.; Steel, M.; Chung, K.F.; Bao, W.; Fowler-Taylor, A.; Matthews, J.; Busse, W.W.; et al. Effects of Treatment with Anti-immunoglobulin E Antibody Omalizumab on Airway Inflammation in Allergic Asthma. Am. J. Respir. Crit. Care Med. 2004, 170, 583–593. [Google Scholar] [CrossRef]
- Lin, H.; Boesel, K.M.; Griffith, D.T.; Prussin, C.; Foster, B.; Romero, F.; Townley, R.; Casale, T.B. Omalizumab rapidly decreases nasal allergic response and FcεRI on basophils. J. Allergy Clin. Immunol. 2004, 113, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Holgate, S.; Casale, T.; Wenzel, S.; Bousquet, J.; Deniz, Y.; Reisner, C. The anti-inflammatory effects of omalizumab confirm the central role of IgE in allergic inflammation. J. Allergy Clin. Immunol. 2005, 115, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Seyed Jafari, S.M.; Gadaldi, K.; Feldmeyer, L.; Yawalkar, N.; Borradori, L.; Schlapbach, C. Effects of Omalizumab on FcεRI and IgE Expression in Lesional Skin of Bullous Pemphigoid. Front. Immunol. 2019, 10, 15–18. [Google Scholar] [CrossRef] [Green Version]
- Saini, S.S.; MacGlashan, D.W.; Sterbinsky, S.A.; Togias, A.; Adelman, D.C.; Lichtenstein, L.M.; Bochner, B.S. Down-regulation of human basophil IgE and FC epsilon RI alpha surface densities and mediator release by anti-IgE-infusions is reversible in vitro and in vivo. J. Immunol. 1999, 162, 5624–5630. [Google Scholar]
- Chan, M.A.; Gigliotti, N.M.; Dotson, A.L.; Rosenwasser, L.J. Omalizumab may decrease IgE synthesis by targeting membrane IgE+ human B cells. Clin. Transl. Allergy 2013, 3, 29. [Google Scholar] [CrossRef] [Green Version]
- Nyborg, A.C.; Zacco, A.; Ettinger, R.; Jack Borrok, M.; Zhu, J.; Martin, T.; Woods, R.; Kiefer, C.; Bowen, M.A.; Suzanne Cohen, E.; et al. Development of an antibody that neutralizes soluble IgE and eliminates IgE expressing B cells. Cell. Mol. Immunol. 2016, 13, 391–400. [Google Scholar] [CrossRef] [Green Version]
- Haba, S.; Nisonoff, A. Effects of syngeneic anti-IgE antibodies on the development of IgE memory and on the secondary IgE response. J. Immunol. 1994, 152, 51–57. [Google Scholar]
- Burt, D.S.; Stanworth, D.R. Inhibition of binding of rat IgE to rat mast cells by synthetic IgE peptides. Eur. J. Immunol. 1987, 17, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Offermann, D.A.; McKendrick, J.E.; Sejberg, J.J.P.; Mo, B.; Holdom, M.D.; Helm, B.A.; Leatherbarrow, R.J.; Beavil, A.J.; Sutton, B.J.; Spivey, A.C. Synthesis and Incorporation into Cyclic Peptides of Tolan Amino Acids and Their Hydrogenated Congeners: Construction of an Array of A–B-loop Mimetics of the Cε3 Domain of Human IgE. J. Org. Chem. 2012, 77, 3197–3214. [Google Scholar] [CrossRef]
- Sandomenico, A.; Monti, S.M.; Marasco, D.; Dathan, N.; Palumbo, R.; Saviano, M.; Ruvo, M. IgE-binding properties and selectivity of peptide mimics of the FcɛRI binding site. Mol. Immunol. 2009, 46, 3300–3309. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.S.; Sandomenico, A.; Severino, V.; Burton, O.T.; Darling, A.; Oettgen, H.C.; Ruvo, M. An IgE receptor mimetic peptide (PepE) protects mice from IgE mediated anaphylaxis. Mol. Biosyst. 2013, 9, 2853. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pahima, H.; Puzzovio, P.G.; Levi-Schaffer, F. 2B4 and CD48: A powerful couple of the immune system. Clin. Immunol. 2019, 204, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Munitz, A.; Bachelet, I.; Finkelman, F.D.; Rothenberg, M.E.; Levi-Schaffer, F. CD48 Is Critically Involved in Allergic Eosinophilic Airway Inflammation. Am. J. Respir. Crit. Care Med. 2007, 175, 911–918. [Google Scholar] [CrossRef]
- Zhang, F.; Huang, G.; Hu, B.; Song, Y.; Shi, Y. A soluble thymic stromal lymphopoietin (TSLP) antagonist, TSLPR-immunoglobulin, reduces the severity of allergic disease by regulating pulmonary dendritic cells. Clin. Exp. Immunol. 2011, 164, 256–264. [Google Scholar] [CrossRef]
- Lin, L.; Zheng, P.; Yuen, J.W.M.; Wang, J.; Zhou, J.; Kong, C.; Peng, X.; Li, J.; Li, L. Prevention and treatment of allergic inflammation by an Fcγ-Der f2 fusion protein in a murine model of dust mite-induced asthma. Immunol. Res. 2012, 52, 276–283. [Google Scholar] [CrossRef]
- Cemerski, S.; Chu, S.Y.; Moore, G.L.; Muchhal, U.S.; Desjarlais, J.R.; Szymkowski, D.E. Suppression of mast cell degranulation through a dual-targeting tandem IgE–IgG Fc domain biologic engineered to bind with high affinity to FcγRIIb. Immunol. Lett. 2012, 143, 34–43. [Google Scholar] [CrossRef]
- Youngblood, B.A.; Brock, E.C.; Leung, J.; Falahati, R.; Bochner, B.S.; Rasmussen, H.S.; Peterson, K.; Bebbington, C.; Tomasevic, N. Siglec-8 antibody reduces eosinophils and mast cells in a transgenic mouse model of eosinophilic gastroenteritis. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [Green Version]
- Youngblood, B.A.; Brock, E.C.; Leung, J.; Falahati, R.; Bryce, P.J.; Bright, J.; Williams, J.; Shultz, L.D.; Greiner, D.L.; Brehm, M.A.; et al. AK002, a Humanized Sialic Acid-Binding Immunoglobulin-Like Lectin-8 Antibody that Induces Antibody-Dependent Cell-Mediated Cytotoxicity against Human Eosinophils and Inhibits Mast Cell-Mediated Anaphylaxis in Mice. Int. Arch. Allergy Immunol. 2019, 180, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Bachelet, I.; Munitz, A.; Levi-Schaffer, F. Abrogation of allergic reactions by a bispecific antibody fragment linking IgE to CD300a. J. Allergy Clin. Immunol. 2006, 117, 1314–1320. [Google Scholar] [CrossRef] [PubMed]
- Bachelet, I.; Munitz, A.; Berent-Maoz, B.; Mankuta, D.; Levi-Schaffer, F. Suppression of Normal and Malignant Kit Signaling by a Bispecific Antibody Linking Kit with CD300a. J. Immunol. 2008, 180, 6064–6069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| MAST CELLS | EOSINOPHILS | BASOPHILS | Refs. | |
|---|---|---|---|---|
| CD300a (also named as CMRF-35H, IRp60, IRC1 or IRC2 in humans and CLM-8, LMIR-1 or MAIR-I in mice) | ✓ Constitutively expressed ✓ Eosinophil-derived MBP and EDN down-regulate CD300a expression on CBMCs ✓ Inhibit IgE/FcεRI dependent activation ✓ Self-regulation mechanism by cis-interaction of CD300a with PS | ✓ Constitutively expressed ✓ Hypoxia and GM-CSF increase CD300a expression ✓ Inhibit Src kinase-dependent activation pathways | ✓ Constitutively expressed ✓ Up-regulated in response to IgE/FcεRI cross-linking and stimulation with fMLP, PMA and PMA + ionomycin ✓ Inhibit IgE/FcεRI dependent activation | [57,62,63,64,65,68,70] |
| CD300b (also named as CD300lb or IREM-3 in humans and CLM-7, LMIR-5, CD300b or mIREM3 in mice) | ✓ Constitutively expressed ✓ Activating role | No available data | No available data | [80] |
| CD300c (also named as CMRF-35A in humans and CLM-6 in mice) | ✓ Constitutively expressed. ✓ Activating role | No available data | ✓ Constitutively expressed ✓ Up-regulated in response to IL-3 ✓ Co-stimulate IgE/FcεRI dependent activation | [42,58] |
| CD300d (also named as CD300ld in humans and CLM-5, LMIR-4 or MAIR-IV in mice) | ✓ Expressed on mice ✓ LPS-mediated stimulation down-regulate CD300d expression ✓ Activating role | No available data | No available data | [74] |
| CD300f (also named as CD300lf, IREM-1 or IgSF13 in humans and CLM-1, DIgR2, LMIR-3 or MAIR-V in mice) | ✓ Constitutively expressed ✓ Allergen challenge increase CD300f expression ✓ Inhibitory and activating roles have been demonstrated | ✓ Constitutively expressed ✓ Allergen challenge increase CD300f expression ✓ Negative regulator of eotaxin-induced responses ✓ Activating role in innate immune activities of eosinophils | ✓ Constitutively expressed | [58,59,74,75,76,77] |
| CD300h (also named as CLM-3 or LMIR-7 in mice) | ✓ Expressed on mice ✓ Activating role | No available data | No available data | [83] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitallé, J.; Terrén, I.; Orrantia, A.; Bilbao, A.; Gamboa, P.M.; Borrego, F.; Zenarruzabeitia, O. The Expression and Function of CD300 Molecules in the Main Players of Allergic Responses: Mast Cells, Basophils and Eosinophils. Int. J. Mol. Sci. 2020, 21, 3173. https://doi.org/10.3390/ijms21093173
Vitallé J, Terrén I, Orrantia A, Bilbao A, Gamboa PM, Borrego F, Zenarruzabeitia O. The Expression and Function of CD300 Molecules in the Main Players of Allergic Responses: Mast Cells, Basophils and Eosinophils. International Journal of Molecular Sciences. 2020; 21(9):3173. https://doi.org/10.3390/ijms21093173
Chicago/Turabian StyleVitallé, Joana, Iñigo Terrén, Ane Orrantia, Agurtzane Bilbao, Pedro M. Gamboa, Francisco Borrego, and Olatz Zenarruzabeitia. 2020. "The Expression and Function of CD300 Molecules in the Main Players of Allergic Responses: Mast Cells, Basophils and Eosinophils" International Journal of Molecular Sciences 21, no. 9: 3173. https://doi.org/10.3390/ijms21093173






