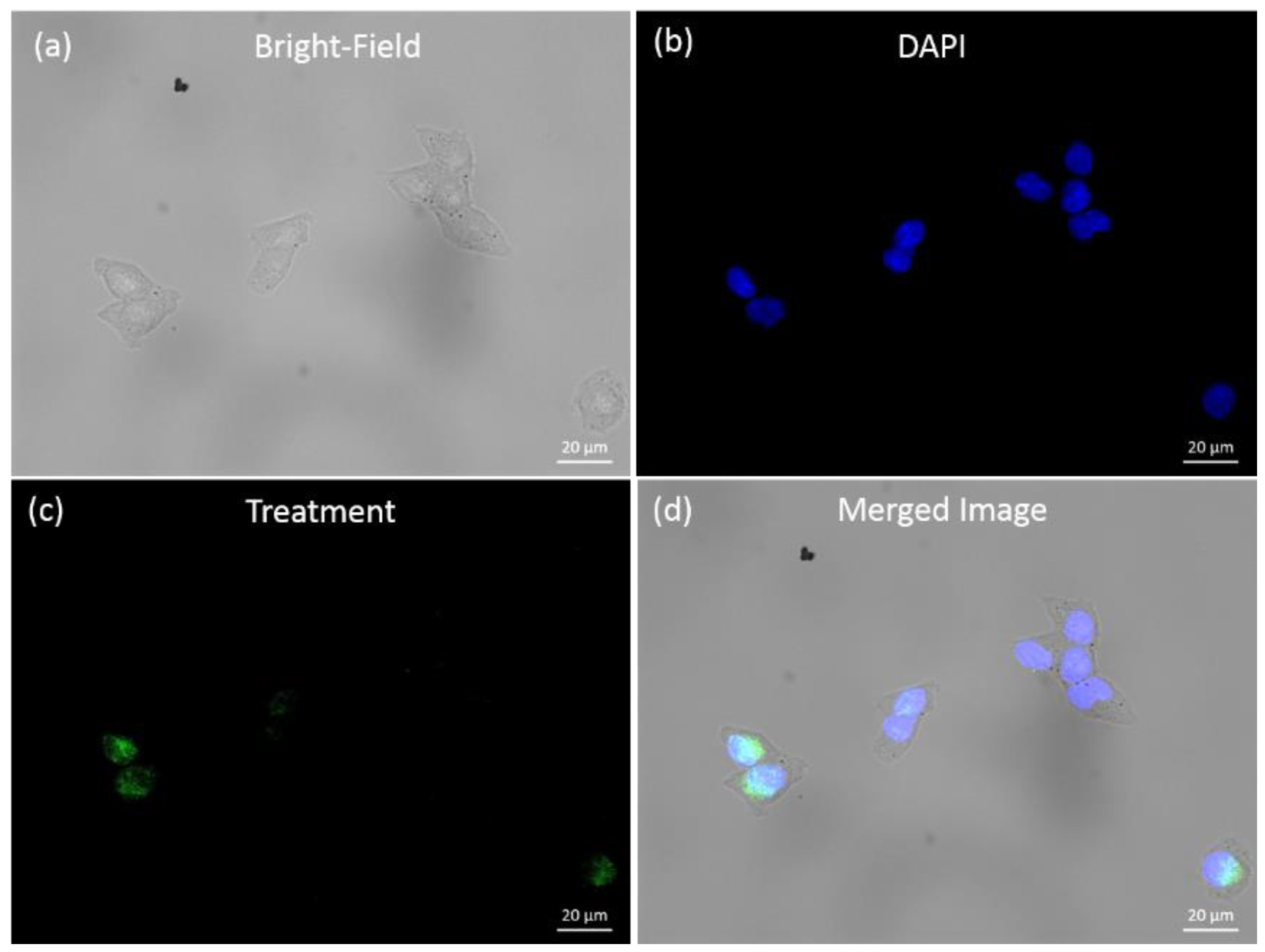
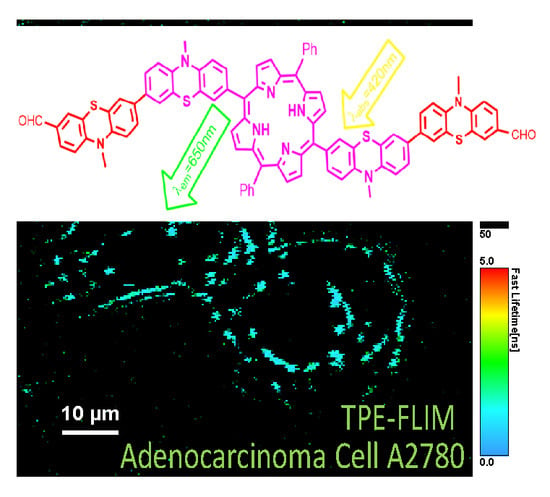
3.2. Reagents
Phenothiazine, 2-chlorophenothiazine and phenylboronic acid were purchased from Sigma Aldrich;
10-methyl-7-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-10H-phenothiazine-3-carbaldehyde was prepared according to the procedure described in the literature [
39].
(4-(9H-carbazol-9-yl)phenyl)boronic acid was prepared according to the procedure described in the literature [
40].
5,10,15,20-tetrakis(4-bromophenyl)-21H,23H-porphine was prepared according to the procedure described in the literature [
41].
8-Cloro-3-formyl-10-methyl-phenothiazine 1b
To a cooled solution (0 °C) containing DMF (10 mL) and POCl3 (12 mL), 2-chloro-10-methyl-10H-phenothiazine (30 g, 0.12 mol) dissolved in 1,2-dichloroethane (150 mL) was added, and the reaction mixture was heated at 85 °C for 6 h. The reaction mixture was poured into cold water (200 mL) and extracted with ethyl-acetate (2 × 200 mL). The extract was dried over anhydrous magnesium sulfate and the solvent was removed till dryness. The product was purified by column chromatography, using a silica gel solid support and toluene as eluent. Yellow powder, yield 48% (16.1g), 1H-NMR (400 MHz, CDCl3) δppm 3.44 (s, 3H, CH3), 6.81(d, 1H, 4J = 1.9 Hz), 6.88 (dd, 1H, 3J = 6.4 Hz, 4J = 1.9 Hz), 6.97 (d 1H, 3J = 6.4 Hz), 7.03 (d, 1H, 3J = 6.8 Hz), 7.60 (d, 1H, 4J = 1.8 Hz), 7.68 (dd, 1H, 3J = 6.8 Hz 4J = 1.8 Hz), 9.81 (s, 1H, CHO); 13C-NMR (100 MHz, CDCl3) δppm 35.9 (N-CH3), 114.1, 115.2, 121.0 (Cq), 123.4 (Cq), 123.8, 127.8, 127.9, 130.6, 131.5 (Cq), 133.7 (Cq), 145.3 (Cq), 150.3 (Cq), 190.0 (CHO).
3.2.1. General Procedure for the Synthesis of AB3 and A2B2-Type Meso-Halogenophenothiazinyl-Porphyrins (2 and 3)
Propionic acid (100 mL) and acetic anhydride (3 mL) were stirred and heated at 110 °C for 1 h. After cooling at room temperature, benzaldehyde (12mmol), 10-methyl-10H-phenothiazinyl-carbaldehyde derivative (12mmol) and pyrrole (24 mmol) were added and the mixture was heated at 110 °C for 4 h. After completion, the obtained purple crystals were collected by filtration and washed with methanol to remove the traces of propionic acid. In the case in which the product did not precipitate, the solvent was removed by reduced pressure distillation and the residue was washed with methanol. The separation by column chromatography on silica gel gave the corresponding meso-halogenophenothiazinyl-porphyrins (2) and (3) respectively.
5,10,15-triphenyl-20-(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 2a
Starting with bromo-phenothiazine carbaldehyde 1a (4 g, 0.012 mol), benzaldehyde (1.3 mL, 0.012 mol), pyrrole (1.7 mL, 0.024 mol), propionic acid (100 mL) and acetic anhydride (3 mL) were added, and after purification by column chromatography using dichloromethane/petrol ether (2:1) as eluent, a purple powder was obtained; yield 18% (0.9 g). 1H-NMR (400 MHz, CDCl3) δppm -2.73 (s, 2H, NH), 3.60 (s, 3H), 6.85 (d, 1H, J = 7.4 Hz), 7.15 (d, 1H, J = 7.4 Hz), 7.37–7.40 (m, 2H), 7.78–7.83 (m, 9H), 8.01–8.05 (m, 2H), 8.25 (d, 6H, J = 7.2 Hz), 8.86–8.89 (m, 6H), 8.92 (d, 2H, J = 4.5 Hz); 13C-NMR (100 MHz, CDCl3) ppm 35.6, 112.5, 115.0, 115.4, 118.7 (Cq), 120.2 (4Cq), 121.2 (Cq), 125.5 (Cq), 126.7 (9C), 127.7 (6C), 129.7, 130.3 (2Cq), 132.9, 134.0, 134.5 (8C), 136.8 (Cq), 142.1 (8Cq), 145.0 (3Cq). HRMS-APCI calcd. for: C51H35BrN5S [M+H]+ 828.1717; found: 828.1791.
5,10,15-triphenyl-20-(2-chloro-10-methyl-10H-phenothiazin-7-yl)-21,23H-porphyrin 2b
Starting with phenothiazine carbaldehyde 1b (4 g, 0.012 mol), benzaldehyde (1.3 mL, 0.012 mol), pyrrole (1.7 mL, 0.024 mol), propionic acid (100 mL) and acetic anhydride (3 mL), a mixture of 2b and 3b was obtained. After separation by column chromatography using chloroform/heptane (2:1) as eluent, 2b was obtained as a purple powder; yield 14% (0.4 g); 1H-NMR (600 MHz, CDCl3) δppm −2.76 (s, 2H, NH), 3.66 (s, 3H), 7.01(s, 1H), 7.04 (d, 1H, J = 7.9 Hz), 7.18 (d, 1H, J = 8.1 Hz), 7.21 (d, 1H, J = 7.9 Hz), 7.78–7.82 (m, 9H,), 8.02–8.06(m, 2H), 8.24 (d, 6H, J = 8.2 Hz), 8.87 (d, 6H, J = 4.2 Hz), 8.91 (d, 2H, J = 4.2 Hz); 13C-NMR (150 MHz, CDCl3) δ ppm 35.7, 112.6, 114.8, 118.7 (Cq), 120.21, 120.23 (Cq), 121.6 (Cq), 121.8 (Cq), 122.5, 126.7 (9C), 127.7 (6C), 127.9 (3Cq), 128.2 (Cq), 129.0 (Cq), 132.9, 133.7 (2Cq), 133.9, 134.5 (8C), 137.0 (2Cq), 142.1 (4Cq), 144.8 (2Cq), 147.0 (2Cq). HRMS-APCI calcd. for: C51H35ClN5S [M+H]+ 783.2223; found: 783.2206.
5,15-diphenyl-10,20-bis(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 3a
Purification by column chromatography using dichloromethane/petrol ether (2:1) as eluent gave a purple powder; yield 12% (0.8 g); 1H-NMR (400 MHz, CDCl3) δ ppm –2.75, (s, 2H, NH), 3.58 (s, 6H), 6.83 (d, 2H, J = 9.2 Hz), 7.14 (d, 2H, J = 8.4 Hz), 7.37–7.41 (m, 4H), 7.77–7.81 (m, 6H), 7.98–8.04 (m, 4H), 8.24 (d, 4H, J = 8.2 Hz), 8.87 (d, 4H, J = 4.2 Hz), 8.91 (d, 4H, J = 4.2 Hz); 13C-NMR (100 MHz, CDCl3) δppm 36.0 (2C), 112.5 (2C), 115.0 (2C), 115.4 (2C), 118.8 (2Cq), 120.2 (2Cq), 121.1 (Cq), 125.3 (Cq), 125.5 (2Cq), 126.7 (4C), 127.7 (2C), 128.2 (2Cq), 129.0 (2C), 129.7 (4C), 130.3 (4C), 132.9 (2C), 134.0 (2C), 134.5 (4C), 136.8 (4Cq), 142.1 (4Cq), 145.0 (4Cq), 145.1 (4Cq). HRMS-APCI calcd. for: C58H39Br2N6S2 [M+H]+ 1043.1018; found: 1043.0968.
5,15-diphenyl-10,20-bis (2-chloro-10-methyl-10H-phenothiazin-7-yl)-21,23H-porphyrin 3b
Purification by column chromatography using dichloromethane/petrol ether (2:1) as eluent gave a purple powder; yield 10% (0.3 g); 1H-NMR (400 MHz, CDCl3) δ ppm-2.77 (s, 2H, NH), 3.65 (s, 6H), 7.03–7.05 (m, 4H), 7.16–7.21 (m, 4H), 7.76–7.81 (m, 6H), 8.02 (d, 4H, J = 7.4Hz), 8.23 (d, 4H, J = 8.4Hz), 8.86–8.90 (m, 8H); 13C-NMR (100 MHz, CDCl3) δ ppm 35.7 (2C), 112.6 (2C), 114.8 (4C), 118.8 (2Cq), 120.2 (2Cq), 121.6 (Cq), 121.7 (2Cq), 122.5 (4C), 126.7 (6C), 127.7 (4Cq), 127.9 (4C), 128.2 (Cq) 132.9 (2Cq), 133.7 (3Cq), 133.9 (3Cq), 134.5 (4C), 136.9 (4C), 142.0 (2Cq), 144.8 (2Cq), 147.0 (4Cq). HRMS-APCI calcd. for: C58H39Cl2N6S2 [M+H]+ 952.1976; found: 952.1905.
3.2.2. General Procedure for the Preparation of Metallo-Phenothiazinyl-Porphyrins
A solution of phenothiazinyl-porphyrin free base (2) or (3) (1 mmol) and Metal(II)-acetate (1.1 mmol) in DMF (50 mL) was heated to reflux for 6 h. The purification was achieved by column chromatography on silica gel, using chloroform/hexane (1:2) as eluent.
5,10,15-triphenyl-20-(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Zn-porphyrin 4a
Purple powder; yield 90% (0.9 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.59 (s, 3H), 6.83 (d, 1H, J = 8.7 Hz), 7.15 (d, 1H, J = 8.1 Hz), 7.32 (s, 1H), 7.38 (d, 1H, J = 8.7 Hz), 7.76–7.82 (m, 9H), 7.97 (s, 1H), 8.02 (d, 1H, J = 8.1 Hz), 8.26 (d, 6H, J = 8.4 Hz), 8.97–9.01 (m, 6H), 9.01 (d, 2H, J = 4.6 Hz); 13C-NMR (150 MHz, CDCl3) δ ppm 35.7, 112.3, 114.9 (Cq), 115.4, 119.7 (Cq), 120.9 (Cq), 121.2 (2Cq), 125.5 (Cq), 126.5 (6C), 126.6 (2C), 127.5 (3C), 129.6, 130.2, 131.8 (2Cq), 132.0 (6C), 132.1 (2C), 132.8, 133.8, 134.43 (2C), 134.45 (2C), 137.4 (Cq), 142.8 (4Cq), 144.8 (Cq), 145.0 (Cq), 150.2 (6Cq). HRMS-APCI calcd. for: C51H33BrN5ZnS [M+H]+ 892.0905; found: 892.0872.
5,10,15-triphenyl-20-(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Pd-porphyrin 4b
Purple powder; yield 80% (0.9 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.60 (s, 3H), 6.85 (d, 1H, J = 8.4 Hz), 7.15 (d, 1H, J = 8.5 Hz), 7.38 (s, 1H, H6), 7.40 (d, 1H, J = 8.4 Hz), 7.74–7.81 (m, 9H), 7.95–7.98 (m, 2H), 8.17–8.21(m, 6H), 8.81–8.85 (m, 6H), 8.89 (d, 2H, J = 4.8 Hz); 13C-NMR (150 MHz, CDCl3) δ ppm 35.7, 112.3 (Cq), 112.4, 114.3 (Cq), 115.0 (Cq), 115.4, 120.3 (Cq), 121.2 (Cq), 121.7 (2Cq), 125.5 (Cq), 126.71 (4C), 126.78 (2C), 127.4 (Cq), 127.7 (3C), 128.2 (Cq), 129.6, 130.3, 130.8 (2Cq), 130.9 (2C), 131.01 (6C), 131.04 (2C), 132.5, 133.4 (Cq), 133.5, 134.0 (2C), 134.1 (2C), 136.4 (Cq), 141.6 (4Cq), 141.7 (2Cq), 145.0 (Cq). HRMS-APCI calcd. for: C51H33BrN5PdS [M+H]+ 934.0648; found: 934.0641.
5,10,15-triphenyl-20-(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Ni-porphyrin 4c
Purple powder; yield 85% (0.9 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.57 (s, 3H), 6.82 (d, 1H, J = 8.2 Hz), 7.10 (d, 1H, J = 7.8 Hz), 7.37 (s, 1H), 7.38 (d, 1H, J = 8.2 Hz), 7.69–7.73 (m, 9H), 7.81 (d, 1H, J = 7.8 Hz), 7.82 (s, 1H), 8.02–8.05 (m, 6H), 8.76–8.78 (m, 6H), 8.80 (d, 2H, J = 4.8 Hz); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (CH3), 112.6 (CH), 115.0 (Cq), 115.4 (CH), 117.6 (Cq), 119.0 (2Cq), 121.3 (Cq), 125.5 (Cq), 126.9 (6CH), 127.7 (3CH), 129.6 (CH), 130.3 (CH), 132.0 (2CH), 132.1 (CH), 132.21 (4CH), 132.23 (2CH), 133.28 (CH), 133.7 (6CH), 135.5 (Cq), 140.8 (4Cq), 142.71 (6Cq), 142.74 (2Cq), 145.0 (Cq), 145.1 (Cq) HRMS-APCI calcd. for: C51H33BrN5NiS [M+H]+ 886.0967; found: 886.0952.
5,10,15-triphenyl-20-(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Cu-porphyrin 4d
Purple powder; yield 80% (0.86 g), HRMS-APCI calcd. for: C51H33BrN5CuS [M+H]+ 889.0930; found: 889.0912.
5,10,15-triphenyl-20-(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Zn-porphyrin 5a
Purple powder; yield 95% (1 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.62 (s, 3H), 6.97 (s, 1H), 7.00 (d, 1H, J = 8.4 Hz), 7.09 (d, 1H, J = 7.9 Hz), 7.16 (d, 1H, J = 8.4 Hz), 7.75–7.82 (m, 9H), 7.97 (s, 1H), 8.02 (d, 1H, J = 7.9 Hz), 8.23–8.25 (m, 6H), 8.96–8.98 (m, 6H), 9.00 (d, 2H, J = 4.6 Hz); 13C-NMR (150 MHz, CDCl3) δ ppm 35.7, 112.4, 114.7, 119.6 (Cq), 121.21 (2Cq), 121.27 (Cq), 121.3 (Cq), 121.7 (Cq), 122.4, 126.51 (4C), 126.57 (2C), 127.5 (3C), 127.8, 131.8 (3Cq), 132.01 (4C), 132.04 (2C), 132.8, 133.6 (Cq), 133.7, 134.41 (4C), 134.44 (4C), 137.6 (Cq), 142.8 (3Cq), 144.5 (Cq), 147.0 (Cq), 150.2 (5Cq). HRMS-APCI calcd. for: C51H33ClN5ZnS [M+H]+ 846.1431; found: 846.1419.
5,10,15-triphenyl-20-(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Pd-porphyrin 5b
Purple powder; yield 90% (1 g); 1H-NMR (400 MHz, CDCl3) δ ppm 3.64 (s, 3H, CH3), 7.02–7.08 (m, 2H, H1,9), 7.15 (d, 1H, 3J = 8.2 Hz, H3), 7.34 (d, 1H, J = 8.2 Hz H8), 7.73–7.79 (m, 9H, Hb,c), 7.95 (d, 1H, J = 8.2 Hz H4), 7.98 (s, 1H, H6), 8.17–8.20 (m, 6H, Ha), 8.82–8.84 (m, 6H, Hβ), 8.89 (d, 2H, 3J = 4.9 Hz, Hβ); 13C-NMR (100 MHz, CDCl3) δ ppm 35.6, 112.3, 114.3, 120.7 (Cq), 121.7 (2Cq), 121.9 (Cq), 122.8 (Cq), 123.3 (Cq), 126.7 (4C), 126.7 (2C), 127.4, 127.7, 127.7 (3C), 131.0 (4C), 131.0 (2C), 132.5, 133.3, 134.4 (4C), 134.4 (4C), 136.0 (Cq), 141.5 (4Cq), 141.6 (2Cq), 141.7 (2Cq), 141.7 (4Cq), 145.5 (Cq), 145.8 (Cq). HRMS-APCI calcd. for: C51H33ClN5PdS [M + H]+ 890.1178; found: 890.1147.
5,10,15-triphenyl-20-(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Ni-porphyrin 5c
Purple powder; yield 88% (0.94 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.58 (s, 3H), 6.96–7.02 (m, 2H), 7.11–7.13 (m, 2H), 7.68–7.73 (m, 9H), 7.81–7.83 (m, 2H), 8.03–8.07 (d, 6H), 8.78–8.81 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6, 112.7, 114.7, 117.6 (Cq), 119.02 (Cq), 119.07, 121.7 (Cq), 121.8 (Cq), 122.5, 126.8 (6C), 127.7 (3C), 127.9, 132.0 (2C), 132.1, 132.24 (4C), 132.29 (2C), 133.1 (2Cq), 133.7 (6C), 135.7 (Cq), 140.9 (4Cq), 142.7 (8Cq), 144.9 (Cq), 146.9 (Cq). HRMS-APCI calcd. for: C51H33ClN5NiS [M+H]+ 840.1493; found: 840.1474.
5,10,15-triphenyl-20-(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Cu-porphyrin 5d
Purple powder; yield 80% (0.86 g); HRMS-APCI Calcd. for: C51H33ClN5CuS [M+H]+ 845.1435; found: 845.1420.
5,15-diphenyl-10,20-bis(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Zn-porphyrin 6a
Purple powder; yield 80% (0.85 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.52 (s, 6H), 6.78 (d, 2H, J = 8.5 Hz), 7.08 (d, 2H, J = 8.2 Hz), 7.26 (s, 2H), 7.34 (d, 2H, J = 8.5 Hz), 7.75–7.79 (m, 6H), 7.90–7.98 (m, 4H), 8.22–8.24 (m, 4H), 8.94–8.96 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (2C), 112.2 (2C), 112.3 (Cq), 114.8 (2Cq), 114.9 (Cq), 115.4 (2C), 119.6 (Cq), 120.7 (Cq), 120.8 (Cq), 121.14 (Cq), 121.17 (2Cq), 125.3 (Cq), 125.4 (Cq), 126.53 (2C), 126.57 (2C), 127.4 (2C), 129.5 (2Cq), 129.6 (Cq), 130.2 (2C), 131.8 (2C), 132.03 (2C), 132.08 (2C), 132.8 (Cq), 132.9 (Cq), 133.82 (2Cq), 133.86 (Cq), 134.5 (2C), 137.5 (2C), 142.9 (2C), 144.7 (4Cq), 145.0 (2C), 150.2 (6Cq). HRMS-APCI calcd. for: C58H37Br2N6ZnS2 [M+H]+ 1105.0153; found: 1105.0118.
5,15-diphenyl-10,20-bis(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Pd-porphyrin 6b
Purple powder; yield 80% (0.88 g); 1H-NMR (400 MHz, CDCl3) δ ppm 3.54 (s, 6H), 6.81 (d, 2H, J = 8.8 Hz), 7.01 (d, 2H, J = 8.4 Hz), 7.20 (d, 2H, J = 8.8 Hz), 7.36 (s, 2H), 7.74–7.78 (m, 6H), 7.90–7.94 (m, 4H), 8.18 (d, 4H, J = 8.6 Hz), 8.81–8.87 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (2C), 112.4 (2C), 114.4 (Cq), 115.0 (2Cq), 115.4 (2C), 120.4 (Cq), 121.9 (Cq), 125.3 (Cq), 125.50 (Cq), 125.54 (Cq), 1263.7 (6C), 127.8 (2C), 129.0 (2C), 129.7 (4C), 130.0 (2C), 130.0 (2C), 130.3 (2C), 131.0 (2C), 131.14 (2C), 131.19 (2C), 132.4 (2Cq), 133.5 (Cq), 134.11 (2Cq), 134.14 (2Cq), 136.3 (Cq), 141.61 (2Cq), 141.67 (2Cq), 141.7 (2Cq), 145.07 (2Cq), 145.09 (2Cq). HRMS-APCI calcd. for: C58H37Br2N6PdS2 [M+H]+ 1146.9871; found: 1146.9896.
5,15-diphenyl-10,20-bis(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Ni-porphyrin 6c
Purple powder; yield 85% (0.89 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.55 (s, 6H), 6.80 (d, 2H, J = 9.2 Hz), 7.08 (d, 2H, J = 8.0 Hz), 7.36–7.37 (m, 4H), 7.68–7.73 (m, 6H), 7.79–7.81 (m, 4H), 8.02 (d, 4H, J = 8.4 Hz), 8.76–8.80 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (2C), 112.6 (2C), 115.0 (2Cq), 115.4 (2C), 117.7 (Cq), 119.1 (Cq), 121.4 (2Cq), 125.4 (2Cq), 126.9 (4C), 127.8 (2Cq), 129.6 (2C), 130.3 (2C), 132.0 (2C), 132.1 (4C), 132.2 (2C), 132.3 (2C), 133.2 (2C), 133.7 (6C), 135.5 (Cq), 140.8 (Cq), 142.70 (5Cq), 142.77 (5Cq), 145.0 (2Cq), 145.1 (2Cq). HRMS-APCI calcd. for: C58H37Br2N6NiS2 [M+H]+ 1099.0215; found: 1099.0195.
5,15-diphenyl-10,20-bis(7-bromo-10-methyl-10H-phenothiazin-3-yl)-21,23-Cu-porphyrin 6d
Purple powder; yield 80% (0.84 g); HRMS-APCI calcd. for: C58H37Br2N6CuS2 [M+H]+ 1104.0157; found: 1104.0133.
5,15-diphenyl-10,20-bis(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Zn-porphyrin 7a
Purple powder; yield 95% (1 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.59 (s, 6H), 6.95 (s, 2H), 6.99 (d, 2H, J = 8.4 Hz), 7.04 (d, 2H, J = 8.2 Hz), 7.14 (d, 2H, J = 8.4 Hz), 7.76–7.81 (m, 6H), 7.91 (d, 2H, J = 8.4 Hz), 7.99 (d, 2H, J = 8.2 Hz), 8.22–8.24 (m, 4H), 8.95–8.99 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (2C), 112.4 (2Cq), 114.7 (4C), 119.74 (Cq), 119.78 (Cq), 121.23 (Cq), 121.28 (Cq), 121.53 (Cq), 121.59 (Cq), 122.4 (4C), 126.5 (4C), 127.5 (2C), 127.8 (2C), 131.8 (2C), 131.9 (2C), 132.0 (4C), 132.7 (Cq), 132.8 (Cq), 133.6 (2Cq), 133.7 (2Cq), 134.4 (2C), 137.5 (2Cq), 142.8 (2Cq), 144.4 (2Cq), 146.9 (2Cq), 150.23 (4Cq), 150.27 (4Cq) HRMS-APCI calcd. for: C58H37Cl2N6ZnS2 [M+H]+ 1017.1154; found: 1017.1118.
5,15-diphenyl-10,20-bis(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Pd-porphyrin 7b
Purple powder; yield 90% (1 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.65 (s, 6H), 7.00 (s, 2H), 7.04 (d, 2H, J = 8.4 Hz), 7.18–7.21 (m, 4H), 7.75–7.81 (m, 6H), 7.96–7.99 (m, 4H), 8.18 (d, 4H, J = 8.6 Hz), 8.82–8.90 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (2C), 112.6 (2Cq), 114.7 (4C), 119.72, 119.77, 121.2 (2Cq), 121.50 (Cq), 121.53 (Cq), 122.5 (4C), 126.7 (4C), 127.5 (2C), 127.8 (2C), 131.8 (2C), 131.9 (2C), 132.0 (2C), 132.7 (Cq), 132.8 (Cq), 133.6 (2Cq), 133.7 (2Cq), 134.7 (4C), 137.5 (2Cq), 142.8 (2Cq), 144.4 (2Cq), 146.9 (2Cq), 150.2 (4Cq), 150.2 (4Cq). HRMS-APCI calcd. for: C58H37Cl2N6PdS2 [M+H]+ 1059.0931; found: 1059.0891.
5,15-diphenyl-10,20-bis(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Ni-porphyrin 7c
Ni-2j Purple powder; yield 85% (0.9 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.53 (s, 6H), 6.93 (s, 2H), 6.98 (d, 2H, J = 8.6 Hz), 7.05 (d, 2H, J = 8.2 Hz), 7.10 (d, 2H, J = 8.6 Hz), 7.68–7.72 (m, 6H), 7.78 (d, 2H, J = 8.2 Hz), 7.82 (s, 2H), 8.04 (d, 4H), 8.77–8.81 (m, 8H,); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6 (2C), 112.6 (2Cq), 114.7 (2C), 119.70, 119.75, 121.2 (2Cq), 121.50 (Cq), 121.53 (Cq), 122.7(4C), 126.8 (4C), 127.3 (2C), 127.9 (2C), 130.7 (2C), 131.4 (2C), 132.5 (2C), 132.9 (2Cq), 133.1 (2Cq), 133.5 (2Cq), 133.6 (2Cq), 134.7 (4C), 137.6 (2Cq), 142.9 (2Cq), 144.4 (2Cq), 146.9 (2Cq), 150.3 (4Cq), 150.4 (4Cq). HRMS-APCI calcd. for: C58H37Cl2N6NiS2 [M+H]+ 1009.1246; found: 1009.1214.
5,15-diphenyl-10,20-bis(8-chloro-10-methyl-10H-phenothiazin-3-yl)-21,23-Cu-porphyrin 7d
Purple powder; yield 80% (0.85 g); HRMS-APCI calcd. for: C58H37Cl2N6CuS2 [M+H]+ 1014.1188; found: 1014.1163.
3.2.3. General Procedure for the Synthesis of Suzuki Cross-Coupling Products
A degassed solution of halogenoMPP porphyrin 2a, 3a or 4a (0.12 mmol 1eq), aryl-boronic pinacolate ester (0.13 mmol, 1.1eq), Pd(PPh3)2Cl2 (0.006 mmol 0.05eq), K2CO3 (0.1 mmol) in DME (30 mL) and water (15), was heated to reflux under argon atmosphere for 18 h. After cooling to room temperature, the obtained precipitate was filtered off and washed with water. The purification of cross-coupling product was achieved by column chromatography on silica gel using DCM or chloroform as eluent.
5,10,15-triphenyl-20-[7-(10′-methyl-10′H-phenothiazine)-10-methyl-10H-phenothiazin-3-yl]-21,23H-porphyrin 8
By starting with 0.15 g 2a, purification of the reaction product by column chromatography using DCM eluent gave: purple powder; yield 50% (0.07 g); 1H-NMR (400 MHz, CDCl3) δ ppm -2.76 (s, 2H, NH), 3.64 (s, 6H), 6.87 (d, 1H, J = 8.3 Hz), 7.19 (d, 1H, J = 7.9 Hz), 7.39–7.42 (m, 3H), 7.69–7.80 (m, 12H), 7.89 (d, 1H, J = 8.2 Hz), 8.02 (s, 1H), 8.04 (d, 1H, J = 7.9 Hz), 8.12 (d, 1H, J = 7.6 Hz), 8.19 (d, 1H, J = 7.6 Hz), 8.21–8.25 (m, 6H), 8.86–8.88 (m, 6H), 8.91 (d, 2H, J = 4.7 Hz); 13C-NMR (100 MHz, CDCl3) δ ppm 35.3, 36.8, 112.2 (Cq), 113.7, 114.0, 114.2 (2C), 115.4 (Cq), 118.8 (Cq), 121.15 (Cq), 121.19 (2Cq), 122.5, 122.7, 123.0 (Cq), 123.8 (Cq), 124.9 (2C), 125.4 (2C), 126.4 (4C), 126.5, 127.2 (2C), 127.4 (2C), 127.5 (2C), 129.6 (Cq), 131.8 (Cq), 131.9 (6C), 132.8 (Cq), 134.41 (6C), 134.48 (3C), 137.6 (Cq), 140.5 (Cq), 142.9 (2Cq), 144.7 (4Cq), 145.1 (2Cq), 145.6 (2Cq), 148.6 (4Cq). HRMS-APCI calcd. for: C64H45N6S2 [M+H]+ 961.3068; found: 961.1229.
5,10,15-triphenyl-20-(7-(10′-methyl-10′H-phenothiazine)-10-methyl-10H-phenothiazin-3-yl)-21,23-Zn-porphyrin 8a
By starting with 0.15 g 4a, purification of the reaction product by column chromatography using DCM eluent gave: purple powder; yield 55% (0.09 g); 1H-NMR (400 MHz, CDCl3) δ ppm 3.39 (s, 3H), 3.60 (s, 3H), 6.81–6.86 (m, 3H), 6.93 (t, 1H, J = 7.4 Hz), 7.13 (s, 1H), 7.17–7.21 (m, 2H), 7.29 (d, 1H, J = 7.4 Hz), 7.31 (s, 1H), 7.34 (d, 1H, J = 7.6 Hz), 7.38 (d, 1H, J = 7.4 Hz), 7.76–7.80 (m, 9H), 7.98 (s, 1H), 8.01 (d, 1H, J = 7.8 Hz), 8.25 (d, 6H), 8.99 (d, 6H), 9.00 (d, 1H, J = 4.5 Hz), 9.03 (d, 1H, J = 4.5 Hz); 13C-NMR (100 MHz, CDCl3) δ ppm 35.3, 40.2, 112.2 (Cq), 114.0 (2C), 114.2 (2C), 115.4 (Cq), 118.8 (Cq), 121.11 (Cq), 121.14 (2Cq), 122.5 (2C), 123.0 (Cq), 123.8 (Cq), 124.9 (2C), 125.4 (2C), 126.4 (4C), 126.5, 127.2 (2C), 127.4 (2C), 127.5 (2C), 129.6 (Cq), 131.8 (Cq), 131.9 (6C), 132.8 (Cq), 134.4 (4C), 134.4 (3C), 137.6 (Cq), 142.9 (2Cq), 144.7 (Cq), 145.1 (Cq), 145.6 (2Cq), 150.2 (8Cq). HRMS-APCI calcd. for: C64H42N6S2Zn [M+H]+ 1022.2203; found: 1022.1968.
5,10,15-triphenyl-20-(7-(3′-formyl-10′-methyl-10H-phenothiazine)-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 9
When starting with 0.1 g 2a, purification of the reaction product by column chromatography with DCM gave a purple powder; yield 60% (0.07 g); 1H-NMR (600 MHz, CDCl3) δ ppm -2.76 (s, 2H, NH), 3.49 (s, 3H), 3.60 (s, 3H), 6.88–6.91 (m, 4H), 7.06–7.10 (m, 4H), 7.63–7.66 (m, 2H), 7.76–7.79 (m, 9H), 8.02–8.05 (m, 2H), 8.24 (d, 6H, J = 7.8 Hz), 8.87–8.93 (m, 8H), 9.83 (s, 1H, CHO); 13C-NMR (150 MHz, CDCl3) δ ppm 35.6, 35.8, 112.4, 113.6 (2C), 114.5, 115.0 (2C), 119.0 (2Cq), 120.2, 121.50 (Cq), 121.53 (Cq), 123.0, 123.6 (2Cq), 123.8 (2Cq), 125.0 (3C), 125.2 (2C), 125.7 (6C), 126.7 (4C), 127.7, 127.9, 130.5 (2C), 131.1 (2C), 133.0, (2Cq) 133.9 (2Cq), 134.2, 134.5 (4C), 135.5 (2Cq), 136.6, 142.1 (4Cq), 143.0 (2Cq), 145.0 (2Cq), 145.2 (3Cq), 150.7 (2Cq), 190.0 (CHO); HRMS (APCI) calcd. for: C65H45N6OS2 [M+H+] 989.3018; found: 989.3071.
5,10,15-triphenyl-20-[7-(4-(9H-carbazole-9-yl)phenyl)-10-methyl-10H-phenothiazin-3-yl]-21,23H-porphyrin 10
When starting with 0.15 g 2a, purification of the reaction product by column chromatography with chloroform/petrol ether 1:1 eluent gave: purple powder; yield 70% (0.12 g); 1H-NMR (400 MHz, CDCl3) δ ppm -2.75 (s, 2H, NH), 3.72 (s, 3H), 7.13 (d, 1H, J = 8.7 Hz), 7.21–7.23 (m, 1H), 7.31–7.35 (m, 3H), 7.44–7.47 (m, 3H, H), 7.51 (d, 2H, J = 8.1 Hz), 7.61 (m, 2H), 7.65 (d, 2H, J = 8.1 Hz), 7.72–7.80 (m, 9H), 8.03 (d, 1H, J = 8.0 Hz), 8.07 (s, 1H), 8.18 (d, 2H, J = 7.7 Hz), 8.24 (d, 6H, J = 7.5 Hz), 8.87 (s, 6H), 8.95 (d, 2H, 3J = 4.4 Hz); 13C-NMR (100 MHz, CDCl3) δ ppm 35.8, 109.8 (2C), 112.4 (2Cq), 114.7 (2Cq), 118.9 (2Cq), 119.9 (2C), 120.2, 121.5, 123.4 (2C), 125.9 (6C), 126.7 (9C), 127.4 (4C), 127.7 (2C), 127.9 (4C), 133.0 (2Cq), 134.0 (2Cq), 134.5 (8C), 135.0 (2Cq), 136.6 (2Cq), 136.7 (2Cq), 140.8 (4Cq), 142.1 (4Cq), 145.2 (Cq), 145.4 (2Cq); HRMS (APCI) calcd. for: C69H47N6S [M+H+] 991.3577; found: 991.3573.
5,10,15-triphenyl-20-(7-phenyl-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 11
By starting with 0.1 g 2a, purification of the reaction product by column chromatography using DCM eluent gave: purple powder; yield 60% (0.06 g); 1H-NMR (400 MHz, CDCl3) δ ppm -2.77 (s, 2H, NH), 3.65 (s, 3H), 7.04 (d, 1H, J = 8.5 Hz), 7.16 (d, 1H, J = 8.1 Hz), 7.30–7.33 (m, 1H, H), 7.42 (t, 2H, J = 7.4 Hz, J = 7.7 Hz), 7.50 (d, 2H, J = 7.6 Hz), 7.57 (d, 2H, J = 7.4 Hz), 7.72–7.75 (m, 9H), 7.99 (d, 1H, J = 8.1 Hz), 8.02 (s, 1H), 8.20 (d, 6H, J = 6.3 Hz), 8.83 (s, 6H), 8.90 (d, 2H, 3J = 4.5 Hz); 13C-NMR (100 MHz, CDCl3) δ ppm 35.9, 112.5, 114.8, 118.8 (Cq), 120.2 (2Cq), 121.4 (Cq), 124.1 (Cq), 126.0, 126.7 (6C), 126.9 (4C), 127.6, 127.7 (3C), 129.6 (Cq), 129.7 (7C), 130.3 (Cq), 130.3 (4C), 131.0 (Cq), 133.0, 133.7 (Cq), 134.0, 134.1 (Cq), 134.2 (Cq), 134.5 (4C), 134.9 (Cq), 136.9 (Cq), 142.1 (4Cq), 144.9 (Cq), 145.8 (Cq), 146.1 (Cq), 150.2 (Cq), 165.4 (Cq); HRMS (APCI) calcd. for: C57H40N5S [M+H+] 826.8334; found: 826.8308.
5,10,15-triphenyl-20-(7-phenyl-10-methyl-10H-phenothiazin-3-yl)-21,23-Zn-porphyrin 11a
By starting with 0.15g 4a, purification of the reaction product by column chromatography using DCM eluent gave a purple powder; yield 66% (0.1 g); 1H-NMR (600 MHz, CDCl3) δ ppm 3.37 (s, 3H), 6.87 (d, 1H, J = 8.7 Hz), 7.10 (d, 1H, J = 8.5 Hz), 7.21 (d, 1H, J = 8.1 Hz), 7.36–7.37 (m, 1H), 7.41 (d, 1H, J = 8.6 Hz), 7.46 (t, 1H, J = 7.3 Hz, J = 8.0 Hz), 7.51 (s, 1H), 7.55 (d, 1H, J = 6.4 Hz), 7.61 (d, 1H, J = 7.4 Hz), 7.76–7.80 (m, 9H), 8.03–8.04 (m, 2H), 8.24–8.25 (m, 6H), 8.97 (s, 6H, Hβ), 9.01 (d, 1H, J = 4.5 Hz), 9.04 (d, 1H, J = 4.5 Hz); 13C-NMR (150 MHz, CDCl3) δ ppm 37.6, 112.9, 115.8, 119.8 (Cq), 120.8 (2Cq), 121.4 (Cq), 123.7, 124.1 (Cq), 126.4 (6C), 126.6 (4C), 128.2, 128.7 (3C), 129.6 (Cq), 129.7 (7C), 130.5 (Cq), 131.9 (4C), 132.0, 133.7 (Cq), 133.8 (Cq), 134.0, 134.1 (Cq), 134.3 (Cq), 134.4 (4C), 137.3 (Cq), 139.2 (Cq), 142.8 (Cq), 145.5 (Cq), 146.4 (Cq), 148.8 (Cq), 150.2 (Cq), 150.4 (2Cq), 150.7 (Cq), 156.6 (Cq); HRMS (APCI) calcd. for: C57H38N5SZn [M+H+] 888.2134; found: 888.2108.
5,10,15-triphenyl-20-(7-(4-formyl-phenyl)-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 12
By starting with 0.15 g 2a, purification of the reaction product by column chromatography using DCM eluent gave: purple powder; yield 40% (0.04 g); 1H-NMR (600 MHz, CDCl3) δ ppm -2.74 (s, 2H, NH), 3.71 (s, 3H), 7.09 (d, 1H, J = 8.4 Hz), 7.22 (d, 1H, J = 7.8 Hz), 7.57–7.59 (m, 2H), 7.78–7.81 (m, 9H), 7.95 (d, 2H, J = 7.9 Hz), 8.04–8.06 (m, 2H), 8.19 (s, 1H), 8.24 (d, 6H, J = 6.4 Hz), 8.83 (d, 1H, J = 6.3 Hz), 8.87–8.93 (m, 8H), 10.06 (s, 1H, CHO); 13C-NMR (150 MHz, CDCl3) δ ppm 35.8, 112.5, 114.6, 118.8 (Cq), 120.2 (2Cq), 121.4 (Cq), 124.1 (Cq), 126.0, 126.7 (6C), 126.9 (4C), 127.7 (3C), 129.6 (Cq), 130.3 (Cq), 129.7 (7C), 130.3 (4C), 131.0 (Cq), 133.0, 133.7 (Cq), 134.0, 134.1 (Cq), 134.2 (Cq), 134.5 (4C), 134.9 (Cq), 136.9 (Cq), 142.1 (4Cq), 144.9 (Cq), 145.8 (Cq), 146.1 (Cq), 150.2 (Cq), 165.4 (2Cq), 191.8 (CHO); HRMS (APCI) calcd. for: C58H40N5OS [M+H+] 854.2948; found: 854.2949.
5,15-diphenyl-15,20-bis(7-(3′-formyl-10′-methyl-10′H-phenothiazin-7yl)-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 13
After starting with 0.1g 3a, purification of the reaction product by column chromatography with DCM gave 13 as a purple powder; yield 65% (0.1 g); 1H-NMR (400 MHz, CDCl3) δ ppm −2.75 (s, 2H, NH), 3.43 (s, 6H), 3.65 (s, 6H), 6.85 (s, 4H), 7.03 (d. 2H, J = 6.2 Hz), 7.18 (d, 2H, J = 8.0 Hz), 7.37–7.39 (m, 4H), 7.45 (s, 4H), 7.63–7.66 (m, 4H), 7.77–7.79 (m, 6H), 8.01 (d, 2H, 3J = 8.0 Hz), 8.04 (s, 2H), 8.23 (d, 4H, J = 6.2 Hz), 8.87–8.92 (m, 8H), 9.81 (s, 2H, CHO); 13C-NMR (100 MHz, CDCl3) δ ppm 35.7 (2C), 35.8 (2C), 112.4 (2Cq), 113.7 (4C), 114.6 (2Cq), 115.0 (4C), 119.0 (2Cq), 120.2 (2Cq), 121.5 (Cq), 123.0 (4C), 123.6 (2Cq), 123.9 (Cq), 125.1 (4C), 125.3 (2Cq), 125.7 (6C), 126.7 (4C), 127.7 (2C), 128.0 (2C), 130.5 (2Cq), 131.1 (4C), 133.0 (Cq), 134.0 (Cq), 134.3 (2Cq), 134.6 (4C), 135.5 (2C), 136.6 (2C), 142.1 (4Cq), 143.0 (4Cq), 145.0 (4Cq), 145.2 (2Cq), 150.8 (4Cq), 190.1 (2CHO); HRMS (APCI) calcd. for: C86H59N8O2S4 [M+H+] 1363.3638; found: 1363.3625.
5,15-diphenyl-15,20-bis [7-(4-(9H-carbazole-9-yl)phenyl)-10-methyl-10H-phenothiazin-3-yl)]-21,23H-porphyrin 14
By starting with 0.1 g 3a, purification of the reaction product by column chromatography with chloroform/petrol ether 1:1 eluent gave a purple powder; yield 70% (0.1 g); 1H-NMR (600 MHz, CDCl3) δ ppm -2.72 (s, 2H, NH), 3.72 (s, 6H), 7.12 (d, 2H, J = 9.0 Hz), 7.22 (d, 2H, J = 8.1 Hz), 7.33 (t, 4H, J = 7.4 Hz, J = 7.2 Hz), 7.46 (t, 4H, J = 7.9 Hz, J = 7.2 Hz), 7.50–7.52 (m, 4H), 7.61–7.62 (m, 2H), 7.62 (s, 2H), 7.66 (d, 4H, J = 8.3 Hz), 7.78–7.83 (m, 10H), 8.05 (s, 2H), 8.09 (s, 2H), 8.10 (d, 4H, J = 7.6 Hz), 8.25 (s, 4H), 8.88–8.96 (m, 8H); 13C-NMR (150 MHz, CDCl3) δ ppm 35.7 (2C), 109.8 (6C), 112.4 (2Cq), 114.7 (2C), 119.0 (Cq), 119.9 (2Cq), 120.0 (4C), 120.2 (2Cq), 120.3 (4C), 121.5 (Cq), 123.4 (6C), 124.0 (2Cq), 125.9 (2C), 125.9 (6C), 126.4 (2Cq), 126.7 (2C), 126.7 (2C), 127.4 (8C), 127.7 (2C), 127.7 (2C), 127.9 (8C), 133.0 (Cq), 133.0 (Cq), 134.0 (Cq), 134.0 (Cq), 134.6 (2Cq), 135.0 (Cq), 136.6 (2Cq), 136.7 (Cq), 139.0 (2Cq), 140.8 (8Cq), 142.1 (2Cq), 145.2 (2Cq), 145.4 (2Cq); HRMS (APCI) calcd. for: C94H63N8S2 [M+H+] 1368.4645; found: 1368.4595.
5,15-diphenyl-15,20-bis(7-(4-formyl-phenyl)-10-methyl-10H-phenothiazin-3-yl)-21,23H-porphyrin 15
Purification by column chromatography with DCM gave: purple powder; yield 50% (0.06 g); 1H-NMR (600 MHz, CDCl3) δ ppm -2.76 (s, 2H, NH), 3.72 (s, 6H), 7.11 (d, 2H, J = 8.0 Hz), 7.23 (d, 2H, J = 7.1 Hz), 7.57 (s, 2H), 7.60 (d, 2H, J = 8.5 Hz), 7.77–7.81 (m, 10H), 7.96 (d, 4H, J = 8.0 Hz), 8.04–8.05 (m, 4H), 8.22–8.24 (m, 4H), 8.86–8.93 (m, 8H), 10.06 (s, 2H, CHO); 13C-NMR (150 MHz, CDCl3) δ ppm 35.8 (2C), 112.5 (2Cq), 112.6 (2Cq), 114.6 (2Cq), 118.8 (Cq), 120.3 (2Cq), 124.0 (Cq), 126.1 (2C), 126.7 (2C), 126.7 (2C), 126.9 (6C), 127.0 (2C), 127.2 (2Cq), 127.7 (2C), 130.4 (4C), 130.4 (2C), 133.0 (2C), 134.0 (2C), 134.2 (2C), 134.5(4C), 134.9 (4C), 136.8 (2C), 142.0 (2Cq), 144.9 (4Cq), 145.8 (4Cq), 146.2 (4Cq), 175.6 (4Cq), 191.8 (2CHO); HRMS (APCI) calcd. for: C72H49N6O2S2 [M+H+] 1093.3352; found: 1093.3356.
5,10,15,20-tetrakis-[4-(3-formyl-10-methyl-10Hphenothiazin-7-yl)phenyl]-21,23H-porphyrin 17
A mixture of 0.1 g BrTPP, 0.23 g 3-formyl-10-methyl-10Hphenothiazin-7-yl boronic pinacolate, 0.025 g Pd(PPh3)2Cl2, 0.12 g K2CO3, 30 mL DME and 15 mL H2O was refluxed for 18 h. Purification by column chromatography with DCM gave a purple powder; yield 64% (0.1 g); 1H-NMR (600 MHz, CDCl3) δ ppm –2.72 (s, 2H, NH), 3.58 (s, 12H), 6.97 (d, 4H, J = 8.2 Hz), 7.08 (d, 4H, J = 8.6 Hz), 7.18–7.20 (m, 4H), 7.72–7.77 (m, 12H), 7.95 (d, 8H, J = 7.9 Hz), 8.11–8.13 (m, 4H), 8.28–8.31 (m, 4H), 8.85–8.95 (m, 8H), 9.87 (s, 4H, CHO); HRMS (APCI) calcd. for: C100H67N8O4S4 [M+H+] 1572.4196; found: 1572.3790.
5,10,15,20-tetrakis-(4′-(9H-carbazole-9yl)-[1,1′-biphenyl]-4yl)-21,23H-porphyrin 18
A mixture of 0.1 g BrTPP, 0.17 g (4-(9H-carbazol-9-yl)phenyl)boronic acid, 0.025 g Pd(PPh3)2Cl2, 0.12 g K2CO3, 30 mL DME and 15 mL H2O was refluxed for 18 h. Purification by column chromatography with DCM gave a purple powder; yield 65% (0.1 g); 1H-NMR (600 MHz, CDCl3) δ ppm -2.70 (s, 2H, NH), 7.38 (t, 6H, J = 7.2 Hz, J = 7.4 Hz), 7.53 (t, 6H, J = 7.5 Hz, J = 7.5 Hz), 7.62 (d, 4H, J = 8.1 Hz), 7.83 (d, 8H, J = 7.8 Hz), 7.91–7.94 (m, 6H), 8.09–8.12 (m, 10H, H), 8.14 (d, 8H, J = 7.9 Hz), 8.24 (d, 8H, J = 7.8 Hz), 8.38 (d, 4H, J = 7.5 Hz), 8.87–8.91 (m, 8H), 9.03 (s, 4H); 13C-NMR (150 MHz, CDCl3) δ ppm 109.9 (8C), 120.1 (16Cq), 120.4 (8C), 123.5 (8C), 125.4 (4Cq), 126.0 (16C), 127.6 (8C), 128.7 (16C), 130.0 (8C), 135.2 (4Cq), 135.8 (4Cq), 135.8 (4Cq), 140.9 (12Cq); HRMS (APCI) calcd. for: C116H75N8 [M+H+] 1580.6142; found: 1580.6138.
5,10,15,20-tetrakis-(4′-formyl-[1,1′-biphenyl]-4yl)-21,23H-porphyrin 19
A mixture of 0.1 g BrTPP, 0.015 g 4-formylphenylboronic acid, 0.025 g Pd(PPh3)2Cl2, 0.12 g K2CO3, 30 mL DME and 15 mL H2O was refluxed for 18 h. Purification by column chromatography with DCM gave a purple powder; yield 48% (0.05 g); 1H-NMR (600 MHz, CDCl3) δ ppm 2.65 (s, 2H, NH), 8.09 (d, 8H, J = 7.6 Hz), 8.12–8.16 (m, 16H), 8.38 (d, 8H, J = 7.7 Hz), 8.98 (s, 8H), 10.19 (s, 4H, CHO); HRMS (APCI) calcd. for: C72H47N4O4 [M+H+] 1031.3591; found: 1031.3591.