Abstract
Metformin is the first-line treatment for many people with type 2 diabetes mellitus (T2DM) and gestational diabetes mellitus (GDM) to maintain glycaemic control. Recent evidence suggests metformin can cross the placenta during pregnancy, thereby exposing the fetus to high concentrations of metformin and potentially restricting placental and fetal growth. Offspring exposed to metformin during gestation are at increased risk of being born small for gestational age (SGA) and show signs of ‘catch up’ growth and obesity during childhood which increases their risk of future cardiometabolic diseases. The mechanisms by which metformin impacts on the fetal growth and long-term health of the offspring remain to be established. Metformin is associated with maternal vitamin B12 deficiency and antifolate like activity. Vitamin B12 and folate balance is vital for one carbon metabolism, which is essential for DNA methylation and purine/pyrimidine synthesis of nucleic acids. Folate:vitamin B12 imbalance induced by metformin may lead to genomic instability and aberrant gene expression, thus promoting fetal programming. Mitochondrial aerobic respiration may also be affected, thereby inhibiting placental and fetal growth, and suppressing mammalian target of rapamycin (mTOR) activity for cellular nutrient transport. Vitamin supplementation, before or during metformin treatment in pregnancy, could be a promising strategy to improve maternal vitamin B12 and folate levels and reduce the incidence of SGA births and childhood obesity. Heterogeneous diagnostic and screening criteria for GDM and the transient nature of nutrient biomarkers have led to inconsistencies in clinical study designs to investigate the effects of metformin on folate:vitamin B12 balance and child development. As rates of diabetes in pregnancy continue to escalate, more women are likely to be prescribed metformin; thus, it is of paramount importance to improve our understanding of metformin’s transgenerational effects to develop prophylactic strategies for the prevention of adverse fetal outcomes.
Keywords:
metformin; diabetes; placenta; folate; vitamin B12; one carbon metabolism; fetal growth; LGA; SGA; fetal programming 1. Introduction
Diabetes and its effect on fetal health are significant to the developmental origins of health and disease (DOHaD) hypothesis. Globally, around 223 million women currently live with diabetes, 60 million of whom are of reproductive age [1]. In addition to pre-existing diabetes, gestational diabetes mellitus (GDM), a form of maternal diabetes typically first diagnosed during weeks 24–28 of pregnancy, currently affects around 1 in 6 births worldwide, equating to approximately 16.8 million pregnancies [1,2,3]. The diagnostic criteria for GDM vary widely in different countries and in turn have led to heterogeneity in screening and trial designs, making it difficult for comparative judgement and unified consensus on its effect on maternal and fetal health [4]. If maternal hyperglycaemia is poorly controlled, this accelerates intrauterine growth and increases the risk of macrosomia, in which birth weight is > 4 kg, or the fetus being born large for gestational age (LGA), in which birth weight is above the 90th percentile. This may cause birth trauma for mother and baby by increasing the risk of preeclampsia, neonatal hypoglycaemia, shoulder dystocia, late stillbirth, or the need for caesarean section or neonatal intensive care [5,6,7]. Although GDM ceases post-parturition, these women are predisposed to an estimated sevenfold increased risk of type 2 diabetes mellitus (T2DM) within 5–10 years post-pregnancy [8,9,10]. Diabetes currently represents 10% of the National Health Service budget and, with the ever increasing prevalence of diabetic pregnancies, including diabetic risk factors such as the obesity epidemic and advanced maternal age [11,12], it is now paramount to refine diagnostic and treatment strategies to improve outcomes for mother and baby.
Insulin therapy is a standard treatment for diabetes to restore glucose homeostasis; however, this therapy is associated with increased maternal weight gain and hypoglycaemia. As rates of diabetes continue to rise, the cost, storage, and administration requirements for insulin have proven to be of increasing concern [13,14], particularly in developing countries where these storage requirements may not be feasible [14]. As such, metformin has been advanced as an alternative first-line therapy for T2DM and GDM in many countries [15,16].
2. Metformin in Pregnancy
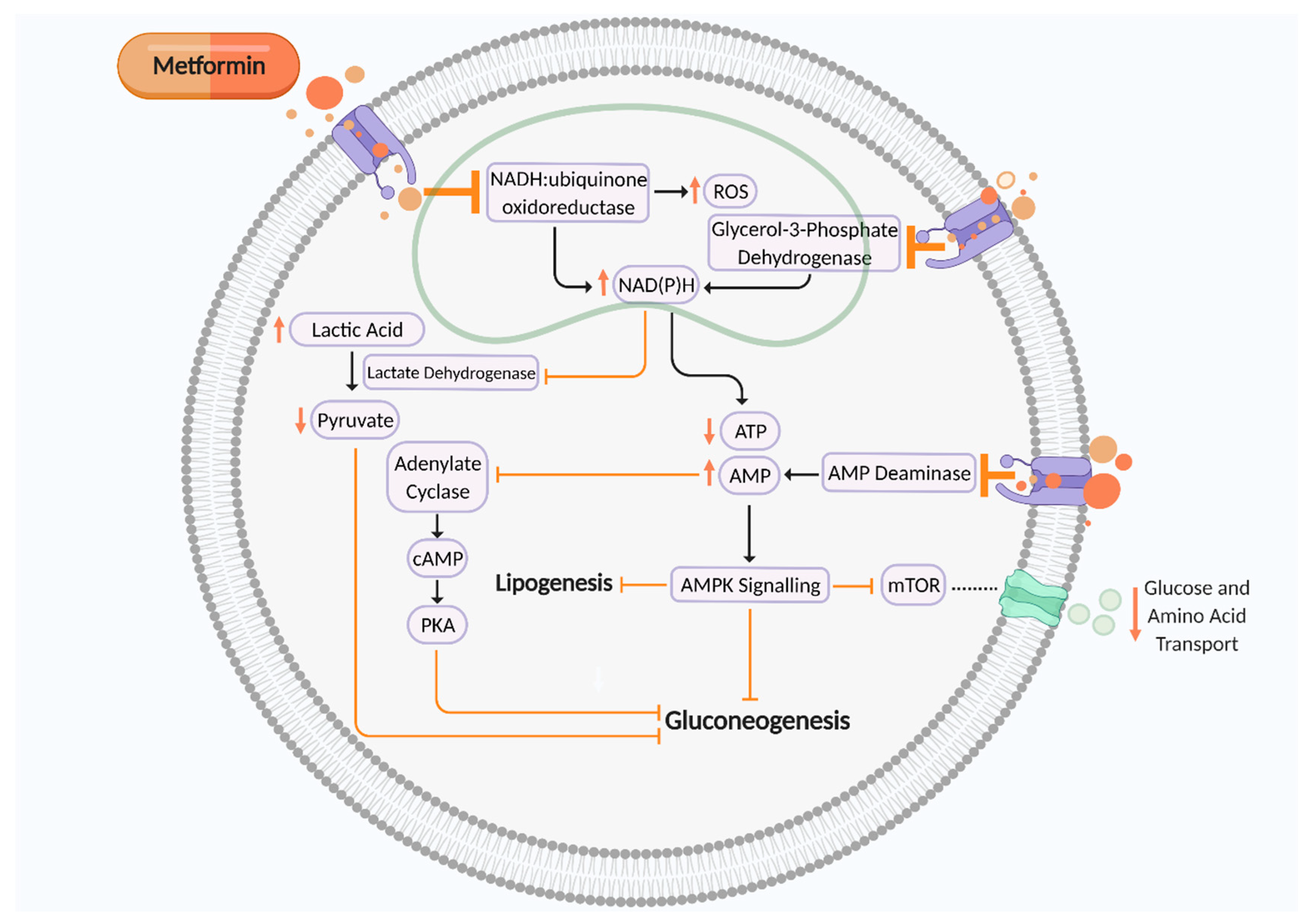
Metformin is an oral synthetic guanidine analogue known as a ‘glucophage’ due to its glucose-lowering abilities by reducing gluconeogenesis and insulin resistance [15,16]. Metformin is a mitochondrial complex I (NADH:ubiquinone oxidoreductase) inhibitor which is transported into the cell to directly influence cellular respiration (Figure 1). Complex I (NADH:ubiquinone oxidoreductase) oxidises NADH synthesised from one carbon metabolism, glycolysis, fatty acid β-oxidation, and the tricarboxylic acid (TCA) cycle for adenosine triphosphate (ATP) production via the electron transport chain [17,18,19]. Thus, metformin-induced suppression of complex I increases NADH accumulation and ROS production and reduces ATP synthesis, thereby elevating the AMP:ATP ratio. This activates AMP-activated protein kinase (AMPK) and leads to inhibition of gluconeogenesis, therefore maintaining glycaemic control [17]. Metformin can also reduce gluconeogenesis by inhibiting AMP deaminase, which further contributes to elevated cellular AMP levels, thus in turn inhibiting adenylate cyclase and cAMP–PKA signalling. Metformin-induced suppression of mitochondrial glycerol 3 phosphate dehydrogenase (G3PDH) also augments cytosolic NAD(P)H concentration, leading to reduced pyruvate levels and a suppression of gluconeogenesis. However, activation of AMPK signalling inhibits mammalian target of rapamycin (mTOR) activity, a nutrient sensor which regulates amino acid transport and glucose storage [17,20,21,22,23] (Figure 1). This mechanism of action leads to improved insulin sensitivity by augmenting insulin receptor tyrosine kinase activity, amplifying glycogenesis and suppressing glycogenolysis, inhibiting lipolysis, enhancing glucose transporter GLUT4 recruitment and activity, and suppressing the activity of hepatic glucose 6 phosphatase. Metformin also heightens insulin release due to enhanced glucagon-like peptide-1 (GLP-1) activity [24].
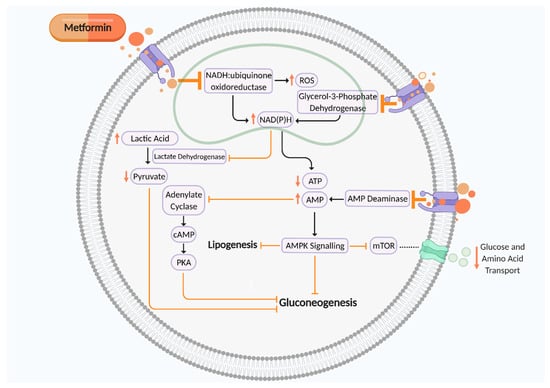
Figure 1.
Putative mechanism of action of metformin on cellular metabolism and mitochondrial aerobic respiration to suppress gluconeogenesis. Metformin is an inhibitor of mitochondrial complex I (NADH:ubiquinone oxidoreductase), AMP deaminase, and mitochondrial glycerol 3 phosphate dehydrogenase (G3PDH), which all contribute towards suppression of cellular gluconeogenesis to maintain glycaemic control. ROS, reactive oxygen species; NAD(P)H, nicotinamide adenine dinucleotide phosphate; ATP, adenosine triphosphate; AMP, adenosine monophosphate; cAMP, cyclic AMP; PKA, protein kinase A; mTOR, mammalian target of rapamycin. Black arrows indicate cellular pathway. Orange arrows indicate putative effects of metformin. Figure created using Biorender.com.
Metformin’s glucose-lowering activity certainly demonstrates beneficial outcomes for maternal health, as it decreases maternal weight gain, inflammation, atherothrombosis, and cardiovascular disease mortality, all of which are diabetic co-morbidities [15,25]. However, studies suggest that its short- and long-term effects on the metabolic health of the offspring may not be as favourable.
Whilst metformin therapy has been shown to significantly reduce the incidence of LGA, it has been reported that it may decrease birth weight to the extreme as metformin use in pregnancy is associated with an increased rate of small for gestational age (SGA) births; that is, those with a birth weight below the 10th percentile or two standard deviations below the mean weight for gestational age [5,7,26]. Notably, SGA offspring exposed to metformin in utero have shown signs of ‘catch-up growth’ during childhood. In the Metformin in Gestational Diabetes: The Offspring Follow Up (MiG: TOFU) study, at two years of age, metformin-exposed offspring demonstrated higher subcutaneous adiposity and larger mid-upper arm circumferences and bicep and subscapular skinfolds than insulin-exposed offspring [27]. By nine years of age, they presented with significantly higher body mass index (BMI) and larger arm and waist circumferences, triceps skinfolds, and abdominal fat volumes compared to insulin-exposed offspring [28]. A follow-up study of children exposed to metformin in utero in pregnancy complicated by polycystic ovarian syndrome also revealed they had higher BMIs at four years old than placebo-treated pregnancies [29]. Another randomised controlled trial showed that infants exposed to metformin during GDM pregnancy were markedly heavier at 12 and 18 months of age compared to insulin-exposed infants [30]. A murine study examining the effects of gestational metformin exposure from days E0.5 to E17.5 also showed that dams exposed to metformin manifested lower fetal weight on E18.5 than untreated dams. When fed a high-fat diet later in development, metformin-exposed fetuses were heavier than untreated fetuses and demonstrated increased mesenteric fat and liver weight. These findings, combined with gene set enrichment analysis of differentially expressed genes in the metformin and untreated murine offspring, reveal that metformin may induce transgenerational effects by way of fetal programming [31]. Accordingly, these studies suggest that metformin therapy in pregnancy may increase the risk of childhood obesity and thus is likely to predispose offspring to cardiometabolic diseases during adulthood. It is therefore crucial to develop our understanding of metformin’s mechanistic activity and its effects on the balance between maternal health and adverse fetal outcomes.
Studies from the literature around the effects of metformin treatment on the placenta suggest that metformin alters placental gene expression and function (Table 1), although the mechanisms remain unclear.

Table 1.
Current literature on the impact of metformin on placental gene expression and function [32,33,34,35,36,37,38,39,40,41,42,43].
2.1. Transplacental Transport of Metformin
As the interface between maternal and fetal circulations, the placenta transports nutrients to the developing fetus. There is also evidence that metformin is transported across the placenta to the fetal circulation. In metformin-exposed pregnancies, serum samples from umbilical cord, placental, and fetal tissues have demonstrated metformin concentrations to be equal or greater than maternal levels, suggesting active transport of metformin from the maternal circulation across the placenta and into fetal tissue [44,45,46]. Metformin is a hydrophilic cation, has a half-life of 5 h, and is not metabolised in humans, but recent evidence suggests that metformin bioavailability, volume of distribution, and clearance may be significantly increased in pregnancy, dependent on dose [47,48]. The mechanisms of how pregnancy alters metformin clearance remain to be established. Although metformin can cross the placenta, it is undetermined how metformin influences placental metabolism to influence gene expression and whether fetal tissues handle metformin in the same way.
The transporter responsible for metformin uptake from the maternal circulation into the placenta is yet to be determined and requires further research. Studies have reported norepinephrine transporter (NET), serotonin transporter (SERT), and organic cation transporter novel type 2 (OCTN2) to be localised on the maternal interface of the placenta at the syncytiotrophoblast apical membrane, which could be responsible [19,49,50,51]. OCT3 has been demonstrated to be the key transporter for fetal metformin uptake and distribution, localised on the fetal interface of the placenta at the syncytiotrophoblast basal membrane and fetal capillaries. Indeed, OCT3-/- pregnant mice show attenuated fetal metformin exposure [19]. However, it is apparent that placental OCT3 expression increases with gestational age, as a murine study demonstrated that placental OCT3 mRNA and protein expression increased by 37-fold and 56-fold, respectively, at day 15 of gestation, and by 46-fold and 128-fold, respectively, at day 19 [52]. Thus, it is possible that metformin may not be reaching fetal tissues with significant concentration until late gestation. Moreover, these findings suggest that, unlike insulin [13], metformin can cross the placenta [21,28] and reach fetal tissue, which could potentially influence fetal growth and programming.
2.2. Impact of Metformin on Placental Nutrient Transport and Nutrient Bioavailabilty
It has been demonstrated that metformin influences fetal growth and nutrient bioavailability by inhibiting mitochondrial complex I, leading to activated AMPK signalling and inhibition of placental mTOR signalling (Figure 1). Attenuated placental mTOR signalling is associated with restricted fetal growth [53]. This mechanism of action may potentially explain the significant relationship between SGA births and metformin exposure in pregnancy. mTOR is highly expressed in the human placenta syncytiotrophoblast layer and mTOR complex 1 (mTORC1) signalling plays a major role in placental nutrient sensing, thus significantly influencing fetal nutrient availability and metabolism. Trophoblast mTORC1 regulates System A and System L amino acid transporters for amino acid uptake, essential for fetal metabolism [54,55]. Preliminary in vitro models of human trophoblast cells with silenced mTORC1 have also demonstrated that placental mTORC1 may regulate a circulating factor or factors, which could influence fetal growth [55,56]. mTORC1 signalling is regulated by placental insulin and IGF I, and fetal glucose, amino acid, and oxygen levels. Diabetes may increase mTORC1 activation due to elevated maternal nutrient and ATP concentrations [54,55]. During early gestation, the embryo mainly expresses immature mitochondria. As gestation develops, the placenta and fetus increase their expression of mature mitochondria, which are more susceptible to metformin inhibition. With this in mind, it is possible metformin may not adversely affect offspring growth until after the first trimester [21].
Transplacental metformin exposure may restrict placental and fetal growth by reducing nutrient bioavailability which could influence fetal programming. Evidence suggests metformin can influence the status of several vitamins and micronutrients, including vitamins B1, B12, and D, folic acid, and magnesium [15,20,57,58,59,60,61,62]. Whilst all of these nutrients are important for fetal growth and development, folate and vitamin B12 are co-factors of one carbon metabolism, essential for cell growth, metabolism, and production of the methyl donor S-adenosyl-methionine (SAM). Furthermore, exposure to metformin and maternal deficiency of both folate and vitamin B12, during pregnancy, lead to similar changes to placental and fetal growth and offspring health [63,64,65,66,67]. We therefore postulated that a potential mechanism by which metformin influences placental and fetal growth and offspring risk of cardiometabolic complications is by affecting the balance between folate and vitamin B12 levels and, therefore, perturbing one carbon metabolism. This review now focuses on this and discusses the evidence that supports this hypothesis.
3. One Carbon Metabolism
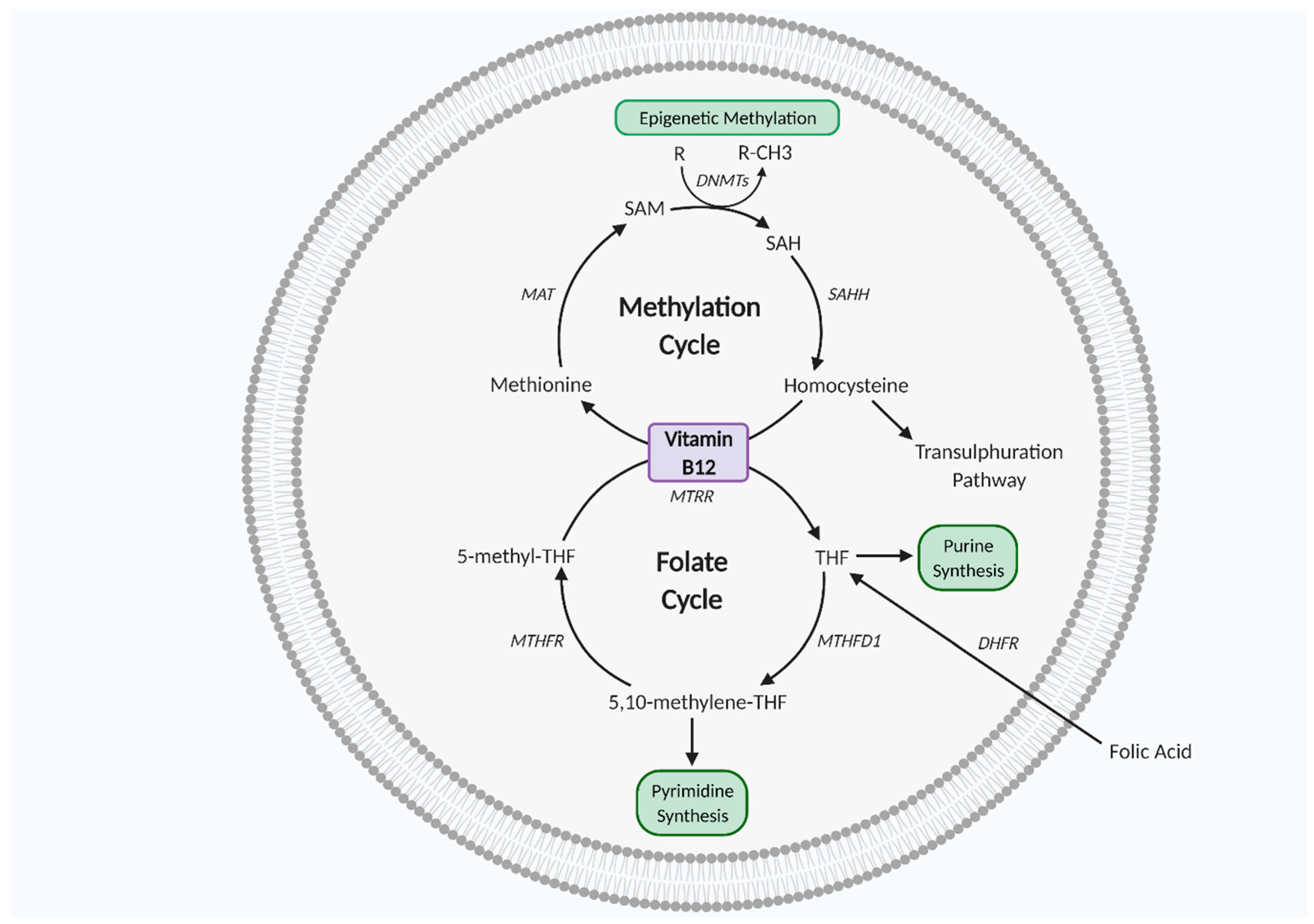
Vitamin B12 and folate work synergistically as co-factors for one carbon metabolism, a biochemical network of methylating reactions and one carbon atom transfer vital for biosynthesis of DNA, RNA, lipids, amino acids, and neurotransmitters [63,68]. One carbon metabolism is also key for histone protein methylation to regulate gene expression and methionine and purine/pyrimidine synthesis to regulate cell growth, proliferation, and differentiation [63,68,69,70,71]. As such, one carbon metabolism is essential for in utero fetal development. One carbon metabolism is particularly important in mitochondrial redox homeostasis, as folate-mediated NADPH production plays a role in redox defence, thereby protective against oxidative stress [72]. In the cell cytoplasm, the folate cycle and methylation cycle work synergistically for purine/pyrimidine, methionine amino acid, and SAM synthesis. SAM is the primary DNA methyl donor synthesised from methionine via methionine adenosyl transferase (MAT) in the methylation cycle and DNA methylation is a crucial regulatory modification that regulates gene expression. SAM is converted to S adenosyl homocysteine (SAH) and then homocysteine (Hcy), which is then followed by regeneration of methionine, which completes the cycle. Hcy conversion to methionine requires synergism with the folate cycle, as 5 methyl tetrahydrofolate (5-methyl-THF) donates its methyl group to Hcy. The folate cycle intersects methionine synthesis via THF production from 5-methyl-THF. THF is then converted to 5-10-methyl-THF, followed by 5-methyl-THF regeneration via methylenetetrahydrofolate reductase (MTHFR) [63,70,71] (Figure 2).
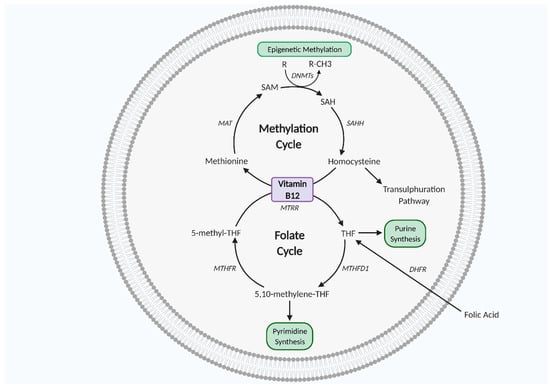
Figure 2.
One carbon metabolism. Vitamin B12 and folate are co-factors of the methylation and folate cycles which interlink to complete the one carbon metabolism, essential for cell proliferation, differentiation, and growth. DHFR, dihydrofolate reductase; DNMTs, DNA methyltransferase; MAT, methionine adenosyltransferase; MTHFD1, methylenetetrahydrofolate dehydrogenase 1; MTHFR, methylenetetrahydrofolate reductase; MTRR, methyltransferase reductase; SAH, S adenosyl L homocysteine; SAHH, adenosylhomocysteinase; SAM, S-adenosyl methionine; THF, tetrahydrofolate. Figure created using Biorender.com.
Folate is found in the diet from fruit and vegetables and can be supplemented with its synthetic form, known as folic acid [68,69]. Folate deficiency elevates Hcy levels. High Hcy concentration is strongly associated with cardiovascular disease and may promote aberrant placental development, as in vitro trophoblast exposure to high Hcy leads to increased apoptosis and decreased human chorionic gonadotropin secretion [73,74]. Vitamin B12 is also a co-factor in one carbon metabolism [63,75]. Vitamin B12 is found in animal-derived foods and is essential for genomic stability and cellular metabolism, key for neurological and haematological developments, which have high cellular turnover rates [76]. Vitamin B12 deficiency inhibits methionine regeneration in one carbon metabolism and leads to Hcy and 5-methyl-THF accumulation [63,75] (Figure 2).
4. One Carbon Metabolism in Pregnancy
The one carbon metabolism cycle is critical for normal growth and development; hence, vitamin B12 and folate are essential micronutrients required for a successful pregnancy.
4.1. Vitamin B12
Heterogeneity exists in the reference ranges used by clinical studies for what is defined as ‘normal’, ‘low’, and ‘deficient’ levels of vitamin B12, as there is no gold standard criteria, thereby making direct comparisons between study results challenging [57]. However, most laboratories tend to use the following definitions of total serum B12 levels: normal (≥250 pmol/L), low (150–249 pmol/L), and acute deficiency (<149 pmol/L) [77].
Vitamin B12 deficiency has been associated with pernicious anaemia, neural tube defects, aberrant neurological development, insulin resistance, and, paradoxically, an increased risk of GDM [76,78,79,80,81,82]. Deficiency in utero, a crucial developmental window, increases the risk of intrauterine growth restriction and has been associated with increased risk of pre-term birth, a key driver of neonatal death and low birth weight or SGA [67,82,83]. This preventable risk is of particular concern in Asian women among whom SGA births are common, as diet is predominantly vegetarian and therefore low in vitamin B12 [84].
4.2. Folate
As defined by the World Health Organisation (WHO), folate deficiency is predicated on Hcy levels and marked as <10 nmol/L serum folate or <340nmol/L RBC folate [85]. Although plasma folate levels are highly influenced by transient diet and folate digestion and metabolism, RBC folate measurement is a good indicator of long-term folate status and has been shown to correlate well with tissue stores [69,86].
Low folate status is significantly associated with cognitive decline [87] and neural tube defects (NTDs) during gestation [88]. Low folate bioavailability is also associated with increased risk of spontaneous abortion and stillbirth [89]. A prospective study of UK pregnant adolescents has revealed that poor folate status is associated with increased prevalence of SGA births [66]; conversely, a positive relationship between RBC folate concentration and birth weight is supported by systematic reviews and meta-analyses in the literature [90,91]. Indeed, poor folate levels in pregnant teenagers have been associated with impaired placental trophoblast cell turnover and system A amino acid transport, demonstrating that low folate restricts placental growth and reduces nutrient transport to the fetus [65].
Low vitamin B12 and folate and perturbed one carbon metabolism have been associated with altered placental gene expression (Table 2) and thus may have a role in fetal programming [92,93,94,95,96]. The adverse impacts of vitamin B12 and folate deficiency on fetal development and birth weight and the similarities of these effects to in utero metformin exposure suggests there may be a link between metformin activity and alterations in the bioavailability or actions of these essential micronutrients.

Table 2.
Effects of folate and vitamin B12 status on the placenta [65,73,97,98,99,100,101,102,103,104,105,106,107].
5. Is Metformin Impacting on Fetal and Placental Development by Perturbing the One Carbon Metabolism Cycle?
5.1. Vitamin B12
It has been long reported that metformin may promote disturbances in vitamin B12 intestinal absorption [108]. Follow-up analysis shows metformin exposure promotes vitamin B12 malabsorption in 10–30% of people [58] and 30% of people may potentially experience deficiency [61,109]. Moreover, a meta-analysis review of 29 studies including a total of 8089 participants revealed people taking metformin were at significantly higher risk of developing vitamin B12 deficiency or insufficiency [110]. Women with GDM taking metformin have demonstrated low total vitamin B12 stores compared to those taking insulin [111]. Vitamin B12 insufficiency is associated with elevated Hcy levels, a phenomenon observed in women with polycystic ovarian syndrome who were taking metformin for 6 months, and also in people at risk of developing T2DM who were taking metformin for 10 years, thus demonstrating that metformin induces vitamin B12 deficiency at the tissue level [58,112].
The underlying pathophysiology of vitamin B12 malabsorption under metformin exposure has not yet been determined. Proposed theories suggest metformin promotes intestinal mobility disorders leading to bacterial overgrowth [113] and/or it may alter intrinsic factor secretion [114]. However, perhaps the most widely accepted theory is that metformin displaces calcium in the ileal surface membrane, leading to disruption in intestinal calcium-dependent vitamin B12 intrinsic factor uptake [115]. Theories on placental metformin vitamin B12 malabsorption are sparse. Several studies have demonstrated vitamin B12 receptors to be expressed in the placenta [116,117,118], which enter the endosomal lysosomal system upon ligand binding. Vitamin B12 is transported through the placenta and the fetal circulation by binding to transcobalamin (TC) proteins I, II, and III in blood [119]. TCs are usually produced in the liver; however, it has been shown that the placenta itself may produce TCs from early gestation [120,121], further demonstrating that vitamin B12 is metabolised by the placenta. As metformin therapy is associated with reduced holoTC levels [59], future studies investigating the effects of metformin on placental and fetal TC concentration may prove useful to determine whether metformin reduces placental and fetal vitamin B12 status by downregulating TC proteins. Pregnant rat models have also demonstrated that transplacental vitamin B12 transport to the fetus increases throughout gestation and placental vitamin B12 levels are consistently higher than maternal plasma and fetal tissue levels at every gestational stage [122]. This finding suggests that vitamin B12 may become increasingly important for fetal and placental growth throughout gestation and metformin-induced reductions in maternal vitamin B12 levels may have a more profound effect during late gestation.
It has been proposed that the placenta can adapt to low vitamin B12 status by upregulating angiogenesis-related genes to increase surface area for fetoplacental nutrient transport. This effect has exclusively been associated with female births. Indeed, placental endoglin (ENG) and vascular endothelial growth factor (VEGF) expression from female SGA and appropriate for gestational age (AGA) births were significantly negatively associated with maternal vitamin B12 status measured during the first trimester [97]. Vitamin B12 status may therefore induce fetal gender specific changes in placental gene expression which could potentially differentially impact fetal programming in male and female births. Controversy remains as to whether metformin promotes angiogenesis in all tissues [123,124,125,126]; however, evidence suggests that metformin may have pro-angiogenic effects in the placenta [37]. Future studies are needed to investigate whether metformin induces similar methylation patterns in placental angiogenesis-related genes as does low vitamin B12 concentration. By finding homologies between the pathogenesis of vitamin B12 deficiency and metformin, this will provide us with a better understanding of metformin’s mechanism of action in evoking adverse transgenerational effects on offspring through fetal programming.
Vitamin B12 deficiency in pregnancy has been associated with increased offspring and maternal metabolic risk due to altered lipid profile. Genome-wide and targeted DNA methylation analysis has shown that vitamin B12 deficiency in cultured human adipocytes leads to hypomethylation of the promoter regions of genes related to cholesterol biosynthesis, low density lipoprotein receptor (LDLR) and sterol regulatory element binding protein 1 (SREBF1), leading to an increase in their expression [127]. Adaikalakoteswari et al. 2017 have also reported that human preadipocyte cell lines exposed to insufficient concentrations of vitamin B12 promotes altered expression of 12 miRNAs associated with adipocyte function and differentiation. This finding was reflected in blood samples of pregnant women with low vitamin B12 status, thereby suggesting vitamin B12 deficiency may increase the risk of maternal insulin resistance and obesity [128]. Perhaps this phenomenon may be reflected in the developing offspring by way of fetal programming. Indeed, maternal vitamin B12 deficiency in Wistar rat models has shown offspring to have adipocyte dysfunction, increased adiposity, and altered lipid metabolism, where they display increased levels of total cholesterol, triglycerides, IL 6, TNF-α, and leptin and reduced levels of adiponectin and IL-1β [129]. Henderson et al. 2018 also showed murine offspring of vitamin B12-deficient mothers supplemented with folic acid have higher adiposity and reduced β cell mass and proliferation. These findings are also demonstrated in children exposed to vitamin B12 deficiency during gestation [70]. Whether metformin therapy induces similar changes in lipid profile, promoter methylation, and miRNA expression, and if these effects extend to the fetus, is currently unknown. Nonetheless, a recent study including 87 women found no significant associations between vitamin B12 measurements at second and third trimesters of pregnancy and insulin resistance, infant weight, and placental weight, respectively [130].
The dose of metformin given to pregnant women varies between 500 and 3000 mg/day [24]; thus, the duration and dose of treatment may significantly influence vitamin B12 status in metformin users (Table 3). As metformin can cross the placenta [21], offspring exposed to higher or longer cumulative doses of metformin could be at increased risk of developing vitamin B12 deficiency, which may lead to pre-term birth and SGA. Routine screening for vitamin B12 deficiency may therefore need to be considered in metformin-exposed pregnancies in women with T2DM and GDM.

Table 3.
Rates of vitamin B12 deficiency in patients treated with metformin [57,58,59,111,131,132,133].
5.2. Folate
It has been demonstrated that metformin therapy reduces plasma and red blood cell (RBC) folate concentration and increases Hcy levels (Table 4).

Table 4.
Impact of metformin on folate, vitamin B12, and Hcy levels [58,69,112,140,141,142,143].
Notably, metabolomic fingerprinting of breast cancer cells has shown that metformin may have tumour suppressor effects by mimicking antifolate activity. Breast cancer cells exposed to metformin demonstrated accumulation of ‘trapped’ 5-formimino-tetrahydrofolate (THF), a folate metabolite and intermediate of one carbon metabolism. Folate-dependent target proteins were also inhibited under metformin exposure, leading to disturbed one carbon metabolism and reduced de novo purine/pyrimidine synthesis [62]. This suggests that metformin exposure in utero may impair fetal growth. However, a prospective cohort study of 336 pregnancies with first trimester metformin exposure revealed no increased risk of major birth defects and spontaneous abortions [134]. Regardless, screening of maternal folate should be considered as part of the therapeutic regimen for metformin-exposed pregnancies to elucidate any associations between placental development and fetal outcomes.
In vitro studies of folate deficient human cytotrophoblast cells show reduced mTOR signalling and amino acid transport, both of which are hallmarks of SGA placentas [103,104,135]. In vivo murine models have also demonstrated that maternal folate deficiency leads to lower fetal weight, reduced placental mTOR signalling and reduced systems A and L amino acid transporter activity [107]. Low folate bioavailability may therefore restrict fetal growth by reducing mTOR activity and placental nutrient transport. Immunohistological analysis has demonstrated reduced mTORC1 phosphorylation signalling in human intrauterine growth restriction (IUGR) placentas compared to appropriate for gestational age (AGA) placentas [136]. Reduced mTOR signalling and expression have also been observed in IUGR cytotrophoblast cells compared to normal term pregnancies [137,138]. Although these studies measured different mTOR phosphorylation targets for signalling activity analysis, both demonstrated downregulated mTOR signalling in association with IUGR. As IUGR- and metformin-exposed pregnancies are characterised by low folate status and similar outcomes as SGA births, these results suggest that metformin may promote aberrant mTOR signalling and nutrient sensing. mTORC1 signalling is regulated via methionine-induced activation from one carbon metabolism. A protein known as SAMTOR binds to SAM in response to methionine levels, which in turn regulates mTORC1 signalling [139]. Whether SAMTOR is affected by folate levels is unknown.
How metformin induces low folate status is yet to be established. Placental folate transport is mediated through folate receptor α (FRα/FOLR1), reduced folate carrier (RFC), and the proton-coupled folate transporter (PCFT/HCPI). FRα and PCFT have been found to be expressed on the syncytiotrophoblast microvillous plasma membrane (MVM) at both first trimester and term placenta. At first trimester, RFC is localised at the MVM and cytotrophoblast plasma membrane and, by term, is found on the MVM and basal plasma membranes of the syncytiotrophoblast [144]. The presence of these transporters in first trimester placenta, as well as FRα knockout shown to be embryonically lethal [145], demonstrate that folate transport is essential for fetal growth from early gestation. Folate transporter protein expression has been shown to be reduced in placentas of SGA offspring [146]. Thus, future studies investigating the interaction of metformin with folate transporters would prove interesting to explore whether metformin may be competing with folate or downregulating transporter expression.
6. Is Metformin Influencing Fetal Programming by Disturbing One Carbon Metabolism?
Maternal folate status has the capacity to influence fetal programming via placental gene expression, which could potentially lead to transgenerational epigenetic inheritance [147]. miRNA array analysis has revealed that folate-sensitive placental microRNAs (miRNAs), miR-222 3p, miR-141 3p, and miR-34b 5p, were downregulated with low maternal folate levels [65]. As metformin induces low maternal folate status, these findings suggest metformin may potentially be targeting the same miRNAs to reduce placental cell turnover and cause placental dysfunction, thereby altering fetal nutrient transport and predisposing offspring to increased disease susceptibility. Indeed, offspring with isolated NTDs, a condition strongly associated with low folate bioavailability, exhibit low placental weight, placental hypermaturity, and pathological oedema in terminal villi compared to offspring without congenital anomalies [148].
In utero development is a critical stage for cell differentiation and proliferation and requires a high turnover rate for growth. Limited bioavailability of methyl donors such as folate and vitamin B12 during this period may promote DNA hypomethylation and thus drive aberrant epigenetic and post-translational regulation by inducing differential gene and promoter methylation patterns. Post-translational methylation of N6-methyladenosine (m6A) on mRNA and primary miRNA transcripts plays a role in gene regulation and may also be altered with limited methyl donor bioavailability [149,150]. m6A methylation requires SAM as a methyl donor and therefore would be impacted by aberrant one carbon metabolism. Low maternal folate and vitamin B12 status in one carbon metabolism may promote genomic instability (Figure 3). Indeed, deficiency of both folate and vitamin B12 results in uracil retention and misincorporation in DNA synthesis rather than thymine, leading to aberrant base pair bonding with adenine, thereby inducing chromosome breaks [151]. Impaired DNA synthesis in utero may compromise placental and fetal development and increase the risk of fetal programming and disease susceptibility in offspring. This was observed by Zheng et al., who demonstrated that a reduction in mitochondrial DNA copy number was associated with insulin resistance in obese people [152].
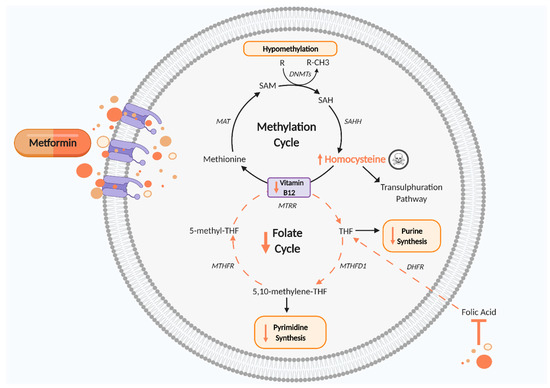
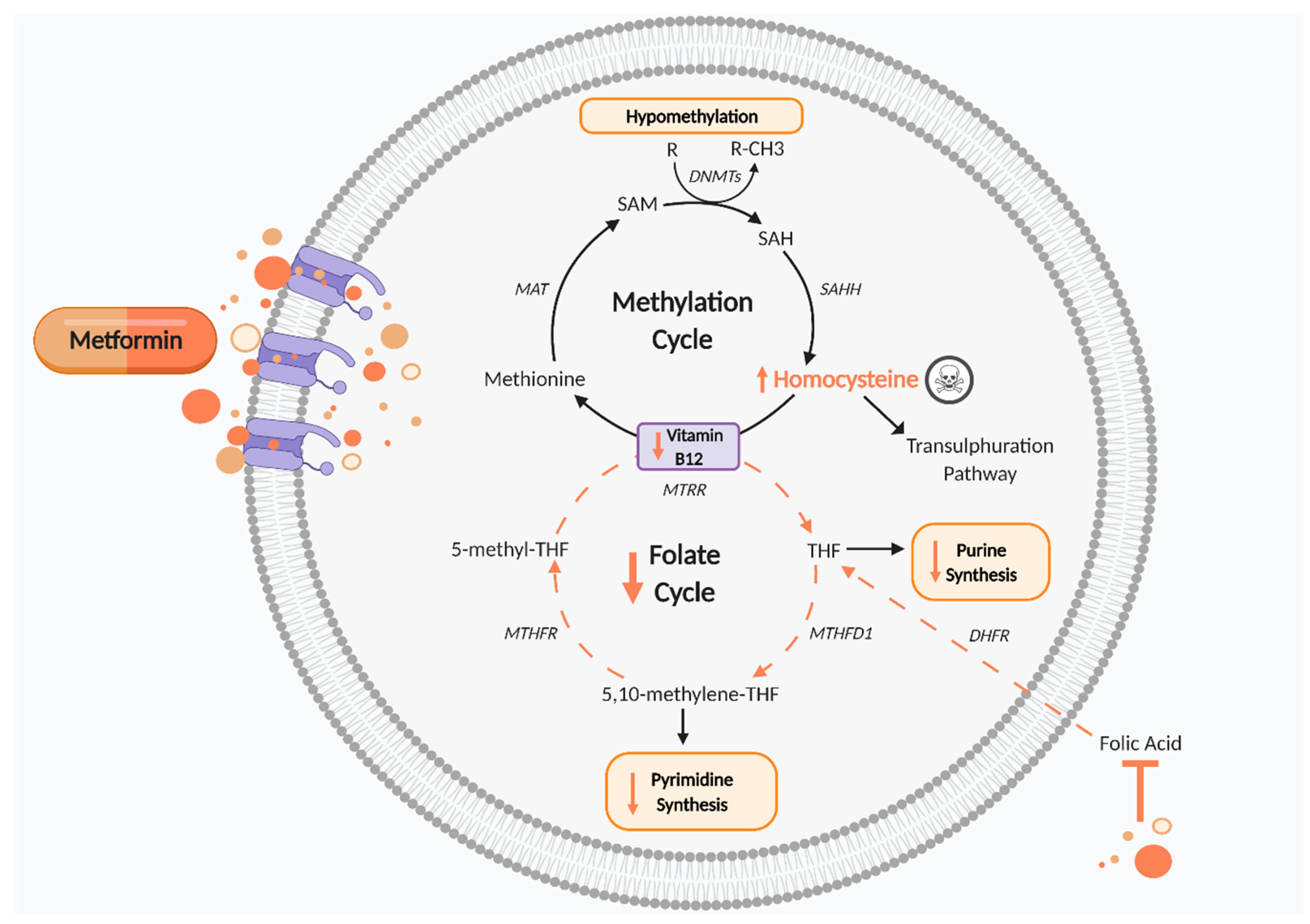
Figure 3.
Disturbed one carbon metabolism induced by metformin. Metformin reduces vitamin B12, thus impairing the methylation cycle and leading to increased Hcy levels, which are cytotoxic, and hypomethylation of proteins and nucleic acids, which may cause epigenetic changes. The folate cycle is also disturbed by metformin’s antifolate-like activity, thereby reducing pyrimidine and purine synthesis and disrupting cell growth and proliferation. DHFR, dihydrofolate reductase; DNMTs, DNA methyltransferase; MAT, methionine adenosyltransferase; MTHFD1, methylenetetrahydrofolate dehydrogenase 1; MTHFR, methylenetetrahydrofolate reductase; MTRR, methyltransferase reductase; SAH, S adenosyl L homocysteine; SAHH, adenosylhomocysteinase; SAM, S-adenosyl methionine; THF, tetrahydrofolate. Black arrows indicate cellular pathway. Orange arrows indicate putative effects of metformin. Figure created using Biorender.com.
Nicotinamide N-methyltransferase (NNMT) is an enzyme that ties one carbon metabolism with the methylation balance and nicotinamide adenine dinucleotide (NAD+) levels of the cell by catalysing nicotinamide (NAM) methylation via SAM. Elevated NNMT expression in murine white adipose tissue (WAT) has been associated with obesity and insulin resistance, whereas WAT NNMT knockout was protective [153,154]. It is already known that the dose of metformin may differentially affect cellular NAD+ levels [155]; however, whether metformin impacts NNMT expression requires further investigation.
Fetal programming induced by post-translational mechanisms from early life suboptimal nutrition has been demonstrated in adipose tissue of prediabetic adult rats and in human adults who were born SGA. It has been reported that, prior to manifestation of metabolic disease, miRNA-483-3p in adipose tissue is upregulated by suboptimal early life nutrition [156]. This miRNA is in intron 2 of the IGF2 gene and represses growth differentiation factor 3 (GDF3) expression, a protein from the transforming growth factor beta (TGFβ) superfamily that is key for lipid accumulation and adipocyte differentiation. Thus, downregulation of GDF3 may limit lipid storage and stimulate lipotoxicity and ectopic triglyceride storage, thereby promoting insulin resistance. Conservation of miRNA-483-3p upregulation in both SGA adult humans and rat suboptimal early life nutrition raises the question of whether this miRNA could be a biomarker for future metabolic syndrome risk [156]. Establishing whether metformin induces hypomethylation of IGF2, which leads to upregulated miRNA-483-3p and repressed GDF3, may give further insight into the possible mechanisms of metformin’s transgenerational programming capacity.
miRNA-483-3p upregulation has also been identified in maternal, placental, and fetal tissues of C57BL/6 mice deficient in both folate and vitamin B12 [105]. The same findings were true for miR-221 [105], an miRNA which has been shown to promote cardiovascular intimal thickening in diabetes [157]. Folate and vitamin B12 deficiency also led to downregulation of miR-133 exclusively in fetal tissues; however, this was tissue- and sex-specific [105]. Repression of miR-133 is a phenomenon associated with cardiomyopathy in diabetes; therefore, folate and vitamin B12 insufficiency in early life could be a risk factor for future disease susceptibility of the offspring. Global DNA methylation was reduced in maternal tissues with deficient levels of folate and vitamin B12 but was increased in fetal tissues. DNA methyltransferases DNMT1, DNMT3A, and DNMT3B were also increased in both maternal and fetal tissues. This strongly represents the significance of folate and vitamin B12 as methyl donors and their influence on gene regulation. Interestingly, RFC, PCFT, and FRα mRNA expression were upregulated in maternal, placenta, and fetal tissues with both folate and vitamin B12 insufficiency [105]. If metformin emulates these changes in epigenetic regulation, as shown by deficient folate and vitamin B12 levels, this will provide further evidence of the consequential downstream effects of metformin’s suppressive activity on methyl donors.
There is a considerable gap in the literature on the potential role of metformin in causing one carbon metabolism disturbance in the placenta and fetus and its impact on fetal programming. Future studies are needed to determine exactly how metformin exerts these disturbances, be it through directly decreasing vitamin B12 and/or folate levels, at a post translational level through miRNA regulation, or by directly targeting one carbon metabolism enzymatic action.
7. Vitamin Supplementation
Vitamin B12 and folate/folic acid supplementation may be an effective prophylactic approach to reduce the adverse effects of metformin on offspring. A cross-sectional study of people with diabetes taking metformin found that vitamin B12 deficiency was significantly decreased in those taking multivitamin supplements. As most multivitamin supplements include 6 to 25 µg of vitamin B12, this dose range may be sufficient for a protective effect for the fetus [60]. Another cross-sectional study of people with T2DM on metformin found multivitamin supplementation was associated with a marked decrease in vitamin B12 deficiency and lower risk of borderline deficiency [57]. The 1999–2006 National Health and Nutrition Examination Survey observed that a daily vitamin B12 dose over 0.6 µg reduced vitamin B12 deficiency and borderline deficiency by two-thirds in the general population, but this dose was not protective in people with T2DM who were taking metformin [61]. More clinical trials are needed to evaluate the protective dose of vitamin B12 supplementation in metformin users, particularly during pregnancy. Follow-up studies of offspring exposed to metformin during gestation and maternal vitamin B12 supplementation are also a priority to improve understanding of the long-term effects of vitamin supplementation on fetal programming. In a rat IUGR model, it has been reported that feeding the first generation (F1) offspring who have metabolic disease with a methyl donor-rich diet leads to prevention of metabolic disease being passed down to the F2 generation [158]. This suggests that postnatal dietary interventions may reverse the epigenetic effects of fetal programming in IUGR offspring and, therefore, postnatal strategies could also be applied to metformin-exposed pregnancies. Although not including people taking metformin, there are two on-going trials currently investigating the effects of vitamin B12 supplementation, pre-conceptually or during early pregnancy, on child development and the risk of diabetes [82,159]; the results of these studies have yet to be published but it will be important to monitor these to assess the potential beneficial effect of pregnancy supplementation.
Folate or folic acid supplementation should also be considered as part of the therapeutic regimen for all metformin-exposed pregnancies. Currently, women with T2DM are advised to take pre-conceptual 5 mg/day of folic acid until 12 weeks gestation, which is more than the normally recommended peri-conceptual dose of 400 µg/day folic acid for non-diabetic women [160,161]. Policies of folate fortification in flour and cereal grain products have already been adopted in many countries to increase the peri-conceptual or early gestational folate exposure of the population. This has shown a reduction in the rate of NTDs and improvement in offspring cognitive function and bone mineral content and density [70,162,163]. This may be of particular importance in early-stage unplanned pregnancies. Indeed, high RBC folate concentration at 28 weeks gestation has been associated with higher birth weight [164], thus suggesting a possible clinical screening time frame and therapeutic window for intervention and maximum protection. However, as it is hard to monitor the intake of folate fortification in the general population, with some subjects also taking additional vitamin supplementation, there is a danger of excess consumption. This may promote adverse health outcomes, such as childhood insulin resistance, childhood asthma, aberrant child neurocognitive development, NK cytotoxicity suppression, and possible progression of premalignant and malignant lesions [162,163,165,166]. Pregnant mice supplemented with excess folic acid have demonstrated embryonic delay, embryonic growth retardation, and thinner embryonic ventricular walls [167]. Risks of folate/folic acid over-supplementation should therefore be considered when treating people with metformin. It has recently been reported that a high choline concentration in Wistar rat gestational diets may mitigate the negative effects of high folate levels on fetal programming in male offspring [168]. This finding may represent a potentially promising therapeutic avenue to be explored in humans who are at risk of folate/folic acid over-supplementation.
Balance and supplementation of both folate and vitamin B12 should be tightly controlled as studies have shown that high maternal folate and low maternal vitamin B12 levels are associated with offspring insulin resistance [163,169]. A murine study investigating the effects of maternal vitamin B12 deficiency and folic acid supplementation demonstrated that offspring from vitamin B12-deficient mothers showed glucose intolerance, fasting hyperglyceamia, and lower β cell mass. Offspring diet also influenced plasma insulin and fasting glucose levels [70]. These findings illustrate how offspring lifestyle and diet may influence disease susceptibility and could be additive ‘hits’ to the fetal reprogramming already induced by maternal stimuli.
8. Future Considerations
Further research is needed to investigate the interaction between metformin and one carbon metabolism. Establishing if metformin directly targets one carbon metabolism enzymatic action or directly targets the cellular levels of both vitamin B12 and folate would provide insight into the causal relationship between metformin and its suppressive influence on one carbon metabolism. Deficient folate and vitamin B12 levels have been shown to promote epigenetic changes in gene methylation patterns and miRNA expression of placental and metabolic genes, which could lead to fetal programming. As such, future research would benefit from exploring the role of metformin in fetal programming by investigating its epigenetic effects in placental, umbilical cord, and fetal tissues. Establishing similarities in gene regulation, demonstrated by both deficient folate and vitamin B12 status, and metformin therapy may provide further insight into metformin’s mechanism of action, which would aid therapeutic innovation strategies.
Emerging studies suggest that another potential mechanism by which metformin-induced impairment of one carbon metabolism may influence events in the placenta and fetus is via regulation of mitochondrial function. Yang et al. 2020 reported a new role for one carbon metabolism, in which it may be linked to mitochondrial respiration via NADH production of serine catabolism [18], whilst Boachie et al. 2021 demonstrated that B12 deficiency impaired mitochondrial respiration [170]. These findings, together with evidence of placental mitochondrial dysfunction in GDM pregnancy [171], suggest that further studies are needed to elucidate whether metformin-mediated B12/folate deficiency exerts a similar impact on mitochondrial function in diabetic pregnancy. However, one carbon metabolism is compartmentalised in the cell, where the cytosol and mitochondria have their own independent one carbon machinery. This may be important to consider when investigating the effects of metformin and whether it predominantly acts at mitochondria [172].
Studying the effects of metformin on placental and fetal growth in women with GDM or T2DM has its challenges. A lack of standardised diagnostic or screening criteria for GDM may leave many women undiagnosed and untreated, which could lead to inconsistencies in clinical trial design [173]. Pregnancy itself carries various contributing factors which may influence metformin activity and placental and fetal development, such as advanced age, weight, ethnicity, stage of diabetes, maternal glycaemic control, diet, lifestyle, pre-conceptual multivitamin intake, and sex of the offspring [174,175]. Metformin can alter the microbiome and lead to lactic acid accumulation, prompting further heterogeneity in vitamin B12 and folate absorption between individuals [15]. Better understanding of these variable factors will improve the efficiency of GDM clinical diagnosis and personalised therapeutic strategies. The dose and duration of metformin use should also be taken into consideration for patient screening regimens to facilitate personalised treatment.
As nutrient biomarkers are rapidly metabolised, this only gives us a transient snapshot of vitamin status and therefore makes it challenging to establish causal relationships of nutrient deficiency [176]. Heterogeneity in the diagnostic criteria and biomarkers for vitamin B12 deficiency promotes inconsistencies between clinical trial results [77], leading to a blurred diagnostic approach and ill-defined nutritional management policy. It is now suggested that other biomarkers, such as holotranscobalamin (active form of vitamin B12) and MMA, should be measured concurrently or as second line tests to validate true B12 deficiency, as serum vitamin B12 may not reflect accurate B12 levels in tissues or cells [176,177]. Heightened patient awareness of vitamin B12 and folate deficiency associated with metformin therapy would encourage patients to eat a more vitamin-rich diet, thus potentially decreasing the risk of adverse fetal health in metformin exposed pregnancy.
9. Conclusions
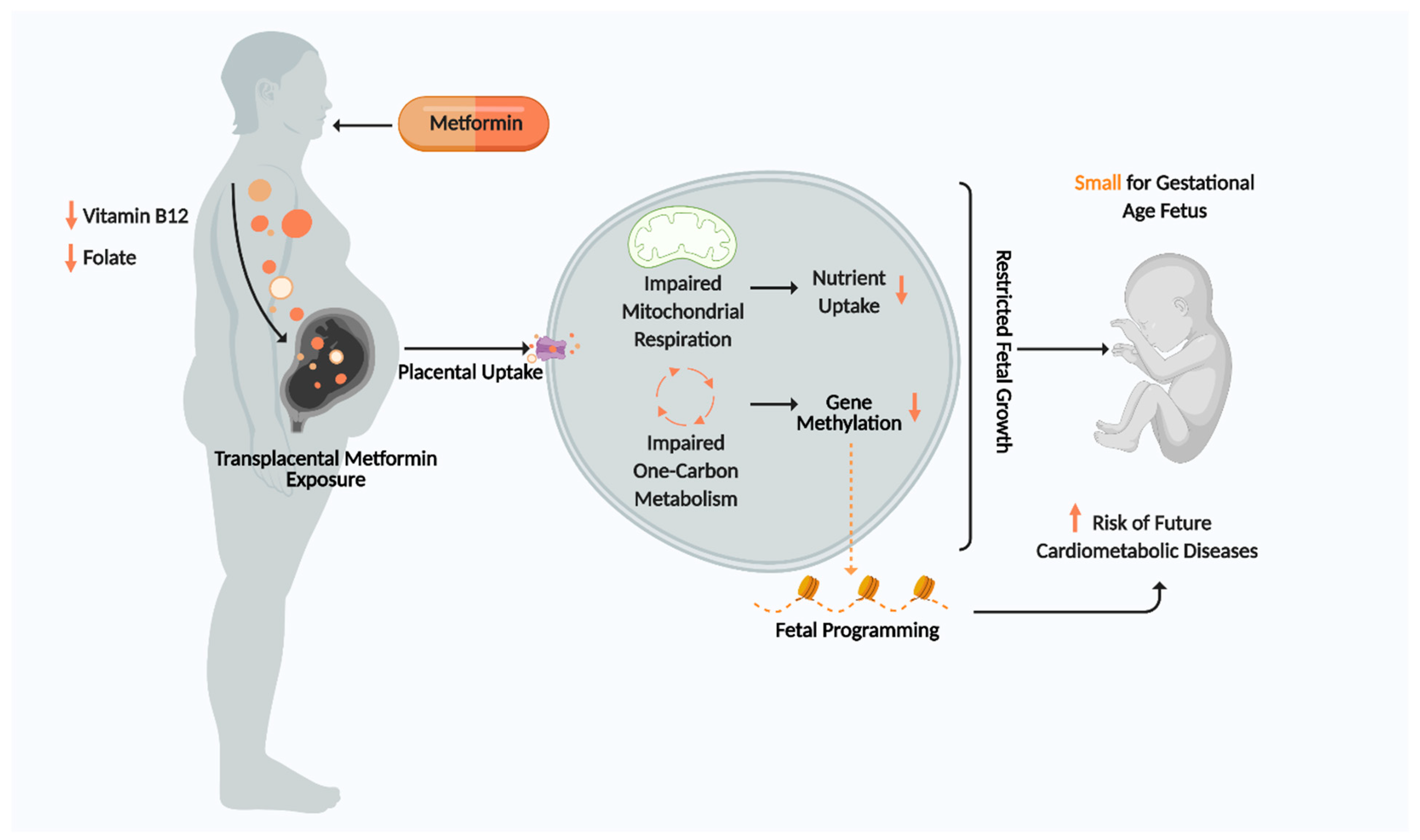
Metformin is a first-line therapy for diabetes in many countries which vastly improves glycaemic control. However, its antifolate-like and vitamin B12-lowering activity may impose adverse transgenerational effects on offspring in pregnant T2DM and GDM women by impairing one carbon metabolism and mitochondrial aerobic respiration. This may restrict placental and fetal growth, thereby promoting SGA births and increasing offspring susceptibility to cardiometabolic diseases in adulthood (Figure 4). Currently, there are no gold standard criteria for GDM diagnosis and no routine tests to measure vitamin status of metformin users during pregnancy, leaving the fetus potentially vulnerable to harmful stimuli for a large part of gestation. Discovering an optimal therapeutic window for vitamin replenishment in metformin-exposed pregnancies may potentially improve fetal health and disease susceptibility. It is paramount that future clinical trials studying the effects of metformin on fetal outcome should have analogous designs and methodologies to allow for more comparable end points and improved data comparison and interpretation. Longer follow-up studies of offspring exposed to metformin are needed to evaluate the long-term effects of metformin on fetal programming and cardiometabolic health. Ultimately, discovering how metformin promotes SGA births and fetal cardiometabolic disease will enable us to design a novel drug that continues to exert the beneficial effects metformin has on maternal health whilst minimising adverse effects on fetal health.
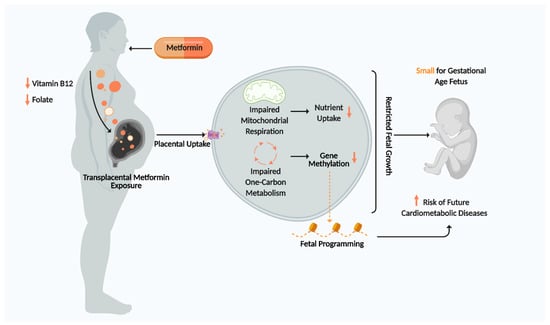
Figure 4.
Summary of Proposed Mechanism for metformin effects on placenta and fetus. Figure created using Biorender.com.
Author Contributions
Conceptualization, K.F. and E.M.S.; Literature Review, M.D.O.; Literature Curation, M.D.O. and B.C.B.; Writing—Original Draft Preparation, M.D.O.; Writing—Review & Editing, B.C.B., E.M.S., and K.F.; Supervision, K.F. and E.M.S.; Funding Acquisition, K.F. and E.M.S. All authors have read and agreed to the published version of the manuscript.
Funding
M.O. is supported by a British Heart Foundation four-year PhD studentship (REF: FS/4yPhD/F/20/34130). We acknowledge funding from the Medical Research Council (REF: MR/T001828/1 (ES) and REF: MR/R023166/1 (KF)). The APC was funded by the British Heart Foundation and Medical Research Council.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- International Diabetes Federation (IDF). World Diabetes Day 2017 to Focus on Women and Diabetes, Belgium. 2017. Available online: https://www.idf.org/news/2:world-dia (accessed on 10 December 2020).
- Melchior, H.; Kurch-Bek, D.; Mund, M. The prevalence of gestational diabetes. Dtsch. Arztebl. Int. 2017, 114, 412–418. [Google Scholar]
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019. [Google Scholar]
- Zhu, Y.; Zhang, C. Prevalence of gestational diabetes and risk of progression to type 2 diabetes: A global perspective. Curr. Diab. Rep. 2016, 16, 7. [Google Scholar] [CrossRef] [PubMed]
- Tarry-Adkins, J.L.; Aiken, C.E.; Ozanne, S.E. Neonatal, infant, and childhood growth following metformin versus insulin treatment for gestational diabetes: A systematic review and meta-analysis. PLOS Med. 2019, 16, e1002848. [Google Scholar] [CrossRef] [PubMed]
- Stacey, T.; Tennant, P.; McCowan, L.; Mitchell, E.; Budd, J.; Li, M.; Thompson, J.; Martin, B.; Roberts, D.; Heazell, A. Gestational diabetes and the risk of late stillbirth: A case–control study from England, UK. BJOG Int. J. Obstet. Gynaecol. 2019, 126, 973–982. [Google Scholar] [CrossRef]
- Feig, D.S.; Donovan, L.E.; Zinman, B.; Sanchez, J.J.; Asztalos, E.; Ryan, E.A.; Fantus, I.G.; Hutton, E.; Armson, A.B.; Lipscombe, L.L.; et al. Metformin in women with type 2 diabetes in pregnancy (MiTy): A multicentre, international, randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2020, 8, 834–844. [Google Scholar] [CrossRef]
- Lee, A.J.; Hiscock, R.J.; Wein, P.; Walker, S.P.; Permezel, M. Gestational diabetes mellitus: Clinical predictors and long-term risk of developing type 2 diabetes. A retrospective cohort study using survival analysis. Diabetes Care 2007, 30, 878–883. [Google Scholar] [CrossRef]
- Engeland, A.; Bjørge, T.; Daltveit, A.K.; Skurtveit, S.; Vangen, S.; Vollset, S.E.; Furu, K. Risk of diabetes after gestational diabetes and preeclampsia. A registry-based study of 230,000 women in Norway. Eur. J. Epidemiol. 2011, 26, 157–163. [Google Scholar] [CrossRef]
- Saravanan, P. Gestational diabetes: Opportunities for improving maternal and child health. Lancet Diabetes Endocrinol. 2020, 8, 793–800. [Google Scholar] [CrossRef]
- Whicher, C.A.; O’Neill, S.; Holt, R.I.G. Diabetes in the UK: 2019. Diabet. Med. 2020, 37, 242–247. [Google Scholar] [CrossRef]
- Murphy, H.R.; Howgate, C.; O’Keefe, J.; Myers, J.; Morgan, M.; Coleman, M.A.; Jolly, M.; Valabhji, J.; Scott, E.M.; Knighton, P.; et al. Characteristics and outcomes of pregnant women with type 1 or type 2 diabetes: A 5-year national population-based cohort study. Lancet Diabetes Endocrinol. 2021, 9, 153–164. [Google Scholar] [CrossRef]
- Kelley, K.W.; Carroll, D.G.; Meyer, A. A review of current treatment strategies for gestational diabetes mellitus. Drugs Context 2015, 4. [Google Scholar] [CrossRef]
- Bahendeka, S.; Kaushik, R.; Swai, A.B.; Otieno, F.; Bajaj, S.; Kalra, S.; Bavuma, C.M.; Karigire, C. EADSG guidelines: Insulin storage and optimisation of injection technique in diabetes management. Diabetes Therapy. 2019, 10, 341–366. [Google Scholar] [CrossRef]
- Wakeman, M.; Archer, D.T. Metformin and micronutrient status in type 2 diabetes: Does polypharmacy involving acid-suppressing medications affect vitamin B12 levels? Diabetes Metab. Syndr. Obes. 2020, 13, 2093–2108. [Google Scholar] [CrossRef]
- Lindsay, R.S.; Loeken, M.R. Metformin use in pregnancy: Promises and uncertainties. Diabetologia 2017, 60, 1612–1619. [Google Scholar] [CrossRef]
- Bridges, H.R.; Jones, A.J.Y.; Pollak, M.N.; Hirst, J. Effects of metformin and other biguanides on oxidative phosphorylation in mitochondria. Biochem. J. 2014, 462, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Garcia Canaveras, J.C.; Chen, Z.; Wang, L.; Liang, L.; Jang, C.; Mayr, J.A.; Zhang, Z.; Ghergurovich, J.M.; Zhan, L.; et al. Serine catabolism feeds NADH when respiration is impaired. Cell Metab. 2020, 31, 809–821. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.; Hebert, M.F.; Wagner, D.J.; Easterling, T.R.; Liang, C.J.; Rice, K.; Wang, J. Organic cation transporter 3 facilitates fetal Exposure to metformin during pregnancy. Mol. Pharmacol. 2018, 94, 1125–1131. [Google Scholar] [CrossRef]
- Cuyàs, E.; Fernández-Arroyo, S.; Buxó, M.; Pernas, S.; Dorca, J.; Álvarez, I.; Martínez, S.; Pérez-Garcia, J.M.; Batista-López, N.; Rodríguez-Sánchez, C.A.; et al. Metformin induces a fasting- and antifolate-mimicking modification of systemic host metabolism in breast cancer patients. Aging 2019, 11, 2874–2888. [Google Scholar] [CrossRef] [PubMed]
- Barbour, L.A.; Scifres, C.; Valent, A.M.; Friedman, J.E.; Buchanan, T.A.; Coustan, D.; Aagaard, K.; Thornburg, K.L.; Catalano, P.M.; Galan, H.L.; et al. A cautionary response to SMFM statement: Pharmacological treatment of gestational diabetes. Am. J. Obstet. Gynecol. 2018, 219. [Google Scholar] [CrossRef] [PubMed]
- Madiraju, A.K.; Erion, D.M.; Rahimi, Y.; Zhang, X.-M.; Braddock, D.T.; Albright, R.A.; Prigaro, B.J.; Wood, J.L.; Bhanot, S.; MacDonald, M.J.; et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature 2014, 510, 542–546. [Google Scholar] [CrossRef]
- An, H.; He, L. Current understanding of metformin effect on the control of hyperglycemia in diabetes. J. Endocrinol. 2016, 228. [Google Scholar] [CrossRef]
- Hyer, S.; Balani, J.; Shehata, H. Metformin in pregnancy: Mechanisms and clinical applications. Int. J. Mol. Sci. 2018, 19, 1954. [Google Scholar] [CrossRef] [PubMed]
- Balani, J.; Hyer, S.L.; Rodin, D.A.; Shehata, H. Pregnancy outcomes in women with gestational diabetes treated with metformin or insulin: A case-control study. Diabet. Med. 2009, 26, 798–802. [Google Scholar] [CrossRef]
- Lee, P.A.; Chernausek, S.D.; Hokken-Koelega, A.C.S.; Czernichow, P. International small for gestational age advisory board consensus development conference statement: Management of short children born small for gestational age, april 24–october 1, 2001. Pediatrics 2003, 111, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Rowan, J.A.; Rush, E.C.; Obolonkin, V.; Battin, M.; Wouldes, T.; Hague, W.M. Metformin in gestational diabetes: The offspring follow-up (MiG TOFU): Body composition at 2 years of age. Diabetes Care 2011, 34, 2279–2284. [Google Scholar] [CrossRef]
- Rowan, J.A.; Rush, E.C.; Plank, L.D.; Lu, J.; Obolonkin, V.; Coat, S.; Hague, W.M. Metformin in gestational diabetes: The offspring follow-up (MiG TOFU): Body composition and metabolic outcomes at 7-9 years of age. BMJ Open Diabetes Res. Care 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Hanem, L.G.E.; Salvesen, Ø.; Juliusson, P.B.; Carlsen, S.M.; Nossum, M.C.F.; Vaage, M.; Ødegård, R.; Vanky, E. Intrauterine metformin exposure and offspring cardiometabolic risk factors (PedMet study): A 5–10 year follow-up of the PregMet randomised controlled trial. Lancet Child. Adolesc. Health 2019, 3, 166–174. [Google Scholar] [CrossRef]
- Ijäs, H.; Vääräsmäki, M.; Saarela, T.; Keravuo, R.; Raudaskoski, T. A follow-up of a randomised study of metformin and insulin in gestational diabetes mellitus: Growth and development of the children at the age of 18 months. BJOG 2015, 122, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Salomäki, H.; Vähätalo, L.H.; Laurila, K.; Jäppinen, N.T.; Penttinen, A.-M.; Ailanen, L.; Ilyasizadeh, J.; Pesonen, U.; Koulu, M. Prenatal metformin exposure in mice programs the metabolic phenotype of the offspring during a high fat diet at adulthood. PLoS ONE 2013, 8, e56594. [Google Scholar] [CrossRef]
- Jamal, A.; Milani, F.; Al-Yasin, A. Evaluation of the effect of metformin and aspirin on utero placental circulation of pregnant women with PCOS. Iran. J. Reprod. Med. 2012, 10, 265–270. [Google Scholar]
- Jiang, S.; Teague, A.M.; Tryggestad, J.B.; Jensen, M.E.; Chernausek, S.D. Role of metformin in epigenetic regulation of placental mitochondrial biogenesis in maternal diabetes. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Brownfoot, F.C.; Hastie, R.; Hannan, N.J.; Cannon, P.; Nguyen, T.V.; Tuohey, L.; Cluver, C.; Tong, S.; Kaitu’u-Lino, T.J. Combining metformin and sulfasalazine additively reduces the secretion of antiangiogenic factors from the placenta: Implications for the treatment of preeclampsia. Placenta 2020, 95, 78–83. [Google Scholar] [CrossRef]
- Cluver, C.; Walker, S.P.; Mol, B.W.; Hall, D.; Hiscock, R.; Brownfoot, F.C.; Kaitu’u-Lino, T.J.; Tong, S. A double blind, randomised, placebo-controlled trial to evaluate the efficacy of metformin to treat preterm pre-eclampsia (PI2 Trial): Study protocol. BMJ Open 2019, 9, e025809. [Google Scholar] [CrossRef]
- Kaitu’u-Lino, T.J.; Brownfoot, F.C.; Beard, S.; Cannon, P.; Hastie, R.; Nguyen, T.V.; Binder, N.K.; Tong, S.; Hannan, N.J. Combining metformin and esomeprazole is additive in reducing sFlt-1 secretion and decreasing endothelial dysfunction—Implications for treating preeclampsia. PLoS ONE 2018, 13, e0188845. [Google Scholar] [CrossRef]
- Brownfoot, F.C.; Hastie, R.; Hannan, N.J.; Cannon, P.; Tuohey, L.; Parry, L.J.; Senadheera, S.; Illanes, S.E.; Kaitu’u-Lino, T.J.; Tong, S. Metformin as a prevention and treatment for preeclampsia: Effects on soluble fms-like tyrosine kinase 1 and soluble endoglin secretion and endothelial dysfunction. Am. J. Obstet. Gynecol. 2016, 214. [Google Scholar] [CrossRef] [PubMed]
- Szukiewicz, D.; Szewczyk, G.; Pyzlak, M.; Stangret, A.; Bachanek, M.; Trojanowski, S.; Alkhalayla, H.; Wejman, J. Anti-inflammatory action of metformin with respect to CX3CL1/CX3CR1 signaling in human placental circulation in normal-glucose versus high-glucose environments. Inflammation 2018, 41, 2246–2264. [Google Scholar] [CrossRef] [PubMed]
- Correia-Branco, A.; Keating, E.; Martel, F. Involvement of mTOR, JNK and PI3K in the negative effect of ethanol and metformin on the human first-trimester extravillous trophoblast HTR-8/SVneo cell line. Eur. J. Pharmacol. 2018, 833, 16–24. [Google Scholar] [CrossRef]
- Arshad, R.; Kanpurwala, M.A.; Karim, N.; Hassan, J.A. Effects of diet and metformin on placental morphology in gestational diabetes mellitus. Pak. J. Med. Sci. 2016, 32, 1522–1527. [Google Scholar] [CrossRef] [PubMed]
- Han, C.S.; Herrin, M.A.; Pitruzzello, M.C.; Mulla, M.J.; Werner, E.F.; Pettker, C.M.; Flannery, C.A.; Abrahams, V.M. Glucose and metformin modulate human first trimester trophoblast function: A model and potential therapy for diabetes-associated uteroplacental insufficiency. Am. J. Reprod. Immunol. 2014, 73, 362–371. [Google Scholar] [CrossRef]
- Wang, F.; Cao, G.; Yi, W.; Li, L.; Cao, X. Effect of metformin on a preeclampsia-like mouse model induced by high-fat diet. BioMed Res. Int. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Alzamendi, A.; Del Zotto, H.; Castrogiovanni, D.; Romero, J.; Giovambattista, A.; Spinedi, E. Oral metformin treatment prevents enhanced insulin demand and placental dysfunction in the pregnant rat fed a fructose-rich diet. ISRN Endocrinol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Eyal, S.; Easterling, T.R.; Carr, D.; Umans, J.G.; Miodovnik, M.; Hankins, G.D.V.; Clark, S.M.; Risler, L.; Wang, J.; Kelly, E.J.; et al. Pharmacokinetics of metformin during pregnancy. Drug Metab. Dispos. 2010, 38, 833–840. [Google Scholar] [CrossRef]
- Charles, B.; Norris, R.; Xiao, X.; Hague, W. Population pharmacokinetics of metformin in late pregnancy. Ther. Drug Monit. 2006, 28, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Vanky, E.; Zahlsen, K.; Spigset, O.; Carlsen, S.M. Placental passage of metformin in women with polycystic ovary syndrome. Fertil. Steril. 2005, 83, 1575–1578. [Google Scholar] [CrossRef]
- Liao, M.Z.; Flood Nichols, S.K.; Ahmed, M.; Clark, S.; Hankins, G.D.; Caritis, S.; Venkataramanan, R.; Haas, D.; Quinney, S.K.; Haneline, L.S.; et al. Effects of pregnancy on the pharmacokinetics of metformin. Drug Metab. Dispos. 2020, 48, 264–271. [Google Scholar] [CrossRef]
- Gormsen, L.C.; Sundelin, E.I.; Jensen, J.B.; Vendelbo, M.H.; Jakobsen, S.; Munk, O.L.; Hougaard Christensen, M.M.; Brøsen, K.; Frøkiær, J.; Jessen, N. In vivo Imaging of human 11C-metformin in peripheral organs: Dosimetry, biodistribution, and kinetic analyses. J. Nucl. Med. 2016, 57, 1920–1926. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, V.; Prasad, P.D. Role of transporters in placental transfer of drugs. Toxicol. Appl. Pharmacol. 2005, 207, 381–387. [Google Scholar] [CrossRef]
- Han, T.; Proctor, W.R.; Costales, C.L.; Cai, H.; Everett, R.S.; Thakker, D.R. Four cation-selective transporters contribute to apical uptake and accumulation of metformin in caco-2 cell monolayers. J. Pharmacol. Exp. Ther. 2015, 352, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Grube, M.; Meyer, Z.; Schwabedissen, H.; Draber, K.; Präger, D.; Möritz, K.U.; Linnemann, K.; Fusch, C.; Jedlitschky, G.; Kroemer, H.K. Expression, localization, and function of the carnitine transporter octn2 (slc22a5) in human placenta. Drug Metab. Dispos. 2005, 33, 31–37. [Google Scholar] [CrossRef]
- Lee, N.; Hebert, M.F.; Prasad, B.; Easterling, T.R.; Kelly, E.J.; Unadkat, J.D.; Wang, J. Effect of gestational age on mRNA and protein expression of polyspecific organic cation transporters during pregnancy. Drug Metab. Dispos. 2013, 41, 2225–2232. [Google Scholar] [CrossRef] [PubMed]
- Grace, M.R.; Dotters-Katz, S.K.; Zhou, C.; Manuck, T.; Boggess, K.; Bae-Jump, V. Effect of a high-fat diet and metformin on placental mtor signaling in mice. AJP Rep. 2019, 9, e138–e143. [Google Scholar] [CrossRef]
- Rosario, F.J.; Powell, T.; Jansson, T. Mechanistic target of rapamycin (mTOR) regulates trophoblast folate uptake by modulating the cell surface expression of FR-α and the RFC. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Jansson, T.; Aye, I.; Goberdhan, D. The emerging role of mTORC1 signaling in placental nutrient-sensing. Placenta 2012, 33, e23–e29. [Google Scholar] [CrossRef] [PubMed]
- Jansson, T.; Eliasson, L.; Rosario, F.; Powell, T.L.; Gupta, M.B. (Eds.) Remote control of fetal metabolism by placental mTOR signaling. In Reproductive Sciences; Sage Publications Inc.: Thousand Oaks, CA, USA, 2012; Volume 19. [Google Scholar]
- Kim, J.; Ahn, C.W.; Fang, S.; Lee, H.S.; Park, J.S. Association between metformin dose and vitamin B12 deficiency in patients with type 2 diabetes. Medicine 2019, 98, e17918. [Google Scholar] [CrossRef] [PubMed]
- Aroda, V.R.; Edelstein, S.L.; Goldberg, R.B.; Knowler, W.C.; Marcovina, S.M.; Orchard, T.J.; Bray, G.A.; Schade, D.S.; Temprosa, M.G.; White, N.H.; et al. Long-term metformin use and vitamin b12 deficiency in the diabetes prevention program outcomes study. J. Clin. Endocrinol. Metab. 2016, 101, 1754–1761. [Google Scholar] [CrossRef] [PubMed]
- Beulens, J.W.J.; Hart, H.E.; Kuijs, R.; Kooijman-Buiting, A.M.J.; Rutten, G.E.H.M. Influence of duration and dose of metformin on cobalamin deficiency in type 2 diabetes patients using metformin. Acta Diabetol. 2015, 52, 47–53. [Google Scholar] [CrossRef]
- Pflipsen, M.C.; Oh, R.C.; Saguil, A.; Seehusen, D.A.; Seaquist, D.; Topolski, R. The prevalence of vitamin B12 deficiency in patients with type 2 diabetes: A cross-sectional study. J. Am. Board Fam. Med. 2009, 22, 528–534. [Google Scholar] [CrossRef]
- Reinstatler, L.; Qi, Y.P.; Williamson, R.S.; Garn, J.V.; Oakley, G.P., Jr. Association of biochemical B12 deficiency with metformin therapy and vitamin B12 supplements: The national health and nutrition examination survey, 1999–2006. Diabetes Care 2012, 35, 327–333. [Google Scholar] [CrossRef]
- Corominas-Faja, B.; Quirantes-Piné, R.; Oliveras-Ferraros, C.; Vazquez-Martin, A.; Cufí, S.; Martin-Castillo, B.; Micol, V.; Joven, J.; Segura-Carretero, A.; Menendez, J.A. Metabolomic fingerprint reveals that metformin impairs one-carbon metabolism in a manner similar to the antifolate class of chemotherapy drugs. Aging 2012, 4, 480–498. [Google Scholar] [CrossRef] [PubMed]
- Rush, E.C.; Katre, P.; Yajnik, C.S. Vitamin B12: One carbon metabolism, fetal growth and programming for chronic disease. Eur. J. Clin. Nutr. 2013, 68, 2–7. [Google Scholar] [CrossRef]
- Maynard, A.G.; Kanarek, N. NADH ties one-carbon metabolism to cellular respiration. Cell Metab. 2020, 31, 660–662. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.C.; Mackie, F.L.; Lean, S.C.; Greenwood, S.L.; Heazell, A.E.P.; Forbes, K.; Jones, R.L. Placental dysfunction is associated with altered microRNA expression in pregnant women with low folate status. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.N.; Wheeler, S.J.; Sanders, T.A.; Thomas, J.E.; Hutchinson, C.J.; Clarke, K.; Berry, J.L.; Jones, R.L.; Seed, P.T.; Poston, L. A prospective study of micronutrient status in adolescent pregnancy. Am. J. Clin. Nutr. 2009, 89, 1114–1124. [Google Scholar] [CrossRef]
- Rogne, T.; Tielemans, M.J.; Chong, M.F.-F.; Yajnik, C.S.; Krishnaveni, G.V.; Poston, L.; Jaddoe, V.W.V.; Steegers, E.A.P.; Joshi, S.; Chong, Y.-S.; et al. Associations of maternal vitamin B12 concentration in pregnancy with the risks of preterm birth and low birth weight: A systematic review and meta-analysis of individual participant data. Am. J. Epidemiol. 2017, 185, 212–223. [Google Scholar] [CrossRef]
- Ducker, G.S.; Rabinowitz, J.D. One-carbon metabolism in health and disease. Cell Metab. 2017, 25, 27–42. [Google Scholar] [CrossRef]
- Malaguarnera, G.; Gagliano, C.; Salomone, S.; Giordano, M.; Bucolo, C.; Pappalardo, A.; Drago, F.; Caraci, F.; Avitabile, T.; Motta, M. Folate status in type 2 diabetic patients with and without retinopathy. Clin. Ophthalmol. 2015, 9, 1437–1442. Available online: http://europepmc.org/abstract/MED/26300625 (accessed on 15 January 2021). [CrossRef] [PubMed]
- Henderson, A.M.; Tai, D.C.; Aleliunas, R.E.; Aljaadi, A.M.; Glier, M.B.; Xu, E.E.; Miller, J.W.; Verchere, C.B.; Green, T.J.; Devlin, A.M. Maternal folic acid supplementation with vitamin B(12) deficiency during pregnancy and lactation affects the metabolic health of adult female offspring but is dependent on offspring diet. FASEB J. 2018, 32, 5039–5050. [Google Scholar] [CrossRef] [PubMed]
- Guéant, J.-L.; Namour, F.; Guéant-Rodriguez, R.-M.; Daval, J.-L. Folate and fetal programming: A play in epigenomics? Trends Endocrinol. Metab. 2013, 24, 279–289. [Google Scholar] [CrossRef]
- Fan, J.; Ye, J.; Kamphorst, J.J.; Shlomi, T.; Thompson, C.B.; Rabinowitz, J.D. Quantitative flux analysis reveals folate-dependent NADPH production. Nature 2014, 510, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Di Simone, N.; Riccardi, P.; Maggiano, N.; Piacentani, A.; D’Asta, M.; Capelli, A.; Caruso, A. Effect of folic acid on homocysteine-induced trophoblast apoptosis. Mol. Hum. Reprod. 2004, 10, 665–669. [Google Scholar] [CrossRef]
- Kumar, A.; Palfrey, H.A.; Pathak, R.; Kadowitz, P.J.; Gettys, T.W.; Murthy, S.N. The metabolism and significance of homocysteine in nutrition and health. Nutr. Metab. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, P.; Yajnik, C.S. Role of maternal vitamin B12 on the metabolic health of the offspring: A contributor to the diabetes epidemic? Br. J. Diabetes Vasc. Dis. 2010, 10, 109–114. [Google Scholar] [CrossRef]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.-L.; Brito, A.; Guéant, J.-L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.-H.; et al. Vitamin B12 deficiency. Nat. Rev. Dis. Prim. 2017, 3, doi. [Google Scholar] [CrossRef] [PubMed]
- Hannibal, L.; Lysne, V.; Bjørke-Monsen, A.-L.; Behringer, S.; Grünert, S.C.; Spiekerkoetter, U.; Jacobsen, D.W.; Blom, H.J. Biomarkers and algorithms for the diagnosis of vitamin B12 deficiency. Front. Mol. Biosci. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Sukumar, N.; Venkataraman, H.; Wilson, S.; Goljan, I.; Selvamoni, S.; Patel, V.; Saravanan, P. Vitamin B12 status among pregnant women in the UK and its association with obesity and gestational diabetes. Nutrients 2016, 8, 768. [Google Scholar] [CrossRef]
- Kouroglou, E.; Anagnostis, P.; Daponte, A.; Bargiota, A. Vitamin B12 insufficiency is associated with increased risk of gestational diabetes mellitus: A systematic review and meta-analysis. Endocrine 2019, 66, 149–156. [Google Scholar] [CrossRef]
- Stewart, C.P.; Christian, P.; Schulze, K.J.; Arguello, M.; LeClerq, S.C.; Khatry, S.K.; West, K.P., Jr. Low maternal vitamin B-12 status is associated with offspring insulin resistance regardless of antenatal micronutrient supplementation in rural nepal. J. Nutr. 2011, 141, 1912–1917. [Google Scholar] [CrossRef]
- Ho, M.; Halim, J.H.; Gow, M.L.; El-Haddad, N.; Marzulli, T.; Baur, L.A.; Cowell, C.T.; Garnett, S.P. Vitamin B12 in obese adolescents with clinical features of insulin resistance. Nutrients 2014, 6, 5611–5618. [Google Scholar] [CrossRef]
- Chandyo, R.K.; Ulak, M.; Kvestad, I.; Shrestha, M.; Ranjitkar, S.; Basnet, S.; Hysing, M.; Shrestha, L.; Strand, T.A. The effects of vitamin B12 supplementation in pregnancy and postpartum on growth and neurodevelopment in early childhood: Study protocol for a randomized placebo controlled trial. BMJ Open 2017, 7, e016434. [Google Scholar] [CrossRef]
- Sukumar, N.; Rafnsson, S.B.; Kandala, N.-B.; Bhopal, R.; Yajnik, C.S.; Saravanan, P. Prevalence of vitamin B-12 insufficiency during pregnancy and its effect on offspring birth weight: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2016, 103, 1232–1251. [Google Scholar] [CrossRef]
- Yajnik, C.S.; Deshpande, S.S.; Panchanadikar, A.V.; Naik, S.S.; Deshpande, J.A.; Coyaji, K.J.; Fall, C.; Refsum, H. Maternal total homocysteine concentration and neonatal size in India. Asia Pac. J. Clin. Nutr. 2005, 14, 179–181. [Google Scholar]
- World Health Organization. Serum and Red Blood Cell Folate Concentrations for Assessing Folate Status in Populations; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Kim, Y.I.; Fawaz, K.; Knox, T.; Lee, Y.M.; Norton, R.; Libby, E.; Mason, J.B. Colonic mucosal concentrations of folate are accurately predicted by blood measurements of folate status among individuals ingesting physiologic quantities of folate. Cancer Epidemiol. Biomark. Prev. 2001, 10, 715–719. [Google Scholar]
- Ramos, M.I.; Allen, L.H.; Mungas, D.M.; Jagust, W.J.; Haan, M.N.; Green, R.; Miller, J.W. Low folate status is associated with impaired cognitive function and dementia in the Sacramento Area Latino Study on Aging. Am. J. Clin. Nutr. 2005, 82, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Zou, R.; El Marroun, H.; Cecil, C.; Jaddoe, V.W.V.; Hillegers, M.; Tiemeier, H.; White, T. Maternal folate levels during pregnancy and offspring brain development in late childhood. Clin. Nutr. 2020. [Google Scholar] [CrossRef]
- Gaskins, A.J.; Rich-Edwards, J.W.; Hauser, R.; Williams, P.L.; Gillman, M.W.; Ginsburg, E.S.; Missmer, S.A.; Chavarro, J.E. Maternal prepregnancy folate intake and risk of spontaneous abortion and stillbirth. Obstet. Gynecol. 2014, 124, 23–31. [Google Scholar] [CrossRef]
- Van Uitert, E.M.; Steegers-Theunissen, R.P. Influence of maternal folate status on human fetal growth parameters. Mol. Nutr. Food Res. 2013, 57, 582–595. [Google Scholar] [CrossRef]
- Hodgetts, V.A.; Morris, R.K.; Francis, A.; Gardosi, J.; Ismail, K.M. Effectiveness of folic acid supplementation in pregnancy on reducing the risk of small-for-gestational age neonates: A population study, systematic review and meta-analysis. BJOG 2015, 122, 478–490. [Google Scholar] [CrossRef]
- Maloney, C.A.; Hay, S.M.; Rees, W.D. Folate deficiency during pregnancy impacts on methyl metabolism without affecting global DNA methylation in the rat fetus. Br. J. Nutr. 2007, 97, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Engeham, S.F.; Haase, A.; Langley-Evans, S.C. Supplementation of a maternal low-protein diet in rat pregnancy with folic acid ameliorates programming effects upon feeding behaviour in the absence of disturbances to the methionine-homocysteine cycle. Br. J. Nutr. 2010, 103, 996–1007. [Google Scholar] [CrossRef]
- Burdge, G. Homocysteine: A role in fetal programming? Br. J. Nutr. 2006, 96. [Google Scholar] [CrossRef]
- Molloy, A.M.; Kirke, P.N.; Brody, L.C.; Scott, J.M.; Mills, J.L. Effects of folate and vitamin B12 deficiencies during pregnancy on fetal, infant, and child development. Food Nutr. Bull. 2008, 29, S101–S111. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, N.; Watson, E.D. Lessons from the one-carbon metabolism: Passing it along to the next generation. Repr. BioMed. Online 2013, 27, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Mani, C.; Kochhar, P.; Ravikumar, G.; Dwarkanath, P.; Sheela, C.N.; George, S.; Thomas, A.; Crasta, J.; Thomas, T.; Kurpad, A.V.; et al. Placental expression of ENG, VEGF, and FLT: Gender-specific associations with maternal vitamin B(12) status. Eur. J. Clin. Nutr. 2020, 74, 176–182. [Google Scholar] [CrossRef]
- Moussa, C.; Ross, N.; Jolette, P.; Macfarlane, A.J. Altered folate metabolism modifies cell proliferation and progesterone secretion in human placental choriocarcinoma JEG-3 cells. Br. J. Nutr. 2015, 114, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Shah, T.; Joshi, K.; Mishra, S.; Otiv, S.; Kumbar, V. Molecular and cellular effects of vitamin B12 forms on human trophoblast cells in presence of excessive folate. Biomed. Pharmacother 2016, 84, 526–534. [Google Scholar] [CrossRef]
- Yin, X.; Gao, R.; Geng, Y.; Chen, X.; Liu, X.; Mu, X.; Ding, Y.; Wang, Y.; He, J. Autophagy regulates abnormal placentation induced by folate deficiency in mice. Mol. Hum. Reprod. 2019, 25, 305–319. [Google Scholar] [CrossRef]
- Carletti, J.V.; Correia-Branco, A.; Silva, C.R.; Andrade, N.; Silva, L.O.P.; Martel, F. The effect of oxidative stress induced by tert-butylhydroperoxide under distinct folic acid conditions: An in vitro study using cultured human trophoblast-derived cells. Reprod. Toxicol. 2018, 77, 33–42. [Google Scholar] [CrossRef]
- Ahmed, T.; Fellus, I.; Gaudet, J.; Macfarlane, A.J.; Fontaine-Bisson, B.; Bainbridge, S.A. Effect of folic acid on human trophoblast health and function in vitro. Placenta 2016, 37, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Rosario, F.J.; Powell, T.L.; Jansson, T. mTOR folate sensing links folate availability to trophoblast cell function. J. Physiol. 2017, 595, 4189–4206. [Google Scholar] [CrossRef]
- Steegers-Theunissen, R.P.; Smith, S.C.; Steegers, E.A.; Guilbert, L.J.; Baker, P.N. Folate affects apoptosis in human trophoblastic cells. BJOG. 2000, 107, 1513–1515. [Google Scholar] [CrossRef]
- Mahajan, A.; Sapehia, D.; Thakur, S.; Mohanraj, P.S.; Bagga, R.; Kaur, J. Effect of imbalance in folate and vitamin B12 in maternal/parental diet on global methylation and regulatory miRNAs. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Shah, T.; Mishra, S.; More, A.; Otiv, S.; Apte, K.; Joshi, K. Combination of vitamin B12 active forms improved fetal growth in Wistar rats through up-regulation of placental miR-16 and miR-21 levels. Life Sci. 2017, 191, 97–103. [Google Scholar] [CrossRef]
- Rosario, F.J.; Nathanielsz, P.W.; Powell, T.L.; Jansson, T. Maternal folate deficiency causes inhibition of mTOR signaling, down-regulation of placental amino acid transporters and fetal growth restriction in mice. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Berchtold, P.; Bolli, P.; Arbenz, U.; Keiser, G. Disturbance of intestinal absorption following metformin therapy (observations on the mode of action of biguanides. Diabetologia 1969, 5, 405–412. [Google Scholar] [CrossRef]
- Kos, E.; Liszek, M.J.; Emanuele, M.A.; Durazo-Arvizu, R.; Camacho, P. Effect of metformin therapy on vitamin D and vitamin B12 levels in patients with type 2 diabetes mellitus. Endocr. Pract. 2012, 18, 179–184. [Google Scholar] [CrossRef]
- Niafar, M.; Hai, F.; Porhomayon, J.; Nader, N.D. The role of metformin on vitamin B12 deficiency: A meta-analysis review. Intern. Emerg. Med. 2015, 10, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Gatford, K.L.; Houda, C.M.; Lu, Z.X.; Coat, S.; Baghurst, P.A.; Owens, J.A.; Sikaris, K.; Rowan, J.A.; Hague, W.M. Vitamin B12 and homocysteine status during pregnancy in the metformin in gestational diabetes trial: Responses to maternal metformin compared with insulin treatment. Diabetes Obes. Metab. 2013, 15, 660–667. [Google Scholar] [CrossRef]
- Esmaeilzadeh, S.; Gholinezhad-Chari, M.; Ghadimi, R. The effect of metformin treatment on the serum levels of homocysteine, folic acid, and vitamin B12 in patients with polycystic ovary syndrome. J. Hum. Reprod. Sci. 2017, 10, 95–101. [Google Scholar] [PubMed]
- Snow, C.F. Laboratory diagnosis of vitamin B12 and folate deficiency: A guide for the primary care physician. Arch. Intern. Med. 1999, 159, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.F.; Clark, J.S.; Ireland, J.T.; Kesson, C.M.; Watson, W.S. Malabsorption of vitamin B12 and intrinsic factor secretion during biguanide therapy. Diabetologia 1983, 24, 16–18. [Google Scholar] [CrossRef]
- Ting, R.Z.-W.; Szeto, C.C.; Chan, M.H.-M.; Ma, K.K.; Chow, K.M. Risk factors of vitamin B12 deficiency in patients receiving metformin. Arch. Intern. Med. 2006, 166, 1975–1979. [Google Scholar] [CrossRef]
- Friedman, P.; Shia, M.; Wallace, J.K. A saturable high affinity binding site for transcobalamin II-vitamin B12 complexes in human placental membrane preparations. J. Clin. Investig. 1977, 59, 51–58. [Google Scholar] [CrossRef]
- Quadros, E.; Sai, P.; Rothenberg, S.P. Characterization of the human placental membrane receptor for transcobalamin II-cobalamin. Arch. Biochem. Biophys. 1994, 308, 192–199. [Google Scholar] [CrossRef]
- Seligman, P.A.; Allen, R.H. Characterization of the receptor for transcobalamin II isolated from human placenta. J. Biol. Chem. 1978, 253, 1766–1772. [Google Scholar] [CrossRef]
- Schneider, H.; Miller, R.K. Receptor-mediated uptake and transport of macromolecules in the human placenta. Int. J. Dev. Biol. 2009, 54, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Ng, W.W.; Catus, R.G.; Miller, R.K. Macromolecule transfer in the human trophoblast: Transcobalamin II-vitamin B12 uptake. Placenta Suppl. 1981, 3, 145–159. [Google Scholar]
- Perez-D’Gregorio, R.E.; Miller, R.K. Transport and endogenous release of vitamin B12 in the dually perfused human placenta. J. Pediatr. 1998, 132, S35–S42. [Google Scholar] [CrossRef]
- Graber, S.E.; Scheffel, U.; Hodkinson, B.; McIntyre, P.A. Placental transport of vitamin B12 in the pregnant rat. J. Clin. Investig. 1971, 50, 1000–1004. [Google Scholar] [CrossRef] [PubMed]
- Dallaglio, K.; Bruno, A.; Cantelmo, A.R.; Esposito, A.I.; Ruggiero, L.; Orecchioni, S.; Calleri, A.; Bertolini, F.; Pfeffer, U.; Noonan, D.M. Paradoxic effects of metformin on endothelial cells and angiogenesis. Carcinogenesis 2014, 35, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Venna, V.R.; Li, J.; Hammond, M.D.; Mancini, N.S.; McCullough, L.D. Chronic metformin treatment improves post-stroke angiogenesis and recovery after experimental stroke. Eur. J. Neurosci. 2014, 39, 2129–2138. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.K.; Adya, R.; Chen, J.; Farhatullah, S.; Heutling, D.; Mitchell, D.; Lehnert, H.; Randeva, H.S. Metformin decreases angiogenesis via NF-κB and Erk1/2/Erk5 pathways by increasing the antiangiogenic thrombospondin-1. Cardiovasc. Res. 2009, 83, 566–574. [Google Scholar] [CrossRef]
- Di Pietro, M.; Parborell, F.; Irusta, G.; Pascuali, N.; Bas, D.; Bianchi, M.S.; Tesone, M.; Abramovich, D. Metformin regulates ovarian angiogenesis and follicular development in a female polycystic ovary syndrome rat model. Endocrinology 2015, 156, 1453–1463. [Google Scholar] [CrossRef]
- Adaikalakoteswari, A.; Finer, S.; Voyias, P.D.; McCarthy, C.M.; Vatish, M.; Moore, J.; Smart-Halajko, M.; Bawazeer, N.; Al-Daghri, N.M.; McTernan, P.G.; et al. Vitamin B12 insufficiency induces cholesterol biosynthesis by limiting s-adenosylmethionine and modulating the methylation of SREBF1 and LDLR genes. Clin. Epigen. 2015, 7. [Google Scholar] [CrossRef]
- Adaikalakoteswari, A.; Vatish, M.; Alam, M.T.; Ott, S.; Kumar, S.; Saravanan, P. Low vitamin b12 in pregnancy is associated with adipose-derived circulating mirs targeting pparγ and insulin resistance. J. Clin. Endocrinol. Metab. 2017, 102, 4200–4209. [Google Scholar] [CrossRef]
- Kumar, K.A.; Lalitha, A.; Pavithra, D.; Padmavathi, I.J.N.; Ganeshan, M.; Rao, K.R.; Venu, L.; Balakrishna, N.; Shanker, N.H.; Reddy, S.U.; et al. Maternal dietary folate and/or vitamin B12 restrictions alter body composition (adiposity) and lipid metabolism in Wistar rat offspring. J. Nutr. Biochem. 2013, 24, 25–31. [Google Scholar] [CrossRef]
- Kamaruddin, M.; Jusuf, E.; Sanusi, H. Correlation of vitamin B12 level with insulin resistance in infant and placenta outcomes. Enfermería Clín. 2020, 30, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.; Muntingh, G.; Rheeder, P. Vitamin B12 deficiency in metformin-treated type-2 diabetes patients, prevalence and association with peripheral neuropathy. BMC Pharmacol. Toxicol. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.H.; Ko, S.H.; Ahn, Y.B.; Song, K.H.; Han, K.D.; Park, Y.M.; Ko, S.H.; Kim, H.S. Association of vitamin B12 deficiency and metformin use in patients with type 2 diabetes. J. Korean Med. Sci. 2014, 29, 965–972. [Google Scholar] [CrossRef]
- Tomkin, G.H.; Hadden, D.R.; Weaver, J.A.; Montgomery, D.A.D. Vitamin-B12 status of patients on long-term metformin therapy. Br. Med. J. 1971, 2, 685–687. [Google Scholar] [CrossRef] [PubMed]
- Scherneck, S.; Schlinke, N.; Beck, E.; Grupe, K.; Weber-Schoendorfer, C.; Schaefer, C. Pregnancy outcome after first-trimester exposure to metformin: A prospective cohort study. Reprod. Toxicol. 2018, 81, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Shibata, E.; Hubel, C.A.; Powers, R.W.; von Versen-Hoeynck, F.; Gammill, H.; Rajakumar, A.; Roberts, J.M. Placental system A amino acid transport is reduced in pregnancies with small for gestational age (SGA) infants but not in preeclampsia with SGA infants. Placenta. 2008, 29, 879–882. [Google Scholar] [CrossRef] [PubMed]
- Roos, S.; Jansson, N.; Palmberg, I.; Säljö, K.; Powell, T.; Jansson, T. Mammalian target of rapamycin in the human placenta regulates leucine transport and is down-regulated in restricted fetal growth. J. Physiol. 2007, 582, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.-H.; Hsieh, T.-T.; Wu, C.-P.; Li, M.-J.; Yeh, Y.-L.; Chen, S.-F. Mammalian target of rapamycin signaling is a mechanistic link between increased endoplasmic reticulum stress and autophagy in the placentas of pregnancies complicated by growth restriction. Placenta 2017, 60, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Yung H-w Calabrese, S.; Hynx, D.; Hemmings, B.A.; Cetin, I.; Charnock-Jones, D.S.; Burton, G.J. Evidence of placental translation inhibition and endoplasmic reticulum stress in the etiology of human intrauterine growth restriction. Am. J. Pathol. 2008, 173, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Orozco, J.M.; Saxton, R.A.; Condon, K.J.; Liu, G.Y.; Krawczyk, P.A.; Scaria, S.M.; Harper, J.W.; Gygi, S.P.; Sabatini, D.M. SAMTOR is an S-adenosylmethionine sensor for the mTORC1 pathway. Science 2017, 358, 813–818. [Google Scholar] [CrossRef]
- Sahin, M.; Tutuncu, N.B.; Ertugrul, D.; Tanaci, N.; Guvener, N.D. Effects of metformin or rosiglitazone on serum concentrations of homocysteine, folate, and vitamin B12 in patients with type 2 diabetes mellitus. J. Diabetes Complicat. 2007, 21, 118–123. [Google Scholar] [CrossRef]
- Pongchaidecha, M.; Srikusalanukul, V.; Chattananon, A.; Tanjariyaporn, S. Effect of metformin on plasma homocysteine, vitamin B12 and folic acid: A cross-sectional study in patients with type 2 diabetes mellitus. J. Med. Assoc. Thai. 2004, 87, 780–787. [Google Scholar] [PubMed]
- Wulffelé, M.G.; Kooy, A.; Lehert, P.; Bets, D.; Ogterop, J.C.; Borger van der Burg, B.; Donker, A.J.M.; Stehouwer, C.D.A. Effects of short-term treatment with metformin on serum concentrations of homocysteine, folate and vitamin B12 in type 2 diabetes mellitus: A randomized, placebo-controlled trial. J. Intern. Med. 2003, 254, 455–463. [Google Scholar] [CrossRef]
- Carlsen, S.M.; Følling, I.; Grill, V.; Bjerve, K.S.; Schneede, J.; Refsum, H. Metformin increases total serum homocysteine levels in non-diabetic male patients with coronary heart disease. Scand. J. Clin. Lab. Invest. 1997, 57, 521–527. [Google Scholar] [CrossRef]
- Solanky, N.; Requena Jimenez, A.; D’Souza, S.W.; Sibley, C.P.; Glazier, J.D. Expression of folate transporters in human placenta and implications for homocysteine metabolism. Placenta 2010, 31, 134–143. [Google Scholar] [CrossRef]
- Piedrahita, J.A.; Oetama, B.; Bennett, G.D.; van Waes, J.; Kamen, B.A.; Richardson, J.; Lacey, S.W.; Anderson, R.G.; Finnell, R.H. Mice lacking the folic acid-binding protein Folbp1 are defective in early embryonic development. Nat. Genet. 1999, 23, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Caviedes, L.; Iñiguez, G.; Hidalgo, P.; Castro, J.J.; Castano, E.; Llanos, M.; Hirsch, S.; Ronco, A.M. Relationship between folate transporters expression in human placentas at term and birth weights. Placenta 2016, 38, 24–28. [Google Scholar] [CrossRef]
- Padmanabhan, N.; Jia, D.; Geary-Joo, C.; Wu, X.; Ferguson-Smith Anne, C.; Fung, E.; Bieda Mark, C.; Snyder Floyd, F.; Gravel Roy, A.; Cross James, C.; et al. Mutation in folate metabolism causes epigenetic instability and transgenerational effects on development. Cell 2013, 155, 81–93. [Google Scholar] [CrossRef] [PubMed]
- White, M.; Grynspan, D.; Van Mieghem, T.; Connor, K.L. Isolated fetal neural tube defects associate with increased risk of placental pathology: Evidence from the collaborative perinatal project. MedRxiv 2021. [Google Scholar] [CrossRef]
- Yue, Y.; Liu, J.; He, C. RNA N6-methyladenosine methylation in post-transcriptional gene expression regulation. Genes Dev. 2015, 29, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, C.R.; Lee, H.; Goodarzi, H.; Halberg, N.; Tavazoie, S.F. N6-methyladenosine marks primary microRNAs for processing. Nature 2015, 519, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Jaiswal, D. One-Carbon Metabolism, Spermatogenesis, and Male Infertility. Reprod. Sci. 2013, 20, 622–630. [Google Scholar] [CrossRef]
- Zheng, L.D.; Linarelli, L.E.; Liu, L.; Wall, S.S.; Greenawald, M.H.; Seidel, R.W.; Estabrooks, P.A.; Almeida, F.A.; Cheng, Z. Insulin resistance is associated with epigenetic and genetic regulation of mitochondrial DNA in obese humans. Clin. Epigen. 2015, 7. [Google Scholar] [CrossRef]
- Kraus, D.; Yang, Q.; Kong, D.; Banks, A.S.; Zhang, L.; Rodgers, J.T.; Pirinen, E.; Pulinilkunnil, T.C.; Gong, F.; Wang, Y.-c.; et al. Nicotinamide N-methyltransferase knockdown protects against diet-induced obesity. Nature 2014, 508, 258–262. [Google Scholar] [CrossRef]
- Roberti, A.; Fernández, A.F.; Fraga, M.F. Nicotinamide N-methyltransferase: At the crossroads between cellular metabolism and epigenetic regulation. Mol. Metab. 2021, 45. [Google Scholar] [CrossRef]
- Alshawi, A.; Agius, L. Low metformin causes a more oxidized mitochondrial NADH/NAD redox state in hepatocytes and inhibits gluconeogenesis by a redox-independent mechanism. J. Biol. Chem. 2019, 294, 2839–5691. [Google Scholar] [CrossRef]
- Ferland-McCollough, D.; Fernandez-Twinn, D.S.; Cannell, I.G.; David, H.; Warner, M.; Vaag, A.A.; Bork-Jensen, J.; Brøns, C.; Gant, T.W.; Willis, A.E.; et al. Programming of adipose tissue miR-483-3p and GDF-3 expression by maternal diet in type 2 diabetes. Cell Death Differ. 2012, 19, 1003–1012. [Google Scholar] [CrossRef]
- Coleman, C.B.; Lightell, D.J., Jr.; Moss, S.C.; Bates, M.; Parrino, P.E.; Woods, T.C. Elevation of miR-221 and-222 in the internal mammary arteries of diabetic subjects and normalization with metformin. Mol. Cell. Endocrinol. 2013, 374, 125–129. [Google Scholar] [CrossRef]
- Goodspeed, D.; Seferovic, M.D.; Holland, W.; Mcknight, R.A.; Summers, S.A.; Branch, D.W.; Lane, R.H.; Aagaard, K.M. Essential nutrient supplementation prevents heritable metabolic disease in multigenerational intrauterine growth-restricted rats. FASEB J. 2015, 29, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, K.; Yajnik, P.; Lubree, H.; Joglekar, C.; Bhat, D.; Katre, P.; Joshi, S.; Ladkat, R.; Fall, C.; Yajnik, C. The pune rural intervention in young adolescents (PRIYA) study: Design and methods of a randomised controlled trial. BMC Nutr. 2017, 3, 1–12. [Google Scholar] [CrossRef]
- Excellence NIfHaC. Diabetes in Pregnancy: Management from Preconception to the Postnatal Period: NICE Guideline, 2015; updated 2020. Available online: https://www.nice.org.uk/guidance/ng3/chapter/Recommendations (accessed on 15 January 2021).
- Hursthouse, N.A.; Gray, A.R.; Miller, J.C.; Rose, M.C.; Houghton, L.A. Folate status of reproductive age women and neural tube defect risk: The effect of long-term folic acid supplementation at doses of 140 µg and 400 µg per day. Nutrients 2011, 3, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Troen, A.M.; Mitchell, B.; Sorensen, B.; Wener, M.H.; Johnston, A.; Wood, B.; Selhub, J.; McTiernan, A.; Yasui, Y.; Oral, E.; et al. Unmetabolized folic acid in plasma is associated with reduced natural killer cell cytotoxicity among postmenopausal women. J. Nutr. 2006, 136, 189–194. [Google Scholar] [CrossRef]
- Krishnaveni, G.V.; Veena, S.R.; Karat, S.C.; Yajnik, C.S.; Fall, C.H.D. Association between maternal folate concentrations during pregnancy and insulin resistance in Indian children. Diabetologia 2014, 57, 110–121. [Google Scholar] [CrossRef]
- Rao, S.; Yajnik, C.S.; Kanade, A.; Fall, C.H.D.; Margetts, B.M.; Jackson, A.A.; Shier, R.; Joshi, S.; Rege, S.; Lubree, H.; et al. Intake of micronutrient-rich foods in rural indian mothers is associated with the size of their babies at birth: Pune maternal nutrition study. J. Nutr. 2001, 131, 1217–1224. [Google Scholar] [CrossRef]
- Whitrow, M.J.; Moore, V.M.; Rumbold, A.R.; Davies, M.J. Effect of supplemental folic acid in pregnancy on childhood asthma: A prospective birth cohort study. Am. J. Epidemiol. 2009, 170, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- Valera-Gran, D.; Navarrete-Muñoz, E.M.; Garcia de la Hera, M.; Fernández-Somoano, A.; Tardón, A.; Ibarluzea, J.; Balluerka, N.; Murcia, M.; González-Safont, L.; Romaguera, D.; et al. Effect of maternal high dosages of folic acid supplements on neurocognitive development in children at 4-5 y of age: The prospective birth cohort infancia y medio ambiente (INMA) study. Am. J. Clin Nutr. 2017, 106, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Pickell, L.; Brown, K.; Li, D.; Wang, X.L.; Deng, L.; Wu, Q.; Selhub, J.; Luo, L.; Jerome-Majewska, L.; Rozen, R. High intake of folic acid disrupts embryonic development in mice. Birth Defects Res. A. Clin. Mol. Teratol. 2011, 91, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Hammoud, R.; Pannia, E.; Kubant, R.; Wasek, B.; Bottiglieri, T.; Malysheva, O.V.; Caudill, M.A.; Anderson, G.H. Choline and folic acid in diets consumed during pregnancy interact to program food intake and metabolic regulation of male wistar rat offspring. J. Nutr. 2021, 151. [Google Scholar] [CrossRef]
- Yajnik, C.S.; Deshpande, S.S.; Jackson, A.A.; Refsum, H.; Rao, S.; Fisher, D.J.; Bhat, D.S.; Naik, S.S.; Coyaji, K.J.; Joglekar, C.V.; et al. Vitamin B12 and folate concentrations during pregnancy and insulin resistance in the offspring: The pune maternal nutrition study. Diabetologia 2008, 51, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Boachie, J.; Adaikalakoteswari, A.; Gázquez, A.; Zammit, V.; Larque, E.; Saravanan, P. Vitamin B12 induces hepatic fatty infiltration through altered fatty acid metabolism. Cell. Physiol. Biochem. 2021, 55, 241–255. [Google Scholar]
- Fisher, J.J.; Vanderpeet, C.L.; Bartho, L.A.; McKeating, D.R.; Cuffe, J.S.M.; Holland, O.J.; Perkins, A.V. Mitochondrial dysfunction in placental trophoblast cells experiencing gestational diabetes mellitus. J. Physiol. 2021, 599, 1291–1305. [Google Scholar] [CrossRef]
- Lawrence, S.A.; Titus, S.A.; Ferguson, J.; Heineman, A.L.; Taylor, S.M.; Moran, R.G. Mammalian mitochondrial and cytosolic folylpolyglutamate synthetase maintain the subcellular compartmentalization of folates. J. Biol Chem. 2014, 289, 29386–29396. [Google Scholar] [CrossRef]
- Behboudi-Gandevani, S.; Amiri, M.; Yarandi, R.B.; Tehrani, F.R. The impact of diagnostic criteria for gestational diabetes on its prevalence: A systematic review and meta-analysis. Diabetol. Metab. Syndr. 2019, 11. [Google Scholar] [CrossRef]
- Jaffe, A.; Giveon, S.; Rubin, C.; Novikov, I.; Ziv, A.; Kalter-Leibovici, O. Gestational diabetes risk in a multi-ethnic population. Acta Diabetologica 2020, 57, 263–269. [Google Scholar] [CrossRef]
- Tarry-Adkins, J.L.; Aiken, C.E.; Ozanne, S.E. Comparative impact of pharmacological treatments for gestational diabetes on neonatal anthropometry independent of maternal glycaemic control: A systematic review and meta-analysis. PLOS Med. 2020, 17, e1003126. [Google Scholar] [CrossRef]
- Sukumar, N.; Saravanan, P. Investigating vitamin B12 deficiency. BMJ 2019, 365. [Google Scholar] [CrossRef] [PubMed]
- Rafnsson, S.B.; Saravanan, P.; Bhopal, R.S.; Yajnik, C.S. Is a low blood level of vitamin B12 a cardiovascular and diabetes risk factor? A systematic review of cohort studies. Eur. J. Nutr. 2011, 50, 97–106. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).