Symptom Severity, Infection Progression and Plant Responses in Solanum Plants Caused by Three Pospiviroids Vary with the Inoculation Procedure
Abstract
:1. Introduction
2. Results
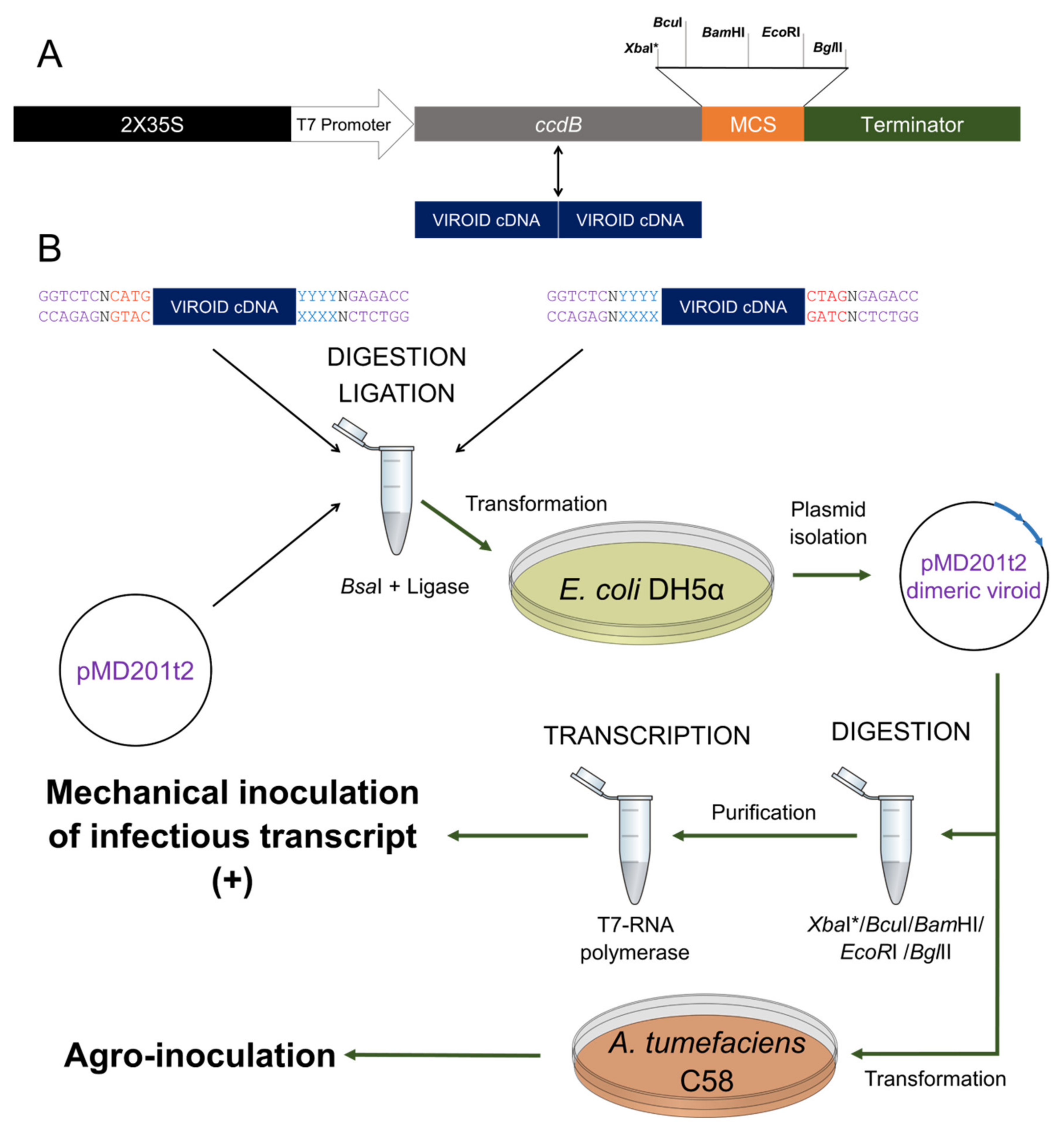
2.1. pMD201t2: A Highly Efficient Plasmid for Both In Vitro Transcription and In Vivo Agro-Inoculation
2.2. Influence of the Inoculation Method on the Symptomatology and Viroid Accumulation in Solanum Lycopersicum and Solanum Melongena
2.3. Effect of the Inoculation Method on the Activation of the Plant Defence Response in Tomato Plants
2.4. Induction of Ribosomal Stress Correlates with the Symptoms-Intensity
3. Discussion
4. Material and Methods
4.1. Binary Vector Construction
4.2. Dimeric Viroid cDNA Construction
4.3. In Vitro Transcription
4.4. Plant Material
4.5. Viroid Inoculation
4.6. RNA Extraction and Northern Blot
4.7. Real-Time PCR
4.8. Extraction and Analysis of SA and GA
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Flores, R.; Minoia, S.; Carbonell, A.; Gisel, A.; Delgado, S.; López-Carrasco, A.; Navarro, B.; Di Serio, F. Viroids, the simplest RNA replicons: How they manipulate their hosts for being propagated and how their hosts react for containing the infection. Virus Res. 2015, 209, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Owens, R.A.; Hammond, R.W. Viroid Pathogenicity: One Process, Many Faces. Viruses 2009, 1, 298–316. [Google Scholar] [CrossRef] [Green Version]
- Flores, R.; Owens, R.; Taylor, J. Pathogenesis by subviral agents: Viroids and hepatitis delta virus. Curr. Opin. Virol. 2016, 17, 87–94. [Google Scholar] [CrossRef]
- Navarro, B.; Gisel, A.; Rodio, M.-E.; Delgado, S.; Flores, R.; Di Serio, F. Viroids: How to infect a host and cause disease without encoding proteins. Biochimie 2012, 94, 1474–1480. [Google Scholar] [CrossRef]
- Daròs, J.-A. Eggplant latent viroid: A friendly experimental system in the family Avsunviroidae. Mol. Plant Pathol. 2016, 17, 1170–1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marquez-Molins, J.; Gomez, G.; Pallas, V. Hop stunt viroid: A polyphagous pathogenic RNA that has shed light on viroid–host interactions. Mol. Plant Pathol. 2021, 22, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Ding, B. The Biology of Viroid-Host Interactions. Annu. Rev. Phytopathol. 2009, 47, 105–131. [Google Scholar] [CrossRef] [PubMed]
- Adkar-Purushothama, C.R.; Perreault, J. Current overview on viroid–host interactions. Wiley Interdiscip. Rev. RNA 2020, 11, e1570. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Li, S.; Hou, W.; Fan, Z.; Xiao, H.; Lu, M.; Sano, T.; Zhang, Z. Global Transcriptomic Changes Induced by Infection of Cucumber (Cucumis sativus L.) with Mild and Severe Variants of Hop Stunt Viroid. Front. Microbiol. 2017, 8, 2427. [Google Scholar] [CrossRef] [Green Version]
- Owens, R.A.; Tech, K.B.; Shao, J.Y.; Sano, T.; Baker, C.J. Global Analysis of Tomato Gene Expression During Potato spindle tuber viroid Infection Reveals a Complex Array of Changes Affecting Hormone Signaling. Mol. Plant-Microbe Interact. 2012, 25, 582–598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez, G.; Castellano, M.; Tortosa, M.; Pallas, V.; Gomez, G. A pathogenic non-coding RNA induces changes in dynamic DNA methylation of ribosomal RNA genes in host plants. Nucleic Acids Res. 2014, 42, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Castellano, M.; Martinez, G.; Pallás, V.; Gómez, G. Alterations in host DNA methylation in response to constitutive expression of Hop stunt viroid RNA in Nicotiana benthamiana plants. Plant Pathol. 2015, 64, 1247–1257. [Google Scholar] [CrossRef]
- Castellano, M.; Pallas, V.; Gomez, G. A pathogenic long noncoding RNA redesigns the epigenetic landscape of the infected cells by subverting host Histone Deacetylase 6 activity. New Phytol. 2016, 211, 1311–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, Y.; Wang, Y.; Ding, B.; Fei, Z. Comprehensive Transcriptome Analyses Reveal that Potato Spindle Tuber Viroid Triggers Genome-Wide Changes in Alternative Splicing, Inducible trans -Acting Activity of Phased Secondary Small Interfering RNAs, and Immune Responses. J. Virol. 2017, 91, e00247-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, J.; Smith, H.N.; Ren, D.; Mudiyanselage, S.D.D.; Dawe, A.L.; Wang, L.; Wang, Y. Potato Spindle Tuber Viroid Modulates Its Replication through a Direct Interaction with a Splicing Regulator. J. Virol. 2018, 92, e01004-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cottilli, P.; Belda-Palazón, B.; Adkar-Purushothama, C.R.; Perreault, J.-P.; Schleiff, E.; Rodrigo, I.; Ferrando, A.; Lisón, P. Citrus exocortis viroid causes ribosomal stress in tomato plants. Nucleic Acids Res. 2019, 47, 8649–8661. [Google Scholar] [CrossRef] [PubMed]
- Prol, F.V.; López-Gresa, M.P.; Rodrigo, I.; Bellés, J.M.; Lisón, P. Ethylene is Involved in Symptom Development and Ribosomal Stress of Tomato Plants upon Citrus Exocortis Viroid Infection. Plants 2020, 9, 582. [Google Scholar] [CrossRef] [PubMed]
- Lisón, P.; Tárraga, S.; López-Gresa, M.P.; Saurí, A.; Torres, C.; Campos, L.; Bellés, J.M.; Conejero, V.; Rodrigo, I. A noncoding plant pathogen provokes both transcriptional and posttranscriptional alterations in tomato. Proteomics 2013, 13, 833–844. [Google Scholar] [CrossRef] [PubMed]
- Granell, A.; Bellés, J.M.; Conejero, V. Induction of pathogenesis-related proteins in tomato by citrus exocortis viroid, silver ion and ethephon. Physiol. Mol. Plant Pathol. 1987, 31, 83–90. [Google Scholar] [CrossRef]
- Bellés, J.M.; Garro, R.; Fayos, J.; Navarro, P.; Primo, J.; Conejero, V. Gentisic Acid as a Pathogen-Inducible Signal, Additional to Salicylic Acid for Activation of Plant Defenses in Tomato. Mol. Plant-Microbe Interact. 1999, 12, 227–235. [Google Scholar] [CrossRef] [Green Version]
- López-Gresa, M.P.; Lisón, P.; Yenush, L.; Conejero, V.; Rodrigo, I.; Bellés, J.M. Salicylic Acid Is Involved in the Basal Resistance of Tomato Plants to Citrus Exocortis Viroid and Tomato Spotted Wilt Virus. PLoS ONE 2016, 11, e0166938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Gresa, M.P.; Payá, C.; Rodrigo, I.; Bellés, J.M.; Barceló, S.; Choi, Y.H.; Verpoorte, R.; Lisón, P. Effect of Benzothiadiazole on the Metabolome of Tomato Plants Infected by Citrus Exocortis Viroid. Viruses 2019, 11, 437. [Google Scholar] [CrossRef] [Green Version]
- Di Serio, F.; Owens, R.A.; Li, S.-F.; Matoušek, J.; Pallás, V.; Randles, J.W.; Sano, T.; Verhoeven, J.T.J.; Vidalakis, G.; Flores, R.; et al. ICTV Virus Taxonomy Profile: Pospiviroidae. J. Gen. Virol. 2021, 102, jgv001543. [Google Scholar] [CrossRef] [PubMed]
- Di Serio, F.; Li, S.-F.; Matoušek, J.; Owens, R.A.; Pall as, V.; Randles, J.W.; Sano, T.; Th Verhoeven, J.J.; Vidalakis, G.; Flores, R. ICTV Virus Taxonomy Profile: Avsunviroidae. J. Gen. Virol. 2018, 99, 611–612. [Google Scholar] [CrossRef] [PubMed]
- Grill, L.K.; Semancik, J.S. RNA sequences complementary to citrus exocortis viroid in nucleic acid preparations from infected Gynura aurantiaca. Proc. Natl. Acad. Sci. USA 1978, 75, 896–900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branch, A.D.; Benenfeld, B.J.; Robertson, H.D. Evidence for a single rolling circle in the replication of potato spindle tuber viroid. Proc. Natl. Acad. Sci. USA 1988, 85, 9128–9132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feldstein, P.A.; Hu, Y.; Owens, R.A. Precisely full length, circularizable, complementary RNA: An infectious form of potato spindle tuber viroid. Proc. Natl. Acad. Sci. USA 1998, 95, 6560–6565. [Google Scholar] [CrossRef] [Green Version]
- Daros, J.-A.; Flores, R. Arabidopsis thaliana has the enzymatic machinery for replicating representative viroid species of the family Pospiviroidae. Proc. Natl. Acad. Sci. USA 2004, 101, 6792–6797. [Google Scholar] [CrossRef] [Green Version]
- Hammond, R.W. Economic Significance of Viroids in Vegetable and Field Crops. In Viroids and Satellites; Elsevier: Amsterdam, The Netherlands, 2017; pp. 5–13. [Google Scholar]
- Diener, T.O. Potato spindle tuber “virus”. IV. A replicating, low molecular weight RNA. Virology 1971, 45, 411–428. [Google Scholar] [CrossRef]
- Minoia, S.; Navarro, B.; Delgado, S.; Di Serio, F.; Flores, R. Viroid RNA turnover: Characterization of the subgenomic RNAs of potato spindle tuber viroid accumulating in infected tissues provides insights into decay pathways operating in vivo. Nucleic Acids Res. 2015, 43, 2313–2325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gramazio, P.; Lerma, M.D.; Villanueva, G.; Vilanova, S.; García-Fortea, E.; Mangino, G.; Figàs, M.R.; Arrones, A.; Alonso, D.; San Bautista, A.; et al. Detection, molecular characterisation and aspects involving the transmission of tomato chlorotic dwarf viroid in eggplant. Ann. Appl. Biol. 2019, 175, 172–183. [Google Scholar] [CrossRef]
- Gómez, G.; Martínez, G.; Pallás, V. Viroid-Induced Symptoms in Nicotiana benthamiana Plants Are Dependent on RDR6 Activity. Plant Physiol. 2008, 148, 414–423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez, G.; Martínez, G.; Pallás, V. Interplay between viroid-induced pathogenesis and RNA silencing pathways. Trends Plant Sci. 2009, 14, 264–269. [Google Scholar] [CrossRef]
- Flores, R.; Navarro, B.; Delgado, S.; Serra, P.; Serio, F. Di Viroid pathogenesis: A critical appraisal of the role of RNA silencing in triggering the initial molecular lesion. FEMS Microbiol. Rev. 2020, 44, 386–398. [Google Scholar] [CrossRef] [PubMed]
- Cress, D.E.; Kiefer, M.C.; Owens, R.A. Construction of infectious potato spindle tuber viroid cDNA clones. Nucleic Acids Res. 1983, 11, 6821–6835. [Google Scholar] [CrossRef] [Green Version]
- Visvader, J.E.; Forster, A.C.; Symons, R.H. Infectivity and in vitro mutagenesis of monomeric cDNA clones of citrus exocortis viroid indicates the site of processing of viroid precursors. Nucleic Acids Res. 1985, 13, 5843–5856. [Google Scholar] [CrossRef] [Green Version]
- Flores, R.; Navarro, J.-A.; De La Peña, M.; Navarro, B.; Ambrós, S.; Vera, A. Viroids with Hammerhead Ribozymes: Some Unique Structural and Functional Aspects with Respect to Other Members of the Group. Biol. Chem. 1999, 380, 849–854. [Google Scholar] [CrossRef] [Green Version]
- Qi, Y.; Pélissier, T.; Itaya, A.; Hunt, E.; Wassenegger, M.; Ding, B. Direct role of a viroid RNA motif in mediating directional RNA trafficking accross a specific cellular boundary. Plant Cell 2004, 16, 1741–1752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, Y.; Ding, B. Replication of Potato spindle tuber viroid in Cultured Cells of Tobacco and Nicotiana benthamiana: The Role of Specific Nucleotides in Determining Replication Levels for Host Adaptation. Viroligy 2002, 302, 445–456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Y.; Qi, Y.; Xun, Y.; Owens, R.; Ding, B. Movement of Potato Spindle Tuber Viroid Reveals Regulatory Points of Phloem-Mediated RNA Traffic. Plant Physiol. 2002, 130, 138–146. [Google Scholar] [CrossRef] [Green Version]
- Gozmanova, M.; Denti, M.A.; Minkov, I.N.; Tsagris, M.; Tabler, M. Characterization of the RNA motif responsible for the specific interaction of potato spindle tuber viroid RNA (PSTVd) and the tomato protein Virp1. Nucleic Acids Res. 2003, 31, 5534–5543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adkar-Purushothama, C.R.; Brosseau, C.; Giguère, T.; Sano, T.; Moffett, P.; Perreault, J.-P. Small RNA Derived from the Virulence Modulating Region of the Potato spindle tuber viroid Silences callose synthase Genes of Tomato Plants. Plant Cell 2015, 27, 2178–2194. [Google Scholar] [CrossRef] [Green Version]
- Marquez-Molins, J.; Navarro, J.A.; Pallas, V.; Gomez, G. Highly efficient construction of infectious viroid-derived clones. Plant Methods 2019, 15, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Duran-Vila, N. Citrus Exocortis Viroid. In Viroids and Satellites; Elsevier: Amsterdam, The Netherlands, 2017; pp. 169–179. [Google Scholar]
- Owens, R.A. Potato spindle tuber viroid: The simplicity paradox resolved? Mol. Plant Pathol. 2007, 8, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Nie, X.; Singh, M. Tomato chlorotic dwarf viroid: An evolutionary link in the origin of pospiviroids. J. Gen. Virol. 1999, 80, 2823–2828. [Google Scholar] [CrossRef]
- Wassenegger, M.; Spieker, R.L.; Thalmeir, S.; Gast, F.-U.; Riedel, L.; Sänger, H.L. A Single Nucleotide Substitution Converts Potato Spindle Tuber Viroid (PSTVd) from a Noninfectious to an Infectious RNA for Nicotiana tabacum. Viroligy 1996, 226, 191–197. [Google Scholar] [CrossRef]
- Ohbayashi, I.; Lin, C.-Y.; Shinohara, N.; Matsumura, Y.; Machida, Y.; Horiguchi, G.; Tsukaya, H.; Sugiyama, M. Evidence for a Role of ANAC082 as a Ribosomal Stress Response Mediator Leading to Growth Defects and Developmental Alterations in Arabidopsis. Plant Cell 2017, 29, 2644–2660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nabeshima, T.; Doi, M.; Hosokawa, M. Agrobacterium-mediated inoculation of chrysanthemum (Chrysanthemum morifolium) plants with chrysanthemum stunt viroid. J. Virol. Methods 2016, 234, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Hammond, R.W. Agrobacterium-Mediated Inoculation of PSTVd cDNAs onto Tomato Reveals the Biological Effect of Apparently Lethal Mutations. Virology 1994, 201, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Gómez, G.; Pallas, V. Hop stunt viroid is processed and translocated in transgenic Nicotiana benthamiana plants. Mol. Plant Pathol. 2006, 7, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Itaya, A.; Ding, B. Transfecting Protoplasts by Electroporation to Study Viroid Replication. Curr. Protoc. Microbiol. 2005, 16D.4.1–16D.4.11. [Google Scholar] [CrossRef]
- Sanz-Carbonell, A.; Marques, M.C.; Bustamante, A.; Fares, M.A.; Rodrigo, G.; Gomez, G. Inferring the regulatory network of the miRNA-mediated response to biotic and abiotic stress in melon. BMC Plant Biol. 2019, 19, 78. [Google Scholar] [CrossRef] [PubMed]
- Fagoaga, C.; Duran-Vila, N. Naturally occurring variants of citrus exocortis viroid in vegetable crops. Plant Pathol. 1996, 45, 45–53. [Google Scholar] [CrossRef]
- Fadda, Z.; Daròs, J.; Flores, R.; Duran-Vila, N. Identification in eggplant of a variant of citrus exocortis viroid (CEVd) with a 96 nucleotide duplication in the right terminal region of the rod-like secondary structure. Virus Res. 2003, 97, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, G.; Gerhardt, N.; Siciliano, F.; Vojnov, A.; Malcuit, I.; Marano, M.R. Salicylic Acid Is Involved in the Nb-Mediated Defense Responses to Potato virus X in Solanum tuberosum. Mol. Plant-Microbe Interact. 2010, 23, 394–405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bellés, J.M.; Garro, R.; Pallas, V.; Fayos, J.; Rodrigo, I.; Conejero, V. Accumulation of gentisic acid as associated with systemic infections but not with the hypersensitive response in plant-pathogen interactions. Planta 2005, 223, 500–511. [Google Scholar] [CrossRef] [PubMed]
- Nagai, A.; Torres, P.B.; Duarte, L.M.L.; Chaves, A.L.R.; Macedo, A.F.; Floh, E.I.S.; De Oliveira, L.F.; Zuccarelli, R.; Dos Santos, D.Y.A.C. Signaling pathway played by salicylic acid, gentisic acid, nitric oxide, polyamines and non-enzymatic antioxidants in compatible and incompatible Solanum-tomato mottle mosaic virus interactions. Plant Sci. 2020, 290, 110274. [Google Scholar] [CrossRef] [PubMed]
- Campos, L.; Granell, P.; Tárraga, S.; López-Gresa, M.P.; Conejero, V.; Bellés, J.M.; Rodrigo, I.; Lisón, P. Salicylic acid and gentisic acid induce RNA silencing-related genes and plant resistance to RNA pathogens. Plant Physiol. Biochem. 2014, 77, 35–43. [Google Scholar] [CrossRef]
- Catalán, P.; Elena, S.F.; Cuesta, J.A.; Manrubia, S. Parsimonious Scenario for the Emergence of Viroid-Like Replicons De Novo. Viruses 2019, 11, 425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Visvader, J.E.; Gould, A.R.; Bruening, G.E.; Symons, R.H. Citrus exocortis viroid: Nucleotide sequence and secondary structure of an Australian isolate. FEBS Lett. 1982, 137, 288–292. [Google Scholar] [CrossRef] [Green Version]
- Gruner, R.; Fels, A.; Qu, F.; Zimmat, R.; Steger, G.; Riesner, D. Interdependence of Pathogenicity and Replicability with Potato Spindle Tuber Viroid. Viroligy 1995, 209, 60–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Genovés, A.; Navarro, J.A.; Pallás, V. Functional analysis of the five melon necrotic spot virus genome-encoded proteins. J. Gen. Viroligy 2006, 87, 2371–2380. [Google Scholar] [CrossRef] [PubMed]
- Herranz, M.C.; Sanchez-Navarro, J.A.; Aparicio, F.; Pallás, V. Simultaneous detection of six stone fruit viruses by non-isotopic molecular hybridization using a unique riboprobe or ‘polyprobe’. J. Virol. Methods 2005, 124, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Gantasala, N.P.; Papolu, P.K.; Thakur, P.K.; Kamaraju, D.; Sreevathsa, R.; Rao, U. Selection and validation of reference genes for quantitative gene expression studies by real-time PCR in eggplant (Solanum melongena L). BMC Res. Notes 2013, 6, 312. [Google Scholar] [CrossRef] [PubMed] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vázquez Prol, F.; Márquez-Molins, J.; Rodrigo, I.; López-Gresa, M.P.; Bellés, J.M.; Gómez, G.; Pallás, V.; Lisón, P. Symptom Severity, Infection Progression and Plant Responses in Solanum Plants Caused by Three Pospiviroids Vary with the Inoculation Procedure. Int. J. Mol. Sci. 2021, 22, 6189. https://doi.org/10.3390/ijms22126189
Vázquez Prol F, Márquez-Molins J, Rodrigo I, López-Gresa MP, Bellés JM, Gómez G, Pallás V, Lisón P. Symptom Severity, Infection Progression and Plant Responses in Solanum Plants Caused by Three Pospiviroids Vary with the Inoculation Procedure. International Journal of Molecular Sciences. 2021; 22(12):6189. https://doi.org/10.3390/ijms22126189
Chicago/Turabian StyleVázquez Prol, Francisco, Joan Márquez-Molins, Ismael Rodrigo, María Pilar López-Gresa, José María Bellés, Gustavo Gómez, Vicente Pallás, and Purificación Lisón. 2021. "Symptom Severity, Infection Progression and Plant Responses in Solanum Plants Caused by Three Pospiviroids Vary with the Inoculation Procedure" International Journal of Molecular Sciences 22, no. 12: 6189. https://doi.org/10.3390/ijms22126189









