Electrospun Nanofibers for Sensing and Biosensing Applications—A Review
Abstract
1. Introduction
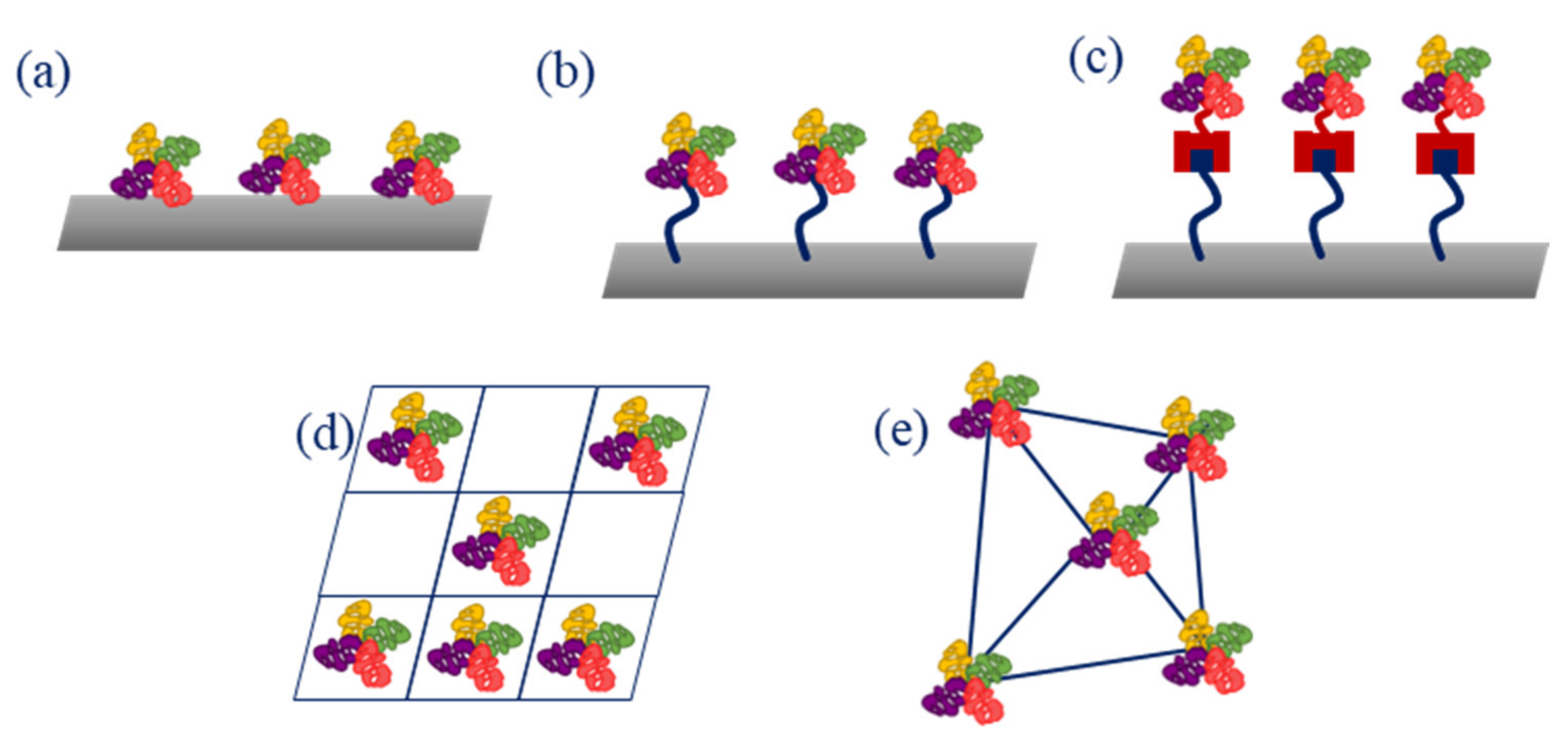
2. Sensors and Biosensors
3. Electrospinning and Nanofibers
3.1. Electrospinning
3.2. Nanofibers
4. Nanofiber-Based Electrochemical Sensors
4.1. Sensing of Glucose
4.2. Sensing of Biomedically Relevant Molecules and Drugs
4.3. Sensing of Metal Ions
5. Nanofiber-Based Optical Sensors
5.1. Sensing of Metal Ions
5.2. Sensing of Biomedically Relevant Compounds
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Agasti, S.S.; Rana, S.; Park, M.H.; Kim, C.K.; You, C.C.; Rotello, V.M. Nanoparticles for detection and diagnosis. Adv. Drug Deliv. Rev. 2010, 62, 316–328. [Google Scholar] [CrossRef]
- Chen, L.; Hwang, E.; Zhang, J. Fluorescent nanobiosensors for sensing glucose. Sensors 2018, 18, 1440. [Google Scholar] [CrossRef]
- Chowdhury, S.; Rooj, B.; Dutta, A.; Mandal, U. Review on Recent Advances in Metal Ions Sensing Using Different Fluorescent Probes. J. Fluoresc. 2018, 28, 999–1021. [Google Scholar] [CrossRef]
- Hulanicki, A.; Glab, S.; Ingman, F. Chemical sensors definitions and classification. Pure Appl. Chem. 1991, 63, 1247–1250. [Google Scholar] [CrossRef]
- Yang, F.; Chang, T.L.; Liu, T.; Wu, D.; Du, H.; Liang, J.; Tian, F. Label-free detection of Staphylococcus aureus bacteria using long-period fiber gratings with functional polyelectrolyte coatings. Biosens. Bioelectron. 2019, 133, 147–153. [Google Scholar] [CrossRef]
- Oloketuyi, S.; Mazzega, E.; Zavašnik, J.; Pungjunun, K.; Kalcher, K.; de Marco, A.; Mehmeti, E. Electrochemical immunosensor functionalized with nanobodies for the detection of the toxic microalgae Alexandrium minutum using glassy carbon electrode modified with gold nanoparticles. Biosens. Bioelectron. 2020, 154. [Google Scholar] [CrossRef]
- Selyanchyn, R.; Wakamatsu, S.; Hayashi, K.; Lee, S.W. A nano-thin film-based prototype QCM sensor array for monitoring human breath and respiratory patterns. Sensors 2015, 15, 18834–18850. [Google Scholar] [CrossRef] [PubMed]
- Sreenivasulu, G.; Qu, P.; Petrov, V.; Qu, H.; Srinivasan, G. Sensitivity enhancement in magnetic sensors based on ferroelectric-bimorphs and multiferroic composites. Sensors 2016, 16, 262. [Google Scholar] [CrossRef]
- Baluta, S.; Zając, D.; Szyszka, A.; Malecha, K.; Cabaj, J. Enzymatic platforms for sensitive neurotransmitter detection. Sensors 2020, 20, 423. [Google Scholar] [CrossRef]
- Dalirirad, S.; Steckl, A.J. Aptamer-based lateral flow assay for point of care cortisol detection in sweat. Sens. Actuators B Chem. 2019, 283, 79–86. [Google Scholar] [CrossRef]
- Tlili, C.; Myung, N.V.; Shetty, V.; Mulchandani, A. Label-free, chemiresistor immunosensor for stress biomarker cortisol in saliva. Biosens. Bioelectron. 2011, 26, 4382–4386. [Google Scholar] [CrossRef]
- Ferro, Y.; Perullini, M.; Jobbagy, M.; Bilmes, S.A.; Durrieu, C. Development of a biosensor for environmental monitoring based on microalgae immobilized in silica hydrogels. Sensors 2012, 12, 16879–16891. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, N.; Jolly, P.; Formisano, N.; Estrela, P. Introduction to biosensors. Essays Biochem. 2016, 60, 1–8. [Google Scholar] [CrossRef]
- Sassolas, A.; Blum, L.J.; Leca-Bouvier, B.D. Immobilization strategies to develop enzymatic biosensors. Biotechnol. Adv. 2012, 30, 489–511. [Google Scholar] [CrossRef]
- Hammond, J.L.; Formisano, N.; Estrela, P.; Carrara, S.; Tkac, J. Electrochemical biosensors and nanobiosensors. Essays Biochem. 2016, 60, 69–80. [Google Scholar] [CrossRef]
- Rackus, D.G.; Shamsi, M.H.; Wheeler, A.R. Electrochemistry, biosensors and microfluidics: A convergence of fields. Chem. Soc. Rev. 2015, 44, 5320–5340. [Google Scholar] [CrossRef]
- Luka, G.; Ahmadi, A.; Najjaran, H.; Alocilja, E.; Derosa, M.; Wolthers, K.; Malki, A.; Aziz, H.; Althani, A.; Hoorfar, M. Microfluidics integrated biosensors: A leading technology towards lab-on-a-chip and sensing applications. Sensors 2015, 15, 30011–30031. [Google Scholar] [CrossRef] [PubMed]
- Damborský, P.; Švitel, J.; Katrlík, J. Optical biosensors. Essays Biochem. 2016, 60, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Nejadmansouri, M.; Majdinasab, M.; Nunes, G.S.; Marty, J.L. An overview of optical and electrochemical sensors and biosensors for analysis of antioxidants in food during the last 5 years. Sensors 2021, 21, 1176. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, B.; Ghani, M.; Shoushtari, A.M.; Rabiee, M. Electrochemical biosensors based on nanofibres for cardiac biomarker detection: A comprehensive review. Biosens. Bioelectron. 2016, 78, 513–523. [Google Scholar] [CrossRef]
- Tucker, N.; Stanger, J.J.; Staiger, M.P.; Razzaq, H.; Hofman, K. The history of the science and technology of electrospinning from 1600 to 1995. J. Eng. Fiber. Fabr. 2012, 7, 63–73. [Google Scholar] [CrossRef]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef] [PubMed]
- Barhoum, A. Handbook of Nanofibers; Barhoum, A., Bechelany, M., Makhlouf, A.S.H., Eds.; Springer Nature: Cham, Switzerland, 2019; ISBN 9783319536545. [Google Scholar]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef]
- Khajavi, R.; Abbasipour, M. Electrospinning as a versatile method for fabricating coreshell, hollow and porous nanofibers. Sci. Iran. 2012, 19, 2029–2034. [Google Scholar] [CrossRef]
- Kenry; Lim, C.T. Nanofiber technology: Current status and emerging developments. Prog. Polym. Sci. 2017, 70, 1–17. [Google Scholar] [CrossRef]
- Haider, A.; Haider, S.; Kang, I.K. A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 2018, 11, 1165–1188. [Google Scholar] [CrossRef]
- Fahmy, A.; Zaid, H.; Ibrahim, M. Optimizing the electrospun parameters which affect the preparation of nanofibers. Biointerface Res. Appl. Chem. 2019, 9, 4463–4473. [Google Scholar] [CrossRef]
- ISO. ISO/TS 80004-2:2015(en), Nanotechnologies—Vocabulary—Part 2: Nano-Objects; ISO: Geneva, Switzerland, 2015. [Google Scholar]
- Terra, I.A.A.; Mercante, L.A.; Andre, R.S.; Correa, D.S. Fluorescent and Colorimetric Electrospun Nanofibers for Heavy-Metal Sensing. Biosensors 2017, 7, 61. [Google Scholar] [CrossRef]
- Balusamy, B.; Senthamizhan, A.; Uyar, T. Functionalized electrospun nanofibers as colorimetric sensory probe for mercury detection: A review. Sensors 2019, 19, 4763. [Google Scholar] [CrossRef] [PubMed]
- Thenmozhi, S.; Dharmaraj, N.; Kadirvelu, K.; Kim, H.Y. Electrospun nanofibers: New generation materials for advanced applications. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2017, 217, 36–48. [Google Scholar] [CrossRef]
- Wang, X.X.; Yu, G.F.; Zhang, J.; Yu, M.; Ramakrishna, S.; Long, Y.Z. Conductive polymer ultrafine fibers via electrospinning: Preparation, physical properties and applications. Prog. Mater. Sci. 2021, 115, 100704. [Google Scholar] [CrossRef]
- Ding, B.; Wang, M.; Wang, X.; Yu, J.; Sun, G. Electrospun nanomaterials for ultrasensitive sensors. Mater. Today 2010, 13, 16–27. [Google Scholar] [CrossRef]
- Merritt, S.R.; Exner, A.A.; Lee, Z.; Von Recum, H.A. Electrospinning and imaging. Adv. Eng. Mater. 2012, 14, 266–278. [Google Scholar] [CrossRef]
- Chen, K.; Chou, W.; Liu, L.; Cui, Y.; Xue, P.; Jia, M. Electrochemical sensors fabricated by electrospinning technology: An overview. Sensors 2019, 19, 3676. [Google Scholar] [CrossRef]
- Sapountzi, E.; Chateaux, J.-F.; Lagarde, F. Combining Electrospinning and Vapor-Phase Polymerization for the Production of Polyacrylonitrile/Polypyrrole Core-Shell Nanofibers and Glucose Biosensor Application. Front. Chem. 2020, 8, 678. [Google Scholar] [CrossRef]
- Rani, S.D.; Ramachandran, R.; Sheet, S.; Aziz, M.A.; Lee, Y.S.; Al-Sehemi, A.G.; Pannipara, M.; Xia, Y.; Tsai, S.Y.; Ng, F.L.; et al. NiMoO4 nanoparticles decorated carbon nanofiber membranes for the flexible and high performance glucose sensors. Sens. Actuators B Chem. 2020, 312, 127886. [Google Scholar] [CrossRef]
- Baek, S.H.; Roh, J.; Park, C.Y.; Kim, M.W.; Shi, R.; Kailasa, S.K.; Park, T.J. Cu-nanoflower decorated gold nanoparticles-graphene oxide nanofiber as electrochemical biosensor for glucose detection. Mater. Sci. Eng. C 2020, 107, 110273. [Google Scholar] [CrossRef]
- Mei, Q.; Fu, R.; Ding, Y.; Li, L.; Wang, A.; Duan, D.; Ye, D. Electrospinning of highly dispersed Ni/CoO carbon nanofiber and its application in glucose electrochemical sensor. J. Electroanal. Chem. 2019, 847, 113075. [Google Scholar] [CrossRef]
- Fu, R.; Lu, Y.; Ding, Y.; Li, L.; Ren, Z.; Si, X.; Wu, Q. A novel non-enzymatic glucose electrochemical sensor based on CNF@Ni-Co layered double hydroxide modified glassy carbon electrode. Microchem. J. 2019, 150, 104106. [Google Scholar] [CrossRef]
- Guo, Q.; Wu, T.; Liu, L.; He, Y.; Liu, D.; You, T. Hierarchically porous NiCo2S4 nanowires anchored on flexible electrospun graphitic nanofiber for high-performance glucose biosensing. J. Alloys Compd. 2020, 819, 153376. [Google Scholar] [CrossRef]
- Adabi, M.; Adabi, M. Electrodeposition of nickel on electrospun carbon nanofiber mat electrode for electrochemical sensing of glucose. J. Dispers. Sci. Technol. 2021, 42, 262–269. [Google Scholar] [CrossRef]
- Yezer, I.; Demirkol, D.O. Cellulose acetate–chitosan based electrospun nanofibers for bio-functionalized surface design in biosensing. Cellulose 2020, 27, 10183–10197. [Google Scholar] [CrossRef]
- Mehdizadeh, B.; Maleknia, L.; Amirabadi, A.; Shabani, M. Glucose sensing by a glassy carbon electrode modified with glucose oxidase/chitosan/graphene oxide nanofibers. Diam. Relat. Mater. 2020, 109, 108073. [Google Scholar] [CrossRef]
- Temoçin, Z. Designing of a stable and selective glucose biosensor by glucose oxidase immobilization on glassy carbon electrode sensitive to H2O2 via nanofiber interface. J. Appl. Electrochem. 2021, 51, 283–293. [Google Scholar] [CrossRef]
- Kafi, A.K.M.; Alim, S.; Jose, R.; Yusoff, M.M. Fabrication of a glucose oxidase/multiporous tin-oxide nanofiber film on Prussian blue–modified gold electrode for biosensing. J. Electroanal. Chem. 2019, 852, 113550. [Google Scholar] [CrossRef]
- Kim, K.O.; Kim, G.J.; Kim, J.H. A cellulose/β-cyclodextrin nanofiber patch as a wearable epidermal glucose sensor. RSC Adv. 2019, 9, 22790–22794. [Google Scholar] [CrossRef]
- Ratlam, C.; Phanichphant, S.; Sriwichai, S. Development of dopamine biosensor based on polyaniline/carbon quantum dots composite. J. Polym. Res. 2020, 27, 183. [Google Scholar] [CrossRef]
- Ozoemena, O.C.; Shai, L.J.; Maphumulo, T.; Ozoemena, K.I. Electrochemical Sensing of Dopamine Using Onion-like Carbons and Their Carbon Nanofiber Composites. Electrocatalysis 2019, 10, 381–391. [Google Scholar] [CrossRef]
- Nathani, A.; Sharma, C.S. Electrospun Mesoporous Poly(Styrene-Block-Methyl- Methacrylate) Nanofibers as Biosensing Platform: Effect of Fibers Porosity on Sensitivity. Electroanalysis 2019, 31, 2138–2144. [Google Scholar] [CrossRef]
- Chauhan, D.; Solanki, P.R. Hydrophilic and Insoluble Electrospun Cellulose Acetate Fiber-Based Biosensing Platform for 25-Hydroxy Vitamin-D 3 Detection. ACS Appl. Polym. Mater. 2019, 1, 1613–1623. [Google Scholar] [CrossRef]
- Bostanci, A.; Tanik, N.A.; Aykut, Y. Cellulose monoacetate/nafion (CMA/N) hybrid nanofibers as interface for electrochemical DNA biosensors. Tekst. Konfeksiyon 2020, 29, 228–236. [Google Scholar] [CrossRef]
- Wang, W.Q.; Yue, H.Y.; Yu, Z.M.; Huang, S.; Song, S.S.; Gao, X.; Guan, E.H.; Zhang, H.J.; Wang, Z. Synthesis and Application of MoS2 Nanosheet Arrays/Carbon Nanofibers for Simultaneous Electrochemical Determination of Levodopa and Uric Acid. IEEE Sens. J. 2019, 19, 5988–5994. [Google Scholar] [CrossRef]
- Paimard, G.; Shahlaei, M.; Moradipour, P.; Karamali, V.; Arkan, E. Impedimetric aptamer based determination of the tumor marker MUC1 by using electrospun core-shell nanofibers. Microchim. Acta 2020, 187, 5. [Google Scholar] [CrossRef]
- Adabi, M.; Esnaashari, S.S.; Adabi, M. An electrochemical immunosensor based on electrospun carbon nanofiber mat decorated with gold nanoparticles and carbon nanotubes for the detection of breast cancer. J. Porous Mater. 2021, 28, 415–421. [Google Scholar] [CrossRef]
- Mirani, A.; Maleknia, L.; Amirabadi, A. Glassy carbon electrode modified with hybrid nanofibers containing carbon nanotubes trapped in chitosan for the voltammetric sensing of nicotine at biological pH. Nanotechnology 2020, 31, 435504. [Google Scholar] [CrossRef] [PubMed]
- Samie, H.A.; Arvand, M. Label-free electrochemical aptasensor for progesterone detection in biological fluids. Bioelectrochemistry 2020, 133, 107489. [Google Scholar] [CrossRef] [PubMed]
- Moradi, S.; Azizi-Lalabadi, M.; Bagheri, V.; Sadeghi, E. Fabrication of electrospun sensor based on a synthesized component doped into PAN (polyacrylonitrile) nanofibers for electrochemical detection of zearalenone mycotoxin in foods simulant. Sens. Bio-Sens. Res. 2020, 28, 100321. [Google Scholar] [CrossRef]
- El-Moghazy, A.Y.; Amaly, N.; Istamboulie, G.; Nitin, N.; Sun, G. A signal-on electrochemical aptasensor based on silanized cellulose nanofibers for rapid point-of-use detection of ochratoxin A. Microchim. Acta 2020, 187, 535. [Google Scholar] [CrossRef]
- Migliorini, F.L.; Sanfelice, R.C.; Mercante, L.A.; Facure, M.H.M.; Correa, D.S. Electrochemical sensor based on polyamide 6/polypyrrole electrospun nanofibers coated with reduced graphene oxide for malathion pesticide detection. Mater. Res. Express 2020, 7, 15601. [Google Scholar] [CrossRef]
- Ozoemena, O.C.; Maphumulo, T.; Shai, J.L.; Ozoemena, K.I. Electrospun Carbon Nanofibers as an Electrochemical Immunosensing Platform for Vibrio cholerae Toxin: Aging Effect of the Redox Probe. ACS Omega 2020, 5, 5762–5771. [Google Scholar] [CrossRef]
- Paimard, G.; Shahlaei, M.; Moradipour, P.; Akbari, H.; Jafari, M.; Arkan, E. An Impedimetric Immunosensor modified with electrospun core-shell nanofibers for determination of the carcinoma embryonic antigen. Sens. Actuators B Chem. 2020, 311, 127928. [Google Scholar] [CrossRef]
- Asmatulu, R.; Veisi, Z.; Uddin, M.N.; Mahapatro, A. Highly Sensitive and Reliable Electrospun Polyaniline Nanofiber Based Biosensor as a Robust Platform for COX-2 Enzyme Detections. Fibers Polym. 2019, 20, 966–974. [Google Scholar] [CrossRef]
- Wang, W.Q.; Yue, H.Y.; Yu, Z.M.; Huang, S.; Song, S.S.; Gao, X.; Guan, E.H.; Zhang, H.J.; Wang, Z. Synthesis of graphene/carbon nanofiber for electrochemical determination of levodopa in the presence of uric acid. Ionics 2019, 25, 2835–2843. [Google Scholar] [CrossRef]
- Ebrahimi Vafaye, S.; Rahman, A.; Safaeian, S.; Adabi, M. An electrochemical aptasensor based on electrospun carbon nanofiber mat and gold nanoparticles for the sensitive detection of Penicillin in milk. J. Food Meas. Charact. 2021, 15, 876–882. [Google Scholar] [CrossRef]
- Afzali, M.; Mostafavi, A.; Shamspur, T. Sensitive detection of colchicine at a glassy carbon electrode modified with magnetic ionic liquid/CuO nanoparticles/carbon nanofibers in pharmaceutical and plasma samples. J. Iran. Chem. Soc. 2020, 17, 1753–1764. [Google Scholar] [CrossRef]
- Hu, W.; Zhang, Z.; Li, L.; Ding, Y.; An, J. Preparation of electrospun SnO2 carbon nanofiber composite for ultra-sensitive detection of APAP and p-Hydroxyacetophenone. Sens. Actuators B Chem. 2019, 299, 127003. [Google Scholar] [CrossRef]
- Bahrami, G.; Ehzari, H.; Mirzabeigy, S.; Mohammadi, B.; Arkan, E. Fabrication of a sensitive electrochemical sensor based on electrospun magnetic nanofibers for morphine analysis in biological samples. Mater. Sci. Eng. C 2020, 106, 110183. [Google Scholar] [CrossRef] [PubMed]
- Salandari-Jolge, N.; Ensafi, A.A.; Rezaei, B. A novel three-dimensional network of CuCr2O4/CuO nanofibers for voltammetric determination of anticancer drug methotrexate. Anal. Bioanal. Chem. 2020, 412, 2443–2453. [Google Scholar] [CrossRef]
- Jahromi, Z.; Mirzaei, E.; Savardashtaki, A.; Afzali, M.; Afzali, Z. A rapid and selective electrochemical sensor based on electrospun carbon nanofibers for tramadol detection. Microchem. J. 2020, 157, 104942. [Google Scholar] [CrossRef]
- Vilian, A.T.E.; Ranjith, K.S.; Lee, S.J.; Umapathi, R.; Hwang, S.K.; Oh, C.W.; Huh, Y.S.; Han, Y.K. Hierarchical dense Ni−Co layered double hydroxide supported carbon nanofibers for the electrochemical determination of metronidazole in biological samples. Electrochim. Acta 2020, 354, 136723. [Google Scholar] [CrossRef]
- Fathi, S.; Saber, R.; Adabi, M.; Rasouli, R.; Douraghi, M.; Morshedi, M.; Farid-Majidi, R. Novel competitive voltammetric aptasensor based on electrospun carbon nanofibers-gold nanoparticles modified graphite electrode for Salmonella enterica serovar detection. Biointerface Res. Appl. Chem. 2021, 11, 8702–8715. [Google Scholar] [CrossRef]
- Liberato, M.S.; Mancini, R.S.N.; Factori, I.M.; Ferreira, F.F.; de Oliveira, V.L.; Carnielli, J.B.T.; Guha, S.; Peroni, L.A.; Oliveira, M.A.L.; Alves, W.A. Peptide-Based Assemblies on Electrospun Polyamide-6/Chitosan Nanofibers for Detecting Visceral Leishmaniasis Antibodies. ACS Appl. Electron. Mater. 2019, 1, 2086–2095. [Google Scholar] [CrossRef]
- Sarabaegi, M.; Roushani, M.; Hosseini, H. Hollow carbon nanocapsules-based nitrogen-doped carbon nanofibers with rosary-like structure as a high surface substrate for impedimetric detection of Pseudomonas aeruginosa. Talanta 2021, 223, 121700. [Google Scholar] [CrossRef]
- Niri, A.D.; Faridi-Majidi, R.; Saber, R.; Khosravani, M.; Adabi, M. Electrospun carbon nanofiber-based electrochemical biosensor for the detection of hepatitis B virus. Biointerface Res. Appl. Chem. 2019, 9, 4022–4026. [Google Scholar] [CrossRef]
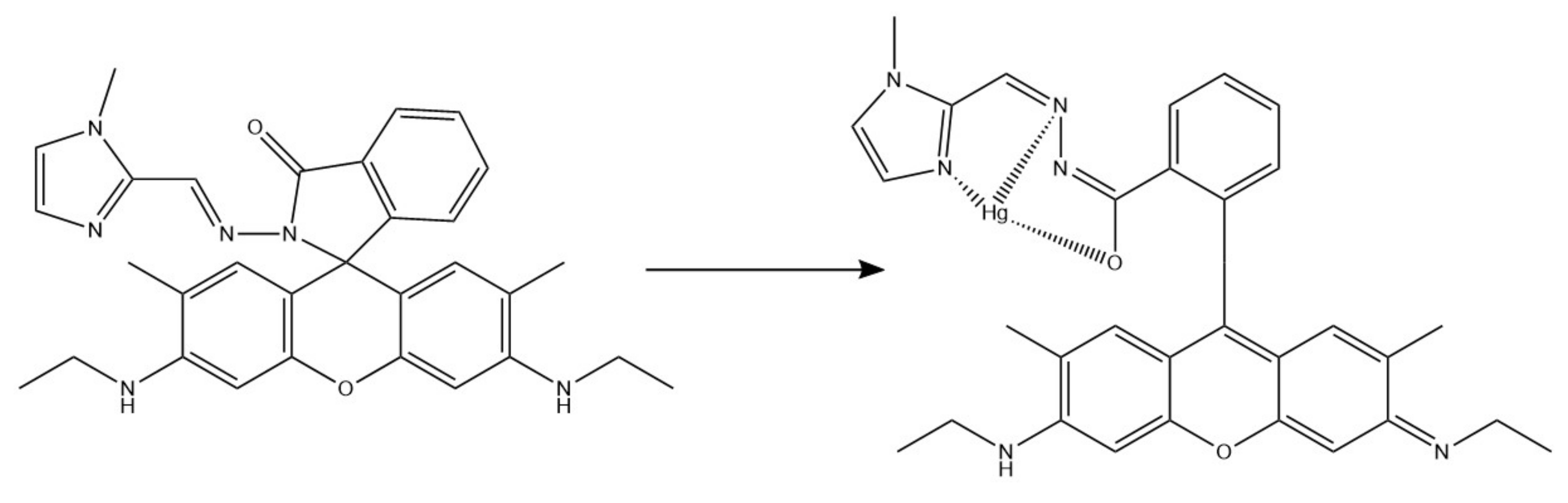
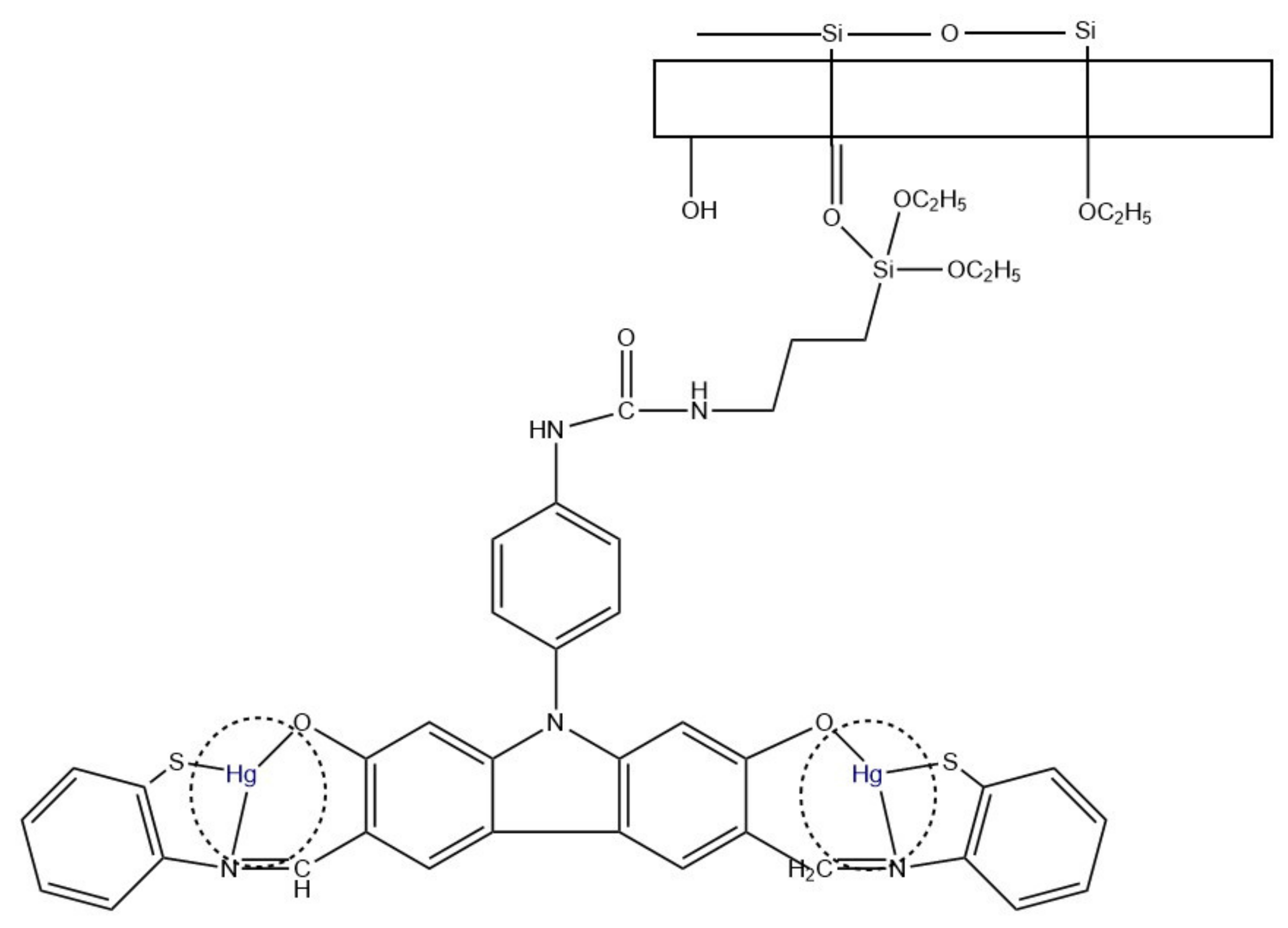
- Ehzari, H.; Safari, M.; Shahlaei, M. A new sensing strategy based on thymine bases–Hg2+–methylene blue coordination on the electrospun PES–QDs platform for detection of Hg2+ in fruit juice samples. J. Iran. Chem. Soc. 2019, 16, 2269–2279. [Google Scholar] [CrossRef]
- Xie, H.; Niu, Y.; Deng, Y.; Cheng, H.; Ruan, C.; Li, G.; Sun, W. Electrochemical aptamer sensor for highly sensitive detection of mercury ion with Au/Pt@carbon nanofiber-modified electrode. J. Chin. Chem. Soc. 2021, 68, 114–120. [Google Scholar] [CrossRef]
- Teodoro, K.B.R.; Migliorini, F.L.; Facure, M.H.M.; Correa, D.S. Conductive electrospun nanofibers containing cellulose nanowhiskers and reduced graphene oxide for the electrochemical detection of mercury(II). Carbohydr. Polym. 2019, 207, 747–754. [Google Scholar] [CrossRef]
- Teodoro, K.B.R.; Migliorini, F.L.; Facure, M.H.M.; Correa, D.S. Corrigendum to “Conductive electrospun nanofibers containing cellulose nanowhiskers and reduced graphene oxide for the electrochemical detection of mercury (II)” (Carbohydrate Polymers (2019) 207 (747–754), (S0144861718314565), (10.1016/j.carbpol.2018.12. Carbohydr. Polym. 2020, 229, 115495. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, X. Preparation of nitrogen doped carbon nanofibers for electrochemical determination of Cd(II) and Pb(II) ions. Int. J. Electrochem. Sci. 2020, 15, 9838–9848. [Google Scholar] [CrossRef]
- Oliveira, V.H.B.; Rechotnek, F.; da Silva, E.P.; de Sousa Marques, V.; Rubira, A.F.; Silva, R.; Lourenço, S.A.; Muniz, E.C. A sensitive electrochemical sensor for Pb2+ ions based on ZnO nanofibers functionalized by L-cysteine. J. Mol. Liq. 2020, 309, 113041. [Google Scholar] [CrossRef]
- Tang, Q.; Zhu, G.; Ge, Y.; Yang, J.; Huang, M.; Liu, J. AuNPs-polyaniline nanosheet array on carbon nanofiber for the determination of As(III). J. Electroanal. Chem. 2020, 873, 114381. [Google Scholar] [CrossRef]
- Rao, P.G.; Saritha, B.; Siva Rao, T. Colorimetric and turn-on Fluorescence Chemosensor for Hg 2+ Ion Detection in Aqueous Media. J. Fluoresc. 2019, 29, 353–360. [Google Scholar] [CrossRef]
- Tahvili, A.; Khalil Poush, M.; Ahmed, M.; Parsaee, Z. New efficient inorganic-organic nanofibers electrospun membrane for fluorescence detection and removal of mercury (II) ions. J. Mol. Struct. 2019, 1179, 242–251. [Google Scholar] [CrossRef]
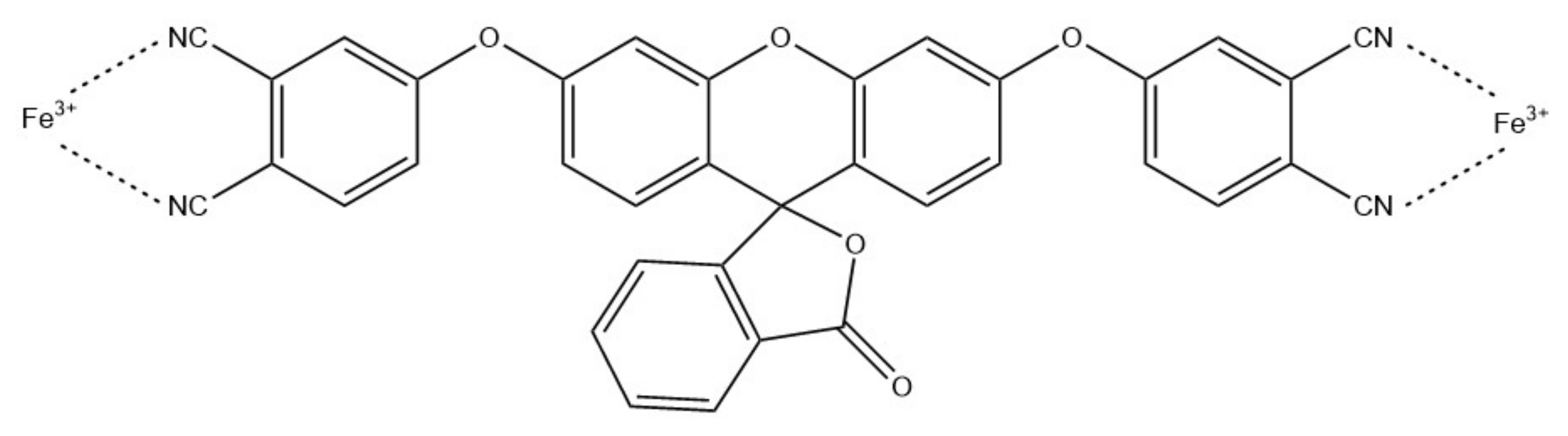
- Rijin, K.K.; Sagitha, P.; Amitha, G.S.; Vasudevan, S.; Sujith, A. 4,4′-Fluoresceinoxy bisphthalonitrile (FPN)-incorporated polycaprolactone electrospun membranes: A portable sensor strip for detection of Fe3+ ions. J. Mater. Sci. 2019, 54, 13433–13444. [Google Scholar] [CrossRef]
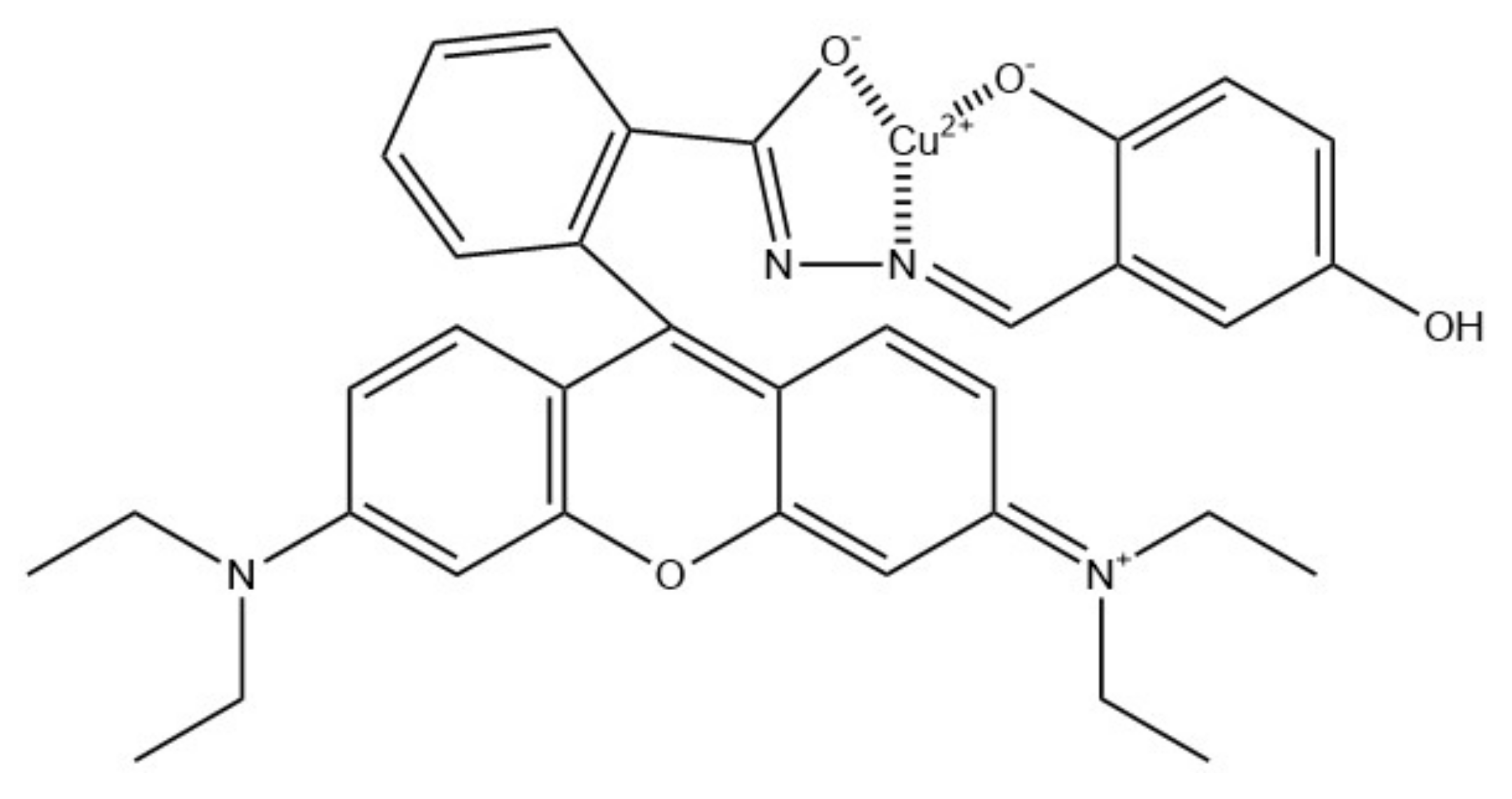
- Zhang, C.; Li, H.; Yu, Q.; Jia, L.; Wan, L.Y. Poly(aspartic acid) Electrospun Nanofiber Hydrogel Membrane-Based Reusable Colorimetric Sensor for Cu(II) and Fe(III) Detection. ACS Omega 2019, 4, 14633–14639. [Google Scholar] [CrossRef] [PubMed]
- Tungsombatvisit, N.; Inprasit, T.; Rohmawati, D.; Pisitsak, P. Rhodamine Derivative- Based Cellulose Acetate Electrospun Colorimetric Sensor for Cu2+ Sensing in Water: Effects of Alkaline Treatment. Fibers Polym. 2019, 20, 481–489. [Google Scholar] [CrossRef]
- Han, T.; Yuan, Y.; Kang, H.; Zhang, Y.; Dong, L. Ultrafast, sensitive and visual sensing of copper ions by a dual-fluorescent film based on quantum dots. J. Mater. Chem. C 2019, 7, 14904–14912. [Google Scholar] [CrossRef]
- Han, T.; Kang, H.; Ye, S.; Yuan, Y.; Zhang, Y.; Dong, L. Ultra-stable fluorescent film sensor based on quantum dots for the real-time detection of Cu2+. Sci. Total Environ. 2020, 746, 141412. [Google Scholar] [CrossRef]
- Li, X.; Wu, Y.; Niu, J.; Jiang, D.; Xiao, D.; Zhou, C. One-step sensitive thrombin detection based on a nanofibrous sensing platform. J. Mater. Chem. B 2019, 7, 5161–5169. [Google Scholar] [CrossRef]
- Rostami, A.; Hadjizadeh, A.; Mahshid, S. Colorimetric determination of dopamine using an electrospun nanofibrous membrane decorated with gold nanoparticles. J. Mater. Sci. 2020, 55, 7969–7980. [Google Scholar] [CrossRef]
- Zhai, M.Y.; Feng, K.; Hu, T.G.; Zong, M.H.; Wu, H. Development of a novel nano-based detection card by electrospinning for rapid and sensitive analysis of pesticide residues. J. Sci. Food Agric. 2020, 100, 4400–4408. [Google Scholar] [CrossRef] [PubMed]
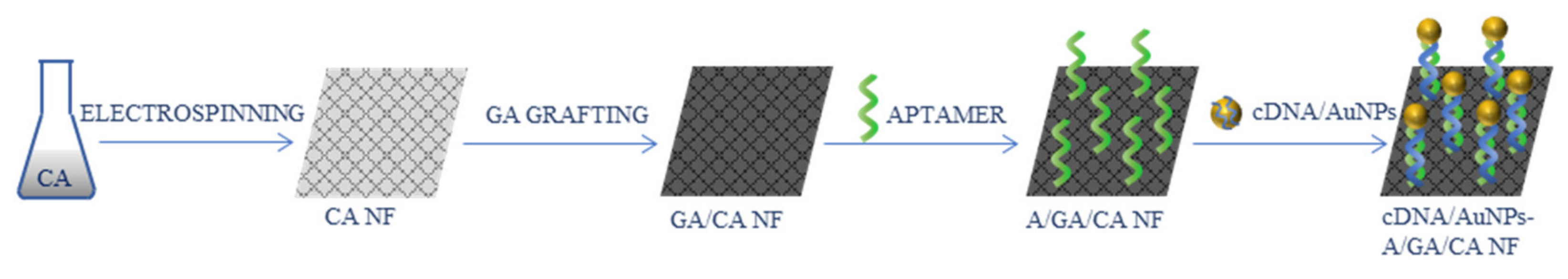
- Abedalwafa, M.A.; Tang, Z.; Qiao, Y.; Mei, Q.; Yang, G.; Li, Y.; Wang, L. An aptasensor strip-based colorimetric determination method for kanamycin using cellulose acetate nanofibers decorated DNA–gold nanoparticle bioconjugates. Microchim. Acta 2020, 187, 360. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.P.; Jannah, F.; Bae, K.; Kim, J.M. A colorimetric and fluorometric polydiacetylene biothiol sensor based on decomposition of a pyridine-mercury complex. Sens. Actuators B Chem. 2020, 309, 127771. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Zhang, J.C.; Yan, J.S.; Zhu, H.; Liu, J.J.; Li, R.; Ramakrishna, S.; Long, Y.Z. Ultrasensitive and reusable upconversion-luminescence nanofibrous indicator paper for in-situ dual detection of single droplet. Chem. Eng. J. 2020, 382, 122779. [Google Scholar] [CrossRef]
- Currie, S.; Shariatzadeh, F.J.; Singh, H.; Logsetty, S.; Liu, S. Highly Sensitive Bacteria-Responsive Membranes Consisting of Core-Shell Polyurethane Polyvinylpyrrolidone Electrospun Nanofibers for In Situ Detection of Bacterial Infections. ACS Appl. Mater. Interfaces 2020, 12, 45859–45872. [Google Scholar] [CrossRef] [PubMed]






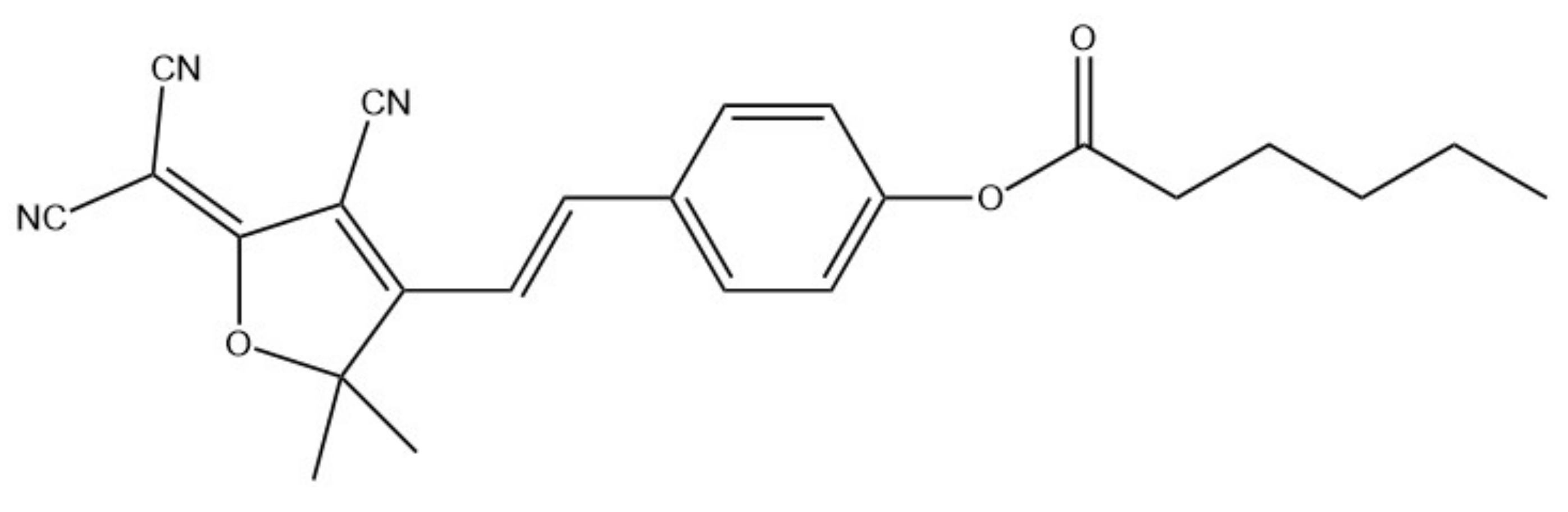
| Sensor | Linear Range | LOD | Ref. |
|---|---|---|---|
| PAN/PPy/PPy3COOH | 20 nM–2 μM | 2 nM | [37] |
| NiMoO4/CNF | 0.0003–4.5 mM | 50 nM | [38] |
| Cu-nanoflower@AuNPs-GO NFs | 0.001–0.1 mM | 0.018 μM | [39] |
| Ni/CoO CNF | 0.25–600 μM | 0.03 μM | [40] |
| CNF@Ni-Co LDH | 1–2000 μM | 0.03 μM | [41] |
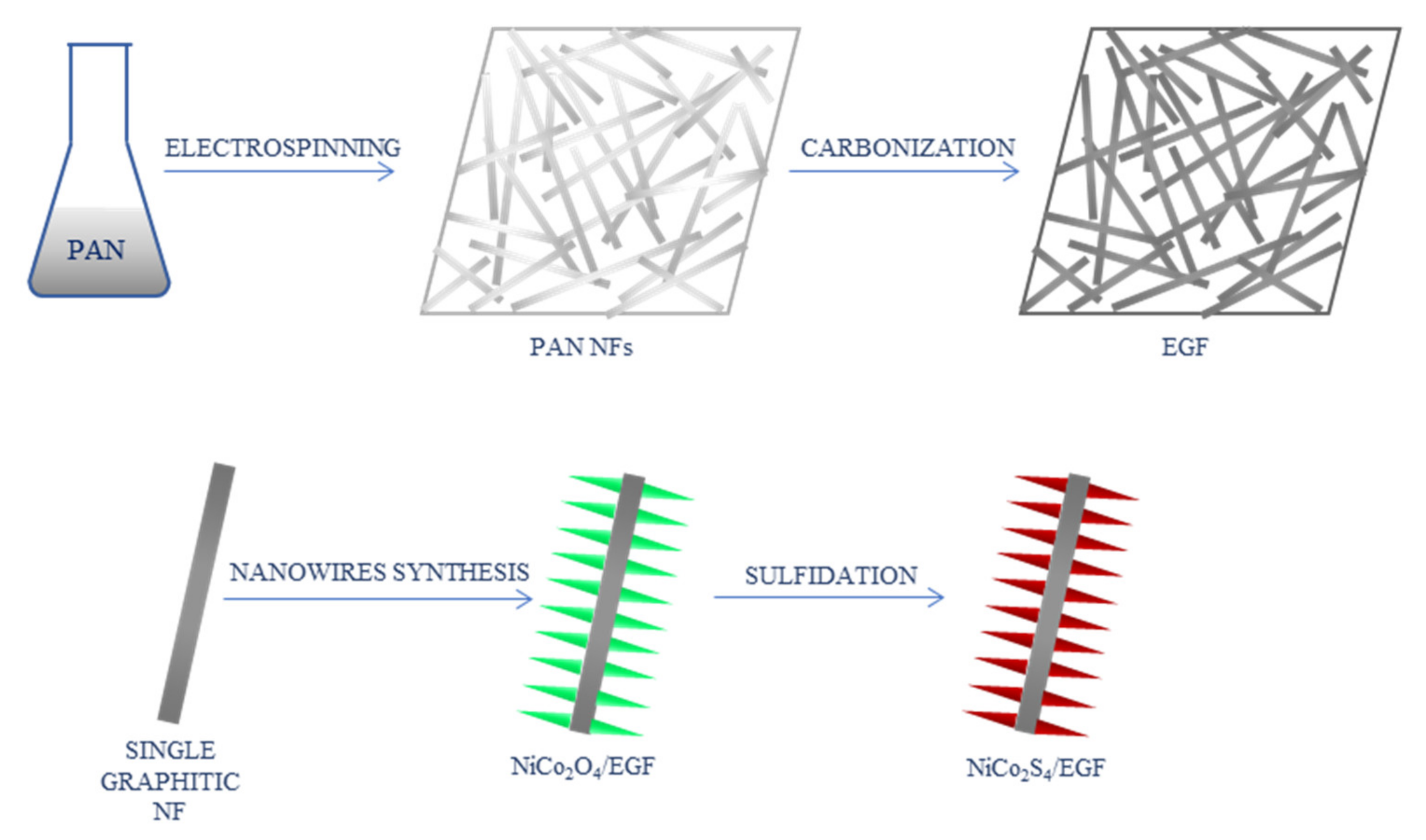
| NiCo2O4/EGF | 0.0005–3.571 mM | 0.167 μM | [42] |
| Ni/CNF | 2 μM–5 mM | 0.57 μM | [43] |
| CA/CS | 5.0 μM–0.75 mM | 4.8 μM | [44] |
| CS/GO | 0.05–20 mM | 0.02 mM | [45] |
| PEI/PVA | 2–8 mM, 10–30 mM | 0.3 mM | [46] |
| SnO2 multiporous NFs | 0.5–5 mM | 0.05 mM | [47] |
| Cellulose/β-CD | 0–1 mM | 9.35 × 10−5 M | [48] |
| Analyte | Sensor | Linear Range | LOD | Ref. |
|---|---|---|---|---|
| Dopamine | PANi/CQDs | 10–90 μM | 0.1013 μM | [49] |
| Dopamine | OLC-CNF | - | 1.42 μM | [50] |
| Streptavidin | PS-b-PMMA | 10 fg/mL–10 ng/mL | 0.37 fg/mL | [51] |
| 25-OHD3 | CA | 10–100 ng/mL | 10 ng/mL | [52] |
| LD and UA | MoS2 NSA/CNF | 1–60 μM | 1 μM | [54] |
| MUC1 cancer marker | Honey/PVA + AuNPs + MWCNTs | 5–115 nM | 2.7 nM | [55] |
| Her-2 cancer marker | Ab/MWCNT/Cys/ | 5–80 ng/mL | 0.45 ng/mL | [56] |
| AuNP/CNF | ||||
| Nicotine | MWCNTs/CS | 0.1–100 μM | 30 nM | [57] |
| Progesterone | GQDs-NiO-Au NFs/MWCNTs | 0.01–1000 nM | 1.86 pM | [58] |
| ZEN | PAN | 5–30 nM 60–100 nM | 1.66 nM | [59] |
| OTA | Silanized cellulose | 0.002–2 ng/mL | 0.81 pg/mL | [60] |
| Malathion | rGO/PA6/PPy | 0.5–20 μg/mL | 0.8 ng/mL | [61] |
| VCT | CNF | 10−13–10−5 g/mL | 0.12 pg/mL | [62] |
| CEA | Honey/PVA + AuNPs + MWCNTs | 0.4–125 ng/mL | 0.09 ng/mL | [63] |
| COX-2 | polyaniline | - | 0.01 pg/mL | [64] |
| LD | Graphene/CNF | 1–60 μM | 1 μM | [65] |
| Analyte | Sensor | Linear Range | LOD | Ref. |
|---|---|---|---|---|
| Penicillin | AuNPs/CNF | 1–400 ng/ml | 0.6 ng/ml | [66] |
| Colchicine | MIL/CuO/CNF | 1–100 nM | 0.25 nM | [67] |
| APAP | SnO2-CNF | 0.5–700 μM | 0.086 μM | [68] |
| p-HAP | 0.2–50 μM | 0.033 μM | ||
| Morphine | Magnetic NFs | 0.033–245 μM | 1.9 nM | [69] |
| Methotrexate | CuCr2O4/CuO | 0.1–300 μM | 25 nM | [70] |
| Tramadol | CNF | 0.05–100 nM | 0.016 nM | [71] |
| Metronidazole | CNF-NiCo-LDH | 3–57 nM | 0.13 nM | [72] |
| Analyte | Sensor | Linear Range | LOD | Ref. |
|---|---|---|---|---|
| Hg2+ | PES/QDs | 0.1–150 nM | 0.02 nM | [77] |
| Hg2+ | AuNPs/PtNPs/CNF | 1 fM–1 μM | 0.33 fM | [78] |
| Hg2+ | CNW/GO/PA6 | 2.5–200 μM | 0.52 μM | [79,80] |
| Cd2+ | ZIF-8NPs/N-doped CNF | 2–100 μg/L | 1.11 μg/L | [81] |
| Pb2+ | 1–100 μg/L | 0.72 μg/L | ||
| Pb2+ | L-Cys/ZnO | 10–140 μg/L | 0.397 μg/L | [82] |
| As3+ | AuNPs/PANi/Fe-CNF | 0.07–5.34 μM | 6.67 nM | [83] |
| Analyte | Sensor | LOD | Ref. |
|---|---|---|---|
| Hg2+ | RIM/polyurethane | - | [84] |
| Hg2+ | PVA/TEOS/S | 0.018 ng/ml | [85] |
| Fe3+ | FPN/PCL | 2.94 nM | [86] |
| Fe3+ | PASP | 0.1 mg/mL | [87] |
| Cu2+ | 0.3 mg/ml | ||
| Cu2+ | RBD/CA | 0.28 mM | [88] |
| Cu2+ | QDs/PEI/PVDF | 2 μM | [89] |
| Cu2+ | QDs/PA6 | 10 μM | [90] |
| Analyte | Sensor | LOD | Ref. |
|---|---|---|---|
| Thrombin | DNA/PS | 1 pM | [91] |
| Dopamine | CQDs/PANi | 0.0801 µM | [49] |
| Dopamine | AuNPs/PET | 0.5 µM | [92] |
| Pesticides | AChE/PVA, IA/PVA | 0.02 mg/L | [93] |
| Kanamycin | cDNA/AuNPs/A/GA/CA | 2.5 nM | [94] |
| Biothiols | DA-Hg-DA/PEO | - | [95] |
| Riboflavin, pH | Nanorods/Ag@SiO2/PS | 0.03 µM | [96] |
| Bacteria | HCy + PU, PU/PVP, PU/PEG | 105 CFU/cm2, | [97] |
| 106 CFU/cm2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Halicka, K.; Cabaj, J. Electrospun Nanofibers for Sensing and Biosensing Applications—A Review. Int. J. Mol. Sci. 2021, 22, 6357. https://doi.org/10.3390/ijms22126357
Halicka K, Cabaj J. Electrospun Nanofibers for Sensing and Biosensing Applications—A Review. International Journal of Molecular Sciences. 2021; 22(12):6357. https://doi.org/10.3390/ijms22126357
Chicago/Turabian StyleHalicka, Kinga, and Joanna Cabaj. 2021. "Electrospun Nanofibers for Sensing and Biosensing Applications—A Review" International Journal of Molecular Sciences 22, no. 12: 6357. https://doi.org/10.3390/ijms22126357
APA StyleHalicka, K., & Cabaj, J. (2021). Electrospun Nanofibers for Sensing and Biosensing Applications—A Review. International Journal of Molecular Sciences, 22(12), 6357. https://doi.org/10.3390/ijms22126357







