Toxicity and Antioxidant Activity of Fullerenol C60,70 with Low Number of Oxygen Substituents
Abstract
:1. Introduction
2. Results and Discussion
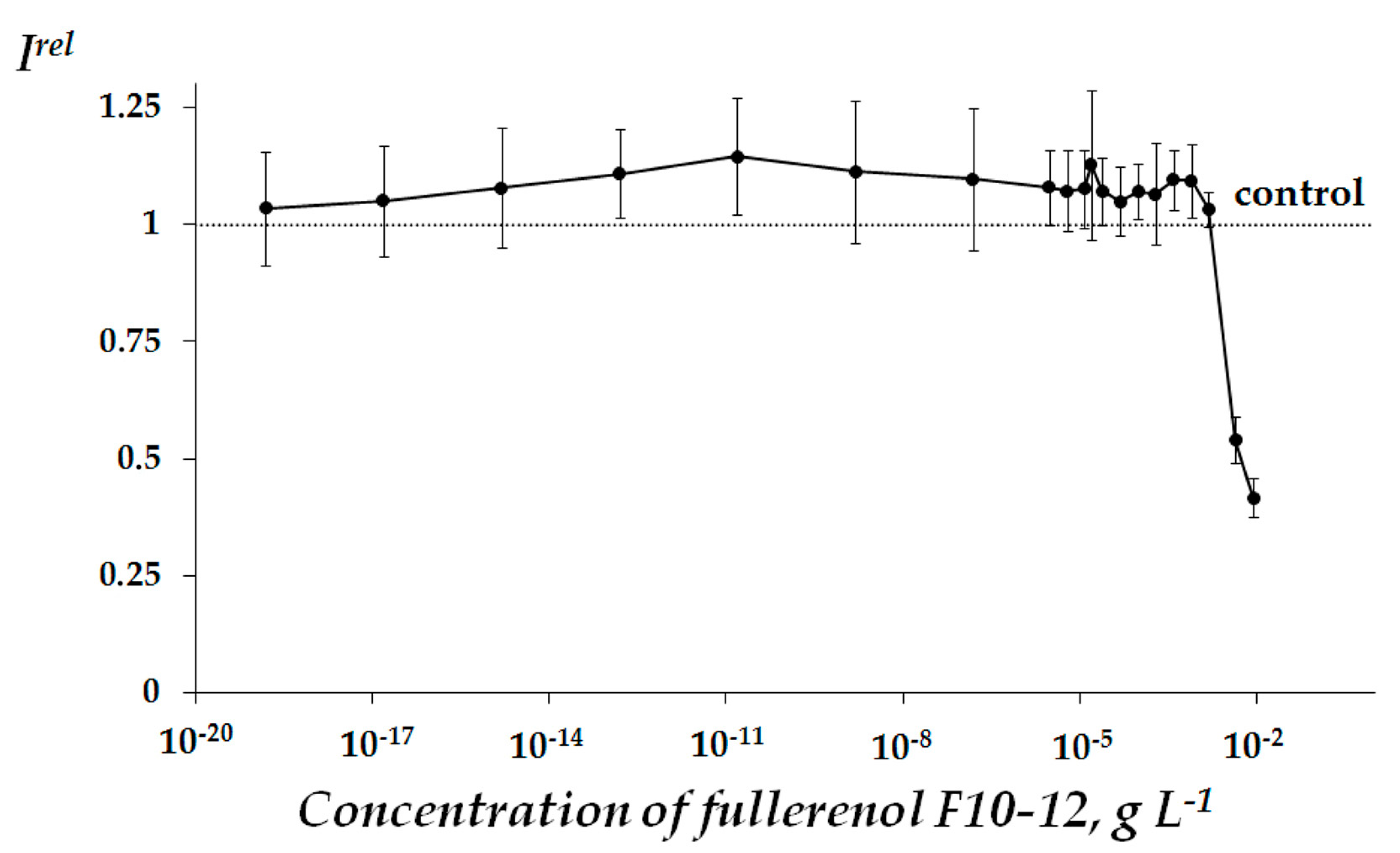
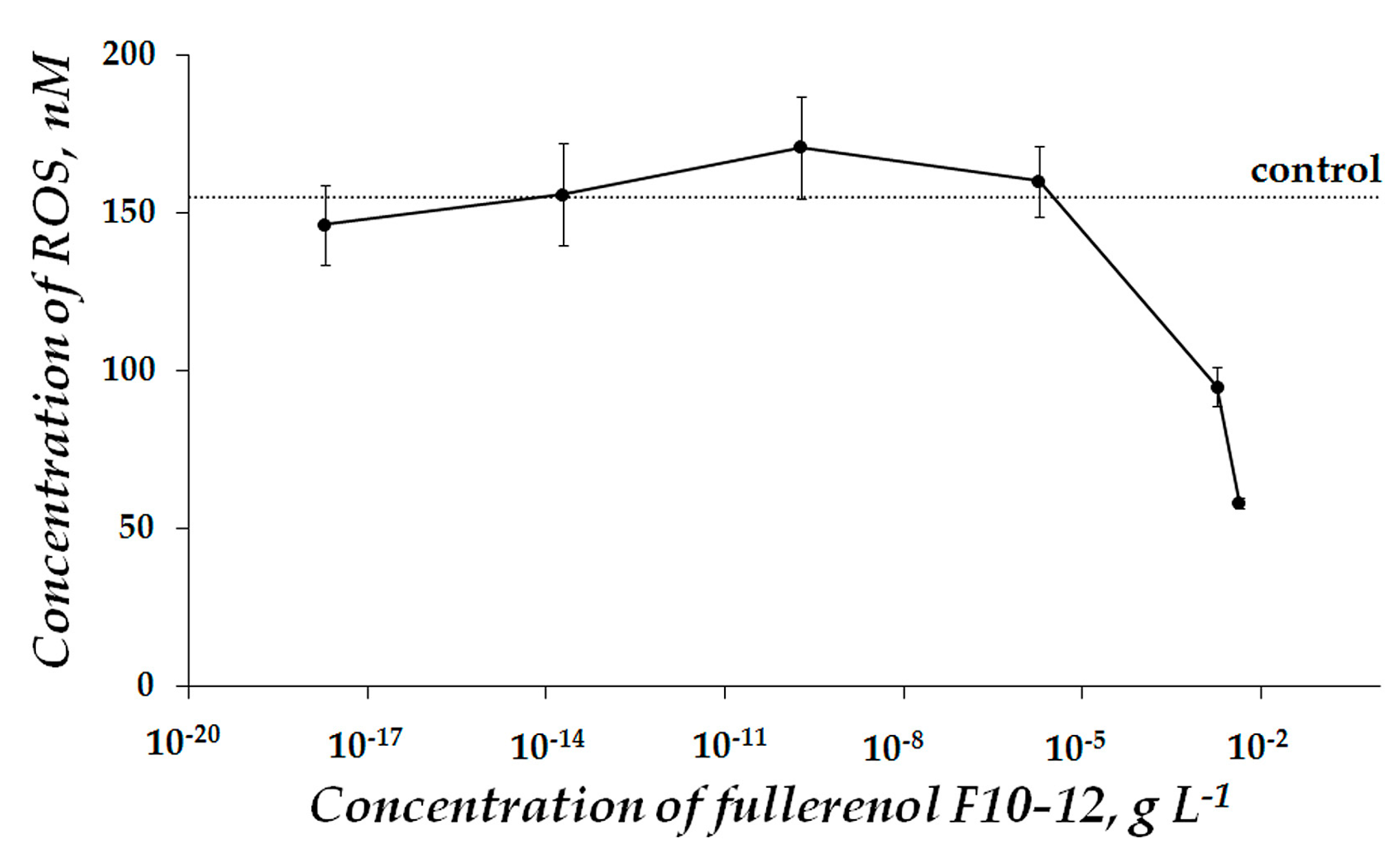
2.1. Fullerenol Toxicity
2.2. Fullerenol Antioxidant Activity
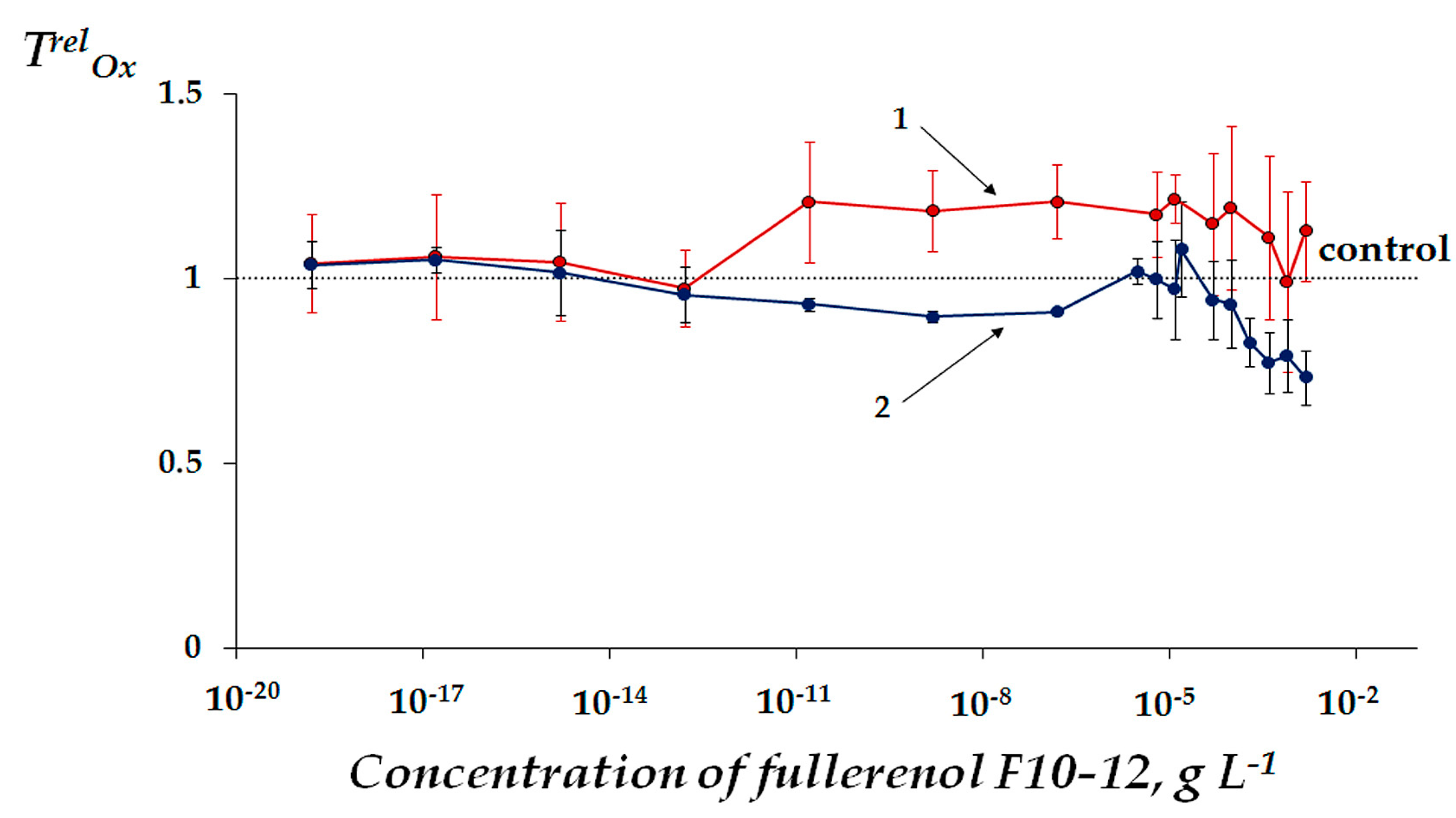
2.2.1. Change of General Toxicity under Conditions of Oxidative Exposure
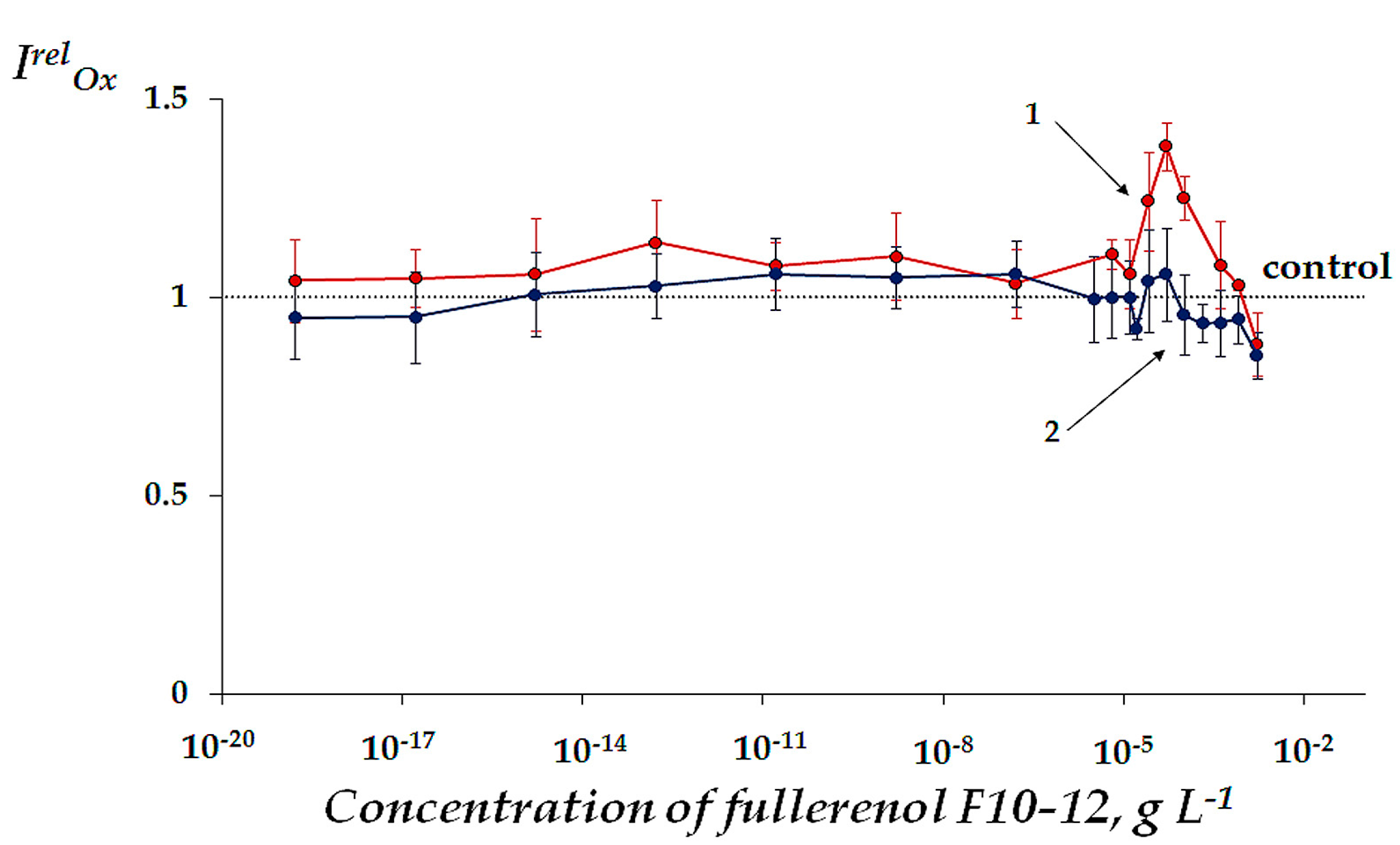
2.2.2. Change of Oxidative Toxicity under Conditions of Oxidative Exposure
3. Materials and Methods
3.1. Preparation and Characterization of Fullerenols
3.2. Bioluminescence Enzymatic Assay and Experimental Data Processing
3.3. Luminol Chemiluminescence Assay
3.4. Statistical Processing
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EC50 | effective concentration of oxidizers or fullerenols inhibited bioluminescence intensity by 50% |
| F10-12 | C60,70Ox(OH)y, where x + y = 10–12, y—even |
| F24-28 | C60,70Ox(OH)y, where x + y = 24-28 |
| F40-42 | C60,70Ox(OH)y, where x + y = 40-42 |
| FMN | flavinmononucleotide |
| NADH | nicotinamide adenine dinucleotide, disodium salt, reduced |
| ROS | reactive oxygen species |
References
- Kroto, H.W.; Health, J.R.; O’Brien, S.C.; Curl, R.F.; Smalley, R.E. C60: Buckminsterfullerene. Nature 1985, 318, 162–163. [Google Scholar] [CrossRef]
- Smalley, R.E. Discovering the fullerenes. Rev. Mod. Phys. 1997, 69, 723–730. [Google Scholar] [CrossRef]
- Krätschmer, W.; Lamb, L.D.; Fostiropoulos, K.; Huffman, D.R. Solid C60: A new form of carbon. Nature 1990, 347, 354–358. [Google Scholar] [CrossRef]
- Bosi, S.; Ros, T.D.; Spalluto, G.; Prato, M. Fullerene derivatives: An attractive tool for biological applications. Eur. J. Med. Chem. 2003, 38, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Afreen, S.; Kokubo, K.; Muthoosamy, K.; Manickam, S. Hydration or hydroxylation: Direct synthesis of fullerenol from pristine fullerene [C60] via acoustic cavitation in the presence of hydrogen peroxide. RSC Adv. 2017, 7, 31930–31939. [Google Scholar] [CrossRef] [Green Version]
- Smalley, R.E.; Yakobson, B.I. The future of the fullerenes. Solid State Commun. 1998, 107, 597–606. [Google Scholar] [CrossRef]
- Krusic, P.J.; Wasserman, E.; Keizer, P.N.; Morton, J.R.; Preston, K.F. Radical reactions of C60. Science 1991, 254, 1183–1185. [Google Scholar] [CrossRef]
- Taylor, R.; Walton, D.R.M. The chemistry of fullerenes. Nature 1993, 363, 685–693. [Google Scholar] [CrossRef]
- Hirsh, A. The Chemistry of Fullerenes, 1st ed.; George Thieme: New York, NY, USA, 1994. [Google Scholar]
- Diederich, F.; Thilgen, C. Covalent fullerene chemistry. Science 1996, 271, 317–324. [Google Scholar] [CrossRef]
- Prato, M. Fullerene Materials. In Fullerenes and Related Structures, 1st ed.; Hirsch, A., Ed.; Springer: Berlin/Heidelberg, Germany, 1999; Volume 199, pp. 173–187. [Google Scholar] [CrossRef]
- Kadish, K.M.; Ruoff, R.S. Fullerenes: Chemistry, Physics, and Technology; John Wiley& Sons: New York, NY, USA, 2000; p. 984. [Google Scholar]
- Hou, J.G.; Zhao, A.D.; Huang, T.; Lu, S. C60-Based Materials. In Encyclopedia of Nanoscience and Nanotechnology; Nalwa, H.S., Ed.; American Scientific: New York, NY, USA, 2014; Volume 1, pp. 409–474. [Google Scholar]
- Martin, N. New challenges in fullerene chemistry. Chem. Commun. 2006, 318, 2093–2104. [Google Scholar] [CrossRef]
- Moussa, F. Fullerene and derivatives for biomedical applications. Nanobiomaterials 2018, 113–136. [Google Scholar] [CrossRef]
- Satoh, M.; Takayanagi, I. Pharmacological studies on fullerene (C60), a novel carbon allotrope, and its derivatives. J. Pharmacol. Sci. 2006, 100, 513–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markovic, Z.; Trajkovic, V. Biomedical potential of the reactive oxygen species generation and quenching by fullerenes (C60). Biomaterials 2008, 29, 3561–3573. [Google Scholar] [CrossRef]
- Rondags, A.; Yuen, W.Y.; Jonkman, M.F.; Horvath, B. Fullerene C60 with cytoprotective and cytotoxic potential: Prospects as a novel treatment agent in Dermatology? Exp. Dermatol. 2016, 26, 220–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Shen, X.; Ma, R.; Hou, Y.; Qian, Y.; Fan, C. Biological and biocompatible characteristics of fullerenols nanomaterials for tissue engineering. Histol. Histopathol. 2021, 18316. [Google Scholar] [CrossRef]
- Sharoyko, V.V.; Ageev, S.V.; Podolsky, N.E.; Petrov, A.V.; Litasovc, E.V.; Vlasov, T.D.; Vasina, L.V.; Murin, I.V.; Piotrovskiy, L.B.; Semenov, K.N. Biologically active water-soluble fullerene adducts: Das Glasperlenspiel (by H. Hesse)? J. Mol. Liq. 2021, 323, 114990. [Google Scholar] [CrossRef]
- Jovic, D.; Jacevic, V.; Kuca, K.; Borišev, I.; Mrdjanovic, J.; Petrovic, D.; Seke, M.; Djordjevic, A. The puzzling potential of carbon nanomaterials: General properties, application, and toxicity. Nanomaterials 2020, 10, 1508. [Google Scholar] [CrossRef]
- Andrievsky, G.V.; Bruskov, V.I.; Tykhomyrov, A.A.; Gudkov, S.V. Peculiarities of the antioxidant and radioprotective effects of hydrated C60 fullerene nanostructures in vitro and in vivo. Free Radic. Biol. Med. 2009, 47, 786–793. [Google Scholar] [CrossRef]
- Andrievsky, G.V.; Klochkov, V.K.; Derevyanchenko, L.I. Is C60 fullerene molecule toxic?! Fuller. Nanotub. Carbon Nanostructures 2005, 13, 363–376. [Google Scholar] [CrossRef]
- Nakamura, E.; Isobe, H. Functionalized fullerenes in water. The first 10 years of their chemistry, biology, and nanoscience. Acc. Chem. Res. 2003, 36, 807–815. [Google Scholar] [CrossRef]
- Eropkin, M.Y.; Melenevskaya, E.Y.; Nasonova, K.V.; Bryazzhikova, T.S.; Eropkina, E.M.; Danilenko, D.M.; Kiselev, O.I. Synthesis and biological activity of fullerenols with various contents of hydroxyl groups. Pharm. Chem. J. 2013, 47, 87–91. [Google Scholar] [CrossRef]
- Nakamura, H.; Nozaki, Y.; Koizumi, Y.; Watano, S. Effect of number of hydroxyl groups of fullerenol C60(OH)n on its interaction with cell membrane. J. Taiwan Inst. Chem. Eng. 2018, 90, 18–24. [Google Scholar] [CrossRef]
- Petrovic, D.; Seke, M.; Srdjenovic, B.; Djordjevic, A. Applications of anti/prooxidant fullerenes in nanomedicine along with fullerenes influence on the immune system. J. Nanomater. 2015, 2015, 565638. [Google Scholar] [CrossRef] [Green Version]
- Grebowski, J.; Kazmierska, P.; Krokosz, A. Fullerenols as a new therapeutic approach in nanomedicine. BioMed Res. Int. 2013, 2013, 751913. [Google Scholar] [CrossRef] [Green Version]
- Cai, X.; Hao, J.; Zhang, X.; Yu, B.; Ren, J.; Luo, C.; Li, Q.; Huang, Q.; Shi, X.; Li, W.; et al. The polyhydroxylated fullerene derivative C60(OH)24 protects mice from ionizing-radiation-induced immune and mitochondrial dysfunction. Toxicol. Appl. Pharmacol. 2010, 243, 27–34. [Google Scholar] [CrossRef]
- Injac, R.; Prijatelj, M.; Strukelj, B. Fullerenol nanoparticles: Toxicity and antioxidant activity. Methods Mol. Biol. 2013, 1028, 75–100. [Google Scholar] [CrossRef]
- Chaban, V.V.; Fileti, E.E. Which fullerenols are water soluble? Systematic atomistic investigation. N. J. Chem. 2017, 41, 184–189. [Google Scholar] [CrossRef]
- Saathoff, J.G.; Inman, A.O.; Xia, X.R.; Riviere, J.E.; Monteiro-Riviere, N.A. In vitro toxicity assessment of three hydroxylated fullerenes in human skin cells. Toxicol. In Vitro 2011, 25, 2105–2112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovel, E.S.; Sachkova, A.S.; Vnukova, N.G.; Churilov, G.N.; Knyazeva, E.M.; Kudryasheva, N.S. Antioxidant activity and toxicity of fullerenols via bioluminescence signaling: Role of oxygen substituents. Int. J. Mol. Sci. 2019, 20, 2324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sergeeva, V.; Kraevaya, O.A.; Ershova, E.; Kameneva, L.; Malinovskaya, E.; Dolgikh, O.; Konkova, M.; Voronov, I.; Zhilenkov, A.; Veiko, N.; et al. Antioxidant properties of fullerene derivatives depend on their chemical structure: A study of two fullerene derivatives on HELFs. Oxid. Med. Cell. Longev. 2019, 2019, 4398695. [Google Scholar] [CrossRef]
- Iohara, D.; Umezaki, Y.; Anraku, M.; Uekama, K.; Hirayama, F. In Vitro and In Vivo Evaluation of Hydrophilic C60(OH)10/2-Hydroxypropyl-β-cyclodextrin Nanoparticles as an Antioxidant. J. Pharm. Sci. 2016, 105, 2959–2965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bulich, A.A.; Isenberg, D.L. Use of the luminescent bacterial system for rapid assessment of aquatic toxicity. ISA Trans. 1981, 20, 29–33. [Google Scholar] [PubMed]
- Girotti, S.; Ferri, E.N.; Fumo, M.G.; Maiolini, E. Monitoring of environmental pollutants by bioluminescent bacteria. Anal. Chim. Acta 2008, 608, 2–29. [Google Scholar] [CrossRef] [PubMed]
- Roda, A.; Pasini, P.; Mirasoni, M.; Michchelini, E.; Guardigli, M. Biotechnological application of bioluminescence and chemiluminescence. Trends Biotech. 2004, 22, 295–303. [Google Scholar] [CrossRef]
- Abbas, M.; Adil, M.; Ehtisham-Ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Tahir, M.A.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef]
- Ismailov, A.D.; Aleskerova, L.E. Photobiosensors containing luminescent bacteria. Biochemistry 2015, 80, 733–744. [Google Scholar] [CrossRef]
- Kratasyuk, V.A.; Esimbekova, E.N. Applications of luminous bacteria enzymes in toxicology. Comb. Chem. High Throughput Screen. 2015, 18, 952–959. [Google Scholar] [CrossRef] [Green Version]
- Vetrova, E.V.; Kudryasheva, N.S.; Kratasyuk, V.A. Redox compounds influence on the NAD(P)H:FMN-oxidoreductase-luciferase bioluminescent system. Photochem. Photobiol. Sci. 2007, 6, 35–40. [Google Scholar] [CrossRef]
- Kudryasheva, N.; Vetrova, E.; Kuznetsov, A.; Kratasyuk, V.; Stom, D. Bioluminescent assays: Effects of quinones and phenols. Ecotoxicol. Environ. Saf. 2002, 53, 221–225. [Google Scholar] [CrossRef]
- Tarasova, A.S.; Stom, D.I.; Kudryasheva, N.S. Effect of humic substances on toxicity of inorganic oxidizer bioluminescent monitoring. Environ. Toxicol. Chem. 2011, 30, 1013–1017. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Tarasova, A.S. Pollutant toxicity and detoxification by humic substances: Mechanisms and quantitative assessment via luminescent biomonitoring. Environ. Sci. Pollut. Res. 2015, 22, 155–167. [Google Scholar] [CrossRef]
- Tarasova, A.S.; Kislan, S.L.; Fedorova, E.S.; Kuznetsov, A.M.; Mogilnaya, O.A.; Stom, D.I.; Kudryasheva, N.S. Bioluminescence as a tool for studying detoxification processes in metal salt solutions involving humic substances. J. Photochem. Photobiol. B 2012, 117, 164–170. [Google Scholar] [CrossRef]
- Tarasova, A.S.; Stom, D.I.; Kudryasheva, N.S. Antioxidant activity of humic substances via bioluminescent monitoring in vitro. Environ. Monit. Assess. 2015, 187, 89. [Google Scholar] [CrossRef] [PubMed]
- Kudryasheva, N.S.; Kovel, E.S.; Sachkova, A.S.; Vorobeva, A.A.; Isakova, V.G.; Churilov, G.N. Bioluminescent enzymatic assay as a tool for studying antioxidant activity and toxicity of bioactive compounds. Photochem. Photobiol. 2017, 93, 536–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Guseynov, O.A.; Bondar, A.A.; Dubinina, I.A.; Kudryasheva, N.S. On mechanism of antioxidant effect of fullerenols. Biochem. Biophys. Rep. 2017, 9, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Stom, D.I.; Kudryasheva, N.S. Biological activity of carbonic nano-structures—comparison via enzymatic bioassay. J. Soils Sediments 2019, 19, 2689–2696. [Google Scholar] [CrossRef] [Green Version]
- Kudryasheva, N.S.; Kovel, E.S. Monitoring of low-intensity exposures via luminescent bioassays of different complexity: Cells, enzyme reactions and fluorescent proteins. Int. J. Mol. Sci. 2019, 20, 4451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachkova, A.S.; Kovel, E.S.; Vorobeva, A.A.; Kudryasheva, N.S. Antioxidant activity of fullerenols. Bioluminescent monitoring in vitro. Procedia Technol. 2017, 27, 230–231. [Google Scholar] [CrossRef]
- Bondarenko, L.S.; Kovel, E.S.; Kydralieva, K.A.; Dzhardimalieva, G.I.; Illé, E.; Tombácz, E.; Kicheeva, A.G.; Kudryasheva, N.S. Effects of modified magnetite nanoparticles on bacterial cells and enzyme reactions. Nanomaterials 2020, 10, 1499. [Google Scholar] [CrossRef]
- McEwen, C.N.; McKay, R.G.; Larsen, B.S. C60 as a radical sponge. J. Am. Chem. Soc. 1992, 114, 4412–4414. [Google Scholar] [CrossRef]
- Brieger, K.; Schiavone, S.; Jr, F.J.M.; Krause, K.-H. Reactive oxygen species: From health to disease. Swiss Med. Wkly. 2012, 142, w13659. [Google Scholar] [CrossRef]
- Weyemi, U.; Dupuy, C. The emerging role of ROS-generating NADPH oxidase NOX4 in DNA-damage responses. Mutat. Res. 2012, 751, 77–81. [Google Scholar] [CrossRef]
- Sedelnikova, O.A.; Redon, C.E.; Dickey, J.S.; Nakamura, A.J.; Georgakilas, A.G.; Bonner, W.M. Role of oxidatively induced DNA lesions in human pathogenesis. Mutat. Res. 2010, 704, 152–159. [Google Scholar] [CrossRef] [Green Version]
- Lambeth, J.D. Nox enzymes, ROS, and chronic disease: An example of antagonistic pleiotropy. Free Radic. Biol. Med. 2007, 43, 332–347. [Google Scholar] [CrossRef] [Green Version]
- Remmel, N.N.; Titova, N.M.; Kratasyuk, V.A. Oxidative stress monitoring in biological samples by bioluminescent method. Bull. Exp. Biol. Med. 2003, 136, 209–211. [Google Scholar] [CrossRef]
- Alexandrova, M.; Rozhko, T.; Vydryakova, G.; Kudryasheva, N. Effect of americium-241 on luminous bacteria. Role of peroxides. J. Environ. Radioact. 2011, 102, 407–411. [Google Scholar] [CrossRef]
- Kudryasheva, N.S. Bioluminescence and exogenous compounds: Physico-chemical basis for bioluminescent assay. J. Photochem. Photobiol. B. 2006, 83, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Keshri, S. Insights into the structural and thermodynamic properties of fullerols [C60(OH)n, n = 12, 14, 16, 18, 20, 22, 24] in aqueous media. Fluid Phase Equilibr. 2020, 525, 112805. [Google Scholar] [CrossRef]
- Nemtseva, E.V.; Kudryasheva, N. The mechanism of electronic excitation in bacterial bioluminescent reaction. Uspekhi Khimii 2007, 76, 101–112. [Google Scholar]
- Lee, J.; Müller, F.; Visser, A.J.W.G. The sensitized bioluminescence mechanism of bacterial luciferase. Photochem. Photobiol. 2019, 95, 679–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rees, J.F.; Wergifosse, B.; Noiset, O.; Dubuisson, M.; Janssens, B.; Thompson, E.M. The origins of marine bioluminescence: Turning oxygen defense mechanisms into deep-sea communication tools. J. Exp. Biol. 1998, 201, 1211–1221. [Google Scholar] [CrossRef]
- Vetrova, E.V.; Kudryasheva, N.S.; Cheng, K.H. Effect of quinone on the fluorescence decay dynamics of endogenous flavin bound to bacterial luciferase. Biophys. Chem. 2009, 141, 59–65. [Google Scholar] [CrossRef]
- Vetrova, E.V.; Kudryasheva, N.S.; Visser, A.J.W.G.; Hoek, A. Characteristics of endogenous flavin fluorescence of Ph. leiognathi luciferase and V. fischeri NAD(P)H:FMN-oxidoreductase. Luminescence 2005, 20, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Meng, F.; Cui, H.; Linc, Y.; Wangc, G.; Wu, J. Ecotoxicity of phenol and cresols to aquatic organisms. Ecotoxicol. Environ. Saf. 2018, 157, 441–456. [Google Scholar] [CrossRef] [PubMed]
- Stasiuk, M.; Kozubek, A. Biological activity of phenolic lipids. Cell. Mol. Life Sci. 2010, 67, 841–860. [Google Scholar] [CrossRef] [PubMed]
- Kamnev, A.A.; Kovács, K.; Kuzmann, E.; Vértes, A. Application of Mössbauer spectroscopy for studying chemical effects of environmental factors on microbial signalling: Redox processes involving iron(III) and some microbial autoinducer molecules. J. Mol. Struct. 2009, 924–926, 131–137. [Google Scholar] [CrossRef]
- Kamnev, A.A.; Dykman, R.L.; Kovács, K.; Pankratov, A.N.; Tugarova, A.V.; Homonnay, Z.; Kuzmann, E. Redox interactions between structurally different alkylresorcinols and iron(III) in aqueous media: Frozen-solution 57Fe Mössbauer spectroscopic studies, redox kinetics and quantum chemical evaluation of the alkylresorcinol reactivities. Struct. Chem. 2014, 25, 649–657. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis: A fundamental concept in biology. Microb. Cell 2014, 1, 145–149. [Google Scholar] [CrossRef] [Green Version]
- Calabrese, E.J. Hormesis: Path and progression to significance. Int. J. Mol. Sci. 2018, 19, 2871. [Google Scholar] [CrossRef] [Green Version]
- Agathokleous, E.; Calabrese, E.J. A global environmental health perspective and optimisation of stress. Sci. Tot. Environ. 2020, 704, 135263. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Leso, V.; Fontana, L.; Calabrese, E.J. Nanoparticle exposure and hormetic dose–responses: An update. Int. J. Mol. Sci. 2018, 19, 805. [Google Scholar] [CrossRef] [Green Version]
- Kovel, E.S. Antioxidant and Toxic Properties of Fullerenes. Bioluminescent Monitoring. Master’s Thesis, Sibirskiy Federal’nyy Universitet, Krasnoyarsk, Russia, 2017. (In Russian). [Google Scholar]
- Fedorova, G.F.; Trofimov, A.V.; Vasil’ev, R.F.; Veprintsev, T.L. Peroxy-radical-mediated chemiluminescence: Mechanistic diversity and fundamentals for antioxidant assay. ARKIVOC 2006, 2007, 163–215. [Google Scholar] [CrossRef] [Green Version]
- Churilov, G.N.; Kratschmer, W.; Osipova, I.V.; Glushenko, G.A.; Vnukova, N.G.; Kolonenko, A.L.; Dudnik, A.I. Sinthesis of fullerenes in a high-frequency arc plasma under elevated helium pressure. Carbon 2013, 62, 389–392. [Google Scholar] [CrossRef]
- Churilov, G.N.; Popov, A.A.; Vnukova, N.G.; Dudnik, A.I.; Glushchenko, G.A.; Samoylova, N.A.; Dubinina, I.A.; Gulyaeva, U.E. A method and apparatus for high-throughput controlled synthesis of fullerenes and endohedral metal fullerenes. Tech. Phys. Lett. 2016, 42, 475–477. [Google Scholar] [CrossRef]
- Andrievsky, G.V.; Kosevich, M.V.; Vovk, O.M.; Shelkovsky, V.S.; Vashchenko, L.A. On the production of an aqueous colloidal solution of fullerenes. J.Chem. Soc. Chem. Commun. 1995, 12, 1281–1282. [Google Scholar] [CrossRef]
- Li, J.; Zhang, M.; Sun, B.; Xing, G.; Song, Y.; Guo, H.; Chang, Y.; Ge, Y.; Zhao, Y. Separation and purification of fullerenols for improved biocompatibility. Carbon 2012, 50, 460–469. [Google Scholar] [CrossRef]
- Li, J.; Wang, T.; Feng, Y.; Zhang, Y.; Zhen, M.; Shu, C.; Jiang, L.; Wang, Y.; Wang, C. A water-soluble gadolinium metallofullerenol: Facile preparation, magnetic properties and magnetic resonance imaging application. Dalton Trans. 2016, 45, 8696–8699. [Google Scholar] [CrossRef]
- Shilin, V.A.; Szhogina, A.A.; Suyasova, M.V.; Sedov, V.P.; Lebedev, V.T.; Kozlov, V.S. Fullerenes and fullerenols survival under irradiation. Nanosyst. Phys. Chem., Math. 2016, 7, 146–152. [Google Scholar] [CrossRef] [Green Version]
- Indeglia, P.A.; Georgieva, A.; Krishna, V.B.; Bonzongo, J.-C.J. Physicochemical characterization of fullerenol and fullerenol synthesis by-products prepared in alkaline media. J. Nanopart. Res. 2014, 16, 2599. [Google Scholar] [CrossRef]
- Georgieva, A.T.; Pappu, V.; Krishna, V.; Georgiev, P.G.; Ghiviriga, I.; Indeglia, P.; Xu, X.; Fan, Z.H.; Koopman, B.; Pardalos, P.M.; et al. Polyhydroxy fullerenes. J. Nanopart. Res. 2013, 15, 1690. [Google Scholar] [CrossRef]
- Kuznetsov, A.M.; Rodicheva, E.K.; Shilova, E.V. Bioassay based on lyophilized bacteria. Biotekhnologiya 1996, 9, 57–61. (In Russian) [Google Scholar]
- Fedorova, E.; Kudryasheva, N.; Kuznetsov, A.; Mogil’naya, O.; Stom, D. Bioluminescent monitoring of detoxification processes: Activity of humic substances in quinone solutions. J. Photochem. Photobiol. B 2007, 88, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Gmurman, V.E. Fundamentals of Probability Theory and Mathematical Statistics; Berenblut, I.I., Ed.; Iliffe Book Ltd.: London, UK, 1968; p. 249. [Google Scholar]
- Fedel, M. Hemocompatibility of Carbon Nanostructures. J. Carbon Res. 2020, 6, 12. [Google Scholar] [CrossRef] [Green Version]
- Johnson-Lyles, D.N.; Peifley, K.; Lockett, S.; Neun, B.W.; Hansen, M.; Clogston, J.; Stern, S.T.; McNeil, S.E. Fullerenol cytotoxicity in kidney cells is associated with cytoskeleton disruption, autophagic vacuole accumulation, and mitochondrial dysfunction. Toxicol. Appl. Pharmacol. 2010, 248, 249–258. [Google Scholar] [CrossRef] [Green Version]
- Isakovic, A.; Markovic, Z.; Todorovic-Markovic, B.; Nikolic, N.; Vranjes-Djuric, S.; Mirkovic, M.; Dramicanin, M.; Harhaji, L.; Raicevic, N.; Nikolic, Z.; et al. Distinct cytotoxic mechanisms of pristine versus hydroxylated fullerene. Toxicol. Sci. 2006, 91, 173–183. [Google Scholar] [CrossRef]
- Prylutska, S.V.; Matyshevska, O.P.; Golub, A.A.; Prylutskyy, Y.I.; Potebnya, G.P.; Ritter, U.; Scharff, P. Study of C60 fullerenes and C60-containing composites cytotoxicity in vitro. Mater. Sci. Eng. 2007, 27, 1121–1124. [Google Scholar] [CrossRef]
- Sayes, C.M.; Fortner, J.D.; Guo, W.; Lyon, D.; Boyd, A.M.; Ausman, K.D.; Tao, Y.J.; Sitharaman, B.; Wilson, L.J.; Hughes, J.B.; et al. The differential cytotoxicity of water-soluble fullerenes. Nano Lett. 2004, 4, 1881–1887. [Google Scholar] [CrossRef]

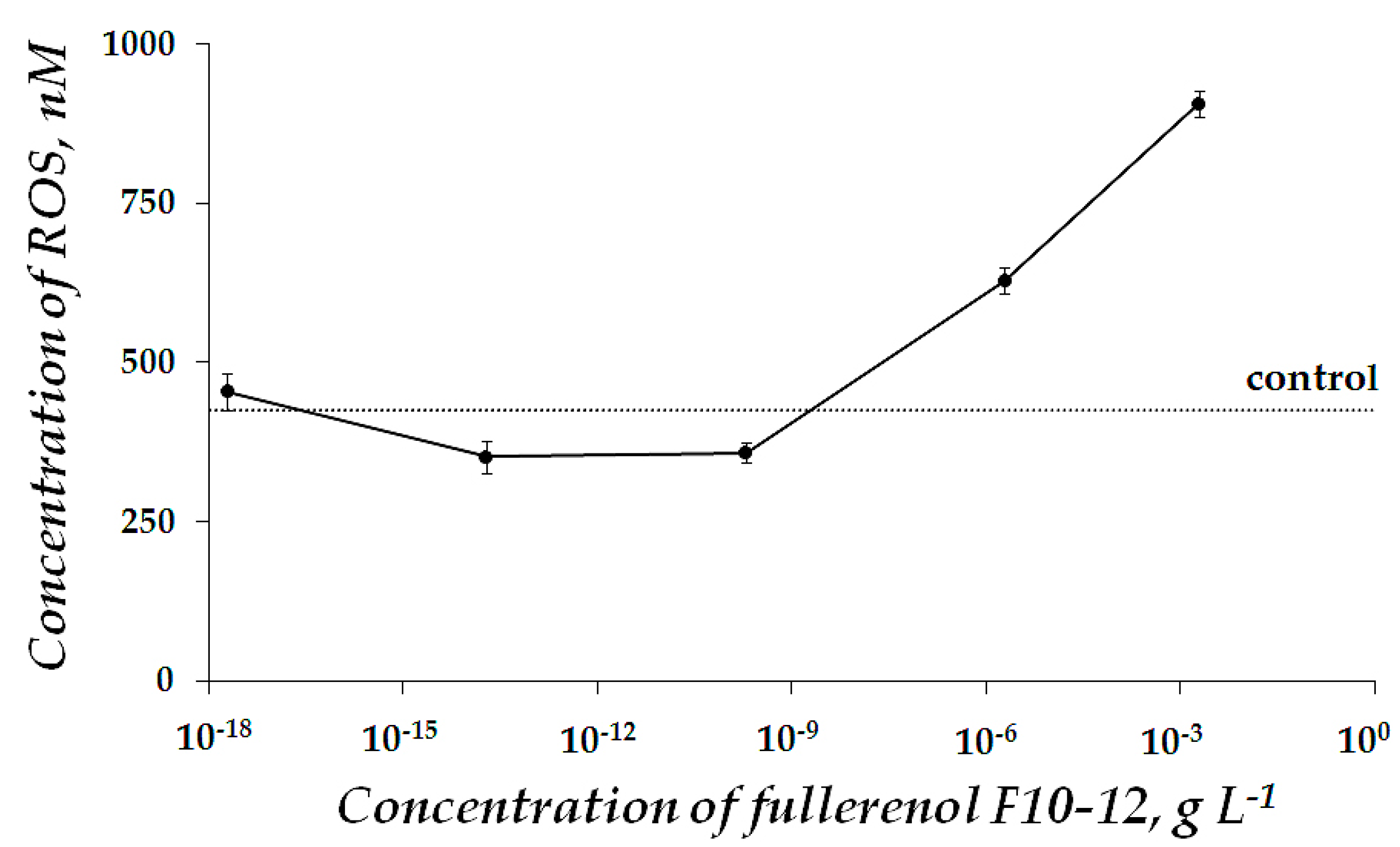
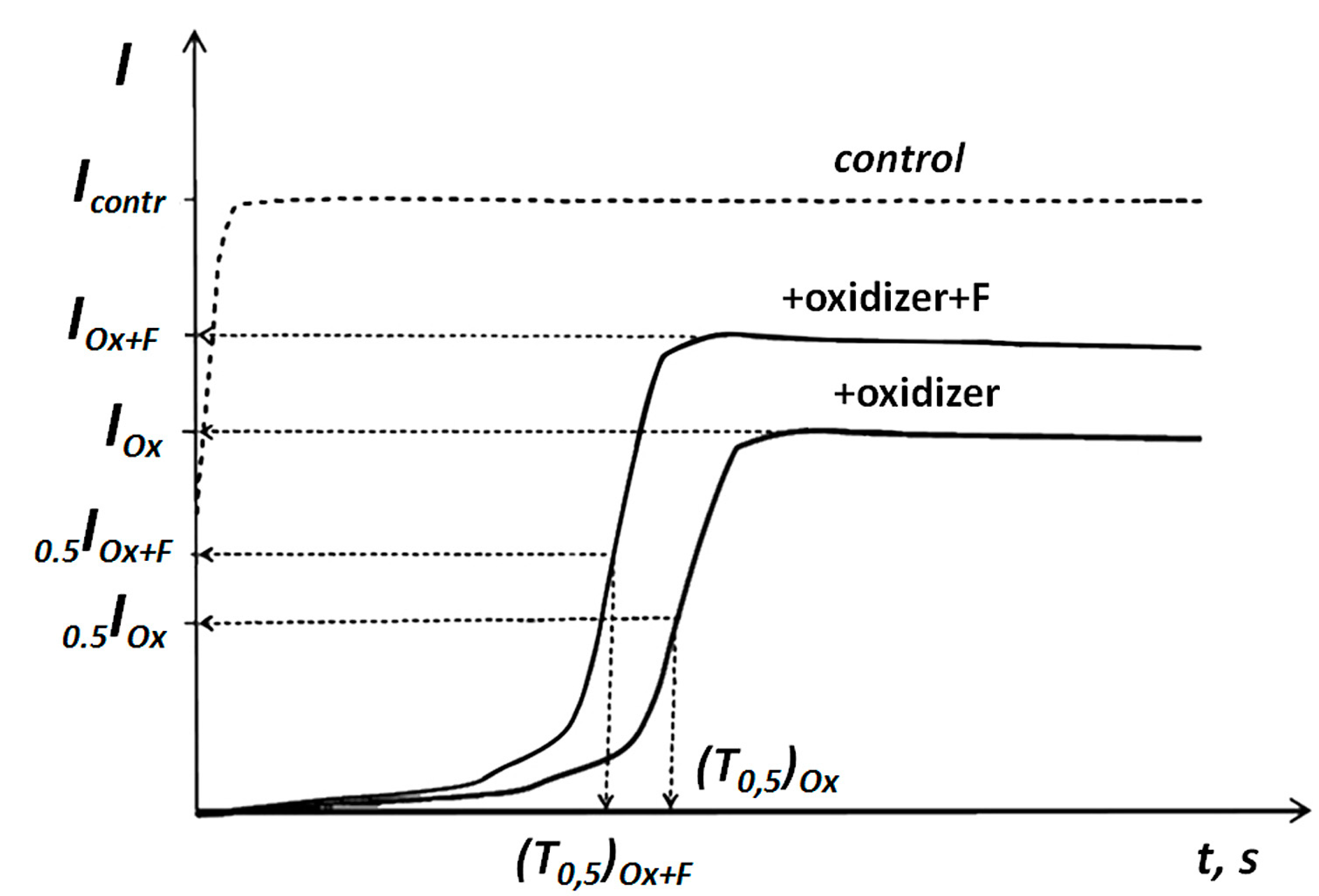
| x + y | Abbreviation |
|---|---|
| 10–12 | F10-12 |
| 24–28 | F24-28 |
| 40–42 | F40-42 |
| Fullerenols | 1,4-benzoquinone | K3[Fe(CN)6] | ||
|---|---|---|---|---|
| Active Concentrations, g L−1 | Maximal Value of IrelOx | Active Concentrations, g L−1 | Maximal Value of IrelOx | |
| F10-12 | 10–5–4·10−4 | 1.38 | - | 1.0 |
| F24-28 [33] | 10–18–10−10 | 1.44 | 10−18–10−4 | 1.2 |
| F40-42 [33] | 10−20–10−3 | 1.44 | - | 1.0 |
| Fullerenols | 1,4-benzoquinone | K3[Fe(CN)6] | ||
|---|---|---|---|---|
| Active Concentrations, g L−1 | Maximal Value of TrelOx | Active Concentrations, g L−1 | Maximal Value of TrelOx | |
| F10-12 | 10−13–5·10−5 | 1.2 | - | 1.0 |
| F24-28 [33] | 10−18–10−4 | 1.9 | 10−18–10−6 | 1.3 |
| F40-42 [33] | - | 1.0 | - | 1.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovel, E.S.; Kicheeva, A.G.; Vnukova, N.G.; Churilov, G.N.; Stepin, E.A.; Kudryasheva, N.S. Toxicity and Antioxidant Activity of Fullerenol C60,70 with Low Number of Oxygen Substituents. Int. J. Mol. Sci. 2021, 22, 6382. https://doi.org/10.3390/ijms22126382
Kovel ES, Kicheeva AG, Vnukova NG, Churilov GN, Stepin EA, Kudryasheva NS. Toxicity and Antioxidant Activity of Fullerenol C60,70 with Low Number of Oxygen Substituents. International Journal of Molecular Sciences. 2021; 22(12):6382. https://doi.org/10.3390/ijms22126382
Chicago/Turabian StyleKovel, Ekaterina S., Arina G. Kicheeva, Natalia G. Vnukova, Grigory N. Churilov, Evsei A. Stepin, and Nadezhda S. Kudryasheva. 2021. "Toxicity and Antioxidant Activity of Fullerenol C60,70 with Low Number of Oxygen Substituents" International Journal of Molecular Sciences 22, no. 12: 6382. https://doi.org/10.3390/ijms22126382








