Pathogenesis-Related Genes of PR1, PR2, PR4, and PR5 Families Are Involved in the Response to Fusarium Infection in Garlic (Allium sativum L.)
Abstract
:1. Introduction
2. Results
2.1. Identification of PR1, PR2, PR4, and PR5 Genes in the A. sativum Genome
2.2. Characterization of Putative PR1, PR2, PR4, and PR5 Proteins in Garlic
2.3. Conserved Domains in AsPR Proteins
2.4. Cis-Acting Elements in the Promoters of the AsPR1, AsPR2, AsPR4, and AsPR5 Genes
2.5. Expression Patterns of AsPR Genes
2.6. AsPR Gene Expression in Response to F. proliferatum Infection
2.6.1. AsPR1 Genes
2.6.2. AsPR2 Genes
2.6.3. AsPR4 Genes
2.6.4. AsPR5 Genes
2.7. Cloning and Characterization of CDSs of AsPR Genes Differentially Expressed in FBR-Sensitive and Resistant Cultivars
3. Discussion
4. Materials and Methods
4.1. In Silico Identification and Structural Characterization of PR1, PR2, PR4, and PR5 Genes in the Allium Sativum Genome
4.2. In Silico mRNA Expression Analysis
4.3. Fungi, Plant Material, and F. proliferatum Infection Assay
4.4. RNA Extraction and Quantitative Real-Time PCR (qRT-PCR) Analysis
4.5. Gene Identification
4.6. Promoter and 5′-UTR Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roux, F.; Voisin, D.; Badet, T.; Balagué, C.; Barlet, X.; Huard-Chauveau, C.; Roby, D.; Raffaele, S. Resistance to phytopathogens e tutti quanti: Placing plant quantitative disease resistance on the map. Mol. Plant Pathol. 2014, 15, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Ganai, B.A.; Kamili, A.N.; Bhat, A.A.; Mir, Z.A.; Bhat, J.A.; Tyagi, A.; Islam, S.T.; Mushtaq, M.; Yadav, P.; et al. Pathogenesis-related proteins and peptides as promising tools for engineering plants with multiple stress tolerance. Microbiol. Res. 2018, 212–213, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Akbudak, M.A.; Yildiz, S.; Filiz, E. Pathogenesis related protein-1 (PR-1) genes in tomato (Solanum lycopersicum L.): Bioinformatics analyses and expression profiles in response to drought stress. Genomics 2020, 112, 4089–4099. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.; Van Kan, J.A.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [Green Version]
- Zipfel, C.; Felix, G. Plants and animals: A different taste for microbes? Curr. Opin. Plant Biol. 2005, 8, 353–360. [Google Scholar] [CrossRef]
- Dangl, J.L.; Jones, J.D.G. Plant pathogens and integrated defence responses to infection. Nature 2001, 411, 826–833. [Google Scholar] [CrossRef]
- Okushima, Y.; Koizumi, N.; Kusano, T.; Sano, H. Secreted proteins of tobacco cultured BY2 cells: Identification of a new member of pathogenesis-related proteins. Plant Mol. Biol. 2000, 42, 479–488. [Google Scholar] [CrossRef]
- Van Loon, L.C.; Pierpont, W.S.; Boller, T.; Conejero, V. Recommendations for naming plant pathogenesis-related proteins. Plant Mol. Biol. Rep. 1994, 12, 245–264. [Google Scholar] [CrossRef]
- Edreva, A. Pathogenesis-related proteins: Research progress in the last 15 years. Gen. Appl. Plant Physiol. 2005, 31, 105–124. [Google Scholar]
- Sinha, M.; Singh, R.P.; Kushwaha, G.S.; Iqbal, N.; Singh, A.; Kaushik, S.; Kaur, P.; Sharma, S.; Singh, T.P. Current overview of allergens of plant pathogenesis related protein families. Sci. World J. 2014, 2014, 543195. [Google Scholar] [CrossRef] [Green Version]
- Guevara-Morato, M.A.; de Lacoba, M.G.; García-Luque, I.; Serra, M.T. Characterization of a pathogenesis-related protein 4 (PR-4) induced in Capsicum chinense L3 plants with dual RNase and DNase activities. J. Exp. Bot. 2010, 61, 3259–3271. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.J.; Geiser, D.M.; Proctor, R.H.; Rooney, A.P.; O’Donnell, K.; Trail, F.; Gardiner, D.M.; Manners, J.M.; Kazan, K. Fusarium pathogenomics. Annu. Rev. Microbiol. 2013, 67, 399–416. [Google Scholar] [CrossRef] [Green Version]
- Summerell, B.A. Resolving Fusarium: Current Status of the Genus. Annu. Rev. Phytopathol. 2019, 57, 323–339. [Google Scholar] [CrossRef]
- Kalman, B.; Abraham, D.; Graph, S.; Perl-Treves, R.; Meller Harel, Y.; Degani, O. Isolation and Identification of Fusarium spp., the Causal Agents of Onion (Allium cepa) Basal Rot in Northeastern Israel. Biology 2020, 9, 69. [Google Scholar] [CrossRef] [Green Version]
- Gagkaeva, T.; Gavrilova, O.; Orina, A.; Lebedin, Y.; Shanin, I.; Petukhov, P.; Eremin, S. Analysis of Toxigenic Fusarium Species Associated with Wheat Grain from Three Regions of Russia: Volga, Ural, and West Siberia. Toxins 2019, 11, 252. [Google Scholar] [CrossRef] [Green Version]
- Masachis, S.; Segorbe, D.; Turrà, D.; Leon-Ruiz, M.; Fürst, U.; El Ghalid, M.; Leonard, G.; López-Berges, M.S.; Richards, T.A.; Felix, G.; et al. A fungal pathogen secretes plant alkalinizing peptides to increase infection. Nat. Microbiol. 2016, 1, 16043. [Google Scholar] [CrossRef]
- Wrobel-Kwiatkowska, M.; Lorenc-Kukula, K.; Starzycki, M.; Oszmianski, J.; Kepczynska, E.; Szopa, J. Expression of b-1,3-glucanase in flax causes increased resistance to fungi. Physiol. Mol. Plant Pathol. 2004, 65, 245–256. [Google Scholar] [CrossRef]
- Samet, M.; Charfeddine, M.; Kamoun, L.; Nouri-Ellouze, O.; Gargouri-Bouzid, R. Effect of compost tea containing phosphogypsum on potato plant growth and protection against Fusarium solani infection. Environ. Sci. Pollut. Res. Int. 2018, 25, 18921–18937. [Google Scholar] [CrossRef]
- Bharti, P.; Jyoti, P.; Kapoor, P.; Sharma, V.; Shanmugam, V.; Yadav, S.K. Host-Induced Silencing of Pathogenicity Genes Enhances Resistance to Fusarium oxysporum Wilt in Tomato. Mol. Biotechnol. 2017, 59, 343–352. [Google Scholar] [CrossRef]
- Li, G.; Zhou, J.; Jia, H.; Gao, Z.; Fan, M.; Luo, Y.; Zhao, P.; Xue, S.; Li, N.; Yuan, Y.; et al. Mutation of a histidine-rich calcium-binding-protein gene in wheat confers resistance to Fusarium head blight. Nat. Genet. 2019, 51, 1106–1112. [Google Scholar] [CrossRef]
- Tan, R.; Collins, P.J.; Wang, J.; Wen, Z.; Boyse, J.F.; Laurenz, R.G.; Gu, C.; Jacobs, J.L.; Song, Q.; Chilvers, M.I.; et al. Different loci associated with root and foliar resistance to sudden death syndrome (Fusarium virguliforme) in soybean. Theor. Appl. Genet. 2019, 132, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.; Zhou, T.; Trick, H.N.; Gill, B.S.; Bockus, W.W.; Muthukrishnan, S. Greenhouse and field testing of transgenic wheat plants stably expressing genes for thaumatin-likeprotein, chitinase and glucanase against Fusarium graminearum. J. Exp. Bot. 2003, 54, 1101–1111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mackintosh, C.A.; Lewis, J.; Radmer, L.E.; Shin, S.; Heinen, S.J.; Smith, L.A.; Wyckoff, M.N.; Dill-Macky, R.; Evans, C.K.; Kravchenko, S.; et al. Overexpression of defense response genes in transgenic wheat enhances resistance to Fusarium head blight. Plant Cell Rep. 2007, 26, 479–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammad, I.A.; Abdel-Razik, A.B.; Soliman, E.R.; Tawfik, E. Transgenic potato (Solanum tuberosum) expressing two antifungal thionin genes confer resistance to Fusarium spp. J. Pharm. Biol. Sci. 2017, 12, 69–79. [Google Scholar] [CrossRef]
- Cramer, C.S. Breeding and genetics of Fusarium basal rot resistance in onion. Euphytica 2000, 115, 159–166. [Google Scholar] [CrossRef]
- Gálvez, L.; Urbaniak, M.; Waśkiewicz, A.; Stępień, Ł.; Palmero, D. Fusarium proliferatum—Causal agent of garlic bulb rot in Spain: Genetic variability and mycotoxin production. Food Microbiol. 2017, 67, 41–48. [Google Scholar] [CrossRef]
- Galván, G.A.; Koning-Boucoiran, C.F.S.; Koopman, W.J.M.; Burger-Meijer, K.; González, P.H.; Waalwijk, C.; Kik, C.; Scholten, O.E. Genetic variation among Fusarium isolates from onion, and resistance to Fusarium basal rot in related Allium species. Eur. J. Plant Pathol. 2008, 121, 499–512. [Google Scholar] [CrossRef]
- Filyushin, M.A.; Anisimova, O.K.; Kochieva, E.Z.; Shchennikova, A.V. Genome-Wide Identification and Expression of Chitinase Class I Genes in Garlic (Allium sativum L.) Cultivars Resistant and Susceptible to Fusarium proliferatum. Plants 2021, 10, 720. [Google Scholar] [CrossRef]
- Chand, S.K.; Nanda, S.; Mishra, R.; Joshi, R.K. Multiple garlic (Allium sativum L.) microRNAs regulate the immunity against the basal rot fungus Fusarium oxysporum f. sp. cepae. Plant Sci. 2017, 257, 9–21. [Google Scholar] [CrossRef]
- Chand, S.K.; Nanda, S.; Joshi, R.K. Regulation of miR394 in Response to Fusarium oxysporum f. sp. cepae (FOC) Infection in Garlic (Allium sativum L.). Front. Plant Sci. 2016, 7, 258. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Zhu, S.; Li, N.; Cheng, Y.; Zhao, J.; Qiao, X.; Lu, L.; Liu, S.; Wang, Y.; Liu, C.; et al. A Chromosome-Level Genome Assembly of Garlic (Allium sativum) Provides Insights into Genome Evolution and Allicin Biosynthesis. Mol. Plant 2020, 13, 1328–1339. [Google Scholar] [CrossRef]
- Van Loon, L.C.; Van Strien, E.A. The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol. Mol. Plant Pathol. 1999, 55, 85–97. [Google Scholar] [CrossRef]
- Bartholomew, E.S.; Black, K.; Feng, Z.; Liu, W.; Shan, N.; Zhang, X.; Wu, L.; Bailey, L.; Zhu, N.; Qi, C.; et al. Comprehensive Analysis of the Chitinase Gene Family in Cucumber (Cucumis sativus L.): From Gene Identification and Evolution to Expression in Response to Fusarium oxysporum. Int. J. Mol. Sci. 2019, 20, 5309. [Google Scholar] [CrossRef] [Green Version]
- Doehlemann, G.; Ökmen, B.; Zhu, W.; Sharon, A. Plant Pathogenic Fungi. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Leyronas, C.; Chrétien, P.L.; Troulet, C.; Duffaud, M.; Villeneuve, F.; Morris, C.E.; Hunyadi, H. First report of Fusarium proliferatum causing garlic clove rot in France. Plant Dis. 2018, 102, 2658. [Google Scholar] [CrossRef]
- Tonti, S.; Prà, M.D.; Nipoti, P.; Prodi, A.; Alberti, I. First Report of Fusarium proliferatum Causing Rot of Stored Garlic Bulbs (Allium sativum L.) in Italy. J. Phytopathol. 2012, 160, 761–763. [Google Scholar] [CrossRef]
- Chen, J.; Piao, Y.; Liu, Y.; Li, X.; Piao, Z. Genome-wide identification and expression analysis of chitinase gene family in Brassica rapa reveals its role in clubroot resistance. Plant Sci. 2018, 270, 257–267. [Google Scholar] [CrossRef]
- Gao, Y.; Jia, S.; Wang, C.; Wang, F.; Wang, F.; Zhao, K. BjMYB1, a transcription factor implicated in plant defence through activating BjCHI1 chitinase expression by binding to a W-box-like element. J. Exp. Bot. 2016, 67, 4647–4658. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Zan, X.; Wu, X.; Yao, L.; Chen, Y.; Jia, S.; Zhao, K. Identification of Fungus-Responsive Cis-Acting Element in the Promoter of Brassica Juncea Chitinase Gene, BjCHI1. Plant Sci. 2014, 215–216, 190–198. [Google Scholar] [CrossRef]
- Verma, V.; Ravindran, P.; Kumar, P.P. Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 2016, 16, 86. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Kracher, B.; Ziegler, J.; Birkenbihl, R.P.; Somssich, I.E. Negative regulation of ABA signaling by WRKY33 is critical for Arabidopsis immunity towards Botrytis cinerea 2100. eLife 2015, 4, e07295. [Google Scholar] [CrossRef]
- Yang, J.; Duan, G.; Li, C.; Liu, L.; Han, G.; Zhang, Y.; Wang, C. The Crosstalks Between Jasmonic Acid and Other Plant Hormone Signaling Highlight the Involvement of Jasmonic Acid as a Core Component in Plant Response to Biotic and Abiotic Stresses. Front. Plant Sci. 2019, 10, 1349. [Google Scholar] [CrossRef] [Green Version]
- Warman, N.M.; Aitken, E.A.B. The Movement of Fusarium oxysporum f. sp. cubense (Sub-Tropical Race 4) in Susceptible Cultivars of Banana. Front. Plant Sci. 2018, 9, 1748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schneiter, R.; Di Pietro, A. The CAP protein superfamily: Function in sterol export and fungal virulence. Biomol. Concepts 2013, 4, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, V.; Vashisht, D.; Cletus, J.; Sakthivel, N. Plant β-1,3-glucanases: Their biological functions and transgenic expression against phytopathogenic fungi. Biotechnol. Lett. 2012, 34, 1983–1990. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Dong, C.; Li, B.; Dai, H. A PR-4 gene identified from Malus domestica is involved in the defense responses against Botryosphaeria dothidea. Plant Physiol. Biochem. 2013, 62, 23–32. [Google Scholar] [CrossRef]
- De Jesús-Pires, C.; Ferreira-Neto, J.R.C.; Pacifico Bezerra-Neto, J.; Kido, E.A.; de Oliveira Silva, R.L.; Pandolfi, V.; Wanderley-Nogueira, A.C.; Binneck, E.; da Costa, A.F.; Pio-Ribeiro, G.; et al. Plant Thaumatin-like Proteins: Function, Evolution and Biotechnological Applications. Curr. Protein Pept. Sci. 2020, 21, 36–51. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0. Molecular biology and evolution. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- Hu, B.; Jin, J.; Guo, A.Y.; Zhang, H.; Luo, J.; Gao, G. GSDS 2.0: An Upgraded Gene Feature Visualization Server. Bioinformatics 2015, 31, 1296–1297. [Google Scholar] [CrossRef] [Green Version]
- Choi, Y.; Chan, A.P. PROVEAN web server: A tool to predict the functional effect of amino acid substitutions and indels. Bioinformatics 2015, 31, 2745–2747. [Google Scholar] [CrossRef] [Green Version]
- Babicki, S.; Arndt, D.; Marcu, A.; Liang, Y.; Grant, J.R.; Maciejewski, A.; Wishart, D.S. Heatmapper: Web-enabled heat mapping for all. Nucl. Acids Res. 2016, 44, W147–W153. [Google Scholar] [CrossRef]
- Anisimova, O.K.; Seredin, T.M.; Danilova, O.A.; Filyushin, M. First Report of Fusarium proliferatum Causing Garlic clove Rot in Russian Federation. Plant Dis. 2021. [Google Scholar] [CrossRef]
- Sugui, J.A.; Deising, H.B. Isolation of infection-specific sequence tags expressed during early stages of maize anthracnose disease development. Mol. Plant Pathol. 2002, 3, 197–203. [Google Scholar] [CrossRef]
- Liu, M.; Wu, Z.; Jiang, F. Selection and validation of garlic reference genes for quantitative real-time PCR normalization. Plant Cell Tissue Organ Cult. 2015, 122, 435–444. [Google Scholar] [CrossRef]
- Schwinn, K.E.; Ngo, H.; Kenel, F.; Brummell, D.A.; Albert, N.W.; McCallum, J.A.; Pither-Joyce, M.; Crowhurst, R.N.; Eady, C.; Davies, K.M. The onion (Allium cepa L.) R2R3-MYB gene MYB1 regulates anthocyanin biosynthesis. Front. Plant Sci. 2016, 7, 1865. [Google Scholar] [CrossRef] [Green Version]
- Lescot, M. PlantCARE, a Database of Plant Cis-Acting Regulatory Elements and a Portal to Tools for in Silico Analysis of Promoter Sequences. Nucl. Acids Res. 2002, 30, 325–327. [Google Scholar] [CrossRef]
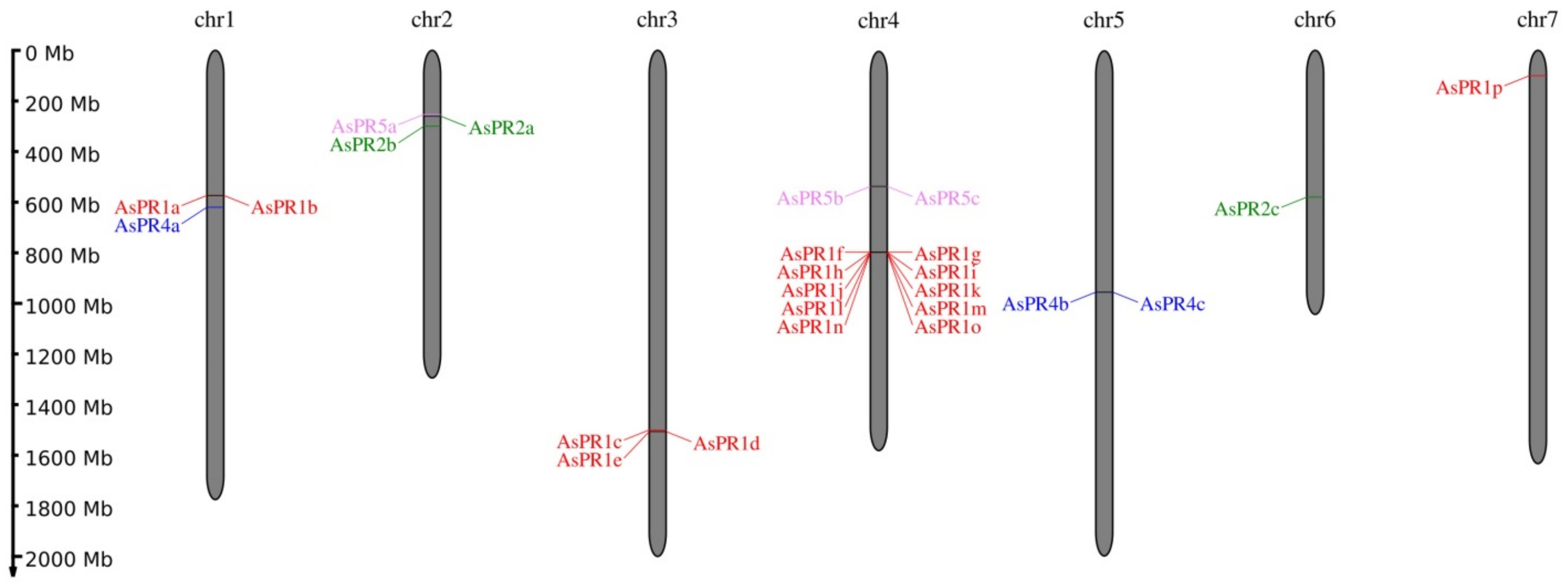





| Gene | Localization | Length (bp) | Number of Exons | CDS (bp) | Protein (aa) | Transcript ID [31] |
|---|---|---|---|---|---|---|
| PR1 family | ||||||
| AsPR1a | ch1:597808382-597808867 (+) | 486 | 1 | 486 | 161 | Asa1G02133.1 |
| AsPR1b | ch1:597852310-597852792 (+) | 483 | 1 | 483 | 160 | Asa1G02134.1 |
| AsPR1c | ch3:1569741035-1569741535(−) | 501 | 1 | 501 | 166 | Asa3G05742.1 |
| AsPR1d | ch3:1574050049-1574050546 (+) | 498 | 1 | 498 | 165 | Asa3G05767.1 |
| AsPR1e | ch3:1575214795-1575215280 (−) | 486 | 1 | 486 | 161 | Asa3G05770.1 |
| AsPR1f | ch4:828639577-828640059 (−) | 483 | 1 | 483 | 160 | Not detected |
| AsPR1g | ch4:828659513-828659995 (−) | 483 | 1 | 483 | 160 | Not detected |
| AsPR1h | ch4:829079571-829080095 (−) | 525 | 1 | 525 | 174 | Asa4G03112.1 |
| AsPR1i | ch4:829255226-829255750 (−) | 525 | 1 | 525 | 174 | Asa4G03113.1 |
| AsPR1j | ch4:829291631-829292125 (−) | 495 | 1 | 495 | 164 | not detected |
| AsPR1k | ch4:831436248-831436742 (−) | 495 | 1 | 495 | 164 | Asa4G03125.1 |
| AsPR1l | ch4:831555457-831555981 (+) | 525 | 1 | 525 | 174 | Asa4G03126.1 |
| AsPR1m | ch4:831614218-831614742 (+) | 525 | 1 | 525 | 174 | Asa4G03127.1 |
| AsPR1n | ch4:831977068-831977562 (+) | 495 | 1 | 495 | 164 | Asa4G03130.1 |
| AsPR1o | ch4:831982650-831983087 (+) | 438 | 1 | 438 | 145 | Asa4G03131.1 |
| AsPR1p | ch7:103854549-103855043(−) | 495 | 1 | 495 | 164 | Asa7G00352.1 |
| PR2 family | ||||||
| AsPR2a | ch2:272017011-272018179 (−) | 1169 | 2 | 1035 | 344 | Asa2G01057.1 |
| AsPR2b | ch2:311576151-311577239 (−) | 1089 | 2 | 990 | 329 | Asa2G01195.1 |
| AsPR2c | ch6:605851704-605853559 (−) | 1856 | 3 | 945 | 314 | Asa6G06180.1 |
| PR4 family | ||||||
| AsPR4a1 | ch1:652401564-652402126 (+) | 563 | 2 | 444 | 147 | Asa1G02345.1 |
| AsPR4b | ch5:998122740-998123259 (+) | 520 | 2 | 444 | 147 | Asa5G03281.1 |
| AsPR4c1 | ch5:999259985-999260547 (+) | 563 | 2 | 444 | 147 | Asa1G02345.1 |
| PR5 family | ||||||
| AsPR5a | ch2:266379091-266379753 (+) | 663 | 1 | 663 | 220 | Asa2G01043.1 |
| AsPR5b | ch4:561214533-561215195 (+) | 663 | 1 | 663 | 220 | Asa4G02099.1 |
| AsPR5c | ch4:561229259-561229921 (+) | 663 | 1 | 663 | 220 | Asa4G02100.1 |
| Protein Symbol | MW (kDa) | pI | AI | GRAVY | Signal Peptide | Catalytic Domain | Subcellular Localization | Biological Process |
|---|---|---|---|---|---|---|---|---|
| PR1 family | ||||||||
| AsPR1a | 17.34 | 7.55 | 76.40 | 0.038 | 1–23 | CAP (31–149) | Secretory | Defense response (GO:0006952), response to biotic stimulus (GO:0009607) |
| AsPR1b | 17.15 | 6.78 | 77.50 | 0.065 | 1–22 | CAP (30–148) | ||
| AsPR1c | 17.84 | 5.99 | 72.77 | −0.135 | 1–27 | CAP (35–154) | ||
| AsPR1d | 17.60 | 6.14 | 75.64 | −0.155 | 1–26 | CAP (34–153) | ||
| AsPR1e | 17.14 | 6.48 | 68.94 | −0.329 | 1–26 | CAP (34–149) | ||
| AsPR1f | 17.51 | 4.50 | 63.44 | −0.319 | 1–19 | CAP (27–145) | ||
| AsPR1g | 17.45 | 4.63 | 63.44 | −0.300 | ||||
| AsPR1h | 19.38 | 5.33 | 61.15 | −0.533 | 1–23 | CAP (31–149) | ||
| AsPR1i | 19.62 | 9.25 | 71.72 | −0.487 | ||||
| AsPR1j | 18.10 | 7.58 | 61.34 | −0.412 | 1–19 | CAP (27–145) | ||
| AsPR1k | 18.04 | 8.20 | 61.34 | −0.393 | ||||
| AsPR1l | 19.58 | 9.21 | 71.72 | −0.485 | 1–23 | CAP (31–149) | ||
| AsPR1m | 19.35 | 6.41 | 64.54 | −0.501 | ||||
| AsPR1n | 18.07 | 8.20 | 59.57 | −0.407 | 1–19 | CAP (27–145) | ||
| AsPR1o | 16.05 | 7.59 | 68.69 | −0.246 | ||||
| AsPR1p | 18.24 | 6.27 | 85.00 | −0.271 | 1–25 | CAP (34–152) | ||
| PR2 family | ||||||||
| AsPR2a | 34.97 | 7.67 | 94.01 | 0.045 | 1–21 | GH17 (22–329) | Secretory | Carbohydrate metabolic process (GO:0005975) |
| AsPR2b | 37.33 | 5.14 | 92.44 | −0.021 | 1–29 | GH17 (30–343) | Secretory | |
| AsPR2c | 33.53 | 6.41 | 91.02 | −0.008 | n/d | GH17 (5–312) | Nucleus | |
| PR4 family | ||||||||
| AsPR4a | 15.55 | 5.54 | 73.74 | −0.078 | 1–25 | Barwin (31–145) | Secretory | Defense response to fungus (GO:0050832), defense response to bacterium (GO:0042742) |
| AsPR4b | 15.5 | 6.22 | 79.05 | −0.029 | ||||
| AsPR4c | 15.55 | 5.54 | 73.74 | −0.078 | ||||
| PR5 family | ||||||||
| AsPR5a | 23.46 | 4.71 | 58.09 | −0.085 | 1–21 | GH64-TLP-SF (28–220) | Secretory | Defense response (GO:0006952) |
| AsPR5b | 23.50 | 4.71 | 55.86 | −0.126 | ||||
| AsPR5c | 23.59 | 4.74 | 55.00 | −0.164 | ||||
| Motif | Response to 1 | Number of Elements in Promoters | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AsPR1a | AsPR1b | AsPR1c | AsPR1d | AsPR1e | AsPR1f | AsPR1g | AsPR1h | AsPR1i | AsPR1j | AsPR1k | AsPR1l | AsPR1m | AsPR1n | AsPR1o | AsPR1p | AsPR2a | AsPR2b | AsPR2c | AsPR4a | AsPR4b | AsPR4c | AsPR5a | AsPR5b | AsPR5c | ||
| Hormone Response | ||||||||||||||||||||||||||
| ABRE | ABA | 1 | 3 | 1 | 2 | 3 | 2 | 2 | 4 | 1 | 3 | 2 | 2 | 2 | ||||||||||||
| ABRE3a | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |||||||||||||||||
| ABRE4 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |||||||||||||||||
| CARE | 1 | |||||||||||||||||||||||||
| AUXRR-core | Auxin | 1 | 1 | |||||||||||||||||||||||
| AuxRE | 1 | 1 | ||||||||||||||||||||||||
| TGA-element | 1 | 1 | 1 | 1 | ||||||||||||||||||||||
| CGTCA-motif | MeJA | 1 | 1 | 1 | 1 | 1 | 2 | 2 | 2 | 1 | 1 | 1 | ||||||||||||||
| TGACG-motif | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 2 | 1 | 1 | 1 | |||||||||||||||
| AS-1 | SA | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 2 | 1 | 1 | 1 | ||||||||||||||
| TCA-element | 2 | 1 | 2 | |||||||||||||||||||||||
| P-box | 2 | 1 | 1 | 1 | ||||||||||||||||||||||
| TATC-box | GA | 1 | ||||||||||||||||||||||||
| GARE-motif | 1 | 1 | 1 | 1 | 1 | |||||||||||||||||||||
| ERE | ET | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | |||||||||||||||||
| Stress Response | ||||||||||||||||||||||||||
| ARE | Anaerobic conditions | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 2 | 1 | 2 | 2 | |||||||||||||
| DRE1/DRE core | Drought | 1 | ||||||||||||||||||||||||
| MBS | 1 | 1 | 1 | 2 | 2 | 3 | 2 | 2 | 1 | 2 | 3 | 3 | 1 | 1 | 1 | 1 | 1 | |||||||||
| LTR | Cold | 1 | 1 | 1 | 1 | |||||||||||||||||||||
| STRE | Heat, osmotic shock, low pH, starvation | 4 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | ||||||||||||
| F-box | Salt, heavy metals | 1 | ||||||||||||||||||||||||
| TC-rich repeats | Defense | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | 1 | ||||||||||||||||
| W-box | Wounding, pathogens | 1 | 2 | |||||||||||||||||||||||
| Wun-motif | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | 1 | ||||||||||||||||
| WRE3 | 1 | 1 | 1 | 1 | 1 | |||||||||||||||||||||
| box S | 1 | 1 | ||||||||||||||||||||||||
| Gene | cv. Sarmat | cv. Strelets |
|---|---|---|
| AsPR1c | c. 481G > A (p. V161I) | c. 379G > C (p. V127L), c. 417T > C, c. 481G > A (p. V161I) |
| AsPR1d | c. 117A > G, c. 200T > G (p. I67R) | |
| AsPR1k | c. 462A > G | |
| AsPR2a | c. 291T > C | c. 35T > C (p. L12S), c. 559A > C p. I187L) |
| AsPR2b | c. 57A > G | |
| AsPR2c | c. 219C > A; c. 855G > C (p. L285F) | |
| AsPR5a | c. 126T > C, c. 159G > C, c. 279A > G, c. 285C > T, c. 403G > A (p. G135S), c. 468G > C, c. 620T > C (p. I207T) | |
| AsPR5c | c. 516T > C, c. 647A > T (p. D216V) | |
| Gene | Primer Sequence (5′→3′) | Application |
|---|---|---|
| AsPR1a, b | ATGGAACACGACACTGGCAG GCATACTGACCAGAGTAACTGG | Gene expression analysis (qRT-PCR) |
| AsPR1c | GGCGGTCCTTATGGTGAAA GCCAGGGTCACATGTGTTA | |
| AsPR1d | GGCGGTCCTTATGGTGAAA CCAGGGTCACATGTGTTGCT | |
| AsPR1f, g | CGATCACCACCGCAGTTCA GCGTAGTTCTGTGCGTAATCAG | |
| AsPR1i | TATGGGGAGAACCTATTCGC AATCTTRACCGACTTAGCCCA | |
| AsPR1k | GTGTCCGAGAAGCGGTACTAT AGCCGCCAGTGTTGCACC | |
| AsPR1p | GTCGCAAAATACGCGCAAAGTT GTACTTCACGACATCGGCATC | |
| AsPR2a | GCTAGAAACCATATCGTTGCCT GCATACCGTAGCATACTCCGA | |
| AsPR2b | GGTCGCATTTCTCCTAGGCAT GCGTCGCCTGCTGATGGAA | |
| AsPR2c | GGCCCATTGTCCAGTTCTTG AGGCGCCGTGAATAATGCGTA | |
| AsPR4a | ATGCCGGCATGTCCCTCG GTCTATGATCCTCACCGTCGTT | |
| AsPR4b | ATGCCGGCATGTCCCTCG GGTCTATGATCCTCACCGTCAA | |
| AsPR5a | CATCCGGACACGGCAGCT TCCATGTACTGCTTCAGAGCG | |
| AsPR5c | GCAAGCAGCTCAACTCAGGA GCCGGTCTGACATCTTCCA | |
| AsPR1c | ATGGGATCAATCAGTAGTTATA AACGACTGAGTACTCTCAGT | Gene amplification |
| AsPR1d | ATGGGATCGACCAGTACTTG TAACGTCGTAGTTGTAACGAC | |
| AsPR1f, g | ATGAAAACGTCATTTCTCTTC ATAAATAGCAGTACACACATAA | |
| AsPR1k | GCTCAAATTACAATGAAAACGTT CTGTCTGTTTCAGCATGCA | |
| AsPR2a | TGTGCACCATCGAATTACCTTC CTCTGTCTCCCTTAATAGTAC | |
| AsPR2b | AAATGCAAGCAAGGAAGCTTG CCCTGGTACATTCATAGTTAAC | |
| AsPR2c | TTGAAATGGTCATGCATGCCT AAACAGGGCACACATGCAAG | |
| AsPR5a | ATGTCGACCCAAATTACAGTC ACTCAATCCAAGAAYACAGTTC | |
| AsPR5c | ATGTCGACCCAAATTACAGTC CTGCAAACTAATTATTCGGTGA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anisimova, O.K.; Shchennikova, A.V.; Kochieva, E.Z.; Filyushin, M.A. Pathogenesis-Related Genes of PR1, PR2, PR4, and PR5 Families Are Involved in the Response to Fusarium Infection in Garlic (Allium sativum L.). Int. J. Mol. Sci. 2021, 22, 6688. https://doi.org/10.3390/ijms22136688
Anisimova OK, Shchennikova AV, Kochieva EZ, Filyushin MA. Pathogenesis-Related Genes of PR1, PR2, PR4, and PR5 Families Are Involved in the Response to Fusarium Infection in Garlic (Allium sativum L.). International Journal of Molecular Sciences. 2021; 22(13):6688. https://doi.org/10.3390/ijms22136688
Chicago/Turabian StyleAnisimova, Olga K., Anna V. Shchennikova, Elena Z. Kochieva, and Mikhail A. Filyushin. 2021. "Pathogenesis-Related Genes of PR1, PR2, PR4, and PR5 Families Are Involved in the Response to Fusarium Infection in Garlic (Allium sativum L.)" International Journal of Molecular Sciences 22, no. 13: 6688. https://doi.org/10.3390/ijms22136688







