Possible Crosstalk of the Immune Cells within the Lung and Mediastinal Fat-Associated Lymphoid Clusters in the Acute Inflammatory Lung Asthma-Like Mouse Model
Abstract
:1. Introduction
2. Results
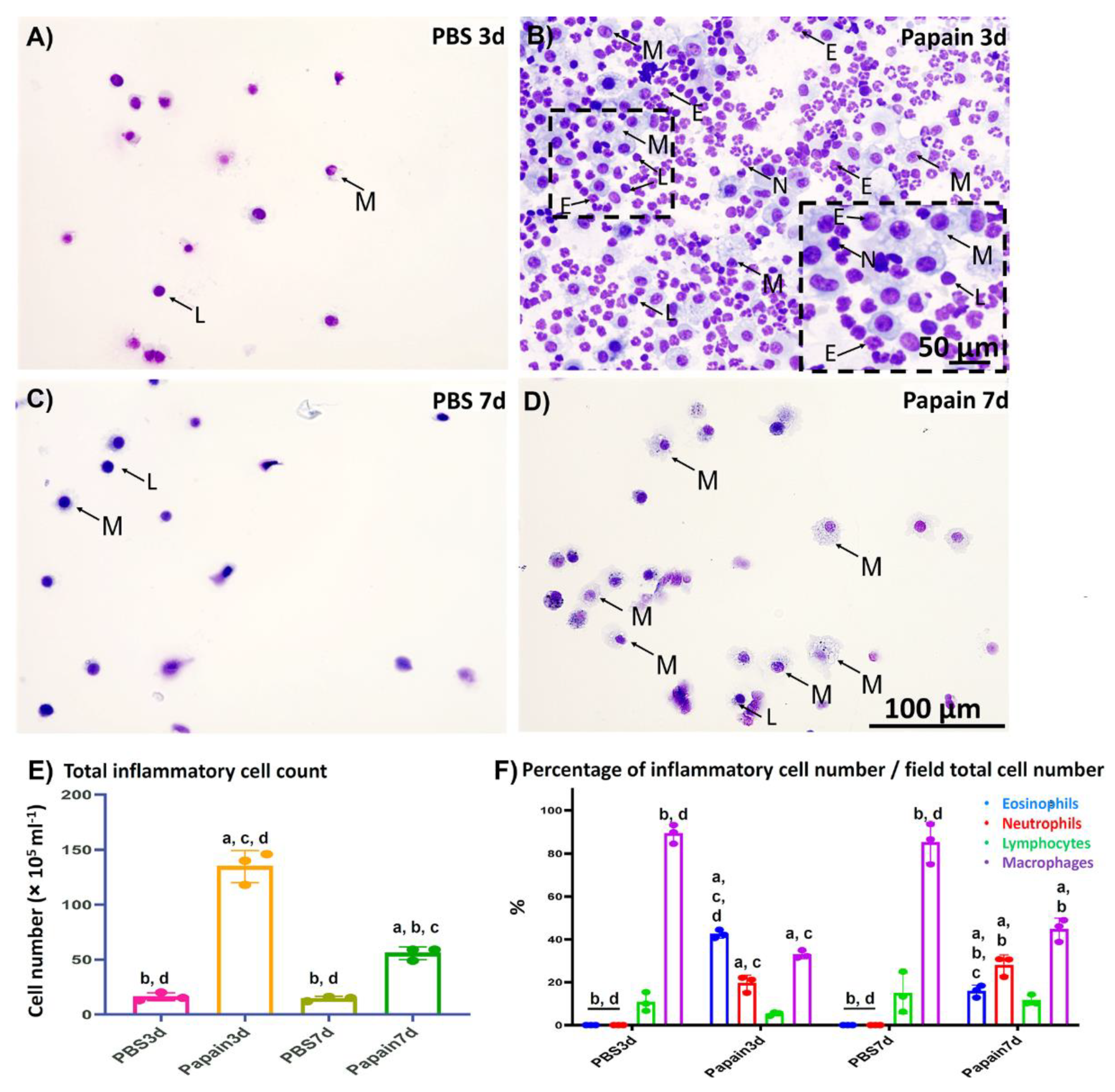
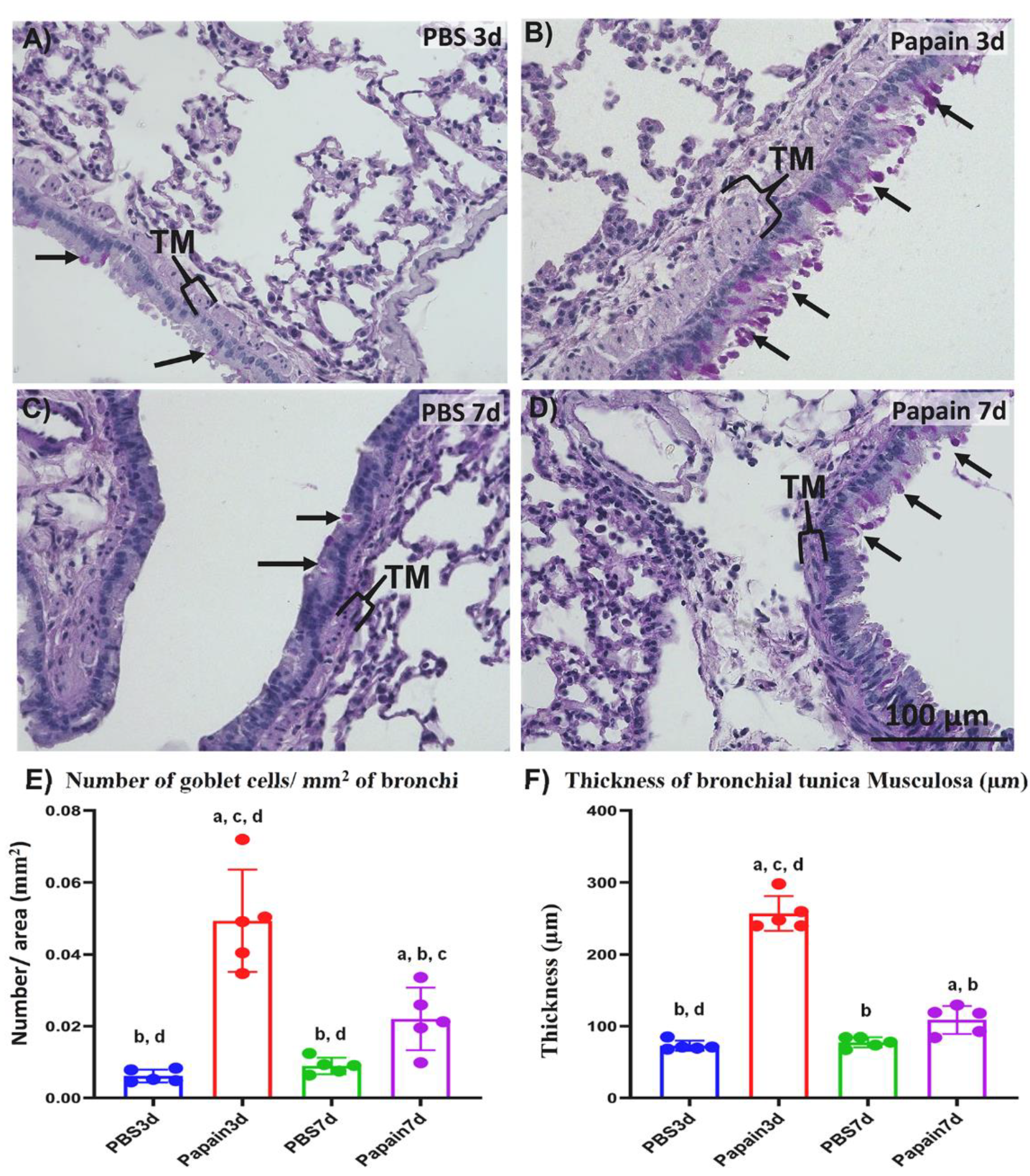
2.1. Indices of Asthma Induction in the Mice Model
2.1.1. Analysis of Inflammatory Cells in the BALF
2.1.2. Morphological and Morphometrical Analysis of Lung Tissue among the Studied Groups
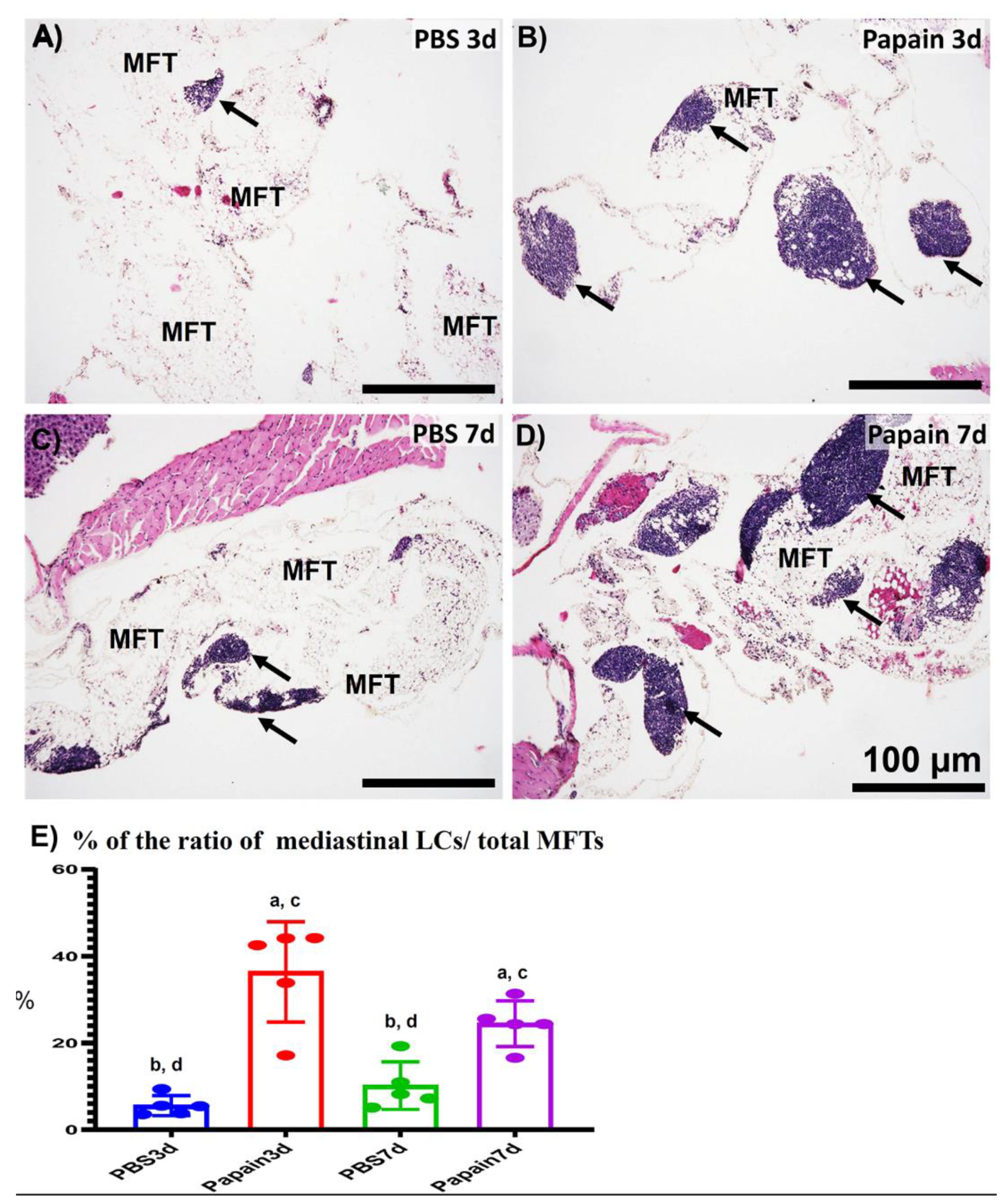
2.2. Morphological Features of MFALCs in the Papain Group vs. Vehicle Group
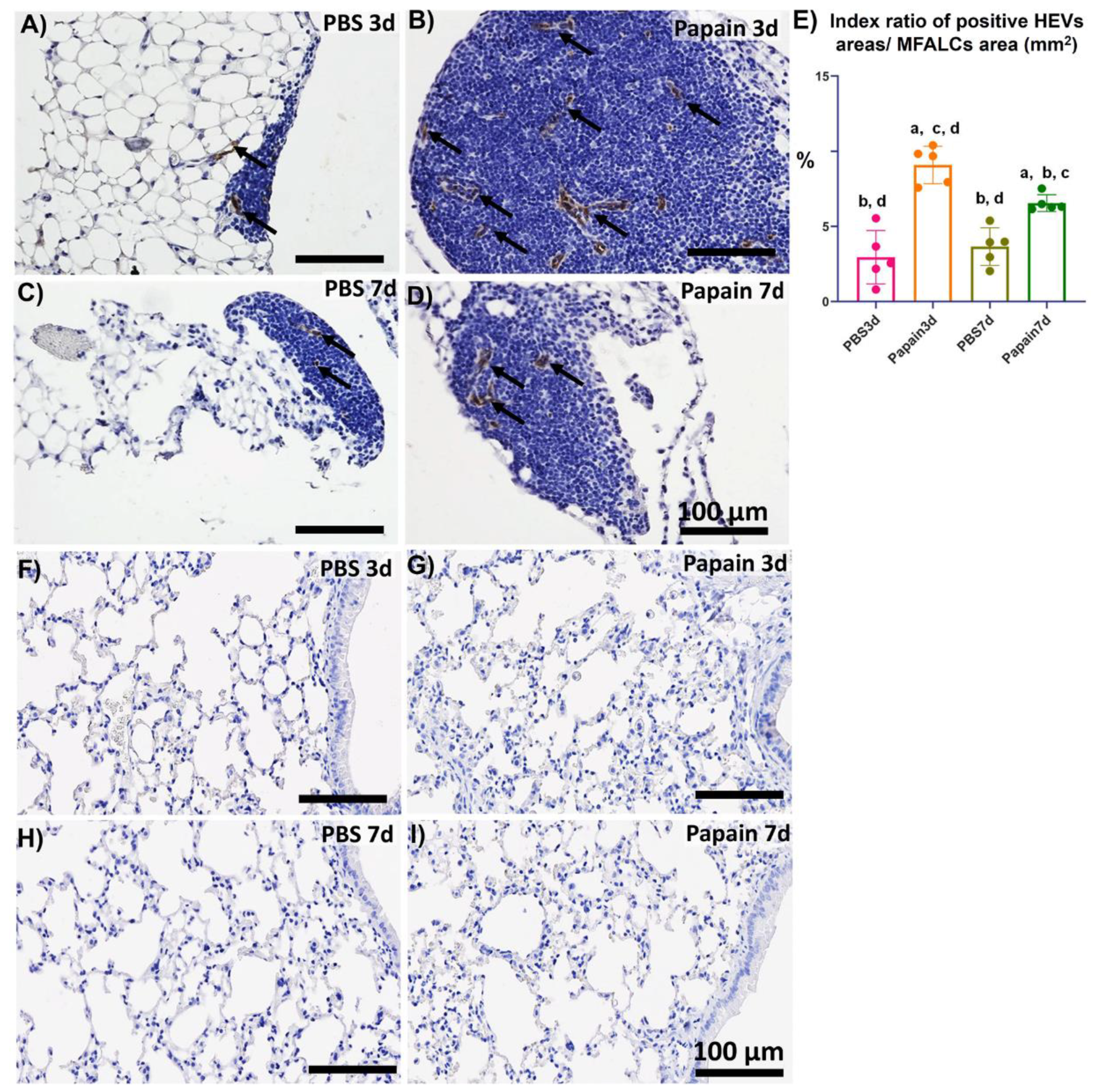
2.3. Analysis of the Role of Peripheral Node Address in (PNAd) Positive High Endothelial Venules in the Development of MFALCs and Lung Asthma
2.4. Analysis of Immune Cells in MFALCs and Lung Tissues of Papain Group and Vehicle Group
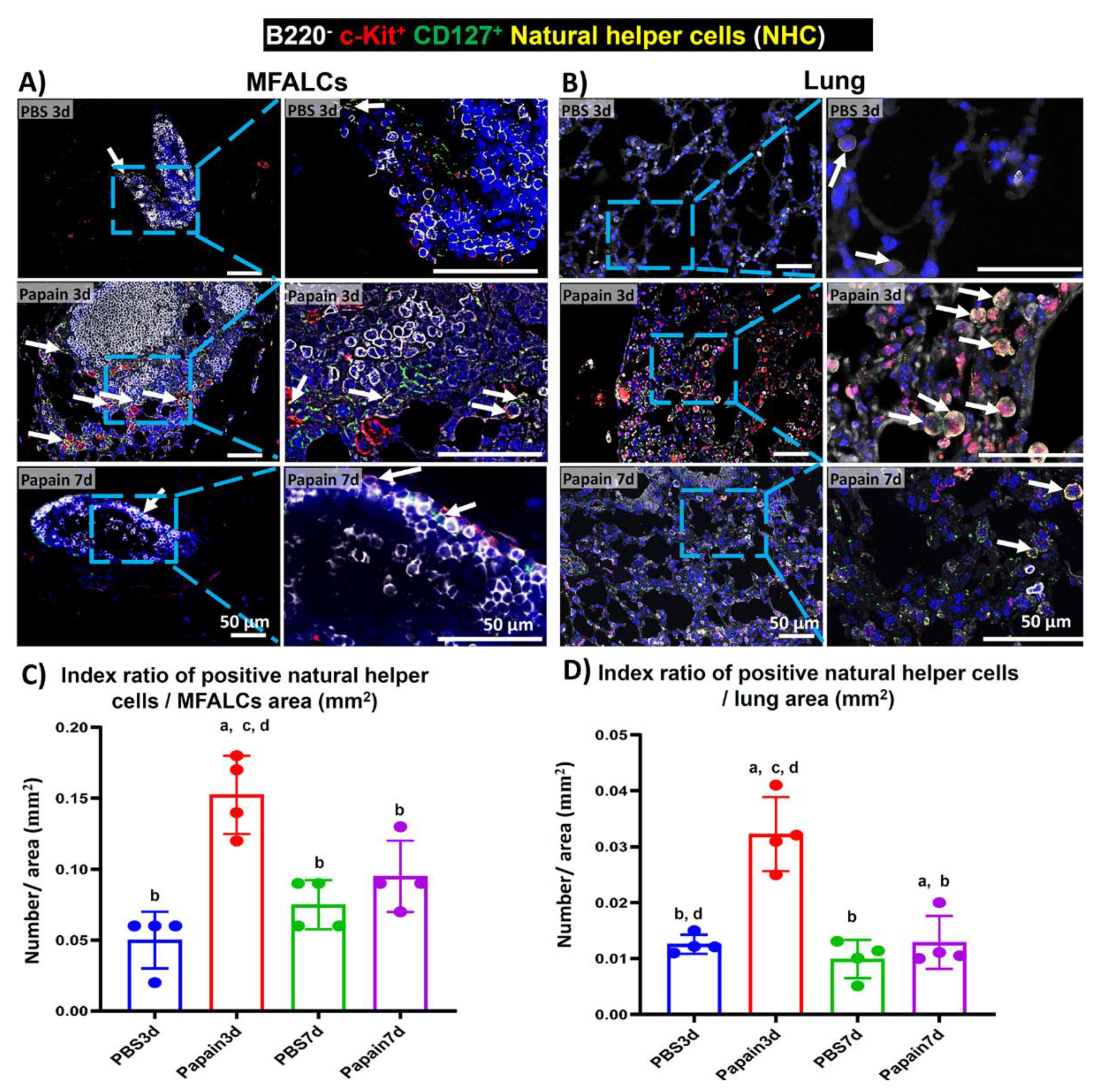
2.5. Analysis of the Occurrence of NHC in MFALCs and Lung Tissues of Papain Group and Vehicle Group
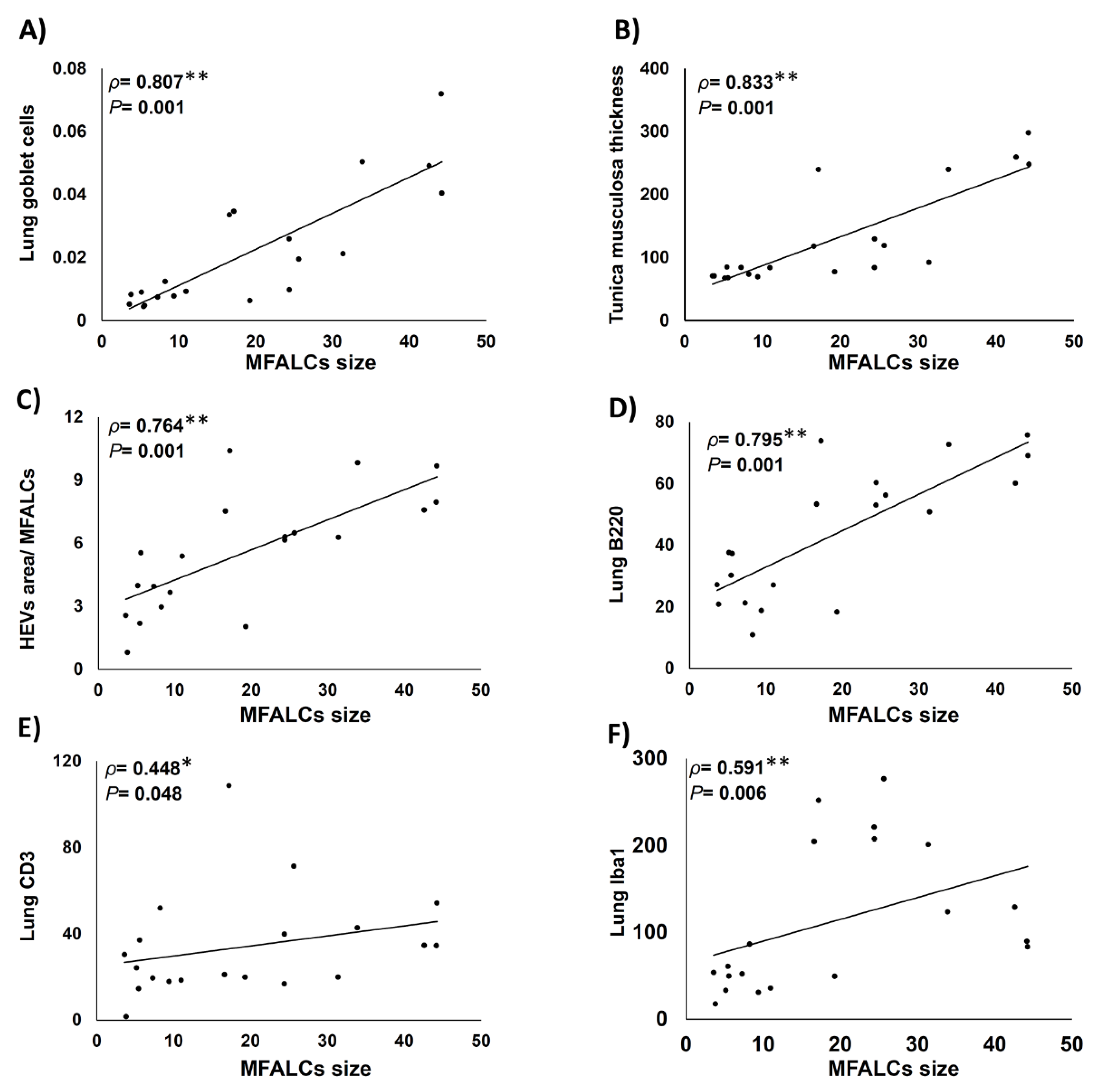
2.6. Histopathological Correlations between MFALCs’ Size, Indices of Lung Asthma Induction, High Endothelial Venules within MFALCs, and Immune Cells’ Infiltration in Lung
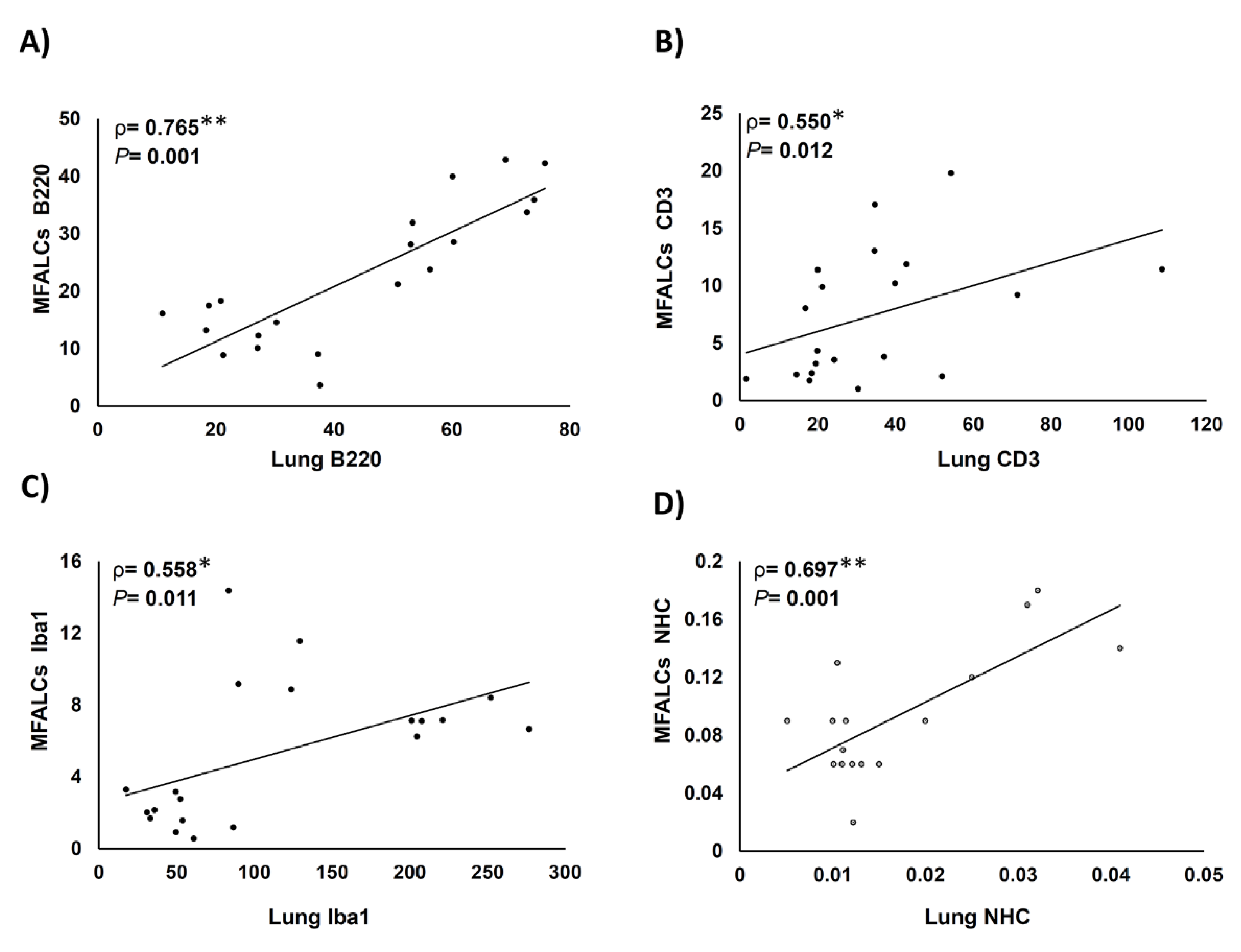
2.7. Histopathological Correlations between Immune Cell Populations in MFALCs and Infiltration in the Lung
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Experimental Design
4.3. BALF Collection and Preparation for Cytological Interpretation
4.4. Morphological and Histopathological Analysis
4.5. Immunohistochemical Staining of High Endothelial Venules and Immune Cells
4.6. Immunofluorescent Staining of NHC
4.7. Histomorphometric Measurements
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BALF | Bronchoalveolar lavage fluid |
| FALCs | Fat-associated lymphoid clusters |
| ILCs | Innate lymphoid cells |
| ILC2s | ILCs group2 |
| Lin | Lineage |
| MFT | Mediastinal fat tissue |
| MFALCs | Mediastinal fat-associated lymphoid clusters |
| NHC | Natural helper cells |
| d | Days |
| PBS | Phosphate buffer saline |
| PAS | Hx-Periodic Acid Schiff-hematoxylin |
| AAALAC | Association for Assessment and Accreditation of Laboratory Animal Care |
References
- Kim, H.Y.; De Kruyff, R.H.; Umetsu, D.T. The many paths to asthma: Phenotype shaped by innate and adaptive immunity. Nat. Immunol. 2010, 11, 577–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambrecht, B.N.; Hammad, H. The immunology of asthma. Nat. Immunol. 2015, 16, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Asher, M.I.; Montefort, S.; Björkstén, B.; Lai, C.K.; Strachan, D.P.; Weiland, S.K.; Williams, H. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet 2006, 368, 733–743. [Google Scholar] [CrossRef]
- Thomson, N.C.; Chaudhuri, R.; Livingston, E. Asthma and cigarette smoking. Eur Respir J. 2004, 24, 822–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milne, J.; Brand, S. Occupational asthma after inhalation of dust of the proteolytic enzyme, papain. Occup. Environ. Med. 1975, 32, 302–307. [Google Scholar] [CrossRef]
- Goeminne, P.; Adams, E.; Deschepper, K.; Valcke, Y.; Nemery, B. Papain-Induced Asthma: A Man with Dyspnea from Dawn Till Dust. Acta Clinica Belgica 2013, 68, 132–134. [Google Scholar] [CrossRef]
- Halim, T.; Krauß, R.H.; Sun, A.C.; Takei, F. Lung Natural Helper Cells Are a Critical Source of Th2 Cell-Type Cytokines in Protease Allergen-Induced Airway Inflammation. Immunity 2012, 36, 451–463. [Google Scholar] [CrossRef] [Green Version]
- Fukuda, K.; Matsuzaki, H.; Hiraishi, Y.; Mikami, Y.; Makita, K.; Nagase, T. Development of papain-induced asthma-COPD overlap mice model. Eur Respir J. 2019, 54, 4070. [Google Scholar]
- Nials, A.T.; Uddin, S. Mouse models of allergic asthma: Acute and chronic allergen challenge. Dis. Model. Mech. 2008, 1, 213–220. [Google Scholar] [CrossRef] [Green Version]
- Holgate, S.T. Pathogenesis of Asthma. Allergy Allergic Dis. 2009, 38, 1608–1631. [Google Scholar] [CrossRef]
- Barnes, P.J. Immunology of asthma and chronic obstructive pulmonary disease. Nat. Rev. Immunol. 2008, 8, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Kay, A. Role of T Cells in Asthma. Mucosal T Cells 1998, 71, 178–191. [Google Scholar] [CrossRef]
- Drake, L.Y.; Iijima, K.; Hara, K.; Kobayashi, T.; Kephart, G.M.; Kita, H. B Cells Play Key Roles in Th2-Type Airway Immune Responses in Mice Exposed to Natural Airborne Allergens. PLoS ONE 2015, 10, e0121660. [Google Scholar] [CrossRef]
- Oboki, K.; Ohno, T.; Kajiwara, N.; Arae, K.; Morita, H.; Ishii, A.; Nambu, A.; Abe, T.; Kiyonari, H.; Matsumoto, K.; et al. IL-33 is a crucial amplifier of innate rather than acquired immunity. Proc. Natl. Acad. Sci. USA 2010, 107, 18581–18586. [Google Scholar] [CrossRef] [Green Version]
- Alisjahbana, A.; Mohammad, I.; Gao, Y.; Evren, E.; Ringqvist, E.; Willinger, T. Human macrophages and innate lymphoid cells: Tissue-resident innate immunity in humanized mice. Biochem. Pharmacol. 2020, 174, 113672. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D.; Kincaid, K.; Alt, J.M.; Heilman, M.J.; Hill, A.M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J. Immunol. 2000, 164, 6166–6173. [Google Scholar] [CrossRef] [Green Version]
- Hirayama, D.; Iida, T.; Nakase, H. The Phagocytic Function of Macrophage-Enforcing Innate Immunity and Tissue Homeo-stasis. Int. J. Mol. Sci. 2017, 19, 92. [Google Scholar] [CrossRef] [Green Version]
- Halim, T.; Steer, C.A.; Mathä, L.; Gold, M.J.; Martinez-Gonzalez, I.; McNagny, K.; McKenzie, A.N.; Takei, F. Group 2 Innate Lymphoid Cells Are Critical for the Initiation of Adaptive T Helper 2 Cell-Mediated Allergic Lung Inflammation. Immunity 2014, 40, 425–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moro, K.; Yamada, T.; Tanabe, M.; Takeuchi, T.; Ikawa, T.; Kawamoto, H.; Furusawa, J.; Ohtani, M.; Fujii, H.; Koyasu, S. Innate production of T(H)2 cytokines by adipose tissue-associated c-Kit (+) Sca-1(+) lymphoid cells. Nature 2010, 463, 540–544. [Google Scholar] [CrossRef]
- Elewa, Y.H.A.; Ichii, O.; Otsuka, S.; Hashimoto, Y.; Kon, Y. Characterization of mouse mediastinal fat-associated lymphoid clusters. Cell Tissue Res. 2014, 357, 731–741. [Google Scholar] [CrossRef]
- Elewa, Y.H.A.; Ichii, O.; Kon, Y. Comparative analysis of mediastinal fat-associated lymphoid cluster development and lung cellular infiltration in murine autoimmune disease models and the corresponding normal control strains. Immunology 2015, 147, 30–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elewa, Y.H.A.; Ichii, O.; Takada, K.; Nakamura, T.; Masum, M.A.; Kon, Y. Histopathological Correlations between Mediastinal Fat-Associated Lymphoid Clusters and the Development of Lung Inflammation and Fibrosis following Bleomycin Admin-istration in Mice. Front. Immunol. 2018, 9, 271. [Google Scholar] [CrossRef] [Green Version]
- Zaffini, R.; Gotte, G.; Menegazzi, M. Asthma and poly (ADP-ribose) polymerase inhibition: A new therapeutic approach. Drug Des. Dev. Ther. 2018, 12, 281–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, J.; Wiley, R.E.; Fattouh, R.; Swirski, F.; Gajewska, B.U.; Coyle, A.J.; Gutierrez-Ramos, J.-C.; Ellis, R.; Inman, M.D.; Jordana, M. Continuous Exposure to House Dust Mite Elicits Chronic Airway Inflammation and Structural Remodeling. Am. J. Respir. Crit. Care Med. 2004, 169, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Rodriguez, S.; Ford, W.; Broadley, K.; Kidd, E. Establishing the phenotype in novel acute and chronic murine models of allergic asthma. Int. Immunopharmacol. 2008, 8, 756–763. [Google Scholar] [CrossRef]
- Bousquet, J.; Jeffery, P.K.; Busse, W.W.; Johnson, M.; Vignola, A.M. Asthma. From bronchoconstriction to airways inflamma-tion and remodeling. Am. J. Respir. Crit. Care Med. 2000, 161, 1720–1745. [Google Scholar] [CrossRef] [Green Version]
- Fish, J.E.; Peters, S.P. Airway remodeling and persistent airway obstruction in asthma. J. Allergy Clin. Immunol. 1999, 104, 509–516. [Google Scholar] [CrossRef]
- Bénézech, C.; Luu, N.T.; Walker, J.A.; Kruglov, A.A.; Loo, Y.; Nakamura, K.; Zhang, Y.; Nayar, S.; Jones, L.H.; Flo-res-Langarica, A.; et al. Inflammation induced formation of fat-associated lymphoid clusters. Nat. Immunol. 2015, 16, 819–828. [Google Scholar] [CrossRef]
- Elewa, Y.H.A.; Ichii, O.; Kon, Y. Sex-related differences in autoimmune-induced lung lesions in MRL/MpJ-faslpr mice are mediated by the development of mediastinal fat-associated lymphoid clusters. Autoimmunity 2017, 50, 306–316. [Google Scholar] [CrossRef]
- Boonyarattanasoonthorn, T.; Elewa, Y.H.A.; Tag-El-Din-Hassan, H.T.; Morimatsu, M.; Agui, T. Profiling of cellular immune responses to Mycoplasma pulmonis infection in C57BL/6 and DBA/2 mice. Infect. Genet. Evol. 2019, 73, 55–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elewa, Y.H.; Ichii, O.; Nakamura, T.; Kon, Y. Pathological Alternations of Mediastinal Fat-Associated Lymphoid Cluster and Lung in a Streptozotocin-Induced Diabetic Mouse Model. Microsc. Microanal. 2021, 27, 187–200. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elewa, Y.H.A.; Abd Elwakil, M.M.; Ichii, O.; Nakamura, T.; Mohamed, S.K.A.; Kon, Y. Possible Crosstalk of the Immune Cells within the Lung and Mediastinal Fat-Associated Lymphoid Clusters in the Acute Inflammatory Lung Asthma-Like Mouse Model. Int. J. Mol. Sci. 2021, 22, 6878. https://doi.org/10.3390/ijms22136878
Elewa YHA, Abd Elwakil MM, Ichii O, Nakamura T, Mohamed SKA, Kon Y. Possible Crosstalk of the Immune Cells within the Lung and Mediastinal Fat-Associated Lymphoid Clusters in the Acute Inflammatory Lung Asthma-Like Mouse Model. International Journal of Molecular Sciences. 2021; 22(13):6878. https://doi.org/10.3390/ijms22136878
Chicago/Turabian StyleElewa, Yaser Hosny Ali, Mahmoud Mansour Abd Elwakil, Osamu Ichii, Teppei Nakamura, Sherif Kh. A. Mohamed, and Yasuhiro Kon. 2021. "Possible Crosstalk of the Immune Cells within the Lung and Mediastinal Fat-Associated Lymphoid Clusters in the Acute Inflammatory Lung Asthma-Like Mouse Model" International Journal of Molecular Sciences 22, no. 13: 6878. https://doi.org/10.3390/ijms22136878





