Emerging Advances of Nanotechnology in Drug and Vaccine Delivery against Viral Associated Respiratory Infectious Diseases (VARID)
Abstract
:1. Introduction
2. Anti-Viral Responses of the Immune System in VARID and Evidence of Nanomedicine
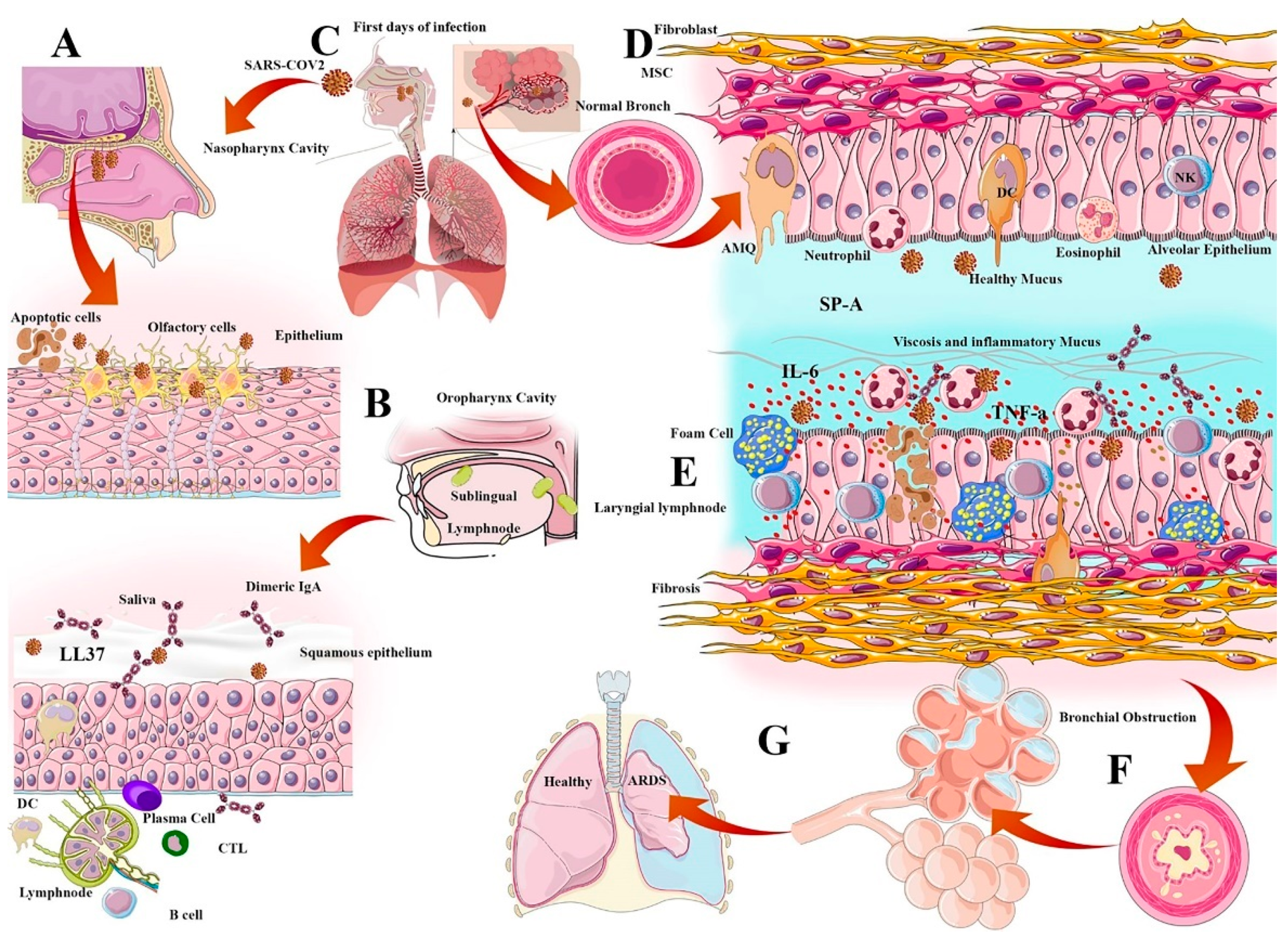
2.1. Physical-Mucosal Barriers from Saliva to Bronchus-Associated Lymphoid Tissue
2.2. Surfactant Role in Viral Infection
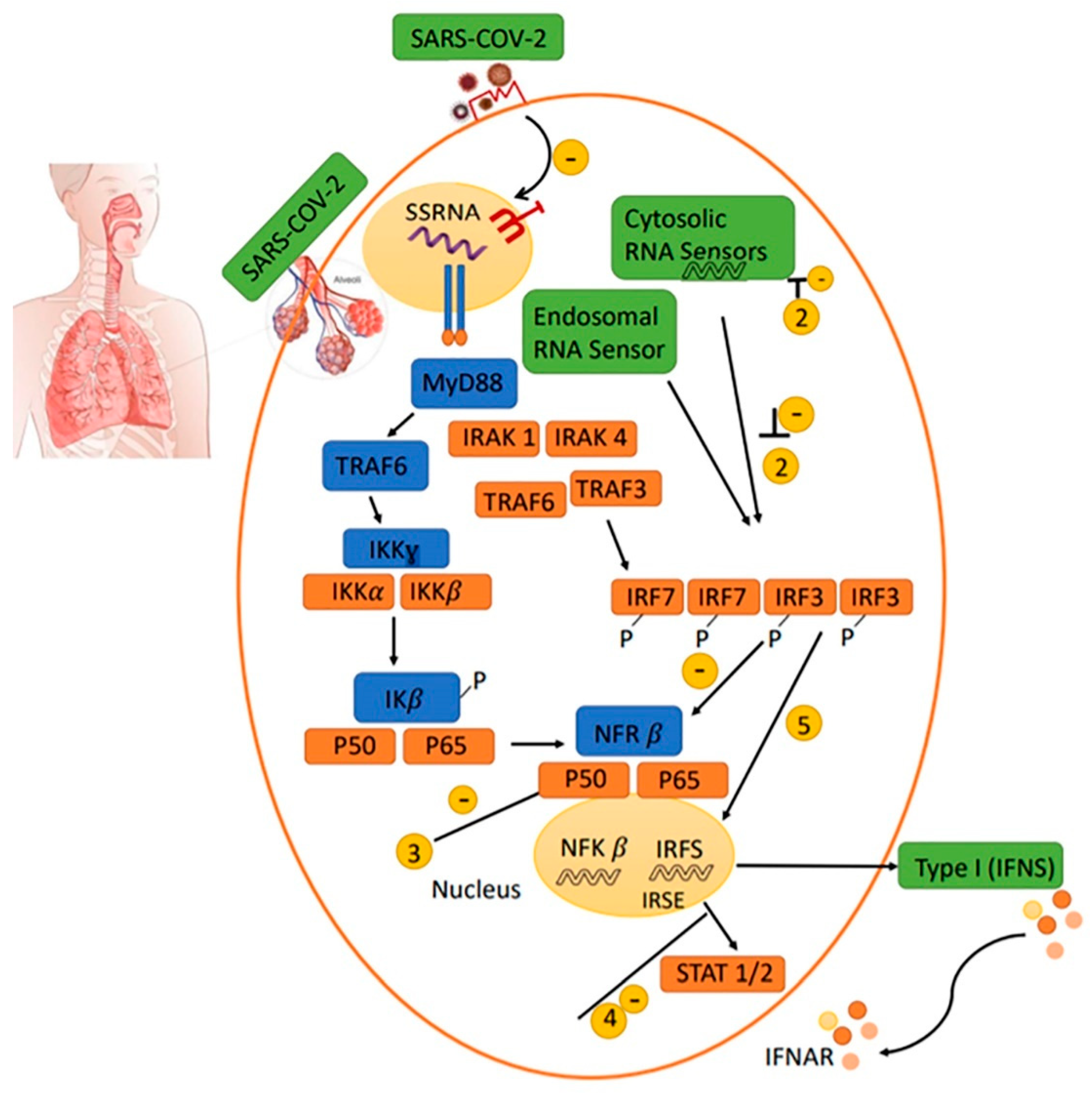
2.3. Antiviral IFN Route
2.4. Natural and Secondary IgA
2.5. Cell Mediated Immunity (CMI)
3. Anti-Viral Systemic or Local Nano-Vaccination and Immunotherapy
3.1. Nanocarriers for Targeted Anti-Viral Drug Delivery and Nano-Vaccine Design
3.1.1. Liposomes
3.1.2. Polymeric Nanoparticles
3.1.3. Dendrimers
3.1.4. Quantum Dots and Inorganic Nanoparticle
3.1.5. SAPNs and VLPs
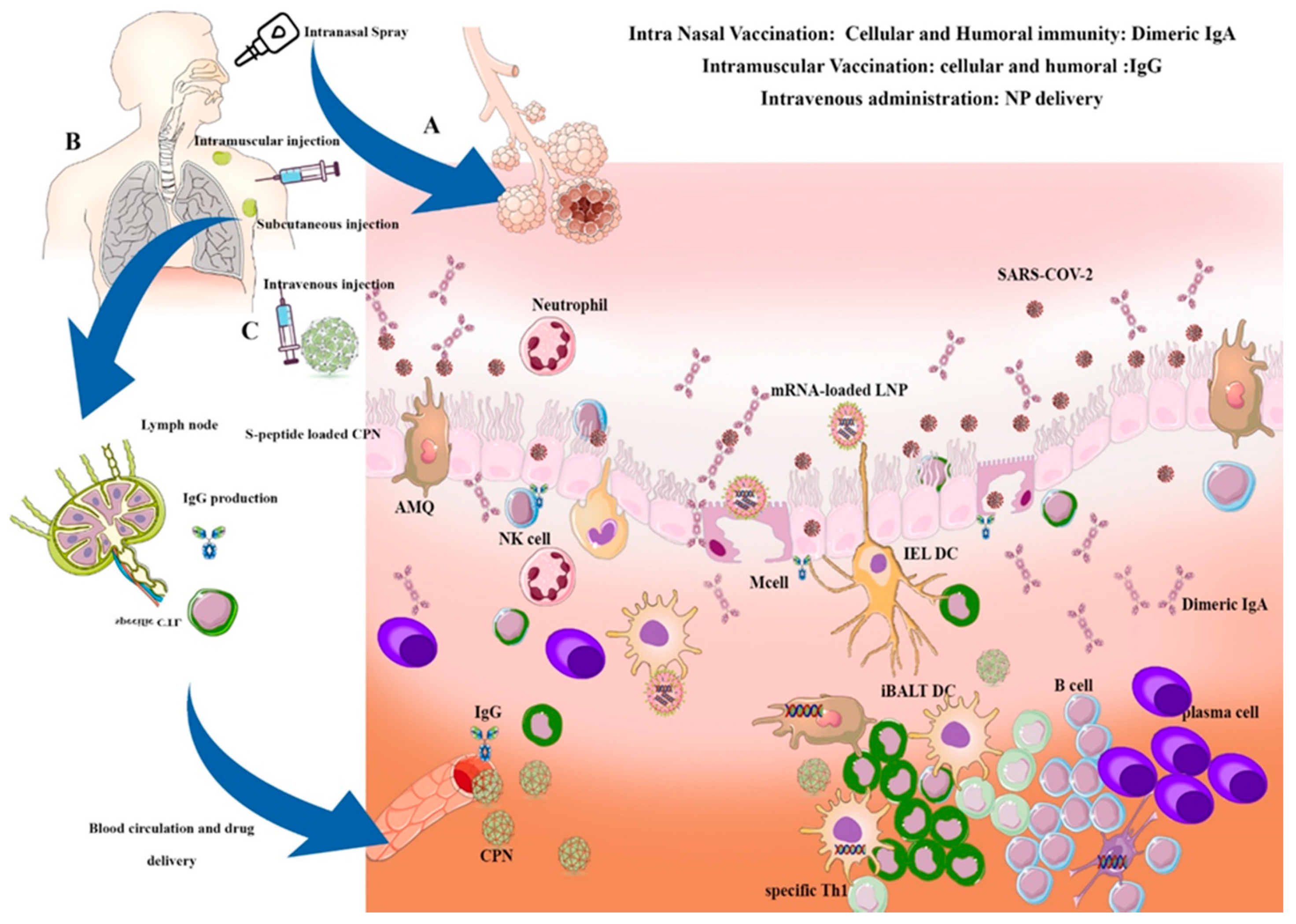
4. Local Airway Delivery of Nanoparticles in VARID
4.1. Intranasal Airway Delivery of Therapeutic Nano-Carriers in VARID
4.2. Intranasal Airway Delivery of Nano-Vaccines in VARID
5. Key Points on Nanomedicine and Nano-Vaccine against COVID-19
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADE | antibody-dependent enhancement |
| APC | antigen-presenting cell |
| CpG | cytosine-guanine rich oligonucleotide |
| DC | dendritic cell |
| EMA | European Medicines Agency |
| FDA | Food and Drug Administration |
| hACE2 | human angiotensin converter enzyme-2 |
| HPMA/NIPAM | N-(2-hydroxypropyl) methacrylamide/N-isopropylacrylamide |
| IEDB | Immune Epitope Database and Analysis Resource |
| IFN-γ | interferon-g |
| IL | interleukin |
| MERS-CoV | Middle East respiratory syndrome coronavirus |
| NALT | nasal-associated lymphoid tissue |
| NLR | nucleotide-binding domain and leucine-rich repeat-containing |
| NLRP3 | NLR Family Pyrin Domain Containing 3 |
| PapMV | papaya mosaic virus |
| PEG | polyethylene glycol |
| PGA | poly-glutamic acid |
| pIgR | polymeric Ig receptor |
| PLGA | poly lactic-co-glycolic acid |
| RBD | receptor-binding domain |
| ROS | reactive oxygen species |
| RSV | respiratory syncytial virus |
| SAPNs | self-assembling protein and peptide nanoparticles |
| SARS | severe acute respiratory syndrome |
| VLPs | virus-like particles |
| CMV | Cytomegalovirus |
References
- Sohrabi, C.; Alsafi, Z.; O’Neill, N.; Khan, M.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int. J. Surg. 2020, 76, 71–76. [Google Scholar] [CrossRef] [PubMed]
- van Doorn, H.R.; Yu, H. Viral Respiratory Infections. Hunt. Trop. Med. Emerg. Infect. Dis. 2020, 284–288. [Google Scholar] [CrossRef]
- Ibricevic, A.; Pekosz, A.; Walter, M.J.; Newby, C.; Battaile, J.T.; Brown, E.G.; Holtzman, M.J.; Brody, S.L. Influenza virus receptor specificity and cell tropism in mouse and human airway epithelial cells. J. Virol. 2006, 80, 7469–7480. [Google Scholar] [CrossRef] [Green Version]
- Sureda, A.; Alizadeh, J.; Nabavi, S.F.; Berindan-Neagoe, I.; Cismaru, C.A.; Jeandet, P.; Los, M.J.; Clementi, E.; Nabavi, S.M.; Ghavami, S. Endoplasmic reticulum as a potential therapeutic target for covid-19 infection management? Eur. J. Pharmacol. 2020, 882, 173288. [Google Scholar] [CrossRef]
- Zhang, H.; Penninger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef] [Green Version]
- Singhal, T. A Review of Coronavirus Disease-2019 (COVID-19). Indian J. Pediatrics 2020, 87, 281–286. [Google Scholar] [CrossRef] [Green Version]
- Newton, A.H.; Cardani, A.; Braciale, T.J. The host immune response in respiratory virus infection: Balancing virus clearance and immunopathology. Semin. Immunopathol. 2016, 38, 471–482. [Google Scholar] [CrossRef] [Green Version]
- Sanders, J.M.; Monogue, M.L.; Jodlowski, T.Z.; Cutrell, J.B. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review. JAMA 2020, 323, 1824–1836. [Google Scholar] [CrossRef]
- Hayden, F. Developing new antiviral agents for influenza treatment: What does the future hold? Clin. Infect. Dis. 2009, 48 (Suppl. 1), S3–S13. [Google Scholar] [CrossRef] [Green Version]
- Shojaei, S.; Suresh, M.; Klionsky, D.J.; Labouta, H.I.; Ghavami, S. Autophagy and SARS-CoV-2 infection: Apossible smart targeting of the autophagy pathway. Virulence 2020, 11, 805–810. [Google Scholar] [CrossRef]
- Chen, L.; Liang, J. An overview of functional nanoparticles as novel emerging antiviral therapeutic agents. Mater. Sci. Eng. C 2020, 112, 110924. [Google Scholar] [CrossRef]
- Mohamed, A.; Pekoz, A.Y.; Ross, K.; Hutcheon, G.A.; Saleem, I.Y. Pulmonary delivery of nanocomposite microparticles (NCMPS) incorporating mir-146a for treatment of COPD. Int. J. Pharm. 2019, 569, 118524. [Google Scholar] [CrossRef]
- Ferreira, R.C.; Neves, H.; Pinto, J.F.; Lopes, C.M. Overview on Inhalable Nanocarriers for Respiratory Immunization. Curr. Pharm. Des. 2017, 23, 6160–6181. [Google Scholar] [CrossRef] [PubMed]
- Zuercher, A.W. Upper Respiratory Tract Immunity. Viral Immunol. 2003, 16, 279–289. [Google Scholar] [CrossRef]
- Lynch, J.P., 3rd; Kajon, A.E. Adenovirus: Epidemiology, Global Spread of Novel Serotypes, and Advances in Treatment and Prevention. Semin. Respir. Crit. Care Med. 2016, 37, 586–602. [Google Scholar] [PubMed] [Green Version]
- Garcia, J.; Espejo, V.; Nelson, M.; Sovero, M.; Villaran, M.V.; Gomez, J.; Barrantes, M.; Sanchez, F.; Comach, G.; Arango, A.E.; et al. Human rhinoviruses and enteroviruses in influenza-like illness in Latin America. Virol. J. 2013, 10, 305. [Google Scholar] [CrossRef] [Green Version]
- Macias, A.E.; McElhaney, J.E.; Chaves, S.S.; Nealon, J.; Nunes, M.C.; Samson, S.I.; Seet, B.T.; Weinke, T.; Yu, H. The disease burden of influenza beyond respiratory illness. Vaccine 2021, 39 (Suppl. 1), A6–A14. [Google Scholar] [CrossRef]
- Williams, J.V. Human metapneumovirus: An important cause of respiratory disease in children and adults. Curr. Infect. Dis. Rep. 2005, 7, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Branche, A.R.; Falsey, A.R. Parainfluenza Virus Infection. Semin. Respir. Crit. Care Med. 2016, 37, 538–554. [Google Scholar] [CrossRef]
- Meng, J.; Stobart, C.C.; Hotard, A.L.; Moore, M.L. An overview of respiratory syncytial virus. PLoS Pathog. 2014, 10, e1004016. [Google Scholar] [CrossRef]
- Gwaltney, J.M., Jr.; Jordan, W.S., Jr. Rhinoviruses and Respiratory Disease. Bacteriol. Rev. 1964, 28, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Tacchi, L.; Musharrafieh, R.; Larragoite, E.T.; Crossey, K.; Erhardt, E.B.; Martin, S.A.M.; LaPatra, S.E.; Salinas, I. Nasal immunity is an ancient arm of the mucosal immune system of vertebrates. Nat. Commun. 2014, 5, 5205. [Google Scholar] [CrossRef]
- Porzia, A.; Cavaliere, C.; Begvarfaj, E.; Masieri, S.; Mainiero, F. Human nasal immune system: A special site for immune response establishment. J. Biol. Regul. Homeost. Agents 2018, 32 (Suppl. 1), 3–8. [Google Scholar]
- Essaidi-Laziosi, M.; Brito, F.; Benaoudia, S.; Royston, L.; Cagno, V.; Fernandes-Rocha, M.; Piuz, I.; Zdobnov, E.; Huang, S.; Constant, S.; et al. Propagation of respiratory viruses in human airway epithelia reveals persistent virus-specific signatures. J. Allergy Clin. Immunol. 2018, 141, 2074–2084. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Pandey, A.N.; Jain, S.K. Nasal-nanotechnology: Revolution for efficient therapeutics delivery. Drug Deliv. 2016, 23, 671–683. [Google Scholar] [CrossRef]
- Moutsopoulos, N.M.; Konkel, J.E. Tissue-Specific Immunity at the Oral Mucosal Barrier. Trends Immunol. 2018, 39, 276–287. [Google Scholar] [CrossRef] [Green Version]
- Feller, L.; Altini, M.; Khammissa, R.A.G.; Chandran, R.; Bouckaert, M.; Lemmer, J. Oral mucosal immunity. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2013, 116, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Osorio, R.; Alfonso-Rodríguez, C.A.; Medina-Castillo, A.L.; Alaminos, M.; Toledano, M. Bioactive Polymeric Nanoparticles for Periodontal Therapy. PLoS ONE 2016, 11, e0166217. [Google Scholar] [CrossRef]
- Corstjens, P.L.A.M.; Abrams, W.R.; Malamud, D. Saliva and viral infections. Periodontology 2000 2016, 70, 93–110. [Google Scholar] [CrossRef]
- Wohlford-Lenane, C.L.; Meyerholz, D.K.; Perlman, S.; Zhou, H.; Tran, D.; Selsted, M.E.; McCray, P.B., Jr. Rhesus theta-defensin prevents death in a mouse model of severe acute respiratory syndrome coronavirus pulmonary disease. J. Virol. 2009, 83, 11385–11390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortega, M.; Guzmán Merino, A.; Fraile-Martínez, O.; Recio-Ruiz, J.; Pekarek, L.; Guijarro, L.G.; García-Honduvilla, N.; Álvarez-Mon, M.; Buján, J.; García-Gallego, S. Dendrimers and Dendritic Materials: From Laboratory to Medical Practice in Infectious Diseases. Pharmaceutics 2020, 12, 874. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, J.; Upadhyay, R.B.; Agrawal, P.; Jaitley, S.; Shekhar, R. Langerhans cells and their role in oral mucosal diseases. N. Am. J. Med. Sci. 2013, 5, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.; Williamson, D.; Titball, R. Vaccine delivery using nanoparticles. Front. Cell Infect. Microbiol. 2013, 3, 13. [Google Scholar] [CrossRef] [Green Version]
- Sousa, F.H.; Casanova, V.; Findlay, F.; Stevens, C.; Svoboda, P.; Pohl, J.; Proudfoot, L.; Barlow, P.G. Cathelicidins display conserved direct antiviral activity towards rhinovirus. Peptides 2017, 95, 76–83. [Google Scholar] [CrossRef]
- Tripathi, S.; Tecle, T.; Verma, A.; Crouch, E.; White, M.; Hartshorn, K.L. The human cathelicidin LL-37 inhibits influenza A viruses through a mechanism distinct from that of surfactant protein D or defensins. J. Gen. Virol. 2013, 94 Pt 1, 40–49. [Google Scholar] [CrossRef]
- Larrick, J.W.; Morgan, J.G.; Palings, I.; Hirata, M.; Yen, M.H. Complementary DNA sequence of rabbit CAP18—A unique lipopolysaccharide binding protein. Biochem. Biophys. Res. Commun. 1991, 179, 170–175. [Google Scholar] [CrossRef]
- Teubl, B.J.; Stojkovic, B.; Docter, D.; Pritz, E.; Leitinger, G.; Poberaj, I.; Prassl, R.; Stauber, R.H.; Fröhlich, E.; Khinast, J.G.; et al. The effect of saliva on the fate of nanoparticles. Clin. Oral Investig. 2018, 22, 929–940. [Google Scholar] [CrossRef]
- Wnorowska, U.; Fiedoruk, K.; Piktel, E.; Prasad, S.V.; Sulik, M.; Janion, M.; Daniluk, T.; Savage, P.B.; Bucki, R. Nanoantibiotics containing membrane-active human cathelicidin LL-37 or synthetic ceragenins attached to the surface of magnetic nanoparticles as novel and innovative therapeutic tools: Current status and potential future applications. J. NanobioTechnol. 2020, 18, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Howell, M.D.; Streib, J.E.; Kim, B.E.; Lesley, L.J.; Dunlap, A.P.; Geng, D.; Feng, Y.; Savage, P.B.; Leung, D.Y. Ceragenins: A class of antiviral compounds to treat orthopox infections. J. Investig. Dermatol. 2009, 129, 2668–2675. [Google Scholar] [CrossRef] [Green Version]
- Niemirowicz, K.; Durnaś, B.; Tokajuk, G.; Piktel, E.; Michalak, G.; Gu, X.; Kułakowska, A.; Savage, P.B.; Bucki, R. Formulation and candidacidal activity of magnetic nanoparticles coated with cathelicidin LL-37 and ceragenin CSA-13. Sci. Rep. 2017, 7, 4610. [Google Scholar] [CrossRef] [PubMed]
- Findlay, F.; Pohl, J.; Svoboda, P.; Shakamuri, P.; McLean, K.; Inglis, N.F.; Proudfoot, L.; Barlow, P.G. Carbon Nanoparticles Inhibit the Antimicrobial Activities of the Human Cathelicidin LL-37 through Structural Alteration. J. Immunol. (Baltim. Md. 1950) 2017, 199, 2483–2490. [Google Scholar] [CrossRef] [Green Version]
- Casanova, V.; Sousa, F.H.; Shakamuri, P.; Svoboda, P.; Buch, C.; D’Acremont, M.; Christophorou, M.A.; Pohl, J.; Stevens, C.; Barlow, P.G. Citrullination Alters the Antiviral and Immunomodulatory Activities of the Human Cathelicidin LL-37 During Rhinovirus Infection. Front. Immunol. 2020, 11, 85. [Google Scholar] [CrossRef] [PubMed]
- Roth, A.; Lütke, S.; Meinberger, D.; Hermes, G.; Sengle, G.; Koch, M.; Streichert, T.; Klatt, A.R. LL-37 fights SARS-CoV-2: The Vitamin D-Inducible Peptide LL-37 Inhibits Binding of SARS-CoV-2 Spike Protein to its Cellular Receptor Angiotensin Converting Enzyme 2 In Vitro. bioRxiv 2020. [Google Scholar] [CrossRef]
- Crane-Godreau, M.A.; Clem, K.J.; Payne, P.; Fiering, S. Vitamin D Deficiency and Air Pollution Exacerbate COVID-19 through Suppression of Antiviral Peptide LL37. Front. Public Health 2020, 8, 232. [Google Scholar] [CrossRef]
- Pradhan, B.; Guha, D.; Murmu, K.C.; Sur, A.; Ray, P.; Das, D.; Aich, P. Comparative efficacy analysis of anti-microbial peptides, LL-37 and indolicidin upon conjugation with CNT, in human monocytes. J. NanobioTechnol. 2017, 15, 44. [Google Scholar] [CrossRef] [PubMed]
- Shannahan, J.H.; Bai, W.; Brown, J.M. Implications of scavenger receptors in the safe development of nanotherapeutics. Recept. Clin. Investig. 2015, 2, e811. [Google Scholar]
- Shannahan, J.H.; Brown, J.M. Engineered nanomaterial exposure and the risk of allergic disease. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Bowden, D.H. The alveolar macrophage. Environ. Health Perspect. 1984, 55, 327–341. [Google Scholar] [CrossRef] [Green Version]
- Rao, L.; Xia, S.; Xu, W.; Tian, R.; Yu, G.; Gu, C.; Pan, P.; Meng, Q.-F.; Cai, X.; Qu, D.; et al. Decoy nanoparticles protect against COVID-19 by concurrently adsorbing viruses and inflammatory cytokines. Proc. Natl. Acad. Sci. USA 2020, 117, 27141–27147. [Google Scholar] [CrossRef]
- Wiley, J.A.; Richert, L.E.; Swain, S.D.; Harmsen, A.; Barnard, D.L.; Randall, T.D.; Jutila, M.; Douglas, T.; Broomell, C.; Young, M.; et al. Inducible Bronchus-Associated Lymphoid Tissue Elicited by a Protein Cage Nanoparticle Enhances Protection in Mice against Diverse Respiratory Viruses. PLoS ONE 2009, 4, e7142. [Google Scholar] [CrossRef] [Green Version]
- Hwang, J.Y.; Randall, T.D.; Silva-Sanchez, A. Inducible Bronchus-Associated Lymphoid Tissue: Taming Inflammation in the Lung. Front. Immunol. 2016, 7, 258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Li, J.; Cho, J.; Malik, A.B. Prevention of vascular inflammation by nanoparticle targeting of adherent neutrophils. Nat. Nanotechnol. 2014, 9, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.K.; Cheung, S.O.; Huang, L.; Niu, J.; Tao, C.; Ho, C.M.; Che, C.M.; Tam, P.K. Further evidence of the anti-inflammatory effects of silver nanoparticles. ChemMedChem 2009, 4, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
- Shoae-Hagh, P.; Rahimifard, M.; Navaei-Nigjeh, M.; Baeeri, M.; Gholami, M.; Mohammadirad, A.; Abdollahi, M. Zinc oxide nanoparticles reduce apoptosis and oxidative stress values in isolated rat pancreatic islets. Biol. Trace Elem. Res. 2014, 162, 262–269. [Google Scholar] [CrossRef]
- Glasser, J.R.; Mallampalli, R.K. Surfactant and its role in the pathobiology of pulmonary infection. Microbes Infect. 2012, 14, 17–25. [Google Scholar] [CrossRef] [Green Version]
- Perino, J.; Crouzier, D.; Spehner, D.; Debouzy, J.C.; Garin, D.; Crance, J.M.; Favier, A.L. Lung surfactant DPPG phospholipid inhibits vaccinia virus infection. Antivir. Res. 2011, 89, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Balakireva, L.; Schoehn, G.; Thouvenin, E.; Chroboczek, J. Binding of adenovirus capsid to dipalmitoyl phosphatidylcholine provides a novel pathway for virus entry. J. Virol. 2003, 77, 4858–4866. [Google Scholar] [CrossRef] [Green Version]
- Davidson, W.J.; Dorscheid, D.; Spragg, R.; Schulzer, M.; Mak, E.; Ayas, N.T. Exogenous pulmonary surfactant for the treatment of adult patients with acute respiratory distress syndrome: Results of a meta-analysis. Crit. Care 2006, 10, R41. [Google Scholar] [CrossRef] [Green Version]
- Kuronuma, K.; Mitsuzawa, H.; Takeda, K.; Nishitani, C.; Chan, E.D.; Kuroki, Y.; Nakamura, M.; Voelker, D.R. Anionic pulmonary surfactant phospholipids inhibit inflammatory responses from alveolar macrophages and U937 cells by binding the lipopolysaccharide-interacting proteins CD14 and MD-2. J. Biol. Chem. 2009, 284, 25488–25500. [Google Scholar] [CrossRef] [Green Version]
- Corbet, A. Clinical Trials of Synthetic Surfactant in the Respiratory Distress Syndrome of Premature Infants. Clin. Perinatol. 1993, 20, 737–760. [Google Scholar] [CrossRef]
- Li, X.; Geng, M.; Peng, Y.; Meng, L.; Lu, S. Molecular immune pathogenesis and diagnosis of COVID-19. J. Pharm Anal. 2020, 10, 102–108. [Google Scholar] [CrossRef]
- Prompetchara, E.; Ketloy, C.; Palaga, T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020, 38, 1–9. [Google Scholar] [PubMed]
- Pilette, C.; Ouadrhiri, Y.; Godding, V.; Vaerman, J.-P.; Sibille, Y. Lung mucosal immunity: Immunoglobulin-A revisited. Eur. Respir. J. 2001, 18, 571–588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brandtzaeg, P. Secretory immunity with special reference to the oral cavity. J. Oral Microbiol. 2013, 5, 20401. [Google Scholar] [CrossRef] [PubMed]
- Lanka, G.K.K.; Yu, J.-J.; Gong, S.; Gupta, R.; Mustafa, S.B.; Murthy, A.K.; Zhong, G.; Chambers, J.P.; Guentzel, M.N.; Arulanandam, B.P. IgA modulates respiratory dysfunction as a sequela to pulmonary chlamydial infection as neonates. Pathog. Dis. 2016, 74, ftv121. [Google Scholar] [CrossRef] [Green Version]
- Sharma, S.; Benson, H.A.; Mukkur, T.K.; Rigby, P.; Chen, Y. Preliminary studies on the development of IgA-loaded chitosan-dextran sulphate nanoparticles as a potential nasal delivery system for protein antigens. J. Microencapsul. 2013, 30, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Jorquera, P.A.; Tripp, R.A. Synthetic Biodegradable Microparticle and Nanoparticle Vaccines against the Respiratory Syncytial Virus. Vaccines 2016, 4, 45. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, M.C. Sol-gel Silica Nanoparticles in Medicine: A Natural Choice. Design, Synthesis and Products. Molecules 2018, 23, 2021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heida, R.; Hinrichs, W.L.; Frijlink, H.W. Inhaled vaccine delivery in the combat against respiratory viruses: A 2021 overview of recent developments and implications for COVID-19. Expert Rev. Vaccines 2021. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Noh, Y.W.; Lim, Y.T. Polymer nanoparticles for cross-presentation of exogenous antigens and enhanced cytotoxic T-lymphocyte immune response. Int. J. Nanomed. 2016, 11, 3753–3764. [Google Scholar]
- Lee, M.-Y.; Yang, J.-A.; Jung, H.S.; Beack, S.; Choi, J.E.; Hur, W.; Koo, H.; Kim, K.; Yoon, S.K.; Hahn, S.K. Hyaluronic Acid–Gold Nanoparticle/Interferon α Complex for Targeted Treatment of Hepatitis C Virus Infection. ACS Nano 2012, 6, 9522–9531. [Google Scholar] [CrossRef]
- Al-Halifa, S.; Gauthier, L.; Arpin, D.; Bourgault, S.; Archambault, D. Nanoparticle-based vaccines against respiratory viruses. Front. Immunol. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gheibi Hayat, S.M.; Darroudi, M. Nanovaccine: A novel approach in immunization. J. Cell Physiol. 2019, 234, 12530–12536. [Google Scholar] [CrossRef] [PubMed]
- Singh Sekhon, B.; Saluja, V. Nanovaccines-an overview. Int. J. Pharm. Front. Res. 2011, 1, 101–109. [Google Scholar]
- Marasini, N.; Skwarczynski, M.; Toth, I. Intranasal delivery of nanoparticle-based vaccines. Ther. Deliv. 2017, 8, 151–167. [Google Scholar] [CrossRef]
- Laval, J.M.; Mazeran, P.E.; Thomas, D. NanobioTechnology and its role in the development of new analytical devices. Analyst 2000, 125, 29–33. [Google Scholar] [CrossRef]
- Utley, T.; Ducharme, N.; Varthakavi, V.; Shepherd, B.; Santangelo, P.; Lindquist, M.; Goldenring, J.; Crowe, J. Respiratory syncytial virus uses a Vps4-independent budding mechanism controlled by Rab11-FIP2. Proc. Natl. Acad. Sci. USA 2008, 105, 10209–10214. [Google Scholar] [CrossRef] [Green Version]
- Daubeney, P.; Taylor, C.; McGaw, J.; Brown, E.; Ghosal, S.; Keeton, B.; Palache, B.; Kerstens, R. Immunogenicity and tolerability of a trivalent influenza subunit vaccine (Influvac) in high-risk children aged 6 months to 4 years. Br. J. Clin. Pract. 1997, 51, 87–90. [Google Scholar]
- Gomes, A.C.; Mohsen, M.; Bachmann, M.F. Harnessing nanoparticles for immunomodulation and vaccines. Vaccines 2017, 5, 6. [Google Scholar] [CrossRef]
- Delore, V.; Salamand, C.; Marsh, G.; Arnoux, S.; Pepin, S.; Saliou, P. Long-term clinical trial safety experience with the inactivated split influenza vaccine, Vaxigrip®. Vaccine 2006, 24, 1586–1592. [Google Scholar] [CrossRef]
- Grohskopf, L.A.; Sokolow, L.Z.; Olsen, S.J.; Bresee, J.S.; Broder, K.R.; Karron, R.A. Prevention and control of influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices, United States, 2015–2016 influenza season. MMWR. Morb. Mortal. Wkly. Rep. 2015, 64, 818. [Google Scholar] [CrossRef] [PubMed]
- Carter, N.J.; Curran, M.P. Live attenuated influenza vaccine (FluMist®; Fluenz™). Drugs 2011, 71, 1591–1622. [Google Scholar] [CrossRef]
- Dhere, R.; Yeolekar, L.; Kulkarni, P.; Menon, R.; Vaidya, V.; Ganguly, M.; Tyagi, P.; Barde, P.; Jadhav, S. A pandemic influenza vaccine in India: From strain to sale within 12 months. Vaccine 2011, 29, A16–A21. [Google Scholar] [CrossRef] [Green Version]
- Chattopadhyay, S.; Chen, J.; Chen, H.; Hu, C. Nanoparticle vaccines adopting virus-like features for enhanced immune potentiation. Nanotheranostics 2017, 1, 244–260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fromen, C.A.; Robbins, G.R.; Shen, T.W.; Kai, M.P.; Ting, J.P.; DeSimone, J.M. Controlled analysis of nanoparticle charge on mucosal and systemic antibody responses following pulmonary immunization. Proc. Natl. Acad. Sci. USA 2015, 112, 488–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahimian, S.; Kleinovink, J.W.; Fransen, M.F.; Mezzanotte, L.; Gold, H.; Wisse, P.; Overkleeft, H.; Amidi, M.; Jiskoot, W.; Löwik, C.W.; et al. Near-infrared labeled, ovalbumin loaded polymeric nanoparticles based on a hydrophilic polyester as model vaccine: In vivo tracking and evaluation of antigen-specific CD8+ T cell immune response. Biomaterials 2015, 37, 469–477. [Google Scholar] [CrossRef]
- Kishimoto, T.K.; Maldonado, R.A. Nanoparticles for the induction of antigen-specific immunological tolerance. Front. Immunol. 2018, 9, 230. [Google Scholar] [CrossRef]
- Tao, W.; Hurst, B.L.; Shakya, A.K.; Uddin, M.J.; Ingrole, R.S.; Hernandez-Sanabria, M.; Arya, R.P.; Bimler, L.; Paust, S.; Tarbet, E.B. Consensus M2e peptide conjugated to gold nanoparticles confers protection against H1N1, H3N2 and H5N1 influenza A viruses. Antivir. Res. 2017, 141, 62–72. [Google Scholar] [CrossRef] [Green Version]
- Tai, W.; He, L.; Zhang, X.; Pu, J.; Voronin, D.; Jiang, S.; Zhou, Y.; Du, L. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: Implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol. Immunol. 2020, in press. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhattacharya, M.; Sharma, A.R.; Patra, P.; Ghosh, P.; Sharma, G.; Patra, B.C.; Lee, S.-S.; Chakraborty, C. Development of epitope-based peptide vaccine against novel coronavirus 2019 (SARS-COV-2): Immunoinformatics approach. J. Med. Virol. 2020, 92, 618–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Svindland, S.C.; Jul-Larsen, Å.; Pathirana, R.; Andersen, S.; Madhun, A.; Montomoli, E.; Jabbal-Gill, I.; Cox, R.J. The mucosal and systemic immune responses elicited by a chitosan-adjuvanted intranasal influenza H5N1 vaccine. Influenza Other Respir. Viruses 2012, 6, 90–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medepalli, K.K. Advanced Nanomaterials for Biomedical Applications; University of Louisville: Louisville, KY, USA, 2008. [Google Scholar]
- Akbarzadeh, A.; Rezaei-Sadabady, R.; Davaran, S.; Joo, S.W.; Zarghami, N.; Hanifehpour, Y.; Samiei, M.; Kouhi, M.; Nejati-Koshki, K. Liposome: Classification, preparation, and applications. Nanoscale Res. Lett. 2013, 8, 102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mallick, S.; Choi, J.S. Liposomes: Versatile and biocompatible nanovesicles for efficient biomolecules delivery. J. Nanosci. Nanotechnol. 2014, 14, 755–765. [Google Scholar] [CrossRef]
- Perrie, Y.; Mohammed, A.R.; Kirby, D.J.; McNeil, S.E.; Bramwell, V.W. Vaccine adjuvant systems: Enhancing the efficacy of sub-unit protein antigens. Int. J. Pharm. 2008, 364, 272–280. [Google Scholar] [CrossRef]
- Nikitina, E.; Larionova, I.; Choinzonov, E.; Kzhyshkowska, J. Monocytes and macrophages as viral targets and reservoirs. Int. J. Mol. Sci. 2018, 19, 2821. [Google Scholar] [CrossRef] [Green Version]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D.; et al. COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Chen, M.; Wang, T. Liposomes used as a vaccine adjuvant-delivery system: From basics to clinical immunization. J. Control. Release 2019, 303, 130–150. [Google Scholar] [CrossRef] [PubMed]
- Henriksen-Lacey, M.; Devitt, A.; Perrie, Y. The vesicle size of DDA: TDB liposomal adjuvants plays a role in the cell-mediated immune response but has no significant effect on antibody production. J. Control. Release 2011, 154, 131–137. [Google Scholar] [CrossRef]
- Corbett, K.S.; Edwards, D.K.; Leist, S.R.; Abiona, O.M.; Boyoglu-Barnum, S.; Gillespie, R.A.; Himansu, S.; Schäfer, A.; Ziwawo, C.T.; DiPiazza, A.T.; et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 2020, 586, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Molecular Depot. Pfizer-BioNTech Moderna and Janssen Vaccines USA: Molecular Depot 2021. Available online: https://moleculardepot.com/2020/12/23/pfizer-biontech-and-moderna-covid-19-vaccines-comparison/ (accessed on 27 June 2021).
- Ulery, B.D.; Kumar, D.; Ramer-Tait, A.E.; Metzger, D.W.; Wannemuehler, M.J.; Narasimhan, B. Design of a protective single-dose intranasal nanoparticle-based vaccine platform for respiratory infectious diseases. PLoS ONE 2011, 6, e17642. [Google Scholar] [CrossRef] [Green Version]
- Vauthier, C.; Bouchemal, K. Methods for the preparation and manufacture of polymeric nanoparticles. Pharm. Res. 2009, 26, 1025–1058. [Google Scholar] [CrossRef] [PubMed]
- Meijer, D.; Jansen, R.; Molema, G. Drug targeting systems for antiviral agents: Options and limitations. Antivir. Res. 1992, 18, 215–258. [Google Scholar] [CrossRef]
- Wibowo, D.; Jorritsma, S.H.T.; Gonzaga, Z.J.; Evert, B.; Chen, S.; Rehm, B.H.A. Polymeric nanoparticle vaccines to combat emerging and pandemic threats. Biomaterials 2021, 268, 120597. [Google Scholar] [CrossRef] [PubMed]
- Lam, H.T.; Le-Vinh, B.; Phan, T.N.Q.; Bernkop-Schnürch, A. Self-emulsifying drug delivery systems and cationic surfactants: Do they potentiate each other in cytotoxicity? J. Pharm. Pharmacol. 2019, 71, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Kik, K.; Bukowska, B.; Sicińska, P. Polystyrene nanoparticles: Sources, occurrence in the environment, distribution in tissues, accumulation and toxicity to various organisms. Environ. Pollut. 2020, 262, 114297. [Google Scholar] [CrossRef]
- Yue, H.; Ma, G. Polymeric micro/nanoparticles: Particle design and potential vaccine delivery applications. Vaccine 2015, 33, 5927–5936. [Google Scholar] [CrossRef] [PubMed]
- Shah, L.K.; Amiji, M.M. Intracellular delivery of saquinavir in biodegradable polymeric nanoparticles for HIV/AIDS. Pharm. Res. 2006, 23, 2638–2645. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.-H.; Patel, N.; Haupt, R.; Zhou, H.; Weston, S.; Hammond, H.; Logue, J.; Portnoff, A.D.; Norton, J.; Guebre-Xabier, M.; et al. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373 immunogenicity in baboons and protection in mice. Nat. Commun. 2021, 12, 372. [Google Scholar] [CrossRef] [PubMed]
- Krasia-Christoforou, T.; Georgiou, T. Polymeric theranostics: Using polymer-based systems for simultaneous imaging and therapy. J. Mater. Chem. B 2013, 1, 3002–3025. [Google Scholar] [CrossRef]
- Tang, Z.; He, C.; Tian, H.; Ding, J.; Hsiao, B.S.; Chu, B.; Chen, X. Polymeric nanostructured materials for biomedical applications. Prog. Polym. Sci. 2016, 60, 86–128. [Google Scholar] [CrossRef] [Green Version]
- Vela-Ramirez, J.E.; Goodman, J.T.; Boggiatto, P.M.; Roychoudhury, R.; Pohl, N.L.B.; Hostetter, J.M.; Wannemuehler, M.J.; Narasimhan, B. Safety and biocompatibility of carbohydrate-functionalized polyanhydride nanoparticles. AAPS J. 2015, 17, 256–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrillo-Conde, B.; Schiltz, E.; Yu, J.; Chris Minion, F.; Phillips, G.J.; Wannemuehler, M.J.; Narasimhan, B. Encapsulation into amphiphilic polyanhydride microparticles stabilizes Yersinia pestis antigens. Acta Biomater. 2010, 6, 3110–3119. [Google Scholar] [CrossRef] [PubMed]
- Petersen, L.K.; Phanse, Y.; Ramer-Tait, A.E.; Wannemuehler, M.J.; Narasimhan, B. Amphiphilic polyanhydride nanoparticles stabilize Bacillus anthracis protective antigen. Mol. Pharm. 2012, 9, 874–882. [Google Scholar] [CrossRef] [Green Version]
- Ulery, B.D.; Petersen, L.K.; Phanse, Y.; Kong, C.S.; Broderick, S.R.; Kumar, D.; Ramer-Tait, A.E.; Carrillo-Conde, B.; Rajan, K.; Wannemuehler, M.J.; et al. Rational design of pathogen-mimicking amphiphilic materials as nanoadjuvants. Sci. Rep. 2011, 1, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ross, K.A.; Haughney, S.L.; Petersen, L.K.; Boggiatto, P.; Wannemuehler, M.J.; Narasimhan, B. Lung deposition and cellular uptake behavior of pathogen-mimicking nanovaccines in the first 48 hours. Adv. Healthc. Mater. 2014, 3, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.; Sahoo, S.K. PLGA nanoparticles containing various anticancer agents and tumour delivery by EPR effect. Adv. Drug Deliv. Rev. 2011, 63, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.L.; Soema, P.C.; Slütter, B.; Ossendorp, F.; Jiskoot, W. PLGA particulate delivery systems for subunit vaccines: Linking particle properties to immunogenicity. Hum. Vaccin. Immunother. 2016, 12, 1056–1069. [Google Scholar] [CrossRef]
- Getts, D.R.; Shea, L.D.; Miller, S.D.; King, N.J.C. Harnessing nanoparticles for immune modulation. Trends Immunol. 2015, 36, 419–427. [Google Scholar] [CrossRef] [Green Version]
- Mansoor, F.; Earley, B.; Cassidy, J.P.; Markey, B.; Doherty, S.; Welsh, M.D. Comparing the immune response to a novel intranasal nanoparticle PLA vaccine and a commercial BPI3V vaccine in dairy calves. BMC Vet. Res. 2015, 11, 220. [Google Scholar] [CrossRef] [Green Version]
- Dhakal, S.; Hiremath, J.; Bondra, K.; Lakshmanappa, Y.S.; Shyu, D.-L.; Ouyang, K.; Kang, K.-I.; Binjawadagi, B.; Goodman, J.; Tabynov, K. Biodegradable nanoparticle delivery of inactivated swine influenza virus vaccine provides heterologous cell-mediated immune response in pigs. J. Control. Release 2017, 247, 194–205. [Google Scholar] [CrossRef] [Green Version]
- Okamoto, S.; Matsuura, M.; Akagi, T.; Akashi, M.; Tanimoto, T.; Ishikawa, T.; Takahashi, M.; Yamanishi, K.; Mori, Y. Poly (γ-glutamic acid) nano-particles combined with mucosal influenza virus hemagglutinin vaccine protects against influenza virus infection in mice. Vaccine 2009, 27, 5896–5905. [Google Scholar] [CrossRef] [PubMed]
- Dhakal, S.; Renu, S.; Ghimire, S.; Shaan Lakshmanappa, Y.; Hogshead, B.T.; Feliciano-Ruiz, N.; Lu, F.; HogenEsch, H.; Krakowka, S.; Lee, C.W. Mucosal immunity and protective efficacy of intranasal inactivated influenza vaccine is improved by chitosan nanoparticle delivery in pigs. Front. Immunol. 2018, 9, 934. [Google Scholar] [CrossRef]
- Liu, Q.; Zheng, X.; Zhang, C.; Shao, X.; Zhang, X.; Zhang, Q.; Jiang, X. Conjugating influenza a (H1N1) antigen to n-trimethylaminoethylmethacrylate chitosan nanoparticles improves the immunogenicity of the antigen after nasal administration. J. Med. Virol. 2015, 87, 1807–1815. [Google Scholar] [CrossRef]
- McGill, J.L.; Kelly, S.M.; Kumar, P.; Speckhart, S.; Haughney, S.L.; Henningson, J.; Narasimhan, B.; Sacco, R.E. Efficacy of mucosal polyanhydride nanovaccine against Respiratory Syncytial Virus infection in the neonatal calf. Sci. Rep. 2018, 8, 3021. [Google Scholar] [CrossRef] [PubMed]
- Gunther, S.C.; Maier, J.D.; Vetter, J.; Podvalnyy, N.; Khanzhin, N.; Hennet, T.; Stertz, S. Antiviral potential of 3’-sialyllactose- and 6’-sialyllactose-conjugated dendritic polymers against human and avian influenza viruses. Sci. Rep. 2020, 10, 768. [Google Scholar] [CrossRef] [Green Version]
- Kandeel, M.; Al-Taher, A.; Park, B.K.; Kwon, H.-J.; Al-Nazawi, M. A pilot study of the antiviral activity of anionic and cationic polyamidoamine dendrimers against the Middle East respiratory syndrome coronavirus. J. Med. Virol. 2020, 92, 1665–1670. [Google Scholar] [CrossRef] [PubMed]
- Khaitov, M.; Nikonova, A.; Shilovskiy, I.; Kozhikhova, K.; Kofiadi, I.; Vishnyakova, L.; Nikolskii, A.; Gattinger, P.; Kovchina, V.; Barvinskaia, E.; et al. Silencing of SARS-CoV-2 with modified siRNA-peptide dendrimer formulation. Allergy 2021. [Google Scholar] [CrossRef]
- Paull, J.R.; Castellarnau, A.; Luscombe, C.A.; Fairley, J.K.; Heery, G.P. Astodrimer sodium, dendrimer antiviral, inhibits replication of SARS-CoV-2 in vitro. Biorxiv 2020. [Google Scholar] [CrossRef]
- Zhang, L.-J.; Wang, S.; Xia, L.; Lv, C.; Tang, H.-W.; Liang, Z.; Xiao, G.; Pang, D.-W. Lipid-specific labeling of enveloped viruses with quantum dots for single-virus tracking. mBio 2020, 11, e00135-20. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Gu, J.; Ye, J.; Fang, B.; Wan, S.; Wang, C.; Ashraf, U.; Li, Q.; Wang, X.; Shao, L. Benzoxazine monomer derived carbon dots as a broad-spectrum agent to block viral infectivity. J. Colloid Interface Sci. 2019, 542, 198–206. [Google Scholar] [CrossRef]
- Loczechin, A.; Séron, K.; Barras, A.; Giovanelli, E.; Belouzard, S.; Chen, Y.-T.; Metzler-Nolte, N.; Boukherroub, R.; Dubuisson, J.; Szunerits, S. Functional Carbon Quantum Dots as Medical Countermeasures to Human Coronavirus (HCoV). ACS Appl. Mater. Interfaces 2019, 11, 42964–42974. [Google Scholar] [CrossRef] [PubMed]
- Roh, C.; Jo, S.K. Quantitative and sensitive detection of SARS coronavirus nucleocapsid protein using quantum dots-conjugated RNA aptamer on chip. J. Chem. Technol. Biotechnol. 2011, 86, 1475–1479. [Google Scholar] [CrossRef] [PubMed]
- Poon, C.; Patel, A.A. Organic and inorganic nanoparticle vaccines for prevention of infectious diseases. Nano Express 2020, 1, 012001. [Google Scholar] [CrossRef]
- Haimov, E.; Weitman, H.; Polani, S.; Schori, H.; Zitoun, D.; Shefi, O. meso-Tetrahydroxyphenylchlorin-conjugated gold nanoparticles as a tool to improve photodynamic therapy. ACS Appl. Mater. Interfaces 2018, 10, 2319–2327. [Google Scholar] [CrossRef]
- Osminkina, L.; Timoshenko, V.Y.; Shilovsky, I.; Kornilaeva, G.; Shevchenko, S.; Gongalsky, M.; Tamarov, K.; Abramchuk, S.; Nikiforov, V.; Khaitov, M. Porous silicon nanoparticles as scavengers of hazardous viruses. J. Nanopart. Res. 2014, 16, 2430. [Google Scholar] [CrossRef]
- Huo, C.; Xiao, J.; Xiao, K.; Zou, S.; Wang, M.; Qi, P.; Liu, T.; Hu, Y. Pre-treatment with zirconia nanoparticles reduces inflammation induced by the pathogenic H5N1 influenza virus. Int. J. Nanomed. 2020, 15, 661. [Google Scholar] [CrossRef] [Green Version]
- Ghaffari, H.; Tavakoli, A.; Moradi, A.; Tabarraei, A.; Bokharaei-Salim, F.; Zahmatkeshan, M.; Farahmand, M.; Javanmard, D.; Kiani, S.J.; Esghaei, M. Inhibition of H1N1 influenza virus infection by zinc oxide nanoparticles: Another emerging application of nanomedicine. J. Biomed. Sci. 2019, 26, 70. [Google Scholar] [CrossRef]
- Mazurkova, N.; Spitsyna, Y.E.; Shikina, N.; Ismagilov, Z.; Zagrebel’nyi, S.; Ryabchikova, E. Interaction of titanium dioxide nanoparticles with influenza virus. Nanotechnol. Russ. 2010, 5, 417–420. [Google Scholar] [CrossRef]
- Ventura, B.D.; Cennamo, M.; Minopoli, A.; Campanile, R.; Censi, S.B.; Terracciano, D.; Portella, G.; Velotta, R. Colorimetric Test for Fast Detection of SARS-CoV-2 in Nasal and Throat Swabs. ACS Sens. 2020, 5, 3043–3048. [Google Scholar] [CrossRef]
- Marques Neto, L.M.; Kipnis, A.; Junqueira-Kipnis, A.P. Role of metallic nanoparticles in vaccinology: Implications for infectious disease vaccine development. Front. Immunol. 2017, 8, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Tao, W.; Ziemer, K.S.; Gill, H.S. Gold nanoparticle-M2e conjugate coformulated with CpG induces protective immunity against influenza A virus. Nanomedicine 2014, 9, 237–251. [Google Scholar] [CrossRef] [Green Version]
- Jazayeri, S.D.; Ideris, A.; Zakaria, Z.; Shameli, K.; Moeini, H.; Omar, A.R. Cytotoxicity and immunological responses following oral vaccination of nanoencapsulated avian influenza virus H5 DNA vaccine with green synthesis silver nanoparticles. J. Control. Release 2012, 161, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Sekimukai, H.; Iwata-Yoshikawa, N.; Fukushi, S.; Tani, H.; Kataoka, M.; Suzuki, T.; Hasegawa, H.; Niikura, K.; Arai, K.; Nagata, N. Gold nanoparticle-adjuvanted S protein induces a strong antigen-specific IgG response against severe acute respiratory syndrome-related coronavirus infection, but fails to induce protective antibodies and limit eosinophilic infiltration in lungs. Microbiol. Immunol. 2020, 64, 33–51. [Google Scholar] [CrossRef] [Green Version]
- Scheerlinck, J.-P.Y.; Greenwood, D.L.V. Virus-sized vaccine delivery systems. Drug Discov. Today 2008, 13, 882–887. [Google Scholar] [CrossRef]
- Schneider-Ohrum, K.; Ross, T. Mucosal Vaccines: Modern Concepts, Strategies, and Challenges; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Park, H.-J.; Lee, E.-Y.; Jung, S.; Ko, H.; Lee, S.-M.; Nam, J.-H. Spike nanoparticle and recombinant adenovirus 5 vaccines induce specific antibodies against the Middle East respiratory syndrome coronavirus (MERS-CoV). J. Immunol. 2017, 198, 225. [Google Scholar]
- Yamashita, I.; Iwahori, K.; Kumagai, S. Ferritin in the field of nanodevices. Biochim. Biophys. Acta 2010, 1800, 846–857. [Google Scholar] [CrossRef]
- Deng, L.; Mohan, T.; Chang, T.Z.; Gonzalez, G.X.; Wang, Y.; Kwon, Y.-M.; Kang, S.-M.; Compans, R.W.; Champion, J.A.; Wang, B.-Z. Double-layered protein nanoparticles induce broad protection against divergent influenza A viruses. Nat. Commun. 2018, 9, 359. [Google Scholar] [CrossRef]
- He, L.; Lin, X.; Wang, Y.; Abraham, C.; Sou, C.; Ngo, T.; Zhang, Y.; Wilson, I.A.; Zhu, J. Single-component, self-assembling, protein nanoparticles presenting the receptor binding domain and stabilized spike as SARS-CoV-2 vaccine candidates. Sci. Adv. 2021, 7, eabf1591. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.-F.; Sun, C.; Zhuang, Z.; Yuan, R.-Y.; Zheng, Q.; Li, J.-P.; Zhou, P.-P.; Chen, X.-C.; Liu, Z.; Zhang, X.; et al. Rapid Development of SARS-CoV-2 Spike Protein Receptor-Binding Domain Self-Assembled Nanoparticle Vaccine Candidates. ACS Nano 2021, 15, 2738–2752. [Google Scholar] [CrossRef] [PubMed]
- Kushnir, N.; Streatfield, S.J.; Yusibov, V. Virus-like particles as a highly efficient vaccine platform: Diversity of targets and production systems and advances in clinical development. Vaccine 2012, 31, 58–83. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, M.O.; Zha, L.; Cabral-Miranda, G.; Bachmann, M.F. Major findings and recent advances in virus–like particle (VLP)-based vaccines. Semin. Immunol. 2017, 34, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, C.; Rioux, G.; Dumas, M.-C.; Leclerc, D. Induction of innate immunity in lungs with virus-like nanoparticles leads to protection against influenza and Streptococcus pneumoniae challenge. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, A.; Zare, F.; Sazegari, S.; Afsharifar, A.; Eskandari, M.; Pormohammad, A. Development of a novel platform of virus-like particle (VLP)-based vaccine against COVID-19 by exposing epitopes: An immunoinformatics approach. New Microbes New Infect. 2020, 38, 100786. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, T.A.P.F.; Yan, Z.; Jeffers, S.A.; Holmes, K.V.; Hodges, R.S.; Burkhard, P. Peptide nanoparticles as novel immunogens: Design and analysis of a prototypic severe acute respiratory syndrome vaccine. Chem. Biol. Drug Des. 2009, 73, 53–61. [Google Scholar] [CrossRef] [Green Version]
- Xu, R.; Shi, M.; Li, J.; Song, P.; Li, N. Construction of SARS-CoV-2 Virus-Like Particles by Mammalian Expression System. Front. Bioeng. Biotechnol. 2020, 8, 862. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Ko, E.; Lee, Y.; Kim, K.; Kim, M.; Lee, Y.; Kang, S. Intranasal vaccination with M2e5x virus-like particles induces humoral and cellular immune responses conferring cross-protection against heterosubtypic influenza viruses. PLoS ONE 2018, 13, e0190868. [Google Scholar] [CrossRef] [Green Version]
- Swann, H.; Sharma, A.; Preece, B.; Peterson, A.; Eldridge, C.; Belnap, D.M.; Vershinin, M.; Saffarian, S. Minimal system for assembly of SARS-CoV-2 virus like particles. Sci. Rep. 2020, 10, 21877. [Google Scholar] [CrossRef]
- Lu, J.; Lu, G.; Tan, S.; Xia, J.; Xiong, H.; Yu, X.; Qi, Q.; Yu, X.; Li, L.; Yu, H. A COVID-19 mRNA vaccine encoding SARS-CoV-2 virus-like particles induces a strong antiviral-like immune response in mice. Cell Res. 2020, 30, 936–939. [Google Scholar] [CrossRef] [PubMed]
- Sawaengsak, C.; Mori, Y.; Yamanishi, K.; Mitrevej, A.; Sinchaipanid, N. Chitosan nanoparticle encapsulated hemagglutinin-split influenza virus mucosal vaccine. AAPS PharmSciTech 2014, 15, 317–325. [Google Scholar] [CrossRef] [Green Version]
- Tatlow, D.; Tatlow, C.; Tatlow, S.; Tatlow, S. A novel concept for treatment and vaccination against Covid-19 with an inhaled chitosan-coated DNA vaccine encoding a secreted spike protein portion. Clin. Exp. Pharmacol. Physiol. 2020, 47, 1874–1878. [Google Scholar] [CrossRef]
- Dabaghian, M.; Latifi, A.M.; Tebianian, M.; NajmiNejad, H.; Ebrahimi, S.M. Nasal vaccination with r4M2e.HSP70c antigen encapsulated into N-trimethyl chitosan (TMC) nanoparticulate systems: Preparation and immunogenicity in a mouse model. Vaccine 2018, 36, 2886–2895. [Google Scholar] [CrossRef]
- Lynn, G.M.; Laga, R.; Darrah, P.A.; Ishizuka, A.S.; Balaci, A.J.; Dulcey, A.E.; Pechar, M.; Pola, R.; Gerner, M.Y.; Yamamoto, A.; et al. In vivo characterization of the physicochemical properties of polymer-linked TLR agonists that enhance vaccine immunogenicity. Nat. Biotechnol. 2015, 33, 1201–1210. [Google Scholar] [CrossRef]
- Roux, X.; Dubuquoy, C.; Durand, G.; Tran-Tolla, T.-L.; Castagné, N.; Bernard, J.; Petit-Camurdan, A.; Eléouët, J.-F.; Riffault, S. Sub-nucleocapsid nanoparticles: A nasal vaccine against Respiratory Syncytial Virus. PLoS ONE 2008, 3, e1766. [Google Scholar] [CrossRef] [Green Version]
- Hervé, P.-L.; Deloizy, C.; Descamps, D.; Rameix-Welti, M.-A.; Fix, J.; McLellan, J.S.; Eléouët, J.-F.; Riffault, S. RSV N-nanorings fused to palivizumab-targeted neutralizing epitope as a nanoparticle RSV vaccine. Nanomedicine 2017, 13, 411–420. [Google Scholar] [CrossRef] [Green Version]
- Hervé, P.-L.; Raliou, M.; Bourdieu, C.; Dubuquoy, C.; Petit-Camurdan, A.; Bertho, N.; Eléouët, J.-F.; Chevalier, C.; Riffault, S. A novel subnucleocapsid nanoplatform for mucosal vaccination against influenza virus that targets the ectodomain of matrix protein 2. J. Virol. 2014, 88, 325–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, M.; Zhang, X.-E.; Sun, X.; Zhang, X.; Yao, Y.; Liu, S.; Chen, Z.; Li, W.; Zhang, Z.; Chen, J.; et al. Intranasal nanovaccine confers homo- and hetero-subtypic influenza protection. Small 2018, 14, 1703207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Chao, C.W.; Tsybovsky, Y.; Abiona, O.M.; Hutchinson, G.B.; Moliva, J.I.; Olia, A.S.; Pegu, A.; Phung, E.; Stewart-Jones, G.B.E.; et al. A platform incorporating trimeric antigens into self-assembling nanoparticles reveals SARS-CoV-2-spike nanoparticles to elicit substantially higher neutralizing responses than spike alone. Sci. Rep. 2020, 10, 18149. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.-S.; Huang, C.; Compans, R.W.; Kang, S.-M. Virus-like particle vaccine induces protective immunity against homologous and heterologous strains of influenza virus. J. Virol. 2007, 81, 3514–3524. [Google Scholar] [CrossRef] [Green Version]
- Kim, A.R.; Lee, D.H.; Lee, S.H.; Rubino, I.; Choi, H.J.; Quan, F.S. Protection induced by virus-like particle vaccine containing tandem repeat gene of respiratory syncytial virus G protein. PLoS ONE 2018, 13, e0191277. [Google Scholar] [CrossRef] [Green Version]
- Coulter, A.; Harris, R.; Davis, R.; Drane, D.; Cox, J.; Ryan, D.; Sutton, P.; Rockman, S.; Pearse, M. Intranasal vaccination with ISCOMATRIX® adjuvanted influenza vaccine. Vaccine 2003, 21, 946–949. [Google Scholar] [CrossRef]
- Wee, J.L.K.; Scheerlinck, J.P.Y.; Snibson, K.J.; Edwards, S.; Pearse, M.; Quinn, C.; Sutton, P. Pulmonary delivery of ISCOMATRIX influenza vaccine induces both systemic and mucosal immunity with antigen dose sparing. Mucosal Immunol. 2008, 1, 489–496. [Google Scholar] [CrossRef] [Green Version]
- Tai, W.; Roberts, L.; Seryshev, A.; Gubatan, J.M.; Bland, C.S.; Zabriskie, R.; Kulkarni, S.; Soong, L.; Mbawuike, I.; Gilbert, B.; et al. Multistrain influenza protection induced by a nanoparticulate mucosal immunotherapeutic. Mucosal Immunol. 2011, 4, 197–207. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.W.; Lee, J.H.; Hung, C.F.; Peng, S.; Roden, R.; Wang, M.C.; Viscidi, R.; Tsai, Y.C.; He, L.; Chen, P.J.; et al. Generation and characterization of DNA vaccines targeting the nucleocapsid protein of severe acute respiratory syndrome coronavirus. J. Virol. 2004, 78, 4638–4645. [Google Scholar] [CrossRef] [Green Version]
- Carroll, T.D.; Jegaskanda, S.; Matzinger, S.R.; Fritts, L.; McChesney, M.B.; Kent, S.J.; Fairman, J.; Miller, C.J. A Lipid/DNA adjuvant-inactivated influenza virus vaccine protects Rhesus Macaques from uncontrolled virus replication after heterosubtypic influenza A virus challenge. J. Infect. Dis 2018, 218, 856–867. [Google Scholar] [CrossRef]
- Reis, C.P.; Neufeld, R.J.; Ribeiro, A.J.; Veiga, F.; Nanoencapsulation, I. Methods for preparation of drug-loaded polymeric nanoparticles. Nanomedicine 2006, 2, 8–21. [Google Scholar] [CrossRef] [Green Version]
- Kumari, A.; Singla, R.; Guliani, A.; Yadav, S.K. Nanoencapsulation for drug delivery. EXCLI J. 2014, 13, 265–286. [Google Scholar]
- Dhand, C.; Dwivedi, N.; Loh, X.J.; Ying, A.N.J.; Verma, N.K.; Beuerman, R.W.; Lakshminarayanan, R.; Ramakrishna, S. Methods and strategies for the synthesis of diverse nanoparticles and their applications: A comprehensive overview. RSC Adv. 2015, 5, 105003–105037. [Google Scholar] [CrossRef]
- Ahmadi, M.; Madrakian, T.; Afkhami, A. Smart nanogels in cancer therapy. In Smart Nanocontainers; Elsevier: Amsterdam, The Netherlands, 2020; pp. 179–193. [Google Scholar]
- Ahmadi, M.; Madrakian, T.; Ghavami, S. Preparation and Characterization of Simvastatin Nanocapsules: Encapsulation of Hydrophobic Drugs in Calcium Alginate. Methods Mol. Biol. 2020, 2125, 47–56. [Google Scholar]
- Beck-Broichsitter, M.; Gauss, J.; Gessler, T.; Seeger, W.; Kissel, T.; Schmehl, T. Pulmonary targeting with biodegradable salbutamol-loaded nanoparticles. J. Aerosol Med. Pulm. Drug Deliv. 2010, 23, 47–57. [Google Scholar] [CrossRef]
- Park, H.; Kim, S.; Kim, S.; Song, Y.; Seung, K.; Hong, D.; Khang, G.; Lee, D. Antioxidant and anti-inflammatory activities of hydroxybenzyl alcohol releasing biodegradable polyoxalate nanoparticles. Biomacromolecules 2010, 11, 2103–2108. [Google Scholar] [CrossRef]
- Kim, S.; Park, H.; Song, Y.; Hong, D.; Kim, O.; Jo, E.; Khang, G.; Lee, D. Reduction of oxidative stress by p-hydroxybenzyl alcohol-containing biodegradable polyoxalate nanoparticulate antioxidant. Biomaterials 2011, 32, 3021–3029. [Google Scholar] [CrossRef] [PubMed]
- Serisier, D.J.; Bilton, D.; De Soyza, A.; Thompson, P.J.; Kolbe, J.; Greville, H.W.; Cipolla, D.; Bruinenberg, P.; Gonda, I. Inhaled, dual release liposomal ciprofloxacin in non-cystic fibrosis bronchiectasis (ORBIT-2): A randomised, double-blind, placebo-controlled trial. Thorax 2013, 68, 812–817. [Google Scholar] [CrossRef] [Green Version]
- Alhajlan, M.; Alhariri, M.; Omri, A. Efficacy and safety of liposomal clarithromycin and its effect on Pseudomonas aeruginosa virulence factors. Antimicrob. Agents Chemother. 2013, 57, 2694–2704. [Google Scholar] [CrossRef] [Green Version]
- Silva, L.F.C.; Kasten, G.; de Campos, C.E.M.; Chinelatto, A.L.; Lemos-Senna, E. Preparation and characterization of quercetin-loaded solid lipid microparticles for pulmonary delivery. Powder Technol. 2013, 239, 183–192. [Google Scholar] [CrossRef]
- Liu, C.; Shi, J.; Dai, Q.; Yin, X.; Zhang, X.; Zheng, A. In-vitro and in-vivo evaluation of ciprofloxacin liposomes for pulmonary administration. Drug Dev. Ind. Pharm. 2015, 41, 272–278. [Google Scholar] [CrossRef]
- Troy, N.M.; Bosco, A. Respiratory viral infections and host responses; insights from genomics. Respir. Res. 2016, 17, 156. [Google Scholar] [CrossRef] [Green Version]
- Mehta, M.; Tewari, D.; Gupta, G.; Awasthi, R.; Singh, H.; Pandey, P.; Chellappan, D.K.; Wadhwa, R.; Collet, T.; Hansbro, P.M.; et al. Oligonucleotide therapy: An emerging focus area for drug delivery in chronic inflammatory respiratory diseases. Chem. Biol. Interact. 2019, 308, 206–215. [Google Scholar] [CrossRef]
- Ibrahim, M.; Verma, R.; Garcia-Contreras, L. Inhalation drug delivery devices: Technology update. Med. Devices (Auckl.) 2015, 8, 131–139. [Google Scholar] [PubMed] [Green Version]
- Bains, B.K.; Birchall, J.C.; Toon, R.; Taylor, G. In vitro reporter gene transfection via plasmid DNA delivered by metered dose inhaler. J. Pharm. Sci. 2010, 99, 3089–3099. [Google Scholar] [CrossRef]
- Conti, D.S.; Bharatwaj, B.; Brewer, D.; da Rocha, S.R. Propellant-based inhalers for the non-invasive delivery of genes via oral inhalation. J. Control. Release Off. J. Control. Release Soc. 2012, 157, 406–417. [Google Scholar] [CrossRef]
- Lam, J.K.-W.; Liang, W.; Chan, H.-K. Pulmonary delivery of therapeutic siRNA. Adv. Drug Deliv. Rev. 2012, 64, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kandil, R.; Merkel, O.M. Pulmonary delivery of siRNA as a novel treatment for lung diseases. Ther. Deliv. 2019, 10, 203–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, J.; Tian, Z.; Yang, X. Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci. Trends 2020, 14, 72–73. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Dong, L.; Hu, S.; Gao, J. Discovering drugs to treat coronavirus disease 2019 (COVID-19). Drug Discov. Ther. 2020, 14, 58–60. [Google Scholar] [CrossRef] [Green Version]
- Gautret, P.; Lagier, J.C.; Parola, P.; Hoang, V.T.; Meddeb, L.; Mailhe, M.; Doudier, B.; Courjon, J.; Giordanengo, V.; Vieira, V.E.; et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents 2020, 56, 105949. [Google Scholar] [CrossRef]
- Liu, C.; Zhou, Q.; Li, Y.; Garner, L.V.; Watkins, S.P.; Carter, L.J.; Smoot, J.; Gregg, A.C.; Daniels, A.D.; Jervey, S.; et al. Research and development on therapeutic aents and vaccines for COVID-19 and related human coronavirus diseases. ACS Cent. Sci. 2020, 6, 315–331. [Google Scholar] [CrossRef]
- Francica, J.R.; Lynn, G.M.; Laga, R.; Joyce, M.G.; Ruckwardt, T.J.; Morabito, K.M.; Chen, M.; Chaudhuri, R.; Zhang, B.; Sastry, M. Thermoresponsive polymer nanoparticles co-deliver RSV F trimers with a TLR-7/8 adjuvant. Bioconjug. Chem. 2016, 27, 2372–2385. [Google Scholar] [CrossRef]
- Chan, W.C. Nano Research for COVID-19; ACS Publications: Washington, DC, USA, 2020. [Google Scholar]
- Taylor, K.M.; Fan, S.J. Liposomes for drug delivery to the respiratory tract. Drug Dev. Ind. Pharm. 1993, 19, 123–142. [Google Scholar] [CrossRef]
- Wyde, P.R.; Six, H.R.; Wilson, S.Z.; Gilbert, B.E.; Knight, V. Activity against rhinoviruses, toxicity, and delivery in aerosol of enviroxime in liposomes. Antimicrob. Agents Chemother. 1988, 32, 890–895. [Google Scholar] [CrossRef] [Green Version]
- Serrano, G.; Kochergina, I.; Albors, A.; Diaz, E.; Oroval, M.; Hueso, G.; Serrano, J.M. Liposomal Lactoferrin as Potential Preventative and Cure for COVID-19. Int. J. Res. Health Sci. 2020, 8, 8–15. [Google Scholar]
- Hassan, A.O.; Kafai, N.M.; Dmitriev, I.P.; Fox, J.M.; Smith, B.K.; Harvey, I.B.; Chen, R.E.; Winkler, E.S.; Wessel, A.W.; Case, J.B.; et al. A Single-Dose Intranasal ChAd Vaccine Protects Upper and Lower Respiratory Tracts against SARS-CoV-2. Cell 2020, 183, 169–184.e13. [Google Scholar] [CrossRef] [PubMed]
- Ulery, B.D.; Phanse, Y.; Sinha, A.; Wannemuehler, M.J.; Narasimhan, B.; Bellaire, B.H. Polymer chemistry influences monocytic uptake of polyanhydride nanospheres. Pharm. Res. 2009, 26, 683–690. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Martinez-Paniagua, M.; Rezvan, A.; Fathi, M.; Singh, S.; Biswas, S.; Pourpak, M.; Yee, C.; Liu, X.; Varadarajan, N. Single-dose intranasal vaccination elicits systemic and mucosal immunity against SARS-CoV-2. bioRxiv 2020. [Google Scholar] [CrossRef]
- Ichinohe, T.; Ainai, A.; Tashiro, M.; Sata, T.; Hasegawa, H. PolyI:polyC12U adjuvant-combined intranasal vaccine protects mice against highly pathogenic H5N1 influenza virus variants. Vaccine 2009, 27, 6276–6279. [Google Scholar] [CrossRef]
- Lycke, N. Recent progress in mucosal vaccine development: Potential and limitations. Nat. Rev. Immunol. 2012, 12, 592–605. [Google Scholar] [CrossRef]
- Chen, L.; Wang, J.; Zganiacz, A.; Xing, Z. Single intranasal mucosal Mycobacterium bovis BCG vaccination confers improved protection compared to subcutaneous vaccination against pulmonary tuberculosis. Infect. Immun. 2004, 72, 238–246. [Google Scholar] [CrossRef] [Green Version]
- Giri, P.K.; Sable, S.B.; Verma, I.; Khuller, G.K. Comparative evaluation of intranasal and subcutaneous route of immunization for development of mucosal vaccine against experimental tuberculosis. FEMS Immunol. Med. Microbiol. 2005, 45, 87–93. [Google Scholar] [CrossRef] [Green Version]
- Mapletoft, J.W.; Latimer, L.; Babiuk, L.A. Intranasal immunization of mice with a bovine respiratory syncytial virus vaccine induces superior immunity and protection compared to those by subcutaneous delivery or combinations of intranasal and subcutaneous prime-boost strategies. Clin. Vaccine Immunol. 2010, 17, 23–35. [Google Scholar] [CrossRef] [Green Version]
- Kharb, S.; Charan, S. Mucosal immunization provides better protection than subcutaneous immunization against Pasteurella multocida (B: 2) in mice preimmunized with the outer membrane proteins. Vet. Res. Commun. 2011, 35, 457–461. [Google Scholar] [CrossRef]
- McCormick, A.A.; Shakeel, A.; Yi, C.; Kaur, H.; Mansour, A.M.; Bakshi, C.S. Intranasal administration of a two-dose adjuvanted multi-antigen TMV-subunit conjugate vaccine fully protects mice against Francisella tularensis LVS challenge. PLoS ONE 2018, 13, e0194614. [Google Scholar] [CrossRef]
- Shao, K.; Singha, S.; Clemente-Casares, X.; Tsai, S.; Yang, Y.; Santamaria, P. Nanoparticle-based immunotherapy for cancer. ACS Nano 2015, 9, 16–30. [Google Scholar] [CrossRef]
- Zilker, C.; Kozlova, D.; Sokolova, V.; Yan, H.; Epple, M.; Überla, K.; Temchura, V. Nanoparticle-based B-cell targeting vaccines: Tailoring of humoral immune responses by functionalization with different TLR-ligands. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Wang, Y.; Zhang, C.; Wang, X.; Wang, H.; Wang, J.; Yuan, Y.; Wang, X.; Zhang, X.; Yu, C. Antigen-Directed Fabrication of a Multifunctional Nanovaccine with Ultrahigh Antigen Loading Efficiency for Tumor Photothermal-Immunotherapy. Adv. Mater. 2018, 30, 1704408. [Google Scholar] [CrossRef]
- Yang, P.; Wang, X. COVID-19: A new challenge for human beings. Cell Mol. Immunol. 2020, 17, 555–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Novavax Press. Novavax Press Release on 2/26/20. 2020. Available online: http://ir.novavax.com/news-releases/news-release-details/novavax-advances-developmentnovel-covid-19-vaccine (accessed on 27 June 2021).
- Le, T.T.; Andreadakis, Z.; Kumar, A.; Román, R.G.; Tollefsen, S.; Saville, M.; Mayhew, S. The COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020, 19, 305–306. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, Q.; Guo, D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J. Med. Virol. 2020, 92, 418–423. [Google Scholar] [CrossRef]
| Virus | VARID | Ref |
|---|---|---|
| Adenoviruses | Common Cold, Pneumonia | [15] |
| Coronaviruses | Common Cold, SARS, MERS, COVID-19 | [6] |
| Enteroviruses | Common Cold | [16] |
| Influenza Virus (Types A and B) | Influenza, Pneumonia | [17] |
| Metapneumovirus | Common Cold, Pneumonia, Bronchiolitis | [18] |
| Parainfluenza Virus (Type 3) | Common Cold, Croup, Pneumonia, Bronchiolitis | [19] |
| Parainfluenza Viruses (Types 1, 2) | Croup | [19] |
| Respiratory Syncytial viruses | Pneumonia, Bronchiolitis | [20] |
| Rhinoviruses | Common Cold | [21] |
| Nanoparticle | Size (nm) | Virus | Antigen/Epitope | Adjuvant | Status | Route of Administration | Ref |
|---|---|---|---|---|---|---|---|
| INORGANIC NANOPARTICLES | |||||||
| Gold | 12 Influenza | M2e | CpG | Preclinical | IN | [88] | |
| 173 ± 2.4 | SARS-COV 2 | S | LPS, P:IC | Preclinical | SC | [145] | |
| POLYMERIC NANOPARTICLES | |||||||
| PLGA | 225.4 | Bovine parainfluenza 3 virus | BPI3V proteins | - | Preclinical | IN | [121] |
| 200–300 | Swine influenza virus (H1N2) | Inactivated virus H1N2 antigen | - | Preclinical | IN | ||
| γ-PGA (Poly-glutamic acid) | 100–200 | Influenza (H1N1) | HA | - | Preclinical | IN | [123] |
| Chitosan | 140 | Influenza (H1N1) | H1N1 antigen | - | Preclinical | IN | [125] |
| 300–350 | Influenza (H1N1) | HA-Split | - | Preclinical | IM | [162] | |
| 571.7 | Swine influenza virus (H1N2) | Killed swine influenza antigen | - | Preclinical | IN | [124] | |
| 200–250 | Influenza (H1N1) | M2e | Heat shock protein 70C | Preclinical | IN | [164] | |
| 125 | SARS-COV2 | S | - | Preclinical | IN | [163] | |
| HPMA/NIPAM | 12–25 | RSV | F protein | TLR-7/8 agonist | Preclinical | IN | [165] |
| Polyanhydride | 200–800 | RSV | F and G glycoproteins | - | Preclinical | IM | [126] |
| SELF-ASSEMBLING PROTEINS AND PEPTIDE-BASED NANOPARTICLES | |||||||
| N nucleocapside protein of RSV | 15 | RSV | RSV phosphoprotein | R192G | Preclinical | IM | [166] |
| 15 | RSV | FsII | MontanideTM Gel 01 | Phase I | IM | [167] | |
| 15 | Influenza (H1N1) | M2e | MontanideTM Gel 01 | Phase I,II | IN | [168] | |
| Ferritin | 12.5 | Influenza (H1N1) | M2e | - | PhaseII | IN | [169] |
| Q11 | - | Influenza (H1N1) | Acid polymerase | - | PhaseI,II | IN | [169] |
| S2G-HR2-RBD | SARS-COV 2 | RBD-S | PhaseII | IM | [151] | ||
| RBD-153-50 | 50.67±0.11 | SARS-COV 2 | RBD | PhaseII | IM | [152] | |
| LuS *-S-F ** | 50 | SARS-COV 2 | S-LuS-F | SAS *** | PhaseII | IN | [170] |
| OTHERS | |||||||
| VLP | 80–120 | Influenza (H1N1) | HA | - | Preclinical | IM | [171] |
| 80–120 | Influenza (H1N1, H3N2, H5N1 | M2e | - | Preclinical | IM | [159] | |
| 80–120 | RSV | F protein and G glycoprotein of RSV and M1 protein of Influenza | - | Preclinical | IM | [172] | |
| 100 | SARS-COV 2 | M-N-S-E | - | Preclinical | IN | [158] | |
| 100 | SARS-COV 2 | M-S-E | - | Preclinical | IM | [160] | |
| ISCOM (Quillaia saponin, cholesterol, phospholipid, and associated antigen) | 40 | Influenza (H1N1) | HA | ISCOMATRIX | Preclinical | IN | [173,174] |
| DLPC liposomes (Dilauroylphosphatidylcholine) | 30–100 | Influenza (H1N1) | M2, HA, NP | MPL and trehalose 6,6′ dimycolate | Preclinical | IN | [175] |
| Surface-linked liposomal peptide | - | Vaccinia virus | SARS-CoV N epitopes | - | Preclinical | IM | [176] |
| Cationic lipid/DNA complex | - | Influenza (H1N1) | whole inactivated IAV vaccine (H1N1, H3N2) | cationic lipid/DNA complex | Preclinical | IM | [177] |
| Type of Nanoparticle | Main Material | Size (nm) | Target Respiratory Virus | Antigen/Epitope | Ref. |
|---|---|---|---|---|---|
| Polymeric | PLGA | 225 | Bovine parainfluenza 3 virus (BPI3V) | BPI3V proteins | [121] |
| PLGA | 200–300 | Swine influenza virus (H1N2) | Inactivated virus H1N2 antigen | [122] | |
| γ-PGA | 100–200 | Influenza (H1N1) | Hemagglutinin | [123] | |
| Chitosan | 140 | Influenza (H1N1) | H1N1 antigen | [125] | |
| Chitosan | 300–350 | Influenza (H1N1) | HA-Split | [162] | |
| Chitosan | 572 | Swine influenza virus (H1N2) | Killed swine influenza antigen | [124] | |
| Chitosan | 200–250 | Influenza (H1N1) | M2e peptide | [164] | |
| HPMA/NIPAM | 12–25 | RSV | F protein | [165] | |
| PEG | 40–500 | RSV | F protein | [202] | |
| SA-CPH copolymer | 348–397 | RSV | Eα peptide | [208] | |
| CPH-CPTEG copolymer | - | RSV | F and G glycoproteins | [126] | |
| Self-assembled proteins and peptides (SANP) | Nucleocapsid (N) protein of RSV | 15 | RSV | RSV phosphoprotein | [167] |
| Nucleocapsid (N) protein of RSV | 15 | RSV | FsII epitope | [167] | |
| Nucleocapsid (N) protein of RSV | 15 | Influenza (H1N1) | M2e peptide | [168] | |
| Ferritin | 12.5 | Influenza (H1N1) | M2e peptide | [169] | |
| Influenza acid polymerase and the Q11 self-assembly domain | - | Influenza (H1N1) | Acid polymerase | [176] | |
| Inorganic | gold | 12 | Influenza (H1N1, H3N2, H5N1) | M2e peptide | [88] |
| VLP | - | - | Influenza (H1N1) | Hemagglutinin | [143] |
| - | 80–120 | Influenza (H1N1, H3N2, H5N1) | M2e5x peptide | [159] | |
| - | 60–80 | RSV | F protein et G glycoprotein of RSV and M1 protein of Influenza | [171] | |
| Liposome | DLPC | 30–100 | Influenza (H1N1) | M2, HA, NP | [175] |
| Liposome, Polymer | 10:1:1:1 of DPPC, DPPG, Cholesterol (Chol), and DPPE-PEG2000 | 89 | SARS-COV 2 | S+ STING agonist | [209] |
| LNP | ChAdenovirus (S) | - | SARS-COV 2 | ChAd-S | [207] |
| Candidate Vaccine | Characteristics | Nano-Composition | Developer-Country | Status |
|---|---|---|---|---|
| mRNA-1273 | mRNA vaccine encoding S protein | SM-102, PEG2000, Tromethamine, | Moderna/USA | FDA- EMA Approved |
| BNT162b2 | mRNA vaccine encoding S protein | ALC-0315, ALC-0159, 1,2-distearoyl-sn-glycero-3-phosphocholine | Pfizer-BioNtech/USA-Germany | FDA-EMA Approved |
| Ad5-nCoV | Adenovirus type 5 vector that expresses S protein | LNP | CanSino Biologicals/China | China-Approved |
| AZD1222 (Covishield) | ChAdOx1-S | AstraZenca/UK-Sweden | FDA-EMA Approved | |
| Ad26.COV2. S | Adenovirus type 26 vector that expresses S protein | Johnson & Johnson (Janssen) | FDA-Approved | |
| INO-4800 | DNA plasmid encoding S protein delivered by electroporation | Inovio Pharmaceuticals | Phase I (NCT04336410) | |
| LV-SMENP-DC | DCs modified with a lentiviral vector expressing synthetic minigene based on domains of selected viral proteins; administered with antigen specific CTLs | Shenzhen Geno-Immune Medical Institute | Phase I (NCT04276896) | |
| Pathogen-specific aAPC | aAPCs modified with a lentiviral vector expressing synthetic minigene based on domains of selected viral proteins | Shenzhen Geno-Immune Medical Institute | Phase I (NCT04299724) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seyfoori, A.; Shokrollahi Barough, M.; Mokarram, P.; Ahmadi, M.; Mehrbod, P.; Sheidary, A.; Madrakian, T.; Kiumarsi, M.; Walsh, T.; McAlinden, K.D.; et al. Emerging Advances of Nanotechnology in Drug and Vaccine Delivery against Viral Associated Respiratory Infectious Diseases (VARID). Int. J. Mol. Sci. 2021, 22, 6937. https://doi.org/10.3390/ijms22136937
Seyfoori A, Shokrollahi Barough M, Mokarram P, Ahmadi M, Mehrbod P, Sheidary A, Madrakian T, Kiumarsi M, Walsh T, McAlinden KD, et al. Emerging Advances of Nanotechnology in Drug and Vaccine Delivery against Viral Associated Respiratory Infectious Diseases (VARID). International Journal of Molecular Sciences. 2021; 22(13):6937. https://doi.org/10.3390/ijms22136937
Chicago/Turabian StyleSeyfoori, Amir, Mahdieh Shokrollahi Barough, Pooneh Mokarram, Mazaher Ahmadi, Parvaneh Mehrbod, Alireza Sheidary, Tayyebeh Madrakian, Mohammad Kiumarsi, Tavia Walsh, Kielan D. McAlinden, and et al. 2021. "Emerging Advances of Nanotechnology in Drug and Vaccine Delivery against Viral Associated Respiratory Infectious Diseases (VARID)" International Journal of Molecular Sciences 22, no. 13: 6937. https://doi.org/10.3390/ijms22136937









