Combinatorial Treatment of Tinzaparin and Chemotherapy Can Induce a Significant Antitumor Effect in Pancreatic Cancer
Abstract
1. Introduction
2. Results
2.1. Tinzaparin Enhances the Anti-Tumor Effect of Nab-Paclitaxel and Gemcitabine in mtKRAS PC Cell Lines
2.2. Stimulation of Apoptotic Pathways Results to Decreased Cell Survival
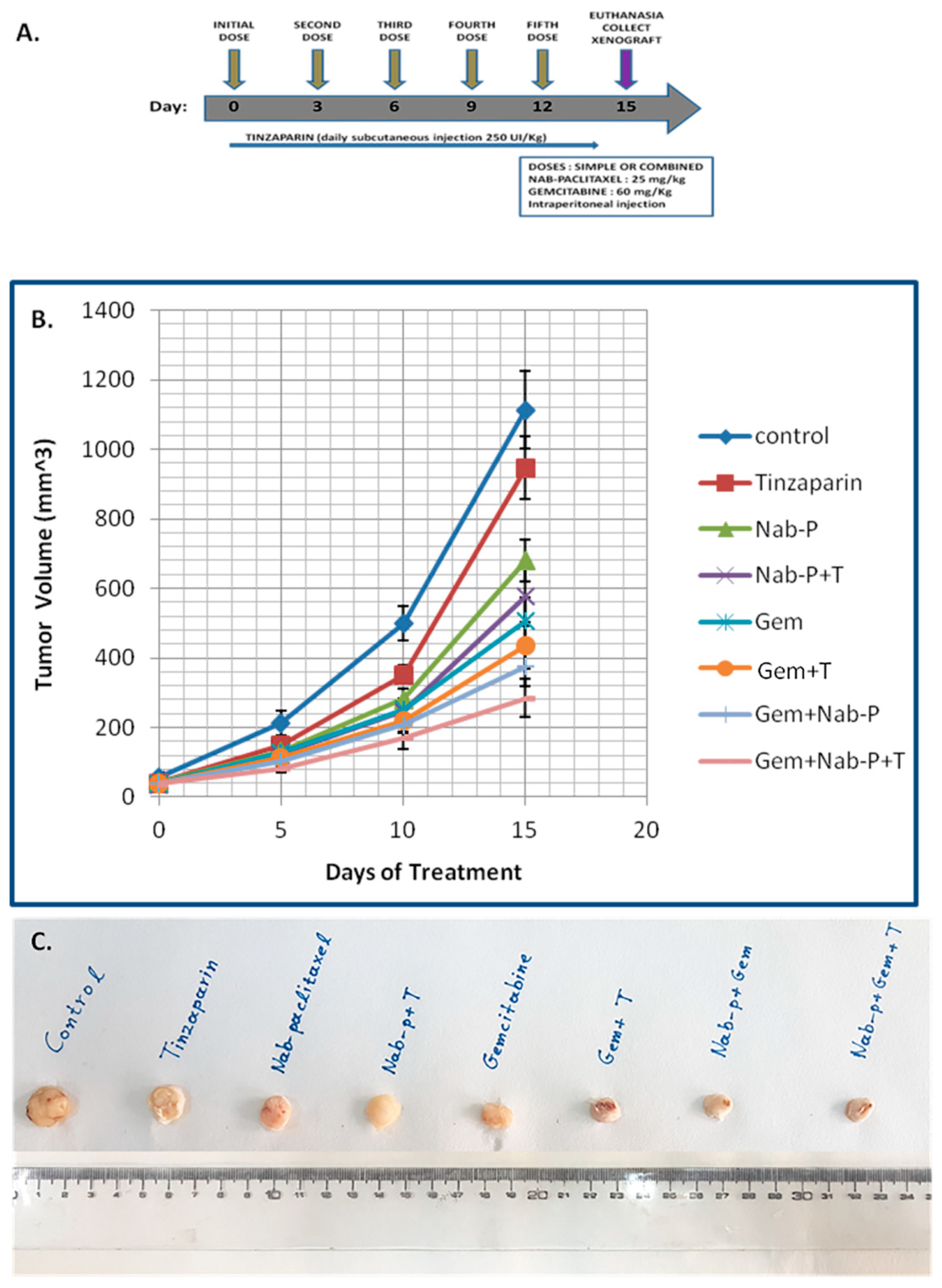
2.3. In Vivo Experiments with N0D/SCID Mouse Model Injected Heterotopically with Pancreatic Cancer Cells, Shows That Tinzaparin Administration Acts Synergistically with Chemotherapeutic Drugs and Provides a Significant Tumor Reduction
2.4. Immunofluorescence with PCNA Shows That the Triple Combination Has Significant Reduction in Cell Proliferation in Mouse Xenografts
2.5. Stimulation of Apoptotic Pathways Is One of the Mechanisms Leading to Tumor Reduction In Vivo
2.6. Triple Drug Combination Negatively Affects Tumor Neoangiogenesis
3. Discussion
4. Materials and Methods
4.1. Cell Lines
4.2. Cell Viability—XTT Assay
4.3. Western Blotting
4.4. In Vivo Experiments
4.5. Immunohistochemistry
4.6. Immunofluorescence
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Pourshams, A.; Sepanlou, S.G.; Ikuta, K.S.; Bisignano, C.; Safiri, S.; Roshandel, G.; Sharif, M.; Khatibian, M.; Fitzmaurice, C.; Nixon, M.R.; et al. The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2019, 4, 934–947. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Gaduputi, V. Epidemiology of Pancreatic Cancer: Global Trends, Etiology and Risk Factors. World J. Oncol. 2019, 10, 10–27. [Google Scholar] [CrossRef]
- Artinyan, A.; Soriano, P.A.; Prendergast, C.; Low, T.; Ellenhorn, J.D.; Kim, J. The anatomic location of pancreatic cancer is a prognostic factor for survival. HPB 2008, 10, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Labori, K.J.; Katz, M.H.; Tzeng, C.W.; Bjørnbeth, B.A.; Cvancarova, M.; Edwin, B.; Kure, E.H.; Eide, T.J.; Dueland, S.; Buanes, T.; et al. Impact of early disease progression and surgical complications on adjuvant chemotherapy completion rates and survival in patients undergoing the surgery first approach for resectable pancreatic ductal adenocarcinoma—A population-based cohort study. Acta Oncol. 2015, 55, 265–277. [Google Scholar] [CrossRef]
- Huang, L.; Balavarca, Y.; Van Der Geest, L.; Lemmens, V.; Van Eycken, L.; De Schutter, H.; Johannesen, T.B.; Zadnik, V.; Primic-Žakelj, M.; Mägi, M.; et al. Development and validation of a prognostic model to predict the prognosis of patients who underwent chemotherapy and resection of pancreatic adenocarcinoma: A large international population-based cohort study. BMC Med. 2019, 17, 66. [Google Scholar] [CrossRef] [PubMed]
- Bates, S.M.; Middeldorp, S.; Rodger, M.; James, A.H.; Greer, I. Guidance for the treatment and prevention of obstetric-associated venous thromboembolism. J. Thromb. Thrombolysis 2016, 41, 92–128. [Google Scholar] [CrossRef]
- Wong, A.; Soo, R.A.; Yong, W.P.; Innocenti, F. Clinical pharmacology and pharmacogenetics of gemcitabine. Drug Metab. Rev. 2009, 41, 77–88. [Google Scholar] [CrossRef]
- Vaccaro, V.; Bria, E.; Sperduti, I.; Gelibter, A.; Moscetti, L.; Mansueto, G.; Ruggeri, E.M.; Gamucci, T.; Cognetti, F.; Milella, M. First-line erlotinib and fixed dose-rate gemcitabine for advanced pancreatic cancer. World J. Gastroenterol. 2013, 19, 4511–4519. [Google Scholar] [CrossRef]
- Al-Hajeili, M.; Azmi, A.S.; Choi, M. Nab-paclitaxel: Potential for the treatment of advanced pancreatic cancer. OncoTargets Ther. 2014, 7, 187–192. [Google Scholar] [CrossRef][Green Version]
- Hoogenboezem, E.N.; Duvall, C.L. Harnessing albumin as a carrier for cancer therapies. Adv. Drug Deliv. Rev. 2018, 130, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Park, C.R.; Jo, J.H.; Song, M.G.; Park, J.Y.; Kim, Y.-H.; Youn, H.; Paek, S.H.; Chung, J.-K.; Jeong, J.M.; Lee, Y.-S.; et al. Secreted protein acidic and rich in cysteine mediates active targeting of human serum albumin in U87MG xenograft mouse models. Theranostics 2019, 9, 7447–7457. [Google Scholar] [CrossRef] [PubMed]
- Sinn, M.; Sinn, B.V.; Striefler, J.K.; Lindner, J.L.; Stieler, J.M.; Lohneis, P.; Bischoff, S.; Bläker, H.; Pelzer, U.; Bahra, M.; et al. SPARC expression in resected pancreatic cancer patients treated with gemcitabine: Results from the CONKO-001 study. Ann. Oncol. 2014, 25, 1025–1032. [Google Scholar] [CrossRef] [PubMed]
- Vishnu, P.; Roy, V. Safety and Efficacy of nab-Paclitaxel in the Treatment of Patients with Breast Cancer. Breast Cancer Basic Clin. Res. 2011, 5, 53–65. [Google Scholar] [CrossRef]
- Dimakakos, E.P.; Vathiotis, I.; Syrigos, K. The Role of Tinzaparin in Oncology. Clin. Appl. Thromb. 2017, 24, 697–707. [Google Scholar] [CrossRef] [PubMed]
- Hemker, H.C.; Al Dieri, R.; Béguin, S. Heparins: A Shift of Paradigm. Front. Med. 2019, 6, 254. [Google Scholar] [CrossRef]
- Dolovich, L.R.; Ginsberg, J.S.; Douketis, J.D.; Holbrook, A.M.; Cheah, G. A Meta-analysis Comparing Low-Molecular-Weight Heparins with Unfractionated Heparin in the Treatment of Venous Thromboembolism: Examining Some Unanswered Questions Regarding Location of Treatment, Product Type and Dosing Frequency. Arch. Intern. Med. 2000, 160, 181–188. [Google Scholar] [CrossRef]
- Bokas, A.; Papakotoulas, P.; Sarantis, P.; Papadimitropoulou, A.; Papavassiliou, A.G.; Karamouzis, M.V. Mechanisms of the Antitumor Activity of Low Molecular Weight Heparins in Pancreatic Adenocarcinomas. Cancers 2020, 12, 432. [Google Scholar] [CrossRef]
- Streiff, M.B.; Agnelli, G.; Connors, J.M.; Crowther, M.; Eichinger, S.; Lopes, R.; McBane, R.D.; Moll, S.; Ansell, J. Guidance for the treatment of deep vein thrombosis and pulmonary embolism. J. Thromb. Thrombolysis 2016, 41, 32–67. [Google Scholar] [CrossRef]
- Petersen, L.J.; Mousa, S.A. Anti-cancer properties of low-molecular-weight heparin: Preclinical evidence. Thromb. Haemost. 2009, 102, 258–267. [Google Scholar] [CrossRef]
- Abdol Razak, N.B.; Jones, G.; Bhandari, M.; Berndt, M.C.; Metharom, P. Cancer-associated thrombosis: An overview of mechanisms, risk factors, and treatment. Cancers 2018, 10, 380. [Google Scholar] [CrossRef] [PubMed]
- Khalil, J.; Bensaid, B.; Elkacemi, H.; Afif, M.; Bensaid, Y.; Kebdani, T.; Benjaafar, N. Venous thromboembolism in cancer patients: An underestimated major health problem. World J. Surg. Oncol. 2015, 13, 204. [Google Scholar] [CrossRef]
- Mousa, S.A. Low-Molecular-Weight Heparins in Thrombosis and Cancer: Emerging Links. Cardiovasc. Drug Rev. 2006, 22, 121–134. [Google Scholar] [CrossRef]
- Bobek, V.; Kovařík, J. Antitumor and antimetastatic effect of warfarin and heparins. Biomed. Pharmacother. 2004, 58, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Borsig, L. Heparin as an inhibitor of cancer progression. Prog. Mol. Biol. Transl. Sci. 2010, 93, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.; Herman, J.; Schulick, R.; Hruban, R.H.; Goggins, M. Pancreatic cancer. Lancet. 2011, 378, 607–620. [Google Scholar] [CrossRef]
- Waters, A.M.; Der, C.J. KRAS: The Critical Driver and Therapeutic Target for Pancreatic Cancer. Cold Spring Harb. Perspect. Med. 2018, 8, a031435. [Google Scholar] [CrossRef]
- Witkiewicz, A.K.; McMillan, E.A.; Balaji, U.; Baek, G.H.; Lin, W.C.; Mansour, J.C.; Mollaee, M.; Wagner, K.U.; Koduru, P.; Yopp, A.C.; et al. Whole-exome sequencing of pancreatic cancer defines genetic diversity and therapeutic targets. Nat. Commun. 2015, 6, 6744. [Google Scholar] [CrossRef]
- Rucki, A.A.; Foley, K.; Zhang, P.; Xiao, Q.; Kleponis, J.; Wu, A.A.; Sharma, R.; Mo, G.; Liu, A.; Van Eyk, J.; et al. Heterogeneous Stromal Signaling within the Tumor Microenvironment Controls the Metastasis of Pancreatic Cancer. Cancer Res. 2017, 77, 41–52. [Google Scholar] [CrossRef]
- Uzunparmak, B.; Sahin, I.H. Pancreatic cancer microenvironment: A current dilemma. Clin. Transl. Med. 2019, 8, 2. [Google Scholar] [CrossRef]
- Koustas, E.; Sarantis, P.; Kyriakopoulou, G.; Papavassiliou, A.G.; Karamouzis, M.V. The Interplay of Autophagy and Tumor Microenvironment in Colorectal Cancer—Ways of Enhancing Immunotherapy Action. Cancers 2019, 11, 533. [Google Scholar] [CrossRef] [PubMed]
- Son, B.; Lee, S.; Youn, H.; Kim, E.; Kim, W.; Youn, B. The role of tumor microenvironment in therapeutic resistance. Oncotarget 2017, 8, 3933–3945. [Google Scholar] [CrossRef]
- Vennin, C.; Murphy, K.J.; Morton, J.P.; Cox, T.R.; Pajic, M.; Timpson, P. Reshaping the Tumor Stroma for Treatment of Pancreatic Cancer. Gastroenterology 2018, 154, 820–838. [Google Scholar] [CrossRef] [PubMed]
- Sarantis, P.; Koustas, E.; Papadimitropoulou, A.; Papavassiliou, A.G.; Karamouzis, M.V. Pancreatic ductal adenocarcinoma: Treatment hurdles, tumor microenvironment and immunotherapy. World J. Gastrointest. Oncol. 2020, 12, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Deer, E.L.; González-Hernández, J.; Coursen, J.D.; Shea, J.E.; Ngatia, J.; Scaife, C.L.; Firpo, M.A.; Mulvihill, S.J. Phenotype and Genotype of Pancreatic Cancer Cell Lines. Pancreas 2010, 39, 425–435. [Google Scholar] [CrossRef]
- Awasthi, N.; Zhang, C.; Schwarz, A.M.; Hinz, S.; Wang, C.; Williams, N.S.; Schwarz, M.A.; Schwarz, R.E. Comparative benefits of Nab-paclitaxel over gemcitabine or polysorbate-based docetaxel in experimental pancreatic cancer. Carcinogenesis 2013, 34, 2361–2369. [Google Scholar] [CrossRef]
- Van Wijk, X.M.R.; Van Kuppevelt, T.H. Heparan sulfate in angiogenesis: A target for therapy. Angiogenesis 2013, 17, 443–462. [Google Scholar] [CrossRef] [PubMed]
- Castelli, R.; Porro, F.; Tarsia, P. The heparins and cancer: Review of clinical trials and biological properties. Vasc. Med. 2004, 9, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Niu, G.; Chen, X. Vascular Endothelial Growth Factor as an Anti-Angiogenic Target for Cancer Therapy. Curr. Drug Targets 2010, 11, 1000–1017. [Google Scholar] [CrossRef]
- Ceci, C.; Atzori, M.G.; Lacal, P.M.; Graziani, G. Role of VEGFs/VEGFR-1 Signaling and Its Inhibition in Modulating Tumor Invasion: Experimental Evidence in Different Metastatic Cancer Models. Int. J. Mol. Sci. 2020, 21, 1388. [Google Scholar] [CrossRef]
- Panicot-Dubois, L.; Mezouar, S.; Plantureux, L.; Crescence, L.; Frere, C.; Dubois, C. PO-34—Optimal doses of tinzaparin to reduce both cancer-associated thrombosis and tumor growth in a mouse model of ectopic pancreatic syngeneic tumor. Thromb. Res. 2016, 140, S189. [Google Scholar] [CrossRef]
- Alyahya, R.; Sudha, T.; Racz, M.; Stain, S.C.; Mousa, S.A. Anti-metastasis efficacy and safety of non-anticoagulant heparin derivative versus low molecular weight heparin in surgical pancreatic cancer models. Int. J. Oncol. 2014, 46, 1225–1231. [Google Scholar] [CrossRef]
- Harvey, J.R.; Mellor, P.; Eldaly, H.; Lennard, T.W.; Kirby, J.A.; Ali, S. Inhibition of CXCR4-Mediated Breast Cancer Metastasis: A Potential Role for Heparinoids? Clin. Cancer Res. 2007, 13, 1562–1570. [Google Scholar] [CrossRef] [PubMed]
- Amirkhosravi, A.; Mousa, S.A.; Amaya, M.; Francis, J.L. Antimetastatic effect of tinzaparin, a low-molecular-weight heparin. J. Thromb. Haemost. 2003, 1, 1972–1976. [Google Scholar] [CrossRef] [PubMed]
- Meyer, G.; Besse, B.; Doubre, H.; Charles-Nelson, A.; Aquilanti, S.; Izadifar, A.; Azarian, R.; Monnet, I.; Lamour, C.; Descourt, R.; et al. Anti-tumour effect of low molecular weight heparin in localised lung cancer: A phase III clinical trial. Eur. Respir. J. 2018, 52, 1801220. [Google Scholar] [CrossRef]
- Longley, D.B.; Harkin, D.P.; Johnston, P.G. 5-Fluorouracil: Mechanisms of action and clinical strategies. Nat. Rev. Cancer 2003, 3, 330–338. [Google Scholar] [CrossRef]
- Faivre, S.; Chan, D.; Salinas, R.; Woynarowska, B.; Woynarowski, J.M. DNA strand breaks and apoptosis induced by oxaliplatin in cancer cells. Biochem. Pharmacol. 2003, 66, 225–237. [Google Scholar] [CrossRef]
- Gupta, E.; Mick, R.; Ramirez, J.; Wang, X.; Lestingi, T.M.; Vokes, E.E.; Ratain, M.J. Pharmacokinetic and pharmacodynamic evaluation of the topoisomerase inhibitor irinotecan in cancer patients. J. Clin. Oncol. 1997, 15, 1502–1510. [Google Scholar] [CrossRef]
- Karamouzis, M.V.; Athanasiadis, I.; Samelis, G.; Vallilas, C.; Bokas, A.; Nikolaidi, A.; Dimitriadou, A.; Sarantis, P.; Pistamaltzian, N.; Schizas, D.; et al. The Impact of Thromboprophylaxis on the Survival of Patients with Advanced Pancreatic Cancer. The Pancreatic Cancer and Tinzaparin (PaCT) Study. Cancers 2021, 13, 2884. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarantis, P.; Bokas, A.; Papadimitropoulou, A.; Koustas, E.; Theocharis, S.; Papakotoulas, P.; Schizas, D.; Papalampros, A.; Felekouras, E.; Papavassiliou, A.G.; et al. Combinatorial Treatment of Tinzaparin and Chemotherapy Can Induce a Significant Antitumor Effect in Pancreatic Cancer. Int. J. Mol. Sci. 2021, 22, 7053. https://doi.org/10.3390/ijms22137053
Sarantis P, Bokas A, Papadimitropoulou A, Koustas E, Theocharis S, Papakotoulas P, Schizas D, Papalampros A, Felekouras E, Papavassiliou AG, et al. Combinatorial Treatment of Tinzaparin and Chemotherapy Can Induce a Significant Antitumor Effect in Pancreatic Cancer. International Journal of Molecular Sciences. 2021; 22(13):7053. https://doi.org/10.3390/ijms22137053
Chicago/Turabian StyleSarantis, Panagiotis, Alexandros Bokas, Adriana Papadimitropoulou, Evangelos Koustas, Stamatios Theocharis, Pavlos Papakotoulas, Dimitrios Schizas, Alexandros Papalampros, Evangelos Felekouras, Athanasios G. Papavassiliou, and et al. 2021. "Combinatorial Treatment of Tinzaparin and Chemotherapy Can Induce a Significant Antitumor Effect in Pancreatic Cancer" International Journal of Molecular Sciences 22, no. 13: 7053. https://doi.org/10.3390/ijms22137053
APA StyleSarantis, P., Bokas, A., Papadimitropoulou, A., Koustas, E., Theocharis, S., Papakotoulas, P., Schizas, D., Papalampros, A., Felekouras, E., Papavassiliou, A. G., & Karamouzis, M. V. (2021). Combinatorial Treatment of Tinzaparin and Chemotherapy Can Induce a Significant Antitumor Effect in Pancreatic Cancer. International Journal of Molecular Sciences, 22(13), 7053. https://doi.org/10.3390/ijms22137053











