Epileptic Mechanisms Shared by Alzheimer’s Disease: Viewed via the Unique Lens of Genetic Epilepsy
Abstract
1. Introduction
2. Major Established Pathological Features of AD
3. Increased Epileptiform Activity or Seizures in AD: An Emergent Common but Under-Recognized Feature
4. Increased EEG Abnormalities in AD Patients
5. Excitatory/Inhibitory (E/I) Imbalance Contributes to Altered Hyperexcitability and Cognitive Decline in AD
6. The Overlapping Mechanisms Identified in GE Associated with Mutations in GABRG2 and AD
7. Insights from GE into Understanding E/I Imbalance in AD
8. Insights from Ketogenic Diet Treatment in GE into Understanding E/I Imbalance and Seizure Occurrence in AD
9. Insights from GE into Understanding Impaired Cognition in AD
10. Endoplasmic Reticulum (ER) Stress Exists in Both GE and AD
11. ER Stress Leads to Impaired Membrane Protein Trafficking and Altered Synaptic Physiology
12. ER Stress, Proteostasis, and Reduced Membrane Protein Trafficking in AD
13. Increased ER Stress Can Cause Increased Neuroinflammation from Very Early on: Evidence from an Epilepsy Mouse Gabrg2+/Q390X
14. Impaired GABAergic Signaling Is a Converging Pathway of Pathophysiology in GE
15. Altered GABAergic Neurotransmission Including GABA Neurotransmitter, Receptors, and Transporters in AD
16. Impaired GABAergic Interneurons in AD
17. Altered GABA Level and Tonic Inhibition in AD
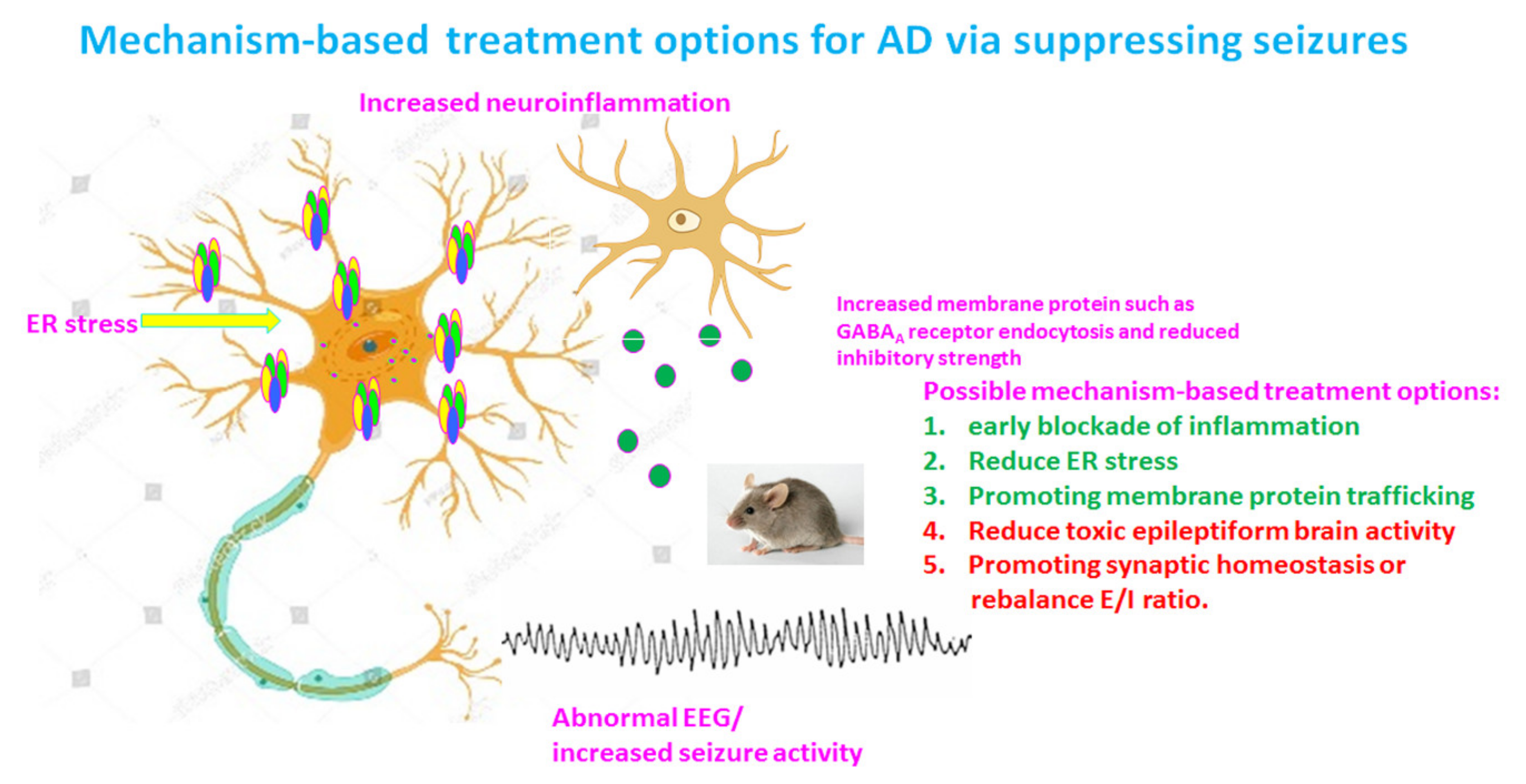
18. Treatment Opportunities for AD
19. Targeting GABAergic Neurotransmission as Treatment for AD
20. Targeting ER Stress as a Treatment Option in AD
21. Targeting Tau as a Treatment Option for AD and Insights from GE
22. Targeting Glutamatergic Neurotransmission as a Treatment Option for AD
23. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Szaruga, M.; Munteanu, B.; Lismont, S.; Veugelen, S.; Horré, K.; Mercken, M.; Saido, T.C.; Ryan, N.S.; De Vos, T.; Savvides, S.N.; et al. Alzheimer’s-Causing Mutations Shift Aβ Length by Destabilizing γ-Secretase-Aβn Interactions. Cell 2017, 170, 443–456. [Google Scholar] [CrossRef]
- Veugelen, S.; Saito, T.; Saido, T.C.; Chavez-Gutierrez, L.; De Strooper, B. Familial Alzheimer’s disease mutations in presenilin generate amyloidogenic Abeta peptide seeds. Neuron 2016, 90, 410–416. [Google Scholar] [CrossRef]
- Cacace, R.; Sleegers, K.; Van Broeckhoven, C. Molecular genetics of early-onset Alzheimer’s disease revisited. Alzheimer’s Dement. 2016, 12, 733–748. [Google Scholar] [CrossRef]
- Benilova, I.; Karran, E.; De Strooper, B. The toxic Abeta oligomer and Alzheimer’s disease: An emperor in need of clothes. Nat. Neurosci. 2012, 15, 349–357. [Google Scholar] [CrossRef]
- Umeda, T.; Kimura, T.; Yoshida, K.; Takao, K.; Fujita, Y.; Matsuyama, S.; Sakai, A.; Yamashita, M.; Yamashita, Y.; Ohnishi, K.; et al. Mutation-induced loss of APP function causes GABAergic depletion in recessive familial Alzheimer’s disease: Analysis of Osaka mutation-knockin mice. Acta Neuropathol. Commun. 2017, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Xia, D.; Watanabe, H.; Wu, B.; Lee, S.H.; Li, Y.; Tsvetkov, E.; Bolshakov, V.Y.; Shen, J.; Kelleher, R.J., 3rd. Presenilin-1 knockin mice reveal loss-of-function mechanism for familial Alzheimer’s disease. Neuron 2015, 85, 967–981. [Google Scholar] [CrossRef] [PubMed]
- Bramblett, G.T.; Goedert, M.; Jakes, R.; Merrick, S.E.; Trojanowski, J.Q.; Lee, V.M. Abnormal tau phosphorylation at Ser396 in Alzheimer’s disease recapitulates development and contributes to reduced microtubule binding. Neuron 1993, 10, 1089–1099. [Google Scholar] [CrossRef]
- Liu, Y.; Guo, C.; Ding, Y.; Long, X.; Li, W.; Ke, D.; Wang, Q.; Liu, R.; Wang, J.-Z.; Zhang, H.; et al. Blockage of AEP attenuates TBI-induced tau hyperphosphorylation and cognitive impairments in rats. Aging 2020, 12, 19421–19439. [Google Scholar] [CrossRef]
- Kempuraj, D.; Mentor, S.; Thangavel, R.; Ahmed, M.E.; Selvakumar, G.P.; Raikwar, S.P.; Dubova, I.; Zaheer, S.; Iyer, S.S.; Zaheer, A. Mast cells in stress, pain, blood-brain barrier, neuroinflammation and Alzheimer’s disease. Front. Cell. Neurosci. 2019, 13, 54. [Google Scholar] [CrossRef]
- Marttinen, M.; Paananen, J.; Neme, A.; Mitra, V.; Takalo, M.; Natunen, T.; Paldanius, K.M.A.; Mäkinen, P.; Bremang, M.; Kurki, M.I.; et al. A multiomic approach to characterize the temporal sequence in Alzheimer’s disease-related pathology. Neurobiol. Dis. 2019, 124, 454–468. [Google Scholar] [CrossRef] [PubMed]
- Convit, A.; de Leon, M.J.; Golomb, J.; George, A.E.; Tarshish, C.Y.; Bobinski, M.; Tsui, W.; De Santi, S.; Wegiel, J.; Wisniewski, H. Hippocampal atrophy in early Alzheimer’s disease: Anatomic specificity and validation. Psychiatr. Q. 1993, 64, 371–387. [Google Scholar] [CrossRef]
- Gomez-Isla, T.; Price, J.L.; McKeel, D.W., Jr.; Morris, J.C.; Growdon, J.H.; Hyman, B.T. Profound loss of layer II entorhinal cortex neurons occurs in very mild Alzheimer’s disease. J. Neurosci. 1996, 16, 4491–4500. [Google Scholar] [CrossRef]
- Delbeuck, X.; Van der Linden, M.; Collette, F. Alzheimer’s disease as a disconnection syndrome? Neuropsychol. Rev. 2003, 13, 79–92. [Google Scholar] [CrossRef]
- Scharfman, H.E. “Untangling” Alzheimer’s disease and epilepsy. Epilepsy Curr. 2012, 12, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Vossel, K.A.; Tartaglia, M.C.; Nygaard, H.B.; Zeman, A.Z.; Miller, B.L. Epileptic activity in Alzheimer’s disease: Causes and clinical relevance. Lancet Neurol. 2017, 16, 311–322. [Google Scholar] [CrossRef]
- Horvath, A.; Szucs, A.; Hidasi, Z.; Csukly, G.; Barcs, G.; Kamondi, A. Prevalence, semiology, and risk factors of epilepsy in Alzheimer’s disease: An ambulatory EEG study. J. Alzheimer’s Dis. 2018, 63, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Szucs, A.; Barcs, G.; Noebels, J.L.; Kamondi, A. Epileptic seizures in Alzheimer disease: A review. Alzheimer Dis. Assoc. Disord. 2016, 30, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Jafarian, M.; Karimzadeh, F.; Alipour, F.; Attari, F.; Lotfinia, A.A.; Speckmann, E.J.; Zarrindast, M.R.; Gorji, A. Cognitive impairments and neuronal injury in different brain regions of a genetic rat model of absence epilepsy. Neuroscience 2015, 298, 161–170. [Google Scholar] [CrossRef]
- Aiba, I.; Noebels, J.L. Spreading depolarization in the brainstem mediates sudden cardiorespiratory arrest in mouse SUDEP models. Sci. Transl. Med. 2015, 7, 282ra46. [Google Scholar] [CrossRef]
- Roberson, E.D.; Halabisky, B.; Yoo, J.W.; Yao, J.; Chin, J.; Yan, F.G.; Wu, T.; Hamto, P.; Devidze, N.; Yu, G.Q. Amyloid-beta/Fyn-induced synaptic, network, and cognitive impairments depend on tau levels in multiple mouse models of Alzheimer’s disease. J. Neurosci. 2011, 31, 700–711. [Google Scholar] [CrossRef]
- Chin, J.; Scharfman, H.E. Shared cognitive and behavioral impairments in epilepsy and Alzheimer’s disease and potential underlying mechanisms. Epilepsy Behav. 2013, 26, 343–351. [Google Scholar] [CrossRef]
- Volicer, L.; Smith, S.; Volicer, B.J. Effect of seizures on progression of dementia of the Alzheimer type. Dementia 1995, 6, 258–263. [Google Scholar] [CrossRef]
- Beagle, A.J.; Darwish, S.M.; Ranasinghe, K.G.; La, A.L.; Karageorgiou, E.; Vossel, K.A. Relative incidence of seizures and myoclonus in Alzheimer’s disease, dementia with Lewy bodies, and frontotemporal dementia. J. Alzheimer’s Dis. 2017, 60, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Vossel, K.A.; Ranasinghe, K.G.; Beagle, A.J.; Mizuiri, D.; Honma, S.M.; Dowling, A.F.; Darwish, S.M.; Van Berlo, V.; Barnes, D.E.; Mantle, M.; et al. Incidence and impact of subclinical epileptiform activity in Alzheimer’s disease. Ann. Neurol. 2016, 80, 858–870. [Google Scholar] [CrossRef]
- Lam, A.D.; Deck, G.; Goldman, A.; Eskandar, E.N.; Noebels, J.; Cole, A.J. Silent hippocampal seizures and spikes identified by foramen ovale electrodes in Alzheimer’s disease. Nat. Med. 2017, 23, 678–680. [Google Scholar] [CrossRef]
- Cretin, B.; Philippi, N.; Dibitonto, L.; Blanc, F. Epilepsy at the prodromal stages of neurodegenerative diseases. Geriatr. Psychol. Neuropsychiatr. Vieil. 2017, 15, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Joutsa, J.; Rinne, J.O.; Hermann, B.; Karrasch, M.; Anttinen, A.; Shinnar, S.; Sillanpää, M. Association between childhood-onset epilepsy and amyloid burden 5 decades later. JAMA Neurol. 2017, 74, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Mi, D.J.; Dixit, S.; Warner, T.A.; Kennard, J.A.; Scharf, D.A.; Kessler, E.S.; Moore, L.M.; Consoli, D.C.; Bown, C.W.; Eugene, A.J.; et al. Altered glutamate clearance in ascorbate deficient mice increases seizure susceptibility and contributes to cognitive impairment in APP/PSEN1 mice. Neurobiol. Aging 2018, 71, 241–254. [Google Scholar] [CrossRef]
- Kang, J.Q.; Shen, W.; Zhou, C.; Xu, D.; Macdonald, R.L. The human epilepsy mutation GABRG2(Q390X) causes chronic subunit accumulation and neurodegeneration. Nat. Neurosci. 2015, 18, 988–996. [Google Scholar] [CrossRef]
- Macdonald, R.L.; Kang, J.Q.; Gallagher, M.J. GABAA Receptor Subunit Mutations and Genetic Epilepsies. In Jasper’s Basic Mechanisms of the Epilepsies; Nathional Center for Biotechnology Information: Bethesda, MD, USA, 2012. [Google Scholar]
- Voglein, J.; Noachtar, S.; McDade, E.; Quaid, K.A.; Salloway, S.; Ghetti, B.; Noble, J.; Berman, S.; Chhatwal, J.; Mori, H.; et al. Seizures as an early symptom of autosomal dominant Alzheimer’s disease. Neurobiol. Aging 2019, 76, 18–23. [Google Scholar] [CrossRef]
- Salloway, S.; Marshall, G.A.; Lu, M.; Brashear, H.R. Long-term safety and efficacy of bapineuzumab in patients with mild-to-moderate Alzheimer’s disease: A phase 2, open-label extension study. Curr. Alzheimer Res. 2018, 15, 1231–1243. [Google Scholar] [CrossRef]
- Martin, R.C.; Gaston, T.E.; Thompson, M.; Ampah, S.P.; Cutter, G.; Bebin, E.M.; Szaflarski, J.P. Cognitive functioning following long-term cannabidiol use in adults with treatment-resistant epilepsy. Epilepsy Behav. 2019, 97, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Musaeus, C.S.; Engedal, K.; Hogh, P.; Jelic, V.; Mørup, M.; Naik, M.; Oeksengaard, A.R.; Snaedal, J.; Wahlund, L.O.; Waldemar, G.; et al. EEG theta power is an early marker of cognitive decline in dementia due to Alzheimer’s disease. J. Alzheimer’s Dis. 2018, 64, 1359–1371. [Google Scholar] [CrossRef] [PubMed]
- Buda, O.; Arsene, D.; Ceausu, M.; Dermengiu, D.; Curca, G.C. Georges Marinesco and the early research in neuropathology. Neurology 2009, 72, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Tonder, N.; Kragh, J.; Finsen, B.R.; Bolwig, T.G.; Zimmer, J. Kindling induces transient changes in neuronal expression of somatostatin, neuropeptide Y, and calbindin in adult rat hippocampus and fascia dentata. Epilepsia 1994, 35, 1299–1308. [Google Scholar] [CrossRef]
- Busche, M.A.; Eichhoff, G.; Adelsberger, H.; Abramowski, D.; Wiederhold, K.H.; Haass, C.; Staufenbiel, M.; Konnerth, A.; Garaschuk, O. Clusters of hyperactive neurons near amyloid plaques in a mouse model of Alzheimer’s disease. Science 2008, 321, 1686–1689. [Google Scholar] [CrossRef]
- Dolev, I.; Fogel, H.; Milshtein, H.; Berdichevsky, Y.; Lipstein, N.; Brose, N.; Gazit, N.; Slutsky, I. Spike bursts increase amyloid-beta 40/42 ratio by inducing a presenilin-1 conformational change. Nat. Neurosci. 2013, 16, 587–595. [Google Scholar] [CrossRef]
- Jayadev, S.; Leverenz, J.B.; Steinbart, E.; Stahl, J.; Klunk, W.; Yu, C.E.; Bird, T.D. Alzheimer’s disease phenotypes and genotypes associated with mutations in presenilin 2. Brain 2010, 133, 1143–1154. [Google Scholar] [CrossRef]
- Rao, S.C.; Dove, G.; Cascino, G.D.; Petersen, R.C. Recurrent seizures in patients with dementia: Frequency, seizure types, and treatment outcome. Epilepsy Behav. 2009, 14, 118–120. [Google Scholar] [CrossRef]
- Armon, C.; Peterson, G.W.; Liwnicz, B.H. Alzheimer’s disease underlies some cases of complex partial status epilepticus. J. Clin. Neurophysiol. 2000, 17, 511–518. [Google Scholar] [CrossRef]
- Povysheva, N.V.; Johnson, J.W. Effects of memantine on the excitation-inhibition balance in prefrontal cortex. Neurobiol. Dis. 2016, 96, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Haider, B.; McCormick, D.A. Rapid neocortical dynamics: Cellular and network mechanisms. Neuron 2009, 62, 171–189. [Google Scholar] [CrossRef]
- Homayoun, H.; Moghaddam, B. NMDA receptor hypofunction produces opposite effects on prefrontal cortex interneurons and pyramidal neurons. J. Neurosci. 2007, 27, 11496–11500. [Google Scholar] [CrossRef]
- Shao, L.R.; Habela, C.W.; Stafstrom, C.E. Pediatric epilepsy mechanisms: Expanding the Paradigm of excitation/inhibition imbalance. Children 2019, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.Q.; Barnes, G. A common susceptibility factor of both autism and epilepsy: Functional deficiency of GABAA receptors. J. Autism Dev. Disord. 2013, 43, 68–79. [Google Scholar] [CrossRef]
- Mody, I.; Pearce, R.A. Diversity of inhibitory neurotransmission through GABA(A) receptors. Trends Neurosci. 2004, 27, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Cope, D.W.; Di, G.G.; Fyson, S.J.; Orbán, G.; Errington, A.C.; Lorincz, M.L.; Gould, T.M.; Carter, D.A.; Crunelli, V. Enhanced tonic GABAA inhibition in typical absence epilepsy. Nat. Med. 2009, 15, 1392–1398. [Google Scholar] [CrossRef] [PubMed]
- Carvill, G.L.; McMahon, J.M.; Schneider, A.; Zemel, M.; Myers, C.T.; Saykally, J.; Nguyen, J.; Robbiano, A.; Zara, F.; Specchio, N.; et al. Mutations in the GABA transporter SLC6A1 cause epilepsy with myoclonic-atonic seizures. Am. J. Hum. Genet. 2015, 96, 808–815. [Google Scholar] [CrossRef]
- Cai, K.; Wang, J.; Eissman, J.; Wang, J.; Nwosu, G.; Shen, W.; Liang, H.C.; Li, X.J.; Zhu, H.X.; Yi, Y.H.; et al. A missense mutation in SLC6A1 associated with Lennox-Gastaut syndrome impairs GABA transporter 1 protein trafficking and function. Exp. Neurol. 2019, 320, 112973. [Google Scholar] [CrossRef]
- Wang, J.; Poliquin, S.; Mermer, F.; Eissman, J.; Delpire, E.; Wang, J.; Shen, W.; Cai, K.; Li, B.M.; Li, Z.Y.; et al. Endoplasmic reticulum retention and degradation of a mutation in SLC6A1 associated with epilepsy and autism. Mol. Brain 2020, 13, 76. [Google Scholar] [CrossRef]
- Mermer, F.; Poliquin, S.; Rigsby, K.; Rastogi, A.; Shen, W.; Romero-Morales, A.; Nwosu, G.; McGrath, P.; Demerast, S.; Aoto, J.; et al. Common molecular mechanisms of SLC6A1 variant-mediated neurodevelopmental disorders in astrocytes and neurons. Brain 2021. [Google Scholar] [CrossRef] [PubMed]
- Rissman, R.A.; Mobley, W.C. Implications for treatment: GABAA receptors in aging, Down syndrome and Alzheimer’s disease. J. Neurochem. 2011, 117, 613–622. [Google Scholar] [CrossRef]
- Fuhrer, T.E.; Palpagama, T.H.; Waldvogel, H.J.; Synek, B.J.L.; Turner, C.; Faull, R.L.; Kwakowsky, A. Impaired expression of GABA transporters in the human Alzheimer’s disease hippocampus, subiculum, entorhinal cortex and superior temporal gyrus. Neuroscience 2017, 351, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Meeker, K.D.; Meabon, J.S.; Cook, D.G. Partial loss of the glutamate transporter GLT-1 alters brain Akt and insulin signaling in a mouse model of Alzheimer’s disease. J. Alzheimer’s Dis. 2015, 45, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Dinamarca, M.C.; Raveh, A.; Schneider, A.; Fritzius, T.; Früh, S.; Rem, P.D.; Stawarski, M.; Lalanne, T.; Turecek, R.; Choo, M.; et al. Complex formation of APP with GABAB receptors links axonal trafficking to amyloidogenic processing. Nat. Commun. 2019, 10, 1331. [Google Scholar] [CrossRef] [PubMed]
- Vico, V.E.; Etter, G.; Williams, S. Excitatory-inhibitory imbalance in Alzheimer’s disease and therapeutic significance. Neurobiol. Dis. 2019, 127, 605–615. [Google Scholar] [CrossRef]
- Macdonald, R.L.; Kang, J.Q. mRNA surveillance and endoplasmic reticulum quality control processes alter biogenesis of mutant GABAA receptor subunits associated with genetic epilepsies. Epilepsia 2012, 53, 59–70. [Google Scholar] [CrossRef]
- Warner, T.A.; Shen, W.; Huang, X.; Liu, Z.; Macdonald, R.L.; Kang, J.Q. Differential molecular and behavioral alterations in mouse models of GABRG2 haploinsufficiency versus dominant negative mutations associated with human epilepsy. Hum. Mol. Genet. 2016, 25, 3192–3207. [Google Scholar] [CrossRef]
- Gonzalez Otarula, K.A.; von Ellenrieder, N.; Cuello-Oderiz, C.; Dubeau, F.; Gotman, J. High-frequency oscillation networks and surgical outcome in adult focal epilepsy. Ann. Neurol. 2019, 85, 485–494. [Google Scholar] [CrossRef]
- Huang, X.; Zhou, C.; Tian, M.; Kang, J.Q.; Shen, W.; Verdier, K.; Pimenta, A.; MacDonald, R.L. Overexpressing wild-type gamma2 subunits rescued the seizure phenotype in Gabrg2+/Q390X Dravet syndrome mice. Epilepsia 2017, 58, 1451–1461. [Google Scholar] [CrossRef] [PubMed]
- Brockmann, K.; Wang, D.; Korenke, C.G.; von Moers, A.; Ho, Y.Y.; Pascual, J.M.; Kuang, K.; Yang, H.; Ma, L.; Kranz-Eble, P.; et al. Autosomal dominant glut-1 deficiency syndrome and familial epilepsy. Ann. Neurol. 2001, 50, 476–485. [Google Scholar] [CrossRef]
- Veggiotti, P.; Teutonico, F.; Alfei, E.; Nardocci, N.; Zorzi, G.; Tagliabue, A.; De Giorgis, V.; Balottin, U. Glucose transporter type 1 deficiency: Ketogenic diet in three patients with atypical phenotype. Brain Dev. 2010, 32, 404–408. [Google Scholar] [CrossRef]
- Pauletti, A.; Terrone, G.; Shekh-Ahmad, T.; Salamone, A.; Ravizza, T.; Rizzi, M.; Pastore, A.; Pascente, R.; Liang, L.P.; Villa, B.R.; et al. Targeting oxidative stress improves disease outcomes in a rat model of acquired epilepsy. Brain 2019, 142, e39. [Google Scholar] [CrossRef] [PubMed]
- Loup, F.; Wieser, H.G.; Yonekawa, Y.; Aguzzi, A.; Fritschy, J.M. Selective alterations in GABAA receptor subtypes in human temporal lobe epilepsy. J. Neurosci. 2000, 20, 5401–5419. [Google Scholar] [CrossRef] [PubMed]
- Loup, F.; Picard, F.; Andre, V.M.; Kehrli, P.; Yonekawa, Y.; Wieser, H.G.; Fritschy, J.M. Altered expression of alpha3-containing GABAA receptors in the neocortex of patients with focal epilepsy. Brain 2006, 129, 3277–3289. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jafarian, M.; Modarres Mousavi, S.M.; Alipour, F.; Aligholi, H.; Noorbakhsh, F.; Ghadipasha, M.; Gharehdaghi, J.; Kellinghaus, C.; Kovac, S.; Ghadiri, M.K.; et al. Cell injury and receptor expression in the epileptic human amygdala. Neurobiol. Dis. 2019, 124, 416–427. [Google Scholar] [CrossRef]
- Soto-Insuga, V.; Lopez, R.G.; Losada-Del Pozo, R.; Rodrigo-Moreno, M.; Cayuelas, E.M.; Giráldez, B.G.; Díaz-Gómez, E.; Sánchez-Martín, G.; García, L.O.; Serratosa, J.M.; et al. Glut1 deficiency is a rare but treatable cause of childhood absence epilepsy with atypical features. Epilepsy Res. 2019, 154, 39–41. [Google Scholar] [CrossRef] [PubMed]
- Mefford, H.C.; Yendle, S.C.; Hsu, C.; Cook, J.; Geraghty, E.; McMahon, J.M.; Eeg-Olofsson, O.; Sadleir, L.G.; Gill, D.; Ben-Zeev, B.; et al. Rare copy number variants are an important cause of epileptic encephalopathies. Ann. Neurol. 2011, 70, 974–985. [Google Scholar] [CrossRef]
- Saitoh, M.; Shinohara, M.; Hoshino, H.; Kubota, M.; Amemiya, K.; Takanashi, J.L.; Hwang, S.K.; Hirose, S.; Mizuguchi, M. Mutations of the SCN1A gene in acute encephalopathy. Epilepsia 2012, 53, 558–564. [Google Scholar] [CrossRef]
- Saitoh, M.; Ishii, A.; Ihara, Y.; Hoshino, A.; Terashima, H.; Kubota, M.; Kikuchi, K.; Yamanaka, G.; Amemiya, K.; Hirose, S.; et al. Missense mutations in sodium channel SCN1A and SCN2A predispose children to encephalopathy with severe febrile seizures. Epilepsy Res. 2015, 117, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.Q.; Macdonald, R.L. Molecular pathogenic basis for GABRG2 mutations associated with a spectrum of epilepsy syndromes, from generalized absence epilepsy to Dravet syndrome. JAMA Neurol. 2016, 73, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.W.; Zhang, Q.; Cai, K.; Poliquin, S.; Shen, W.; Winters, N.; Yi, Y.H.; Wang, J.; Hu, N.; Macdonald, R.L.; et al. Synaptic clustering differences due to different GABRB3 mutations cause variable epilepsy syndromes. Brain 2019, 142, 3028–3044. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Li, M.; Wu, M.; Shi, K.; Fang, B.; Wang, J. FAD-linked Presenilin-1 V97L mutation impede tranport regulation and intracellular Ca(2+) homeostasis under ER stress. Int. J. Clin. Exp. Med. 2015, 8, 20742–20750. [Google Scholar]
- Gerakis, Y.; Hetz, C. A decay of the adaptive capacity of the unfolded protein response exacerbates Alzheimer’s disease. Neurobiol. Aging 2018, 63, 162–164. [Google Scholar] [CrossRef]
- Kang, J.Q.; Shen, W.; Macdonald, R.L. Trafficking-deficient mutant GABRG2 subunit amount may modify epilepsy phenotype. Ann. Neurol. 2013, 74, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Xia, G.; Pourali, P.; Warner, T.A.; Zhang, C.Q.; Macdonald, L.; Kang, J.Q. Altered GABAA receptor expression in brainstem nuclei and SUDEP in Gabrg2(+/Q390X) mice associated with epileptic encephalopathy. Epilepsy Res. 2016, 123, 50–54. [Google Scholar] [CrossRef]
- Shen, W.; Poliquin, S.; Macdonald, R.L.; Dong, M.; Kang, J.Q. Endoplasmic reticulum stress increases inflammatory cytokines in an epilepsy mouse model Gabrg2(+/Q390X) knockin: A link between genetic and acquired epilepsy? Epilepsia 2020, 61, 2301–2312. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Cao, J.; Yang, S.S.; Fu, Z.Q.; Zeng, P.; Chu, J.; Ning, L.N.; Zhang, T.; Shi, Y.; Tian, Q.; et al. Endoplasmic reticulum stress induces spatial memory deficits by activating GSK-3. J. Cell. Mol. Med. 2018, 22, 3489–3502. [Google Scholar] [CrossRef]
- Wang, J.Z.; Grundke-Iqbal, I.; Iqbal, K. Glycosylation of microtubule-associated protein tau: An abnormal posttranslational modification in Alzheimer’s disease. Nat. Med. 1996, 2, 871–875. [Google Scholar] [CrossRef]
- Yu, A.; Fox, S.G.; Cavallini, A.; Kerridge, C.; O’Neill, M.J.; Wolak, J.; Bose, S.; Morimoto, R.I. Tau protein aggregates inhibit the protein-folding and vesicular trafficking arms of the cellular proteostasis network. J. Biol. Chem. 2019, 294, 7917–7930. [Google Scholar] [CrossRef]
- Poirier, Y.; Grimm, A.; Schmitt, K.; Eckert, A. Link between the unfolded protein response and dysregulation of mitochondrial bioenergetics in Alzheimer’s disease. Cell. Mol. Life Sci. 2019, 76, 1419–1431. [Google Scholar] [CrossRef]
- Perez, A.; Garcia-Penton, L.; Canales-Rodriguez, E.J.; Lerma-Usabiaga, G.; Iturria-Medina, Y.; Román, F.J.; Davidson, D.; Alemán-Gómez, Y.; Acha, J.; Carreiras, M. Brain morphometry of Dravet syndrome. Epilepsy Res. 2014, 108, 1326–1334. [Google Scholar] [CrossRef]
- Perez, S.E.; Miguel, J.C.; He, B.; Malek-Ahmadi, M.; Abrahamson, E.E.; Ikonomovic, M.D.; Lott, I.; Doran, E.; Alldred, M.J.; Ginsberg, S.D.; et al. Frontal cortex and striatal cellular and molecular pathobiology in individuals with Down syndrome with and without dementia. Acta Neuropathol. 2019, 137, 413–436. [Google Scholar] [CrossRef]
- Meisler, M.H.; Kearney, J.A. Sodium channel mutations in epilepsy and other neurological disorders. J. Clin. Investig. 2005, 115, 2010–2017. [Google Scholar] [CrossRef] [PubMed]
- Czirr, E.; Wyss-Coray, T. The immunology of neurodegeneration. J. Clin. Investig. 2012, 122, 1156–1163. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; Nicoll, J.A.; Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol. 2010, 6, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Marklund, N.; Vedung, F.; Lubberink, M.; Tegner, Y.; Johansson, J.; Blennow, K.; Zetterberg, H.; Fahlström, M.; Haller, S.; Stenson, S.; et al. Tau aggregation and increased neuroinflammation in athletes after sports-related concussions and in traumatic brain injury patients—A PET/MR study. Neuroimage Clin. 2021, 30, 102665. [Google Scholar] [CrossRef]
- Kang, J.Q. Defects at the crossroads of GABAergic signaling in generalized genetic epilepsies. Epilepsy Res. 2017, 137, 9–18. [Google Scholar] [CrossRef]
- Frahm, C.; Draguhn, A. GAD and GABA transporter (GAT-1) mRNA expression in the developing rat hippocampus. Brain Res. Dev. Brain Res. 2001, 132, 1–13. [Google Scholar] [CrossRef]
- Powell, E.M. Interneuron development and epilepsy: Early genetic defects cause long-term consequences in seizures and susceptibility. Epilepsy Curr. 2013, 13, 172–176. [Google Scholar] [CrossRef]
- Hunt, R.F.; Baraban, S.C. Interneuron transplantation as a treatment for epilepsy. Cold Spring Harb. Perspect. Med. 2015, 5, a022376. [Google Scholar] [CrossRef]
- Hunt, R.F.; Girskis, K.M.; Rubenstein, J.L.; Alvarez-Buylla, A.; Baraban, S.C. GABA progenitors grafted into the adult epileptic brain control seizures and abnormal behavior. Nat. Neurosci. 2013, 16, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Saito, T.; Saido, T.C.; Mody, I. Novel quantitative analyses of spontaneous synaptic events in cortical pyramidal cells reveal subtle parvalbumin-expressing interneuron dysfunction in a knock-in mouse model of Alzheimer’s disease. eNeuro 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Losa, M.; Tracy, T.E.; Ma, K.; Tegner, Y.; Johansson, J.; Blennow, K.; Zetterberg, H.; Fahlström, M.; Haller, S.; Stenson, S.; et al. Nav1.1-overexpressing interneuron transplants restore brain rhythms and cognition in a mouse model of Alzheimer’s disease. Neuron 2018, 98, 75–89. [Google Scholar] [CrossRef]
- Jo, S.; Yarishkin, O.; Hwang, Y.J.; Chun, Y.E.; Park, M.; Woo, D.H.; Bae, J.Y.; Kim, T.; Lee, J.; Chun, H.; et al. GABA from reactive astrocytes impairs memory in mouse models of Alzheimer’s disease. Nat. Med. 2014, 20, 886–896. [Google Scholar] [CrossRef]
- Wu, Z.; Guo, Z.; Gearing, M.; Chen, G. Tonic inhibition in dentate gyrus impairs long-term potentiation and memory in an Alzheimer’s [corrected] disease model. Nat. Commun. 2014, 5, 4159. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yoon, B.E.; Berglund, K.; Oh, S.J.; Park, H.; Shin, H.S.; Augustine, G.J.; Lee, C.J. Channel-mediated tonic GABA release from glia. Science 2010, 330, 790–796. [Google Scholar] [CrossRef]
- Heja, L.; Barabas, P.; Nyitrai, G.; Kékesi, K.A.; Lasztóczi, B.; Toke, O.; Tárkányi, G.; Madsen, K.; Schousboe, A.; Dobolyi, A.; et al. Glutamate uptake triggers transporter-mediated GABA release from astrocytes. PLoS ONE 2009, 4, e7153. [Google Scholar] [CrossRef]
- Oh, W.C.; Lutzu, S.; Castillo, P.E.; Kwon, H.B. De novo synaptogenesis induced by GABA in the developing mouse cortex. Science 2016, 353, 1037–1040. [Google Scholar] [CrossRef]
- Abbas, G.; Mahmood, W.; Kabir, N. Recent progress on the role of GABAergic neurotransmission in the pathogenesis of Alzheimer’s disease. Rev. Neurosci. 2016, 27, 449–455. [Google Scholar] [CrossRef]
- Gacsalyi, I.; Moricz, K.; Gigler, G.; Megyeri, K.; Machado, P.; Antoni, F.A. Persistent therapeutic effect of a novel alpha5-GABAA receptor antagonist in rodent preclinical models of vascular cognitive impairment. Eur. J. Pharmacol. 2018, 834, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Pilipenko, V.; Narbute, K.; Amara, I.; Trovato, A.; Scuto, M.; Pupure, J.; Jansone, B.; Poikans, J.; Bisenieks, E.; Klusa, V.; et al. GABA-containing compound gammapyrone protects against brain impairments in Alzheimer’s disease model male rats and prevents mitochondrial dysfunction in cell culture. J. Neurosci. Res. 2019, 97, 708–726. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Iwamura, Y. AC-3933, a benzodiazepine partial inverse agonist, improves memory performance in MK-801-induced amnesia mouse model. Pharmacol. Biochem. Behav. 2016, 144, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, S.; Ishii, A.; Kamano, N.; Watamura, N.; Saito, T.; Ohshima, T.; Yokosuka, M.; Saido, T.C. Endoplasmic reticulum stress responses in mouse models of Alzheimer’s disease: Overexpression paradigm versus knockin paradigm. J. Biol. Chem. 2018, 293, 3118–3125. [Google Scholar] [CrossRef] [PubMed]
- Hatayama, Y.; Hashimoto, T.; Kohayakawa, H.; Kiyoshi, T.; Nakamichi, K.; Kinoshita, T.; Yoshida, N. In vivo pharmacological characterization of AC-3933, a benzodiazepine receptor partial inverse agonist for the treatment of Alzheimer’s disease. Neuroscience 2014, 265, 217–225. [Google Scholar] [CrossRef]
- Holth, J.K.; Bomben, V.C.; Reed, J.G.; Inoue, T.; Younkin, L.; Younkin, S.G.; Pautler, R.G.; Botas, J.; Noebels, J.L. Tau loss attenuates neuronal network hyperexcitability in mouse and Drosophila genetic models of epilepsy. J. Neurosci. 2013, 33, 1651–1659. [Google Scholar] [CrossRef]
- Ittner, L.M.; Ke, Y.D.; Delerue, F.; Bi, M.; Gladbach, A.; van Eersel, J.; Wölfing, H.; Chieng, B.C.; Christie, M.J.; Napier, I.A.; et al. Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer’s disease mouse models. Cell 2010, 142, 387–397. [Google Scholar] [CrossRef]
- Gheyara, A.L.; Ponnusamy, R.; Djukic, B.; Craft, R.J.; Ho, K.; Guo, W.; Finucane, M.M.; Sanchez, P.E.; Mucke, L. Tau reduction prevents disease in a mouse model of Dravet syndrome. Ann. Neurol. 2014, 76, 443–456. [Google Scholar] [CrossRef]
- Marwick, K.; Skehel, P.; Hardingham, G.; Wyllie, D. Effect of a GRIN2A de novo mutation associated with epilepsy and intellectual disability on NMDA receptor currents and Mg(2+) block in cultured primary cortical neurons. Lancet 2015, 385, S65. [Google Scholar] [CrossRef]
- Ogden, K.K.; Chen, W.; Swanger, S.A.; McDaniel, M.J.; Fan, L.Z.; Hu, C.; Tankovic, A.; Kusumoto, H.; Kosobucki, G.J.; Schulien, A.J.; et al. Molecular mechanism of disease-associated mutations in the pre-M1 helix of NMDA receptors and potential rescue pharmacology. PLoS Genet. 2017, 13, e1006536. [Google Scholar] [CrossRef]
- Makino, M.; Takahashi-Ito, K.; Murasawa, H.; Pawlak, A.; Kashimoto, Y.; Kitano, Y. Memantine ameliorates learning and memory disturbance and the behavioral and psychological symptoms of dementia in thiamine-deficient mice. Pharmacol. Biochem. Behav. 2019, 183, 6–13. [Google Scholar] [CrossRef]
- Thancharoen, O.; Limwattananon, C.; Waleekhachonloet, O.; Rattanachotphanit, T.; Limwattananon, P.; Limpawattana, P. Ginkgo biloba extract (EGb761), cholinesterase inhibitors, and memantine for the treatment of mild-to-moderate Alzheimer’s disease: A network meta-analysis. Drugs Aging 2019, 36, 435–452. [Google Scholar] [CrossRef] [PubMed]
- Lipton, S.A. Pathologically-activated therapeutics for neuroprotection: Mechanism of NMDA receptor block by memantine and S-nitrosylation. Curr. Drug Targets 2007, 8, 621–632. [Google Scholar] [CrossRef]
- Zadori, D.; Veres, G.; Szalardy, L.; Klivenyi, P.; Toldi, J.; Vecsei, L. Glutamatergic dysfunctioning in Alzheimer’s disease and related therapeutic targets. J. Alzheimer’s Dis. 2014, 42, S177–S187. [Google Scholar] [CrossRef]
- Kang, J.Q.; Shen, W.; Macdonald, R.L. Why does fever trigger febrile seizures? GABAA receptor gamma2 subunit mutations associated with idiopathic generalized epilepsies have temperature-dependent trafficking deficiencies. J. Neurosci. 2006, 9, 2590–2597. [Google Scholar] [CrossRef]
- Kang, J.Q.; Shen, W.; Lee, M.; Gallagher, M.J.; Macdonald, R.L. Slow degradation and aggregation in vitro of mutant GABAA receptor gamma2(Q351X) subunits associated with epilepsy. J. Neurosci. 2010, 41, 13895–13905. [Google Scholar] [CrossRef]
- Tian, M.; Macdonald, R.L. The intronic GABRG2 mutation, IVS6+2T->G, associated with childhood absence epilepsy altered subunit mRNA intron splicing, activated nonsense-mediated decay, and produced a stable truncated γ2 subunit. J. Neurosci. 2012, 17, 5937–5952. [Google Scholar] [CrossRef]
- Shen, D.; Hernandez, C.C.; Shen, W.; Hu, N.; Poduri, A.; Shiedley, B.; Rotenberg, A.; Datta, A.N.; Leiz, S.; Patzer, S.; et al. De novo GABRG2 mutations associated with epileptic encephalopathies. Brain 2017, 1, 49–67. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Mei, D.; Freri, E.; Hernandez, C.C.; Granata, T.; Shen, W.; Macdonald, R.L.; Guerrini, R. Impaired surface αβγ GABA(A) receptor expression in familial epilepsy due to a GABRG2 frameshift mutation. Neurobiol. Dis. 2013, 50, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, M.J.; Shen, W.; Song, L.; Macdonald, R.L. Endoplasmic reticulum retention and associated degradation of a GABAA receptor epilepsy mutation that inserts an aspartate in the M3 transmembrane segment of the alpha1 subunit. J. Biol. Chem. 2005, 45, 37995–80004. [Google Scholar] [CrossRef] [PubMed]
- Poliquin, S.; Hughes, I.; Shen, W.; Mermer, F.; Wang, J.; Mack, T.; Xu, D.; Kang, J.-Q. Genetic mosaicism, intrafamilial phenotypic heterogeneity, and molecular defects of a novel missense SLC6A1 mutation associated with epilepsy and ADHD. Exp. Neurol. 2021, 342. [Google Scholar] [CrossRef] [PubMed]

| Gene | Mutation/Variants | Models | Postulated Mechanisms | Channel Function | Phenotypes | References |
|---|---|---|---|---|---|---|
| GABRG2 | Q390X | cells | Impaired oligomerization, ER retention | reduced | GEFS+, DS | [30] |
| GABRG2 | R82Q | cells | Impaired oligomerization, ER Retention | reduced | FS, CAE | [117] |
| GABRG2 | Q390X, W429X, W461X | cells | Impaired oligomerization, ER Retention | reduced | FS, GEFS+, DS | [77] |
| GABRG2 | Q390X | mice | ER rentention, dominant negative suppression | reduced | [62] | |
| GABRG2 | Q390X | Mice | protein accumulation, aggregation | reduced | GEFS+, DS | [118] |
| GABRG2 | Q390X | mice | increased neuroinflamamtion | reduced | DS | [79] |
| GABRG2 | IVS6+2T->G | cell mice | NMD, ERAD | reduced | CAE, FS | [119] |
| GABRG2 | A106T, I107T, P282S, etc. | cell | ERAD, ER retention | reduced | DEE | [120] |
| GABRG2 | S443delC | cells | ERAD, ER retention? | reduced | GEFS+ | [121] |
| GABRB3 | N328D | cells | ERAD, ER retention | reduced | LGS | [74] |
| GABRB3 | E357K | cells | ERAD, ER retention | reduced | JAE | [74] |
| GABRA1 | A322D | cells | ERAD, ER retention | reduced | JME | [122] |
| SLC6A1 | G234S | cells | ERAD, ER retention | reduced | DS | [51] |
| SLC6A1 | P361T | cells | ERAD, ER retention | reduced | autism, CAE | [52] |
| SLC6A1 | V125M | cells | ERAD, ER Retention | reduced | CAE, ADHD | [123] |
| SLC6A1 | 22 mutations | cells | ER Retention, dominant negative effect? | reduced | various phenotypes | [53] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.-Q. Epileptic Mechanisms Shared by Alzheimer’s Disease: Viewed via the Unique Lens of Genetic Epilepsy. Int. J. Mol. Sci. 2021, 22, 7133. https://doi.org/10.3390/ijms22137133
Kang J-Q. Epileptic Mechanisms Shared by Alzheimer’s Disease: Viewed via the Unique Lens of Genetic Epilepsy. International Journal of Molecular Sciences. 2021; 22(13):7133. https://doi.org/10.3390/ijms22137133
Chicago/Turabian StyleKang, Jing-Qiong. 2021. "Epileptic Mechanisms Shared by Alzheimer’s Disease: Viewed via the Unique Lens of Genetic Epilepsy" International Journal of Molecular Sciences 22, no. 13: 7133. https://doi.org/10.3390/ijms22137133
APA StyleKang, J.-Q. (2021). Epileptic Mechanisms Shared by Alzheimer’s Disease: Viewed via the Unique Lens of Genetic Epilepsy. International Journal of Molecular Sciences, 22(13), 7133. https://doi.org/10.3390/ijms22137133





