Novel Hybrid Compounds Containing Benzofuroxan and Aminothiazole Scaffolds: Synthesis and Evaluation of Their Anticancer Activity
Abstract
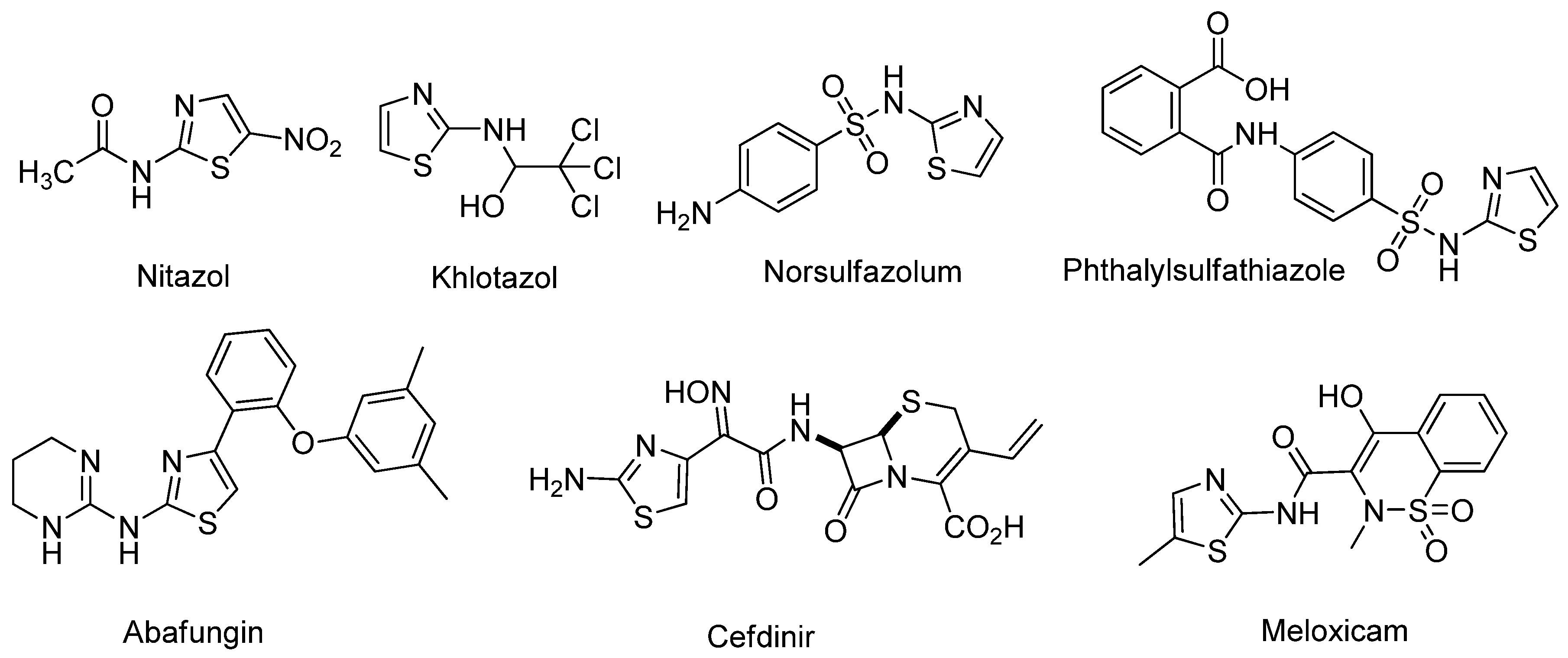
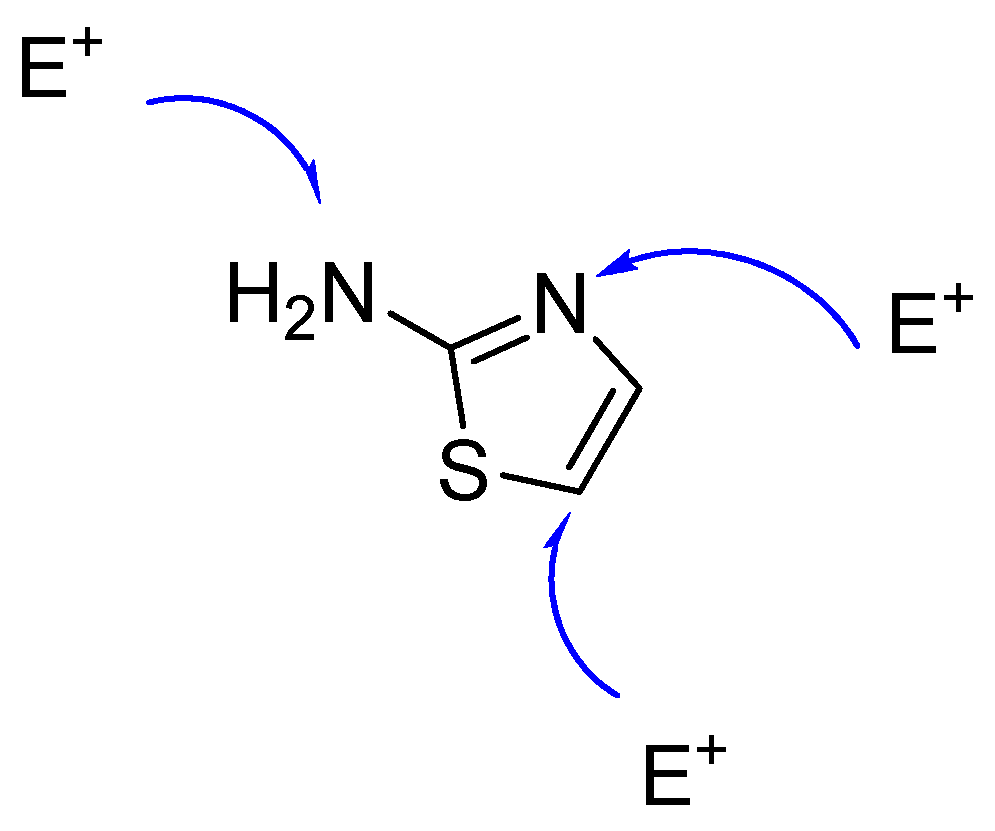
1. Introduction
2. Results and Discussion
2.1. Chemistry
2.1.1. Quantum Chemical Calculations
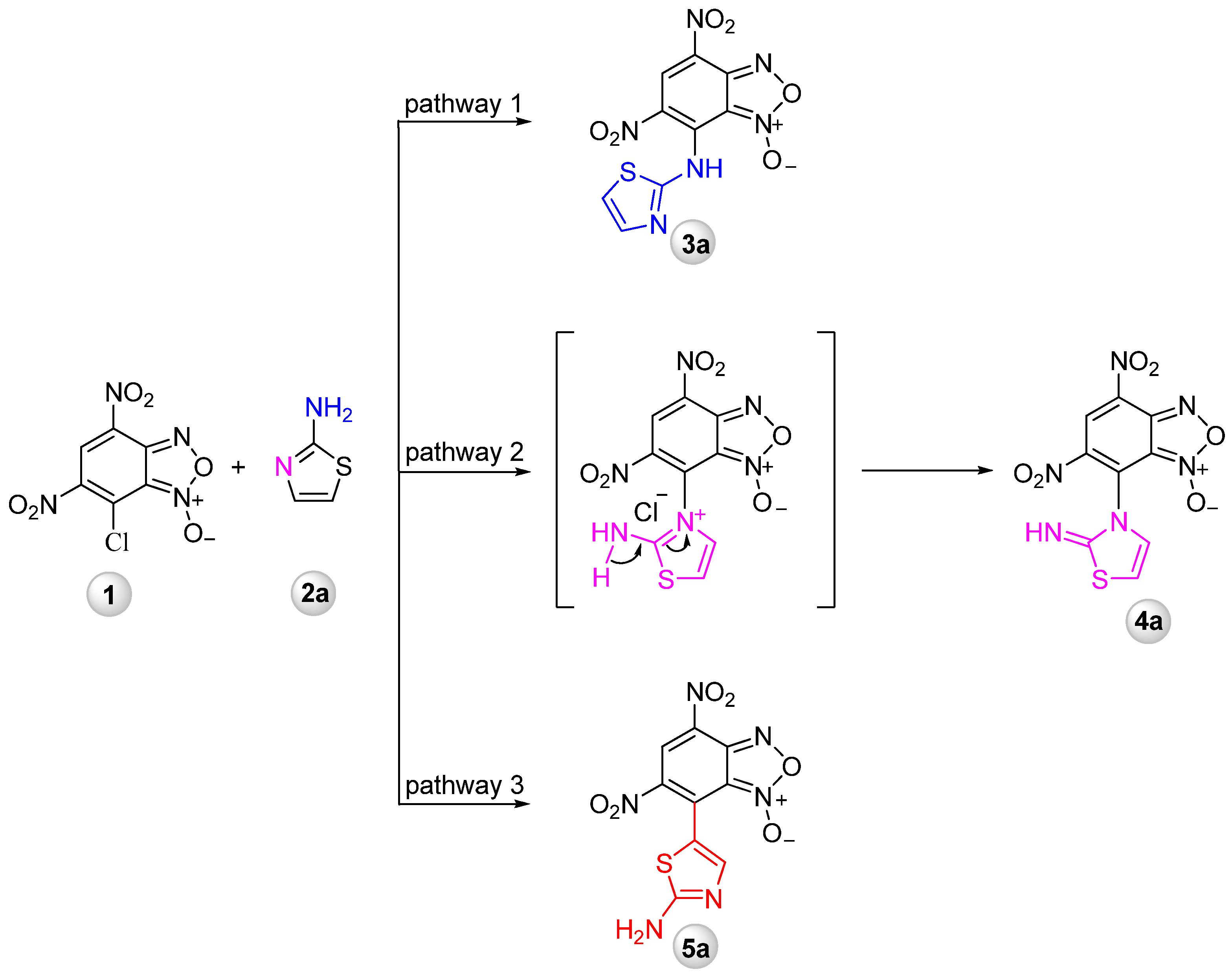
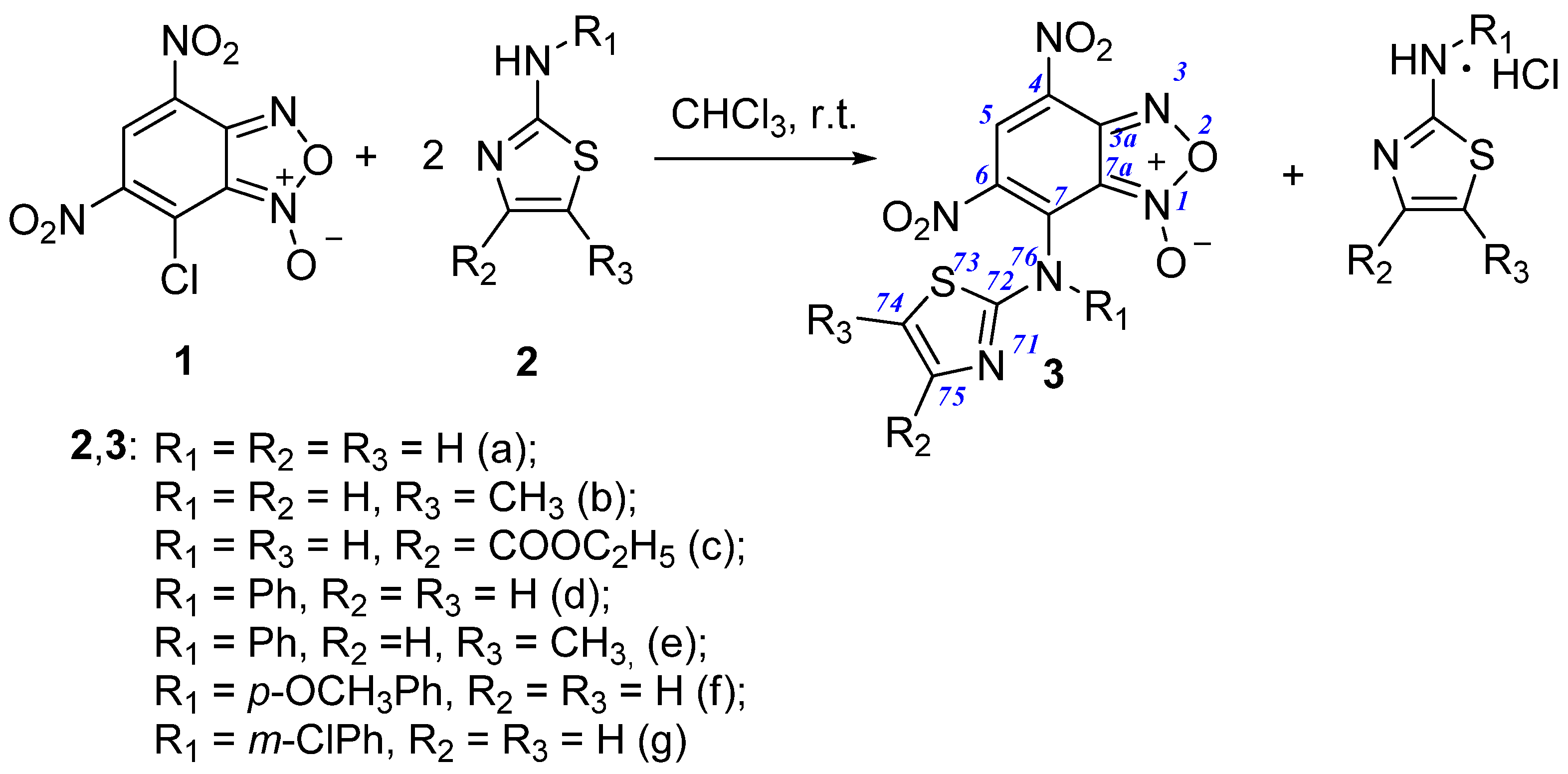
2.1.2. Synthesis
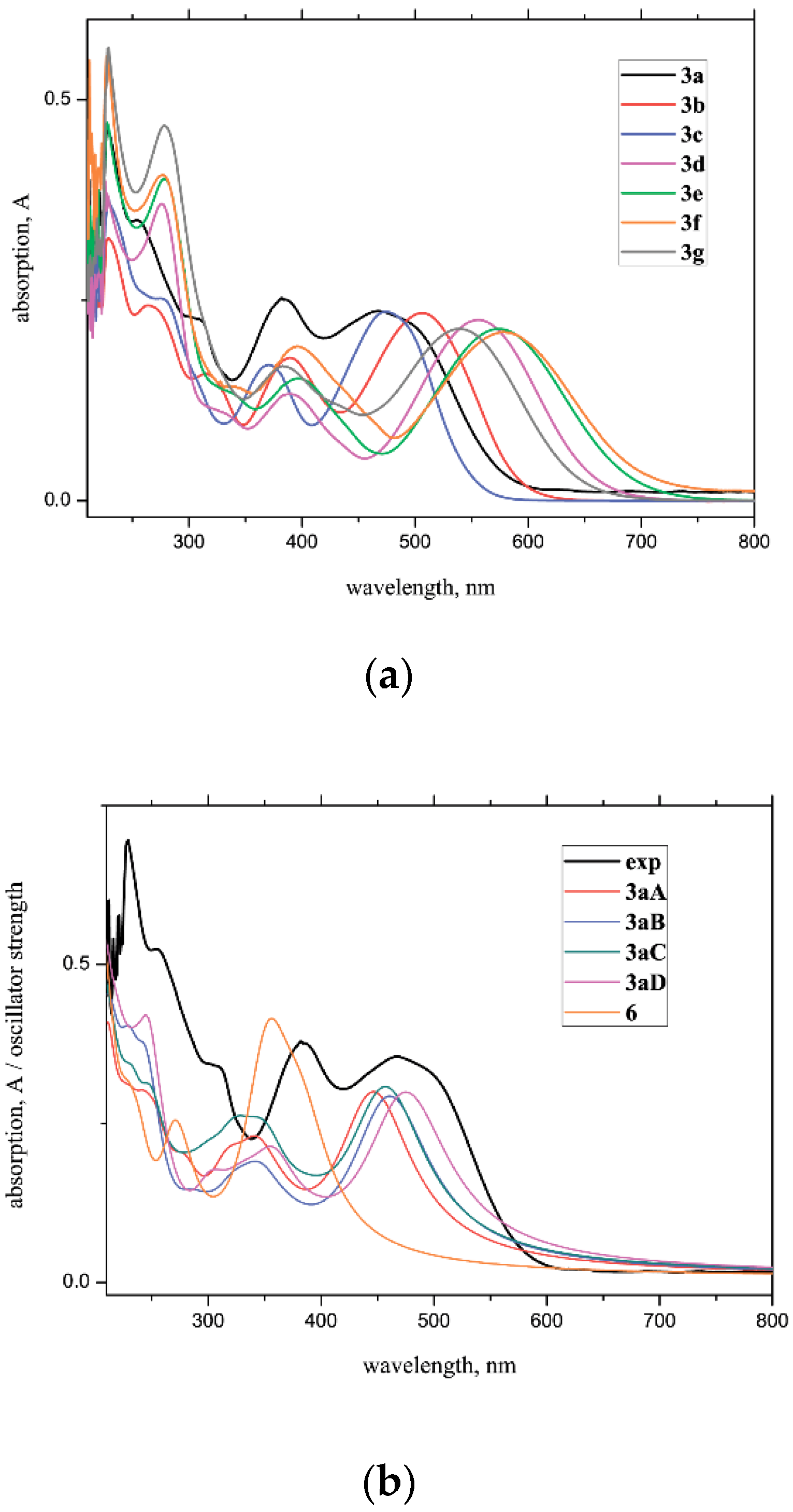
2.1.3. UV/Vis Spectral Analysis
2.2. Biological Studies
2.2.1. Cytotoxic Assay
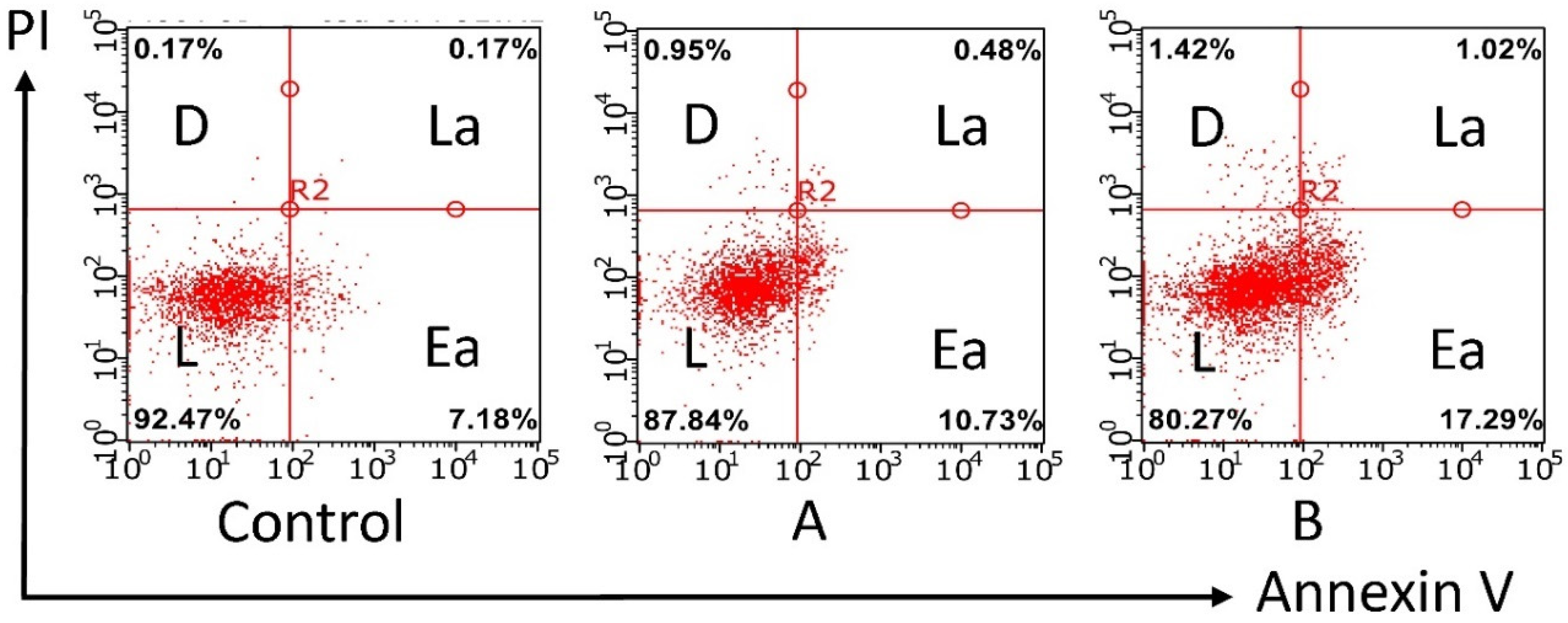
2.2.2. Induction of Apoptotic Effects by Test Compounds
2.2.3. Effects on the Mitochondrial Membrane Potential (Δψm)
2.2.4. Cell Cycle Analysis
2.2.5. Absorption, Distribution, Metabolism, Excretion, and Toxicity Data (ADMET) Predictions
3. Conclusions
4. Materials and Methods
4.1. Chemistry
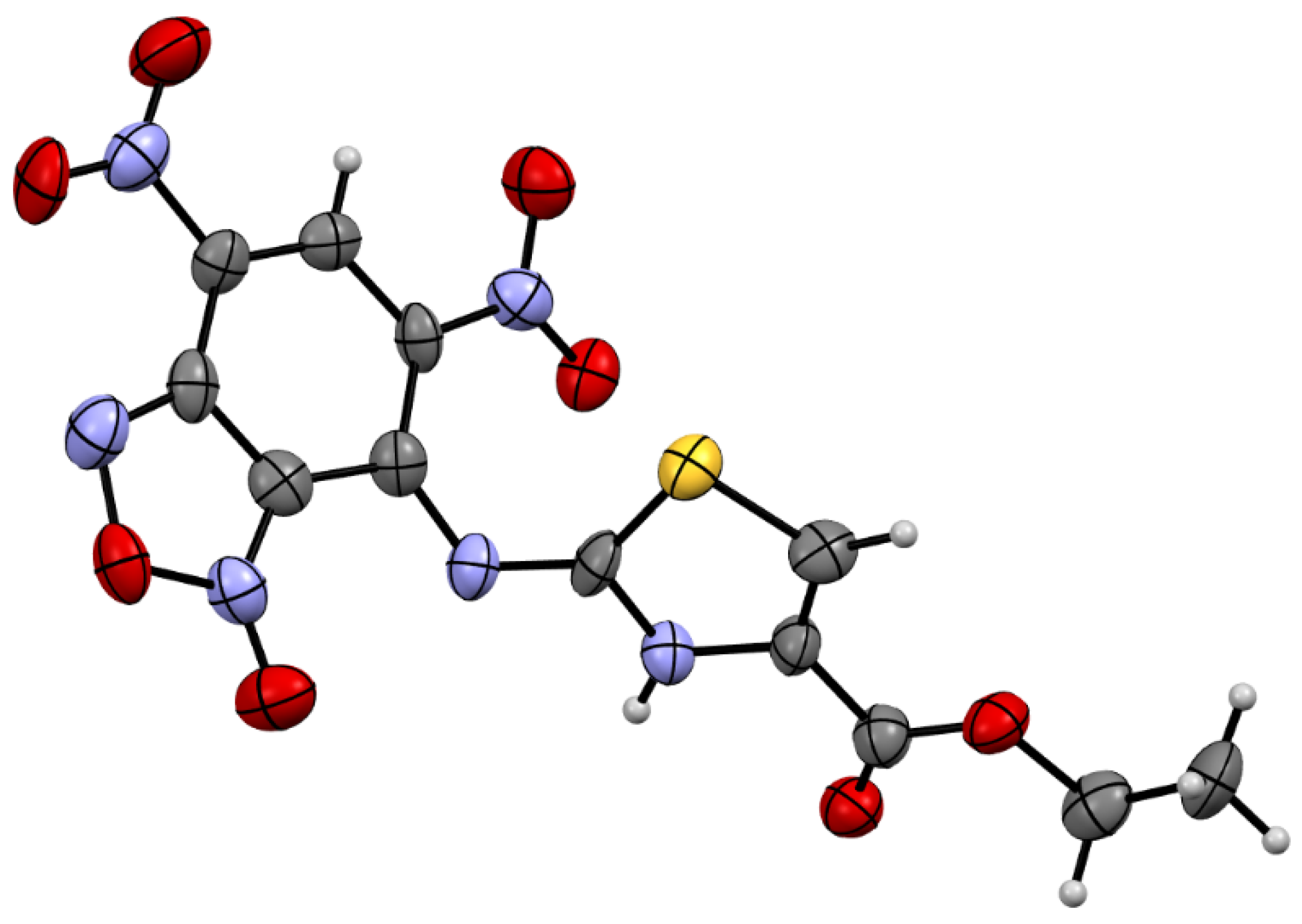
4.1.1. X-ray Crystallography Data
4.1.2. Quantum-Chemical Computations
4.2. Biological Studies
4.2.1. Cytotoxicity Assay
4.2.2. Flow Cytometry Assay
4.2.3. Mitochondrial Membrane Potential
4.2.4. Cell Cycle Analysis
4.2.5. Statistical Analysis
4.2.6. ADMET Predictions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Falzone, L.; Salomone, S.; Libra, M. Evolution of Cancer Pharmacological Treatments at the Turn of the Third Millennium. Front. Pharmacol. 2018, 9, 1300. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Ju, D.-T.; Chang, C.-F.; Muralidhar Reddy, P.; Velmurugan, B.K. A review on the effects of current chemotherapy drugs and natural agents in treating non-small cell lung cancer. BioMedicine 2017, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C.; Patel, S.K.S.; Cho, B.-K.; Wood, T.K.; Lee, J.-K. Emerging applications of bacteria as antitumor agents. Semin. Cancer Biol. 2021. [Google Scholar] [CrossRef]
- Weber, G.F. DNA Damaging Drugs. Mol. Ther. Cancer 2014, 9–112. [Google Scholar] [CrossRef]
- Dilshad, E.; Bibi, M.; Sheikh, N.A.; Tamrin, K.F.; Mansoor, Q.; Maqbool, Q.; Nawaz, M. Synthesis of Functional Silver Nanoparticles and Microparticles with Modifiers and Evaluation of Their Antimicrobial, Anticancer, and Antioxidant Activity. J. Funct. Biomater. 2020, 11, 76. [Google Scholar] [CrossRef]
- Otari, S.V.; Kumar, M.; Anwar, M.Z.; Thorat, N.D.; Patel, S.K.S.; Lee, D.; Lee, J.H.; Lee, J.-K.; Kang, Y.C.; Zhang, L. Rapid synthesis and decoration of reduced graphene oxide with gold nanoparticles by thermostable peptides for memory device and photothermal applications. Sci. Rep. 2017, 7, 10980. [Google Scholar] [CrossRef]
- Otari, S.V.; Pawar, S.H.; Patel, S.K.S.; Singh, R.K.; Kim, S.-Y.; Lee, J.H.; Zhang, L.; Lee, J.-K. Canna edulis Leaf Extract-Mediated Preparation of Stabilized Silver Nanoparticles: Characterization, Antimicrobial Activity, and Toxicity Studies. J. Microbiol. Biotechnol. 2017, 27, 731–738. [Google Scholar] [CrossRef]
- Singh, K.; Singh, S.; Taylor, J.A. Monoazo disperse dyes—Part 1: Synthesis, spectroscopic studies and technical evaluation of monoazo disperse dyes derived from 2-aminothiazoles. Dye. Pigment. 2002, 54, 189–200. [Google Scholar] [CrossRef]
- Trivette, C.D., Jr.; Morita, E.; Young, E.J. 2-Mercaptothiazole and Derivatives as Vulcanization Accelerators. Rubber Chem. Technol. 1962, 35, 1360–1429. [Google Scholar] [CrossRef]
- Khan, E.; Khan, A.; Gul, Z.; Ullah, F.; Tahir, M.N.; Khalid, M.; Asif, H.M.; Asim, S.; Braga, A.A.C. Molecular salts of terephthalic acids with 2-aminopyridine and 2-aminothiazole derivatives as potential antioxidant agents; Base-Acid-Base type architectures. J. Mol. Struct. 2020, 1200, 127126. [Google Scholar] [CrossRef]
- Venkatachalam, T.K.; Mao, C.; Uckun, F. Effect of stereochemistry on the anti-HIV activity of chiral thiourea compounds. Bioorg. Med. Chem. 2004, 12, 4275–4284. [Google Scholar] [CrossRef]
- Chugunova, E.; Boga, C.; Sazykin, I.; Cino, S.; Micheletti, G.; Mazzanti, A.; Sazykina, M.; Burilov, A.; Khmelevtsova, L.; Kostina, N. Synthesis and antimicrobial activity of novel structural hybrids of benzofuroxan and benzothiazole derivatives. Eur. J. Med. Chem. 2015, 93, 349–359. [Google Scholar] [CrossRef]
- Mjambili, F.; Njoroge, M.; Naran, K.; De Kock, C.; Smith, P.J.; Mizrahi, V.; Warner, D.; Chibale, K. Synthesis and biological evaluation of 2-aminothiazole derivatives as antimycobacterial and antiplasmodial agents. Bioorg. Med. Chem. Lett. 2014, 24, 560–564. [Google Scholar] [CrossRef]
- Wan, Y.; Long, J.; Gao, H.; Tang, Z. 2-Aminothiazole: A privileged scaffold for the discovery of anti-cancer agents. Eur. J. Med. Chem. 2021, 210, 112953. [Google Scholar] [CrossRef] [PubMed]
- Hussein, A.H.M.; Khames, A.A.; El-Adasy, A.-B.A.; Atalla, A.A.; Abdel-Rady, M.; Hassan, M.I.A.; Nemr, M.T.M.; Elshaier, Y.A.A.M. Design, synthesis and biological evaluation of new 2-aminothiazole scaffolds as phosphodiesterase type 5 regulators and COX-1/COX-2 inhibitors. RSC Adv. 2020, 10, 29723–29736. [Google Scholar] [CrossRef]
- Chugunova, E.A.; Akylbekov, N.I.; Voloshina, A.D.; Kulik, N.V.; Zobov, V.V.; Babaev, V.M.; Gavrilov, N.V.; Burilov, A.R. Synthesis and investigation of antimicrobial activity of compounds derived from benzo[c][1,2,5]oxadiazole-1-oxides and phenolates. Synth. Commun. 2016, 46, 1560–1565. [Google Scholar] [CrossRef]
- Serkov, I.V.; Chugunova, E.A.; Burilov, A.R.; Bachurin, S.O. Synthesis of amino acid derivatives of benzofuroxan. Dokl. Chem. 2013, 450, 149–151. [Google Scholar] [CrossRef]
- Chugunova, E.A.; Akylbekov, N.I.; Appazov, N.O.; Makhrus, E.M.; Burilov, A.R. Synthesis of the first tertiary ammonium derivative of 6-chloro-5-nitrobenzofuroxan. Russ. J. Org. Chem. 2016, 52, 920–921. [Google Scholar] [CrossRef]
- Boga, C.; Del Vecchio, E.; Forlani, L.; Goumont, R.; Terrier, F.; Tozzi, S. Evidence for the intermediacy of Wheland-Meisenheimer complexes in SEAr reactions of aminothiazoles with 4,6-dinitrobenzofuroxan. Chemistry 2007, 13, 9600–9607. [Google Scholar] [CrossRef] [PubMed]
- Micheletti, G.; Telese, D.; Boga, C. 4,6-Dinitro-7-(thiazol-2-ylamino)benzo[c][1,2,5]oxadiazole 1-oxide. Molbank 2020, 2020, M1165. [Google Scholar] [CrossRef]
- Forlani, L.; Boga, C.; Mazzanti, A.; Zanna, N. Trapping and Analysing Wheland–Meisenheimer σ Complexes, Usually Labile and Escaping Intermediates. Eur. J. Org. Chem. 2012, 2012, 1123–1129. [Google Scholar] [CrossRef]
- Alenzi, R.A.; El Guesmi, N.; Shaaban, M.R.; Asghar, B.H.; Farghaly, T.A. Assessing the nucleophilic character of 2-amino-4-arylthiazoles through coupling with 4,6-dinitrobenzofuroxan: Experimental and theoretical approaches based on structure-reactivity relationships. J. Saudi Chem. Soc. 2020, 24, 754–764. [Google Scholar] [CrossRef]
- El Guesmi, N.; Hussein, E.M.; Asghar, B.H.; Obaid, R.J.; Jassas, R.S.; Alharbi, A.; Altass, H.M.; Althagafi, I.I.; Morad, M.; Moussa, Z.; et al. Nucleophilicity and solvent effects on the kinetics of 4-(pyren-1-yl)thiazol-2-amine interaction with 4,6-dinitrobenzofuroxan. Arab. J. Chem. 2020, 13, 3702–3713. [Google Scholar] [CrossRef]
- Forlani, L.; De Maria, P.; Foresti, E.; Pradella, G. Reactivity of 2-aminothiazole toward 2,4-dinitrofluorobenzene. Products and structures. J. Org. Chem. 1981, 46, 3178–3181. [Google Scholar] [CrossRef]
- Chugunova, E.; Akylbekov, N.; Shakirova, L.; Dobrynin, A.; Syakaev, V.; Latypov, S.; Bukharov, S.; Burilov, A. Synthesis of hybrids of benzofuroxan and N-, S-containing sterically hindered phenols derivatives. Tautomerism. Tetrahedron 2016, 72, 6415–6420. [Google Scholar] [CrossRef]
- Micheletti, G.; Iannuzzo, L.; Calvaresi, M.; Bordoni, S.; Telese, D.; Chugunova, E.; Boga, C. Intriguing enigma of nitrobenzofuroxan’s “Sphinx”: Boulton-Katritzky rearrangement or unusual evidence of theN-1/N-3-oxide rearrangement? RSC Adv. 2020, 10. [Google Scholar] [CrossRef]
- Forlani, L.; De Maria, P. Tautomerism of aminothiazoles. pKBH+ Values of 2-aminothiazoles and of some model imines. J. Chem. Soc. Perkin Trans. 1982, 2, 535–537. [Google Scholar] [CrossRef]
- Phukan, N.; Baruah, J.B. Imine-tautomers of aminothiazole derivatives: Intriguing aspects of chemical reactivities. CrystEngComm 2016, 18, 3877–3890. [Google Scholar] [CrossRef]
- Chugunova, E.A.; Akylbekov, N.I.; Mahrous, E.M.; Voloshina, A.D.; Kulik, N.V.; Zobov, V.V.; Strelnik, A.G.; Gerasimova, T.P.; Dobrynin, A.B.; Burilov, A.R. Synthesis and study of antimicrobial activity of quaternary ammonium benzofuroxan salts. Monatshefte Fur Chemie Chem. Mon. 2018, 149, 119–126. [Google Scholar] [CrossRef]
- Chugunova, E.; Akylbekov, N.; Bulatova, A.; Gavrilov, N.; Voloshina, A.; Kulik, N.; Zobov, V.; Dobrynin, A.; Syakaev, V.; Burilov, A. Synthesis and biological evaluation of novel structural hybrids of benzofuroxan derivatives and fluoroquinolones. Eur. J. Med. Chem. 2016, 116, 165–172. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Green, D.R.; Llambi, F. Cell Death Signaling. Cold Spring Harb. Perspect. Biol. 2015, 7, a006080. [Google Scholar] [CrossRef] [PubMed]
- Garifullin, B.F.; Strobykina, I.Y.; Khabibulina, L.R.; Sapunova, A.S.; Voloshina, A.D.; Sharipova, R.R.; Khairutdinov, B.I.; Zuev, Y.F.; Kataev, V.E. Synthesis and cytotoxicity of the conjugates of diterpenoid isosteviol and N-acetyl-D-glucosamine. Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Han, J.; Kurita, Y.; Isoda, H. Genistein-induced G2/M cell cycle arrest of human intestinal colon cancer Caco-2 cells is associated with Cyclin B1 and Chk2 down-regulation. Cytotechnology 2013, 65, 973–978. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chugunova, E.A.; Voloshina, A.D.; Mukhamatdinova, R.E.; Serkov, I.V.; Proshin, A.N.; Gibadullina, E.M.; Burilov, A.R.; Kulik, N.V.; Zobov, V.V.; Krivolapov, D.B.; et al. The Study of the Biological Activity of Amino-Substituted Benzofuroxans. Lett. Drug Des. Discov. 2014, 11, 502–512. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and Computational Approaches to Estimate Solubility and Permeability in Drug Discovery and Development Settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Ghose, A.K.; Viswanadhan, V.N.; Wendoloski, J.J. A Knowledge-Based Approach in Designing Combinatorial or Medicinal Chemistry Libraries for Drug Discovery. 1. A Qualitative and Quantitative Characterization of Known Drug Databases. J. Comb. Chem. 1999, 1, 55–68. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. A. 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Macrae, C.F.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Shields, G.P.; Taylor, R.; Towler, M.; van de Streek, J. Mercury: Visualization and analysis of crystal structures. J. Appl. Crystallogr. 2006, 39, 453–457. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision B.01; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Becke, A.D. Density Functional Thermochemistry III The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Hehre, W.J.; Ditchfield, R.; Pople, J.A. Self—Consistent Molecular Orbital Methods. XII. Further Extensions of Gaussian—Type Basis Sets for Use in Molecular Orbital Studies of Organic Molecules. J. Chem. Phys. 1972, 56, 2257–2261. [Google Scholar] [CrossRef]
- Ditchfield, R.; Hehre, W.J.; Pople, J.A. Self-consistent molecular-orbital methods. IX. An extended gaussian-type basis for molecular-orbital studies of organic molecules. J. Chem. Phys. 1971, 54, 724. [Google Scholar] [CrossRef]
- Petersson, G.A.; Bennett, A.; Tensfeldt, T.G.; Al-Laham, M.A.; Shirley, W.A.; Mantzaris, J. A complete basis set model chemistry. I. The total energies of closed-shell atoms and hydrides of the first-row elements. J. Chem. Phys. 1988. [Google Scholar] [CrossRef]
- Norris, W.P.; Chafin, A.; Spear, R.J.; Read, R.W. Synthesis and thermal rearrangement of 5-chloro-4,6-dinitrobenzofuroxan. Heterocycles 1984, 22, 271–274. [Google Scholar] [CrossRef]
- Smolobochkin, A.; Gazizov, A.; Sazykina, M.; Akylbekov, N.; Chugunova, E.; Sazykin, I.; Gildebrant, A.; Voronina, J.; Burilov, A.; Karchava, S.; et al. Synthesis of Novel 2-(Het)arylpyrrolidine Derivatives and Evaluation of Their Anticancer and Anti-Biofilm Activity. Molecules 2019, 24, 3086. [Google Scholar] [CrossRef]
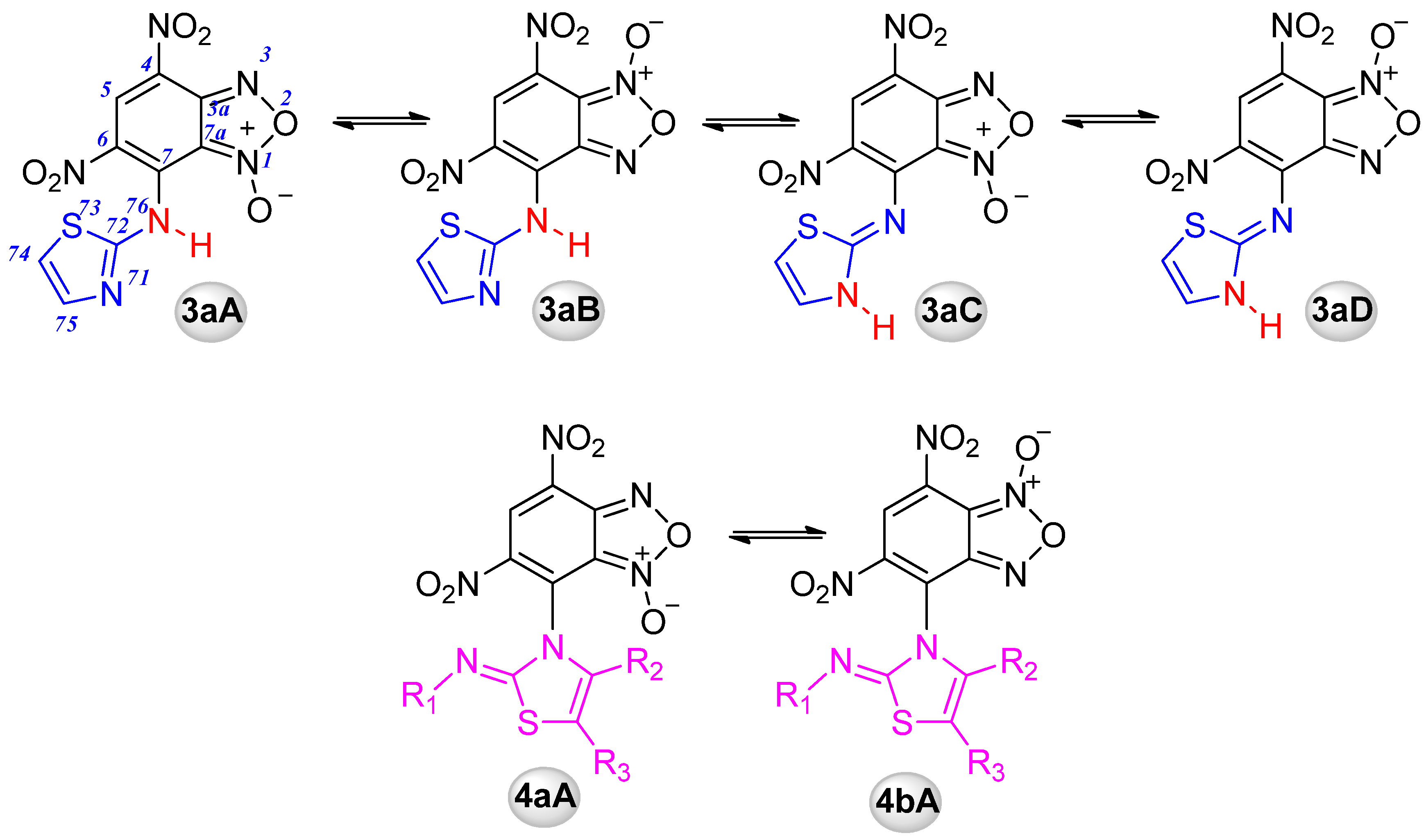




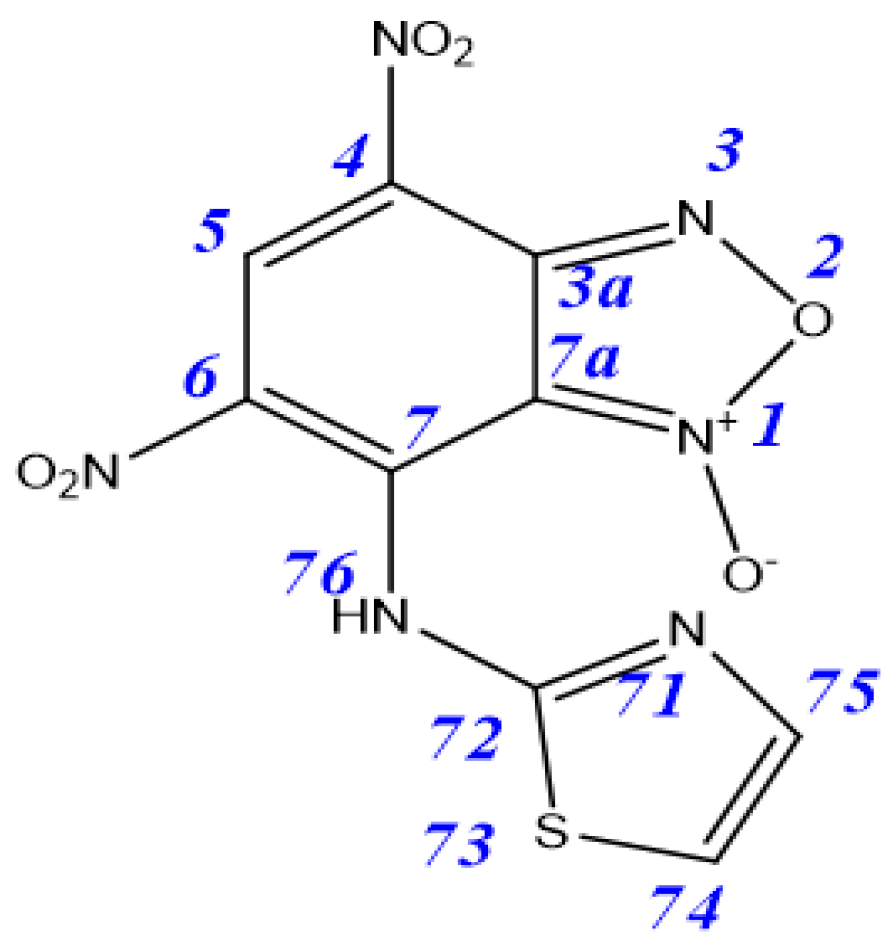
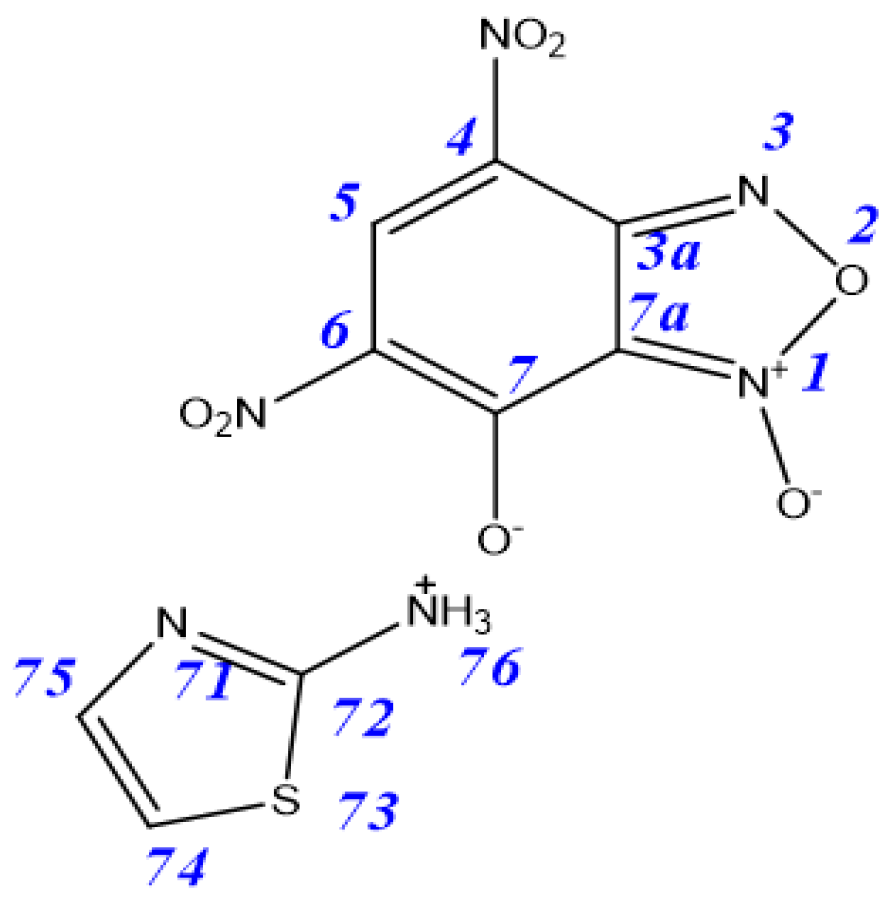
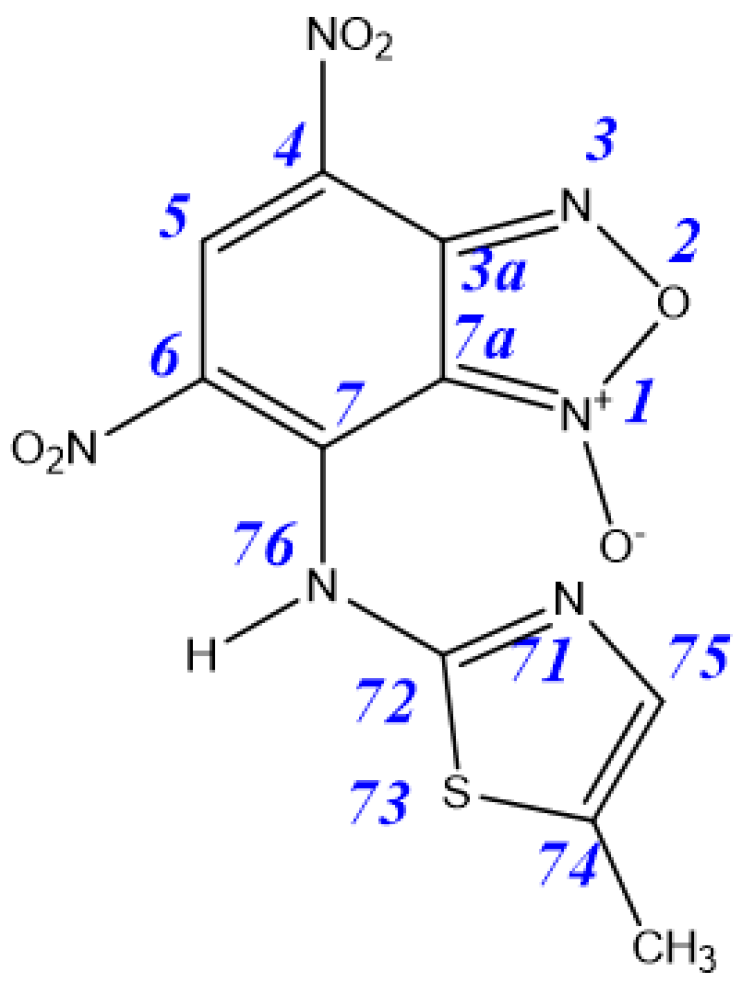
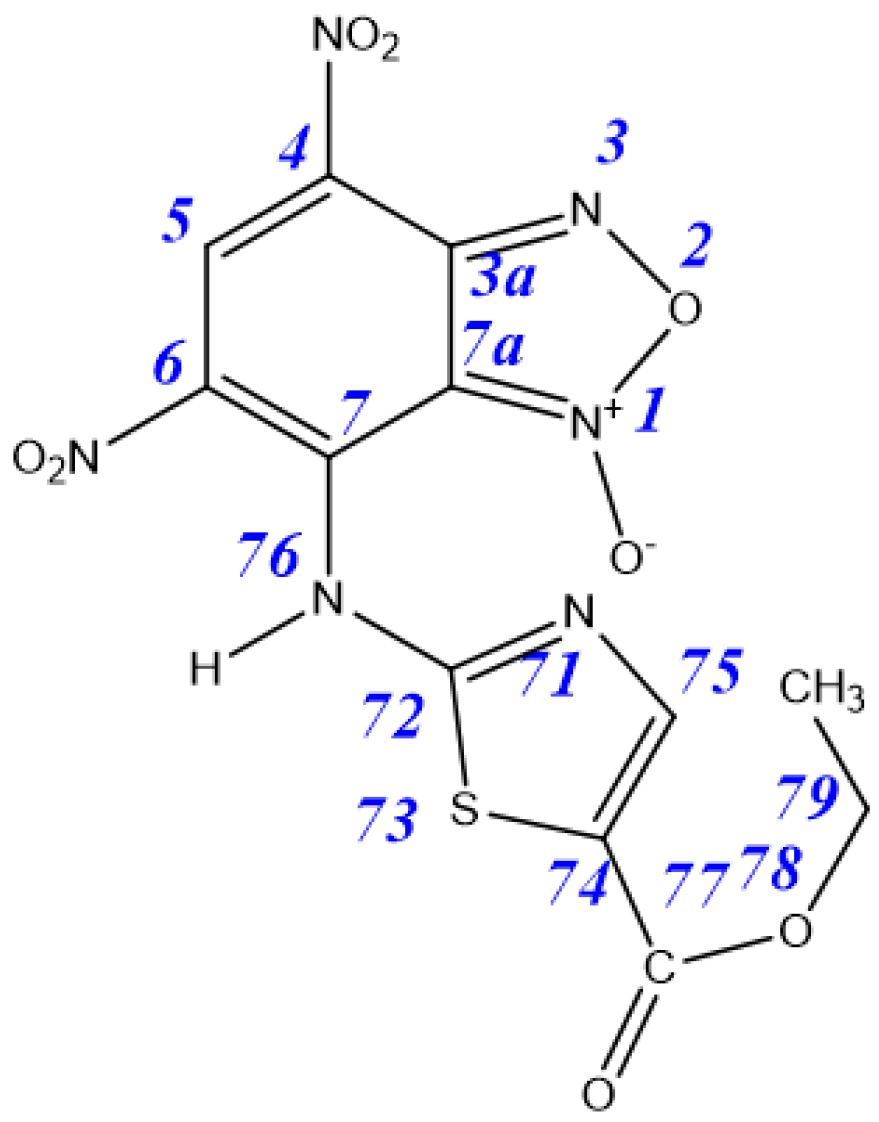
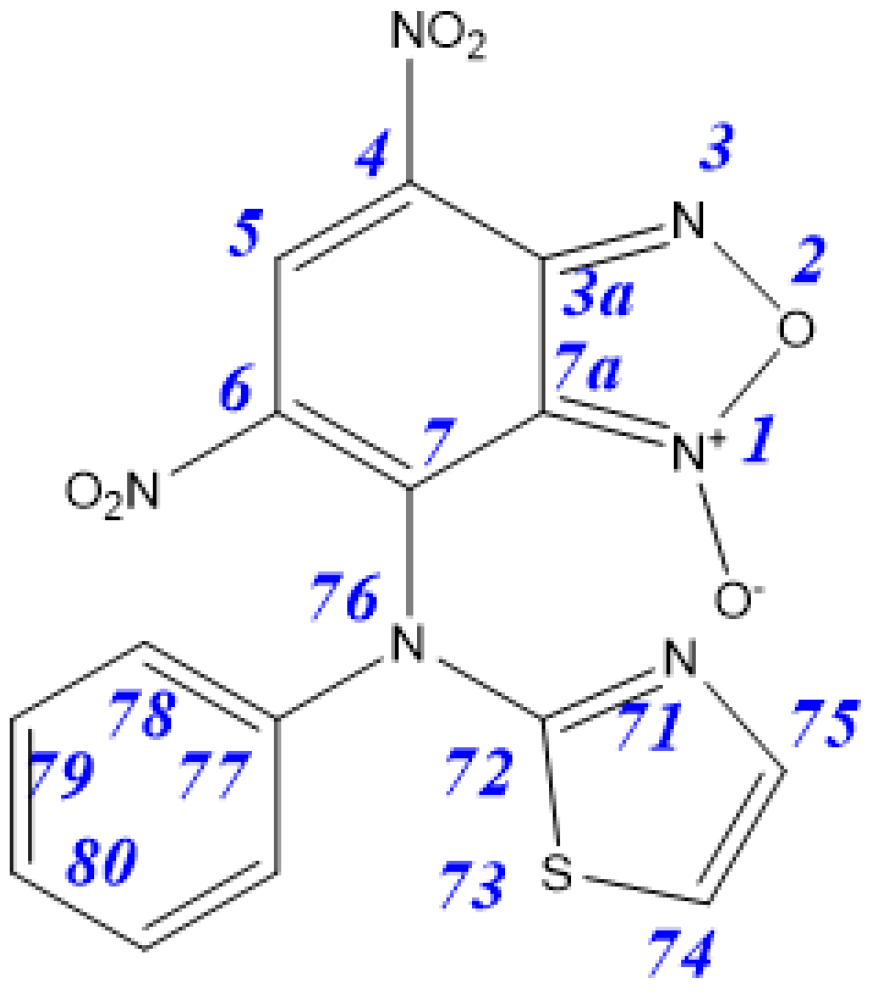
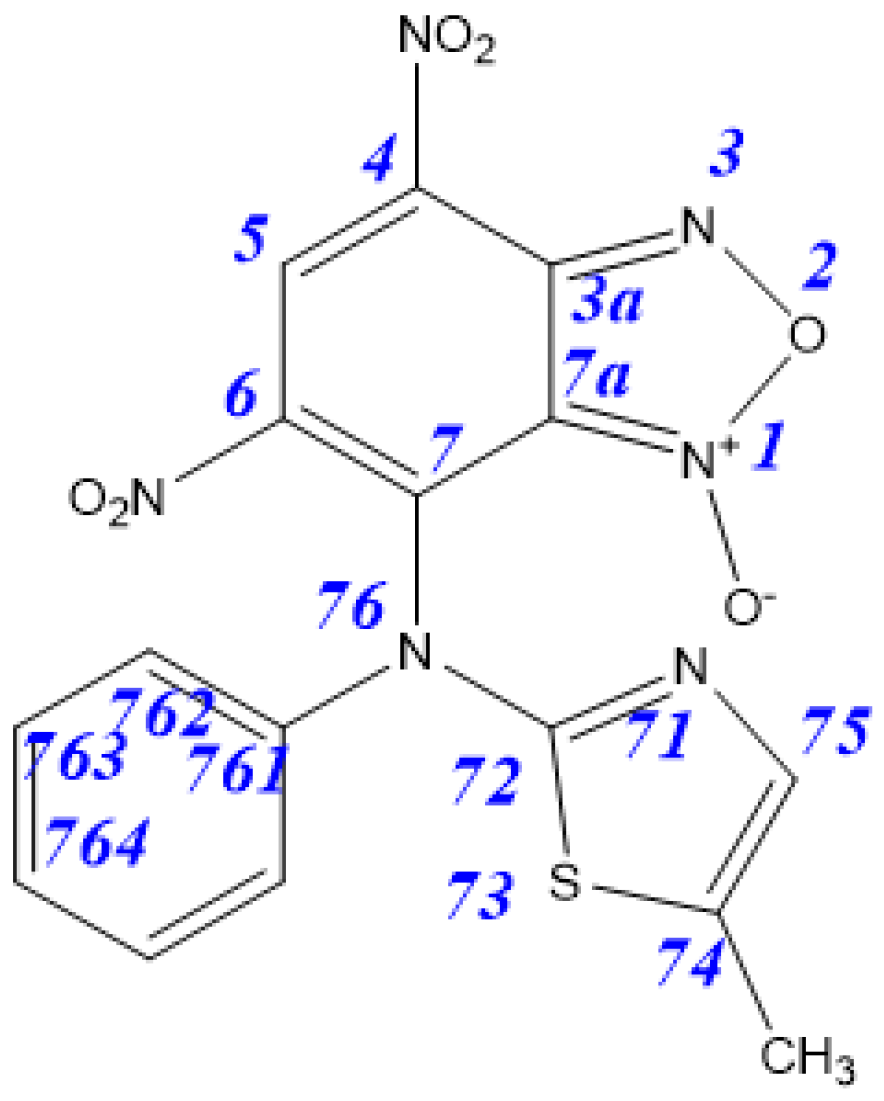

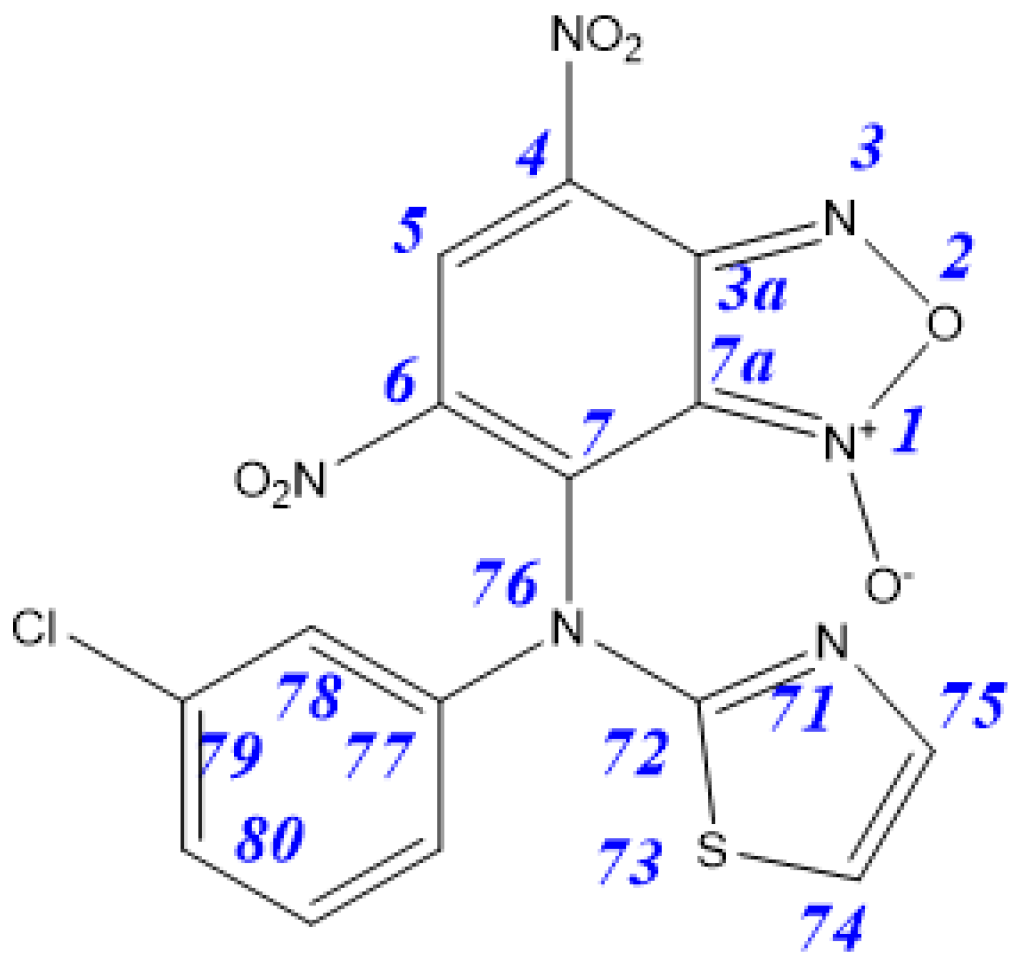
| Tautomer | 3aA | 3aB | 3aC | 3aD | 4aA | 4aB |
|---|---|---|---|---|---|---|
| ΔE | 0.9 | 4.6 | 0 | 3.9 | 14.6 | 16.3 |
| 3a | 3b | 3c | 3d | 3e | 3f | 3g | |
|---|---|---|---|---|---|---|---|
| Experiment | 467/494 | 507 | 475 | 556 | 574 | 581 | 540 |
| Computations | 457 | 461 | 456 | 558 | 570 | 597 | 549 |
| Test Compound | Cancer Cell Line | Normal Cell Line | ||
|---|---|---|---|---|
| M-HeLa | HuTu 80 | PANC-1 | Chang Liver | |
| 1 | 94.1 ± 8.6 | >100 | >100 | >100 |
| 2a | 91 ± 8.3 | >100 | >100 | >100 |
| 3a | 61.0 ± 5.4 | >100 | >100 | >100 |
| 2b | 92.0 ± 7.9 | >100 | >100 | >100 |
| 3b | 70.0 ± 6.2 | >100 | >100 | >100 |
| 2c | 63.3 ± 5.7 | >100 | >100 | 95.4 ± 8.7 |
| 3c | 95.8 ± 8.4 | >100 | >100 | 100 ± 9.4 |
| 3d | 61.0 ± 5.6 | >100 | >100 | 86.1 ± 7.8 |
| 2e | 92 ± 8.2 | >100 | >100 | >100 |
| 3e | 79 ± 6.3 | >100 | >100 | 85.8 ±7.4 |
| 2f | 93.8 ± 8.4 | >100 | >100 | >100 |
| 3f | 56.6 ± 4.7 | >100 | >100 | >100 |
| Tamoxifen | 28.0 ± 2.5 | - | - | 46.2 ± 3.5 |
| 5-Fluorouracil | 62.0 ± 4.9 | 65.2 ± 5.5 | 68.3 ± 5.7 | 72.4 ± 6.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chugunova, E.; Micheletti, G.; Telese, D.; Boga, C.; Islamov, D.; Usachev, K.; Burilov, A.; Tulesinova, A.; Voloshina, A.; Lyubina, A.; et al. Novel Hybrid Compounds Containing Benzofuroxan and Aminothiazole Scaffolds: Synthesis and Evaluation of Their Anticancer Activity. Int. J. Mol. Sci. 2021, 22, 7497. https://doi.org/10.3390/ijms22147497
Chugunova E, Micheletti G, Telese D, Boga C, Islamov D, Usachev K, Burilov A, Tulesinova A, Voloshina A, Lyubina A, et al. Novel Hybrid Compounds Containing Benzofuroxan and Aminothiazole Scaffolds: Synthesis and Evaluation of Their Anticancer Activity. International Journal of Molecular Sciences. 2021; 22(14):7497. https://doi.org/10.3390/ijms22147497
Chicago/Turabian StyleChugunova, Elena, Gabriele Micheletti, Dario Telese, Carla Boga, Daut Islamov, Konstantin Usachev, Alexander Burilov, Alena Tulesinova, Alexandra Voloshina, Anna Lyubina, and et al. 2021. "Novel Hybrid Compounds Containing Benzofuroxan and Aminothiazole Scaffolds: Synthesis and Evaluation of Their Anticancer Activity" International Journal of Molecular Sciences 22, no. 14: 7497. https://doi.org/10.3390/ijms22147497
APA StyleChugunova, E., Micheletti, G., Telese, D., Boga, C., Islamov, D., Usachev, K., Burilov, A., Tulesinova, A., Voloshina, A., Lyubina, A., Amerhanova, S., Gerasimova, T., Gilfanova, A., & Syakaev, V. (2021). Novel Hybrid Compounds Containing Benzofuroxan and Aminothiazole Scaffolds: Synthesis and Evaluation of Their Anticancer Activity. International Journal of Molecular Sciences, 22(14), 7497. https://doi.org/10.3390/ijms22147497








