Abstract
Medical staff represent the largest group of workers occupationally exposed to ionizing radiation (IR). Chronic exposure to low-dose IR may result in DNA damage and genotoxicity associated with increased risk of cancer. This review aims to identify the genotoxicity biomarkers that are the most elevated in IR-exposed vs. unexposed health workers. A systematic review of the literature was performed to retrieve relevant studies with various biomarkers of genotoxicity. Subsequent meta-analyses produced a pooled effect size for several endpoints. The search procedure yielded 65 studies. Chromosome aberrations (CA) and micronuclei (MN) frequencies were significantly different between IR-exposed and unexposed workers (θpooled = 3.19, 95% CI 1.46–4.93; and θpooled = 1.41, 95% CI 0.97–1.86, for total aberrant cells and MN frequencies, respectively), which was not the case for ring chromosomes and nucleoplasmic bridges. Although less frequently used, stable translocations, sister chromatid exchanges (SCE) and comet assay endpoints were also statistically different between IR-exposed and unexposed workers. This review confirms the relevance of CA and MN as genotoxicity biomarkers that are consistently elevated in IR-exposed vs. unexposed workers. Other endpoints are strong candidates but require further studies to validate their usefulness. The integration of the identified biomarkers in future prospective epidemiological studies is encouraged.
1. Introduction
Exposure of humans to ionizing radiation (IR) during medical procedures is the greatest contributor to annual radiation exposure dose from all artificial IR sources. Diagnostic and therapeutic use of IR has substantially increased in the last decades, exemplified by a range of new imaging techniques [1,2] and new targeted irradiation therapeutic modalities [3]. Accordingly, medical workers comprise the largest professional human group that are exposed to occupational IR at low doses and low dose-rates (7.35 million worldwide, representing 75% of workers exposed to artificial sources of radiation) [4]. Exposure to moderate-to-high doses of IR induces genotoxic effects that can lead to carcinogenesis [5]. However, whether such detrimental effects can be produced after exposure to low-dose IR is still debated [6]. To this end, the value of biomarkers has been recognized as a complementary tool to conventional epidemiology that can facilitate understanding the health effects of low-dose IR exposures [7]. They may fill important gaps in the understanding of the biological mechanisms that link IR exposure and disease. IR induces many types of deoxyribonucleic acid (DNA) lesions, of which DNA double-strand breaks (DSB) are recognized as the ones that have the greatest role in radiation-induced genomic instability and subsequently carcinogenesis [8,9]. Unrepaired or mis-repaired DSB can lead to the formation of chromosome aberrations (CA), a broad class of DNA mutations that are linked to various health risks [10]. Increased rates of CA in peripheral blood lymphocytes (PBL) have been associated with an increased risk of cancer [11,12,13]. CA can therefore be considered as potential bioindicators of cancer risk.
Numerous types of CA can be broadly categorized into stable (e.g., inversions and translocations) and unstable (e.g., acentric fragments, dicentrics, and rings) aberrations. The former are non-lethal for cells and can persist for years, whereas the latter cause cell death during mitosis, thus are considered short lived genotoxic events that decline with time after the triggering insult [10]. Detection and quantification of unstable CA is simple and employed shortly after IR exposure, e.g., for biodosimetry [14]. Persistence of stable CA allows for the evaluation of the long-term effects of single IR exposures or the cumulative effects of protracted chronic exposures [15]. However, the detection of this type of CA requires more elaborated and costly techniques, such as fluorescence in situ hybridization (FISH). Sister chromatid exchanges (SCE) are reciprocal exchange of segments between two identical sister chromatids, resulting from damaged DNA and involving several mechanisms during the S-phase. SCE are frequently reported as a marker of spontaneous and induced chromosomal instability in cells [16]. Although acute IR exposure is not efficient in producing SCE [17], increased SCE levels were seen in populations chronically exposed to IR [18]. Micronuclei (MN) originate from chromosome fragments or whole chromosomes that, upon nuclear division, lag behind the anaphase and are not properly segregated into daughter cell nuclei. Therefore, they represent chromosomal instability and can also serve as an indicator of chromosomal damage induced by IR [19]. Additionally, MN frequency was shown to be strongly correlated with the length of telomeres (the terminal structures of linear chromosomes), whose role is to protect chromosomes and participate in the integrity of the genetic heritage [20]. Interestingly, recent studies show that IR exposure causes telomere length shortening in Chernobyl accident recovery workers that could potentiate carcinogenesis [21]. Thus, the measurement of telomere lengths in people exposed to IR bears a potential as a biomarker of the risk of cancer and other age-related diseases [22]. Molecular damage to DNA, such as single-strand breaks (SSB) and DSB, can also be measured using various assays such as the comet assay for SSB and the detection of immunofluorescently labeled phosphorylated H2AX (designated as γH2AX) foci.
Although there exist a great number of studies examining genotoxicity biomarkers in IR-exposed medical professionals, results are often inconsistent or conflicting [23,24]. These studies often employ small size human cohorts causing low statistical power to discriminate IR-exposed and unexposed workers and discrepancies in results. This provides motivation for conducting a quantitative meta-analysis of the published results in an attempt to summarize and analyze the available knowledge and to reveal the most sensitive and reliable biomarkers. Therefore, the objective of the present work was to carry out a systematic review and meta-analysis to identify the type(s) of genotoxicity biomarkers that are most elevated in IR-exposed compared to unexposed medical workers. These biomarkers, as most sensitive to IR exposure, would then provide a possibility for future focused prospective epidemiological studies to examine the association between these biomarkers and long-term health outcomes, primarily cancer.
2. Methods
These systematic review and meta-analyses have been made according the PRISMA guidelines, as a basis for reporting systematic reviews. Registration has been recorded on the PROSPERO database (ID CRD42020182636).
2.1. Online Searches
The literature search was conducted in the PubMed, Google Scholar, Scopus and Web of Science databases, in April 2020. The following query has been used: (((ionizing radiation) AND medical workers) AND (chromosome OR biomarker OR genetic OR DNA)). Relevant publications and international reports such as BEIR VII and UNSCEAR 2006 were also screened for additional references. Duplicates from the different databases were removed. Based on the results of this research, a first selection was conducted by two independent reviewers (MGA and CB) who reviewed all titles and abstracts according to predefined selection criteria (see below), and disagreements were resolved by a third reviewer (MOB). A second selection was conducted based on full-text screening. For all articles eventually included in the review, relevant information was collected including study title, first author, journal, year of publication, study design, population, inclusion and exclusion criteria, sample size, objectives, and endpoints. The systematic literature review was then supplemented by meta-analyses.
2.2. Selection Criteria
To be eligible, studies had to be observational (longitudinal/cohort, case-control or cross-sectional), in English, and published between 1 January 2000 and 31 March 2020, The rational for the use of this time range was the marked growth of the nuclear medicine domain and the development of new and the refinement of old genotoxicity biomarkers and their detection. Studies with 15 participants or less in one of the exposed/unexposed groups were excluded. The meta-analyses included only the studies with common and complete quantitative information (measurement data of the evaluated endpoint such as frequencies, length or score, for both IR-exposed and unexposed workers groups) where only studies with exposed/unexposed design were considered.
Selection criteria for relevant studies have been made following the PECO statements (Table 1).

Table 1.
Selection criteria (PECO statements).
The target population (P) were all medical radiation workers potentially exposed to IR: radiological technologists, radiologists, interventional cardiologists, nurses and laboratory technicians.
The exposure (E) comprised IR from all medical sources (external irradiation or internal contamination). When available, dose estimates were reported either as absorbed doses to organs (mGy) or effective doses (mSv). Otherwise, surrogates were also considered (occupational radiological risk score (ORRS), index of cumulative radiological score, number of years at exposed work, reconstruction of the lifetime cumulative professional exposure, etc.).
Comparisons (C) between exposed and unexposed workers, or between different categories of exposure were studied.
For the outcomes (O), this review focused on biomarkers in peripheral blood lymphocytes: cytogenetic biomarkers such as CA (stable and unstable: chromosome and/or chromatid breaks, inversions, insertions, deletion, gaps, aneuploidy, dicentrics, acentric fragments, rings, and translocations), MN, nucleoplasmic bridges (NPB), SCE, or premature centromere divisions (PCD), and markers of DNA integrity such as mutated gene frequencies, γH2AX foci, comet tail length/moment, telomere length (TL), and DNA strand breaks. As most of the CA studies presented percent or fraction of aberrant cells or number of cells with at least one CA, the total of aberrant cells has also been analyzed making it possible to pool results from studies using different CA types for the purpose of meta-analysis. Studies involving oxidative stress and inflammation biomarkers, apoptosis, cell cycle, and gene expression biomarkers, or genetic susceptibility biomarkers (influence of SNPs on biomarkers of effect or cancer risk) were excluded.
2.3. Quality Assessment
To assess the quality of the included studies, the Newcastle–Ottawa Scale was used, which is the tool most commonly used nowadays for observational studies. The assessment is based on eight items categorized into three groups: selection of study groups, comparability of groups and determination of exposure or outcome of interest for case-control or cohort/cross-sectional studies. A study can be assigned a maximum of one star for each of the eight items (up to two stars for the comparability group item). A final score between 0 and 9 is obtained by adding up all the stars. Studies with a score <3 were excluded.
2.4. Statistical Analysis
Since most studies presented continuous variables for biomarkers (frequencies, comet tail or telomere length, etc.) in both IR-exposed and unexposed workers, we computed Hedges’s g [25] standardized mean differences for individual studies. Briefly, Hedges’s g is defined as the unbiased difference between two means (m1 and m2) divided by a pooled weighted standard deviation (s* calculated from standard deviations s1 and s2) for two populations to be compared (population sizes n1 and n2):
with .
Then, standardized mean differences were pooled together to generate and plot an overall effect size using the DerSimonian–Laird random-effect method, involving the assumption that the effects estimated in the different studies are not equal. A Z-test was used to assess the null hypothesis whereby the overall effect size would not be significantly different from 0. The between-study heterogeneity was reported using the Cochrane’s homogeneity test (Q) and the I2 statistic which allows to quantify the proportion of the total variation due to that heterogeneity [26]. Finally, small-study effects and publication bias were visually and numerically explored using Egger’s test.
Data were analyzed using Stata 16 software (StataCorp. 2019. Stata Statistical Software: Release 16. StataCorp LLC: College Station, TX, USA) using the meta command. Statistical significance was defined by p < 0.05.
3. Results
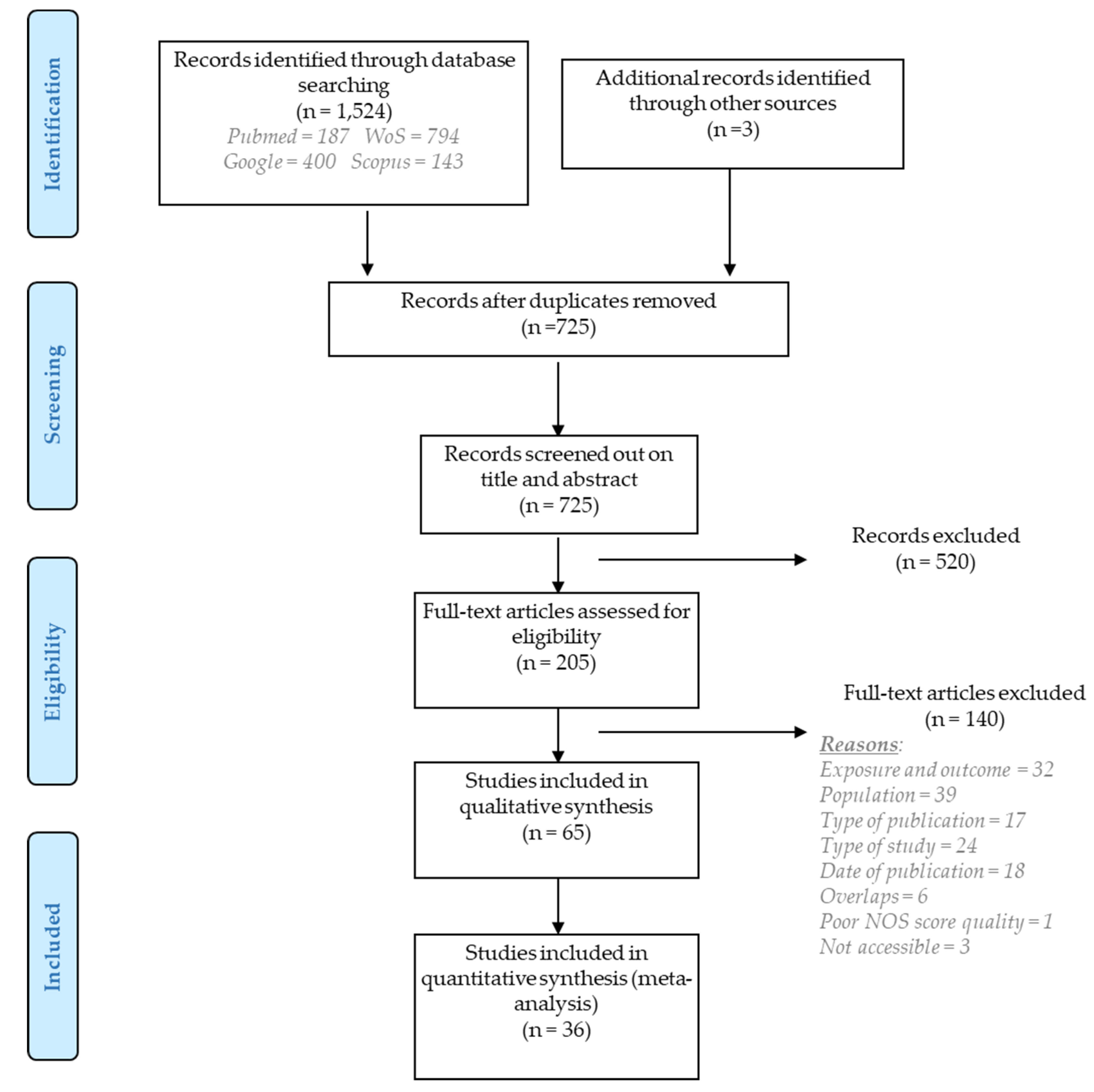
The systematic search produced 1524 records. Three additional records were identified from reference screening in relevant papers or reports. After removing duplicates, 725 titles and abstracts were screened using PECO, and eventually 205 articles were selected for a full-text analysis. Exclusion of 134 articles based on full-text screening resulted in 65 studies suitable for inclusion in the qualitative synthesis. Of those 65, 36 articles contained sufficient information and data to be included in a quantitative meta-analysis (Figure 1).
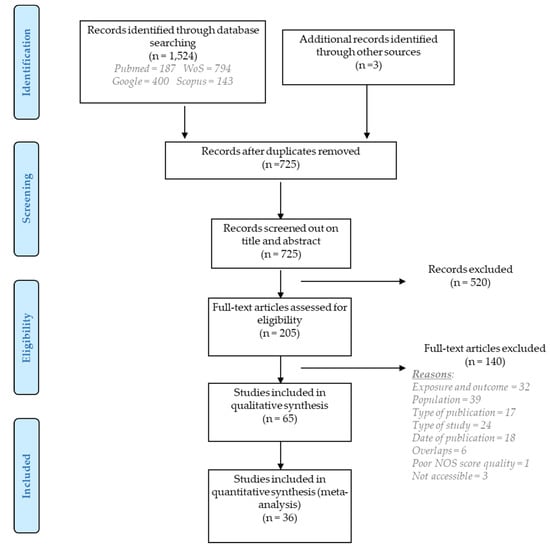
Figure 1.
PRISMA flow diagram for the results of literature search, screening, and selection of relevant studies.
Reasons for exclusion after full-text evaluation included: exposure and outcome did not meet the PECO criteria (n = 32), the population exposed to IR did not involve medical workers or was <15 in number (n = 39), some publications were books/conference proceedings/systematic reviews (n = 17), the type of study was experimental (n = 24), the NOS quality assessment was <3 (n = 1), the articles were published before 2000 (n = 18) or were not accessible (n = 3) or overlapped with others (n = 6). For the purpose of maintaining structured presentation of results, the various endpoints were grouped into two large categories: cytogenetic and DNA integrity biomarkers.
3.1. Cytogenetic Biomarkers
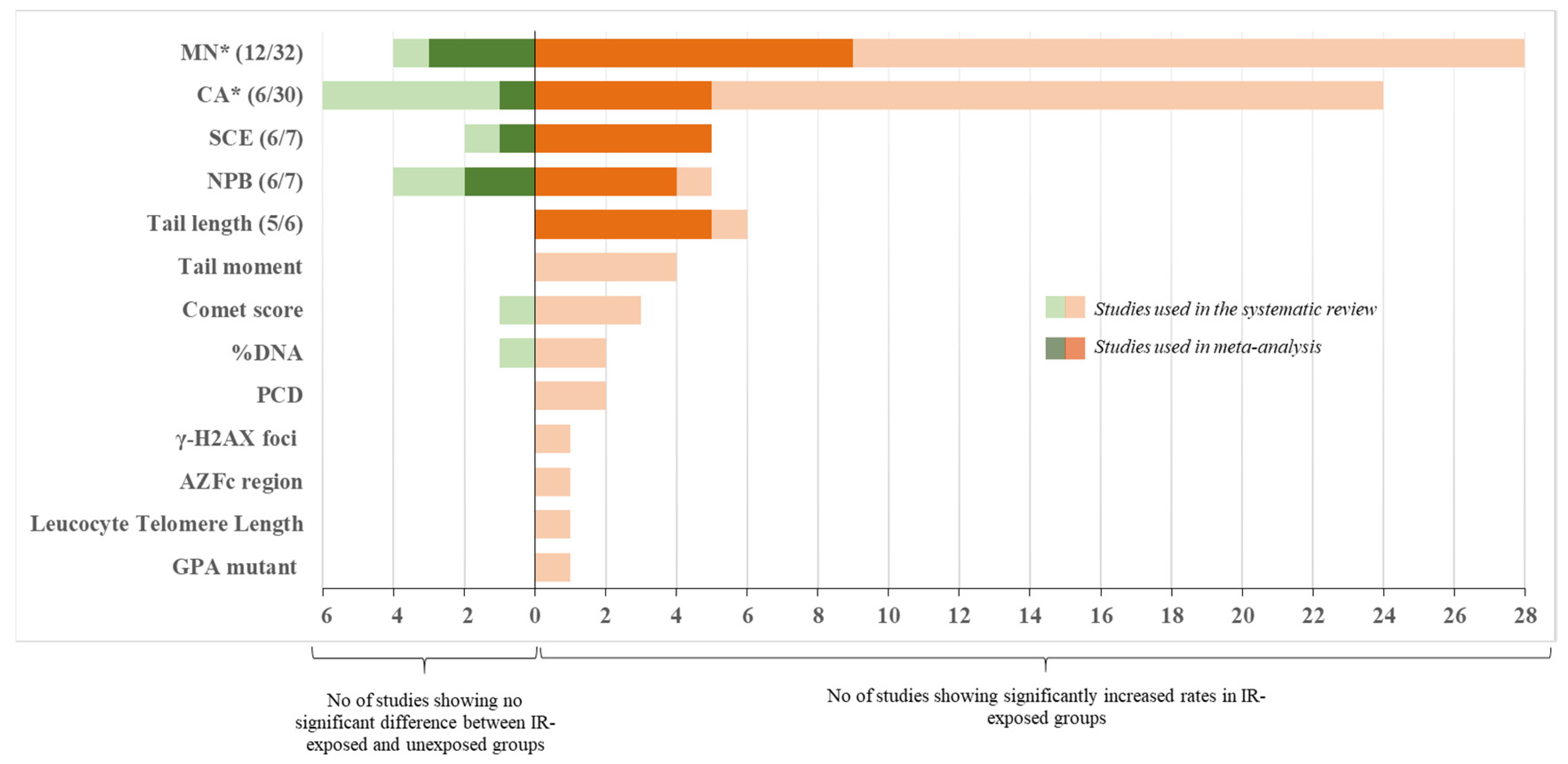
Fifty-three out of the 65 studies included in the systematic review investigated cytogenetic endpoints to examine differences between IR-exposed and non-exposed professionals. CA frequencies was the focus of 30 studies, including at least dicentrics, acentric fragments, and/or rings for 24, 14, and 14 studies respectively. Out of the 65 studies included in the present systematic review, MN frequencies, NPB, SCE and PCD were the focus of 32, 7, 7, and 2 studies respectively.
Overall, most of the studies reported significantly higher frequencies of CA frequencies in IR-exposed compared to unexposed workers (Table 2, Figure 2).

Table 2.
List and characteristics of the included studies.
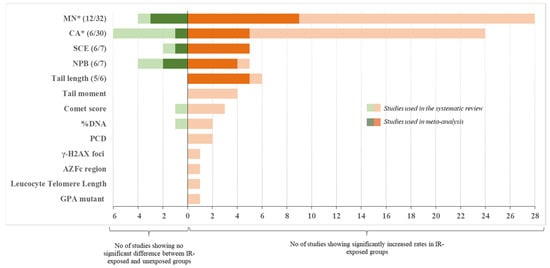
Figure 2.
Summary chart of the included studies. Shown are numbers of studies broken by endpoint. Studies that reported significant increase in endpoints in IR-exposed groups are plotted to the right of the X = 0, whereas studies showing no difference are plotted to the left. Studies used in meta-analysis are shown in dark color and their numbers are also shown in the brackets (e.g., 6/7 means 6 out of 7 total studies qualified and were used in meta-analysis). * Correlation of the endpoint with other confounding factors (alcohol consumption, smoking) were reported (see text for detail). AZFc: azoospermia factor region c; CA: chromosome aberration; GPA: glycophorin A; MN: micronucleus; NPB: nucleoplasmic bridge; PCD: premature centromere divisions; SCE: sister chromatid exchange.
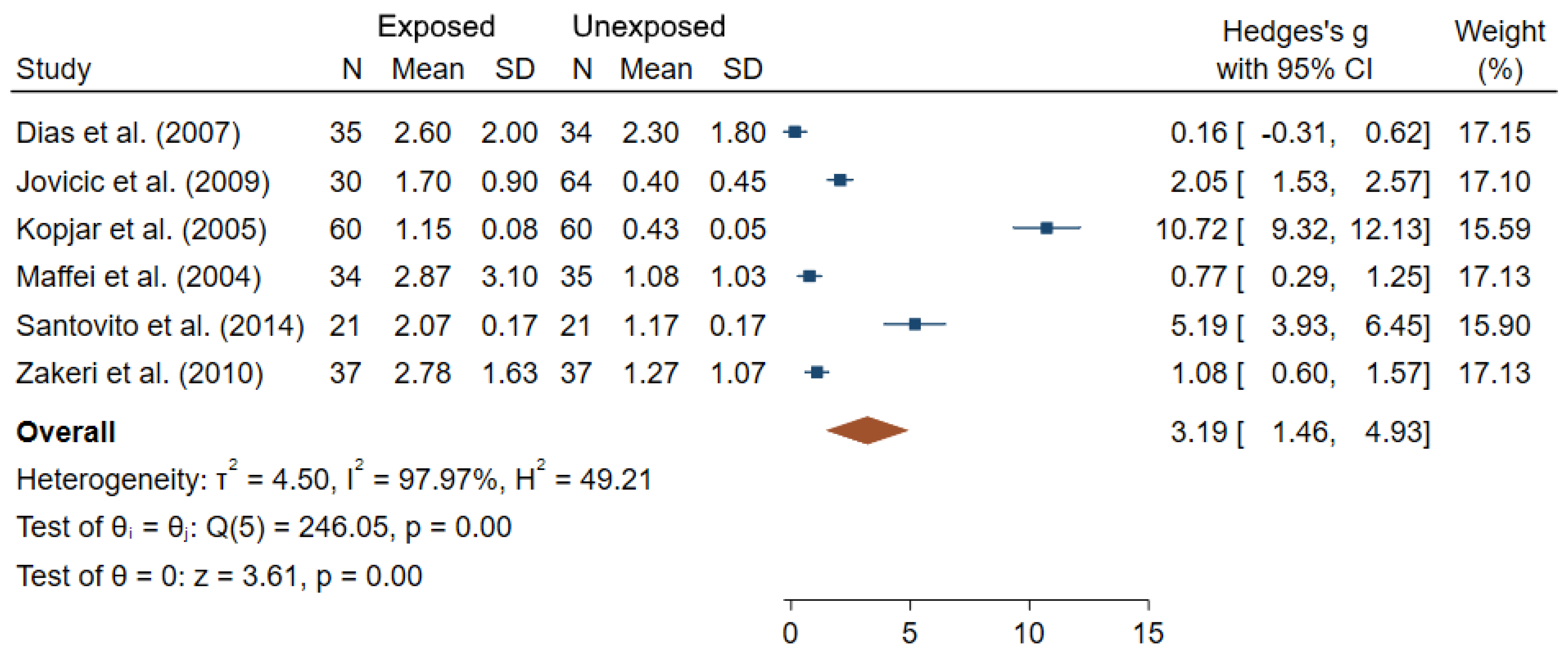
Few studies investigated dose-response analyses between low-dose IR exposure and CA frequencies in medical workers, with no clear relationships reported [53,55,90]. In Figure 3, results of the six studies with complete information are summarized in a form of a Forest plot showing the differences in the rates of aberrant cells between IR-exposed and unexposed workers [35,48,52,57,73,88]. An estimated overall standardized mean difference (θpooled = 3.19; 95% CI 1.46–4.93) was significantly different from 0 (Z = 3.61, p < 10−3). A high heterogeneity between studies was observed (I2 = 97.97%; Q = 246.05, p < 10−3). Small-study effects and publication bias were found using the Egger’s test (p < 10−3).
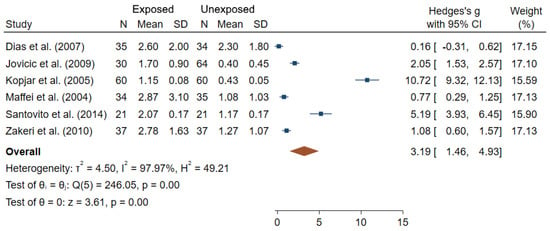
Figure 3.
Forest plot of mean differences in fraction of cells with CA between IR exposed and unexposed workers. The blue squares represent the differences in standardized means between IR-exposed and non-exposed workers for each study individually (with their associated blue bar corresponding to the confidence interval of each mean), while the brown diamond below corresponds to the estimated overall standardized mean difference (placed in the center of the diamond, with the bounds of the confidence interval being at the extreme points of the diamond).
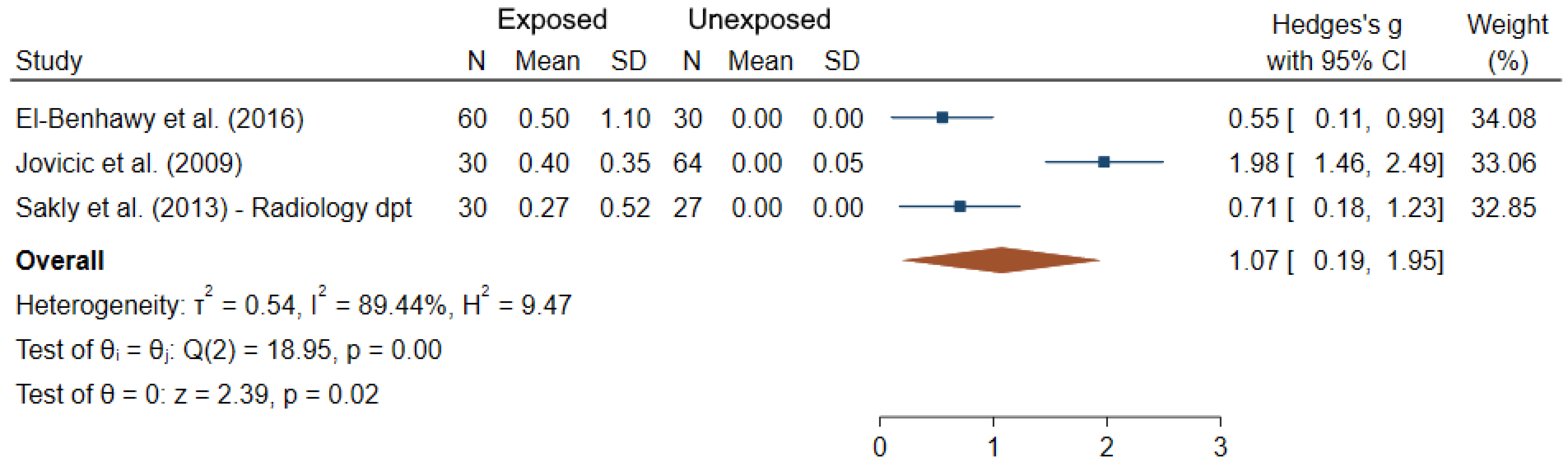
Among the 24 studies which focused on dicentrics, only three provided sufficient data for meta-analysis (Figure 4). It can be noted that the rates of dicentrics in controls were zero; and the mean difference between IR-exposed and unexposed workers was significantly greater than 0 (θpooled = 1.07; 95% CI 0.19–1.95; Z = 2.39, p = 0.02), results being heterogeneous between studies (I2 = 89.44%; Q = 18.95, p < 10−3), but the Egger regression-based test did not show publication bias and small-study effects (p = 0.61).
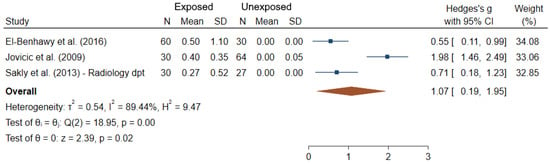
Figure 4.
Forest plot of mean differences in dicentrics between IR exposed and unexposed workers.
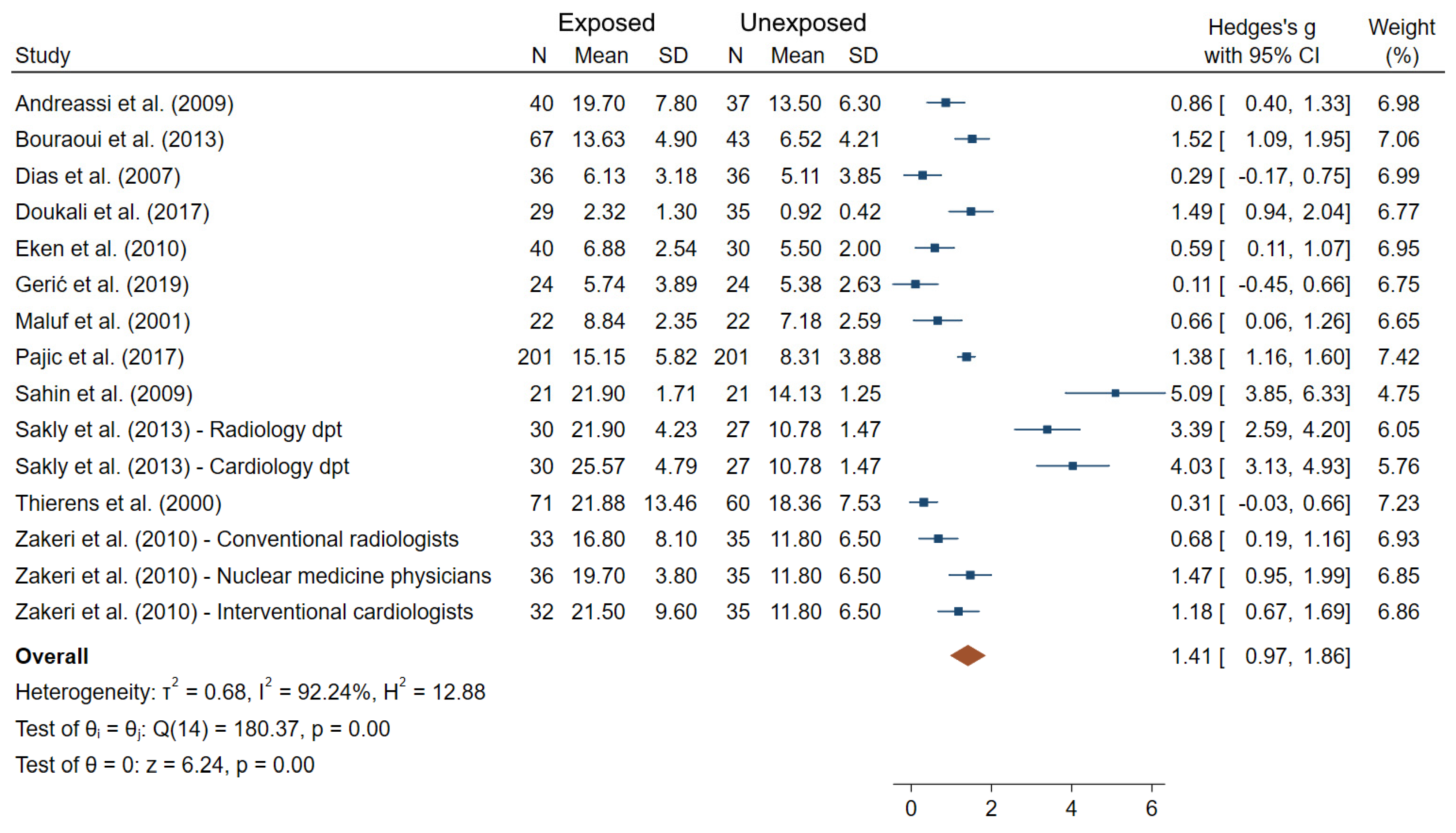
With respect to the studies dealing with MN frequencies, which is the most widely used endpoints among studies, 28 out of the 32 did show significantly higher frequencies in exposed compared to unexposed workers using comparisons tests between groups (Figure 2). Additionally, among the 32 MN studies, 23 evaluated the relationship (correlation or dose-response) between IR exposure and MN frequencies using two alternative indicators of exposure (using one or both indicators): cumulative dose from personal dosimeters (14 studies) and/or duration of exposure in years (16 studies). Among those, a significant association/correlation between cumulative dose or duration of exposure and MN frequencies were reported in 5/14 and 9/16 studies, respectively [23,32,38,61,64,65,74,77,82]. Additionally, in professionals working in a nuclear medicine department, both MN and SCE levels were significantly higher during their occupational exposure compared to levels immediately after vacation period [70]. Twelve out of 32 studies satisfied the criteria for inclusion into meta-analysis. Figure 5 shows an overall mean difference of MN frequencies comparisons between IR-exposed and unexposed workers (θpooled = 1.41; 95% CI 0.97–1.86) which was significantly different from 0 (Z = 6.24, p < 10−3), with high heterogeneity between study-specific effect sizes (I2 = 92.24%; Q = 180.37, p < 10−3). The Egger regression-based test revealed small-study effects and publication bias (p < 10−3).
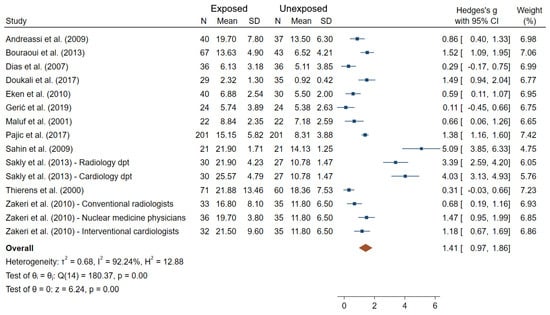
Figure 5.
Forest plot of mean differences in MN frequencies between IR exposed and unexposed workers.
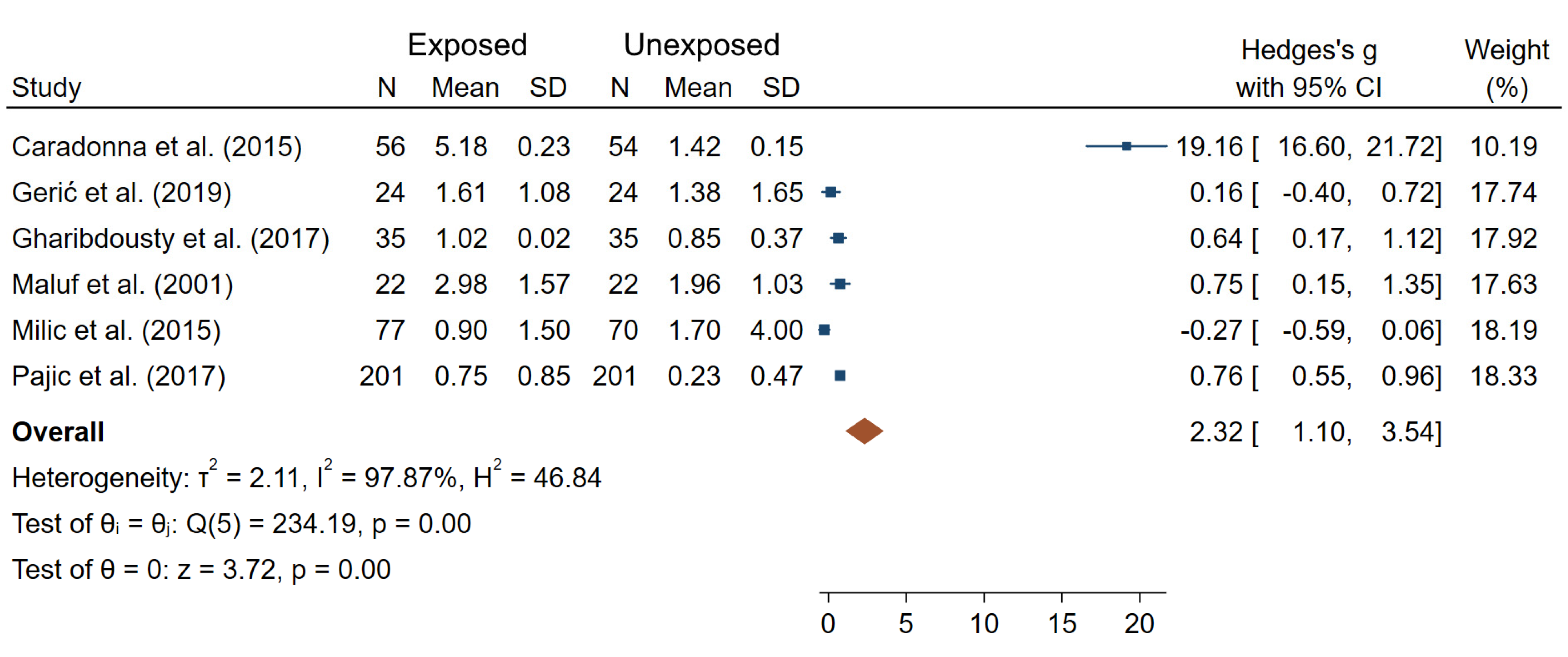
In terms of NPB frequencies, four studies reported a lack of a significant difference between IR-exposed and unexposed workers [24,45,61,69], and three studies showed significantly elevated NPB frequencies in IR-exposed medical workers compared to controls [33,58,65]. Meta-analysis carried out on the data from six studies with complete information (Figure 6) reported a significant overall difference in NPB frequencies between IR exposed and unexposed workers (θpooled = 2.32; 95% CI 1.10–3.54; Z = 3.72, p < 10−3; I2 = 97.87%; Q = 234.19, p < 10−3). Small-study effects and publication bias (p < 10−3) were observed.
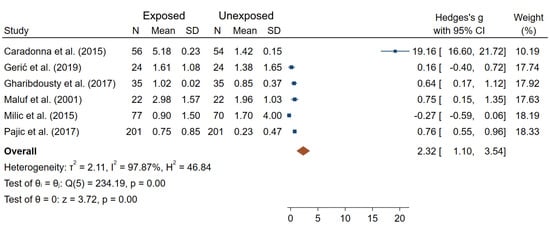
Figure 6.
Forest plot of mean differences in NPB frequencies between IR exposed and unexposed workers.
The mean number of SCE per cell was significantly higher in five out of seven studies. While SCE number was significantly higher in a group of workers with a duration of employment ≥15 years compared to a group with <15 years of employment [38], only two studies conducted linear regression and reported a positive but non-significant β-coefficient for SCE frequency with an increase in IR exposure assessed by duration of employment [73] or exposure dose [39].
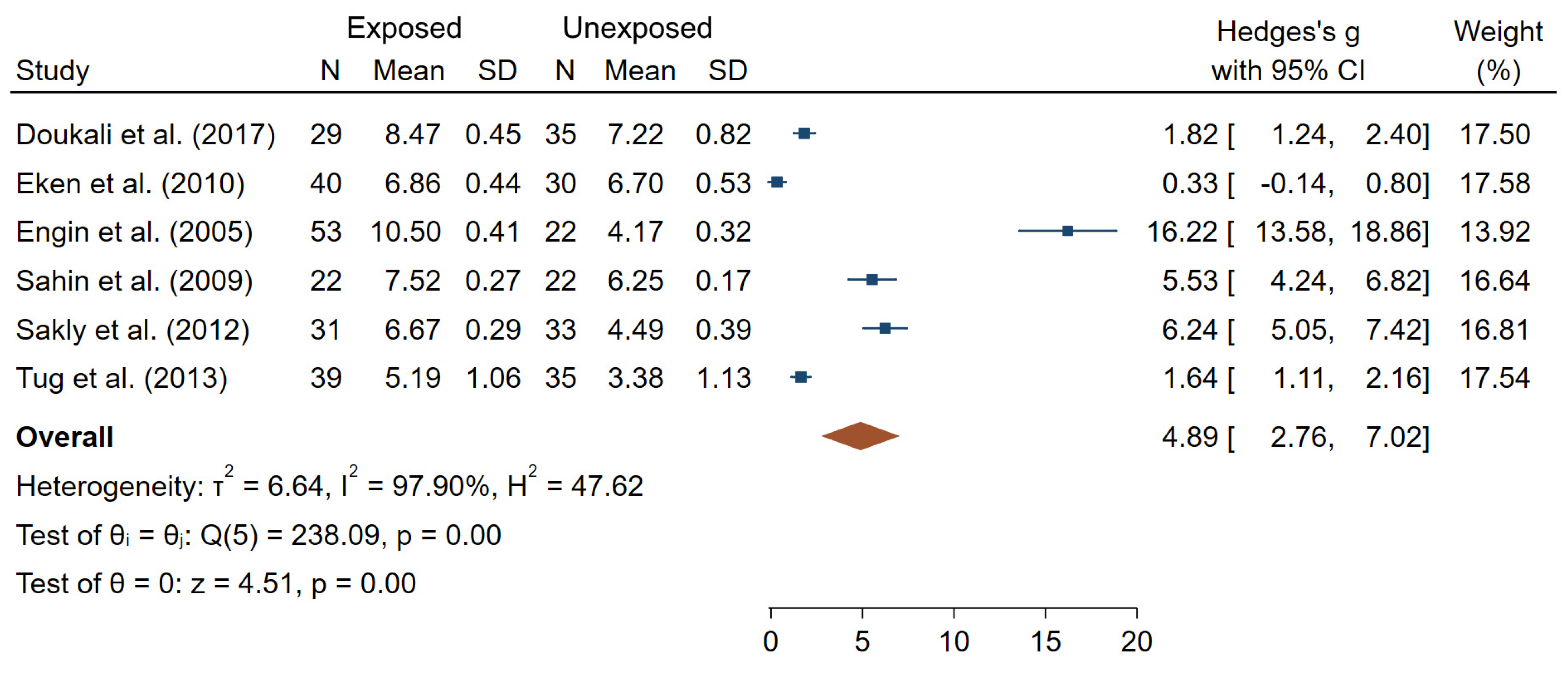
Figure 7 reports overall mean differences in SCE frequencies between IR-exposed and unexposed workers for the six studies included in meta-analysis (θpooled = 4.89; 95% CI 2.76–7.02), which was significantly different from 0 (Z = 4.51, p < 10−3). High heterogeneity (I2 = 97.90%; Q = 238.09, p < 10−3), small-study effects and publication bias were observed (p < 10−3).
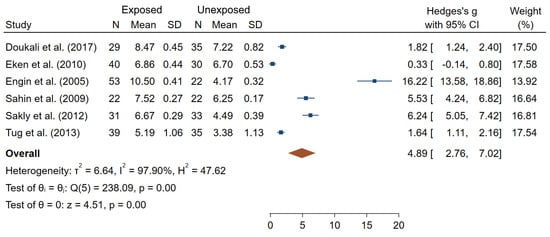
Figure 7.
Forest plot of mean differences in SCE frequencies between IR exposed and unexposed workers.
Lastly, the only two studies that focused on PCD reported significantly higher frequencies in IR-exposed workers compared to unexposed, regardless of a specific PCD readout used [49,64].
3.2. DNA Integrity Biomarkers
Only 18 out of the 65 studies evaluated various DNA integrity endpoints as genotoxicity biomarkers. These were comet tail length (TL; six studies), comet tail moment (TM; four studies), comet score (DNA damage extent, CS; four studies), %DNA in the tail (%DNA; three studies), DNA strand breaks (SBs; one study), glycophorin A (GPA) mutant (one study), leucocyte telomere length (LTL; one study), copy number variation in AZFc region (one study), and γ-H2AX foci (1 study), with almost no overlap within a single study.
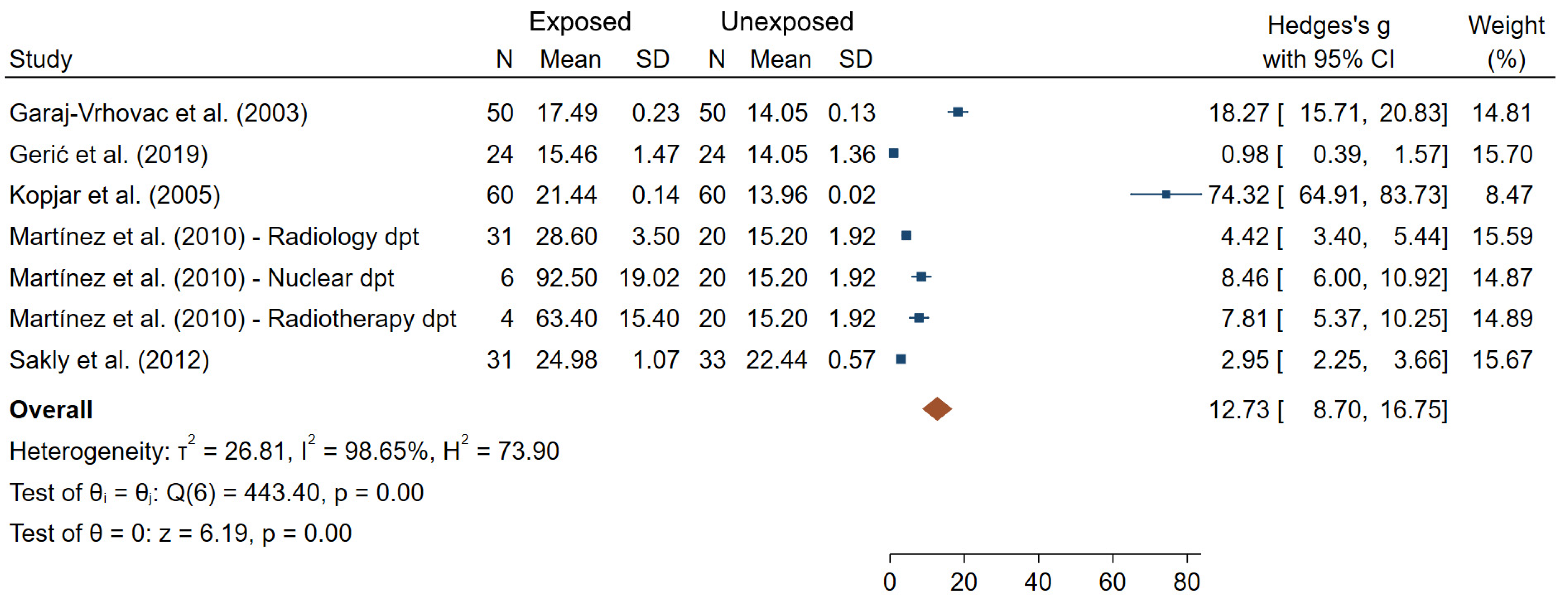
TL was significantly greater in IR-exposed compared to unexposed workers in all the six studies (out of the six reporting results for this endpoint). Additionally, TL was found to increase significantly at the end of a work day in IR-exposed individuals, but not in unexposed workers [59]. It can also be noted that differences in TL were found between various hospital departments and working places; however, no common pattern across studies was seen [44,52,59,82]. Figure 8 presents an overall mean difference for TL between IR-exposed and unexposed workers (θpooled = 12.73; 95% CI 8.70–16.75) which was significantly different from 0 (Z = 6.19, p < 10−3). However, large heterogeneities (I2 = 98.65%; Q = 443.40, p < 10−3), small-study effects and publication bias (p < 10−3) have been observed.
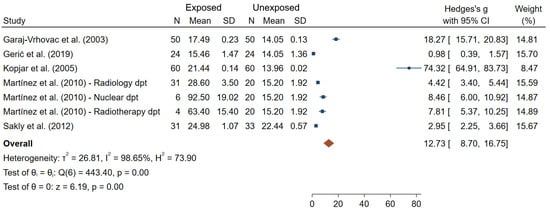
Figure 8.
Forest plot of mean differences in tail length between IR exposed and unexposed workers.
A higher %DNA in the comet tail was reported in IR-exposed compared to unexposed workers (statistically significant in two studies [37,42], and non-significant in one study [45]).
TM, which is an integral damage parameter derived from TL and %DNA, was found to be significantly higher in IR-exposed workers compared to unexposed workers in all 4 studies reporting TM [37,42,44,82]. Whereas no correlation between TM or %DNA and effective yearly dose measured by individual dosimeters was observed in nuclear medicine workers [37], an increased gradient in TM and in %DNA was found according to a 10-year increase in X-ray exposure duration [42].
Assigning arbitrary grades (0–4) to the extent of damage based on the size/shape of comets, as opposed to a direct measurement of TL in µm, is an alternative way of quantifying DNA damage by the comet assay. This technique was used in four included studies, three of which demonstrate that the level of DNA damage was significantly higher in the IR-exposed compared to unexposed workers [51,58,78]. However, one study did not show any difference in DNA damage between 20 IR-exposed and 20 unexposed workers [66].
Gaetani et al. observed no difference between IR-exposed and unexposed workers for three types of DNA lesions: SBs, oxidized purines and oxidized pyrimidines assessed by the conventional and enzyme modified comet assay in peripheral blood cells [43].
A significant dose-response relationship was found between cumulative IR exposure dose and glycophorin A (GPA) mutation frequency in red blood cells of hospital workers, both for NO and NN variants (β = 1.88 × 10−6/cGy, p = 0.003; and β = 2.23 × 10−6/cGy, p = 0.0001, respectively) [46].
Using polymerase chain reaction (PCR), relative leukocyte telomere length (LTL) (as the ratio of telomere repeats to a single-copy gene, relative to a reference sample) was shown to decrease significantly with an increase in lifetime radiation dose (r = −0.319; p = 0.03), but also with an increase of occupational radiological risk score (r = −0.267; p = 0.002) [28]. The latter exposure parameter takes into account the number of years in catheterization laboratory, the number of procedures per year, and the distance from the source of radiation. Recently, using quantitative real-time PCR, a significantly higher rate of microdeletion and microduplication as assessed by copy number variation (CNV) in the SY1197 sequence-tagged site of the Y-chromosome azoospermia factor region c (AZFc) was shown in male Cath lab workers compared to unexposed controls (CNV = 1.53 ± 0.8 vs. CNV = 1.02 ± 0.4 respectively, p = 0.0005) [29]. However, a non-significant difference was shown for the SY579 site CNV in AZFc region between exposed and unexposed workers. A sequence-tagged site (STS) is relatively short (200–500 base pairs) and can be specifically amplified by PCR. In clinical setting, the STSs (such as SY579 and SY1197) have been used to detect microdeletions (and infertility) in Azoospermia (AZF) genes in men.
Lastly, the frequency of γ-H2AX foci, a marker of DNA double-strand breaks, was found to be significantly higher in IR-exposed workers compared to healthy volunteers [68].
4. Discussion
Potential genotoxic effect of medical occupational exposure to IR is a widely recognized concern that has been scrutinized in many studies in the last two decades. Although a great variety of genotoxicity endpoints has been assessed in these studies helping generate new knowledge, there are still inconsistencies in the results, making it difficult to interpret. In this systematic review and meta-analysis, we attempted to consolidate knowledge using commonly accepted methods with the overall objective to identify the biomarkers of genotoxicity that are most reliably and commonly observed at elevated levels in medical workers occupationally exposed to IR compared to unexposed cohorts. The approach implemented in our work produced a list of 65 studies (Table 2).
The results of our work confirm the relevance of CA and MN as genotoxicity biomarkers that are consistently elevated in IR-exposed vs. unexposed workers. SCE, stable translocations and the comet assay endpoints are strong candidates and require further studies to validate their usefulness.
Thus, the most commonly reported cytogenetic biomarkers were unstable CA (mainly dicentrics and acentric fragments) and MN, and both were significantly elevated in IR-exposed workers compared to controls in the majority of the studies (Table 3, Figure 2).

Table 3.
Summary of the biomarkers used in the included studies.
Our meta-analyses of the data from studies with complete information confirmed that IR-exposed medical workers had significantly elevated frequencies of blood lymphocytes with CA and/or MN. Particularly, dicentrics were previously reported to be “the biomarker of choice for investigating recent exposure to IR” which is typically little confounded by other factors [22] and is a standard endpoint for radiation biodosimetry applications [91]. Because of its unstable nature and continuous renewal of PBL, the frequency of dicentrics decreases with time after exposure. This may explain why decreases in unstable CA frequencies were found in workers upon removal from IR exposure [48]. Interestingly, such periods without IR exposure (vacation, break or change to non-IR professional activities) may have contributed to the failure to show dose-response relationships for unstable CA in nuclear medical workers [48,60]. However, regardless of the endpoint, establishing dose-response for genotoxicity biomarkers in human studies is a very challenging task due to several factors, such as uncertainty in dose estimates, type of IR, mode of exposure, time, etc.). Dose-responses for these endpoints are well established for controlled ex vivo irradiation of human PBL, but they poorly compare to the studies reviewed here in terms of lowest dose resolution. Indeed, most of the occupational exposures are below the lowest resolution power observed in ex vivo cytogenetic assays [92,93,94], but previous studies did show a significant increase in cytogenetic endpoints in patients who received radiation exposure during a single medical examination, such as a CT scan or catheterization procedure, where the radiation dose is far lower than 100 mSv [95,96,97]. Similarly the effect of confounding factors on CA frequencies was inconsistent between studies, with some authors reporting the effect of certain factors such as age, smoking and gender [54,57,60,72], while others showing no influence [62,64,76,87]. Furthermore, statistically significant differences in CA frequencies were reported between cohorts employed at different working places, with specificity of certain types of CA depending on the job. Translocations have not been extensively studied (6 studies out of the 30 studies dealing with CA), but showed a good consistency and reliability, with significantly higher values in IR-exposed workers in almost all studies. Unlike dicentrics, translocations are stable CA in PBL and can therefore be considered as retrospective biomarkers of exposures [14]. However, translocations were reported to be hypersensitive to other factors (e.g., age, smoking habits, mode of exposure, diet and exposure to other clastogenic agents) that can influence their accumulation and persistence [98].
Compared to CA, MN are much easier to score, either manually or using automated systems [14]. The MN assay can also be viewed as an alternative method to dicentric chromosome assay and presents the advantage that it can be assessed in lymphocytes (fresh or frozen), cell lines, erythrocytes, epithelial buccal cells, nasal mucosa or urine-derived cells using standardized protocols [99]. Thus, MN frequencies from epithelial buccal cells have recently been shown to be significantly higher in IR-exposed compared to unexposed health workers (studies not included in the present systematic review) [100,101]. The inconsistency between the sensitivity seen in ex vivo (20 mGy or higher) and in vivo studies (typically <20 mGy) for CA and dicentrics is also evident for the MN assay that is considered not to be very sensitive at low doses in ex vivo studies (~200 mGy lowest dose resolution) [93,102]. MN can be induced by other environmental agents suggesting MN is a non-specific biomarker of IR exposure [22]. Although the results suggest that MN is one of the best discriminators between IR-exposed and unexposed medicine workers (Figure 2, Figure 5), careful consideration of the named potentially confounding factors should be included in new studies, as well as in the analyses of published results.
SCE are easy to detect and score and were shown to be a sensitive and reliable endpoint of a genotoxic potential of chemical carcinogens and mutagens [103,104]. However, such sensitivity to chemical and relative insensitivity to IR [17] suggests that SCE measurements in low-dose IR-exposed humans should be carefully examined for potential co-exposure to chemical mutagens or other DNA damaging factors.
With regards to NPB frequencies, only three out of seven studies reported significantly higher NPB frequencies in IR-exposed medical workers compared to controls [33,58,65]. Furthermore, out of the three studies, Caradonna et al. reported extremely high values for NPB frequency compared to the rest of studies for this endpoint [33]. NPB are indicators of the presence of dicentric chromosomes and therefore their frequency are expected to be similar to that of dicentric chromosomes, which was not the case in this study. Additionally, the mean age in exposed and non-exposed groups in Caradonna et al. were different, 42 vs. 30 years old, and no adjustment was made in contrast to other studies. The overall mean difference between IR-exposed and unexposed workers was no longer significant when the study of Caradonna et al. was excluded (Figure S1). PCD were used in only two studies, which does not allow judging its usefulness as a biomarker in further studies. Lastly, ring chromosome frequencies were assessed in 14 studies, in which non-significant differences between IR-exposed and unexposed workers were found for most of the studies, suggesting that this endpoint can be excluded from the list of potential IR exposure biomarkers.
Molecular biomarkers, grouped here as DNA integrity endpoints, were dominated by the parameters that are measured using the comet assay. Thus, 12 out of 18 studies in this category used the comet assay to measure tail length, tail moment, %DNA in the tail or an arbitrary comet score or index, showing a good consistency and reliability, with significantly higher values in IR-exposed workers in most of the studies. It is not clear what parameter is the most relevant for genotoxicity assessment, and this has been the subject of debate because each comes with its advantages and limitations. It was first assumed that tail moment (product of %DNA and tail length) provides a better description of DNA integrity compared to tail length only [105]. However, tails with different lengths, numbers of fragments and relative amounts of DNA may have the same tail moment, which can be considered a counter-argument for the use of this descriptor [106]. An alternative way of scoring DNA damage by the comet assay, suitable for low budget and fast assessment, is a manual classification of comets into five categories based on their appearance [107]. Noteworthily, in the studies included in this review, these comet assay parameters were found to have little or no sensitivity to confounding factors such as gender, age, smoking status and alcohol consumption, suggesting potential specificity to IR exposure [52,59,71]. Nevertheless, it can be noted that differences in TL were found between various hospital departments and working places; however, no common pattern across studies was seen [44,52,59,82]. It should be pointed that the types of DNA lesions detected by the comet assay (SSB, oxidized and alkali-labile sites) are highly abundant in cells due to oxygen metabolism and replication which has to be considered while interpreting results. These lesions are rapidly repaired after irradiation (minutes to hours) and thus should not be interpreted as the actual DNA damage induced directly by protracted low-dose IR exposure [108]. Instead, the detected increases in DNA damage levels in lymphocytes of IR-exposed subjects may reflect secondary effects due to altered repair machinery, genomic instability or additional production of reactive oxygen species. Moreover, one of the main limitations of the comet assay is the inter-laboratory variability in protocols, affecting results and, subsequently, the comparability between studies.
Glycophorin A (GPA) mutant, leukocyte telomere length, copy number variation in AZFc region, and γH2AX foci were assessed only in one study for each endpoint, and were found to be elevated in IR-exposed cohorts. However, no correlation between age or duration of occupational exposure and γ-H2AX foci frequency in IR-exposed medical workers were seen. Each of these endpoints is relevant to a very specific type of DNA lesions and, when examined separately, may be poor indicators of the overall burden of genotoxicity.
The biomarkers included in this work have been widely investigated by previous molecular epidemiology studies to assess environmental, occupational and medical exposure to IR. Although there are still uncertainties with respect to their sensitivity or specificity to detect low level of IR exposures in human biomonitoring [7], it is worth considering these important aspects in the interpretation of the results. To this end, Table 4 shows a summary of the dose detection limit and specificity for each biomarker used in our study.

Table 4.
Detection limit dose and specificity for biomarkers.
Relevance to an adverse health outcome is an important attribute of a genotoxicity biomarker since inferring a potential health risk is one of the main objectives of measuring such a biomarker in IR exposed individuals. The greater relevance, the higher value of such assessment. Although high frequencies of CA in PBL have long been linked to an increased risk of cancer using association [11,12,13,109] and incidental evidence [110], the causal relationship between CA in PBL and cancer has not been established. Recent results of the analysis of genomic characteristics of thyroid cancer in IR-exposed vs. unexposed patients (I-131 from Chernobyl nuclear accident) suggest that radiation exposure was associated with increased frequencies of small insertions/deletions and other small structural chromosome variants originating from DSB [9]. Although indirectly, these results highlight the importance of CA in tumorigenesis and thus their value as a genotoxicity biomarker in nuclear medicine workers.
Like unstable CA, MN represent a lethal abnormality that typically results in cell death during mitosis, thus preventing potential neoplastic transformation of the cell and suggesting a poor link to cancer risk. However, recent findings suggest that MN may not be passive outcomes of earlier DNA damage events, but exert biological activity triggering hypermutation and pro-inflammatory signaling [111,112]. Since lymphocytes are known to actively penetrate tissues, it is feasible to hypothesize that pro-inflammatory signaling by micronucleated lymphocytes may contribute to chronic tissue inflammation which in turn would increase the risk of tumorigenesis [113]. Together with these recent highlights on the role of MN in mutagenesis and tissue homeostasis, our results confirming that MN are reliably detected at elevated rates in PBL of IR-exposed medical workers highlight the potential value of this biomarker.
Unfortunately, our systematic review showed a limited number of studies assessing dose-responses, resulting in inability to carry out quantitative dose-response meta-analysis. Furthermore, given that non-significant results are less likely to be reported [114], it can be assumed that the dose–response relationship between occupational medical IR exposure and cancer biomarkers is poorly understood [27,64,71,74]. Issues associated with dose records include for example underestimation of dose when personal badges are not properly worn as observed in cardiologists studies [115], which could affect corresponding dose-response estimates [116]. However, IR-exposure assessed by the duration of employment as proxy did not show more conclusive results [24,30,72]. Biological factors that can affect the shape of a dose-response for genotoxicity endpoints include a large variety of adaptive stress responses that can be induced at low doses of IR and used by cells to effectively eliminate genotoxic damage, predominantly by activation of DNA repair [117,118,119]. Low-dose IR was also shown to trigger anti-inflammatory processes [120,121] and immune activation [122,123], both capable of affecting the shape of the dose-response. The decrease in CA frequencies found in medical workers after vacation or removal from IR exposure [48,60] are consistent with these compensatory biological mechanisms. Interestingly, an alternative explanation of these results could be a withdrawal from the chronic work-/life-related stress also known to cause accumulation of DNA and chromosomal damage [124,125]. Indeed, chronic stress is known to cause DNA damage [126]. Similarly, exam-associated stress can lead to increased DNA damage in university students [127] and stress-relieving hormonal therapy was associated with a marked reduction of cancer risk [128]. Therefore, in complex real-life situation studies such as those carried out on nuclear medical workers, it is very important to consider a multitude of variables and factors. These and other factors discussed above that can potentially affect the results of the assessment of genotoxicity biomarkers may have contributed to high heterogeneity revealed by our meta-analyses. However, we did not examine the interaction effect between IR and potentially confounding factors that—as shown above for many endpoints—can influence the results. In fact, the number of studies where the endpoints were measured separately in populations stratified by other factors (e.g., in smoker vs. non-smokers) was very limited, preventing the interaction effect tests. Such analyses are arguably very important in future studies and meta-analyses to understand the causal relationship between a genotoxic endpoint and IR exposure.
Further, a complex dependence of the endpoints measured on a type of a medical department of employment or occupation was reported, certainly because of the type of radiation and the distance with the source [37,52,67], constituting another potential factor of data heterogeneity. A limited number of studies stratifying by department/occupation made it impossible to account for this information and/or detect a pattern.
Genetic susceptibility may account for inter-individual differences in radiation sensitivity [27,30,61,71,82] and further contribute to data variability. Specifically, the genetic polymorphism of DNA repair and xenobiotic-metabolizing enzymes may play a crucial role in determining an individual’s ability to repair cellular DNA after IR exposure, and therefore, to influence the biological endpoint and the dose-response relationship [27,30,61,71,82,129]. Progress in understanding the interaction of the genotype and genotoxic insults, such as IR, and how it affects cancer risks may pave the way towards future personalized radiation protection principles and approaches.
To our knowledge, this work is the first systematic review and meta-analysis of literature assessing genotoxicity biomarkers in medical workers exposed occupationally to IR. We included a broad range of endpoints ranging from molecular DNA events to cytogenetic rearrangement, resulting in a large number of studies covered. All included studies met the previously defined criteria according to the PRISMA recommendations, allowing robust and exhaustive analysis while maintaining focus on the main research question. In spite of all the revealed limitations of the reviewed studies, which is partially due to a broad range of covered genotoxicity endpoints, using the combination of both qualitative and quantitative descriptions, we were able to provide an overview of the status-quo in the area of genotoxicity biomarkers in healthcare professionals occupationally exposed to IR. Although the quality of the various analyses used for the measurement of genotoxicity in individual studies was not assessed (e.g., see such assessment in [130]), we applied the Newcastle–Ottawa Scale to assess the quality of the included studies, resulting in the exclusion of only one study with a score <3, thus underlining the good overall level of the considered studies.
Finally, in an attempt to account for confounding factors potentially correlated with certain endpoints, most studies did match the IR-exposed and unexposed groups by age and gender (and sometimes by smoking habits) and reported crude values, which we used in our analyses. This made it possible to handle confusion bias in our work.
5. Conclusions
We reviewed all available data on genotoxicity biomarkers in health workers exposed to IR occupationally using systematic review and meta-analyses. Our qualitative and quantitative results suggest that CA (mainly dicentrics and acentric fragments) and MN are the best discriminators and correctly reflect the interaction between the biological system of healthcare workers and low-dose IR exposure. In contrast, ring chromosomes and nucleoplasmic bridges appear to correlate poorly with medical occupational IR exposure and can be excluded from the list of potential IR exposure biomarkers. Among the DNA integrity biomarkers, the comet assay endpoints showed good correlation with IR exposure, however, it is DNA DSB, not SSB or single nucleotide variants that are thought to contribute to IR-induced cancer. Thus, the relevance of the comet assay results to long-term health conditions is unclear. Several factors that can contribute to the measured value of a genotoxicity marker exist (increasing variability and uncertainty of results) and should be better accounted for in future work; these include time away from IR exposure, repair mechanisms, age and life style. Other new biomarkers and techniques, such as telomere length as well as gene array techniques, may be highly useful to improve overall biological understanding of low dose radiation exposure and the likelihood of subsequent disease as well as to identify underlying factors that modulate radiation sensitivity.
Lastly, our review revealed a shortage of studies with accurate dosimetric information, thus emphasizing the need for dose evaluation to facilitate the construction of dose-responses. Our results warrant and inform future studies aiming at examining the role of specific types of CA and MN in long-term health outcomes, with prospective epidemiological studies of proper design being instrumental for achieving this daunting goal.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/ijms22147504/s1.
Author Contributions
C.B., M.G.A. and M.-O.B. designed the study; C.B. and M.G.A. made the systematic research and independently performed the selection of studies according to the PECO criteria; disagreements have been discussed and resolved by M.-O.B.; C.B. performed the qualitative and quantitative syntheses of the studies; C.B., M.G.A., M.-O.B. and D.K. drafted the initial report, they take responsibility for the integrity of the data and the accuracy of the data analysis. All authors have read and agreed to the published version of the manuscript.
Funding
The study was funded by an intramural program of IRSN.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors are grateful to all the authors of the studies included in this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fazel, R.; Krumholz, H.M.; Wang, Y.; Ross, J.S.; Chen, J.; Ting, H.H.; Shah, N.D.; Nasir, K.; Einstein, A.J.; Nallamothu, B.K. Exposure to Low-Dose Ionizing Radiation from Medical Imaging Procedures in the United States. N. Engl. J. Med. 2009, 361, 849–857. [Google Scholar] [CrossRef]
- Brenner, D.J.; Hall, E.J. Computed Tomography—An Increasing Source of Radiation Exposure. N. Engl. J. Med. 2007, 357, 2277–2284. [Google Scholar] [CrossRef] [PubMed]
- Tafreshi, N.K.; Doligalski, M.L.; Tichacek, C.J.; Pandya, D.N.; Budzevich, M.M.; El-Haddad, G.; Khushalani, N.I.; Moros, E.G.; McLaughlin, M.L.; Wadas, T.J.; et al. Development of Targeted Alpha Particle Therapy for Solid Tumors. Molecules 2019, 24, 4314. [Google Scholar] [CrossRef]
- UNSCEAR. Sources and Effects of Ionizing Radiation, Report to the General Assembly with Scientific Annexes; UNSCEAR: New York, NY, USA, 2008. [Google Scholar]
- Grosse, Y.; Baan, R.; Straif, K.; Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Galichet, L.; Cogliano, V.; et al. A Review of Human Carcinogens--Part A: Pharmaceuticals. Lancet Oncol. 2009, 10, 13–14. [Google Scholar] [CrossRef]
- Kreuzer, M.; Auvinen, A.; Cardis, E.; Durante, M.; Harms-Ringdahl, M.; Jourdain, J.R.; Madas, B.G.; Ottolenghi, A.; Pazzaglia, S.; Prise, K.M.; et al. Multidisciplinary European Low Dose Initiative (MELODI): Strategic Research Agenda for Low Dose Radiation Risk Research. Radiat. Environ. Biophys. 2018, 57, 5–15. [Google Scholar] [CrossRef]
- Hall, J.; Jeggo, P.A.; West, C.; Gomolka, M.; Quintens, R.; Badie, C.; Laurent, O.; Aerts, A.; Anastasov, N.; Azimzadeh, O.; et al. Ionizing Radiation Biomarkers in Epidemiological Studies—An Update. Mutat. Res. 2017, 771, 59–84. [Google Scholar] [CrossRef] [PubMed]
- Little, J.B. Radiation Carcinogenesis. Carcinogenesis 2000, 21, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Morton, L.M.; Karyadi, D.M.; Stewart, C.; Bogdanova, T.I.; Dawson, E.T.; Steinberg, M.K.; Dai, J.; Hartley, S.W.; Schonfeld, S.J.; Sampson, J.N.; et al. Radiation-Related Genomic Profile of Papillary Thyroid Cancer after the Chernobyl Accident. Science 2021. [Google Scholar] [CrossRef]
- Natarajan, A.T.; Boei, J.J.W.A. Formation of Chromosome Aberrations: Insights from FISH. Mutat. Res./Rev. Mutat. Res. 2003, 544, 299–304. [Google Scholar] [CrossRef]
- Boffetta, P.; van der Hel, O.; Norppa, H.; Fabianova, E.; Fucic, A.; Gundy, S.; Lazutka, J.; Cebulska-Wasilewska, A.; Puskailerova, D.; Znaor, A.; et al. Chromosomal Aberrations and Cancer Risk: Results of a Cohort Study from Central Europe. Am. J. Epidemiol. 2007, 165, 36–43. [Google Scholar] [CrossRef]
- Bonassi, S.; Norppa, H.; Ceppi, M.; Strömberg, U.; Vermeulen, R.; Znaor, A.; Cebulska-Wasilewska, A.; Fabianova, E.; Fucic, A.; Gundy, S.; et al. Chromosomal Aberration Frequency in Lymphocytes Predicts the Risk of Cancer: Results from a Pooled Cohort Study of 22 358 Subjects in 11 Countries. Carcinogenesis 2008, 29, 1178–1183. [Google Scholar] [CrossRef]
- Hagmar, L.; Strömberg, U.; Bonassi, S.; Hansteen, I.-L.; Knudsen, L.E.; Lindholm, C.; Norppa, H. Impact of Types of Lymphocyte Chromosomal Aberrations on Human Cancer Risk: Results from Nordic and Italian Cohorts. Cancer Res. 2004, 64, 2258–2263. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Energy Agency (IAEA). Cytogenetic Dosimetry: Applications in Preparedness for and Response to Radiation Emergencies; International Atomic Energy Agency: Vienna, Austria, 2011. [Google Scholar]
- Fučić, A.; Želježić, D.; Kašuba, V.; Kopjar, N.; Rozgaj, R.; Lasan, R.; Mijić, A.; Hitrec, V.; Lucas, J.N. Stable and Unstable Chromosome Aberrations Measured after Occupational Exposure to Ionizing Radiation and Ultrasound. Croat. Med. J. 2007, 48, 371–377. [Google Scholar]
- De Pascalis, I.; Pilato, B.; Mazzotta, A.; Dell’Endice, T.S.; Rubini, V.; Simone, G.; Paradiso, A.; Aiello, V.; Mangia, A. Sister Chromatid Exchange: A Possible Approach to Characterize Familial Breast Cancer Patients. Oncol. Rep. 2015, 33, 930–934. [Google Scholar] [CrossRef] [PubMed]
- Perry, P.; Evans, H.J. Cytological Detection of Mutagen-Carcinogen Exposure by Sister Chromatid Exchange. Nature 1975, 258, 121–125. [Google Scholar] [CrossRef]
- Lazutka, J.R.; Lekevicius, R.; Dedonyte, V.; Maciuleviciute-Gervers, L.; Mierauskiene, J.; Rudaitiene, S.; Slapsyte, G. Chromosomal Aberrations and Sister-Chromatid Exchanges in Lithuanian Populations: Effects of Occupational and Environmental Exposures. Mutat. Res. 1999, 445, 225–239. [Google Scholar] [CrossRef]
- Gutiérrez-Enríquez, S.; Ramón y Cajal, T.; Alonso, C.; Corral, A.; Carrasco, P.; Cornet, M.; Sanz, J.; Ribas, M.; Baiget, M.; Diez, O. Ionizing Radiation or Mitomycin-Induced Micronuclei in Lymphocytes of BRCA1 or BRCA2 Mutation Carriers. Breast Cancer Res. Treat. 2011, 127, 611–622. [Google Scholar] [CrossRef]
- Blackburn, E.H. Switching and Signaling at the Telomere. Cell 2001, 106, 661–673. [Google Scholar] [CrossRef]
- Reste, J.; Zvigule, G.; Zvagule, T.; Kurjane, N.; Eglite, M.; Gabruseva, N.; Berzina, D.; Plonis, J.; Miklasevics, E. Telomere Length in Chernobyl Accident Recovery Workers in the Late Period after the Disaster. J. Radiat. Res. 2014, 55, 1089–1100. [Google Scholar] [CrossRef][Green Version]
- Pernot, E.; Hall, J.; Baatout, S.; Benotmane, M.A.; Blanchardon, E.; Bouffler, S.; El Saghire, H.; Gomolka, M.; Guertler, A.; Harms-Ringdahl, M.; et al. Ionizing Radiation Biomarkers for Potential Use in Epidemiological Studies. Mutat. Res./Rev. Mutat. Res. 2012, 751, 258–286. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Dong, X.; Liu, T.; Zhang, L.; Ao, L. Antioxidant Status and Cytogenetic Damage in Hospital Workers Occupationally Exposed to Low Dose Ionizing Radiation. Mutat. Res./Genet. Toxicol. Environ. Mutagenesis 2020, 850–851, 503152. [Google Scholar] [CrossRef] [PubMed]
- Gharibdousty Low Levels of Ionizing Radiation Exposure and Cytogenetic Effects in Radiopharmacists. BBRC 2017. [CrossRef]
- Hedges, L.V. Distribution Theory for Glass’s Estimator of Effect Size and Related Estimators. J. Educ. Stat. 1981, 6, 107–128. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring Inconsistency in Meta-Analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Andreassi, M.G.; Foffa, I.; Manfredi, S.; Botto, N.; Cioppa, A.; Picano, E. Genetic Polymorphisms in XRCC1, OGG1, APE1 and XRCC3 DNA Repair Genes, Ionizing Radiation Exposure and Chromosomal DNA Damage in Interventional Cardiologists. Mutat. Res. 2009, 666, 57–63. [Google Scholar] [CrossRef]
- Andreassi, M.G.; Piccaluga, E.; Gargani, L.; Sabatino, L.; Borghini, A.; Faita, F.; Bruno, R.M.; Padovani, R.; Guagliumi, G.; Picano, E. Subclinical Carotid Atherosclerosis and Early Vascular Aging from Long-Term Low-Dose Ionizing Radiation Exposure: A Genetic, Telomere, and Vascular Ultrasound Study in Cardiac Catheterization Laboratory Staff. JACC Cardiovasc. Interv. 2015, 8, 616–627. [Google Scholar] [CrossRef]
- Andreassi, M.G.; Borghini, A.; Vecoli, C.; Piccaluga, E.; Guagliumi, G.; Del Greco, M.; Gaita, F.; Picano, E. Reproductive Outcomes and Y Chromosome Instability in Radiation-Exposed Male Workers in Cardiac Catheterization Laboratory. Environ. Mol. Mutagen. 2020, 61, 361–368. [Google Scholar] [CrossRef]
- Angelini, S.; Kumar, R.; Carbone, F.; Maffei, F.; Forti, G.C.; Violante, F.S.; Lodi, V.; Curti, S.; Hemminki, K.; Hrelia, P. Micronuclei in Humans Induced by Exposure to Low Level of Ionizing Radiation: Influence of Polymorphisms in DNA Repair Genes. Mutat. Res. 2005, 570, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, P.; Preston, D.L.; Doody, M.M.; Hauptmann, M.; Kampa, D.; Alexander, B.H.; Petibone, D.; Simon, S.L.; Weinstock, R.M.; Bouville, A.; et al. Retrospective Biodosimetry among United States Radiologic Technologists. Radiat. Res. 2007, 167, 727–734. [Google Scholar] [CrossRef]
- Bouraoui, S.; Mougou, S.; Drira, A.; Tabka, F.; Bouali, N.; Mrizek, N.; Elghezal, H.; Saad, A. A Cytogenetic Approach to the Effects of Low Levels of Ionizing Radiation (IR) on the Exposed Tunisian Hospital Workers. Int. J. Occup. Med. Environ. Health 2013, 26, 144–154. [Google Scholar] [CrossRef]
- Caradonna, F. Nucleoplasmic Bridges and Acrocentric Chromosome Associations as Early Markers of Exposure to Low Levels of Ionising Radiation in Occupationally Exposed Hospital Workers. Mutagenesis 2015, 30, 269–275. [Google Scholar] [CrossRef]
- Cigarrán, S.; Barquinero, J.F.; Barrios, L.; Ribas, M.; Egozcue, J.; Caballín, M.R. Cytogenetic Analyses by Fluorescence In Situ Hybridization (FISH) in Hospital Workers Occupationally Exposed to Low Levels of Ionizing Radiation. Radiat. Res. 2001, 155, 417–423. [Google Scholar] [CrossRef]
- Dias, F.L.; Antunes, L.M.G.; Rezende, P.A.; Carvalho, F.E.S.; Silva, C.M.D.; Matheus, J.M.; Oliveira, J.V.; Lopes, G.P.; Pereira, G.A.; Balarin, M.A.S. Cytogenetic Analysis in Lymphocytes from Workers Occupationally Exposed to Low Levels of Ionizing Radiation. Environ. Toxicol. Pharmacol. 2007, 23, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Djokovic-Davidovic, J.; Milovanovic, A.; Milovanovic, J.; Antic, V.; Gajic, M. Analysis of Chromosomal Aberrations Frequency, Haematological Parameters and Received Doses by Nuclear Medicine Professionals. J. BUON 2016, 21, 1307–1315. [Google Scholar]
- Dobrzyńska, M.M.; Pachocki, K.A.; Gajowik, A.; Radzikowska, J.; Sackiewicz, A. The Effect Occupational Exposure to Ionizing Radiation on the DNA Damage in Peripheral Blood Leukocytes of Nuclear Medicine Personnel. J. Occup. Health 2014, 56, 379–386. [Google Scholar] [CrossRef]
- Doukali, H.; Ben Salah, G.; Ben Rhouma, B.; Hajjaji, M.; Jaouadi, A.; Belguith-Mahfouth, N.; Masmoudi, M.-L.; Ammar-Keskes, L.; Kamoun, H. Cytogenetic Monitoring of Hospital Staff Exposed to Ionizing Radiation: Optimize Protocol Considering DNA Repair Genes Variability. Int. J. Radiat. Biol. 2017, 93, 1283–1288. [Google Scholar] [CrossRef]
- Eken, A.; Aydin, A.; Erdem, O.; Akay, C.; Sanal, H.T.; Soykut, B.; Sayal, A.; Somuncu, I. Cytogenetic Analysis of Peripheral Blood Lymphocytes of Hospital Staff Occupationally Exposed to Low Doses of Ionizing Radiation. Toxicol. Ind. Health 2010, 26, 273–280. [Google Scholar] [CrossRef] [PubMed]
- El-Benhawy, S.A.; Sadek, N.A.; Behery, A.K.; Issa, N.M.; Ali, O.K. Chromosomal Aberrations and Oxidative DNA Adduct 8-Hydroxy-2-Deoxyguanosine as Biomarkers of Radiotoxicity in Radiation Workers. J. Radiat. Res. Appl. Sci. 2016, 9, 249–258. [Google Scholar] [CrossRef]
- Engin, A.B.; Ergun, M.A.; Yurtcu, E.; Kan, D.; Sahin, G. Effect of Ionizing Radiation on the Pteridine Metabolic Pathway and Evaluation of Its Cytotoxicity in Exposed Hospital Staff. Mutat. Res. 2005, 585, 184–192. [Google Scholar] [CrossRef]
- Fang, L.; Li, J.; Li, W.; Mao, X.; Ma, Y.; Hou, D.; Zhu, W.; Jia, X.; Qiao, J. Assessment of Genomic Instability in Medical Workers Exposed to Chronic Low-Dose X-Rays in Northern China. Dose Response 2019, 17, 1559325819891378. [Google Scholar] [CrossRef]
- Gaetani, S.; Monaco, F.; Bracci, M.; Ciarapica, V.; Impollonia, G.; Valentino, M.; Tomasetti, M.; Santarelli, L.; Amati, M. DNA Damage Response in Workers Exposed to Low-Dose Ionising Radiation. Occup. Environ. Med. 2018, 75, 724–729. [Google Scholar] [CrossRef]
- Garaj-Vrhovac, V.; Kopjar, N. The Alkaline Comet Assay as Biomarker in Assessment of DNA Damage in Medical Personnel Occupationally Exposed to Ionizing Radiation. Mutagenesis 2003, 18, 265–271. [Google Scholar] [CrossRef]
- Gerić, M.; Popić, J.; Gajski, G.; Garaj-Vrhovac, V. Cytogenetic Status of Interventional Radiology Unit Workers Occupationally Exposed to Low-Dose Ionising Radiation: A Pilot Study. Mutat. Res. 2019, 843, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Yoo, K.-Y.; Cho, S.-H. Glycophorin A Mutant Frequency in Radiation Workers at the Nuclear Power Plants and a Hospital. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 2002, 501, 45–56. [Google Scholar] [CrossRef]
- Joseph, L.J.; Patwardhan, U.N.; Samuel, A.M. Frequency of Micronuclei in Peripheral Blood Lymphocytes from Subjects Occupationally Exposed to Low Levels of Ionizing Radiation. Mutat. Res. 2004, 564, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Jovicic, D.; Milacic, S.; Milic, N.; Bukvic, N.; Vukov, T.D. Chromosomal Aberrations in Subjects Exposed to Ionizing Radiation. J. Environ. Pathol. Toxicol. Oncol. 2009, 28, 75–82. [Google Scholar] [CrossRef]
- Jovicić, D.; Milacić, S.; Vukov, T.D.; Rakić, B.; Stevanović, M.; Drakulić, D.; Rakić, R.; Bukvić, N. Detection of Premature Segregation of Centromeres in Persons Exposed to Ionizing Radiation. Health Phys. 2010, 98, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Kasuba, V.; Rozgaj, R.; Jazbec, A. Chromosome Aberrations in Peripheral Blood Lymphocytes of Croatian Hospital Staff Occupationally Exposed to Low Levels of Ionising Radiation. Arh. Hig. Rada Toksikol. 2008, 59, 251–259. [Google Scholar] [CrossRef]
- Khisroon, M.; Khan, A.; Naseem, M.; Ali, N.; Khan, S.; Rasheed, S.B. Evaluation of DNA Damage in Lymphocytes of Radiology Personnel by Comet Assay. J. Occup. Health 2015, 57, 268–274. [Google Scholar] [CrossRef]
- Kopjar, N.; Garaj-Vrhovac, V. Assessment of DNA Damage in Nuclear Medicine Personnel—Comparative Study with the Alkaline Comet Assay and the Chromosome Aberration Test. Int. J. Hyg. Environ. Health 2005, 208, 179–191. [Google Scholar] [CrossRef]
- Kumar, D.; Kumari, S.; Salian, S.R.; Uppangala, S.; Kalthur, G.; Challapalli, S.; Chandraguthi, S.G.; Kumar, P.; Adiga, S.K. Genetic Instability in Lymphocytes Is Associated With Blood Plasma Antioxidant Levels in Health Care Workers Occupationally Exposed to Ionizing Radiation. Int. J. Toxicol. 2016, 35, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Lalić, H.; Lekić, A.; Radosević-Stasić, B. Comparison of Chromosome Aberrations in Peripheral Blood Lymphocytes from People Occupationally Exposed to Ionizing and Radiofrequency Radiation. Acta Med. Okayama 2001, 55, 117–127. [Google Scholar] [CrossRef]
- Little, M.P.; Kwon, D.; Doi, K.; Simon, S.L.; Preston, D.L.; Doody, M.M.; Lee, T.; Miller, J.S.; Kampa, D.M.; Bhatti, P.; et al. Association of Chromosome Translocation Rate with Low Dose Occupational Radiation Exposures in U.S. Radiologic Technologists. Radiat. Res. 2014, 182, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Maffei, F.; Angelini, S.; Forti, G.C.; Lodi, V.; Violante, F.S.; Mattioli, S.; Hrelia, P. Micronuclei Frequencies in Hospital Workers Occupationally Exposed to Low Levels of Ionizing Radiation: Influence of Smoking Status and Other Factors. Mutagenesis 2002, 17, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Maffei, F.; Angelini, S.; Forti, G.C.; Violante, F.S.; Lodi, V.; Mattioli, S.; Hrelia, P. Spectrum of Chromosomal Aberrations in Peripheral Lymphocytes of Hospital Workers Occupationally Exposed to Low Doses of Ionizing Radiation. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 2004, 547, 91–99. [Google Scholar] [CrossRef]
- Maluf, S.W.; Passos, D.F.; Bacelar, A.; Speit, G.; Erdtmann, B. Assessment of DNA Damage in Lymphocytes of Workers Exposed to X-Radiation Using the Micronucleus Test and the Comet Assay. Environ. Mol. Mutagen. 2001, 38, 311–315. [Google Scholar] [CrossRef]
- Martínez, A.; Coleman, M.; Romero-Talamás, C.A.; Frias, S. An Assessment of Immediate DNA Damage to Occupationally Exposed Workers to Low Dose Ionizing Radiation by Using the Comet Assay. Rev. Invest. Clin. 2010, 62, 23–30. [Google Scholar] [PubMed]
- Milacic, S. Frequency of Chromosomal Lesions and Damaged Lymphocytes of Workers Occupationally Exposed to x Rays. Health Phys. 2005, 88, 334–339. [Google Scholar] [CrossRef]
- Milić, M.; Rozgaj, R.; Kašuba, V.; Jazbec, A.-M.; Starčević, B.; Lyzbicki, B.; Ravegnini, G.; Zenesini, C.; Musti, M.; Hrelia, P.; et al. Polymorphisms in DNA Repair Genes: Link with Biomarkers of the CBMN Cytome Assay in Hospital Workers Chronically Exposed to Low Doses of Ionising Radiation. Arh. Hig. Rada Toksikol. 2015, 66, 109–120. [Google Scholar] [CrossRef]
- Movafagh, A.; Farajolah, A.M.; Fadaie, S.; Azargashb, E. Movafagh Persistent Unstable Chromosomal Aberrations in Lymphocytes Of Radiotherapy Workers After 1(St) Mitotic Division In Tehran, Iran. Pak. J. Med. Sci. 2007, 23, 254–258. [Google Scholar]
- Mrdjanović, J.; Jakimov, D.; Tursijan, S.; Bogdanović, G. Evaluation of Sister Chromatid Exchanges, Micronuclei, And Proliferating Rate Index In Hospital Workers Chronically Exposed To Ionizing Radiation. J. BUON 2005, 10, 99–103. [Google Scholar]
- Pajic, J.; Rakic, B.; Jovicic, D.; Milovanovic, A. A Cytogenetic Study of Hospital Workers Occupationally Exposed to Radionuclides in Serbia: Premature Centromere Division as Novel Biomarker of Exposure? Int. Arch. Occup. Environ. Health 2016, 89, 477–484. [Google Scholar] [CrossRef]
- Pajic, J.; Jovicic, D.; Ps Milovanovic, A. Micronuclei as a Marker for Medical Screening of Subjects Continuously Occupationally Exposed to Low Doses of Ionizing Radiation. Biomarkers 2017, 22, 439–445. [Google Scholar] [CrossRef]
- Pakniat, F.; Mozdarani, H.; Nasirian, B.; Faeghi, F. Radioadaptive Response in Peripheral Blood Leukocytes of Occupationally Exposed Medical Staff with Investigation of DNA Damage by the Use of Neutral Comet Assay. Int. J. Radiat. Res. 2013, 11, 91–97. [Google Scholar]
- Qian, Q.-Z.; Cao, X.-K.; Shen, F.-H.; Wang, Q. Effects of Ionising Radiation on Micronucleus Formation and Chromosomal Aberrations in Chinese Radiation Workers. Radiat. Prot. Dosim. 2016, 168, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Raavi, V.; Basheerudeen, S.A.S.; Jagannathan, V.; Joseph, S.; Chaudhury, N.K.; Venkatachalam, P. Frequency of Gamma H2AX Foci in Healthy Volunteers and Health Workers Occupationally Exposed to X-Irradiation and Its Relevance in Biological Dosimetry. Radiat. Environ. Biophys. 2016, 55, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Ropolo, M.; Balia, C.; Roggieri, P.; Lodi, V.; Nucci, M.C.; Violante, F.S.; Silingardi, P.; Colacci, A.; Bolognesi, C. The Micronucleus Assay as a Biological Dosimeter in Hospital Workers Exposed to Low Doses of Ionizing Radiation. Mutat. Res. 2012, 747, 7–13. [Google Scholar] [CrossRef]
- Sahin, A.; Tatar, A.; Oztas, S.; Seven, B.; Varoglu, E.; Yesilyurt, A.; Ayan, A.K. Evaluation of the Genotoxic Effects of Chronic Low-Dose Ionizing Radiation Exposure on Nuclear Medicine Workers. Nucl. Med. Biol. 2009, 36, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Sakly, A.; Gaspar, J.F.; Kerkeni, E.; Silva, S.; Teixeira, J.P.; Chaari, N.; Ben Cheikh, H. Genotoxic Damage in Hospital Workers Exposed to Ionizing Radiation and Metabolic Gene Polymorphisms. J. Toxicol. Environ. Health Part A 2012, 75, 934–946. [Google Scholar] [CrossRef] [PubMed]
- Sakly, A.; Ayed, Y.; Chaari, N.; Akrout, M.; Bacha, H.; Cheikh, H.B. Assessment of Chromosomal Aberrations and Micronuclei in Peripheral Lymphocytes from Tunisian Hospital Workers Exposed to Ionizing Radiation. Genet. Test. Mol. Biomark. 2013, 17, 650–655. [Google Scholar] [CrossRef]
- Santovito, A.; Cervella, P.; Delpero, M. Increased Frequency of Chromosomal Aberrations and Sister Chromatid Exchanges in Peripheral Lymphocytes of Radiology Technicians Chronically Exposed to Low Levels of Ionizing Radiations. Environ. Toxicol. Pharmacol. 2014, 37, 396–403. [Google Scholar] [CrossRef]
- Sari-Minodier, I.; Orsière, T.; Auquier, P.; Martin, F.; Botta, A. Cytogenetic Monitoring by Use of the Micronucleus Assay among Hospital Workers Exposed to Low Doses of Ionizing Radiation. Mutat. Res. 2007, 629, 111–121. [Google Scholar] [CrossRef]
- Scarpato, R.; Antonelli, A.; Ballardin, M.; Cipollini, M.; Fallahi, P.; Tomei, A.; Traino, C.; Barale, R. Analysis of Chromosome Damage in Circulating Lymphocytes of Radiological Workers Affected by Thyroid Nodules. Mutat. Res./Genet. Toxicol. Environ. Mutagenesis 2006, 606, 21–26. [Google Scholar] [CrossRef]
- Shafiee, M.; Borzoueisileh, S.; Rashidfar, R.; Dehghan, M.; Jaafarian Sisakht, Z. Chromosomal Aberrations in C-Arm Fluoroscopy, CT-Scan, Lithotripsy, and Digital Radiology Staff. Mutat. Res. 2020, 849, 503131. [Google Scholar] [CrossRef]
- Siama, Z.; Zosang-Zuali, M.; Vanlalruati, A.; Jagetia, G.C.; Pau, K.S.; Kumar, N.S. Chronic Low Dose Exposure of Hospital Workers to Ionizing Radiation Leads to Increased Micronuclei Frequency and Reduced Antioxidants in Their Peripheral Blood Lymphocytes. Int. J. Radiat. Biol. 2019, 95, 697–709. [Google Scholar] [CrossRef]
- Silva, R.G.; Oliviera Barros Alencar, M.V.; Silva Teixeira, J.; Rodrigues e Silva, R.; Paz, M.F.C.J.; de Castro e Sousa, J.M.; de Aguiar, R.P.S.; de Carvalho, R.M.; Gomerz, A.L., Jr.; da Mata, A.M.O.F.; et al. Genotoxicity and DNA Repair Indicative in Blood Cells after Occupational Exposure to Ionizing Radiation. Int. Arch. Med. 2016, 9. [Google Scholar] [CrossRef]
- Surniyantoro, H.N.E.; Lusiyanti, Y.; Rahardjo, T.; Nurhayati, S.; Tetriana, D. Association between XRCC1 Exon 10 (Arg399Gln) Gene Polymorphism and Micronucleus as a Predictor of DNA Damage among Radiation Workers. Biodiversitas J. Biol. Divers. 2018, 19, 1676–1682. [Google Scholar] [CrossRef]
- Thierens, H.; Vral, A.; Morthier, R.; Aousalah, B.; de Ridder, L. Cytogenetic Monitoring of Hospital Workers Occupationally Exposed to Ionizing Radiation Using the Micronucleus Centromere Assay. Mutagenesis 2000, 15, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Tug, E.; Kayhan, G.; Kan, D.; Guntekin, S.; Ergun, M.A. The Evaluation of Long-Term Effects of Ionizing Radiation through Measurement of Current Sister Chromatid Exchange (SCE) Rates in Radiology Technologists, Compared with Previous SCE Values. Mutat. Res. 2013, 757, 28–30. [Google Scholar] [CrossRef]
- Vellingiri, B.; Shanmugam, S.; Subramaniam, M.D.; Balasubramanian, B.; Meyyazhagan, A.; Alagamuthu, K.; Prakash, V.; Shafiahammedkhan, M.; Kathannan, S.; Pappuswamy, M.; et al. Cytogenetic Endpoints and Xenobiotic Gene Polymorphism in Lymphocytes of Hospital Workers Chronically Exposed to Ionizing Radiation in Cardiology, Radiology and Orthopedic Laboratories. Ecotoxicol. Environ. Saf. 2014, 100, 266–274. [Google Scholar] [CrossRef]
- Vral, A.; Decorte, V.; Depuydt, J.; Wambersie, A.; Thierens, H. A Semi-Automated FISH-Based Micronucleus-Centromere Assay for Biomonitoring of Hospital Workers Exposed to Low Doses of Ionizing Radiation. Mol. Med. Rep. 2016, 14, 103–110. [Google Scholar] [CrossRef]
- Wang, H.; Chen, Q.; Liu, G.; Tian, Y.; Zhang, F.; Qu, J.; Lim, D.; Feng, Z. The Comparison of Health Status Between Male and Female Medical Radiation Workers in China. Radiat. Prot. Dosim. 2017, 175, 508–516. [Google Scholar] [CrossRef]
- Zakeri, F.; Assaei, R.; Varzegar, R. Chromosomal Aberrations in Workers Occupationally Exposed to Chronic Low-Level Ionizing Radiation. Occup. Environ. Med. 2003, 9, 33–38. [Google Scholar]
- Zakeri, F.; Assaei, R.G. Cytogenetic Monitoring of Personnel Working in Angiocardiography Laboratories in Iran Hospitals. Mutat. Res. 2004, 562, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zakeri, F.; Hirobe, T. A Cytogenetic Approach to the Effects of Low Levels of Ionizing Radiations on Occupationally Exposed Individuals. Eur. J. Radiol. 2010, 73, 191–195. [Google Scholar] [CrossRef]
- Zakeri, F.; Hirobe, T.; Akbari Noghabi, K. Biological Effects of Low-Dose Ionizing Radiation Exposure on Interventional Cardiologists. Occup. Med. 2010, 60, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.D.; Yao, L.; Guo, K.M.; Lu, C.W. Cytogenetic Evaluation of Cataract Patients Occupationally Exposed to Ionizing Radiation in Northeast China. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Bhatti, P.; Doody, M.M.; Preston, D.L.; Kampa, D.; Ron, E.; Weinstock, R.W.; Simon, S.; Edwards, A.A.; Sigurdson, A.J. Increased Frequency of Chromosome Translocations Associated with Diagnostic X-Ray Examinations. Radiat. Res. 2008, 170, 149–155. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oestreicher, U.; Samaga, D.; Ainsbury, E.; Antunes, A.C.; Baeyens, A.; Barrios, L.; Beinke, C.; Beukes, P.; Blakely, W.F.; Cucu, A.; et al. RENEB Intercomparisons Applying the Conventional Dicentric Chromosome Assay (DCA). Int. J. Radiat. Biol. 2017, 93, 20–29. [Google Scholar] [CrossRef]
- Silva-Barbosa, I.; Pereira-Magnata, S.; Amaral, A.; Sotero, G.; Melo, H.C. Dose Assessment by Quantification of Chromosome Aberrations and Micronuclei in Peripheral Blood Lymphocytes from Patients Exposed to Gamma Radiation. Genet. Mol. Biol. 2005, 28, 452–457. [Google Scholar] [CrossRef]
- Rothkamm, K.; Beinke, C.; Romm, H.; Badie, C.; Balagurunathan, Y.; Barnard, S.; Bernard, N.; Boulay-Greene, H.; Brengues, M.; de Amicis, A.; et al. Comparison of Established and Emerging Biodosimetry Assays. Radiat. Res. 2013, 180, 111–119. [Google Scholar] [CrossRef][Green Version]
- Iwasaki, T.; Takashima, Y.; Suzuki, T.; Yoshida, M.A.; Hayata, I. The Dose Response of Chromosome Aberrations in Human Lymphocytes Induced in Vitro by Very Low-Dose γ Rays. Radiat. Res. 2011, 175, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Abe, Y.; Miura, T.; Yoshida, M.A.; Ujiie, R.; Kurosu, Y.; Kato, N.; Katafuchi, A.; Tsuyama, N.; Ohba, T.; Inamasu, T.; et al. Increase in Dicentric Chromosome Formation after a Single CT Scan in Adults. Sci. Rep. 2015, 5, 13882. [Google Scholar] [CrossRef]
- Golfier, S.; Jost, G.; Pietsch, H.; Lengsfeld, P.; Eckardt-Schupp, F.; Schmid, E.; Voth, M. Dicentric Chromosomes and Gamma-H2AX Foci Formation in Lymphocytes of Human Blood Samples Exposed to a CT Scanner: A Direct Comparison of Dose Response Relationships. Radiat. Prot. Dosim. 2009, 134, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Fujioka, K.; Sakurai-Ozato, N.; Fukumoto, W.; Satoh, K.; Sun, J.; Awazu, A.; Tanaka, K.; Ishida, M.; Ishida, T.; et al. Chromosomal Abnormalities in Human Lymphocytes after Computed Tomography Scan Procedure. Radiat. Res. 2018, 190, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Tucker, J.D. Low-Dose Ionizing Radiation and Chromosome Translocations: A Review of the Major Considerations for Human Biological Dosimetry. Mutat. Res. 2008, 659, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Sommer, S.; Buraczewska, I.; Kruszewski, M. Micronucleus Assay: The State of Art, and Future Directions. Int. J. Mol. Sci. 2020, 21, 1534. [Google Scholar] [CrossRef] [PubMed]
- Aguiar Torres, L.; dos Santos Rodrigues, A.; Linhares, D.; Camarinho, R.; Nunes Páscoa Soares Rego, Z.M.; Ventura Garcia, P. Buccal Epithelial Cell Micronuclei: Sensitive, Non-Invasive Biomarkers of Occupational Exposure to Low Doses of Ionizing Radiation. Mutat. Res./Genet. Toxicol. Environ. Mutagenesis 2019, 838, 54–58. [Google Scholar] [CrossRef]
- Padilla-Raygoza, N.; Del Rocio Adame Gutiérrez, M.; Martínez, I.Z.M.; Beltran-Campos, V.; Del Carmen Delgado-Sandoval, S.; de Lourdes Garcia-Campos, M.; Sosa-Aquino, M.A.; Cordova-Fraga, T.; Guzman-Cabrera, R. Evaluation of Micronuclei in Oral Mucosa of Individuals Exposed to Ionizing Radiation: A Pilot Study from Celaya, México. Cent. Asian J. Glob. Health 2019, 8, 331. [Google Scholar] [CrossRef]
- Vral, A.; Fenech, M.; Thierens, H. The Micronucleus Assay as a Biological Dosimeter of in Vivo Ionising Radiation Exposure. Mutagenesis 2011. [Google Scholar] [CrossRef]
- Nakanishi, Y.; Schneider, E.L. In Vivo Sister-Chromatid Exchange: A Sensitive Measure of DNA Damage. Mutat. Res. 1979, 60, 329–337. [Google Scholar] [CrossRef]
- Lialiaris, T.S. Sister Chromatid Exchange. In Brenner’s Encyclopedia of Genetics, 2nd ed.; Maloy, S., Hughes, K., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 454–457. ISBN 978-0-08-096156-9. [Google Scholar]
- Olive, P.L.; Banáth, J.P.; Durand, R.E. Heterogeneity in Radiation-Induced DNA Damage and Repair in Tumor and Normal Cells Measured Using the “Comet” Assay. Radiat. Res. 1990, 122, 86–94. [Google Scholar] [CrossRef]
- Møller, P.; Loft, S.; Ersson, C.; Koppen, G.; Dusinska, M.; Collins, A. On the Search for an Intelligible Comet Assay Descriptor. Front. Genet. 2014, 5. [Google Scholar] [CrossRef]
- Gedik, C.M.; Ewen, S.W.; Collins, A.R. Single-Cell Gel Electrophoresis Applied to the Analysis of UV-C Damage and Its Repair in Human Cells. Int. J. Radiat. Biol. 1992, 62, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.R.; Dobson, V.; Dušinská, M.; Kennedy, G.; Štětina, R. The Comet Assay: What Can It Really Tell Us? Mutat. Res./Fundam. Mol. Mech. Mutagenesis 1997, 375, 183–193. [Google Scholar] [CrossRef]
- Hagmar, L.; Brøgger, A.; Hansteen, I.L.; Heim, S.; Högstedt, B.; Knudsen, L.; Lambert, B.; Linnainmaa, K.; Mitelman, F.; Nordenson, I. Cancer Risk in Humans Predicted by Increased Levels of Chromosomal Aberrations in Lymphocytes: Nordic Study Group on the Health Risk of Chromosome Damage. Cancer Res. 1994, 54, 2919–2922. [Google Scholar]
- Vodicka, P.; Polivkova, Z.; Sytarova, S.; Demova, H.; Kucerova, M.; Vodickova, L.; Polakova, V.; Naccarati, A.; Smerhovsky, Z.; Ambrus, M.; et al. Chromosomal Damage in Peripheral Blood Lymphocytes of Newly Diagnosed Cancer Patients and Healthy Controls. Carcinogenesis 2010, 31, 1238–1241. [Google Scholar] [CrossRef][Green Version]
- Fenech, M.; Knasmueller, S.; Bolognesi, C.; Holland, N.; Bonassi, S.; Kirsch-Volders, M. Micronuclei as Biomarkers of DNA Damage, Aneuploidy, Inducers of Chromosomal Hypermutation and as Sources of pro-Inflammatory DNA in Humans. Mutat. Res. 2020, 786, 108342. [Google Scholar] [CrossRef] [PubMed]
- Kirsch-Volders, M.; Bolognesi, C.; Ceppi, M.; Bruzzone, M.; Fenech, M. Micronuclei, Inflammation and Auto-Immune Disease. Mutat. Res. 2020, 786, 108335. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Pieper, D.; Buechter, R.; Jerinic, P.; Eikermann, M. Overviews of Reviews Often Have Limited Rigor: A Systematic Review. J. Clin. Epidemiol. 2012, 65, 1267–1273. [Google Scholar] [CrossRef]
- Jacob, S.; Scanff, P.; Bertrand, A.; Laurier, D.; Bernier, M.-O. Use of Personal Radiation Protection Tools and Individual Dosimetric Monitoring in a Sample of Interventional Cardiologists in France, 2005–2009. Radioprotection 2014, 49, 257–260. [Google Scholar] [CrossRef]
- Andreassi, M.G.; Cioppa, A.; Botto, N.; Joksic, G.; Manfredi, S.; Federici, C.; Ostojic, M.; Rubino, P.; Picano, E. Somatic DNA Damage in Interventional Cardiologists: A Case-Control Study. FASEB J. 2005, 19, 998–999. [Google Scholar] [CrossRef]
- Bauchinger, M. Quantification of Low-Level Radiation Exposure by Conventional Chromosome Aberration Analysis. Mutat. Res./Rev. Genet. Toxicol. 1995, 339, 177–189. [Google Scholar] [CrossRef]
- Olivieri, G.; Bodycote, J.; Wolff, S. Adaptive Response of Human Lymphocytes to Low Concentrations of Radioactive Thymidine. Science 1984, 223, 594–597. [Google Scholar] [CrossRef]
- Hou, J.; Wang, F.; Kong, P.; Yu, P.K.N.; Wang, H.; Han, W. Gene Profiling Characteristics of Radioadaptive Response in AG01522 Normal Human Fibroblasts. PLoS ONE 2015, 10, e0123316. [Google Scholar] [CrossRef]
- Ebrahimian, T.G.; Beugnies, L.; Surette, J.; Priest, N.; Gueguen, Y.; Gloaguen, C.; Benderitter, M.; Jourdain, J.R.; Tack, K. Chronic Exposure to External Low-Dose Gamma Radiation Induces an Increase in Anti-Inflammatory and Anti-Oxidative Parameters Resulting in Atherosclerotic Plaque Size Reduction in ApoE-/- Mice. Radiat. Res. 2018, 189, 187–196. [Google Scholar] [CrossRef]
- Frey, B.; Hehlgans, S.; Rödel, F.; Gaipl, U.S. Modulation of Inflammation by Low and High Doses of Ionizing Radiation: Implications for Benign and Malign Diseases. Cancer Lett. 2015, 368, 230–237. [Google Scholar] [CrossRef]
- Luzhna, L.; Kovalchuk, O. Low Dose Irradiation Profoundly Affects Transcriptome and MicroRNAme in Rat Mammary Gland Tissues. Oncoscience 2014, 1, 751–762. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rödel, F.; Frey, B.; Manda, K.; Hildebrandt, G.; Hehlgans, S.; Keilholz, L.; Seegenschmiedt, M.H.; Gaipl, U.S.; Rödel, C. Immunomodulatory Properties and Molecular Effects in Inflammatory Diseases of Low-Dose x-Irradiation. Front. Oncol. 2012, 2, 120. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.R.; Kovacs, J.J.; Whalen, E.J.; Rajagopal, S.; Strachan, R.T.; Grant, W.; Towers, A.J.; Williams, B.; Lam, C.M.; Xiao, K.; et al. A Stress Response Pathway Regulates DNA Damage through β 2-Adrenoreceptors and β-Arrestin-1. Nature 2011, 477, 349–353. [Google Scholar] [CrossRef]
- Huang, P.; Huang, B.; Weng, H.; Nakayama, K.; Morimoto, K. Effects of Lifestyle on Micronuclei Frequency in Human Lymphocytes in Japanese Hard-Metal Workers. Prev. Med. 2009, 48, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Flint, M.S.; Baum, A.; Chambers, W.H.; Jenkins, F.J. Induction of DNA Damage, Alteration of DNA Repair and Transcriptional Activation by Stress Hormones. Psychoneuroendocrinology 2007, 32, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Forlenza, M.J.; Latimer, J.J.; Baum, A. The Effects of Stress on DNA Repair Capacity. Psychol. Health 2000, 15, 881–891. [Google Scholar] [CrossRef]
- Antoni, M.H.; Lutgendorf, S.K.; Cole, S.W.; Dhabhar, F.S.; Sephton, S.E.; McDonald, P.G.; Stefanek, M.; Sood, A.K. The Influence of Bio-Behavioural Factors on Tumour Biology: Pathways and Mechanisms. Nat. Rev. Cancer 2006, 6, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Niazi, Y.; Thomsen, H.; Smolkova, B.; Vodickova, L.; Vodenkova, S.; Kroupa, M.; Vymetalkova, V.; Kazimirova, A.; Barancokova, M.; Volkovova, K.; et al. DNA Repair Gene Polymorphisms and Chromosomal Aberrations in Healthy, Nonsmoking Population. DNA Repair 2021, 101, 103079. [Google Scholar] [CrossRef]
- Scholten, B.; Vlaanderen, J.; Stierum, R.; Portengen, L.; Rothman, N.; Lan, Q.; Pronk, A.; Vermeulen, R. A Quantitative Meta-Analysis of the Relation between Occupational Benzene Exposure and Biomarkers of Cytogenetic Damage. Environ. Health Perspect. 2020, 128, 87004. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).