Perspective of the Relationship between the Susceptibility to Initial SARS-CoV-2 Infectivity and Optimal Nasal Conditioning of Inhaled Air
Abstract
:1. Background to SARS-CoV-2 and COVID-19
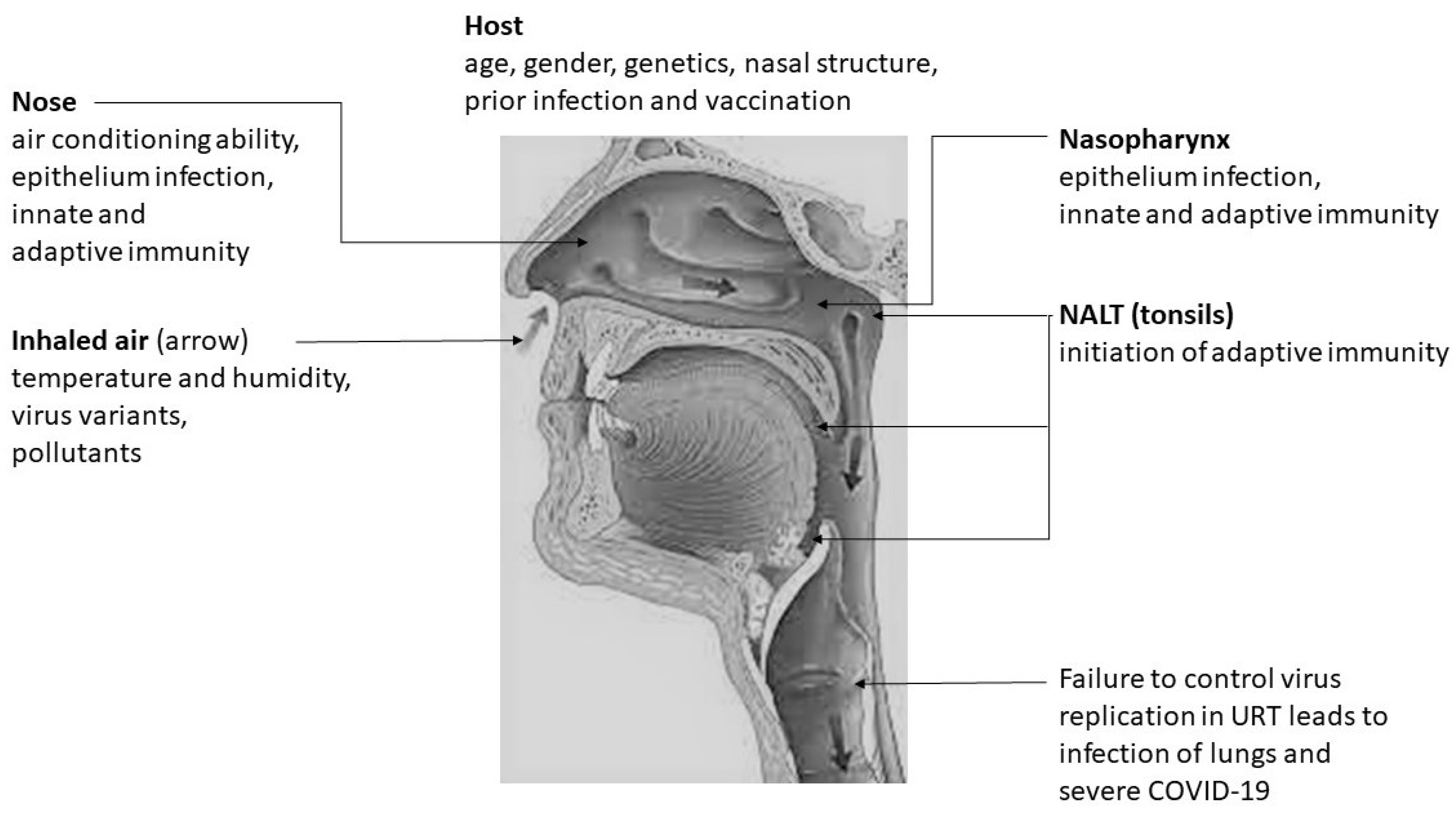
2. SARS-CoV-2 Infection in the Upper Respiratory Tract
3. Physiological Importance of the Nasal Conditioning of Inspired Air
4. Innate and Adaptive Immune Response in the Upper Respiratory Tract in Protection against SARS-CoV-2 Infection
5. Humidity of Inspired Air and Protection against SARS-CoV-2 Infection
6. Temperature of Inspired Air and Protection against SARS-CoV-2 Infection
7. Nasal Air Conditioning and Genetic Differences in Susceptibility to SARS-CoV-2 Infection
8. Differences in Nasal Air Conditioning and the Age and Gender Differences in Susceptibility to SARS-CoV-2 Infection
9. Other Factors Influencing Susceptibility to SARS-CoV-2 Infection
10. Conclusions
Funding
Conflicts of Interest
References
- Johns Hopkins University of Medicine, Coronavirus Resource Center. Global Map. Available online: https://coronavirus.jhu.edu/map.html (accessed on 21 June 2021).
- Hartenian, E.; Nandakumar, D.; Lari, A.; Ly, M.; Tucker, J.M.; Glaunsinger, B.A. The molecular virology of coronaviruses. J. Biol. Chem. 2020, 295, 12910–12934. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 2020, 181, 281–292. [Google Scholar] [CrossRef]
- Papa, G.; Mallery, D.L.; Albecka, A.; Welch, L.G.; Cattin-Ortolá, J.; Luptak, J.; Paul, D.; McMahon, H.T.; Goodfellow, I.G.; Carter, A.; et al. Furin cleavage of SARS-CoV-2 Spike promotes but is not essential for infection and cell-cell fusion. PLoS Pathog. 2021, 17, e1009246. [Google Scholar] [CrossRef]
- Jones, R.M. Relative contributions of transmission routes for COVID-19 among healthcare personnel providing patient care. J. Occup. Environ. Hyg. 2020, 17, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Lednicky, J.A.; Lauzardo, M.; Fan, Z.H.; Jutla, A.; Tilly, T.B.; Gangwar, M.; Usmani, M.; Shankar, S.N.; Mohamed, K.; Eiguren-Fernandez, A.; et al. Viable SARS-CoV-2 in the air of a hospital room with COVID-19 patients. Int. J. Infect. Dis. 2020, 100, 476–482. [Google Scholar] [CrossRef]
- Santarpia, J.L.; Rivera, D.N.; Herrera, V.L.; Morwitzer, M.J.; Creager, H.M.; Santarpia, G.W.; Crown, K.K.; Brett-Major, M.; Schnaubelt, R.; Broadhurst, J.; et al. Aerosol and surface contamination of SARS-CoV-2 observed in quarantine and isolation care. Sci. Rep. 2020, 10, 12732. [Google Scholar] [CrossRef]
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Müller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 581, 465–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, L.; Yao, M.; Zhang, X.; Hu, B.; Li, X.; Chen, H.; Zhang, L.; Liu, Y.; Du, M.; Sun, B.; et al. Breath-, air- and surface-borne SARS-CoV-2 in hospitals. J. Aerosol. Sci. 2020, 152, 105693. [Google Scholar] [CrossRef] [PubMed]
- Sette, A.; Crotty, S. Adaptive immunity to SARS-CoV-2 and COVID-19. Cell 2021, 184, 861–880. [Google Scholar] [CrossRef]
- Cao, X. COVID-19: Immunopathology and its implications for therapy. Nat. Rev. Immunol. 2020, 20, 269–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denney, L.; Ho, L.-P. The role of respiratory epithelium in host defence against influenza virus infection. Biomed. J. 2018, 41, 218–233. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, R. Immunity to human influenza A—An overview. Brunei Darussalam J. Health. 2010, 4, 1–8. [Google Scholar]
- Nguyen, T.H.O.; Koutsakos, M.; van de Sandt, C.E.; Crawford, J.C.; Loh, L.; Sant, S.; Grzelak, L.; Allen, E.K.; Brahm, T.; Clemens, E.B.; et al. Immune cellular networks underlying recovery from influenza virus infection in acute hospitalized patients. Nat. Commun. 2021, 12, 1–17. [Google Scholar] [CrossRef]
- Shah, J.; Liu, S.; Potula, H.-H.; Bhargava, P.; Cruz, I.; Force, D.; Bazerbashi, A.; Ramasamy, R. IgG and IgM antibody formation to spike and nucleocapsid proteins in COVID-19 characterized by multiplex immunoblot assays. BMC Infect. Dis. 2021, 21, 325. [Google Scholar] [CrossRef]
- Tamerius, J.D.; Shaman, J.; Alonso, W.; Bloom-Feshbach, K.; Uejio, C.K.; Comrie, A.; Viboud, C. Environmental predictors of seasonal influenza epidemics across temperate and tropical climates. PLOS Pathog. 2013, 9, e1003194. [Google Scholar] [CrossRef]
- Paynter, S. Humidity and respiratory virus transmission in tropical and temperate settings. Epidemiol. Infect. 2015, 143, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, M.; Hugentobler, W.J.; Iwasaki, A. Seasonality of respiratory viral infections. Annu. Rev. Virol. 2020, 7, 83–101. [Google Scholar] [CrossRef]
- Baker, R.E.; Yang, W.; Vecchi, G.A.; Metcalf, C.J.E.; Grenfell, B.T. Assessing the influence of climate on wintertime SARS-CoV-2 outbreaks. Nat. Commun. 2021, 12, 846. [Google Scholar] [CrossRef]
- Cunningham, L.; Nicholson, P.J.; O’Connor, J.; McFadden, J.P. Cold working environments as an occupational risk factor for COVID-19. Occup. Med. 2020, 195, kqaa195. [Google Scholar] [CrossRef] [PubMed]
- Livadiotis, G. Statistical analysis of the impact of environmental temperature on the exponential growth rate of cases infected by COVID-19. PLoS ONE 2020, 15, e0233875. [Google Scholar] [CrossRef] [PubMed]
- Kaplin, A.; Junker, C.; Kumar, A.; Ribeiro, M.A.; Yu, E.; Wang, M.; Smith, T.; Rai, S.N.; Bhatnagar, A. Evidence and magnitude of the effects of meteorological changes on SARS-CoV-2 transmission. PLoS ONE 2021, 16, e0246167. [Google Scholar] [CrossRef]
- Mathur, R.; Rentsch, C.T.; E Morton, C.; Hulme, W.J.; Schultze, A.; MacKenna, B.; Eggo, R.M.; Bhaskaran, K.; Wong, A.Y.S.; Williamson, E.J.; et al. Ethnic differences in SARS-CoV-2 infection and COVID-19-related hospitalisation, intensive care unit admission, and death in 17 million adults in England: An observational cohort study using the OpenSAFELY platform. Lancet 2021, 397, 1711–1724. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. COVID-19 in Racial and Ethnic Minority Groups. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/racial-ethnic-minorities.html (accessed on 4 May 2021).
- O’Driscoll, M.; Dos Santos, G.R.; Wang, L.; Cummings, D.A.T.; Azman, A.S.; Palreau, J.; Fontanet, A.; Cauchemez, S.; Salje, H. Age-specific mortality and immunity patterns of SARS-CoV-2. Nature 2021, 590, 140–145. [Google Scholar] [CrossRef] [PubMed]
- The British Society for Immunology. The Ageing Immune System and COVID-19. Available online: https://www.immunology.org/sites/default/files/BSI_Ageing_COVID-19_Report_Nov2020_FINAL.pdf. (accessed on 17 May 2021).
- Du, P.; Li, D.; Wang, A.; Shen, S.; Ma, Z.; Li, X. A systematic review and meta-analysis of risk factors associated with severity and death in COVID-19 patients. Can. J. Infect. Dis. Med. Microbiol. 2021, 2021, 1–12. [Google Scholar] [CrossRef]
- Ramasamy, R. Nasal conditioning of inspired air, innate immunity in the respiratory tract and SARS-CoV-2 infectivity. Open Sci. Forum 2020. [Google Scholar] [CrossRef]
- Hou, Y.J.; Okuda, K.; Edwards, C.E.; Martinez, D.R.; Asakura, T.; Dinnon, K.H.; Kato, T.; Lee, R.E.; Yount, B.L.; Mascenik, T.M.; et al. SARS-CoV-2 reverse genetics reveals a variable infection gradient in the respiratory tract. Cell 2020, 182, 429–446.e14. [Google Scholar] [CrossRef] [PubMed]
- Sungnak, W.; Huang, N.; Bécavin, C.; Berg, M.; Queen, R.; Litvinukova, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Sampaziotis, F.; et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat. Med. 2020, 26, 681–687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zou, L.; Ruan, F.; Huang, M.; Liang, L.; Huang, H.; Hong, Z.; Yu, J.; Kang, M.; Song, Y.; Xia, J.; et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N. Engl. J. Med. 2020, 382, 1177–1179. [Google Scholar] [CrossRef]
- Richard, M.; Brand, J.M.A.V.D.; Bestebroer, T.M.; Lexmond, P.; De Meulder, D.; Fouchier, R.; Lowen, A.C.; Herfst, S. Influenza A viruses are transmitted via the air from the nasal respiratory epithelium of ferrets. Nat. Commun. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Gaeckle, N.T.; A Pragman, A.; Pendleton, K.M.; Baldomero, A.K.; Criner, G.J. The oral-lung axis: The impact of oral health on lung health. Respir. Care 2020, 65, 1211–1220. [Google Scholar] [CrossRef]
- Elad, D.; Wolf, M.; Keck, T. Air-conditioning in the human nasal cavity. Respir. Physiol. Neurobiol. 2008, 163, 121–127. [Google Scholar] [CrossRef]
- Williams, R.; Rankin, N.; Smith, T.; Galler, D.; Seakins, P. Relationship between the humidity and temperature of inspired gas and the function of the airway mucosa. Crit. Care Med. 1996, 24, 1920–1929. [Google Scholar] [CrossRef] [PubMed]
- Abbott, D.J.; Baroody, F.M.; Naureckas, E.; Naclerio, R.M. Elevation of nasal mucosal temperature increases the ability of the nose to warm and humidify air. Am. J. Rhinol. 2001, 15, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Foxman, E.F.; Molony, R.D. Early local immune defences in the respiratory tract. Nat. Rev. Immunol. 2017, 17, 7–20. [Google Scholar] [CrossRef] [Green Version]
- Takaki, H.; Ichimiya, S.; Matsumoto, M.; Seya, T. Mucosal immune response in nasal-associated lymphoid tissue upon intranasal administration by adjuvants. J. Innate Immun. 2018, 10, 515–521. [Google Scholar] [CrossRef] [PubMed]
- McMahan, K.; Yu, J.; Mercado, N.B.; Loos, C.; Tostanoski, L.H.; Chandrashekar, A.; Liu, J.; Peter, L.; Atyeo, C.; Zhu, A.; et al. Correlates of protection against SARS-CoV-2 in rhesus macaques. Nature 2021, 590, 630–634. [Google Scholar] [CrossRef]
- Barrett, J.R.; Belij-Rammerstorfer, S.; Dold, C.; Ewer, K.J.; Folegatti, P.M.; Gilbride, C.; Halkerston, R.; Hill, J.; Jenkin, D.; Stockdale, L.; et al. Phase 1/2 trial of SARS-CoV-2 vaccine ChAdOx1 nCoV-19 with a booster dose induces multifunctional antibody responses. Nat. Med. 2021, 27, 279–288. [Google Scholar] [CrossRef]
- Ewer, K.J.; Barrett, J.R.; Belij-Rammerstorfer, S.; Sharpe, H.; Makinson, R.; Morter, R.; Flaxman, A.; Wright, D.; Bellamy, D.; Bittaye, M.; et al. T cell and antibody responses induced by a single dose of ChAdOx1 nCoV-19 (AZD1222) vaccine in a phase 1/2 clinical trial. Nat. Med. 2021, 27, 270–278. [Google Scholar] [CrossRef]
- Ramasamy, M.N.; Minassian, A.M.; Ewer, K.J.; Flaxman, A.L.; Folegatti, P.M.; Owens, D.R.; Voysey, M.; Aley, P.K.; Angus, B.; Babbage, G.; et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): A single-blind, randomised, controlled, phase 2/3 trial. Lancet 2020, 396, 1979–1993. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, E.Q.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D.; et al. COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T-cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA vaccine against SARS-CoV-2—preliminary report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 2020, 181, 1489–1501.e1415. [Google Scholar] [CrossRef] [PubMed]
- Nelde, A.; Bilich, T.; Heitmann, J.S.; Maringer, Y.; Salih, H.R.; Roerden, M.; Lübke, M.; Bauer, J.; Rieth, J.; Wacker, M.; et al. SARS-CoV-2-derived peptides define heterologous and COVID-19-induced T cell recognition. Nat. Immunol. 2021, 22, 74–85. [Google Scholar] [CrossRef]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Yu, E.D.; Faliti, C.E.; Grifoni, A.; Ramirez, S.I.; Haupt, S.; Frazier, A.; et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef]
- Kudo, E.; Song, E.; Yockey, L.J.; Rakib, T.; Wong, P.W.; Homer, R.J.; Iwasaki, A. Low ambient humidity impairs barrier function and innate resistance against influenza infection. Proc. Natl. Acad. Sci. USA 2019, 116, 10905–10910. [Google Scholar] [CrossRef] [Green Version]
- Matson, M.J.; Yinda, C.K.; Seifert, S.N.; Bushmaker, T.; Fischer, R.J.; Van Doremalen, N.; Lloyd-Smith, J.O.; Munster, V.J. Effect of environmental conditions on SARS-CoV-2 stability in human nasal mucus and sputum. Emerg. Infect. Dis. 2020, 26, 2276–2278. [Google Scholar] [CrossRef] [PubMed]
- Lazear, H.M.; Schoggins, J.W.; Diamond, M.S. Shared and distinct functions of Type I and Type III interferons. Immunity 2019, 50, 907–923. [Google Scholar] [CrossRef] [PubMed]
- Sa Ribero, M.; Jouvenet, N.; Dreux, M.; Nisole, S. Interplay between SARS-CoV-2 and the type I interferon response. PLoS Pathog. 2020, 16, e1008737. [Google Scholar] [CrossRef] [PubMed]
- Foxman, E.F.; Storer, J.A.; Fitzgerald, M.E.; Wasik, B.R.; Hou, L.; Zhao, H.; Turner, P.E.; Pyle, A.M.; Iwasaki, A. Temperature-dependent innate defense against the common cold virus limits viral replication at warm temperature in mouse airway cells. Proc. Natl. Acad. Sci. USA 2015, 112, 827–832. [Google Scholar] [CrossRef] [Green Version]
- Foxman, E.F.; Storer, J.A.; Vanaja, K.; Levchenko, A.; Iwasaki, A. Two interferon-independent double-stranded RNA-induced host defense strategies suppress the common cold virus at warm temperature. Proc. Natl. Acad. Sci. USA 2016, 113, 8496–8501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- V’Kovski, P.; Gultom, M.; Kelly, J.N.; Steiner, S.; Russeil, J.; Mangeat, B.; Cora, E.; Pezoldt, J.; Holwerda, M.; Kratzel, A.; et al. Disparate temperature-dependent virus–host dynamics for SARS-CoV-2 and SARS-CoV in the human respiratory epithelium. PLoS Biol. 2021, 19, e3001158. [Google Scholar] [CrossRef] [PubMed]
- Vanderheiden, A.; Ralfs, P.; Chirkova, T.; Upadhyay, A.A.; Zimmerman, M.G.; Bedoya, S.; Aoued, H.; Tharp, G.M.; Pellegrini, K.L.; Manfredi, C.; et al. Type I and type III interferons restrict SARS-CoV-2 infection of human airway epithelial cultures. J. Virol. 2020, 94. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Bastard, P.; Liu, Z.; Le Pen, J.; Moncada-Velez, M.; Chen, J.; Ogishi, M.; Sabli, I.K.D.; Hodeib, S.; Korol, C.; et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science 2020, 370, eabd4570. [Google Scholar] [CrossRef]
- Pairo-Castineira, E.; Clohisey, S.; Klaric, L.; Bretherick, A.D.; Rawlik, K.; Pasko, D.; Walker, S.; Parkinson, N.; Fourman, M.H.; Russell, C.D.; et al. Genetic mechanisms of critical illness in COVID-19. Nature 2021, 591, 92–98. [Google Scholar] [CrossRef]
- Noback, M.L.; Harvati, K.; Spoor, F. Climate-related variation of the human nasal cavity. Am. J. Phys. Anthr. 2011, 145, 599–614. [Google Scholar] [CrossRef]
- Zeberg, H.; Pääbo, S. The major genetic risk factor for severe COVID-19 is inherited from Neanderthals. Nat. Cell Biol. 2020, 587, 610–612. [Google Scholar] [CrossRef] [PubMed]
- Zeberg, H.; Pääbo, S. A genomic region associated with protection against severe COVID-19 is inherited from Neandertals. Proc. Natl. Acad. Sci. USA 2021, 118, 2026309118. [Google Scholar] [CrossRef] [PubMed]
- Chapman, S.J.; Hill, A.V.S. Human genetic susceptibility to infectious disease. Nat. Rev. Genet. 2012, 13, 175–188. [Google Scholar] [CrossRef]
- Allison, A.C. Protection afforded by sickle-cell trait against subtertian malareal infection. Br. Med. J. 1954, 1, 290–294. [Google Scholar] [CrossRef] [Green Version]
- Ramasamy, R. Zoonotic malaria-global overview and research and policy needs. Front. Public Health. 2014, 2, 123. [Google Scholar] [CrossRef] [Green Version]
- Ramasamy, R. Mosquito vector proteins homologous to α1-3 galactosyl transferases of tick vectors in the context of protective immunity against malaria and hypersensitivity to vector bites. Parasites Vectors 2021, 14, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shelton, J.F.; Shastri, A.J.; Ye, C.; Weldon, C.H.; Filshtein-Sonmez, T.; Coker, D.; Symons, A.; Esparza-Gordillo, J.; Aslibekyan, S.; Auton, A.; et al. Trans-ancestry analysis reveals genetic and nongenetic associations with COVID-19 susceptibility and severity. Nat. Genet. 2021, 53, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Maddux, S.D.; Butaric, L.; Yokley, T.R.; Franciscus, R.G. Ecogeographic variation across morphofunctional units of the human nose. Am. J. Phys. Anthr. 2016, 162, 103–119. [Google Scholar] [CrossRef]
- Zaidi, A.A.; Mattern, B.C.; Claes, P.; McEcoy, B.; Hughes, C.; Shriver, M. Investigating the case of human nose shape and climate adaptation. PLoS Genet. 2017, 13, e1006616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiner, J.S. Nose shape and climate. Am. J. Phys. Anthr. 1954, 12, 615–618. [Google Scholar] [CrossRef]
- Lindemann, J.; Sannwald, D.; Wiesmiller, K. Age-related changes in intranasal air conditioning in the elderly. Laryngoscope 2008, 118, 1472–1475. [Google Scholar] [CrossRef]
- Ganjaei, K.; Soler, Z.; Mappus, E.; Worley, M.; Rowan, N.; Garcia, G.; Matthews, L.; Dubno, J.; Eckert, M.; Schlosser, R. Radiologic changes in the aging nasal cavity. Rhinol. J. 2019, 57, 117–124. [Google Scholar] [CrossRef]
- Gadi, N.; Wu, S.C.; Spihlman, A.P.; Moulton, V.R. What’s sex got to do with COVID-19? Gender-based differences in the host immune response to coronaviruses. Front. Immunol. 2020, 11, 2147. [Google Scholar] [CrossRef] [PubMed]
- Filippini, T.; Rothman, K.J.; Cocchio, S.; Narne, E.; Mantoan, D.; Saia, M.; Goffi, A.; Ferrari, F.; Maffeis, G.; Orsini, N.; et al. Associations between mortality from COVID-19 in two Italian regions and outdoor air pollution as assessed through tropospheric nitrogen dioxide. Sci. Total. Environ. 2021, 760, 143355. [Google Scholar] [CrossRef]
- Suzaki, I.; Kobayashi, H. Coronavirus disease 2019 and nasal conditions: a review of current evidence. In Vivo 2021, 35, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Francisco, D.; Conway, M.; Martinez, F.D.; Vercelli, D.; Polverino, F.; Billheimer, D.; Kraft, M. Type 2 inflammation modulates ACE2 and TMPRSS2 in airway epithelial cells. J. Allergy Clin. Immunol. 2020, 146, 80–88.e8. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Ginn, H.M.; Dejnirattisai, W.; Supasa, P.; Wang, B.; Tuekprakhon, A.; Nutalai, R.; Zhou, D.; Mentzer, A.J.; Zhao, Y.; et al. Reduced neutralization of SARS-CoV-2 B.1.617 by vaccine and convalescent serum. Cell 2021. [Google Scholar] [CrossRef]
| Induction | Effector Cell or Molecule | Effector Mechanism |
|---|---|---|
| - | Naturally occurring mucins, defensins and collectins | Bind virion and prevent cell binding and entry |
| Altered surface of the virion and virus-infected cells | Complement | Activation through the alternate or lectin pathway to promote lysis and opsonisation, inflammation |
| Pathogen associated molecular pattern (PAMP) recognition by pattern recognition receptors (PRRs) | Type 1 (α,β) and Type 3 (λ) interferons (IFNs) | Induction of anti-viral state in infected and neighbouring cells through inhibition of protein synthesis and mRNA degradation. Activation of phagocytic cells and dendritic cells |
| PRR | Inflammasome in macrophages and dendritic cells | Production of IL-1, IL-6 and TNF that promote an inflammatory response in tissue, fever and the synthesis of acute phase proteins |
| PRR | Macrophage and dendritic cell synthesis of IL-12, IL-18 | Activation of NK cells to lyse virus infected cells and enhancement of adaptive immune response |
| Stress molecules expressed by infected cells | γδT cells secreting Type 2 IFNγ | Activation of NK cells, phagocytes, dendritic cells and the adaptive immune response |
| Effector Molecule or Cell | Mechanism of Action |
|---|---|
| Secreted IgA antibodies in mucus | Prevention of virion binding to epithelial cells by agglutination and neutralization of virions |
| IgG and IgM antibodies in mucosa and blood, including anti-A and anti-B blood group antibodies | Prevention of virion binding to host cells through agglutination and neutralization, activation of complement through the classical pathway, promoting opsonisation and phagocytosis, assisting NK cell killing through Fcγ receptors |
| CD4+ TH lymphocytes | Activation of B cells, promoting immunoglobulin class switching and affinity maturation, secretion of cytokines like IFNγ that activate phagocytes and NK cells and upregulate major histocompatibility complex molecules. |
| CD8+ cytotoxic lymphocytes | Apoptosis of virus-infected cells by granzyme, perforin, etc. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramasamy, R. Perspective of the Relationship between the Susceptibility to Initial SARS-CoV-2 Infectivity and Optimal Nasal Conditioning of Inhaled Air. Int. J. Mol. Sci. 2021, 22, 7919. https://doi.org/10.3390/ijms22157919
Ramasamy R. Perspective of the Relationship between the Susceptibility to Initial SARS-CoV-2 Infectivity and Optimal Nasal Conditioning of Inhaled Air. International Journal of Molecular Sciences. 2021; 22(15):7919. https://doi.org/10.3390/ijms22157919
Chicago/Turabian StyleRamasamy, Ranjan. 2021. "Perspective of the Relationship between the Susceptibility to Initial SARS-CoV-2 Infectivity and Optimal Nasal Conditioning of Inhaled Air" International Journal of Molecular Sciences 22, no. 15: 7919. https://doi.org/10.3390/ijms22157919






