Molecular and Cellular Insights into the Development of Uterine Fibroids
Abstract
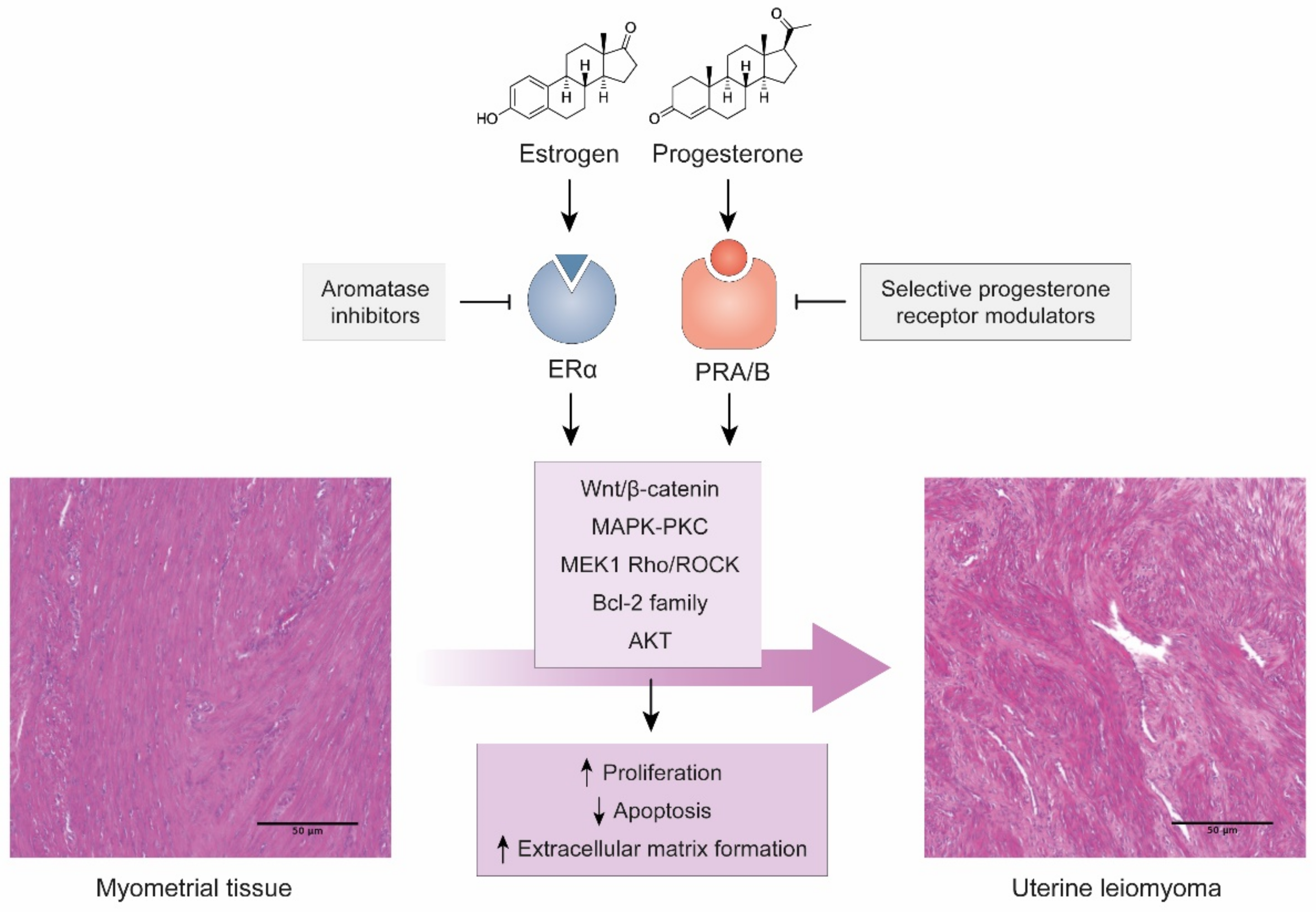
:1. Introduction
2. Uterine Leiomyoma Etiopathology
2.1. Hormone Features
2.2. Genetics
2.2.1. Inherited Susceptibility
2.2.2. Point Mutations
2.2.3. Chromosomal Abnormalities
- Chromothripsis
- Target genes of chromosomal alterations
2.3. Epigenetics
2.4. Cell Origin
3. Molecular Tools in Clinical Management of Uterine Leiomyomas
3.1. Detection–Putative Biomarkers
3.2. Classification–Gene Expression Profiling
3.3. Treatment–Targetable Dysregulated Pathways
3.3.1. Wnt/β-Catenin Pathway
3.3.2. PI3K/AKT Pathway
3.3.3. TGF-β Pathway
4. Clinical Translation and Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baird, D.; Dunson, D.; Hill, M.; Cousins, D.; Schectman, J. High cumulative incidence of uterine leiomyoma in black and white women: Ultrasound evidence. Am. J. Obstet. Gynecol. 2003, 188, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Stewart, E.A.; Cookson, C.L.; Gandolfo, R.A.; Schulze-Rath, R. Epidemiology of uterine fibroids: A systematic review. BJOG 2017, 124, 1501–1512. [Google Scholar] [CrossRef]
- Marsh, E.E.; Ekpo, G.E.; Cardozo, E.R.; Brocks, M.; Dune, T.; Cohen, L.S. Racial differences in fibroid prevalence and ultrasound findings in asymptomatic young women (18–30 years old): A pilot study. Fertil. Steril. 2013, 99, 1951–1957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marsh, E.E.; Al-Hendy, A.; Kappus, D.; Galitsky, A.; Stewart, E.A.; Kerolous, M. Burden, Prevalence, and Treatment of Uterine Fibroids: A Survey of U.S. Women. J. Women’s Health 2018, 27, 1359–1367. [Google Scholar] [CrossRef] [Green Version]
- Spies, J.B.; Myers, E.R.; Worthington-Kirsch, R.; Mulgund, J.; Goodwin, S.; Mauro, M. The FIBROID Registry: Symptom and quality-of-life status 1 year after therapy. Obstet. Gynecol. 2005, 106, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Downes, E.; Sikirica, V.; Gilabert-Estelles, J.; Bolge, S.C.; Dodd, S.L.; Maroulis, C.; Subramanian, D. The burden of uterine fibroids in five European countries. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 152, 96–102. [Google Scholar] [CrossRef]
- Friedman, A.A.; Letai, A.; Fisher, D.E.; Flaherty, K.T. Precision medicine for cancer with next-generation functional diagnostics. Nat. Rev. Cancer 2015, 15, 747–756. [Google Scholar] [CrossRef]
- Prasad, V.; Fojo, T.; Brada, M. Precision oncology: Origins, optimism, and potential. Lancet Oncol. 2016, 17, e81–e86. [Google Scholar] [CrossRef]
- Siravegna, G.; Marsoni, S.; Siena, S.; Bardelli, A. Integrating liquid biopsies into the management of cancer. Nat. Rev. Clin. Oncol. 2017, 14, 531–548. [Google Scholar] [CrossRef] [PubMed]
- Lethaby, A.; Vollenhoven, B. Fibroids (uterine myomatosis, leiomyomas). BMJ Clin. Evid. 2015, 2015, 814. [Google Scholar]
- Marsh, E.E.; Bulun, S.E. Steroid hormones and leiomyomas. Obstet. Gynecol. Clin. N. Am. 2006, 33, 59–67. [Google Scholar] [CrossRef]
- Deng, L.; Wu, T.; Chen, X.Y.; Xie, L.; Yang, J. Selective estrogen receptor modulators (SERMs) for uterine leiomyomas. Cochrane Database Syst. Rev. 2012, 10, Cd005287. [Google Scholar] [CrossRef] [PubMed]
- Tristan, M.; Orozco, L.J.; Steed, A.; Ramírez-Morera, A.; Stone, P. Mifepristone for uterine fibroids. Cochrane Database Syst. Rev. 2012, 8, Cd007687. [Google Scholar] [CrossRef] [PubMed]
- Sangkomkamhang, U.S.; Lumbiganon, P.; Pattanittum, P. Progestogens or progestogen-releasing intrauterine systems for uterine fibroids (other than preoperative medical therapy). Cochrane Database Syst. Rev. 2020, 11, Cd008994. [Google Scholar] [CrossRef]
- D’Aloisio, A.A.; Baird, D.D.; DeRoo, L.A.; Sandler, D.P. Association of intrauterine and early-life exposures with diagnosis of uterine leiomyomata by 35 years of age in the Sister Study. Environ. Health Perspect. 2010, 118, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.A.; Palmer, J.R.; Harlow, B.L.; Spiegelman, D.; Stewart, E.A.; Adams-Campbell, L.L.; Rosenberg, L. Reproductive factors, hormonal contraception, and risk of uterine leiomyomata in African-American women: A prospective study. Am. J. Epidemiol. 2004, 159, 113–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Velez Edwards, D.R.; Baird, D.D.; Hartmann, K.E. Association of age at menarche with increasing number of fibroids in a cohort of women who underwent standardized ultrasound assessment. Am. J. Epidemiol. 2013, 178, 426–433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wise, L.A.; Laughlin-Tommaso, S.K. Epidemiology of Uterine Fibroids: From Menarche to Menopause. Clin. Obstet. Gynecol. 2016, 59, 2–24. [Google Scholar] [CrossRef] [Green Version]
- Ciavattini, A.; Di Giuseppe, J.; Stortoni, P.; Montik, N.; Giannubilo, S.R.; Litta, P.; Islam, M.S.; Tranquilli, A.L.; Reis, F.M.; Ciarmela, P. Uterine fibroids: Pathogenesis and interactions with endometrium and endomyometrial junction. Obstet. Gynecol. Int. 2013, 2013, 173184. [Google Scholar] [CrossRef]
- Reis, F.M.; Bloise, E.; Ortiga-Carvalho, T.M. Hormones and pathogenesis of uterine fibroids. Best Pract. Res. Clin. Obstet. Gynecol. 2016, 34, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, S.K.; Herring, A.H.; Savitz, D.A.; Olshan, A.F.; Fielding, J.R.; Hartmann, K.E.; Baird, D.D. Pregnancy-related fibroid reduction. Fertil. Steril. 2010, 94, 2421–2423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maruo, T.; Ohara, N.; Wang, J.; Matsuo, H. Sex steroidal regulation of uterine leiomyoma growth and apoptosis. Hum. Reprod. Update 2004, 10, 207–220. [Google Scholar] [CrossRef] [Green Version]
- Sabry, M.; Al-Hendy, A. Innovative oral treatments of uterine leiomyoma. Obstet. Gynecol. Int. 2012, 2012, 943635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Omar, M.; Laknaur, A.; Al-Hendy, A.; Yang, Q. Myometrial progesterone hyper-responsiveness associated with increased risk of human uterine fibroids. BMC Womens Health 2019, 19, 92. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Ishi, K.; Serna, V.A.; Kakazu, R.; Bulun, S.E.; Kurita, T. Progesterone is essential for maintenance and growth of uterine leiomyoma. Endocrinology 2010, 151, 2433–2442. [Google Scholar] [CrossRef] [Green Version]
- Yin, P.; Lin, Z.; Cheng, Y.H.; Marsh, E.E.; Utsunomiya, H.; Ishikawa, H.; Xue, Q.; Reierstad, S.; Innes, J.; Thung, S.; et al. Progesterone receptor regulates Bcl-2 gene expression through direct binding to its promoter region in uterine leiomyoma cells. J. Clin. Endocrinol. Metab. 2007, 92, 4459–4466. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Feng, Q.; Wang, Z.; Liu, P.; Cui, S. Progesterone receptor antagonist provides palliative effects for uterine leiomyoma through a Bcl-2/Beclin1-dependent mechanism. Biosci. Rep. 2019, 39, BSR20190094. [Google Scholar] [CrossRef] [Green Version]
- Hoekstra, A.V.; Sefton, E.C.; Berry, E.; Lu, Z.; Hardt, J.; Marsh, E.; Yin, P.; Clardy, J.; Chakravarti, D.; Bulun, S.; et al. Progestins activate the AKT pathway in leiomyoma cells and promote survival. J. Clin. Endocrinol. Metab. 2009, 94, 1768–1774. [Google Scholar] [CrossRef]
- Cordeiro Mitchell, C.N.; Islam, M.S.; Afrin, S.; Brennan, J.; Psoter, K.J.; Segars, J.H. Mechanical stiffness augments ligand-dependent progesterone receptor B activation via MEK 1/2 and Rho/ROCK-dependent signaling pathways in uterine fibroid cells. Fertil. Steril. 2021, 116, 255–265. [Google Scholar] [CrossRef]
- Engman, M.; Granberg, S.; Williams, A.R.; Meng, C.X.; Lalitkumar, P.G.; Gemzell-Danielsson, K. Mifepristone for treatment of uterine leiomyoma. A prospective randomized placebo controlled trial. Hum. Reprod. 2009, 24, 1870–1879. [Google Scholar] [CrossRef] [Green Version]
- Murji, A.; Whitaker, L.; Chow, T.L.; Sobel, M.L. Selective progesterone receptor modulators (SPRMs) for uterine fibroids. Cochrane Database Syst. Rev. 2017, 4, Cd010770. [Google Scholar] [CrossRef] [PubMed]
- Hodges, L.C.; Houston, K.D.; Hunter, D.S.; Fuchs-Young, R.; Zhang, Z.; Wineker, R.C.; Walker, C.L. Transdominant suppression of estrogen receptor signaling by progesterone receptor ligands in uterine leiomyoma cells. Mol. Cell. Endocrinol. 2002, 196, 11–20. [Google Scholar] [CrossRef]
- Hassan, M.H.; Salama, S.A.; Arafa, H.M.; Hamada, F.M.; Al-Hendy, A. Adenovirus-mediated delivery of a dominant-negative estrogen receptor gene in uterine leiomyoma cells abrogates estrogen- and progesterone-regulated gene expression. J. Clin. Endocrinol. Metab. 2007, 92, 3949–3957. [Google Scholar] [CrossRef] [Green Version]
- Barbarisi, A.; Petillo, O.; Di Lieto, A.; Melone, M.A.; Margarucci, S.; Cannas, M.; Peluso, G. 17-beta estradiol elicits an autocrine leiomyoma cell proliferation: Evidence for a stimulation of protein kinase-dependent pathway. J. Cell. Physiol. 2001, 186, 414–424. [Google Scholar] [CrossRef]
- Hermon, T.L.; Moore, A.B.; Yu, L.; Kissling, G.E.; Castora, F.J.; Dixon, D. Estrogen receptor alpha (ERalpha) phospho-serine-118 is highly expressed in human uterine leiomyomas compared to matched myometrium. Virchows Arch. 2008, 453, 557–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, N.; Guan, Q.; Zheng, L.; Qu, X.; Dai, H.; Cheng, Z. Estrogen-mediated activation of fibroblasts and its effects on the fibroid cell proliferation. Transl. Res. 2014, 163, 232–241. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, G.; Li, B.; Qu, J.; Zhang, Y. LncRNA APTR Promotes Uterine Leiomyoma Cell Proliferation by Targeting ERα to Activate the Wnt/β-Catenin Pathway. Front. Oncol. 2021, 11, 536346. [Google Scholar] [CrossRef]
- Song, H.; Lu, D.; Navaratnam, K.; Shi, G. Aromatase inhibitors for uterine fibroids. Cochrane Database Syst. Rev. 2013, 10, Cd009505. [Google Scholar] [CrossRef] [Green Version]
- Luoto, R.; Kaprio, J.; Rutanen, E.M.; Taipale, P.; Perola, M.; Koskenvuo, M. Heritability and risk factors of uterine fibroids--the Finnish Twin Cohort study. Maturitas 2000, 37, 15–26. [Google Scholar] [CrossRef]
- Ge, T.; Chen, C.Y.; Neale, B.M.; Sabuncu, M.R.; Smoller, J.W. Phenome-wide heritability analysis of the UK Biobank. PLoS Genet. 2017, 13, e1006711. [Google Scholar] [CrossRef]
- Bray, M.J.; Davis, L.K.; Torstenson, E.S.; Jones, S.H.; Edwards, T.L.; Edwards, D.R.V. Estimating Uterine Fibroid SNP-Based Heritability in European American Women with Imaging-Confirmed Fibroids. Hum. Hered. 2019, 84, 73–81. [Google Scholar] [CrossRef]
- Snieder, H.; MacGregor, A.J.; Spector, T.D. Genes control the cessation of a woman’s reproductive life: A twin study of hysterectomy and age at menopause. J. Clin. Endocrinol. Metab. 1998, 83, 1875–1880. [Google Scholar] [CrossRef]
- Vikhlyaeva, E.M.; Khodzhaeva, Z.S.; Fantschenko, N.D. Familial predisposition to uterine leiomyomas. Int. J. Gynecol. Obstet. 1995, 51, 127–131. [Google Scholar] [CrossRef]
- Sato, F.; Mori, M.; Nishi, M.; Kudo, R.; Miyake, H. Familial aggregation of uterine myomas in Japanese women. J. Epidemiol. 2002, 12, 249–253. [Google Scholar] [CrossRef] [Green Version]
- Van Voorhis, B.J.; Romitti, P.A.; Jones, M.P. Family history as a risk factor for development of uterine leiomyomas. Results of a pilot study. J. Reprod. Med. 2002, 47, 663–669. [Google Scholar] [PubMed]
- Okolo, S.O.; Gentry, C.C.; Perrett, C.W.; Maclean, A.B. Familial prevalence of uterine fibroids is associated with distinct clinical and molecular features. Hum. Reprod. 2005, 20, 2321–2324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ooi, A. Advances in hereditary leiomyomatosis and renal cell carcinoma (HLRCC) research. Semin. Cancer Biol. 2020, 61, 158–166. [Google Scholar] [CrossRef]
- Launonen, V.; Vierimaa, O.; Kiuru, M.; Isola, J.; Roth, S.; Pukkala, E.; Sistonen, P.; Herva, R.; Aaltonen, L.A. Inherited susceptibility to uterine leiomyomas and renal cell cancer. Proc. Natl. Acad. Sci. USA 2001, 98, 3387–3392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomlinson, I.P.; Alam, N.A.; Rowan, A.J.; Barclay, E.; Jaeger, E.E.; Kelsell, D.; Leigh, I.; Gorman, P.; Lamlum, H.; Rahman, S.; et al. Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat. Genet. 2002, 30, 406–410. [Google Scholar] [CrossRef]
- Linehan, W.M.; Rouault, T.A. Molecular pathways: Fumarate hydratase-deficient kidney cancer--targeting the Warburg effect in cancer. Clin. Cancer Res. 2013, 19, 3345–3352. [Google Scholar] [CrossRef] [Green Version]
- Cha, P.C.; Takahashi, A.; Hosono, N.; Low, S.K.; Kamatani, N.; Kubo, M.; Nakamura, Y. A genome-wide association study identifies three loci associated with susceptibility to uterine fibroids. Nat. Genet. 2011, 43, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.A.; Ruiz-Narvaez, E.A.; Palmer, J.R.; Cozier, Y.C.; Tandon, A.; Patterson, N.; Radin, R.G.; Rosenberg, L.; Reich, D. African ancestry and genetic risk for uterine leiomyomata. Am. J. Epidemiol. 2012, 176, 1159–1168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bondagji, N.S.; Morad, F.A.; Al-Nefaei, A.A.; Khan, I.A.; Elango, R.; Abdullah, L.S.; Al-Mansouri, N.M.; Sabir, J.; Banaganapalli, B.; Edris, S.; et al. Replication of GWAS loci revealed the moderate effect of TNRC6B locus on susceptibility of Saudi women to develop uterine leiomyomas. J. Obstet. Gynecol. Res. 2017, 43, 330–338. [Google Scholar] [CrossRef]
- Eggert, S.L.; Huyck, K.L.; Somasundaram, P.; Kavalla, R.; Stewart, E.A.; Lu, A.T.; Painter, J.N.; Montgomery, G.W.; Medland, S.E.; Nyholt, D.R.; et al. Genome-wide linkage and association analyses implicate FASN in predisposition to Uterine Leiomyomata. Am. J. Hum. Genet. 2012, 91, 621–628. [Google Scholar] [CrossRef] [Green Version]
- Aissani, B.; Zhang, K.; Wiener, H. Evaluation of GWAS candidate susceptibility loci for uterine leiomyoma in the multi-ethnic NIEHS uterine fibroid study. Front. Genet. 2015, 6, 241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aissani, B.; Zhang, K.; Wiener, H. Genetic determinants of uterine fibroid size in the multiethnic NIEHS uterine fibroid study. Int. J. Mol. Epidemiol. Genet. 2015, 6, 9–19. [Google Scholar]
- Gallagher, C.S.; Morton, C.C. Genetic Association Studies in Uterine Fibroids: Risk Alleles Presage the Path to Personalized Therapies. Semin. Reprod. Med. 2016, 34, 235–241. [Google Scholar] [CrossRef]
- Ponomarenko, I.; Reshetnikov, E.; Polonikov, A.; Verzilina, I.; Sorokina, I.; Yermachenko, A.; Dvornyk, V.; Churnosov, M. Candidate Genes for Age at Menarche Are Associated With Uterine Leiomyoma. Front. Genet. 2020, 11, 512940. [Google Scholar] [CrossRef]
- Makinen, N.; Mehine, M.; Tolvanen, J.; Kaasinen, E.; Li, Y.; Lehtonen, H.J.; Gentile, M.; Yan, J.; Enge, M.; Taipale, M.; et al. MED12, the mediator complex subunit 12 gene, is mutated at high frequency in uterine leiomyomas. Science 2011, 334, 252–255. [Google Scholar] [CrossRef]
- Lee, M.; Cheon, K.; Chae, B.; Hwang, H.; Kim, H.K.; Chung, Y.J.; Song, J.Y.; Cho, H.H.; Kim, J.H.; Kim, M.R. Analysis of MED12 Mutation in Multiple Uterine Leiomyomas in South Korean patients. Int. J. Med. Sci. 2018, 15, 124–128. [Google Scholar] [CrossRef] [Green Version]
- Park, M.J.; Shen, H.; Spaeth, J.M.; Tolvanen, J.H.; Failor, C.; Knudtson, J.F.; McLaughlin, J.; Halder, S.K.; Yang, Q.; Bulun, S.E.; et al. Oncogenic exon 2 mutations in Mediator subunit MED12 disrupt allosteric activation of cyclin C-CDK8/19. J. Biol. Chem. 2018, 293, 4870–4882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ajabnoor, G.M.A.; Mohammed, N.A.; Banaganapalli, B.; Abdullah, L.S.; Bondagji, O.N.; Mansouri, N.; Sahly, N.N.; Vaidyanathan, V.; Bondagji, N.; Elango, R.; et al. Expanded Somatic Mutation Spectrum of MED12 Gene in Uterine Leiomyomas of Saudi Arabian Women. Front. Genet. 2018, 9, 552. [Google Scholar] [CrossRef] [Green Version]
- Klatt, F.; Leitner, A.; Kim, I.V.; Ho-Xuan, H.; Schneider, E.V.; Langhammer, F.; Weinmann, R.; Müller, M.R.; Huber, R.; Meister, G.; et al. A precisely positioned MED12 activation helix stimulates CDK8 kinase activity. Proc. Natl. Acad. Sci. USA 2020, 117, 2894–2905. [Google Scholar] [CrossRef]
- Srivastava, S.; Kulshreshtha, R. Insights into the regulatory role and clinical relevance of mediator subunit, MED12, in human diseases. J. Cell. Physiol. 2021, 236, 3163–3177. [Google Scholar] [CrossRef]
- Mäkinen, N.; Vahteristo, P.; Bützow, R.; Sjöberg, J.; Aaltonen, L.A. Exomic landscape of MED12 mutation-negative and -positive uterine leiomyomas. Int. J. Cancer. 2014, 134, 1008–1012. [Google Scholar] [CrossRef] [PubMed]
- Kampjarvi, K.; Park, M.J.; Mehine, M.; Kim, N.H.; Clark, A.D.; Butzow, R.; Bohling, T.; Bohm, J.; Mecklin, J.P.; Jarvinen, H.; et al. Mutations in Exon 1 highlight the role of MED12 in uterine leiomyomas. Hum. Mutat. 2014, 35, 1136–1141. [Google Scholar] [CrossRef] [PubMed]
- Hodge, J.C.; Kim, T.M.; Dreyfuss, J.M.; Somasundaram, P.; Christacos, N.C.; Rousselle, M.; Quade, B.J.; Park, P.J.; Stewart, E.A.; Morton, C.C. Expression profiling of uterine leiomyomata cytogenetic subgroups reveals distinct signatures in matched myometrium: Transcriptional profilingof the t(12;14) and evidence in support of predisposing genetic heterogeneity. Hum. Mol. Genet. 2012, 21, 2312–2329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mittal, P.; Shin, Y.H.; Yatsenko, S.A.; Castro, C.A.; Surti, U.; Rajkovic, A. Med12 gain-of-function mutation causes leiomyomas and genomic instability. J. Clin. Investig. 2015, 125, 3280–3284. [Google Scholar] [CrossRef]
- Al-Hendy, A.; Laknaur, A.; Diamond, M.P.; Ismail, N.; Boyer, T.G.; Halder, S.K. Silencing Med12 Gene Reduces Proliferation of Human Leiomyoma Cells Mediated via Wnt/β-Catenin Signaling Pathway. Endocrinology 2017, 158, 592–603. [Google Scholar] [CrossRef]
- El Andaloussi, A.; Al-Hendy, A.; Ismail, N.; Boyer, T.G.; Halder, S.K. Introduction of Somatic Mutation in MED12 Induces Wnt4/β-Catenin and Disrupts Autophagy in Human Uterine Myometrial Cell. Reprod. Sci. 2020, 27, 823–832. [Google Scholar] [CrossRef]
- Asano, R.; Asai-Sato, M.; Matsukuma, S.; Mizushima, T.; Taguri, M.; Yoshihara, M.; Inada, M.; Fukui, A.; Suzuki, Y.; Miyagi, Y.; et al. Expression of erythropoietin messenger ribonucleic acid in wild-type MED12 uterine leiomyomas under estrogenic influence: New insights into related growth disparities. Fertil. Steril. 2019, 111, 178–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinonen, H.R.; Sarvilinna, N.S.; Sjöberg, J.; Kämpjärvi, K.; Pitkänen, E.; Vahteristo, P.; Mäkinen, N.; Aaltonen, L.A. MED12 mutation frequency in unselected sporadic uterine leiomyomas. Fertil. Steril. 2014, 102, 1137–1142. [Google Scholar] [CrossRef]
- Heinonen, H.R.; Pasanen, A.; Heikinheimo, O.; Tanskanen, T.; Palin, K.; Tolvanen, J.; Vahteristo, P.; Sjöberg, J.; Pitkänen, E.; Bützow, R.; et al. Multiple clinical characteristics separate MED12-mutation-positive and -negative uterine leiomyomas. Sci. Rep. 2017, 7, 1015. [Google Scholar] [CrossRef]
- Yatsenko, S.A.; Mittal, P.; Wood-Trageser, M.A.; Jones, M.W.; Surti, U.; Edwards, R.P.; Sood, A.K.; Rajkovic, A. Highly heterogeneous genomic landscape of uterine leiomyomas by whole exome sequencing and genome-wide arrays. Fertil. Steril. 2017, 107, 457–466. [Google Scholar] [CrossRef] [Green Version]
- Shaik, N.A.; Lone, W.G.; Khan, I.A.; Vaidya, S.; Rao, K.P.; Kodati, V.L.; Hasan, Q. Detection of somatic mutations and germline polymorphisms in mitochondrial DNA of uterine fibroids patients. Genet. Test. Mol. Biomark. 2011, 15, 537–541. [Google Scholar] [CrossRef]
- Dal Cin, P.; Moerman, P.; Deprest, J.; Brosens, I.; Van den Berghe, H. A new cytogenetic subgroup in uterine leiomyoma is characterized by a deletion of the long arm of chromosome 3. Genes Chromosomes Cancer 1995, 13, 219–220. [Google Scholar] [CrossRef]
- Vanharanta, S.; Wortham, N.C.; Laiho, P.; Sjoberg, J.; Aittomaki, K.; Arola, J.; Tomlinson, I.P.; Karhu, A.; Arango, D.; Aaltonen, L.A. 7q deletion mapping and expression profiling in uterine fibroids. Oncogene 2005, 24, 6545–6554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandberg, A.A. Updates on the cytogenetics and molecular genetics of bone and soft tissue tumors: Leiomyoma. Cancer Genet. Cytogenet. 2005, 158, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Surti, U. Subgroups of uterine leiomyomas based on cytogenetic analysis. Hum. Pathol. 1991, 22, 1009–1016. [Google Scholar] [CrossRef]
- Rein, M.S.; Friedman, A.J.; Barbieri, R.L.; Pavelka, K.; Fletcher, J.A.; Morton, C.C. Cytogenetic abnormalities in uterine leiomyomata. Obstet. Gynecol. 1991, 77, 923–926. [Google Scholar]
- El-Gharib, M.N.; Elsobky, E.S. Cytogenetic aberrations and the development of uterine leiomyomata. J. Obstet. Gynecol. Res. 2010, 36, 101–107. [Google Scholar] [CrossRef]
- Hodge, J.C.; Pearce, K.E.; Clayton, A.C.; Taran, F.A.; Stewart, E.A. Uterine cellular leiomyomata with chromosome 1p deletions represent a distinct entity. Am. J. Obstet. Gynecol. 2014, 210, 572.e1–572.e7. [Google Scholar] [CrossRef] [Green Version]
- Stephens, P.J.; Greenman, C.D.; Fu, B.; Yang, F.; Bignell, G.R.; Mudie, L.J.; Pleasance, E.D.; Lau, K.W.; Beare, D.; Stebbings, L.A.; et al. Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 2011, 144, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Koltsova, A.S.; Pendina, A.A.; Efimova, O.A.; Chiryaeva, O.G.; Kuznetzova, T.V.; Baranov, V.S. On the Complexity of Mechanisms and Consequences of Chromothripsis: An Update. Front. Genet. 2019, 10, 393. [Google Scholar] [CrossRef] [PubMed]
- Mehine, M.; Mäkinen, N.; Heinonen, H.R.; Aaltonen, L.A.; Vahteristo, P. Genomics of uterine leiomyomas: Insights from high-throughput sequencing. Fertil. Steril. 2014, 102, 621–629. [Google Scholar] [CrossRef]
- Mehine, M.; Kaasinen, E.; Makinen, N.; Katainen, R.; Kampjarvi, K.; Pitkanen, E.; Heinonen, H.R.; Butzow, R.; Kilpivaara, O.; Kuosmanen, A.; et al. Characterization of uterine leiomyomas by whole-genome sequencing. N. Engl. J. Med. 2013, 369, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Holzmann, C.; Saager, C.; Mechtersheimer, G.; Koczan, D.; Helmke, B.; Bullerdiek, J. Malignant transformation of uterine leiomyoma to myxoid leiomyosarcoma after morcellation associated with ALK rearrangement and loss of 14q. Oncotarget 2018, 9, 27595–27604. [Google Scholar] [CrossRef] [Green Version]
- Pendina, A.A.; Koltsova, A.S.; Efimova, O.A.; Malysheva, O.V.; Osinovskaya, N.S.; Sultanov, I.Y.; Tikhonov, A.V.; Shved, N.Y.; Chiryaeva, O.G.; Simareva, A.D.; et al. Case of chromothripsis in a large solitary non-recurrent uterine leiomyoma. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 219, 134–136. [Google Scholar] [CrossRef]
- Markowski, D.N.; Bullerdiek, J. Chromothripsis in uterine leiomyomas. N. Engl. J. Med. 2013, 369, 2160. [Google Scholar] [CrossRef]
- Gross, K.L.; Neskey, D.M.; Manchanda, N.; Weremowicz, S.; Kleinman, M.S.; Nowak, R.A.; Ligon, A.H.; Rogalla, P.; Drechsler, K.; Bullerdiek, J.; et al. HMGA2 expression in uterine leiomyomata and myometrium: Quantitative analysis and tissue culture studies. Genes Chromosomes Cancer 2003, 38, 68–79. [Google Scholar] [CrossRef]
- Klemke, M.; Meyer, A.; Nezhad, M.H.; Bartnitzke, S.; Drieschner, N.; Frantzen, C.; Schmidt, E.H.; Belge, G.; Bullerdiek, J. Overexpression of HMGA2 in uterine leiomyomas points to its general role for the pathogenesis of the disease. Genes Chromosomes Cancer 2009, 48, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qiang, W.; Griffin, B.B.; Gao, T.; Chakravarti, D.; Bulun, S.; Kim, J.J.; Wei, J.J. HMGA2-mediated tumorigenesis through angiogenesis in leiomyoma. Fertil. Steril. 2020, 114, 1085–1096. [Google Scholar] [CrossRef]
- Liu, B.; Chen, G.; He, Q.; Liu, M.; Gao, K.; Cai, B.; Qu, J.; Lin, S.; Geng, A.; Li, S.; et al. An HMGA2-p62-ERα axis regulates uterine leiomyomas proliferation. FASEB J. 2020, 34, 10966–10983. [Google Scholar] [CrossRef] [PubMed]
- Schoenmakers, E.F.; Huysmans, C.; Van de Ven, W.J. Allelic knockout of novel splice variants of human recombination repair gene RAD51B in t(12;14) uterine leiomyomas. Cancer Res. 1999, 59, 19–23. [Google Scholar]
- Mehine, M.; Kaasinen, E.; Heinonen, H.R.; Makinen, N.; Kampjarvi, K.; Sarvilinna, N.; Aavikko, M.; Vaharautio, A.; Pasanen, A.; Butzow, R.; et al. Integrated data analysis reveals uterine leiomyoma subtypes with distinct driver pathways and biomarkers. Proc. Natl. Acad. Sci. USA 2016, 113, 1315–1320. [Google Scholar] [CrossRef] [Green Version]
- Sornberger, K.S.; Weremowicz, S.; Williams, A.J.; Quade, B.J.; Ligon, A.H.; Pedeutour, F.; Vanni, R.; Morton, C.C. Expression of HMGIY in three uterine leiomyomata with complex rearrangements of chromosome 6. Cancer Genet. Cytogenet. 1999, 114, 9–16. [Google Scholar] [CrossRef]
- Nezhad, M.H.; Drieschner, N.; Helms, S.; Meyer, A.; Tadayyon, M.; Klemke, M.; Belge, G.; Bartnitzke, S.; Burchardt, K.; Frantzen, C.; et al. 6p21 rearrangements in uterine leiomyomas targeting HMGA1. Cancer Genet. Cytogenet. 2010, 203, 247–252. [Google Scholar] [CrossRef]
- Vanni, R.; Schoenmakers, E.F.; Andria, M. Deletion 7q in uterine leiomyoma: Fluorescence in situ hybridization characterization on primary cytogenetic preparations. Cancer Genet. Cytogenet. 1999, 113, 183–187. [Google Scholar] [CrossRef]
- Vanharanta, S.; Wortham, N.C.; Langford, C.; El-Bahrawy, M.; van der Spuy, Z.; Sjöberg, J.; Lehtonen, R.; Karhu, A.; Tomlinson, I.P.; Aaltonen, L.A. Definition of a minimal region of deletion of chromosome 7 in uterine leiomyomas by tiling-path microarray CGH and mutation analysis of known genes in this region. Genes Chromosomes Cancer 2007, 5, 451–458. [Google Scholar] [CrossRef]
- Saito, E.; Okamoto, A.; Saito, M.; Shinozaki, H.; Takakura, S.; Yanaihara, N.; Ochiai, K.; Tanaka, T. Genes associated with the genesis of leiomyoma of the uterus in a commonly deleted chromosomal region at 7q22. Oncol. Rep. 2005, 13, 469–472. [Google Scholar]
- Ptacek, T.; Song, C.; Walker, C.L.; Sell, S.M. Physical mapping of distinct 7q22 deletions in uterine leiomyoma and analysis of a recently annotated 7q22 candidate gene. Cancer Genet. Cytogenet. 2007, 174, 116–120. [Google Scholar] [CrossRef]
- Hodge, J.C.; Park, P.J.; Dreyfuss, J.M.; Assil-Kishawi, I.; Somasundaram, P.; Semere, L.G.; Quade, B.J.; Lynch, A.M.; Stewart, E.A.; Morton, C.C. Identifying the molecular signature of the interstitial deletion 7q subgroup of uterine leiomyomata using a paired analysis. Genes Chromosomes Cancer 2009, 48, 865–885. [Google Scholar] [CrossRef] [Green Version]
- Zeng, W.R.; Scherer, S.W.; Koutsilieris, M.; Huizenga, J.J.; Filteau, F.; Tsui, L.C.; Nepveu, A. Loss of heterozygosity and reduced expression of the CUTL1 gene in uterine leiomyomas. Oncogene 1997, 14, 2355–2365. [Google Scholar] [CrossRef]
- Moon, N.S.; Rong Zeng, W.; Premdas, P.; Santaguida, M.; Bérubé, G.; Nepveu, A. Expression of N-terminally truncated isoforms of CDP/CUX is increased in human uterine leiomyomas. Int. J. Cancer 2002, 100, 429–432. [Google Scholar] [CrossRef]
- Schoenmakers, E.F.; Bunt, J.; Hermers, L.; Schepens, M.; Merkx, G.; Janssen, B.; Kersten, M.; Huys, E.; Pauwels, P.; Debiec-Rychter, M.; et al. Identification of CUX1 as the recurrent chromosomal band 7q22 target gene in human uterine leiomyoma. Genes Chromosomes Cancer 2013, 52, 11–23. [Google Scholar] [CrossRef]
- Zhou, J.; Mochizuki, T.; Smeets, H.; Antignac, C.; Laurila, P.; de Paepe, A.; Tryggvason, K.; Reeders, S.T. Deletion of the paired alpha 5(IV) and alpha 6(IV) collagen genes in inherited smooth muscle tumors. Science 1993, 261, 1167–1169. [Google Scholar] [CrossRef]
- Heidet, L.; Dahan, K.; Zhou, J.; Xu, Z.; Cochat, P.; Gould, J.D.; Leppig, K.A.; Proesmans, W.; Guyot, C.; Guillot, M.; et al. Deletions of both alpha 5(IV) and alpha 6(IV) collagen genes in Alport syndrome and in Alport syndrome associated with smooth muscle tumours. Hum. Mol. Genet. 1995, 4, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Nozu, K.; Minamikawa, S.; Yamada, S.; Oka, M.; Yanagita, M.; Morisada, N.; Fujinaga, S.; Nagano, C.; Gotoh, Y.; Takahashi, E.; et al. Characterization of contiguous gene deletions in COL4A6 and COL4A5 in Alport syndrome-diffuse leiomyomatosis. J. Hum. Genet. 2017, 62, 733–735. [Google Scholar] [CrossRef]
- Yang, Q.; Mas, A.; Diamond, M.P.; Al-Hendy, A. The Mechanism and Function of Epigenetics in Uterine Leiomyoma Development. Reprod. Sci. 2016, 23, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Vergara, D.; Favilli, A.; La Rosa, V.L.; Tinelli, A.; Gerli, S.; Noventa, M.; Vitagliano, A.; Triolo, O.; Rapisarda, A.M.C.; et al. Epigenetic and genetic landscape of uterine leiomyomas: A current view over a common gynecological disease. Arch. Gynecol. Obstet. 2017, 296, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Välimäki, N.; Kuisma, H.; Pasanen, A.; Heikinheimo, O.; Sjöberg, J.; Bützow, R.; Sarvilinna, N.; Heinonen, H.R.; Tolvanen, J.; Bramante, S.; et al. Genetic predisposition to uterine leiomyoma is determined by loci for genitourinary development and genome stability. Elife 2018, 7, e37110. [Google Scholar] [CrossRef]
- Martinez-Jimenez, C.P.; Sandoval, J. Epigenetic crosstalk: A molecular language in human metabolic disorders. Front. Biosci. Sch. 2015, 7, 46–57. [Google Scholar]
- Miozzo, M.; Vaira, V.; Sirchia, S.M. Epigenetic alterations in cancer and personalized cancer treatment. Future Oncol. 2015, 11, 333–348. [Google Scholar] [CrossRef]
- Asada, H.; Yamagata, Y.; Taketani, T.; Matsuoka, A.; Tamura, H.; Hattori, N.; Ohgane, J.; Shiota, K.; Sugino, N. Potential link between estrogen receptor-alpha gene hypomethylation and uterine fibroid formation. Mol. Hum. Reprod. 2008, 14, 539–545. [Google Scholar] [CrossRef] [Green Version]
- Navarro, A.; Yin, P.; Monsivais, D.; Lin, S.M.; Du, P.; Wei, J.J.; Bulun, S.E. Genome-wide DNA methylation indicates silencing of tumor suppressor genes in uterine leiomyoma. PLoS ONE 2012, 7, e33284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maekawa, R.; Sato, S.; Yamagata, Y.; Asada, H.; Tamura, I.; Lee, L.; Okada, M.; Tamura, H.; Takaki, E.; Nakai, A.; et al. Genome-wide DNA methylation analysis reveals a potential mechanism for the pathogenesis and development of uterine leiomyomas. PLoS ONE 2013, 8, e66632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamagata, Y.; Maekawa, R.; Asada, H.; Taketani, T.; Tamura, I.; Tamura, H.; Ogane, J.; Hattori, N.; Shiota, K.; Sugino, N. Aberrant DNA methylation status in human uterine leiomyoma. Mol. Hum. Reprod. 2009, 15, 259–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Styer, A.K.; Rueda, B.R. The Epidemiology and Genetics of Uterine Leiomyoma. Best Pract. Res. Clin. Obstet. Gynecol. 2016, 34, 3–12. [Google Scholar] [CrossRef]
- George, J.W.; Fan, H.; Johnson, B.; Carpenter, T.J.; Foy, K.K.; Chatterjee, A.; Patterson, A.L.; Koeman, J.; Adams, M.; Madaj, Z.B.; et al. Integrated Epigenome, Exome, and Transcriptome Analyses Reveal Molecular Subtypes and Homeotic Transformation in Uterine Fibroids. Cell Rep. 2019, 29, 4069–4085.e4066. [Google Scholar] [CrossRef] [Green Version]
- Kol’tsova, A.S.; Pendina, A.A.; Efimova, O.A.; Kaminskaya, A.N.; Tikhonov, A.V.; Osinovskaya, N.S.; Sultanov, I.Y.; Shved, N.Y.; Kakhiani, M.I.; Baranov, V.S. Differential DNA Hydroxymethylation in Human Uterine Leiomyoma Cells Depending on the Phase of Menstrual Cycle and Presence of MED12 Gene Mutations. Bull. Exp. Biol. Med. 2017, 163, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yin, P.; Kujawa, S.A.; Coon, J.S.t.; Okeigwe, I.; Bulun, S.E. Progesterone receptor integrates the effects of mutated MED12 and altered DNA methylation to stimulate RANKL expression and stem cell proliferation in uterine leiomyoma. Oncogene 2019, 38, 2722–2735. [Google Scholar] [CrossRef] [Green Version]
- Vaiman, D. Towards an Epigenetic Treatment of Leiomyomas? Endocrinology 2020, 161, bqaa172. [Google Scholar] [CrossRef] [PubMed]
- Albert, M.; Helin, K. Histone methyltransferases in cancer. Semin. Cell Dev. Biol. 2010, 21, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.; Crabtree, G.R. Chromatin remodelling during development. Nature 2010, 463, 474–484. [Google Scholar] [CrossRef]
- Cao, R.; Wang, L.; Wang, H.; Xia, L.; Erdjument-Bromage, H.; Tempst, P.; Jones, R.S.; Zhang, Y. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 2002, 298, 1039–1043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viré, E.; Brenner, C.; Deplus, R.; Blanchon, L.; Fraga, M.; Didelot, C.; Morey, L.; Van Eynde, A.; Bernard, D.; Vanderwinden, J.M.; et al. The Polycomb group protein EZH2 directly controls DNA methylation. Nature 2006, 439, 871–874. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.H.; Torng, P.L.; Hsiao, S.M.; Jeng, Y.M.; Chen, M.W.; Chen, C.A. Histone deacetylase 6 regulates estrogen receptor alpha in uterine leiomyoma. Reprod. Sci. 2011, 18, 755–762. [Google Scholar] [CrossRef]
- Borahay, M.A.; Al-Hendy, A.; Kilic, G.S.; Boehning, D. Signaling Pathways in Leiomyoma: Understanding Pathobiology and Implications for Therapy. Mol. Med. 2015, 21, 242–256. [Google Scholar] [CrossRef]
- Wang, J.; Bhutani, M.; Pathak, A.K.; Lang, W.; Ren, H.; Jelinek, J.; He, R.; Shen, L.; Issa, J.P.; Mao, L. Delta DNMT3B variants regulate DNA methylation in a promoter-specific manner. Cancer Res. 2007, 67, 10647–10652. [Google Scholar] [CrossRef] [Green Version]
- Karmon, A.E.; Cardozo, E.R.; Rueda, B.R.; Styer, A.K. MicroRNAs in the development and pathobiology of uterine leiomyomata: Does evidence support future strategies for clinical intervention? Hum. Reprod. Update 2014, 20, 670–687. [Google Scholar] [CrossRef]
- Segars, J.; Al-Hendy, A. Uterine Leiomyoma: New Perspectives on an Old Disease. Semin. Reprod. Med. 2017, 35, 471–472. [Google Scholar] [CrossRef]
- Ono, M.; Yin, P.; Navarro, A.; Moravek, M.B.; Coon, V.J.; Druschitz, S.A.; Gottardi, C.J.; Bulun, S.E. Inhibition of canonical WNT signaling attenuates human leiomyoma cell growth. Fertil. Steril. 2014, 101, 1441–1449. [Google Scholar] [CrossRef]
- Chuang, T.D.; Rehan, A.; Khorram, O. Functional role of the long noncoding RNA X-inactive specific transcript in leiomyoma pathogenesis. Fertil. Steril. 2021, 115, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Ciarmela, P.; Petraglia, F. New epigenetic mechanism involved in leiomyoma formation. Fertil. Steril. 2021, 115, 94–95. [Google Scholar] [CrossRef]
- Linder, D.; Gartler, S.M. Glucose-6-phosphate dehydrogenase mosaicism: Utilization as a cell marker in the study of leiomyomas. Science 1965, 150, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Maruyama, T.; Masuda, H.; Kajitani, T.; Nagashima, T.; Arase, T.; Ito, M.; Ohta, K.; Uchida, H.; Asada, H.; et al. Side population in human uterine myometrium displays phenotypic and functional characteristics of myometrial stem cells. Proc. Natl. Acad. Sci. USA 2007, 104, 18700–18705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mas, A.; Cervello, I.; Gil-Sanchis, C.; Faus, A.; Ferro, J.; Pellicer, A.; Simon, C. Identification and characterization of the human leiomyoma side population as putative tumor-initiating cells. Fertil. Steril. 2012, 98, 741–751.e746. [Google Scholar] [CrossRef] [PubMed]
- Moravek, M.B.; Yin, P.; Ono, M.; Coon, V.J.S.; Dyson, M.T.; Navarro, A.; Marsh, E.E.; Chakravarti, D.; Kim, J.J.; Wei, J.-J.; et al. Ovarian steroids, stem cells and uterine leiomyoma: Therapeutic implications. Hum. Reprod. Update 2015, 21, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Mas, A.; Stone, L.; O’Connor, P.M.; Yang, Q.; Kleven, D.; Simon, C.; Walker, C.L.; Al-Hendy, A. Developmental Exposure to Endocrine Disruptors Expands Murine Myometrial Stem Cell Compartment as a Prerequisite to Leiomyoma Tumorigenesis. Stem. Cells 2017, 35, 666–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ono, M.; Qiang, W.; Serna, V.A.; Yin, P.; Coon, J.S.t.; Navarro, A.; Monsivais, D.; Kakinuma, T.; Dyson, M.; Druschitz, S.; et al. Role of stem cells in human uterine leiomyoma growth. PLoS ONE 2012, 7, e36935. [Google Scholar] [CrossRef] [PubMed]
- Mas, A.; Cervello, I.; Gil-Sanchis, C.; Simon, C. Current understanding of somatic stem cells in leiomyoma formation. Fertil. Steril. 2014, 102, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Mas, A.; Cervelló, I.; Fernández-Álvarez, A.; Faus, A.; Díaz, A.; Burgués, O.; Casado, M.; Simón, C. Overexpression of the truncated form of High Mobility Group A proteins (HMGA2) in human myometrial cells induces leiomyoma-like tissue formation. Mol. Hum. Reprod. 2015, 21, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Bertsch, E.; Qiang, W.; Zhang, Q.; Espona-Fiedler, M.; Druschitz, S.; Liu, Y.; Mittal, K.; Kong, B.; Kurita, T.; Wei, J.J. MED12 and HMGA2 mutations: Two independent genetic events in uterine leiomyoma and leiomyosarcoma. Mod. Pathol. 2014, 27, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Yi, T.; Shen, K.; Zhang, B.; Huang, F.; Zhao, X. Hypoxia: The driving force of uterine myometrial stem cells differentiation into leiomyoma cells. Med. Hypotheses 2011, 77, 985–986. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, T.; Ono, M.; Yoshimura, Y. Somatic stem cells in the myometrium and in myomas. Semin. Reprod. Med. 2013, 31, 77–81. [Google Scholar] [CrossRef]
- Chen, D.C.; Liu, J.Y.; Wu, G.J.; Ku, C.H.; Su, H.Y.; Chen, C.H. Serum vascular endothelial growth factor165 levels and uterine fibroid volume. Acta Obstet. Gynecol. Scand. 2005, 84, 317–321. [Google Scholar] [CrossRef]
- Dingiloglu, B.S.; Gungor, T.; Ozdal, B.; Cavkaytar, S.; Bilge, U.; Mollamahmutoglu, L. Serum leptin levels in women with uterine leiomyomas. Taiwan, J. Obstet. Gynecol. 2007, 46, 33–37. [Google Scholar] [CrossRef] [Green Version]
- Levy, G.; Hill, M.J.; Plowden, T.C.; Catherino, W.H.; Armstrong, A.Y. Biomarkers in uterine leiomyoma. Fertil. Steril. 2013, 99, 1146–1152. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.P.; Chen, Y.W.; Liu, W.H.; Chou, H.C.; Chang, Y.P.; Lin, S.T.; Li, J.M.; Jian, S.F.; Lee, Y.R.; Chan, H.L. Proteomic identification of plasma biomarkers in uterine leiomyoma. Mol. Biosyst. 2012, 8, 1136–1145. [Google Scholar] [CrossRef]
- Santulli, P.; Borghese, B.; Lemaréchal, H.; Leconte, M.; Millischer, A.E.; Batteux, F.; Chapron, C.; Borderie, D. Increased serum oxidative stress markers in women with uterine leiomyoma. PLoS ONE 2013, 8, e72069. [Google Scholar] [CrossRef]
- Caglayan, A.; Katlan, D.C.; Tuncer, Z.S.; Yuce, K.; Sayal, H.B.; Kocer-Gumusel, B. Assessment of oxidant-antioxidant status alterations with tumor biomarkers and reproductive system hormones in uterine MYOMAS. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 229, 1–7. [Google Scholar] [CrossRef]
- Ciebiera, M.; Włodarczyk, M.; Słabuszewska-Jóźwiak, A.; Nowicka, G.; Jakiel, G. Influence of vitamin D and transforming growth factor β3 serum concentrations, obesity, and family history on the risk for uterine fibroids. Fertil. Steril. 2016, 106, 1787–1792. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, W.; Wang, J.; Wei, H. Serum YKL-40 Level Positively Correlates With Uterine Leiomyomas. Reprod. Sci. 2016, 23, 1559–1564. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, D.; Sheng, J.; Luo, L.; Zhang, W. Identification of TRADD as a potential biomarker in human uterine leiomyoma through iTRAQ based proteomic profiling. Mol. Cell. Probes. 2017, 36, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Kamalipooya, S.; Zarezadeh, R.; Latifi, Z.; Nouri, M.; Fattahi, A.; Salemi, Z. Serum transforming growth factor β and leucine-rich α-2-glycoprotein 1 as potential biomarkers for diagnosis of uterine leiomyomas. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 102037. [Google Scholar] [CrossRef]
- Trovik, J.; Salvesen, H.B.; Cuppens, T.; Amant, F.; Staff, A.C. Growth differentiation factor-15 as biomarker in uterine sarcomas. Int. J. Gynecol. Cancer 2014, 24, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Juang, C.M.; Yen, M.S.; Horng, H.C.; Twu, N.F.; Yu, H.C.; Hsu, W.L. Potential role of preoperative serum CA125 for the differential diagnosis between uterine leiomyoma and uterine leiomyosarcoma. Eur. J. Gynaecol. Oncol. 2006, 27, 370–374. [Google Scholar]
- Basta, P.; Mach, P.; Pitynski, K.; Bednarek, W.; Klimek, M.; Zietek, J.; Zajac, K.; Wicherek, L. Differences in the blood serum levels of soluble HLA-G concentrations between the menstrual cycle phases and menopause in patients with ovarian endometriosis and uterine leiomyoma. Neuro-Endocrinol. Lett. 2009, 30, 91–98. [Google Scholar]
- Makinen, N.; Kampjarvi, K.; Frizzell, N.; Butzow, R.; Vahteristo, P. Characterization of MED12, HMGA2, and FH alterations reveals molecular variability in uterine smooth muscle tumors. Mol. Cancer 2017, 16, 101. [Google Scholar] [CrossRef] [Green Version]
- Kämpjärvi, K.; Mäkinen, N.; Mehine, M.; Välipakka, S.; Uimari, O.; Pitkänen, E.; Heinonen, H.R.; Heikkinen, T.; Tolvanen, J.; Ahtikoski, A.; et al. MED12 mutations and FH inactivation are mutually exclusive in uterine leiomyomas. Br. J. Cancer 2016, 114, 1405–1411. [Google Scholar] [CrossRef] [Green Version]
- Markowski, D.N.; Bartnitzke, S.; Löning, T.; Drieschner, N.; Helmke, B.M.; Bullerdiek, J. MED12 mutations in uterine fibroids--their relationship to cytogenetic subgroups. Int. J. Cancer 2012, 131, 1528–1536. [Google Scholar] [CrossRef]
- Holzmann, C.; Markowski, D.N.; Bartnitzke, S.; Koczan, D.; Helmke, B.M.; Bullerdiek, J. A rare coincidence of different types of driver mutations among uterine leiomyomas (UL). Mol. Cytogenet. 2015, 8, 76. [Google Scholar] [CrossRef] [Green Version]
- Galindo, L.J.; Hernández-Beeftink, T.; Salas, A.; Jung, Y.; Reyes, R.; de Oca, F.M.; Hernández, M.; Almeida, T.A. HMGA2 and MED12 alterations frequently co-occur in uterine leiomyomas. Gynecol. Oncol. 2018, 150, 562–568. [Google Scholar] [CrossRef]
- Adams, C.L.; Dimitrova, I.; Post, M.D.; Gibson, L.; Spillman, M.A.; Behbakht, K.; Bradford, A.P. Identification of a novel diagnostic gene expression signature to discriminate uterine leiomyoma from leiomyosarcoma. Exp. Mol. Pathol. 2019, 110, 104284. [Google Scholar] [CrossRef]
- Mas, A.; Simon, C. Molecular differential diagnosis of uterine leiomyomas and leiomyosarcomas. Biol. Reprod. 2019, 101, 1115–1123. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X. Targeting the Wnt/β-catenin signaling pathway in cancer. J. Hematol. Oncol. 2020, 13, 165. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yin, P.; Dotts, A.J.; Kujawa, S.A.; Coon, V.J.; Wei, J.J.; Chakravarti, D.; Bulun, S.E. Activation of protein kinase B by WNT4 as a regulator of uterine leiomyoma stem cell function. Fertil. Steril. 2020, 114, 1339–1349. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Tsuda, Y.; Yabuki, K.; Shiba, E.; Uchihashi, K.; Matsuyama, A.; Fujino, Y.; Hachisuga, T.; Hisaoka, M. Inhibition of WNT/β-catenin signaling under serum starvation and hypoxia induces adipocytic transdifferentiation in human leiomyoma cells. Lab. Investig. 2018, 98, 439–448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Hendy, A.; Diamond, M.P.; Boyer, T.G.; Halder, S.K. Vitamin D3 Inhibits Wnt/β-Catenin and mTOR Signaling Pathways in Human Uterine Fibroid Cells. J. Clin. Endocrinol. Metab. 2016, 101, 1542–1551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corachán, A.; Ferrero, H.; Aguilar, A.; Garcia, N.; Monleon, J.; Faus, A.; Cervelló, I.; Pellicer, A. Inhibition of tumor cell proliferation in human uterine leiomyomas by vitamin D via Wnt/β-catenin pathway. Fertil. Steril. 2019, 111, 397–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martini, M.; De Santis, M.C.; Braccini, L.; Gulluni, F.; Hirsch, E. PI3K/AKT signaling pathway and cancer: An updated review. Ann. Med. 2014, 46, 372–383. [Google Scholar] [CrossRef]
- Kovács, K.A.; Lengyel, F.; Vértes, Z.; Környei, J.L.; Gocze, P.M.; Sumegi, B.; Szabó, I.; Vértes, M. Phosphorylation of PTEN (phosphatase and tensin homologue deleted on chromosome ten) protein is enhanced in human fibromyomatous uteri. J. Steroid. Biochem. Mol. Biol. 2007, 103, 196–199. [Google Scholar] [CrossRef]
- Karra, L.; Shushan, A.; Ben-Meir, A.; Rojansky, N.; Klein, B.Y.; Shveiky, D.; Levitzki, R.; Ben-Bassat, H. Changes related to phosphatidylinositol 3-kinase/Akt signaling in leiomyomas: Possible involvement of glycogen synthase kinase 3alpha and cyclin D2 in the pathophysiology. Fertil. Steril. 2010, 93, 2646–2651. [Google Scholar] [CrossRef] [PubMed]
- Ben-Sasson, H.; Ben-Meir, A.; Shushan, A.; Karra, L.; Rojansky, N.; Klein, B.Y.; Levitzki, R.; Ben-Bassat, H. All-trans-retinoic acid mediates changes in PI3K and retinoic acid signaling proteins of leiomyomas. Fertil. Steril. 2011, 95, 2080–2086. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Choi, Y.S.; Park, J.H.; Kim, H.; Lee, I.; Won, Y.B.; Yun, B.H.; Seo, S.K.; Lee, B.S.; Cho, S. MiR-150-5p May Contribute to Pathogenesis of Human Leiomyoma via Regulation of the Akt/p27(Kip1) Pathway In Vitro. Int. J. Mol. Sci. 2019, 20, 2684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bifulco, G.; Miele, C.; Pellicano, M.; Trencia, A.; Ferraioli, M.; Paturzo, F.; Tommaselli, G.A.; Beguinot, F.; Nappi, C. Molecular mechanisms involved in GnRH analogue-related apoptosis for uterine leiomyomas. Mol. Hum. Reprod. 2004, 10, 43–48. [Google Scholar] [CrossRef] [Green Version]
- Ciebiera, M.; Włodarczyk, M.; Wrzosek, M.; Męczekalski, B.; Nowicka, G.; Łukaszuk, K.; Słabuszewska-Jóźwiak, A.; Jakiel, G. Role of Transforming Growth Factor β in Uterine Fibroid Biology. Int. J. Mol. Sci. 2017, 18, 2435. [Google Scholar] [CrossRef] [Green Version]
- Lee, B.S.; Nowak, R.A. Human leiomyoma smooth muscle cells show increased expression of transforming growth factor-beta 3 (TGF beta 3) and altered responses to the antiproliferative effects of TGF beta. J. Clin. Endocrinol. Metab. 2001, 86, 913–920. [Google Scholar] [CrossRef] [Green Version]
- Luo, X.; Ding, L.; Xu, J.; Chegini, N. Gene expression profiling of leiomyoma and myometrial smooth muscle cells in response to transforming growth factor-beta. Endocrinology 2005, 146, 1097–1118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolańska, M.; Bańkowski, E. Transforming growth factor beta and platelet-derived growth factor in human myometrium and in uterine leiomyomas at various stages of tumour growth. Eur. J. Obstet. Gynecol. Reprod. Biol. 2007, 130, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wu, Y.; Lu, Q.; Zhang, P.; Ren, M. Transforming growth factor-β signaling pathway cross-talking with ERα signaling pathway on regulating the growth of uterine leiomyoma activated by phenolic environmental estrogens in vitro. Tumour. Biol. 2016, 37, 455–462. [Google Scholar] [CrossRef]
- Chegini, N.; Luo, X.; Ding, L.; Ripley, D. The expression of Smads and transforming growth factor beta receptors in leiomyoma and myometrium and the effect of gonadotropin releasing hormone analogue therapy. Mol. Cell. Endocrinol. 2003, 209, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Włodarczyk, M.; Wrzosek, M.; Słabuszewska-Jóźwiak, A.; Nowicka, G.; Jakiel, G. Ulipristal acetate decreases transforming growth factor β3 serum and tumor tissue concentrations in patients with uterine fibroids. Fertil. Steril. 2018, 109, 501–507.e502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, T.D.; Malik, M.; Britten, J.; Parikh, T.; Cox, J.; Catherino, W.H. Ulipristal acetate decreases active TGF-β3 and its canonical signaling in uterine leiomyoma via two novel mechanisms. Fertil. Steril. 2019, 111, 806–815.e801. [Google Scholar] [CrossRef] [PubMed]
- Chudasama, P.; Mughal, S.S.; Sanders, M.A.; Hubschmann, D.; Chung, I.; Deeg, K.I.; Wong, S.H.; Rabe, S.; Hlevnjak, M.; Zapatka, M.; et al. Integrative genomic and transcriptomic analysis of leiomyosarcoma. Nat. Commun. 2018, 9, 144. [Google Scholar] [CrossRef] [Green Version]
- Stratton, K.L.; Chang, S.S. Locally advanced prostate cancer: The role of surgical management. BJU Int. 2009, 104, 449–454. [Google Scholar] [CrossRef]
- Shibue, T.; Weinberg, R.A. EMT, CSCs, and drug resistance: The mechanistic link and clinical implications. Nat. Rev. Clin. Oncol. 2017, 14, 611–629. [Google Scholar] [CrossRef] [Green Version]
- Picelli, S.; Björklund, Å.K.; Faridani, O.R.; Sagasser, S.; Winberg, G.; Sandberg, R. Smart-seq2 for sensitive full-length transcriptome profiling in single cells. Nat. Methods 2013, 10, 1096–1098. [Google Scholar] [CrossRef]
- Navin, N.E. The first five years of single-cell cancer genomics and beyond. Genome Res. 2015, 25, 1499–1507. [Google Scholar] [CrossRef] [Green Version]
- Tanay, A.; Regev, A. Scaling single-cell genomics from phenomenology to mechanism. Nature 2017, 541, 331–338. [Google Scholar] [CrossRef] [Green Version]
- Svensson, V.; Vento-Tormo, R.; Teichmann, S.A. Exponential scaling of single-cell RNA-seq in the past decade. Nat. Protoc. 2018, 13, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Moffitt, J.R.; Bambah-Mukku, D.; Eichhorn, S.W.; Vaughn, E.; Shekhar, K.; Perez, J.D.; Rubinstein, N.D.; Hao, J.; Regev, A.; Dulac, C.; et al. Molecular, spatial, and functional single-cell profiling of the hypothalamic preoptic region. Science 2018, 362, eaau5324. [Google Scholar] [CrossRef] [Green Version]
- Angelidis, I.; Simon, L.M.; Fernandez, I.E.; Strunz, M.; Mayr, C.H.; Greiffo, F.R.; Tsitsiridis, G.; Ansari, M.; Graf, E.; Strom, T.M.; et al. An atlas of the aging lung mapped by single cell transcriptomics and deep tissue proteomics. Nat. Commun. 2019, 10, 963. [Google Scholar] [CrossRef] [Green Version]
- Plasschaert, L.W.; Žilionis, R.; Choo-Wing, R.; Savova, V.; Knehr, J.; Roma, G.; Klein, A.M.; Jaffe, A.B. A single-cell atlas of the airway epithelium reveals the CFTR-rich pulmonary ionocyte. Nature 2018, 560, 377–381. [Google Scholar] [CrossRef]
- Ali, H.; Kilic, G.; Vincent, K.; Motamedi, M.; Rytting, E. Nanomedicine for uterine leiomyoma therapy. Ther. Deliv. 2013, 4, 161–175. [Google Scholar] [CrossRef] [Green Version]
- Jiang, J.; Bischof, J. Effect of timing, dose and interstitial versus nanoparticle delivery of tumor necrosis factor alpha in combinatorial adjuvant cryosurgery treatment of ELT-3 uterine fibroid tumor. Cryo. Lett. 2010, 31, 50–62. [Google Scholar]
- Shalaby, S.M.; Khater, M.K.; Perucho, A.M.; Mohamed, S.A.; Helwa, I.; Laknaur, A.; Lebedyeva, I.; Liu, Y.; Diamond, M.P.; Al-Hendy, A.A. Magnetic nanoparticles as a new approach to improve the efficacy of gene therapy against differentiated human uterine fibroid cells and tumor-initiating stem cells. Fertil. Steril. 2016, 105, 1638–1648.e1638. [Google Scholar] [CrossRef] [Green Version]
- Moyo, M.B.; Parker, J.B.; Chakravarti, D. Altered chromatin landscape and enhancer engagement underlie transcriptional dysregulation in MED12 mutant uterine leiomyomas. Nat. Commun. 2020, 11, 1019. [Google Scholar] [CrossRef] [PubMed]
- Grossman, S.R.; Zhang, X.; Wang, L.; Engreitz, J.; Melnikov, A.; Rogov, P.; Tewhey, R.; Isakova, A.; Deplancke, B.; Bernstein, B.E.; et al. Systematic dissection of genomic features determining transcription factor binding and enhancer function. Proc. Natl. Acad. Sci. USA 2017, 114, e1291–e1300. [Google Scholar] [CrossRef] [Green Version]



| Molecular Tools in the Clinical Management of uLM | Detection: Putative biomarkers | ↑ serum levels in uLM | Biomarkers for differential diagnosis | ||||
| • Leptin | • CA125 | • AOPP | • GDF-15 | ||||
| • VEGF | • Vitamin D3 | • LRG1 | • CA125 | ||||
| • Ghrelin | • TGF-β | • YKL40 | • HLA-G | ||||
| Classification: Gene expression profiling | Molecular subclassification | Differential diagnosis | |||||
| Mutational status of driver genes | uLM versus uterine leiomyosarcoma | ||||||
| Similar cellular processes involved | Clearly distinct transcriptomic profiles | ||||||
| • MED12 | High number of genes involved | ||||||
| • HMGA2 | • BRCA2 | • FOXM1 | • PAX3 | ||||
| Treatment: Targetable pathways | WNT-β catenin pathway | PI3K-AKT pathway | TGFβ pathway | ||||
| ↑ proliferation | ↑ growth | Overexpression of cytokines | |||||
| ↑ stem cell function | Involved in development | ↑ growth | |||||
| Inhibited by vitamin D3 | Inhibited by GnRH analogues and UPA | Inhibited by GnRH analogues and UPA | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Machado-Lopez, A.; Simón, C.; Mas, A. Molecular and Cellular Insights into the Development of Uterine Fibroids. Int. J. Mol. Sci. 2021, 22, 8483. https://doi.org/10.3390/ijms22168483
Machado-Lopez A, Simón C, Mas A. Molecular and Cellular Insights into the Development of Uterine Fibroids. International Journal of Molecular Sciences. 2021; 22(16):8483. https://doi.org/10.3390/ijms22168483
Chicago/Turabian StyleMachado-Lopez, Alba, Carlos Simón, and Aymara Mas. 2021. "Molecular and Cellular Insights into the Development of Uterine Fibroids" International Journal of Molecular Sciences 22, no. 16: 8483. https://doi.org/10.3390/ijms22168483







