Beneficial Effects of Soybean-Derived Bioactive Peptides
Abstract
1. Introduction
2. Production of Soy Bioactive Peptide
3. Characteristics of Soybean Peptides
4. Functionality of Soy Peptides
4.1. Neuroprotective Effects and Improvement of Cognitive Impairment
4.2. Modulation of the Cardiovascular System and Blood Pressure
4.3. Inhibition of Chronic Kidney Disease Progression
4.4. Immunoregulatory Effects
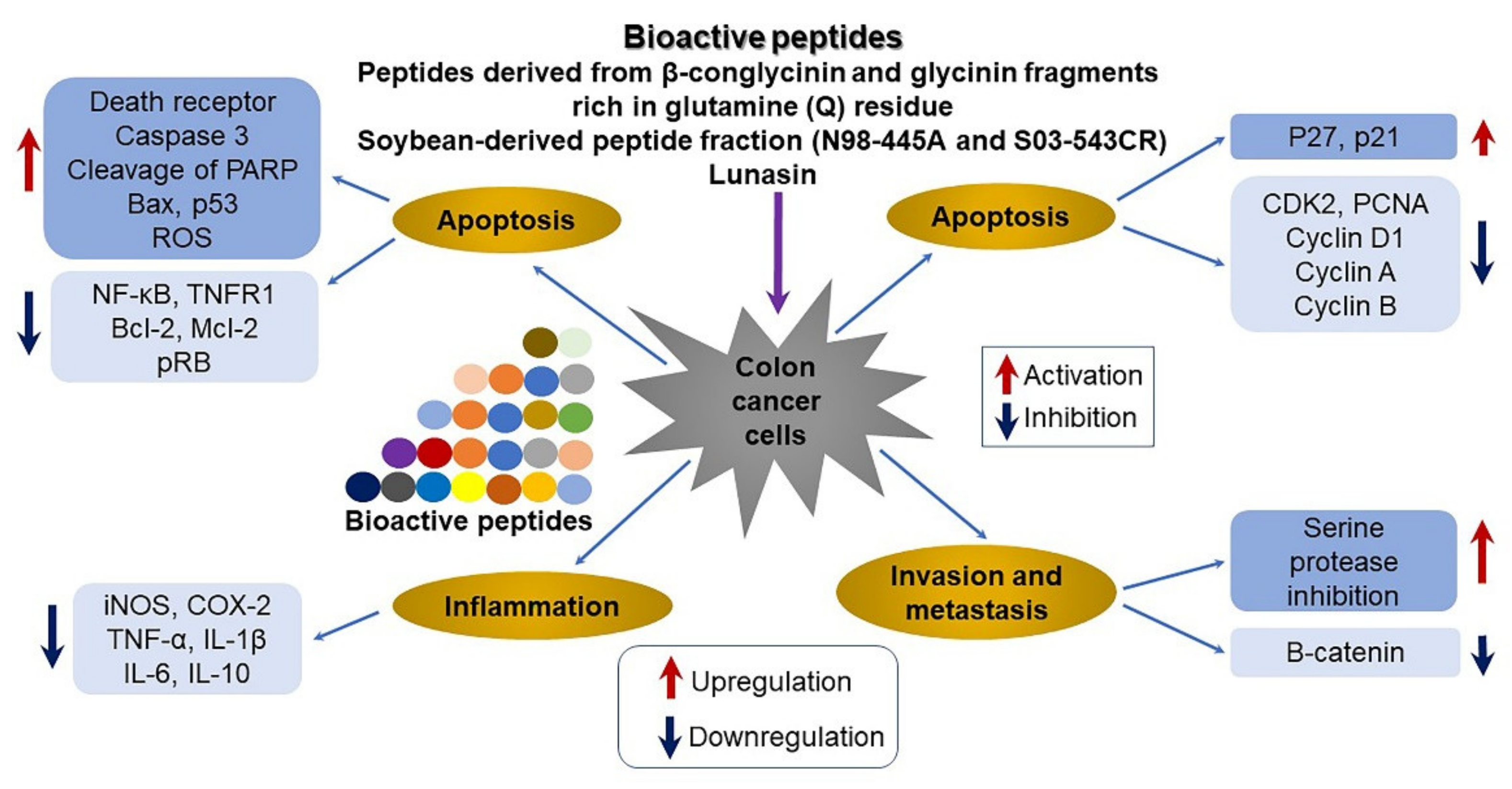
4.5. Inhibition of Cancer Cell Proliferation by SPs
4.6. Anti-Inflammation Properties
4.7. Modulation of Lipid Metabolism
4.8. Antiobesity Effects
4.9. Antiarteriosclerosis Effects
4.10. Antidiabetic Effects
4.11. Skin Protection Effect of Soybean Oligopeptides against Ultraviolet Radiation
4.12. Antioxidative Properties
4.13. Soybean Proteins and Control of Increased Gut Microbial Activity Caused by Probiotics
4.14. Antimicrobial and Antiviral Effects
5. Economic Feasibility and Application of Soybean-Derived Bioactive Peptides
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Giri, S.K.; Mangaraj, S. Processing influences on composition and quality attributes of soymilk and its powder. Food Eng. Rev. 2012, 4, 149–164. [Google Scholar] [CrossRef]
- Chandrasekara, A.; Josheph Kumar, T. Roots and tuber crops as functional foods: A review on phytochemical constituents and their potential health benefits. Int. J. Food Sci. 2016, 2016, 3631647. [Google Scholar] [CrossRef] [PubMed]
- Capuano, E. The behavior of dietary fiber in the gastrointestinal tract determines its physiological effect. Crit. Rev. Food Sci. Nutr. 2017, 57, 3543–3564. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.S.; Kim, C.H.; Yang, W.S. Physiologically active molecules and functional properties of soybeans in human health-A current perspective. Int. J. Mol. Sci. 2021, 22, 4054. [Google Scholar] [CrossRef]
- Kim, I.S.; Hwang, C.W.; Yang, W.S.; Kim, C.H. Current perspectives on the physiological activities of fermented soybean-derived cheonggukjang. Int. J. Mol. Sci. 2021, 22, 5746. [Google Scholar] [CrossRef]
- Hao, Y.; Fan, X.; Guo, H.; Yao, Y.; Ren, G.; Lv, X.; Yang, X. Overexpression of the bioactive lunasin peptide in soybean and evaluation of its anti-inflammatory and anti-cancer activities in vitro. J. Biosci. Bioeng. 2020, 129, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.W. Health effects of soy protein and isoflavones in humans. J. Nutr. 2008, 138, 1244S–1249S. [Google Scholar] [CrossRef] [PubMed]
- Ramdath, D.D.; Padhi, E.M.; Sarfaraz, S.; Renwick, S.; Duncan, A.M. Beyond the cholesterol-lowering effect of soy protein: A review of the effects of dietary soy and its constituents on risk factors for cardiovascular disease. Nutrients 2017, 9, 324. [Google Scholar] [CrossRef]
- Zheng, X.; Lee, S.K.; Chun, O.K. Soy isoflavones and osteoporotic bone loss: A review with an emphasis on modulation of bone remodeling. J. Med. Food 2016, 19, 1–14. [Google Scholar] [CrossRef]
- Chatterjee, C.; Gleddie, S.; Xiao, C.W. Soybean bioactive peptides and their functional properties. Nutrients 2018, 10, 1211. [Google Scholar] [CrossRef] [PubMed]
- Agyei, D. Bioactive proteins and peptides from soybeans. Recent Pat. Food Nutr. Agric. 2015, 7, 100–107. [Google Scholar] [CrossRef]
- Jung, S.J.; Park, S.H.; Choi, E.K.; Cha, Y.S.; Cho, B.H.; Kim, Y.G.; Kim, M.G.; Song, W.O.; Park, T.S.; Ko, J.K.; et al. Beneficial effects of Korean traditional diets in hypertensive and type 2 diabetic patients. J. Med. Food 2014, 17, 161–171. [Google Scholar] [CrossRef]
- Van Vliet, S.; Kronberg, S.L.; Provenza, F.D. Plant-based meats, human health, and climate change. Front. Sustain. Food Syst. 2020, 4, 128. [Google Scholar] [CrossRef]
- Messina, M.; Messina, V. The role of soy vegetarian diet. Nutrients 2010, 2, 855–888. [Google Scholar] [CrossRef]
- Montgomery, K.S. Soy protein. J. Perinat. Educ. 2003, 12, 42–45. [Google Scholar] [CrossRef]
- Messina, M. Soy and health update: Evaluation of the clinical and epidemiologic literature. Nutrients 2016, 8, 754. [Google Scholar] [CrossRef]
- He, F.J.; Chen, J.Q. Consumption of soybean, soy foods, soy isoflavones and breast cancer incidence: Differences between Chinese women and women in Western countries and possible mechanisms. Food Sci. Hum. Wellness 2013, 2, 146–161. [Google Scholar] [CrossRef]
- Boccellino, M.; D’Angelo, S. Anti-obesity effects of polyphenol intake: Current status and future possibilities. Int. J. Mol. Sci. 2020, 21, 5642. [Google Scholar] [CrossRef]
- Singh, M.; Thrimawithana, T.; Shukla, R.; Adhikari, B. Managing obesity through natural polyphenols: A review. Future Food. 2020, 1–2, 100002. [Google Scholar] [CrossRef]
- Lule, V.K.; Garg, S.; Pophaly, S.D.; Hitesh; Tomar, S.K. Potential health benefits of lunasin: A multifaceted soy-derived bioactive peptide. J. Food Sci. 2015, 80, R485–R494. [Google Scholar] [CrossRef]
- Peighambardoust, S.H.; Karami, Z.; Pateiro, M.; Lorenzo, J.M. A review on health-promoting, biological, and functional aspects of bioactive peptides in food applications. Biomolecules 2021, 11, 631. [Google Scholar] [CrossRef]
- Karami, Z.; Akbari-Adergani, B. Bioactive food derived peptides: A review on correlation between structure of bioactive peptides and their functional properties. J. Food Sci. Technol. 2019, 56, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Gorguc, A.; Gencdag, E.; Yilmaz, F.M. Bioactive peptides derived from plant origin by-products: Biological activities and techno-functional utilizations in food developments—A review. Food Res. Int. 2020, 136, 109504. [Google Scholar] [CrossRef] [PubMed]
- Nasri, M. Protein hydrolysates and biopeptides: Production, biological activities, and applications in foods and health benefits. A review. Adv. Food Nutr. Res. 2017, 81, 109–159. [Google Scholar] [PubMed]
- Wang, W.; De Mejia, E.G. A new frontier in soy bioactive peptides that may prevent age-related chronic diseases. Compr. Rev. Food Sci. Food Saf. 2005, 4, 63–78. [Google Scholar] [CrossRef]
- Barati, M.; Javanmardi, F.; Mousavi Jazayeri, S.M.H.; Jabbari, M.; Rahmani, J.; Barati, F.; Nickho, H.; Davoodi, S.H.; Roshanravan, N.; Mousavi Khaneghah, A. Techniques, perspectives, and challenges of bioactive peptide generation: A comprehensive systematic review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1488–1520. [Google Scholar] [CrossRef]
- Tu, Z.; Chen, L.; Wang, H.; Ruan, C.; Zhang, L.; Kou, Y. Effect of fermentation and dynamic high pressure microfluidization on dietary fibre of soybean residue. J. Food Sci. Technol. 2014, 51, 3285–3292. [Google Scholar] [CrossRef]
- Rekha, C.R.; Vijayalakshmi, G. Influence of processing parameters on the quality of soycurd (tofu). J. Food Sci. Technol. 2013, 50, 176–180. [Google Scholar] [CrossRef]
- Matemu, A.O.; Katayama, S.; Kayahara, H.; Murasawa, H.; Nakamura, S. Improving surface functional properties of tofu whey-derived peptides by chemical modification with fatty acids. J. Food Sci. 2012, 77, C333–C339. [Google Scholar] [CrossRef] [PubMed]
- Jang, C.H.; Oh, J.; Lim, J.S.; Kim, H.J.; Kim, J.S. Fermented soy products: Beneficial potential in neurodegenerative diseases. Foods 2021, 10, 636. [Google Scholar] [CrossRef]
- Rivero-Pino, F.; Espejo-Carpio, F.J.; Guadix, E.M. Antidiabetic food-derived peptides for functional feeding: Production, functionality and in vivo evidences. Foods 2020, 9, 983. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liao, A.; Hou, Y.; Pan, L.; Yu, G.; Du, J.; Yang, C.; Li, X.; Huang, J. Digestive characteristics and peptide release from wheat embryo proteins in vitro. Food Funct. 2021, 12, 2257–2269. [Google Scholar] [CrossRef]
- Nath, A.; Kailo, G.G.; Mednyanszky, Z.; Kisko, G.; Csehi, B.; Pasztorne-Huszar, K.; Gerencser-Berta, R.; Galambos, I.; Pozsgai, E.; Banvolgyi, S.; et al. Antioxidant and antibacterial peptides from soybean milk through enzymatic- and membrane-based technologies. Bioengineering 2019, 7, 5. [Google Scholar] [CrossRef]
- Li, X.; Liu, X.; Hua, Y.; Chen, Y.; Kong, X.; Zhang, C. Effects of water absorption of soybean seed on the quality of soymilk and the release of flavor compounds. RSC Adv. 2019, 9, 2906–2918. [Google Scholar] [CrossRef]
- Amigo, L.; Hernandez-Ledesma, B. Current evidence on the bioavailability of food bioactive peptides. Molecules 2020, 25, 4479. [Google Scholar] [CrossRef] [PubMed]
- Daher, D.; Deracinois, B.; Baniel, A.; Wattez, E.; Dantin, J.; Froidevaux, R.; Chollet, S.; Flahaut, C. Principal component analysis from mass spectrometry data combined to a sensory evaluation as a suitable method for assessing bitterness of enzymatic hydrolysates produced from micellar casein proteins. Foods 2020, 9, 1354. [Google Scholar] [CrossRef]
- Rizzo, G.; Baroni, L. Soy, soy foods and their role in vegetarian diets. Nutrients 2018, 10, 43. [Google Scholar] [CrossRef]
- Coscueta, E.R.; Campos, D.A.; Osorio, H.; Nerli, B.B.; Pintado, M. Enzymatic soy protein hydrolysis: A tool for biofunctional food ingredient production. Food Chem. X 2019, 1, 100006. [Google Scholar] [CrossRef]
- Caponio, G.R.; Wang, D.Q.; Di Ciaula, A.; De Angelis, M.; Portincasa, P. Regulation of cholesterol metabolism by bioactive components of soy proteins: Novel translational evidence. Int. J. Mol. Sci. 2020, 22, 227. [Google Scholar] [CrossRef]
- Jesch, E.D.; Carr, T.P. Food ingredients that inhibit cholesterol absorption. Prev. Nutr. Food Sci. 2017, 22, 67–80. [Google Scholar]
- Martínez-Sánchez, S.M.; Gabaldón-Hernández, J.A.; Montoro-García, S. Unravelling the molecular mechanisms associated with the role of food-derived bioactive peptides in promoting cardiovascular health. J. Funct. Foods 2020, 64, 103645. [Google Scholar] [CrossRef]
- Nagaoka, S. Structure-function properties of hypolipidemic peptides. J. Food Biochem. 2019, 43, e12539. [Google Scholar] [CrossRef]
- Sánchez, A.; Vázquez, A. Bioactive peptides: A review. Food Qual. Saf. 2017, 1, 29–46. [Google Scholar] [CrossRef]
- Maestri, E.; Pavlicevic, M.; Montorsi, M.; Marmiroli, N. Meta-Analysis for correlating structure of bioactive peptides in foods of animal origin with regard to effect and stability. Compr. Rev. Food Sci. Food Saf. 2019, 18, 3–30. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Castilla, J.; Hernández-Álvarez, A.J.; Jiménez-Martínez, C.; Gutiérrez-López, G.F.; Dávila-Ortiz, G. Use of proteomics and peptidomics methods in food bioactive peptide science and engineering. Food Eng. Rev. 2012, 4, 224–243. [Google Scholar] [CrossRef]
- Hay, M.; Barnes, C.; Huentelman, M.; Brinton, R.; Ryan, L. Hypertension and age-related cognitive impairment: Common risk factors and a role for precision aging. Curr. Hypertens. Rep. 2020, 22, 80. [Google Scholar] [CrossRef] [PubMed]
- Mohd Sairazi, N.S.; Sirajudeen, K.N.S. Natural products and their bioactive compounds: Neuroprotective potentials against neurodegenerative diseases. Evid. Based Complement. Altern. Med. 2020, 2020, 6565396. [Google Scholar] [CrossRef]
- Ju, D.T.; K, A.K.; Kuo, W.W.; Ho, T.J.; Chang, R.L.; Lin, W.T.; Day, C.H.; Viswanadha, V.V.P.; Liao, P.H.; Huang, C.Y. Bioactive peptide VHVV upregulates the long-term memory-related biomarkers in adult spontaneously hypertensive rats. Int. J. Mol. Sci. 2019, 20, 3069. [Google Scholar] [CrossRef]
- Huang, L.K.; Chao, S.P.; Hu, C.J. Clinical trials of new drugs for Alzheimer disease. J. Biomed. Sci. 2020, 27, 18. [Google Scholar] [CrossRef]
- Katayama, S.; Imai, R.; Sugiyama, H.; Nakamura, S. Oral administration of soy peptides suppresses cognitive decline by induction of neurotrophic factors in SAMP8 mice. J. Agric. Food Chem. 2014, 62, 3563–3569. [Google Scholar] [CrossRef]
- Gu, Y.; Wu, J. LC-MS/MS coupled with QSAR modeling in characterising of angiotensin I-converting enzyme inhibitory peptides from soybean proteins. Food Chem. 2013, 141, 2682–2690. [Google Scholar] [CrossRef] [PubMed]
- Kodera, T.; Nio, N. Identification of an angiotensin I-converting enzyme inhibitory peptides from protein hydrolysates by a soybean protease and the antihypertensive effects of hydrolysates in 4 spontaneously hypertensive model rats. J. Food Sci. 2006, 71, C164–C173. [Google Scholar] [CrossRef]
- Fujita, H.; Usui, H.; Kurahashi, K.; Yoshikawa, M. Isolation and characterization of ovokinin, a bradykinin B1 agonist peptide derived from ovalbumin. Peptides 1995, 16, 785–790. [Google Scholar] [CrossRef]
- Matoba, N.; Usui, H.; Fujita, H.; Yoshikawa, M. A novel anti-hypertensive peptide derived from ovalbumin induces nitric oxide-mediated vasorelaxation in an isolated SHR mesenteric artery. FEBS Lett. 1999, 452, 181–184. [Google Scholar] [CrossRef]
- Kuba, M.; Tanaka, K.; Tawata, S.; Takeda, Y.; Yasuda, M. Angiotensin I-converting enzyme inhibitory peptides isolated from tofuyo fermented soybean food. Biosci. Biotechnol. Biochem. 2003, 67, 1278–1283. [Google Scholar] [CrossRef]
- Singh, B.P.; Vij, S. Growth and bioactive peptides production potential of Lactobacillus plantarum strain C2 in soy milk: A LC-MS/MS based revelation for peptides biofunctionality. LWT 2017, 86, 293–301. [Google Scholar] [CrossRef]
- Korhonen, H.; Pihlanto, A. Food-derived bioactive peptides—Opportunities for designing future foods. Curr. Pharm. Des. 2003, 9, 1297–1308. [Google Scholar] [CrossRef] [PubMed]
- Kitts, D.D.; Weiler, K. Bioactive proteins and peptides from food sources. Applications of bioprocesses used in isolation and recovery. Curr. Pharm. Des. 2003, 9, 1309–1323. [Google Scholar] [CrossRef] [PubMed]
- Tsuruki, T.; Kishi, K.; Takahashi, M.; Tanaka, M.; Matsukawa, T.; Yoshikawa, M. Soymetide, an immunostimulating peptide derived from soybean β-conglycinin, is an fMLP agonist. FEBS Lett. 2003, 540, 206–210. [Google Scholar] [CrossRef]
- Egusa, S.; Otani, H. Characterization of a cellular immunostimulating peptide from a soybean protein fraction digested with peptidase R. J. Nutr. Sci. Vitam. 2009, 55, 428–433. [Google Scholar] [CrossRef]
- Guijarro-Diez, M.; Garcia, M.C.; Marina, M.L.; Crego, A.L. LC-ESI-TOF MS method for the evaluation of the immunostimulating activity of soybeans via the determination of the functional peptide soymetide. J. Agric. Food Chem. 2013, 61, 3611–3618. [Google Scholar] [CrossRef]
- McConnell, E.J.; Devapatla, B.; Yaddanapudi, K.; Davis, K.R. The soybean-derived peptide lunasin inhibits non-small cell lung cancer cell proliferation by suppressing phosphorylation of the retinoblastoma protein. Oncotarget 2015, 6, 4649–4662. [Google Scholar] [CrossRef]
- Fernandez-Tome, S.; Xu, F.; Han, Y.; Hernandez-Ledesma, B.; Xiao, H. Inhibitory effects of peptide lunasin in colorectal cancer HCT-116 cells and their tumorsphere-derived subpopulation. Int. J. Mol. Sci. 2020, 21, 537. [Google Scholar] [CrossRef] [PubMed]
- Guha, S.; Majumder, K. Structural-features of food-derived bioactive peptides with anti-inflammatory activity: A brief review. J. Food Biochem. 2019, 43, e12531. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. Biomed. Res. Int. 2014, 2014, 608979. [Google Scholar] [CrossRef]
- Gu, L.; Wang, Y.; Xu, Y.; Tian, Q.; Lei, G.; Zhao, C.; Gao, Z.; Pan, Q.; Zhao, W.; Nong, L.; et al. Lunasin functionally enhances LDL uptake via inhibiting PCSK9 and enhancing LDLR expression in vitro and in vivo. Oncotarget 2017, 8, 80826–80840. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Montoya, M.; Hernandez-Ledesma, B.; Silvan, J.M.; Mora-Escobedo, R.; Martinez-Villaluenga, C. Peptides derived from in vitro gastrointestinal digestion of germinated soybean proteins inhibit human colon cancer cells proliferation and inflammation. Food Chem. 2018, 242, 75–82. [Google Scholar] [CrossRef]
- Pan, F.; Wang, L.; Cai, Z.; Wang, Y.; Wang, Y.; Guo, J.; Xu, X.; Zhang, X. Soybean peptide QRPR activates autophagy and attenuates the inflammatory response in the RAW264.7 cell model. Protein Pept. Lett. 2019, 26, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Cam, A.; Sivaguru, M.; Gonzalez de Mejia, E. Endocytic mechanism of internalization of dietary peptide lunasin into macrophages in inflammatory condition associated with cardiovascular disease. PLoS ONE 2013, 8, e72115. [Google Scholar] [CrossRef]
- Nagaoka, S.; Nakamura, A.; Shibata, H.; Kanamaru, Y. Soystatin (VAWWMY), a novel bile acid-binding peptide, decreased micellar solubility and inhibited cholesterol absorption in rats. Biosci. Biotechnol. Biochem. 2010, 74, 1738–1741. [Google Scholar] [CrossRef]
- Nagaoka, S. Mystery of cholesterol-lowering peptides, lactostatin and soystatin. J. Agric. Food Chem. 2018, 66, 3993–3994. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, S.; Futamura, Y.; Miwa, K.; Awano, T.; Yamauchi, K.; Kanamaru, Y.; Tadashi, K.; Kuwata, T. Identification of novel hypocholesterolemic peptides derived from bovine milk beta-lactoglobulin. Biochem. Biophys. Res. Commun. 2001, 281, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, K.; Kondo, I.; Kanamaru, Y.; Nagaoka, S. A novel regulatory pathway for cholesterol degradation via lactostatin. Biochem. Biophys. Res. Commun. 2007, 352, 697–702. [Google Scholar] [CrossRef]
- Yamauchi, R.; Ohinata, K.; Yoshikawa, M. Beta-lactotensin and neurotensin rapidly reduce serum cholesterol via NT2 receptor. Peptides 2003, 24, 1955–1961. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Bartley, G.E.; Zhang, H.; Jing, W.; Fagerquist, C.K.; Zhong, F.; Yokoyama, W. Peptides identified in soybean protein increase plasma cholesterol in mice on hypercholesterolemic diets. J. Agric. Food Chem. 2013, 61, 8389–8395. [Google Scholar] [CrossRef]
- Lammi, C.; Zanoni, C.; Arnoldi, A.; Vistoli, G. Two peptides from soy beta-conglycinin induce a hypocholesterolemic effect in HepG2 cells by a statin-like mechanism: Comparative in vitro and in silico modeling studies. J. Agric. Food Chem. 2015, 63, 7945–7951. [Google Scholar] [CrossRef]
- Wu, Y.J.; Hughes, D.; Lin, L.; Braymer, D.H.; York, D.A. Comparative study of enterostatin sequence in five rat strains and enterostatin binding proteins in rat and chicken serum. Peptides 2002, 23, 537–544. [Google Scholar] [CrossRef]
- Takenaka, Y.; Nakamura, F.; Yamamoto, T.; Yoshikawa, M. Enterostatin (VPDPR) and its peptide fragment DPR reduce serum cholesterol levels after oral administration in mice. Biosci. Biotechnol. Biochem. 2003, 67, 1620–1622. [Google Scholar] [CrossRef]
- Takenaka, Y.; Shimano, T.; Mori, T.; Hou, I.C.; Ohinata, K.; Yoshikawa, M. Enterostatin reduces serum cholesterol levels by way of a CCK(1) receptor-dependent mechanism. Peptides 2008, 29, 2175–2178. [Google Scholar] [CrossRef]
- Marques, M.R.; Fontanari, G.G.; Pimenta, D.C.; Soares-Freitas, R.M.; Arêas, J.A.G. Proteolytic hydrolysis of cowpea proteins is able to release peptides with hypocholesterolemic activity. Food Res. Int. 2015, 77, 43–48. [Google Scholar] [CrossRef]
- Lammi, C.; Zanoni, C.; Arnoldi, A. IAVPGEVA, IAVPTGVA, and LPYP, three peptides from soy glycinin, modulate cholesterol metabolism in HepG2 cells through the activation of the LDLR-SREBP2 pathway. J. Funct. Foods 2015, 14, 469–478. [Google Scholar] [CrossRef]
- Lammi, C.; Zanoni, C.; Aiello, G.; Arnoldi, A.; Grazioso, G. Lupin peptides modulate the protein-protein interaction of PCSK9 with the low density lipoprotein receptor in HepG2 Cells. Sci. Rep. 2016, 6, 29931. [Google Scholar] [CrossRef] [PubMed]
- Lammi, C.; Aiello, G.; Vistoli, G.; Zanoni, C.; Arnoldi, A.; Sambuy, Y.; Ferruzza, S.; Ranaldi, G. A multidisciplinary investigation on the bioavailability and activity of peptides from lupin protein. J. Funct. Foods 2016, 24, 297–306. [Google Scholar] [CrossRef]
- Zanoni, C.; Aiello, G.; Arnoldi, A.; Lammi, C. Investigations on the hypocholesterolaemic activity of LILPKHSDAD and LTFPGSAED, two peptides from lupin β-conglutin: Focus on LDLR and PCSK9 pathways. J. Funct. Foods 2017, 32, 1–8. [Google Scholar] [CrossRef]
- Kagawa, K.; Matsutaka, H.; Fukuhama, C.; Fujino, H.; Okuda, H. Suppressive effect of globin digest on postprandial hyperlipidemia in male volunteers. J. Nutr. 1998, 128, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Kim, J.H.; Nam, B.; Kim, J.; Lee, J.H.; Hwang, K.Y.; Lee, S.J. The dipeptide H-Trp-Glu-OH (WE) shows agonistic activity to peroxisome proliferator-activated protein-alpha and reduces hepatic lipid accumulation in lipid-loaded H4IIE cells. Bioorg. Med. Chem. Lett. 2014, 24, 2957–2962. [Google Scholar] [CrossRef]
- Nagase, H.; Bray, G.A.; York, D.A. Effect of galanin and enterostatin on sympathetic nerve activity to interscapular brown adipose tissue. Brain Res. 1996, 709, 44–50. [Google Scholar] [CrossRef]
- Yamada, Y.; Muraki, A.; Oie, M.; Kanegawa, N.; Oda, A.; Sawashi, Y.; Kaneko, K.; Yoshikawa, M.; Goto, T.; Takahashi, N.; et al. Soymorphin-5, a soy-derived mu-opioid peptide, decreases glucose and triglyceride levels through activating adiponectin and PPARalpha systems in diabetic KKAy mice. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E433–E440. [Google Scholar] [CrossRef] [PubMed]
- Navab, M.; Anantharamaiah, G.M.; Reddy, S.T.; Hama, S.; Hough, G.; Frank, J.S.; Grijalva, V.R.; Ganesh, V.K.; Mishra, V.K.; Palgunachari, M.N.; et al. Oral small peptides render HDL antiinflammatory in mice and monkeys and reduce atherosclerosis in ApoE null mice. Circ. Res. 2005, 97, 524–532. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Rupasinghe, S.G.; Schuler, M.A.; Gonzalez de Mejia, E. Peptides from purified soybean beta-conglycinin inhibit fatty acid synthase by interaction with the thioesterase catalytic domain. FEBS J. 2010, 277, 1481–1493. [Google Scholar] [CrossRef] [PubMed]
- Tsou, M.J.; Kao, F.J.; Lu, H.C.; Kao, H.C.; Chiang, W.D. Purification and identification of lipolysis-stimulating peptides derived from enzymatic hydrolysis of soy protein. Food Chem. 2013, 138, 1454–1460. [Google Scholar] [CrossRef] [PubMed]
- Inoue, N.; Nagao, K.; Sakata, K.; Yamano, N.; Gunawardena, P.E.; Han, S.Y.; Matsui, T.; Nakamori, T.; Furuta, H.; Takamatsu, K.; et al. Screening of soy protein-derived hypotriglyceridemic di-peptides in vitro and in vivo. Lipids Health Dis. 2011, 10, 85. [Google Scholar] [CrossRef] [PubMed]
- Kolonin, M.G.; Saha, P.K.; Chan, L.; Pasqualini, R.; Arap, W. Reversal of obesity by targeted ablation of adipose tissue. Nat. Med. 2004, 10, 625–632. [Google Scholar] [CrossRef]
- Van Lenten, B.J.; Wagner, A.C.; Anantharamaiah, G.M.; Navab, M.; Reddy, S.T.; Buga, G.M.; Fogelman, A.M. Apolipoprotein A-I mimetic peptides. Curr. Atheroscler. Rep. 2009, 11, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Sato, M.; Tanaka, M.; Yamada, Y.; Watanabe, S.; Fujimoto, Y.; Imaizumi, K.; Matsumoto, K. Vasodilating dipeptide Trp-His can prevent atherosclerosis in apo E-deficient mice. Br. J. Nutr. 2010, 103, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Lammi, C.; Bollati, C.; Ferruzza, S.; Ranaldi, G.; Sambuy, Y.; Arnoldi, A. Soybean- and lupin-derived peptides inhibit DPP-IV activity on in situ human intestinal Caco-2 cells and ex vivo human serum. Nutrients 2018, 10, 1082. [Google Scholar] [CrossRef] [PubMed]
- Mojica, L.; Gonzalez de Mejia, E.; Granados-Silvestre, M.Á.; Menjivar, M. Evaluation of the hypoglycemic potential of a black bean hydrolyzed protein isolate and its pure peptides using in silico, in vitro and in vivo approaches. J. Funct. Foods 2017, 31, 274–286. [Google Scholar] [CrossRef]
- Jiang, H.; Feng, J.; Du, Z.; Zhen, H.; Lin, M.; Jia, S.; Li, T.; Huang, X.; Ostenson, C.G.; Chen, Z. Oral administration of soybean peptide Vglycin normalizes fasting glucose and restores impaired pancreatic function in Type 2 diabetic Wistar rats. J. Nutr. Biochem. 2014, 25, 954–963. [Google Scholar] [CrossRef]
- Shin, Z.I.; Yu, R.; Park, S.A.; Chung, D.K.; Ahn, C.W.; Nam, H.S.; Kim, K.S.; Lee, H.J. His-His-Leu, an angiotensin I converting enzyme inhibitory peptide derived from Korean soybean paste, exerts antihypertensive activity in vivo. J. Agric. Food Chem. 2001, 49, 3004–3009. [Google Scholar] [CrossRef]
- Ma, H.; Liu, R.; Zhao, Z.; Zhang, Z.; Cao, Y.; Ma, Y.; Guo, Y.; Xu, L. A novel peptide from soybean protein isolate significantly enhances resistance of the organism under oxidative stress. PLoS ONE 2016, 11, e0159938. [Google Scholar] [CrossRef]
- Wang, R.; Zhao, H.; Pan, X.; Orfila, C.; Lu, W.; Ma, Y. Preparation of bioactive peptides with antidiabetic, antihypertensive, and antioxidant activities and identification of alpha-glucosidase inhibitory peptides from soy protein. Food Sci. Nutr. 2019, 7, 1848–1856. [Google Scholar] [CrossRef]
- Cavaliere, C.; Montone, A.M.I.; Aita, S.E.; Capparelli, R.; Cerrato, A.; Cuomo, P.; Lagana, A.; Montone, C.M.; Piovesana, S.; Capriotti, A.L. Production and characterization of medium-sized and short antioxidant peptides from soy flour-simulated gastrointestinal hydrolysate. Antioxidants 2021, 10, 734. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; He, H.; Xiang, J.; Li, B.; Zhao, M.; Hou, T. Selenium-containing soybean antioxidant peptides: Preparation and comprehensive comparison of different selenium supplements. Food Chem. 2021, 358, 129888. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Kong, X.; Chen, Y.; Zhang, C.; Hua, Y.; Li, X. Selective extraction and antioxidant properties of thiol-containing peptides in soy glycinine hydrolysates. Molecules 2018, 23, 1909. [Google Scholar] [CrossRef]
- Zhang, Q.; Tong, X.; Qi, B.; Wang, Z.; Li, Y.; Sui, X.; Jiang, L. Changes in antioxidant activity of Alcalase-hydrolyzed soybean hydrolysate under simulated gastrointestinal digestion and transepithelial transport. J. Funct. Foods 2018, 42, 298–305. [Google Scholar] [CrossRef]
- Dhayakaran, R.; Neethirajan, S.; Weng, X. Investigation of the antimicrobial activity of soy peptides by developing a high throughput drug screening assay. Biochem. Biophys. Rep. 2016, 6, 149–157. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cheng, A.C.; Lin, H.L.; Shiu, Y.L.; Tyan, Y.C.; Liu, C.H. Isolation and characterization of antimicrobial peptides derived from Bacillus subtilis E20-fermented soybean meal and its use for preventing Vibrio infection in shrimp aquaculture. Fish Shellfish Immunol. 2017, 67, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Padhi, S.; Sanjukta, S.; Chourasia, R.; Labala, R.K.; Singh, S.P.; Rai, A.K. A multifunctional peptide from Bacillus fermented soybean for effective inhibition of SARS-CoV-2 S1 receptor binding domain and modulation of Toll like receptor 4: A molecular docking study. Front. Mol. Biosci. 2021, 8, 636647. [Google Scholar] [CrossRef]
- Mafra, D.; Borges, N.; Alvarenga, L.; Esgalhado, M.; Cardozo, L.; Lindholm, B.; Stenvinkel, P. Dietary components that may influence the disturbed gut microbiota in chronic kidney disease. Nutrients 2019, 11, 496. [Google Scholar] [CrossRef] [PubMed]
- Devries, M.C.; Sithamparapillai, A.; Brimble, K.S.; Banfield, L.; Morton, R.W.; Phillips, S.M. Changes in kidney function do not differ between healthy adults consuming higher- compared with lower- or normal-protein diets: A systematic review and meta-analysis. J. Nutr. 2018, 148, 1760–1775. [Google Scholar] [CrossRef]
- He, L.X.; Abdolmaleky, H.M.; Yin, S.; Wang, Y.; Zhou, J.R. Dietary fermented soy extract and oligo-lactic acid alleviate chronic kidney disease in mice via inhibition of inflammation and modulation of gut microbiota. Nutrients 2020, 12, 2376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shi, W.; He, H.; Cao, R.; Hou, T. Hypolipidemic effects and mechanisms of Val-Phe-Val-Arg-Asn in C57BL/6J mice and 3T3-L1 cell models. J. Funct. Foods 2020, 73, 104100. [Google Scholar] [CrossRef]
- Kojima-Yuasa, A.; Huang, X.; Matsui-Yuasa, I. Synergistic anticancer activities of natural substances in human hepatocellular carcinoma. Diseases 2015, 3, 260–281. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Yao, Y.; Shi, Z.; Everaert, N.; Ren, G. Synergistic effect of bioactive anticarcinogens from soybean on anti-proliferative activity in MDA-MB-231 and MCF-7 human breast cancer cells in vitro. Molecules 2018, 23, 1557. [Google Scholar] [CrossRef] [PubMed]
- Aviles-Gaxiola, S.; Gutierrez-Grijalva, E.P.; Leon-Felix, J.; Angulo-Escalante, M.A.; Heredia, J.B. Peptides in colorectal cancer: Current state of knowledge. Plant Foods Hum. Nutr. 2020, 75, 467–476. [Google Scholar] [CrossRef]
- Rayaprolu, S.J.; Hettiarachchy, N.S.; Chen, P.; Kannan, A.; Mauromostakos, A. Peptides derived from high oleic acid soybean meals inhibit colon, liver and lung cancer cell growth. Food Res. Int. 2013, 50, 282–288. [Google Scholar] [CrossRef]
- Nagaoka, S.; Takeuchi, A.; Banno, A. Plant-derived peptides improving lipid and glucose metabolism. Peptides 2021, 142, 170577. [Google Scholar] [CrossRef] [PubMed]
- Kohno, M.; Hirotsuka, M.; Kito, M.; Matsuzawa, Y. Decreases in serum triacylglycerol and visceral fat mediated by dietary soybean beta-conglycinin. J. Atheroscler. Thromb. 2006, 13, 247–255. [Google Scholar] [CrossRef]
- Hashidume, T.; Kato, A.; Tanaka, T.; Miyoshi, S.; Itoh, N.; Nakata, R.; Inoue, H.; Oikawa, A.; Nakai, Y.; Shimizu, M.; et al. Single ingestion of soy beta-conglycinin induces increased postprandial circulating FGF21 levels exerting beneficial health effects. Sci. Rep. 2016, 6, 28183. [Google Scholar] [CrossRef]
- Goto, T.; Mori, A.; Nagaoka, S. Soluble soy protein peptic hydrolysate stimulates adipocyte differentiation in 3T3-L1 cells. Mol. Nutr. Food Res. 2013, 57, 1435–1445. [Google Scholar] [CrossRef]
- Tachibana, N.; Yamashita, Y.; Nagata, M.; Wanezaki, S.; Ashida, H.; Horio, F.; Kohno, M. Soy beta-conglycinin improves glucose uptake in skeletal muscle and ameliorates hepatic insulin resistance in Goto-Kakizaki rats. Nutr. Res. 2014, 34, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zeng, Y.; Hou, W.; Zhang, S.; Li, L.; Luo, X.; Xi, W.; Chen, Z.; Xiang, M. The soybean peptide aglycin regulates glucose homeostasis in type 2 diabetic mice via IR/IRS1 pathway. J. Nutr. Biochem. 2012, 23, 1449–1457. [Google Scholar] [CrossRef]
- Zhou, B.R.; Ma, L.W.; Liu, J.; Zhang, J.A.; Xu, Y.; Wu, D.; Permatasari, F.; Luo, D. Protective effects of soy oligopeptides in ultraviolet B-induced acute photodamage of human skin. Oxid. Med. Cell. Longev. 2016, 2016, 5846865. [Google Scholar] [CrossRef]
- Kwak, J.H.; Kim, M.; Lee, E.; Lee, S.H.; Ahn, C.W.; Lee, J.H. Effects of black soy peptide supplementation on blood pressure and oxidative stress: A randomized controlled trial. Hypertens. Res. 2013, 36, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.Y.; Zhang, J.T.; Miyakawa, T.; Li, G.M.; Gu, R.Z.; Tanokura, M. Antioxidant properties and inhibition of angiotensin-converting enzyme by highly active peptides from wheat gluten. Sci. Rep. 2021, 11, 5206. [Google Scholar] [CrossRef]
- Matemu, A.; Nakamura, S.; Katayama, S. Health benefits of antioxidative peptides derived from legume proteins with a high amino acid score. Antioxidants 2021, 10, 316. [Google Scholar] [CrossRef]
- Jimenez-Ruiz, E.I.; Calderon de la Barca, A.M.; Sotelo-Mundo, R.R.; Arteaga-Mackinney, G.E.; Valenzuela-Melendez, M.; Pena-Ramos, E.A. Partial characterization of ultrafiltrated soy protein hydrolysates with antioxidant and free radical scavenging activities. J. Food Sci. 2013, 78, C1152–C1158. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jiang, B.; Zhang, T.; Mu, W.; Liu, J. Antioxidant and free radical-scavenging activities of chickpea protein hydrolysate (CPH). Food Chem. 2008, 106, 444–450. [Google Scholar] [CrossRef]
- Shi, Z.; Yao, Y.; Zhu, Y.; Ren, G. Nutritional composition and antioxidant activity of twenty mung bean cultivars in China. Crop J. 2016, 4, 398–406. [Google Scholar] [CrossRef]
- Onuh, J.O.; Girgih, A.T.; Aluko, R.E.; Aliani, M. In vitro antioxidant properties of chicken skin enzymatic protein hydrolysates and membrane fractions. Food Chem. 2014, 150, 366–373. [Google Scholar] [CrossRef]
- Chen, C.; Chi, Y.J.; Zhao, M.Y.; Lv, L. Purification and identification of antioxidant peptides from egg white protein hydrolysate. Amino Acids 2012, 43, 457–466. [Google Scholar] [CrossRef]
- Hernandez-Ledesma, B.; Hsieh, C.C.; de Lumen, B.O. Antioxidant and anti-inflammatory properties of cancer preventive peptide lunasin in RAW 264.7 macrophages. Biochem. Biophys. Res. Commun. 2009, 390, 803–808. [Google Scholar] [CrossRef]
- Moure, A.; Domínguez, H.; Parajó, J.C. Antioxidant properties of ultrafiltration-recovered soy protein fractions from industrial effluents and their hydrolysates. Process Biochem. 2006, 41, 447–456. [Google Scholar] [CrossRef]
- Wang, H.; Jenner, A.M.; Lee, C.Y.; Shui, G.; Tang, S.Y.; Whiteman, M.; Wenk, M.R.; Halliwell, B. The identification of antioxidants in dark soy sauce. Free Radic. Res. 2007, 41, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Marcela, G.M.; Eva, R.G.; Del Carmen, R.M.; Rosalva, M.E. Evaluation of the antioxidant and antiproliferative effects of three peptide fractions of germinated soybeans on breast and cervical cancer cell lines. Plant Foods Hum. Nutr. 2016, 71, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Ji, X.; Canning, C.; Sun, S.; Zhou, K. Comparison of different strategies for soybean antioxidant extraction. J. Agric. Food Chem. 2010, 58, 4508–4512. [Google Scholar] [CrossRef]
- Chen, C.; Sun-Waterhouse, D.; Zhang, Y.; Zhao, M.; Sun, W. The chemistry behind the antioxidant actions of soy protein isolate hydrolysates in a liposomal system: Their performance in aqueous solutions and liposomes. Food Chem. 2020, 323, 126789. [Google Scholar] [CrossRef]
- Guo, Y.; Ma, M.; Jiang, F.; Jiang, W.; Wang, H.; Du, S.K. Protein quality and antioxidant properties of soymilk derived from black soybean after in vitro simulated gastrointestinal digestion. Int. J. Food Sci. Technol. 2019, 55, 720–728. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, J.; Zhu, H.; Xiong, Y.L. Mass spectrometric evidence of malonaldehyde and 4-hydroxynonenal adductions to radical-scavenging soy peptides. J. Agric. Food Chem. 2012, 60, 9727–9736. [Google Scholar] [CrossRef]
- Marazza, J.A.; Nazareno, M.A.; de Giori, G.S.; Garro, M.S. Enhancement of the antioxidant capacity of soymilk by fermentation with Lactobacillus rhamnosus. J. Funct. Foods 2012, 4, 594–601. [Google Scholar] [CrossRef]
- Wang, Y.C.; Yu, R.C.; Chou, C.C. Antioxidative activities of soymilk fermented with lactic acid bacteria and bifidobacteria. Food Microbiol. 2006, 23, 128–135. [Google Scholar] [CrossRef]
- Singh, B.P.; Bhushan, B.; Vij, S. Antioxidative, ACE inhibitory and antibacterial activities of soy milk fermented by indigenous strains of lactobacilli. Legume Sci. 2020, 2, e54. [Google Scholar] [CrossRef]
- Yamamoto, N.; Shoji, M.; Hoshigami, H.; Watanabe, K.; Watanabe, K.; Takatsuzu, T.; Yasuda, S.; Igoshi, K.; Kinoshita, H. Antioxidant capacity of soymilk yogurt and exopolysaccharides produced by lactic acid bacteria. Biosci. Microbiota Food Health 2019, 38, 97–104. [Google Scholar] [CrossRef]
- Li, G.; Long, X.; Pan, Y.; Zhao, X.; Song, J.L. Study on soybean milk fermented by Lactobacillus plantarum YS-1 reduced the H2O2-induced oxidative damage in Caco-2 cells. Biomed. Res. 2018, 29, 357–364. [Google Scholar] [CrossRef]
- Miri, S.; Hajihosseini, R.; Saedi, H.; Vaseghi, M.; Rasooli, A. Fermented soybean meal extract improves oxidative stress factors in the lung of inflammation/infection animal model. Ann. Microbiol. 2019, 69, 1507–1515. [Google Scholar] [CrossRef]
- De Oliveira, C.F.; Corrêa, A.P.F.; Coletto, D.; Daroit, D.J.; Cladera-Olivera, F.; Brandelli, A. Soy protein hydrolysis with microbial protease to improve antioxidant and functional properties. J. Food Sci. Technol. 2014, 52, 2668–2678. [Google Scholar] [CrossRef] [PubMed]
- Bai, G.; Tsuruta, T.; Nishino, N. Dietary soy, meat, and fish proteins modulate the effects of prebiotic raffinose on composition and fermentation of gut microbiota in rats. Int. J. Food Sci. Nutr. 2018, 69, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Davoodi, S.H.; Shahbazi, R.; Esmaeili, S.; Sohrabvandi, S.; Mortazavian, A.; Jazayeri, S.; Aghdas Taslimi, A. Health-related aspects of milk proteins. Iran. J. Pharm. Res. 2016, 15, 573–591. [Google Scholar] [PubMed]
- Zheng, L.; Regenstein, J.M.; Teng, F.; Li, Y. Tofu products: A review of their raw materials, processing conditions, and packaging. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3683–3714. [Google Scholar] [CrossRef]
- Henchion, M.; Hayes, M.; Mullen, A.M.; Mark Fenelon, M.; Brijesh Tiwari, B. Future protein supply and demand: Strategies and factors influencing a sustainable equilibrium. Foods 2017, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Ko, B.B.; Davaatseren, M.; Hong, G.P. Effects of soy protein hydrolysates prepared by varying subcritical media on the physicochemical properties of pork patties. Food Sci. Anim. Resour. 2016, 36, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Tokudome, Y.; Nakamura, K.; Kage, M.; Todo, H.; Sugibayashi, K.; Hashimoto, F. Effects of soybean peptide and collagen peptide on collagen synthesis in normal human dermal fibroblasts. Int. J. Food Sci. Nutr. 2012, 63, 689–695. [Google Scholar] [CrossRef]
- Park, J.; Bae, S.J.; Lee, T.J.; Son, K.B. Marketing and pricing strategies of blockbuster drugs in the South Korean market: A 15-year retrospective cohort study for choline alfoscerate. Front. Pharm. 2020, 11, 232. [Google Scholar] [CrossRef] [PubMed]
- Sismondo, S. Access to medicines, access to markets. Front. Sociol. 2020, 5, 58. [Google Scholar] [CrossRef]
- Zhang, J.; Li, W.; Ying, Z.; Di Zhao, D.; Yi, G.; Li, H.; Liu, X. Soybean protein-derived peptide nutriment increases negative nitrogen balance in burn injury-induced inflammatory stress response in aged rats through the modulation of white blood cells and immune factors. Food Nutr. Res. 2020, 64. [Google Scholar] [CrossRef] [PubMed]
- Amaya-Farfan, J. Denaturation of proteins, generation of bioactive peptides, and alterations of amino acids. In Chemical Changes during Processing and Storage of Foods: Implication for Food Quality and Human Health, 1st ed.; Rodriguez-Amaya, D.B., Amaya-Farfan, J., Eds.; Academic Press: London, UK, 2021; pp. 21–84. [Google Scholar]

| Peptide Sequence | Biological Effect | Reference |
|---|---|---|
| VHVV | Neuroprotection | [48] |
| IVF, LLF, LNF, LSW, LEF | ACE inhibitory | [51] |
| NWGPLV | ACE inhibitory | [52] |
| FRADHPFL, RADHPF | Lowering blood pressure and vasorelaxation | [53,54] |
| IFL, WL | ACE inhibitory | [55] |
| HSYNLRQSQVSELKYEGNWGPLV NPESQQGSPRV | ACE inhibitory and antioxidant | [56] |
| FRADHPFL, RADHPF | Lowering blood pressure and vasorelaxation | [53,54] |
| LLPVFK, RLPKPW | Anti-hypertensive | [57] |
| PGTAVPK | Anti-hypertensive | [58] |
| MITLAIPVNKPGR | Immunoregulatory | [59] |
| QQQQQQKSHGGR | Immunoregulatory | [60] |
| LITLAIPVNKPGR | Immunoregulatory | [61] |
| Lunasin (KWQHQQSCRKQLQGVNLTPCDD DDDDDDDEKHIMEKIQGRGDDDD DDD DDEKH IMEKIQ) | Anticancer, anti-inflammatory, antioxidant and cholesterol regulation | [62,63,64,65,66] |
| QQQQQGGSQSQ, QEPQESQQ, QQQQQGGSQSQSQKG, PETMQQQQQQ, | Anti-cancer | [67] |
| QRPR | Anti-inflammatory | [68] |
| RGD | Anti-inflammatory | [65,69] |
| Soystatin (VAWWMY) | Cholesterol regulation | [42,70] |
| Lactostatin (IIAEK) | Cholesterol regulation | [71,72,73] |
| Lactostensin (HIRL) | Cholesterol regulation | [74] |
| LPYPR, WGAPSL | Cholesterol regulation | [75] |
| IAVPGEVA, IAVPTGVA, LPYP | Cholesterol regulation and lipid metabolism | [76] |
| HIRL, DPR, FVVNATSN | Hypocholesterolemic | [75] |
| YVVNPDNDEN, YVVNPDNNEN | Hypocholesterolemic | [76] |
| DPR, VPDPR, APGPR, VPGPR | Hypocholesterolemic | [77,78,79] |
| GCTLN, QDF | Hypolipidemic | [80] |
| IAVPGEVA, IAVPTGVA, LPYP | Hypolipidemic | [81] |
| Lupin peptides (LIPKHSDAD, LTFPGSAED, GDEQSHQDEGVIVR) | Hypolipidemic and hypocholesterolemic | [82,83,84] |
| VVVP, YPFVV | Hypotriglyceridemic | [42,85] |
| WE | Hypocholesterolemic and hypotriglyceridemic | [86] |
| Enterostatin (VPDPR) | Hypocholesterolemic and hypotriglyceridemic | [87] |
| Soymorphin-5 (YPFVV) | Hypotriglyceridemic and anti-diabetic | [88] |
| KRES, FREL | Hypotriglyceridemic antioxidant, anti-inflammatory and anti-atherogenic | [42,89] |
| KNPQLR, EIPEKNPQLR, RKQEEDEDEEQQRE | FAS inhibitor | [90] |
| KNPQLR, EIPEKNPQLR, RKQEEDEDEEQQRE | Hypotriglyceridemic and FAS inhibitor | [42] |
| ILL, LLL, VHVV | Lipolysis and anti-obesity | [91] |
| SY | Anti-obesity | [92] |
| CKGGRAKDC | Anti-obesity | [93] |
| DWFKAFYDKVAEKFKEAF | Anti-arteriosclerosis | [94] |
| FREL | Anti-arteriosclerosis | [89] |
| WH | Anti-arteriosclerosis | [95] |
| IAVPTGVA | Anti-diabetic | [96] |
| AKSPLF, ATNPLF, FEELN, LSVSVL | Anti-diabetic | [97] |
| Vglycin (VSCNGVCSPFEMPPCGSS ACRCIPYGLVVGNCRHPSG) | Anti-diabetic | [98] |
| HHL | ACE inhibitory and antioxidant | [99] |
| FDPAL | Antioxidant | [100] |
| LLPLPVLK, SWLRL, WLRL | Antioxidant | [101] |
| LH, VNPESQQGSPR | Antioxidant | [102] |
| Selenium (Se)-containing peptide [SFQ(K)SeM and SeCPEE] | Antioxidant | [103] |
| Thiol-containing peptides containing Tyr, Met, His, Lys, and Cys | Antioxidant | [104] |
| GNPDIEHPE, TNDRPSIG, SVIKPPTDE, VIKPPTDE, GNPDIEHPET, LVPPQESQ, EITPEKNPQ, TLVNNDDRDS, NSQHPEL, FEEPQQPQ | Antioxidant | [105] |
| PGTAVFK, IKAFKEATKVDKVVVLWTA | Antimicrobial | [106] |
| KHPHGRSYKTKLRILA, LRFRAPAPVLRRIAKR, HTSKALLDMLKRLGK | Antimicrobial | [107] |
| ALPEEVIQHTFNLKSQ | Antiviral | [108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, I.-S.; Yang, W.-S.; Kim, C.-H. Beneficial Effects of Soybean-Derived Bioactive Peptides. Int. J. Mol. Sci. 2021, 22, 8570. https://doi.org/10.3390/ijms22168570
Kim I-S, Yang W-S, Kim C-H. Beneficial Effects of Soybean-Derived Bioactive Peptides. International Journal of Molecular Sciences. 2021; 22(16):8570. https://doi.org/10.3390/ijms22168570
Chicago/Turabian StyleKim, Il-Sup, Woong-Suk Yang, and Cheorl-Ho Kim. 2021. "Beneficial Effects of Soybean-Derived Bioactive Peptides" International Journal of Molecular Sciences 22, no. 16: 8570. https://doi.org/10.3390/ijms22168570
APA StyleKim, I.-S., Yang, W.-S., & Kim, C.-H. (2021). Beneficial Effects of Soybean-Derived Bioactive Peptides. International Journal of Molecular Sciences, 22(16), 8570. https://doi.org/10.3390/ijms22168570







