Brain Atrophy and White Matter Damage Linked to Peripheral Bioenergetic Deficits in the Neurodegenerative Disease FXTAS
Abstract
:1. Introduction
2. Results
2.1. Demographic Characteristics of Participants Included in This Study
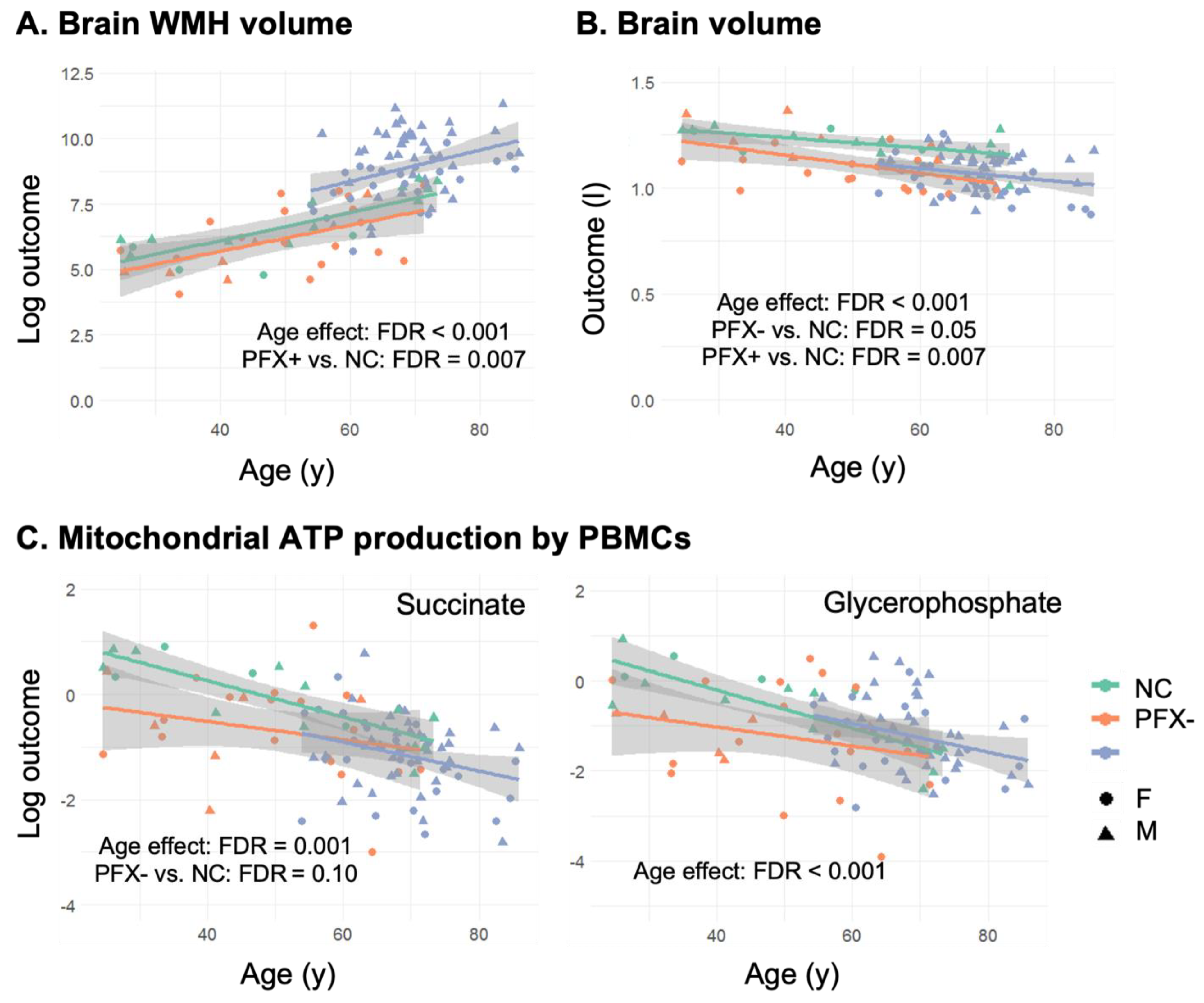
2.2. Smaller Brain Volume in Premutation Carriers Compared to Non-Carriers, and Higher WMHV in Carriers with FXTAS Stages ≥ 2
2.3. Lower Overall Oxygen-Linked ATP Production Fueled by Succinate in Premutation PBMCs but Higher with Glycerophosphate as Substrate in Carriers with FXTAS Stages ≥ 2
2.4. Age-Dependent Negative Correlation between WMHV and Oxygen-Linked ATP Production, and Positive Correlation between Brain Volume and ATP Production and Mitochondrial Mass in the Premutation Carriers
3. Discussion
4. Materials and Methods
4.1. Research Participants
4.2. PBMCs Preparation
4.3. Mitochondrial Outcomes
4.4. MRI Acquisition and Processing
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bickford, P.C.; Flowers, A.; Grimmig, B. Aging leads to altered microglial function that reduces brain resiliency increasing vulnerability to neurodegenerative diseases. Exp. Gerontol. 2017, 94, 4–8. [Google Scholar] [CrossRef]
- Ishii, R.; Canuet, L.; Aoki, Y.; Hata, M.; Iwase, M.; Ikeda, S.; Nishida, K.; Ikeda, M. Healthy and Pathological Brain Aging: From the Perspective of Oscillations, Functional Connectivity, and Signal Complexity. Neuropsychobiology 2017, 75, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Jan, A.T.; Azam, M.; Rahman, S.; Almigeiti, A.; Choi, D.H.; Lee, E.J.; Haq, Q.M.R.; Choi, I. Perspective Insights into Disease Progression, Diagnostics, and Therapeutic Approaches in Alzheimer’s disease: A Judicious Update. Front. Aging Neurosci. 2017, 9, 356. [Google Scholar] [CrossRef]
- Childs, B.G.; Gluscevic, M.; Baker, D.J.; Laberge, R.-M.; Marquess, D.; Dananberg, J.; Van Deursen, J.M. Senescent cells: An emerging target for diseases of ageing. Nat. Rev. Drug Discov. 2017, 16, 718–735. [Google Scholar] [CrossRef]
- Wallace, C.D.; Fan, W. Energetics, epigenetics, mitochondrial genetics. Mitochondrion 2010, 10, 12–31. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.C.; Fan, W.; Procaccio, V. Mitochondrial Energetics and Therapeutics. Annu. Rev. Pathol. Mech. Dis. 2010, 5, 297–348. [Google Scholar] [CrossRef]
- Wallace, D.C. A mitochondrial bioenergetic etiology of disease. J. Clin. Investig. 2013, 123, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- Bouzier-Sore, A.-K.; Voisin, P.; Bouchaud, V.; Bezancon, E.; Franconi, J.-M.; Pellerin, L. Competition between glucose and lactate as oxidative energy substrates in both neurons and astrocytes: A comparative NMR study. Eur. J. Neurosci. 2006, 24, 1687–1694. [Google Scholar] [CrossRef] [PubMed]
- Khatri, N.; Man, H.-Y. Synaptic Activity and Bioenergy Homeostasis: Implications in Brain Trauma and Neurodegenerative Diseases. Front. Neurol. 2013, 4. [Google Scholar] [CrossRef]
- Kim, H.A.; Miller, A.A.; Drummond, G.R.; Thrift, A.G.; Arumugam, T.; Phan, T.G.; Srikanth, V.K.; Sobey, C.G. Vascular cognitive impairment and Alzheimer’s disease: Role of cerebral hypoperfusion and oxidative stress. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2012, 385, 953–959. [Google Scholar] [CrossRef]
- Mosconi, L.; Sorbi, S.; De Leon, M.J.; Li, Y.; Nacmias, B.; Myoung, P.S.; Tsui, W.; Ginestroni, A.; Bessi, V.; Fayyazz, M.; et al. Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer’s disease. J. Nucl. Med. 2006, 47, 1778–1786. [Google Scholar]
- Ossenkoppele, R.; van der Flier, W.M.; Zwan, M.D.; Adriaanse, S.F.; Boellaard, R.; Windhorst, A.D.; Barkhof, F.; Lammertsma, A.A.; Scheltens, P.; van Berckel, B.N. Differential effect of APOE genotype on amyloid load and glucose metabolism in AD dementia. Neurology 2012, 80, 359–365. [Google Scholar] [CrossRef]
- Reiman, E.M.; Caselli, R.J.; Yun, L.S.; Chen, K.; Bandy, D.; Minoshima, S.; Thibodeau, S.N.; Osborne, D. Preclinical evidence of Alzheimer’s disease in persons homozygous for the epsilon 4 allele for apolipoprotein E. N. Engl. J. Med. 1996, 334, 752–758. [Google Scholar] [CrossRef]
- Small, G.W.; Mazziotta, J.C.; Collins, M.T.; Baxter, L.R.; Phelps, M.E.; Mandelkern, M.A.; Kaplan, A.; La Rue, A.; Adamson, C.F.; Chang, L.; et al. Apolipoprotein E type 4 allele and cerebral glucose metabolism in relatives at risk for familial Alzheimer disease. JAMA 1995, 273, 942–947. [Google Scholar] [CrossRef] [PubMed]
- Chiaravalloti, A.; Martorana, A.; Koch, G.; Toniolo, S.; Di Biagio, D.; Di Pietro, B.; Schillaci, O. Functional correlates of t-Tau, p-Tau and Aβ1–42 amyloid cerebrospinal fluid levels in Alzheimer’s disease: A 18F-FDG PET/CT study. Nucl. Med. Commun. 2015, 36, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Friedland, R.P.; Budinger, T.F.; Ganz, E.; Yano, Y.; Mathis, C.A.; Koss, B.; Ober, B.A.; Huesman, R.H.; Derenzo, S.E. Regional cerebral metabolic alterations in dementia of the Alzheimer type: Positron emission tomography with [18F]fluorodeoxyglucose. J. Comput. Assist. Tomogr. 1983, 7, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Minoshima, S.; Giordani, B.; Berent, S.; Frey, K.A.; Foster, N.L.; Kuhl, D.E. Metabolic reduction in the posterior cingulate cortex in very early Alzheimer’s disease. Ann. Neurol. 1997, 42, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Giulivi, C.; Napoli, E.; Tassone, F.; Halmai, J.; Hagerman, R. Plasma metabolic profile delineates roles for neurodegeneration, pro-inflammatory damage and mitochondrial dysfunction in the FMR1 premutation. Biochem. J. 2016, 473, 3871–3888. [Google Scholar] [CrossRef]
- Giulivi, C.; Napoli, E.; Tassone, F.; Halmai, J.; Hagerman, R. Plasma Biomarkers for Monitoring Brain Pathophysiology in FMR1 Premutation Carriers. Front. Mol. Neurosci. 2016, 9, 71. [Google Scholar] [CrossRef]
- Napoli, E.; Ross-Inta, C.; Song, G.; Wong, S.; Hagerman, R.; Gane, L.W.; Smilowitz, J.T.; Tassone, F.; Giulivi, C. Premutation in the Fragile X Mental Retardation 1 (FMR1) Gene Affects Maternal Zn-milk and Perinatal Brain Bioenergetics and Scaffolding. Front. Neurosci. 2016, 10, 159. [Google Scholar] [CrossRef]
- Napoli, E.; Ross-Inta, C.; Wong, S.; Omanska-Klusek, A.; Barrow, C.; Iwahashi, C.; Garcia-Arocena, D.; Sakaguchi, D.; Berry-Kravis, E.; Hagerman, R.; et al. Altered zinc transport disrupts mitochondrial protein processing/import in fragile X-associated tremor/ataxia syndrome. Hum. Mol. Genet. 2011, 20, 3079–3092. [Google Scholar] [CrossRef]
- Napoli, E.; Song, G.; Schneider, A.; Hagerman, R.; Eldeeb, M.A.A.A.; Azarang, A.; Tassone, F.; Giulivi, C. Warburg effect linked to cognitive—Executive deficits in FMR1 premutation. FASEB J. 2016, 30, 3334–3351. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Song, G.; Wong, S.; Hagerman, R.; Giulivi, C. Altered Bioenergetics in Primary Dermal Fibroblasts from Adult Carriers of the FMR1 Premutation Before the Onset of the Neurodegenerative Disease Fragile X-Associated Tremor/Ataxia Syndrome. Cerebellum 2016, 15, 552–564. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Wong, S.; Hung, C.; Ross-Inta, C.; Bomdica, P.; Giulivi, C. Defective mitochondrial disulfide relay system, altered mitochondrial morphology and function in Huntington’s disease. Hum. Mol. Genet. 2013, 22, 989–1004. [Google Scholar] [CrossRef] [PubMed]
- Armand-Ugon, M.; Ansoleaga, B.; Berjaoui, S.; Ferrer, I. Reduced mitochondrial activity is early and steady in the entorhinal cortex but it is mainly unmodified in the frontal cortex in Alzheimer’s disease. Curr. Alzheimer Res. 2017, 14, 1327–1334. [Google Scholar] [CrossRef] [PubMed]
- Ishii, K.; Sasaki, M.; Kitagaki, H.; Yamaji, S.; Sakamoto, S.; Matsuda, K.; Mori, E. Reduction of cerebellar glucose metabolism in advanced Alzheimer’s disease. J. Nucl. Med. 1997, 38, 925–928. [Google Scholar]
- Bubber, P.; Haroutunian, V.; Fisch, G.; Blass, J.P.; Gibson, G.E. Mitochondrial abnormalities in Alzheimer brain: Mechanistic implications. Ann. Neurol. 2005, 57, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, F.; Iqbal, K.; Grundke-Iqbal, I.; Gong, C.-X. Decreased glucose transporters correlate to abnormal hyperphosphorylation of tau in Alzheimer disease. FEBS Lett. 2008, 582, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Trimmer, P.A.; Borland, M.K. Differentiated Alzheimer’s disease transmitochondrial cybrid cell lines exhibit reduced organelle movement. Antioxid. Redox Signal. 2005, 7, 1101–1109. [Google Scholar] [CrossRef]
- Ghosh, D.; LeVault, K.R.; Barnett, A.J.; Brewer, G.J. A reversible early oxidized redox state that precedes macromolecular ROS damage in aging nontransgenic and 3xTg-AD mouse neurons. J. Neurosci. 2012, 32, 5821–5832. [Google Scholar] [CrossRef]
- Kitazawa, M.; Cheng, D.; LaFerla, F.M. Chronic copper exposure exacerbates both amyloid and tau pathology and selectively dysregulates cdk5 in a mouse model of AD. J. Neurochem. 2009, 108, 1550–1560. [Google Scholar] [CrossRef]
- Smith, M.A.; Sayre, L.M.; Anderson, V.E.; Harris, P.L.; Beal, M.F.; Kowall, N.; Perry, G. Cytochemical Demonstration of Oxidative Damage in Alzheimer Disease by Immunochemical Enhancement of the Carbonyl Reaction with 2,4-Dinitrophenylhydrazine. J. Histochem. Cytochem. 1998, 46, 731–735. [Google Scholar] [CrossRef] [PubMed]
- Walls, K.C.; Coskun, P.; Gallegos-Perez, J.-L.; Zadourian, N.; Freude, K.; Rasool, S.; Blurton-Jones, M.; Green, K.N.; LaFerla, F.M. Swedish Alzheimer Mutation Induces Mitochondrial Dysfunction Mediated by HSP60 Mislocalization of Amyloid Precursor Protein (APP) and Beta-Amyloid. J. Biol. Chem. 2012, 287, 30317–30327. [Google Scholar] [CrossRef] [PubMed]
- Brownlow, M.L.; Joly-Amado, A.; Azam, S.; Elza, M.; Selenica, M.-L.; Pappas, C.; Small, B.; Engelman, R.; Gordon, M.N.; Morgan, D. Partial rescue of memory deficits induced by calorie restriction in a mouse model of tau deposition. Behav. Brain Res. 2014, 271, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Yang, T.; Ho, L.; Zhao, Z.; Wang, J.; Chen, L.; Zhao, W.; Thiyagarajan, M.; MacGrogan, D.; Rodgers, J.T.; et al. Neuronal SIRT1 Activation as a Novel Mechanism Underlying the Prevention of Alzheimer Disease Amyloid Neuropathology by Calorie Restriction. J. Biol. Chem. 2006, 281, 21745–21754. [Google Scholar] [CrossRef] [PubMed]
- Rühlmann, C.; Wölk, T.; Blümel, T.; Stahn, L.; Vollmar, B.; Kuhla, A. Long-term caloric restriction in ApoE-deficient mice results in neuroprotection via Fgf21-induced AMPK/mTOR pathway. Aging 2016, 8, 2777–2789. [Google Scholar] [CrossRef]
- Schafer, M.J.; Alldred, M.J.; Lee, S.H.; Calhoun, M.E.; Petkova, E.; Mathews, P.M.; Ginsberg, S.D. Reduction of beta-amyloid and gamma-secretase by calorie restriction in female Tg2576 mice. Neurobiol. Aging 2015, 36, 1293–1302. [Google Scholar] [CrossRef]
- Karuppagounder, S.S.; Pinto, J.T.; Xu, H.; Chen, H.L.; Beal, M.F.; Gibson, G.E. Dietary supplementation with resveratrol reduces plaque pathology in a transgenic model of Alzheimer’s disease. Neurochem. Int. 2009, 54, 111–118. [Google Scholar] [CrossRef]
- Lu, C.-H.; Cheng-Yang, H.; Li, C.-Y.; Hsieh, C.-Y.; Ou, H.-T. Lower risk of dementia with pioglitazone, compared with other second-line treatments, in metformin-based dual therapy: A population-based longitudinal study. Diabetologia 2017, 61, 562–573. [Google Scholar] [CrossRef]
- Liu, W.; Zhuo, P.; Li, L.; Jin, H.; Lin, B.; Zhang, Y.; Liang, S.; Wu, J.; Huang, J.; Wang, Z.; et al. Activation of brain glucose metabolism ameliorating cognitive impairment in APP/PS1 transgenic mice by electroacupuncture. Free Radic. Biol. Med. 2017, 112, 174–190. [Google Scholar] [CrossRef]
- Baur, J.; Pearson, K.J.; Price, N.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; López-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef]
- Harrison, D.E.; Strong, R.; Sharp, Z.D.; Nelson, J.F.; Astle, C.M.; Flurkey, K.; Nadon, N.L.; Wilkinson, J.E.; Frenkel, K.; Carter, C.S.; et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009, 460, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Mattison, J.A.; Colman, R.J.; Beasley, T.M.; Allison, D.B.; Kemnitz, J.W.; Roth, G.S.; Ingram, D.K.; Weindruch, R.; De Cabo, R.; Anderson, R.M. Caloric restriction improves health and survival of rhesus monkeys. Nat. Commun. 2017, 8, 14063. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Schneider, A.; Hagerman, R.; Song, G.; Wong, S.; Tassone, F.; Giulivi, C. Impact of FMR1 Premutation on Neurobehavior and Bioenergetics in Young Monozygotic Twins. Front. Genet. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Schneider, A.; Wang, J.Y.; Trivedi, A.; Carrillo, N.R.; Tassone, F.; Rogawski, M.; Hagerman, R.J.; Giulivi, C. Allopregnanolone Treatment Improves Plasma Metabolomic Profile Associated with GABA Metabolism in Fragile X-Associated Tremor/Ataxia Syndrome: A Pilot Study. Mol. Neurobiol. 2018, 56, 3702–3713. [Google Scholar] [CrossRef]
- Song, G.; Napoli, E.; Wong, S.; Hagerman, R.; Liu, S.; Tassone, F.; Giulivi, C. Altered Redox Mitochondrial Biology in the Neurodegenerative Disorder Fragile X-Tremor/Ataxia Syndrome: Use of Antioxidants in Precision Medicine. Mol. Med. 2016, 22, 548–559. [Google Scholar] [CrossRef]
- Kaplan, E.S.; Cao, Z.; Hulsizer, S.; Tassone, F.; Berman, R.F.; Hagerman, P.J.; Pessah, I.N. Early mitochondrial abnormalities in hippocampal neurons cultured fromFmr1pre-mutation mouse model. J. Neurochem. 2012, 123, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Loesch, D.Z.; Annesley, S.J.; Trost, N.; Bui, M.; Lay, S.T.; Storey, E.; De Piazza, S.W.; Sanislav, O.; Francione, L.M.; Hammersley, E.M.; et al. Novel Blood Biomarkers Are Associated with White Matter Lesions in Fragile X- Associated Tremor/Ataxia Syndrome. Neurodegener. Dis. 2016, 17, 22–30. [Google Scholar] [CrossRef]
- Robinson, B.H. [39] Use of fibroblast and lymphoblast cultures for detection of respiratory chain defects. Methods Enzymol. 1996, 264, 454–464. [Google Scholar] [CrossRef]
- Bernier, F.; Boneh, A.; Dennett, X.; Chow, C.; Cleary, M.; Thorburn, D. Diagnostic criteria for respiratory chain disorders in adults and children. Neurology 2002, 59, 1406–1411. [Google Scholar] [CrossRef]
- Jeon, J.P.; Shim, S.M.; Nam, H.Y.; Baik, S.Y.; Kim, J.W.; Han, B.G. Copy number increase of 1p36.33 and mitochondrial genome amplification in Epstein-Barr virus-transformed lymphoblastoid cell lines. Cancer Genet. Cytogenet. 2007, 173, 122–130. [Google Scholar] [CrossRef]
- Petronis, A.; Vincent, J.B.; Surh, L.C.; Ashizawa, T.; Kennedy, J.L. Polyglutamine-containing proteins in schizophrenia: An effect of lymphoblastoid cells? Mol. Psychiatry 2000, 5, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, G.; Zeuthen, J. Complex forms of mitochondrial DNA in human B cells transformed by Epstein-Barr virus (EBV). J. Cancer Res. Clin. Oncol. 1983, 105, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Cahir-McFarland, E.D.; Davidson, D.M.; Schauer, S.L.; Duong, J.; Kieff, E. NF-kappa B inhibition causes spontaneous apoptosis in Epstein-Barr virus-transformed lymphoblastoid cells. Proc. Natl. Acad. Sci. USA 2000, 97, 6055–6060. [Google Scholar] [CrossRef] [PubMed]
- Lázló, L.; Tuckwell, J.; Self, T.; Lowe, J.; Landon, M.; Smith, S.; Hawthorne, J.N.; Mayer, R.J. The latent membrane protein-1 in Epstein-Barr virus-transformed lymphoblastoid cells is found with ubiquitin-protein conjugates and heat-shock protein 70 in lysosomes oriented around the microtubule organizing centre. J. Pathol. 1991, 164, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Park, G.B.; Kim, Y.S.; Lee, H.-K.; Cho, D.-H.; Kim, D.; Hur, D.Y. CD80 (B7.1) and CD86 (B7.2) induce EBV-transformed B cell apoptosis through the Fas/FasL pathway. Int. J. Oncol. 2013, 43, 1531–1540. [Google Scholar] [CrossRef] [PubMed]
- Srimatkandada, P.; Loomis, R.; Carbone, R.; Srimatkandada, S.; Lacy, J. Combined proteasome and Bcl-2 inhibition stimulates apoptosis and inhibits growth in EBV-transformed lymphocytes: A potential therapeutic approach to EBV-associated lymphoproliferative diseases. Eur. J. Haematol. 2008, 80, 407–418. [Google Scholar] [CrossRef]
- Bourgeron, T.; Chrétien, D.; Rotig, A.; Munnich, A.; Rustin, P. Isolation and characterization of mitochondria from human B lymphoblastoid cell lines. Biochem. Biophys. Res. Commun. 1992, 186, 16–23. [Google Scholar] [CrossRef]
- Chakrabarty, S.; D’Souza, R.R.; Kabekkodu, S.P.; Gopinath, P.M.; Rossignol, R.; Satyamoorthy, K. Upregulation of TFAM and mitochondria copy number in human lymphoblastoid cells. Mitochondrion 2014, 15, 52–58. [Google Scholar] [CrossRef]
- Cohen, S.; Masyn, K.; Adams, J.; Hessl, D.; Rivera, S.; Tassone, F.; Brunberg, J.; DeCarli, C.; Zhang, L.; Cogswell, J.; et al. Molecular and imaging correlates of the fragile X-associated tremor/ataxia syndrome. Neurology 2006, 67, 1426–1431. [Google Scholar] [CrossRef]
- Bacalman, S.; Farzin, F.; Bourgeois, J.A.; Cogswell, J.; Goodlin-Jones, B.L.; Gane, L.W.; Grigsby, J.; Leehey, M.A.; Tassone, F.; Hagerman, R.J. Psychiatric phenotype of the fragile X-associated tremor/ataxia syndrome (FXTAS) in males: Newly described fronto-subcortical dementia. J. Clin. Psychiatry 2006, 67, 87–94. [Google Scholar] [CrossRef]
- Brunberg, J.A.; Jacquemont, S.; Hagerman, R.J.; Berry-Kravis, E.M.; Grigsby, J.; Leehey, M.A.; Tassone, F.; Brown, W.T.; Greco, C.M.; Hagerman, P.J. Fragile X Premutation Carriers: Characteristic MR Imaging Findings of Adult Male Patients with Progressive Cerebellar and Cognitive Dysfunction. Am. J. Neuroradiol. 2002, 23, 1757–1766. [Google Scholar]
- Hall, D.A.; Birch, R.C.; Anheim, M.; Jønch, A.E.; Pintado, E.; O’Keefe, J.; Trollor, J.N.; Stebbins, G.T.; Hagerman, R.J.; Fahn, S.; et al. Emerging topics in FXTAS. J. Neurodev. Disord. 2014, 6, 31. [Google Scholar] [CrossRef]
- Ross-Inta, C.; Omanska-Klusek, A.; Wong, S.; Barrow, C.; Garcia-Arocena, D.; Iwahashi, C.; Berry-Kravis, E.; Hagerman, R.J.; Hagerman, P.J.; Giulivi, C. Evidence of mitochondrial dysfunction in fragile X-associated tremor/ataxia syndrome. Biochem. J. 2010, 429, 545–552. [Google Scholar] [CrossRef]
- Wang, J.Y.; Hessl, D.; Hagerman, R.J.; Simon, T.J.; Tassone, F.; Ferrer, E.; Rivera, S.M. Abnormal trajectories in cerebellum and brainstem volumes in carriers of the fragile X premutation. Neurobiol. Aging 2017, 55, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Sabayan, B.; van der Grond, J.; Westendorp, R.G.; van Buchem, M.A.; de Craen, A.J. Accelerated progression of white matter hyperintensities and subsequent risk of mortality: A 12-year follow-up study. Neurobiol. Aging 2015, 36, 2130–2135. [Google Scholar] [CrossRef] [PubMed]
- Boyle, P.A.; Yu, L.; Fleischman, D.A.; Leurgans, S.; Yang, J.; Wilson, R.S.; Schneider, J.A.; Arvanitakis, Z.; Arfanakis, K.; Bennett, D.A. White matter hyperintensities, incident mild cognitive impairment, and cognitive decline in old age. Ann. Clin. Transl. Neurol. 2016, 3, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Moura, A.R.; Lee, S.; Habeck, C.; Razlighi, Q.; Stern, Y. The relationship between white matter hyperintensities and cognitive reference abilities across the life span. Neurobiol. Aging 2019, 83, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Tsapanou, A.; Habeck, C.; Gazes, Y.; Razlighi, Q.; Sakhardande, J.; Stern, Y.; Salthouse, T.A. Brain biomarkers and cognition across adulthood. Hum. Brain Mapp. 2019, 40, 3832–3842. [Google Scholar] [CrossRef]
- Yatawara, C.; Lee, D.; Ng, K.P.; Chander, R.; Ng, D.; Ji, F.; Shim, H.Y.; Hilal, S.; Venketasubramanian, N.; Chen, C.; et al. Mechanisms Linking White Matter Lesions, Tract Integrity, and Depression in Alzheimer Disease. Am. J. Geriatr. Psychiatry 2019, 27, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Adams, P.E.; Nguyen, D.; Brunberg, J.A.; Tassone, F.; Zhang, W.; Koldewyn, K.; Rivera, S.M.; Grigsby, J.; Zhang, L.; et al. Volumetric brain changes in females with fragile X-associated tremor/ataxia syndrome (FXTAS). Neurology 2007, 69, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Nargund, A.M.; Fiorese, C.J.; Pellegrino, M.W.; Deng, P.; Haynes, C.M. Mitochondrial and nuclear accumulation of the transcription factor ATFS-1 promotes OXPHOS recovery during the UPR(mt). Mol. Cell 2015, 58, 123–133. [Google Scholar] [CrossRef]
- Yoneda, T.; Benedetti, C.; Urano, F.; Clark, S.G.; Harding, H.; Ron, D. Compartment-specific perturbation of protein handling activates genes encoding mitochondrial chaperones. J. Cell Sci. 2004, 117, 4055–4066. [Google Scholar] [CrossRef] [PubMed]
- Robinson, B.H. Lacticacidemia. Biochemical, clinical, and genetic considerations. Adv. Hum. Genet. 1989, 18, 151–179. [Google Scholar]
- Robinson, B.H. Lacticacidemia. Biochim. Biophys. Acta 1993, 1182, 231–244. [Google Scholar] [CrossRef]
- Čapková, M.; Houštěk, J.; Hansíková, H.; Hainer, V.; Kunešová, M.; Zeman, J. Activities of cytochrome c oxidase and citrate synthase in lymphocytes of obese and normal-weight subjects. Int. J. Obes. 2002, 26, 1110–1117. [Google Scholar] [CrossRef] [PubMed]
- Gropman, A.L. Neuroimaging in Mitochondrial Disorders. Neurotherapeutics 2012, 10, 273–285. [Google Scholar] [CrossRef]
- Finsterer, J.; Zarrouk-Mahjoub, S. Cerebral imaging in paediatric mitochondrial disorders. Neuroradiol. J. 2018, 31, 596–608. [Google Scholar] [CrossRef]
- Liu, B.; Li, Y.; Stackpole, E.E.; Novak, A.; Gao, Y.; Zhao, Y.; Zhao, X.; Richter, J.D. Regulatory discrimination of mRNAs by FMRP controls mouse adult neural stem cell differentiation. Proc. Natl. Acad. Sci. USA 2018, 115, E11397–E11405. [Google Scholar] [CrossRef] [PubMed]
- Weisz, E.D.; Towheed, A.; Monyak, R.E.; Toth, M.S.; Wallace, D.C.; Jongens, T.A. Loss of Drosophila FMRP leads to alterations in energy metabolism and mitochondrial function. Hum. Mol. Genet. 2017, 27, 95–106. [Google Scholar] [CrossRef]
- Napoli, E.; McLennan, Y.A.; Schneider, A.; Tassone, F.; Hagerman, R.J.; Giulivi, C. Characterization of the Metabolic, Clinical and Neuropsychological Phenotype of Female Carriers of the Premutation in the X-Linked FMR1 Gene. Front. Mol. Biosci. 2020, 7, 578640. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Flores, A.; Mansuri, Y.; Hagerman, R.J.; Giulivi, C. Sulforaphane improves mitochondrial metabolism in fibroblasts from patients with fragile X-associated tremor and ataxia syndrome. Neurobiol. Dis. 2021, 157, 105427. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Tassone, F.; Wong, S.; Angkustsiri, K.; Simon, T.J.; Song, G.; Giulivi, C. Mitochondrial Citrate Transporter-dependent Metabolic Signature in the 22q11.2 Deletion Syndrome. J. Biol. Chem. 2015, 290, 23240–23253. [Google Scholar] [CrossRef]
- Napoli, E.; Wong, S.; Giulivi, C. Evidence of reactive oxygen species-mediated damage to mitochondrial DNA in children with typical autism. Mol. Autism 2013, 4, 1–8. [Google Scholar] [CrossRef]
- Napoli, E.; Hung, C.; Wong, S.; Giulivi, C. Toxicity of the Flame-Retardant BDE-49 on Brain Mitochondria and Neuronal Progenitor Striatal Cells Enhanced by a PTEN-Deficient Background. Toxicol. Sci. 2013, 132, 196–210. [Google Scholar] [CrossRef]
- Giulivi, C.; Napoli, E.; Schwartzer, J.; Careaga, M.; Ashwood, P. Gestational Exposure to a Viral Mimetic Poly(I:C) Results in Long-Lasting Changes in Mitochondrial Function by Leucocytes in the Adult Offspring. Mediat. Inflamm. 2013, 2013, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Ross-Inta, C.; Wong, S.; Hung, C.; Fujisawa, Y.; Sakaguchi, D.; Angelastro, J.; Omanska-Klusek, A.; Schoenfeld, R.; Giulivi, C. Mitochondrial Dysfunction in Pten Haplo-Insufficient Mice with Social Deficits and Repetitive Behavior: Interplay between Pten and p. PLoS ONE 2012, 7, e42504. [Google Scholar] [CrossRef]
- Giulivi, C.; Zhang, Y.-F.; Omanska-Klusek, A.; Ross-Inta, C.; Wong, S.; Hertz-Picciotto, I.; Tassone, F.; Pessah, I.N. Mitochondrial Dysfunction in Autism. JAMA 2010, 304, 2389–2396. [Google Scholar] [CrossRef]
- Nolin, S.; Napoli, E.; Flores, A.; Hagerman, R.; Giulivi, C. Deficits in Prenatal Serine Biosynthesis Underlie the Mitochondrial Dysfunction Associated with the Autism-Linked FMR1 Gene. Int. J. Mol. Sci. 2021, 22, 5886. [Google Scholar] [CrossRef]
- Tustison, N.J.; Avants, B.B.; Cook, P.A.; Zheng, Y.; Egan, A.; Yushkevich, P.A.; Gee, J.C. N4ITK: Improved N3 Bias Correction. IEEE Trans. Med. Imaging 2010, 29, 1310–1320. [Google Scholar] [CrossRef]
- Smith, S.; Zhanga, Y.; Jenkinson, M.; Chenab, J.; Matthews, P.M.; Federico, A.; de Stefano, N. Accurate, Robust, and Automated Longitudinal and Cross-Sectional Brain Change Analysis. NeuroImage 2002, 17, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, P.; Gaser, C.; Arsic, M.; Buck, D.; Förschler, A.; Berthele, A.; Hoshi, M.; Ilg, R.; Schmid, V.J.; Zimmer, C.; et al. An automated tool for detection of FLAIR-hyperintense white-matter lesions in Multiple Sclerosis. NeuroImage 2012, 59, 3774–3783. [Google Scholar] [CrossRef]
- Yushkevich, P.A.; Piven, J.; Hazlett, H.C.; Smith, R.G.; Ho, S.; Gee, J.C.; Gerig, G. User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. NeuroImage 2006, 31, 1116–1128. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]


| Measures | N | Age | PFX− vs. Controls | PFX+ vs. Controls | PFX+ vs. PFX− | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β (SD) | P | FDR | β ** (SD) | P | FDR | β ** (SD) | P | FDR | β ** (SD) | P | FDR | ||
| MRI | |||||||||||||
| WMHV (log) | 96 | 0.050 (0.011) | <0.001 | <0.001 | 0.005 (0.45) | 0.99 | 0.99 | 1.54 (0.52) | 0.004 | 0.007 | 1.53 (0.36) | <0.001 | <0.001 |
| BV (ml) | 96 | −3.18 (0.62) | <0.001 | <0.001 | −52.2 (24.4) | 0.035 | 0.046 | −86.8 (28.6) | 0.003 | 0.007 | −34.7 (19.6) | 0.08 | 0.09 |
| Mitochondria | |||||||||||||
| CS * | 103 | −0.026 (0.006) | <0.001 | <0.001 | −0.46 (0.24) | 0.06 | 0.18 | −0.30 (0.23) | 0.20 | 0.39 | 0.16 (0.22) | 0.45 | 0.61 |
| NADH-dep. ATP prod. * | 102 | −0.021 (0.006) | 0.001 | 0.008 | −0.47 (0.23) | 0.045 | 0.16 | −0.25 (0.22) | 0.26 | 0.46 | 0.22 (0.21) | 0.30 | 0.47 |
| FADH2-dep. ATP prod. * | 103 | −0.027 (0.006) | <0.001 | 0.001 | −0.57 (0.24) | 0.020 | 0.10 | −0.55 (0.23) | 0.020 | 0.10 | 0.03 (0.22) | 0.91 | 0.94 |
| GP * | 97 | −0.032 (0.007) | <0.001 | <0.001 | −0.62 (0.28) | 0.029 | 0.12 | 0.012 (0.27) | 0.97 | 0.97 | 0.64 (0.26) | 0.016 | 0.10 |
| CCO * | 102 | −0.035 (0.008) | <0.001 | <0.001 | −0.22 (0.21) | 0.31 | 0.47 | −0.44 (0.22) | 0.051 | 0.17 | −0.22 (0.21) | 0.20 | 0.39 |
| Basal * | 95 | −0.031 (0.008) | <0.001 | <0.001 | −0.52 (0.30) | 0.08 | 0.23 | −0.17 (0.29) | 0.55 | 0.68 | 0.35 (0.27) | 0.19 | 0.39 |
| RCRu * | 95 | −0.003 (0.005) | 0.51 | 0.65 | −0.27 (0.18) | 0.14 | 0.35 | −0.38 (0.18) | 0.03 | 0.13 | −0.11 (0.16) | 0.49 | 0.64 |
| SRC * | 95 | 0 (0.004) | 0.92 | 0.94 | 0.03 (0.16) | 0.85 | 0.93 | −0.14 (0.15) | 0.35 | 0.51 | −0.17 (0.14) | 0.22 | 0.41 |
| PL/ROS * | 95 | 0.001 (0.005) | 0.88 | 0.94 | 0.27 (0.20) | 0.19 | 0.39 | 0.31 (0.20) | 0.12 | 0.33 | 0.04 (0.18) | 0.83 | 0.93 |
| IRC | 95 | 0.001 (0.003) | 0.69 | 0.79 | −0.09 (0.10) | 0.35 | 0.51 | −0.13 (0.10) | 0.16 | 0.38 | −0.04 (0.09) | 0.64 | 0.75 |
| RCR | 95 | −0.006 (0.007) | 0.38 | 0.53 | −0.30 (0.29) | 0.30 | 0.47 | −0.43 (0.28) | 0.13 | 0.33 | −0.13 (0.26) | 0.62 | 0.75 |
| Mitochondrial Outcomes | N | Semi-Partial r | P | FDR | N | Semi-Partial r | P | FDR |
|---|---|---|---|---|---|---|---|---|
| With log white matter hyperintensity volume | With whole brain volume | |||||||
| Citrate synthase activity * | 87 | −0.227 | 0.036 | 0.13 | 87 | 0.249 | 0.021 | 0.08 |
| NADH-linked ATP production * | 86 | −0.172 | 0.115 | 0.25 | 86 | 0.158 | 0.149 | 0.33 |
| FADH2-linked ATP production * | 87 | −0.264 | 0.014 | 0.08 | 87 | 0.246 | 0.023 | 0.08 |
| Glycerophosphate-linked ATP production * | 79 | −0.091 | 0.427 | 0.67 | 79 | 0.064 | 0.576 | 0.63 |
| Cytochrome oxidase activity * | 86 | −0.176 | 0.107 | 0.25 | 86 | 0.323 | 0.003 | 0.03 |
| Glucose-Gln-fueled ATP production * | 81 | −0.330 | 0.003 | 0.03 | 81 | 0.213 | 0.058 | 0.16 |
| RCRu * | 81 | −0.023 | 0.842 | 0.84 | 81 | 0.098 | 0.385 | 0.61 |
| SRC * | 81 | −0.105 | 0.354 | 0.65 | 81 | 0.020 | 0.862 | 0.86 |
| PL/ROS * | 81 | −0.040 | 0.727 | 0.84 | 81 | −0.070 | 0.540 | 0.63 |
| IRC | 81 | 0.026 | 0.819 | 0.84 | 81 | 0.082 | 0.470 | 0.63 |
| RCR | 81 | −0.060 | 0.599 | 0.82 | 81 | 0.135 | 0.231 | 0.42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.Y.; Napoli, E.; Kim, K.; McLennan, Y.A.; Hagerman, R.J.; Giulivi, C. Brain Atrophy and White Matter Damage Linked to Peripheral Bioenergetic Deficits in the Neurodegenerative Disease FXTAS. Int. J. Mol. Sci. 2021, 22, 9171. https://doi.org/10.3390/ijms22179171
Wang JY, Napoli E, Kim K, McLennan YA, Hagerman RJ, Giulivi C. Brain Atrophy and White Matter Damage Linked to Peripheral Bioenergetic Deficits in the Neurodegenerative Disease FXTAS. International Journal of Molecular Sciences. 2021; 22(17):9171. https://doi.org/10.3390/ijms22179171
Chicago/Turabian StyleWang, Jun Yi, Eleonora Napoli, Kyoungmi Kim, Yingratana A. McLennan, Randi J. Hagerman, and Cecilia Giulivi. 2021. "Brain Atrophy and White Matter Damage Linked to Peripheral Bioenergetic Deficits in the Neurodegenerative Disease FXTAS" International Journal of Molecular Sciences 22, no. 17: 9171. https://doi.org/10.3390/ijms22179171







