Netrin-1 as a Multitarget Barrier Stabilizer in the Peripheral Nerve after Injury
Abstract
:1. Introduction
2. Results
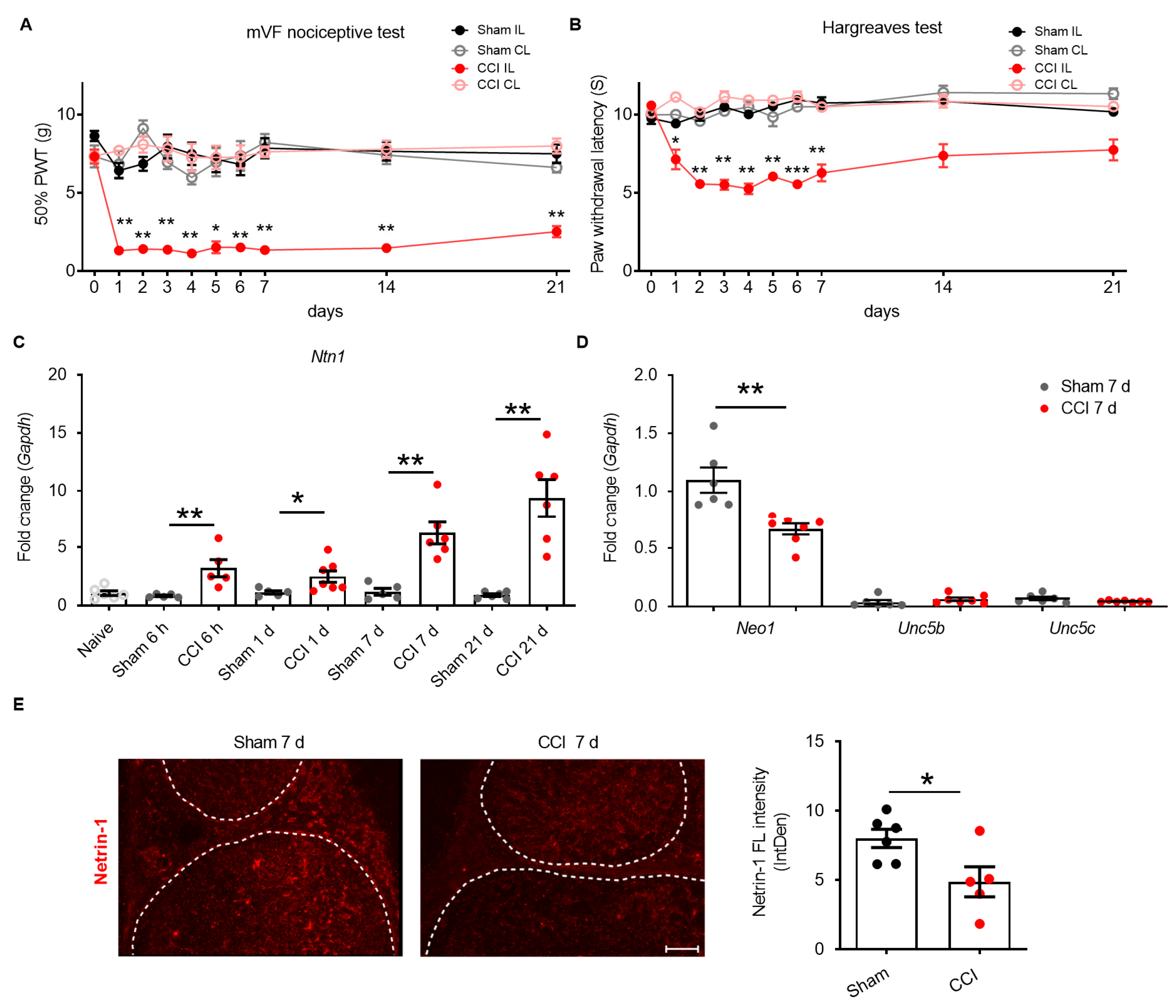
2.1. Netrin-1 Dysregulation in the Sciatic Nerve after CCI
2.2. Exogenous Recombinant Netrin-1 Treatment Rescues Neuropathic Pain via Neo1
2.3. Netrin-1 as A Multitarget Barrier Stabilizer in the Sciatic Nerve
3. Discussion
4. Materials and Methods
4.1. Chronic Constriction Injury (CCI) and Nociceptive Tests
4.2. Nociceptive Behavioral Assays
4.2.1. Thermal Nociceptive Behavioral Responses
4.2.2. Static Mechanical Hypersensitivity
4.3. Drugs and siRNA Treatment
4.4. Nerve Permeability
4.4.1. Permeability of the BNB (Perineurium)
4.4.2. Permeability of the Myelin Barrier
4.5. RNA Extraction and Quantitative PCR
4.6. Western Blotting
4.7. Immunofluorescence
4.8. Fluorescence Quantification
4.9. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paradisi, A.; Mehlen, P. Netrin-1, a missing link between chronic inflammation and tumor progression. Cell Cycle 2010, 9, 1253–1262. [Google Scholar] [CrossRef] [Green Version]
- Ko, S.Y.; Dass, C.R.; Nurgali, K. Netrin-1 in the developing enteric nervous system and colorectal cancer. Trends Mol. Med. 2012, 18, 544–554. [Google Scholar] [CrossRef]
- Podjaski, C.; Alvarez, J.I.; Bourbonniere, L.; Larouche, S.; Terouz, S.; Bin, J.M.; Lecuyer, M.A.; Saint-Laurent, O.; Larochelle, C.; Darlington, P.J.; et al. Netrin 1 regulates blood-brain barrier function and neuroinflammation. Brain 2015, 138, 1598–1612. [Google Scholar] [CrossRef] [Green Version]
- Miloudi, K.; Binet, F.; Wilson, A.; Cerani, A.; Oubaha, M.; Menard, C.; Henriques, S.; Mawambo, G.; Dejda, A.; Nguyen, P.T.; et al. Truncated netrin-1 contributes to pathological vascular permeability in diabetic retinopathy. J. Clin. Investig. 2016, 126, 3006–3022. [Google Scholar] [CrossRef] [Green Version]
- Peltonen, S.; Alanne, M.; Peltonen, J. Barriers of the peripheral nerve. Tissue Barriers 2013, 1, e24956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinhold, A.K.; Rittner, H.L. Barrier function in the peripheral and central nervous system—A review. Pflugers Arch. 2017, 469, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Reinhold, A.K.; Rittner, H.L. Characteristics of the nerve barrier and the blood dorsal root ganglion barrier in health and disease. Exp. Neurol. 2020, 327, 113244. [Google Scholar] [CrossRef] [PubMed]
- Alanne, M.H.; Pummi, K.; Heape, A.M.; Grenman, R.; Peltonen, J.; Peltonen, S. Tight junction proteins in human Schwann cell autotypic junctions. J. Histochem. Cytochem. 2009, 57, 523–529. [Google Scholar] [CrossRef]
- Lim, T.K.Y.; Shi, X.Q.; Martin, H.C.; Huang, H.; Luheshi, G.; Rivest, S.; Zhang, J. Blood-nerve barrier dysfunction contributes to the generation of neuropathic pain and allows targeting of injured nerves for pain relief. Pain 2014, 155, 954–967. [Google Scholar] [CrossRef]
- Moreau, N.; Dieb, W.; Mauborgne, A.; Bourgoin, S.; Villanueva, L.; Pohl, M.; Boucher, Y. Hedgehog Pathway-Mediated Vascular Alterations Following Trigeminal Nerve Injury. J. Dent. Res. 2017, 96, 450–457. [Google Scholar] [CrossRef]
- Moreau, N.; Mauborgne, A.; Bourgoin, S.; Couraud, P.O.; Romero, I.A.; Weksler, B.B.; Villanueva, L.; Pohl, M.; Boucher, Y. Early alterations of Hedgehog signaling pathway in vascular endothelial cells after peripheral nerve injury elicit blood-nerve barrier disruption, nerve inflammation, and neuropathic pain development. Pain 2016, 157, 827–839. [Google Scholar] [CrossRef]
- Reinhold, A.K.; Schwabe, J.; Lux, T.J.; Salvador, E.; Rittner, H.L. Quantitative and Microstructural Changes of the Blood-Nerve Barrier in Peripheral Neuropathy. Front. Neurosci. 2018, 12, 936. [Google Scholar] [CrossRef]
- Wang, X.; Miao, Y.; Ni, J.; Wang, Y.; Qian, T.; Yu, J.; Liu, Q.; Wang, P.; Yi, S. Peripheral Nerve Injury Induces Dynamic Changes of Tight Junction Components. Front. Physiol. 2018, 9, 1519. [Google Scholar] [CrossRef]
- Richner, M.; Ferreira, N.; Dudele, A.; Jensen, T.S.; Vaegter, C.B.; Goncalves, N.P. Functional and Structural Changes of the Blood-Nerve-Barrier in Diabetic Neuropathy. Front. Neurosci. 2018, 12, 1038. [Google Scholar] [CrossRef] [Green Version]
- Shubayev, V.I.; Strongin, A.Y.; Yaksh, T.L. Role of myelin auto-antigens in pain: A female connection. Neural Regen. Res. 2016, 11, 890–891. [Google Scholar] [CrossRef] [PubMed]
- Jessen, K.R.; Mirsky, R.; Lloyd, A.C. Schwann Cells: Development and Role in Nerve Repair. Cold Spring Harb. Perspect. Biol. 2015, 7, a020487. [Google Scholar] [CrossRef]
- Kim, H.A.; Mindos, T.; Parkinson, D.B. Plastic fantastic: Schwann cells and repair of the peripheral nervous system. Stem Cells Transl. Med. 2013, 2, 553–557. [Google Scholar] [CrossRef] [PubMed]
- Dun, X.P.; Parkinson, D.B. Role of Netrin-1 Signaling in Nerve Regeneration. Int. J. Mol. Sci. 2017, 18, 491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.K.; Seo, I.A.; Seo, E.; Seo, S.Y.; Lee, H.J.; Park, H.T. Netrin-1 induces proliferation of Schwann cells through Unc5b receptor. Biochem. Biophys. Res. Commun. 2007, 362, 1057–1062. [Google Scholar] [CrossRef]
- Lv, J.; Sun, X.; Ma, J.; Ma, X.; Zhang, Y.; Li, F.; Li, Y.; Zhao, Z. Netrin-1 induces the migration of Schwann cells via p38 MAPK and PI3K-Akt signaling pathway mediated by the UNC5B receptor. Biochem. Biophys. Res. Commun. 2015, 464, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Ke, X.; Li, Q.; Xu, L.; Zhang, Y.; Li, D.; Ma, J.; Mao, X. Netrin-1 overexpression in bone marrow mesenchymal stem cells promotes functional recovery in a rat model of peripheral nerve injury. J. Biomed. Res. 2015, 29, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Reinhold, A.K.; Yang, S.; Chen, J.T.; Hu, L.; Sauer, R.S.; Krug, S.M.; Mambretti, E.M.; Fromm, M.; Brack, A.; Rittner, H.L. Tissue plasminogen activator and neuropathy open the blood-nerve barrier with upregulation of microRNA-155-5p in male rats. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 1160–1169. [Google Scholar] [CrossRef]
- Hackel, D.; Krug, S.M.; Sauer, R.S.; Mousa, S.A.; Bocker, A.; Pflucke, D.; Wrede, E.J.; Kistner, K.; Hoffmann, T.; Niedermirtl, B.; et al. Transient opening of the perineurial barrier for analgesic drug delivery. Proc. Natl. Acad. Sci. USA 2012, 109, E2018–E2027. [Google Scholar] [CrossRef] [Green Version]
- Sauer, R.S.; Kirchner, J.; Yang, S.; Hu, L.; Leinders, M.; Sommer, C.; Brack, A.; Rittner, H.L. Blood-spinal cord barrier breakdown and pericyte deficiency in peripheral neuropathy. Ann. N. Y. Acad. Sci. 2017, 1405, 71–88. [Google Scholar] [CrossRef]
- Nitta, T.; Hata, M.; Gotoh, S.; Seo, Y.; Sasaki, H.; Hashimoto, N.; Furuse, M.; Tsukita, S. Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J. Cell Biol. 2003, 161, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Ben-Kraiem, A.; Sauer, R.S.; Norwig, C.; Popp, M.; Bettenhausen, A.L.; Atalla, M.S.; Brack, A.; Blum, R.; Doppler, K.; Rittner, H.L. Selective blood-nerve barrier leakiness with claudin-1 and vessel-associated macrophage loss in diabetic polyneuropathy. J. Mol. Med. 2021, 99, 1237–1250. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, T.; Morita, K.; Takemoto, D.; Takeuchi, K.; Kitano, Y.; Miyakawa, T.; Nakayama, K.; Okamura, Y.; Sasaki, H.; Miyachi, Y.; et al. Tight junctions in Schwann cells of peripheral myelinated axons: A lesson from claudin-19-deficient mice. J. Cell Biol. 2005, 169, 527–538. [Google Scholar] [CrossRef]
- Guo, J.; Wang, L.; Zhang, Y.; Wu, J.; Arpag, S.; Hu, B.; Imhof, B.A.; Tian, X.; Carter, B.D.; Suter, U.; et al. Abnormal junctions and permeability of myelin in PMP22-deficient nerves. Ann. Neurol. 2014, 75, 255–265. [Google Scholar] [CrossRef] [Green Version]
- Colloca, L.; Ludman, T.; Bouhassira, D.; Baron, R.; Dickenson, A.H.; Yarnitsky, D.; Freeman, R.; Truini, A.; Attal, N.; Finnerup, N.B.; et al. Neuropathic pain. Nat. Rev. Dis. Primers 2017, 3, 17002. [Google Scholar] [CrossRef] [Green Version]
- Inoue, K.; Tsuda, M. Microglia in neuropathic pain: Cellular and molecular mechanisms and therapeutic potential. Nat. Rev. Neurosci. 2018, 19, 138–152. [Google Scholar] [CrossRef]
- Jolivalt, C.G.; Mizisin, L.M.; Nelson, A.; Cunha, J.M.; Ramos, K.M.; Bonke, D.; Calcutt, N.A. B vitamins alleviate indices of neuropathic pain in diabetic rats. Eur. J. Pharmacol. 2009, 612, 41–47. [Google Scholar] [CrossRef]
- Topp, K.S.; Tanner, K.D.; Levine, J.D. Damage to the cytoskeleton of large diameter sensory neurons and myelinated axons in vincristine-induced painful peripheral neuropathy in the rat. J. Comp. Neurol. 2000, 424, 563–576. [Google Scholar] [CrossRef]
- Yu, J.; Li, C.; Ding, Q.; Que, J.; Liu, K.; Wang, H.; Liao, S. Netrin-1 Ameliorates Blood-Brain Barrier Impairment Secondary to Ischemic Stroke via the Activation of PI3K Pathway. Front. Neurosci. 2017, 11, 700. [Google Scholar] [CrossRef] [Green Version]
- Yao, L.L.; Hu, J.X.; Li, Q.; Lee, D.; Ren, X.; Zhang, J.S.; Sun, D.; Zhang, H.S.; Wang, Y.G.; Mei, L.; et al. Astrocytic neogenin/netrin-1 pathway promotes blood vessel homeostasis and function in mouse cortex. J. Clin. Investig. 2020, 130, 6490–6509. [Google Scholar] [CrossRef]
- Xie, Z.; Enkhjargal, B.; Reis, C.; Huang, L.; Wan, W.; Tang, J.; Cheng, Y.; Zhang, J.H. Netrin-1 Preserves Blood-Brain Barrier Integrity Through Deleted in Colorectal Cancer/Focal Adhesion Kinase/RhoA Signaling Pathway Following Subarachnoid Hemorrhage in Rats. J. Am. Heart Assoc. 2017, 6, e005198. [Google Scholar] [CrossRef] [PubMed]
- Aherne, C.M.; Collins, C.B.; Masterson, J.C.; Tizzano, M.; Boyle, T.A.; Westrich, J.A.; Parnes, J.A.; Furuta, G.T.; Rivera-Nieves, J.; Eltzschig, H.K. Neuronal guidance molecule netrin-1 attenuates inflammatory cell trafficking during acute experimental colitis. Gut 2012, 61, 695–705. [Google Scholar] [CrossRef] [Green Version]
- Madison, R.D.; Zomorodi, A.; Robinson, G.A. Netrin-1 and peripheral nerve regeneration in the adult rat. Exp. Neurol. 2000, 161, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Webber, C.A.; Christie, K.J.; Cheng, C.; Martinez, J.A.; Singh, B.; Singh, V.; Thomas, D.; Zochodne, D.W. Schwann cells direct peripheral nerve regeneration through the Netrin-1 receptors, DCC and Unc5H2. Glia 2011, 59, 1503–1517. [Google Scholar] [CrossRef] [PubMed]
- Takizawa, Y.; Kishimoto, H.; Kitazato, T.; Tomita, M.; Hayashi, M. Changes in protein and mRNA expression levels of claudin family after mucosal lesion by intestinal ischemia/reperfusion. Int. J. Pharm. 2012, 426, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Kress, L.; Hofmann, L.; Klein, T.; Klug, K.; Saffer, N.; Spitzel, M.; Bar, F.; Sommer, C.; Karl, F.; Uceyler, N. Differential impact of keratinocytes and fibroblasts on nociceptor degeneration and sensitization in small fiber neuropathy. Pain 2021, 162, 1262–1272. [Google Scholar] [CrossRef]
- Fortelny, N.; Overall, C.M.; Pavlidis, P.; Freue, G.V.C. Can we predict protein from mRNA levels? Nature 2017, 547, E19–E20. [Google Scholar] [CrossRef]
- Vogel, C.; Marcotte, E.M. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat. Rev. Genet. 2012, 13, 227–232. [Google Scholar] [CrossRef]
- Remacle, A.G.; Hullugundi, S.K.; Dolkas, J.; Angert, M.; Chernov, A.V.; Strongin, A.Y.; Shubayev, V.I. Acute- and late-phase matrix metalloproteinase (MMP)-9 activity is comparable in female and male rats after peripheral nerve injury. J. Neuroinflamm. 2018, 15, 89. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Chen, Q.; Yi, S.; Liu, Q.; Zhang, R.; Wang, P.; Qian, T.; Li, S. The microRNAs let-7 and miR-9 down-regulate the axon-guidance genes Ntn1 and Dcc during peripheral nerve regeneration. J. Biol. Chem. 2019, 294, 3489–3500. [Google Scholar] [CrossRef] [Green Version]
- Rosenberger, P.; Schwab, J.M.; Mirakaj, V.; Masekowsky, E.; Mager, A.; Morote-Garcia, J.C.; Unertl, K.; Eltzschig, H.K. Hypoxia-inducible factor-dependent induction of netrin-1 dampens inflammation caused by hypoxia. Nat. Immunol. 2009, 10, 195–202. [Google Scholar] [CrossRef]
- Toque, H.A.; Fernandez-Flores, A.; Mohamed, R.; Caldwell, R.B.; Ramesh, G.; Caldwell, R.W. Netrin-1 is a novel regulator of vascular endothelial function in diabetes. PLoS ONE 2017, 12, e0186734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faguer, S.; Chauveau, D.; Cintas, P.; Tack, I.; Cointault, O.; Rostaing, L.; Vargas-Poussou, R.; Ribes, D. Renal, ocular, and neuromuscular involvements in patients with CLDN19 mutations. Clin. J. Am. Soc. Nephrol. 2011, 6, 355–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadj-Rabia, S.; Baala, L.; Vabres, P.; Hamel-Teillac, D.; Jacquemin, E.; Fabre, M.; Lyonnet, S.; De Prost, Y.; Munnich, A.; Hadchouel, M.; et al. Claudin-1 gene mutations in neonatal sclerosing cholangitis associated with ichthyosis: A tight junction disease. Gastroenterology 2004, 127, 1386–1390. [Google Scholar] [CrossRef] [PubMed]
- Schubert, A.L.; Held, M.; Sommer, C.; Uceyler, N. Reduced gene expression of netrin family members in skin and sural nerve specimens of patients with painful peripheral neuropathies. J. Neurol. 2019, 266, 2812–2820. [Google Scholar] [CrossRef] [PubMed]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 1994, 53, 55–63. [Google Scholar] [CrossRef]





| Name | Forward | Reverse |
|---|---|---|
| Gapdh | 5′-AGTCTACTGGCGTCTTCAC-3′ | 5′-TCATATTTCTCGTGGTTCAC-3′ |
| Cldn1 | 5′-GGGACAACATCGTGACTGCT-3′ | 5′-CCACTAATGTCGCCAGACCTG-3′ |
| Cldn5 | 5′-AAATTCTGGGTCTGGTGCTG-3′ | 5′-GCCGGTCAAGGTAACAAAGA-3′ |
| Cldn12 | 5′-AACTGGCCAAGTGTCTGGTC-3′ | 5′-AGACCCCCTGAGCTAGCAAT-3′ |
| Cldn19 | 5′-TGCTGAAGGACCCATCTG-3′ | 5′-TGTGCTTGCTGTGAGAACTG-3′ |
| Ntn1 | 5′-CAGGAAGGACTATGCTGTCCA-3′ | 5′-TACGACTTGTGCCCTGCTTG-3′ |
| Neo1 | 5′-TGTGATGGTGACCAAAGGCA-3′ | 5′-GGAGGCTGCCAGTTCACTATT-3′ |
| Unc5b | 5′-CGACCCTAAAAGCCGCCCC-3′ | 5′-GGGATCTTGTCGGCAGAGTCC-3′ |
| Unc5c | 5′-AGGCTGCTCCTGACTCAGATG-3′ | 5′-GGGTCTAGAATTGGAGAATTGG-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.T.-C.; Schmidt, L.; Schürger, C.; Hankir, M.K.; Krug, S.M.; Rittner, H.L. Netrin-1 as a Multitarget Barrier Stabilizer in the Peripheral Nerve after Injury. Int. J. Mol. Sci. 2021, 22, 10090. https://doi.org/10.3390/ijms221810090
Chen JT-C, Schmidt L, Schürger C, Hankir MK, Krug SM, Rittner HL. Netrin-1 as a Multitarget Barrier Stabilizer in the Peripheral Nerve after Injury. International Journal of Molecular Sciences. 2021; 22(18):10090. https://doi.org/10.3390/ijms221810090
Chicago/Turabian StyleChen, Jeremy Tsung-Chieh, Lea Schmidt, Christina Schürger, Mohammed K. Hankir, Susanne M. Krug, and Heike L. Rittner. 2021. "Netrin-1 as a Multitarget Barrier Stabilizer in the Peripheral Nerve after Injury" International Journal of Molecular Sciences 22, no. 18: 10090. https://doi.org/10.3390/ijms221810090








