Zone-Dependent Architecture and Biochemical Composition of Decellularized Porcine Nasal Cartilage Modulate the Activity of Adipose Tissue-Derived Stem Cells in Cartilage Regeneration
Abstract
:1. Introduction
2. Results
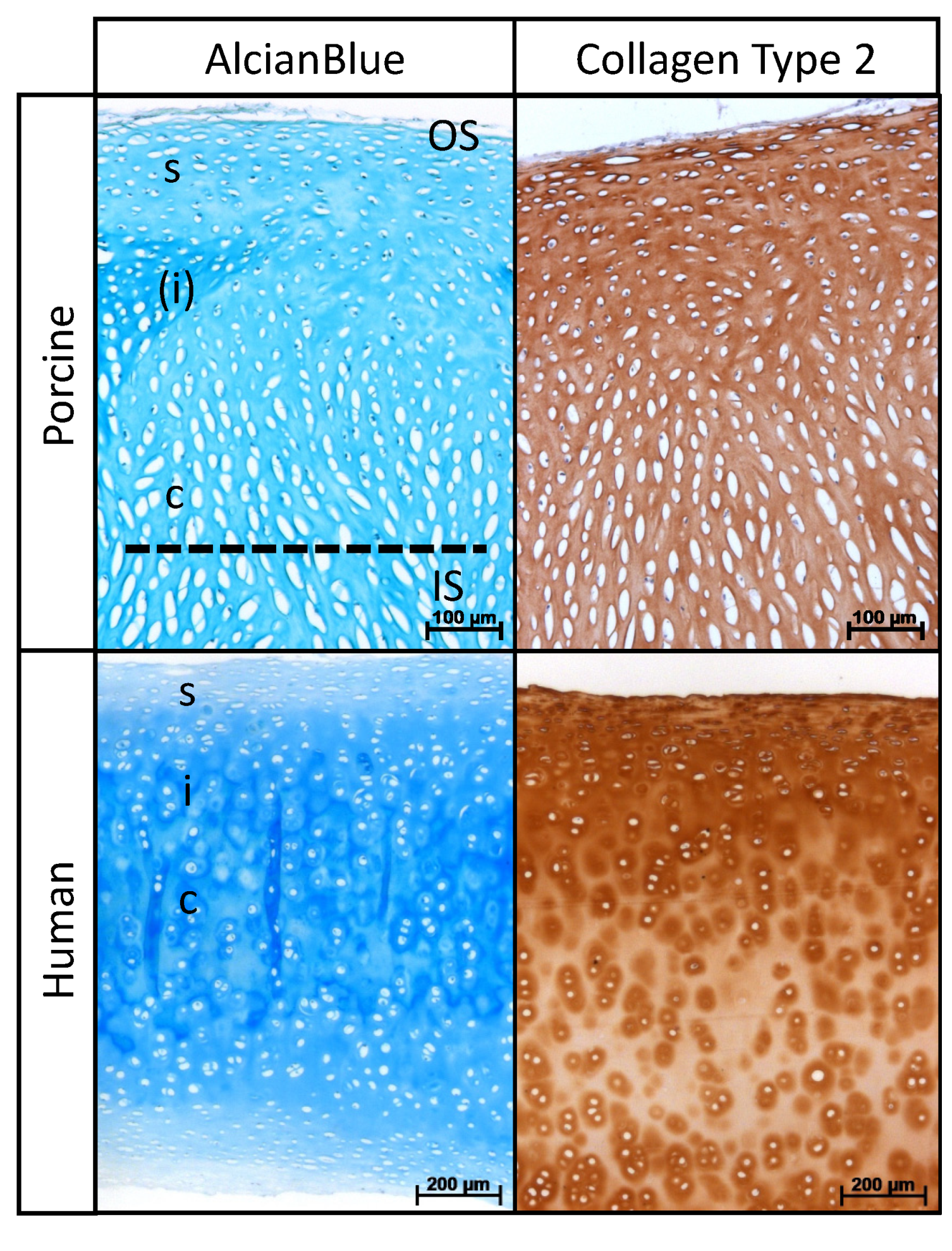
2.1. Histological Analysis of Native Nasal Cartilage
2.2. Evaluation of Seeding Success, Cell Distribution and Cell Vitality on DPNC Surfaces
2.3. ASC Metabolism and Proliferation
2.4. Biochemical Analysis of ASC Chondrogenesis
2.5. Histological Analysis and Cell Migration
2.6. Gene Expression Analysis
3. Discussion
4. Material and Methods
4.1. Donors
4.2. Cell Isolation and Culture
4.3. Manufacturing and Handling of DPNC
4.4. Histological and Immunohistochemical Analyses of Native porcine and Human Nasal Cartilage
4.5. Static Seeding on Different DPNC Zones
4.6. Evaluation of Cell Vitality and Cell Seeding Efficiency (CSE)
4.7. Influence of Matrix Zones on Metabolic Activity and Proliferation Capacity of ASCs
4.8. Biochemical Analysis of Chondrogenic Differentiation
4.9. Histological Analyses of DPNCs
4.10. QPCR
4.11. Statistical Evaluation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Mendez, B.M.; Thornton, J.F. Current basal and squamous cell skin cancer management. Plast. Reconstr. Surg. 2018, 142, 373E–387E. [Google Scholar] [CrossRef]
- Vogt, P.M.; Gohritz, A.; Haubitz, M.; Steiert, A. Reconstruction of Nasal Deformity in Wegener’s Granulomatosis: Contraindication or Benefit? Aesthet. Plast. Surg. 2011, 35, 156–161. [Google Scholar] [CrossRef]
- Prasad, B.K.; Mokamati, S. Archives of Otolaryngology and Rhinology Tertiary Nasal Syphilis: Rare But Still a Reality. Arch. Otolaryngol. Rhinol. 2016, 2, 13–15. [Google Scholar] [CrossRef] [Green Version]
- Rettinger, G. Risiken und Komplikationen der Rhinoplastik. Laryngo-Rhino-Otol. 2007, 86, 40–54. [Google Scholar] [CrossRef]
- Rohrich, R.J.; Ahmad, J. A Practical Approach to Rhinoplasty. Plast. Reconstr. Surg. 2016, 137, 725e–746e. [Google Scholar] [CrossRef]
- Ohara, K.; Nakamura, K.; Ohta, E. Chest wall deformities and thoracic scoliosis after costal cartilage graft harvesting. Plast. Reconstr. Surg. 1997, 99, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Lavernia, L.; Brown, W.E.; Wong, B.J.F.; Hu, J.C.; Athanasiou, K.A. Acta Biomaterialia Toward tissue-engineering of nasal cartilages. Acta Biomater. 2019, 88, 42–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langer, R.; Vacanti, J. Advances in tissue engineering. J. Pediatr. Surg. 2016, 51, 8–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langer, R.; Vacanti, J.P. Tissue Engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiggenhauser, P.S.; Schantz, J.T.; Rotter, N. Cartilage engineering in reconstructive surgery: Auricular, nasal and tracheal engineering from a surgical perspective. Regen. Med. 2017, 12, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, P.; Lombardi, F.; Siragusa, G.; Cifone, M.G.; Cinque, B.; Giuliani, M. Methods of isolation, characterization and expansion of human adipose-derived stem cells (ASCs): An overview. Int. J. Mol. Sci. 2018, 19, 1897. [Google Scholar] [CrossRef] [Green Version]
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International So. Cytotherapy 2013, 15, 641–648. [Google Scholar] [CrossRef] [Green Version]
- Buschmann, J.; Gao, S.; Härter, L.; Hemmi, S.; Welti, M.; Werner, C.M.L.; Calcagni, M.; Cinelli, P.; Wanner, G.A. Yield and proliferation rate of adipose-derived stromal cells as a function of age, body mass index and harvest site-increasing the yield by use of adherent and supernatant fractions? Cytotherapy 2013, 15, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
- Im, G.I.; Ko, J.Y.; Lee, J.H. Chondrogenesis of adipose stem cells in a porous polymer scaffold: Influence of the pore size. Cell Transplant. 2012, 21, 2397–2405. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A. The Adipose-derived Stem Cell: Looking Back and Looking Ahead. Mol. Biol. Cell 2010, 21, 1783–1787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiggenhauser, P.S.; Kuhlmann, C.; Blum, J.; Giunta, R.E.; Schenck, T. Influence of software parameters on measurements in automatized image-based analysis of fat tissue histology. Acta Histochem. 2020, 122, 151537. [Google Scholar] [CrossRef] [PubMed]
- Karkhaneh, A.; Naghizadeh, Z.; Shokrgozar, M.A.; Bonakdar, S.; Solouk, A.; Haghighipour, N. Effects of hydrostatic pressure on biosynthetic activity during chondrogenic differentiation of MSCs in hybrid scaffolds. Int. J. Artif. Organs 2014, 37, 142–148. [Google Scholar] [CrossRef]
- Zhu, H.; Gong, H.; Liu, Q.; Chhen, H.H. Three-dimensional Bioprinting for Cartilage Regeneration. Tissue Eng. Nanotheranostics 2017, 49–74. [Google Scholar] [CrossRef]
- Brizzi, M.F.; Tarone, G.; Defilippi, P. Extracellular matrix, integrins, and growth factors as tailors of the stem cell niche. Curr. Opin. Cell Biol. 2012, 24, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.L.; Wagers, A.J. No place like home: Anatomy and function of the stem cell niche. Nat. Rev. Mol. Cell Biol. 2008, 9, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Guneta, V.; Zhou, Z.; Tan, N.S.; Sugii, S.; Wong, M.T.C.; Choong, C. Recellularization of decellularized adipose tissue-derived stem cells: Role of the cell-secreted extracellular matrix in cellular differentiation. Biomater. Sci. 2018, 6, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Vinatier, C.; Mrugala, D.; Jorgensen, C.; Guicheux, J.; Noël, D. Cartilage engineering: A crucial combination of cells, biomaterials and biofactors. Trends Biotechnol. 2009, 27, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.; Koerber, L.; Elsaesser, A.F.; Goldberg-Bockhorn, E.; Seitz, A.M.; Dürselen, L.; Ignatius, A.; Walther, P.; Breiter, R.; Rotter, N. Decellularized Cartilage Matrix as a Novel Biomatrix for Cartilage Tissue-Engineering Applications. Tissue Eng. Part A 2012, 18, 2195–2209. [Google Scholar] [CrossRef]
- Schwarz, S.; Elsaesser, A.F.; Koerber, L.; Goldberg-Bockhorn, E.; Seitz, A.M.; Bermueller, C.; Dürselen, L.; Ignatius, A.; Breiter, R.; Rotter, N. Processed xenogenic cartilage as innovative biomatrix for cartilage tissue engineering: Effects on chondrocyte differentiation and function. J. Tissue Eng. Regen. Med. 2015, 9, E239–E251. [Google Scholar] [CrossRef]
- Elsaesser, A.F.; Bermueller, C.; Schwarz, S.; Koerber, L.; Breiter, R.; Rotter, N. In Vitro Cytotoxicity and In Vivo Effects of a Decellularized Xenogeneic Collagen Scaffold in Nasal Cartilage Repair. Tissue Eng. Part A 2014, 20, 1668–1678. [Google Scholar] [CrossRef] [PubMed]
- von Bomhard, A.; Elsaesser, A.; Riepl, R.; Pippich, K.; Faust, J.; Schwarz, S.; Koerber, L.; Breiter, R.; Rotter, N. Cartilage regeneration using decellularized cartilage matrix: Long-term comparison of subcutaneous and intranasal placement in a rabbit model. J. Cranio-Maxillofac. Surg. 2019, 47, 682–694. [Google Scholar] [CrossRef]
- Kuhlmann, C.; Schenck, T.L.; Tluczynski, K.; Aszodi, A.; Metzger, P.; Giunta, R.; Wiggenhauser, P.S. Experimental approach to nasal septal cartilage regeneration with adipose tissue-derived stem cells and decellularized porcine septal cartilage. Xenotransplantation 2020, 28, e12660. [Google Scholar] [CrossRef]
- Lu, P.; Weaver, V.M.; Werb, Z. The extracellular matrix: A dynamic niche in cancer progression. J. Cell Biol. 2012, 196, 395–406. [Google Scholar] [CrossRef]
- Egeblad, M.; Nakasone, E.S.; Werb, Z. Tumors as organs: Complex tissues that interface with the entire organism. Dev. Cell 2010, 18, 884–901. [Google Scholar] [CrossRef] [Green Version]
- Gattazzo, F.; Urciuolo, A.; Bonaldo, P. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochim. Biophys. Acta-Gen. Subj. 2014, 1840, 2506–2519. [Google Scholar] [CrossRef]
- Hynes, R.O. Extracellular matrix: Not just pretty fibrils Richard. Science 2009, 326, 1216–1219. [Google Scholar] [CrossRef] [Green Version]
- Mammoto, T.; Ingber, D.E. Mechanical control of tissue and organ development. Development 2010, 137, 1407–1420. [Google Scholar] [CrossRef] [Green Version]
- Folkman, J.; Moscona, A. Role of cell shape in growth control. Nature 1978, 273, 345–349. [Google Scholar] [CrossRef]
- Ingber, D. Integrins As mechanochemical transducers. Curr. Opin. Cell Biol. 1991, 3, 841–848. [Google Scholar] [CrossRef]
- Von der Mark, K.; Gauss, V.; Von der Mark, H.; Müller, P. Relationship between shape and type of collagen synthesized as chondrocytes lose their cartilage phenotype in culture. Nature 1977, 270, 572. [Google Scholar] [CrossRef]
- Schnabel, M.; Marlovits, S.; Eckhoff, G.; Fichtel, I.; Gotzen, L.; Vécsei, V.; Schlegel, J. Dedifferentiation-associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthr. Cartil. 2002, 10, 62–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caron, M.M.J.; Emans, P.J.; Coolsen, M.M.E.; Voss, L.; Surtel, D.A.M.; Cremers, A.; van Rhijn, L.W.; Welting, T.J.M. Redifferentiation of dedifferentiated human articular chondrocytes: Comparison of 2D and 3D cultures. Osteoarthr. Cartil. 2012, 20, 1170–1178. [Google Scholar] [CrossRef] [Green Version]
- Khoshgoftar, M.; Torzilli, P.A.; Maher, S.A. Influence of the pericellular and extracellular matrix structural properties on chondrocyte mechanics. J. Orthop. Res. 2018, 36, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Guilak, F.; Nims, R.J.; Dicks, A.; Wu, C.L.; Meulenbelt, I. Osteoarthritis as a disease of the cartilage pericellular matrix. Matrix Biol. 2018, 71–72, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Alexopoulos, L.G.; Setton, L.A.; Guilak, F. The biomechanical role of the chondrocyte pericellular matrix in articular cartilage. Acta Biomater. 2005, 1, 317–325. [Google Scholar] [CrossRef]
- Popko, M.; Bleys, R.L.A.W.; De Groot, J.W.; Huizing, E.H. Histological structure of the nasal cartilages and their perichondrial envelope. I. The septal and lobular cartilage. Rhinology 2007, 45, 148–152. [Google Scholar] [PubMed]
- Homicz, M.R.; McGowan, K.B.; Lottman, L.M.; Beh, G.; Sah, R.L.; Watson, D. A compositional analysis of human nasal septal cartilage. Arch. Facial Plast. Surg. 2003, 5, 53–58. [Google Scholar] [CrossRef]
- Wiggenhauser, P.S.; Schwarz, S.; Rotter, N. The distribution patterns of COMP and matrilin-3 in septal, alar and triangular cartilages of the human nose. Histochem. Cell Biol. 2018, 150, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Gouttenoire, J.; Bougault, C.; Aubert-Foucher, E.; Perrier, E.; Ronzière, M.C.; Sandell, L.; Lundgren-Akerlund, E.; Mallein-Gerin, F. BMP-2 and TGF-β1 differentially control expression of type II procollagen and α10 and α11 integrins in mouse chondrocytes. Eur. J. Cell Biol. 2010, 89, 307–314. [Google Scholar] [CrossRef] [Green Version]
- Bi, W.; Deng, J.M.; Zhang, Z.; Behringer, R.R.; De Crombrugghe, B. Sox9 is required for cartilage formation. Nat. Genet. 1999, 22, 85–89. [Google Scholar] [CrossRef]
- Nakayama, N.; Pothiawala, A.; Lee, J.Y.; Matthias, N.; Umeda, K.; Ang, B.K.; Huard, J.; Huang, Y.; Sun, D. Human pluripotent stem cell-derived chondroprogenitors for cartilage tissue engineering. Cell. Mol. Life Sci. 2020, 77, 2543–2563. [Google Scholar] [CrossRef] [PubMed]
- Barry, F.; Boynton, R.E.; Liu, B.; Murphy, J.M. Chondrogenic differentiation of mesenchymal stem cells from bone marrow: Differentiation-dependent gene expression of matrix components. Exp. Cell Res. 2001, 268, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.H.; Kudva, A.K.; Guckert, N.L.; Linse, K.D.; Roy, K. Unique biomaterial compositions direct bone marrow stem cells into specific chondrocytic phenotypes corresponding to the various zones of articular cartilage. Biomaterials 2011, 32, 1327–1338. [Google Scholar] [CrossRef]
- Varas, L.; Ohlsson, L.B.; Honeth, G.; Olsson, A.; Bengtsson, T.; Wiberg, C.; Bockermann, R.; Järnum, S.; Richter, J.; Pennington, D.; et al. α10 Integrin expression is up-regulated on fibroblast growth factor-2-treated mesenchymal stem cells with improved chondrogenic differentiation potential. Stem Cells Dev. 2007, 16, 965–978. [Google Scholar] [CrossRef]
- Vinod, E.; Kachroo, U.; Amirtham, S.M.; Ramasamy, B.; Sathishkumar, S. Comparative analysis of fresh chondrocytes, cultured chondrocytes and chondroprogenitors derived from human articular cartilage. Acta Histochem. 2020, 122, 151462. [Google Scholar] [CrossRef]
- Jayasuriya, C.T.; Chen, Q. Potential benefits and limitations of utilizing chondroprogenitors in cell-based cartilage therapy. Connect. Tissue Res. 2015, 56, 265–271. [Google Scholar] [CrossRef] [Green Version]
- Holtzer, H.; Abbott, J.; Lash, J.; Holtzer, S. The Loss of Phenotypic Traits By Differentiated Cells in Vitro, I. Dedifferentiation of Cartilage Cells. Proc. Natl. Acad. Sci. USA 2006, 46, 1533–1542. [Google Scholar] [CrossRef] [Green Version]
- Wuest, S.L.; Caliò, M.; Wernas, T.; Tanner, S.; Giger-Lange, C.; Wyss, F.; Ille, F.; Gantenbein, B.; Egli, M. Influence of mechanical unloading on articular chondrocyte dedifferentiation. Int. J. Mol. Sci. 2018, 19, 1289. [Google Scholar] [CrossRef] [Green Version]
- Zanetti, N.C.; Solursh, M. Induction of chondrogenesis in limb mesenchymal cultures by disruption of the actin cytoskeleton. J. Cell Biol. 1984, 99, 115–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benya, P.D.; Shaffer, J.D. Dedifferentiated chondrocytes reexpress the differentiated collagen phenotype when cultured in agarose gels. Cell 1982, 30, 215–224. [Google Scholar] [CrossRef]
- Benya, P.D.; Padilla, S.R. Dihydrocytochalasin B enhances transforming growth factor-β-induced reexpression of the differentiated chondrocyte phenotype without stimulation of collagen synthesis. Exp. Cell Res. 1993, 204, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Woods, A.; Wang, G.; Beier, F. Regulation of chondrocyte differentiation by the act in cytoskeleton and adhesive interactions. J. Cell. Physiol. 2007, 213, 1–8. [Google Scholar] [CrossRef]
- Glowacki, J.; Trepman, E.; Folkman, J. Cell Shape and Phenotypic Expression in Chondrocytes. Exp. Biol. Med. 2013, 172, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Tekari, A.; Luginbuehl, R.; Hofstetter, W.; Egli, R.J. Chondrocytes expressing intracellular collagen type II enter the cell cycle and co-express collagen type i in monolayer culture. J. Orthop. Res. 2014, 32, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Barlič, A.; Drobnič, M.; Maličev, E.; Kregar-Velikonja, N. Quantitative analysis of gene expression in human articular chondrocytes assigned for autologous implantation. J. Orthop. Res. 2008, 26, 847–853. [Google Scholar] [CrossRef]
- Haudenschild, D.R.; Chen, J.; Pang, N.; Lotz, M.K.; D’Lima, D.D. Rho kinase-dependent activation of SOX9 in chondrocytes. Arthritis Rheum. 2010, 62, 191–200. [Google Scholar] [CrossRef] [Green Version]
- Lu, Z.; Doulabi, B.Z.; Huang, C.; Bank, R.A.; Helder, M.N. Collagen type II enhances chondrogenesis in adipose tissue-derived stem cells by affecting cell shape. Tissue Eng.-Part A 2010, 16, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Levorson, E.J.; Hu, O.; Mountziaris, P.M.; Kasper, F.K.; Mikos, A.G. Cell-derived polymer/extracellular matrix composite scaffolds for cartilage regeneration, part 2: Construct devitalization and determination of chondroinductive capacity. Tissue Eng.-Part C Methods 2014, 20, 358–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wise, J.K.; Yarin, A.L.; Megaridis, C.M.; Cho, M. Chondrogenic differentiation of human mesenchymal stem cells on oriented nanofibrous scaffolds: Engineering the superficial zone of articular cartilage. Tissue Eng.-Part A 2009, 15, 913–921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbosa, I.; Garcia, S.; Barbier-Chassefière, V.; Caruelle, J.P.; Martelly, I.; Papy-García, D. Improved and simple micro assay for sulfated glycosaminoglycans quantification in biological extracts and its use in skin and muscle tissue studies. Glycobiology 2003, 13, 647–653. [Google Scholar] [CrossRef] [Green Version]





| Gene | Primer (Left) | Primer (Right) | Nt | RefSeq |
|---|---|---|---|---|
| Target Genes | ||||
| ACAN | cctccccttcacgtgtaaaa | gctccgcttctgtagtctgc | 64 | NM_001135.3 |
| COL10A1 | caccttctgcactgctcatc | ggcagcatattctcagatgga | 104 | NM_000493.3 |
| COL1A1 | ggattccctggacctaaag | ggaacacctcgctctcca | 63 | NM_000088.3 |
| COL2A1 | gtgaacctggtgtctctggtc | tttccaggttttccagcttc | 94 | NM_001844.4 |
| ITGA10 | cttttcctcgcacgtggt | gctccattccagtcataggc | 70 | NM_001004439.1 |
| ITGA11 | cttttcctcgcacgtggt | gctccattccagtcataggc | 69 | NM_001004439.1 |
| MKI67 | ccaaccaaaagaaagtctctgg | tgatggttgaggctgttcct | 78 | NM_001145966.1 |
| MMP13 | ccagtctccgaggagaaaca | aaaaacagctccgcatcaac | 85 | NM_002427.3 |
| PPARG | tgacaggaaagacaacagacaaa | gaggactcagggtggttcag | 126 | NM_001330615.1 |
| RUNX2 | cagtgacaccatgtcagcaa | gctcacgtcgctcattttg | 104 | NM_001015051.3 |
| SOX9 | gtacccgcacttgcacaac | tctcgctctcgttcagaagtc | 74 | NM_000346.3 |
| Housekeeping Gene | ||||
| HPRT1 | tgaccttgatttattttgcatacc | cgagcaagacgttcagtcct | 102 | NM_000194.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuhlmann, C.; Schenck, T.L.; Aszodi, A.; Giunta, R.E.; Wiggenhauser, P.S. Zone-Dependent Architecture and Biochemical Composition of Decellularized Porcine Nasal Cartilage Modulate the Activity of Adipose Tissue-Derived Stem Cells in Cartilage Regeneration. Int. J. Mol. Sci. 2021, 22, 9917. https://doi.org/10.3390/ijms22189917
Kuhlmann C, Schenck TL, Aszodi A, Giunta RE, Wiggenhauser PS. Zone-Dependent Architecture and Biochemical Composition of Decellularized Porcine Nasal Cartilage Modulate the Activity of Adipose Tissue-Derived Stem Cells in Cartilage Regeneration. International Journal of Molecular Sciences. 2021; 22(18):9917. https://doi.org/10.3390/ijms22189917
Chicago/Turabian StyleKuhlmann, Constanze, Thilo L. Schenck, Attila Aszodi, Riccardo E. Giunta, and Paul Severin Wiggenhauser. 2021. "Zone-Dependent Architecture and Biochemical Composition of Decellularized Porcine Nasal Cartilage Modulate the Activity of Adipose Tissue-Derived Stem Cells in Cartilage Regeneration" International Journal of Molecular Sciences 22, no. 18: 9917. https://doi.org/10.3390/ijms22189917
APA StyleKuhlmann, C., Schenck, T. L., Aszodi, A., Giunta, R. E., & Wiggenhauser, P. S. (2021). Zone-Dependent Architecture and Biochemical Composition of Decellularized Porcine Nasal Cartilage Modulate the Activity of Adipose Tissue-Derived Stem Cells in Cartilage Regeneration. International Journal of Molecular Sciences, 22(18), 9917. https://doi.org/10.3390/ijms22189917






