Paternal Methyl Donor Supplementation in Rats Improves Fertility, Physiological Outcomes, Gut Microbial Signatures and Epigenetic Markers Altered by High Fat/High Sucrose Diet
Abstract
:1. Introduction
2. Results
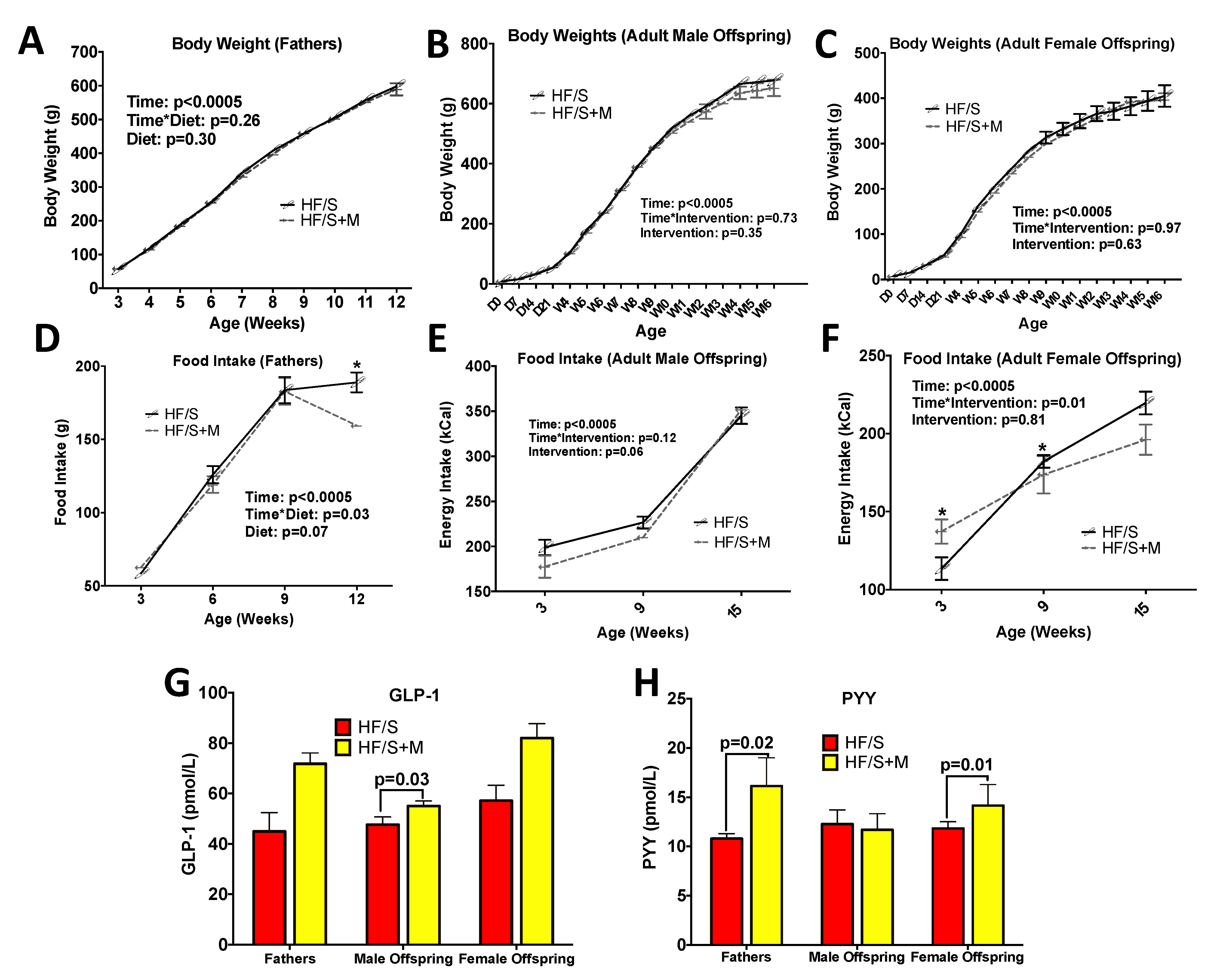
2.1. Paternal HF/S+M Decreases Adiposity in Adult Male and Female Offspring
2.2. Paternal HF/S+M Reduces Fasting Insulin and Insulin Resistance
2.3. HF/S+M Improved Markers of Reproductive Performance in Fathers
2.4. Paternal HF/S+M Consumption Improved Epigenetic Markers in Fathers and Offspring
2.5. Gut Microbiota and Short Chain Fatty Acids Are Impacted by Paternal HF/S+M Consumption Intergenerationally
3. Discussion
4. Materials and Methods
4.1. Animal Model and Dietary Treatment
4.2. Body Weight, Food Intake and Body Composition
4.3. Oral Glucose Tolerance Test (OGTT) and Insulin Tolerance Test (ITT)
4.4. Tissue Harvest and Blood Insulin, GLP-1, PYY and HOMA-IR
4.5. Hepatic Triglyceride Analysis
4.6. Gut Microbiota 16S rRNA Gene Sequencing
4.7. Cecal Short Chain Fatty Acids
4.8. RT-PCR of DNA Methyltransferase mRNA and microRNAs
4.9. Statistical and Bioinformatics Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DNMT | DNA methyltransferase |
| HF/S | High fat/high sucrose |
| HF/S+M | High fat/high sucrose supplemented with methyl donor cocktail |
| Hcy | Homocysteine |
| HOMA-IR | Homeostatic Model Assessment of Insulin Resistance |
| miR | microRNA |
| ncRNAs | Non-coding RNAs |
| SAM | S-adenosylmethionine |
| SCFAs | Short chain fatty acids |
References
- Liu, Y.; Ding, Z. Obesity, a Serious Etiologic Factor for Male Subfertility in Modern Society. Reproduction 2017, 154, R123–R131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giahi, L.; Mohammadmoradi, S.; Javidan, A.; Sadeghi, M.R. Nutritional Modifications in Male Infertility: A Systematic Review Covering 2 Decades. Nutr. Rev. 2016, 74, 118–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salas-Huetos, A.; Bulló, M.; Salas-Salvadó, J. Dietary Patterns, Foods and Nutrients in Male Fertility Parameters and Fecundability: A Systematic Review of Observational Studies. Hum. Reprod. Update 2017, 23, 371–389. [Google Scholar] [CrossRef] [PubMed]
- Crean, A.J.; Senior, A.M. High-Fat Diets Reduce Male Reproductive Success in Animal Models: A Systematic Review and Meta-Analysis. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2019, 20, 921–933. [Google Scholar] [CrossRef] [PubMed]
- Szendroedi, J.; Roden, M. Ectopic Lipids and Organ Function. Curr. Opin. Lipidol. 2009, 20, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Stinkens, R.; Goossens, G.H.; Jocken, J.W.E.; Blaak, E.E. Targeting Fatty Acid Metabolism to Improve Glucose Metabolism. Obes. Rev. 2015, 16, 715–757. [Google Scholar] [CrossRef]
- Campos-Silva, P.; Furriel, A.; Costa, W.S.; Sampaio, F.J.B.; Gregório, B.M.; Campos-Silva, P.; Furriel, A.; Costa, W.S.; Sampaio, F.J.B.; Gregório, B.M. Metabolic and Testicular Effects of the Long-Term Administration of Different High-Fat Diets in Adult Rats. Int. Braz. J. Urol. 2015, 41, 569–575. [Google Scholar] [CrossRef] [Green Version]
- Fan, Y.; Liu, Y.; Xue, K.; Gu, G.; Fan, W.; Xu, Y.; Ding, Z. Diet-Induced Obesity in Male C57BL/6 Mice Decreases Fertility as a Consequence of Disrupted Blood-Testis Barrier. PLoS ONE 2015, 10, e0120775. [Google Scholar] [CrossRef]
- Eacker, S.M.; Agrawal, N.; Qian, K.; Dichek, H.L.; Gong, E.-Y.; Lee, K.; Braun, R.E. Hormonal Regulation of Testicular Steroid and Cholesterol Homeostasis. Mol. Endocrinol. 2008, 22, 623–635. [Google Scholar] [CrossRef] [Green Version]
- Sèdes, L.; Thirouard, L.; Maqdasy, S.; Garcia, M.; Caira, F.; Lobaccaro, J.-M.A.; Beaudoin, C.; Volle, D.H. Cholesterol: A Gatekeeper of Male Fertility? Front. Endocrinol. 2018, 9. [Google Scholar] [CrossRef]
- Peschansky, V.J.; Wahlestedt, C. Non-Coding RNAs as Direct and Indirect Modulators of Epigenetic Regulation. Epigenetics 2014, 9, 3–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cescon, M.; Chianese, R.; Tavares, R.S. Environmental Impact on Male (In)Fertility via Epigenetic Route. J. Clin. Med. 2020, 9, 2520. [Google Scholar] [CrossRef] [PubMed]
- Papaioannou, M.D.; Nef, S. MicroRNAs in the Testis: Building up Male Fertility. J. Androl. 2010, 31, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Minatel, B.C.; Martinez, V.D.; Ng, K.W.; Sage, A.P.; Tokar, T.; Marshall, E.A.; Anderson, C.; Enfield, K.S.S.; Stewart, G.L.; Reis, P.P.; et al. Large-Scale Discovery of Previously Undetected MicroRNAs Specific to Human Liver. Hum. Genom. 2018, 12, 16. [Google Scholar] [CrossRef] [PubMed]
- Gujar, H.; Weisenberger, D.J.; Liang, G. The Roles of Human DNA Methyltransferases and Their Isoforms in Shaping the Epigenome. Genes 2019, 10, 172. [Google Scholar] [CrossRef] [Green Version]
- Mandaviya, P.R.; Stolk, L.; Heil, S.G. Homocysteine and DNA Methylation: A Review of Animal and Human Literature. Mol. Genet. Metab. 2014, 113, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Kang, S. Functional Implications of DNA Methylation in Adipose Biology. Diabetes 2019, 68, 871–878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- King, S.E.; Skinner, M.K. Epigenetic Transgenerational Inheritance of Obesity Susceptibility. Trends Endocrinol. Metab. 2020, 31, 478–494. [Google Scholar] [CrossRef]
- Ducker, G.S.; Rabinowitz, J.D. One-Carbon Metabolism in Health and Disease. Cell Metab. 2017, 25, 27–42. [Google Scholar] [CrossRef] [Green Version]
- Radziejewska, A.; Muzsik, A.; Milagro, F.I.; Martínez, J.A.; Chmurzynska, A. One-Carbon Metabolism and Nonalcoholic Fatty Liver Disease: The Crosstalk between Nutrients, Microbiota, and Genetics. Lifestyle Genom. 2020, 13, 53–63. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Stojanovic, I.; Stoimenov, T.J.; Pavlovic, D.; Kocic, G.; Bjelakovic, G.B.; Sokolovic, D.; Basic, J. Polyamines, Folic Acid Supplementation and Cancerogenesis. Pteridines 2017, 28, 115–131. [Google Scholar] [CrossRef]
- Lurz, E.; Horne, R.G.; Määttänen, P.; Wu, R.Y.; Botts, S.R.; Li, B.; Rossi, L.; Johnson-Henry, K.C.; Pierro, A.; Surette, M.G.; et al. Vitamin B12 Deficiency Alters the Gut Microbiota in a Murine Model of Colitis. Front. Nutr. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Nazki, F.H.; Sameer, A.S.; Ganaie, B.A. Folate: Metabolism, Genes, Polymorphisms and the Associated Diseases. Gene 2014, 533, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Chiuve, S.E.; Giovannucci, E.L.; Hankinson, S.E.; Zeisel, S.H.; Dougherty, L.W.; Willett, W.C.; Rimm, E.B. The Association between Betaine and Choline Intakes and the Plasma Concentrations of Homocysteine in Women. Am. J. Clin. Nutr. 2007, 86, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Choline, Other Methyl-Donors and Epigenetics. Nutrients 2017, 9, 445. [Google Scholar] [CrossRef] [PubMed]
- Salbaum, J.M.; Kappen, C. Genetic and Epigenomic Footprints of Folate. Prog. Mol. Biol. Transl. Sci. 2012, 108, 129–158. [Google Scholar] [CrossRef] [Green Version]
- Kok, D.E.G.; Dhonukshe-Rutten, R.A.M.; Lute, C.; Heil, S.G.; Uitterlinden, A.G.; van der Velde, N.; van Meurs, J.B.J.; van Schoor, N.M.; Hooiveld, G.J.E.J.; de Groot, L.C.P.G.M.; et al. The Effects of Long-Term Daily Folic Acid and Vitamin B12 Supplementation on Genome-Wide DNA Methylation in Elderly Subjects. Clin. Epigenetics 2015, 7, 121. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, E.; Matte, A.; Perfilyev, A.; de Mello, V.D.; Käkelä, P.; Pihlajamäki, J.; Ling, C. Epigenetic Alterations in Human Liver From Subjects With Type 2 Diabetes in Parallel With Reduced Folate Levels. J. Clin. Endocrinol. Metab. 2015, 100, E1491–E1501. [Google Scholar] [CrossRef]
- Ly, A.; Hoyt, L.; Crowell, J.; Kim, Y.-I. Folate and DNA Methylation. Antioxid. Redox Signal. 2012, 17, 302–326. [Google Scholar] [CrossRef]
- Romano, K.A.; Vivas, E.I.; Amador-Noguez, D.; Rey, F.E. Intestinal Microbiota Composition Modulates Choline Bioavailability from Diet and Accumulation of the Proatherogenic Metabolite Trimethylamine-N-Oxide. mBio 2015, 6. [Google Scholar] [CrossRef] [Green Version]
- Mayengbam, S.; Chleilat, F.; Reimer, R.A. Dietary Vitamin B6 Deficiency Impairs Gut Microbiota and Host and Microbial Metabolites in Rats. Biomedicines 2020, 8, 469. [Google Scholar] [CrossRef] [PubMed]
- Kalhan, S.C. One Carbon Metabolism in Pregnancy: Impact on Maternal, Fetal and Neonatal Health. Mol. Cell. Endocrinol. 2016, 435, 48–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, K.; Jaiswal, K. One-Carbon Metabolism, Spermatogenesis, and Male Infertility. Reprod. Sci. 2013, 20, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Sujit, K.M.; Sarkar, S.; Singh, V.; Pandey, R.; Agrawal, N.K.; Trivedi, S.; Singh, K.; Gupta, G.; Rajender, S. Genome-Wide Differential Methylation Analyses Identifies Methylation Signatures of Male Infertility. Hum. Reprod. 2018, 33, 2256–2267. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Yang, L. Transgenerational Impaired Spermatogenesis with Sperm H19 and Gtl2 Hypomethylation Induced by the Endocrine Disruptor p,p′-DDE. Toxicol. Lett. 2018, 297, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Irani, M.; Amirian, M.; Sadeghi, R.; Lez, J.L.; Roudsari, R.L. The Effect of Folate and Folate Plus Zinc Supplementation on Endocrine Parameters and Sperm Characteristics in Sub-Fertile Men: A Systematic Review and Meta-Analysis. Urol. J. 2017, 14, 4069–4078. [Google Scholar] [CrossRef] [PubMed]
- Carlin, J.; George, R.; Reyes, T.M. Methyl Donor Supplementation Blocks the Adverse Effects of Maternal High Fat Diet on Offspring Physiology. PLoS ONE 2013, 8, e63549. [Google Scholar] [CrossRef]
- Zeisel, S.H. Importance of Methyl Donors during Reproduction. Am. J. Clin. Nutr. 2009, 89, 673S–677S. [Google Scholar] [CrossRef]
- Tang, M.; Xu, W.; Wang, Q.; Xiao, W.; Xu, R. Potential of DNMT and Its Epigenetic Regulation for Lung Cancer Therapy. Curr. Genom. 2009, 10, 336–352. [Google Scholar] [CrossRef]
- Benito-Vicente, A.; Uribe, K.B.; Rotllan, N.; Ramírez, C.M.; Jebari-Benslaiman, S.; Goedeke, L.; Canfrán-Duque, A.; Galicia-García, U.; Saenz De Urturi, D.; Aspichueta, P.; et al. MiR-27b Modulates Insulin Signaling in Hepatocytes by Regulating Insulin Receptor Expression. Int. J. Mol. Sci. 2020, 21, 8675. [Google Scholar] [CrossRef]
- Jiménez-Chillarón, J.C.; Díaz, R.; Martínez, D.; Pentinat, T.; Ramón-Krauel, M.; Ribó, S.; Plösch, T. The Role of Nutrition on Epigenetic Modifications and Their Implications on Health. Biochimie 2012, 94, 2242–2263. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.; Hyppönen, E.; Berry, D.; Power, C. Associations between Parental and Offspring Adiposity up to Midlife: The Contribution of Adult Lifestyle Factors in the 1958 British Birth Cohort Study. Am. J. Clin. Nutr. 2010, 92, 946–953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Law, C.; Lo Conte, R.; Power, C. Intergenerational Influences on Childhood Body Mass Index: The Effect of Parental Body Mass Index Trajectories. Am. J. Clin. Nutr. 2009, 89, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, H.; Mitsui, T.; Eguchi, T.; Tamada, S.; Hiramatsu, Y. The Effects of Paternal High-Fat Diet Exposure on Offspring Metabolism with Epigenetic Changes in the Mouse Adiponectin and Leptin Gene Promoters. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E236–E245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bannister, A.J.; Kouzarides, T. Regulation of Chromatin by Histone Modifications. Cell Res. 2011, 21, 381. [Google Scholar] [CrossRef] [PubMed]
- Cedar, H.; Bergman, Y. Linking DNA Methylation and Histone Modification: Patterns and Paradigms. Nat. Rev. Genet. 2009, 10, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Pigeyre, M.; Yazdi, F.T.; Kaur, Y.; Meyre, D. Recent Progress in Genetics, Epigenetics and Metagenomics Unveils the Pathophysiology of Human Obesity. Clin. Sci. Lond. Engl. 1979 2016, 130, 943–986. [Google Scholar] [CrossRef] [Green Version]
- van Dijk, S.J.; Tellam, R.L.; Morrison, J.L.; Muhlhausler, B.S.; Molloy, P.L. Recent Developments on the Role of Epigenetics in Obesity and Metabolic Disease. Clin. Epigenetics 2015, 7, 66. [Google Scholar] [CrossRef] [Green Version]
- Yao, H.; Fan, C.; Lu, Y.; Fan, X.; Xia, L.; Li, P.; Wang, R.; Tang, T.; Wang, Y.; Qi, K. Alteration of Gut Microbiota Affects Expression of Adiponectin and Resistin through Modifying DNA Methylation in High-Fat Diet-Induced Obese Mice. Genes Nutr. 2020, 15, 12. [Google Scholar] [CrossRef]
- Lu, Y.; Fan, C.; Liang, A.; Fan, X.; Wang, R.; Li, P.; Qi, K. Effects of SCFA on the DNA Methylation Pattern of Adiponectin and Resistin in High-Fat-Diet-Induced Obese Male Mice. Br. J. Nutr. 2018, 120, 385–392. [Google Scholar] [CrossRef] [Green Version]
- Elsner, V.; Cechinel, L.; de Meireles, L.F.; Bertoldi, K.; Siqueira, I. Epigenetic Marks Are Modulated by Gender and Time of the Day in the Hippocampi of Adolescent Rats: A Preliminary Study. Neural Regen. Res. 2018, 13, 2160. [Google Scholar] [CrossRef] [PubMed]
- Maschietto, M.; Bastos, L.C.; Tahira, A.C.; Bastos, E.P.; Euclydes, V.L.V.; Brentani, A.; Fink, G.; de Baumont, A.; Felipe-Silva, A.; Francisco, R.P.V.; et al. Sex Differences in DNA Methylation of the Cord Blood Are Related to Sex-Bias Psychiatric Diseases. Sci. Rep. 2017, 7, 44547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheung, O.K.-W.; Cheng, A.S.-L. Gender Differences in Adipocyte Metabolism and Liver Cancer Progression. Front. Genet. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.; Barnes, S.; Demark-Wahnefried, W.; Morrow, C.; Salvador, C.; Skibola, C.; Tollefsbol, T.O. Influences of Diet and the Gut Microbiome on Epigenetic Modulation in Cancer and Other Diseases. Clin. Epigenetics 2015, 7, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mischke, M.; Plösch, T. The Gut Microbiota and Their Metabolites: Potential Implications for the Host Epigenome. Adv. Exp. Med. Biol. 2016, 902, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Magnúsdóttir, S.; Ravcheev, D.; de Crécy-Lagard, V.; Thiele, I. Systematic Genome Assessment of B-Vitamin Biosynthesis Suggests Co-Operation among Gut Microbes. Front. Genet. 2015, 6. [Google Scholar] [CrossRef] [Green Version]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut Microbiota Functions: Metabolism of Nutrients and Other Food Components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef] [Green Version]
- Paßlack, N.; Vahjen, W.; Zentek, J. Impact of Dietary Cellobiose on the Fecal Microbiota of Horses. J. Equine Vet. Sci. 2020, 91, 103106. [Google Scholar] [CrossRef]
- Hang, I.; Rinttila, T.; Zentek, J.; Kettunen, A.; Alaja, S.; Apajalahti, J.; Harmoinen, J.; de Vos, W.M.; Spillmann, T. Effect of High Contents of Dietary Animal-Derived Protein or Carbohydrates on Canine Faecal Microbiota. BMC Vet. Res. 2012, 8, 90. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Daza, M.-C.; Roquim, M.; Dudonné, S.; Pilon, G.; Levy, E.; Marette, A.; Roy, D.; Desjardins, Y. Berry Polyphenols and Fibers Modulate Distinct Microbial Metabolic Functions and Gut Microbiota Enterotype-Like Clustering in Obese Mice. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Yuan, X.; Chen, R.; Zhang, Y.; Lin, X.; Yang, X. Gut Microbiota: Effect of Pubertal Status. BMC Microbiol. 2020, 20, 334. [Google Scholar] [CrossRef] [PubMed]
- Cady, N.; Peterson, S.R.; Freedman, S.N.; Mangalam, A.K. Beyond Metabolism: The Complex Interplay Between Dietary Phytoestrogens, Gut Bacteria, and Cells of Nervous and Immune Systems. Front. Neurol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Song, X.; Zhou, H.; Zhou, X.; Xia, Y.; Dong, X.; Zhong, W.; Tang, S.; Wang, L.; Wen, S.; et al. Gut Microbiome Associates With Lipid-Lowering Effect of Rosuvastatin in Vivo. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hung, Y.-H.; Kanke, M.; Kurtz, C.L.; Cubitt, R.; Bunaciu, R.P.; Miao, J.; Zhou, L.; Graham, J.L.; Hussain, M.M.; Havel, P.; et al. Acute Suppression of Insulin Resistance-Associated Hepatic MiR-29 in Vivo Improves Glycemic Control in Adult Mice. Physiol. Genom. 2019, 51, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.-L.; Novo-Veleiro, I.; Manzanedo, L.; Alvela-Suárez, L.; Macías, R.; Laso, F.-J.; Marcos, M. Role of MicroRNAs in Alcohol-Induced Liver Disorders and Non-Alcoholic Fatty Liver Disease. World J. Gastroenterol. 2018, 24, 4104–4118. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-E.; Fu, T.; Seok, S.; Kim, D.-H.; Yu, E.; Lee, K.-W.; Kang, Y.; Li, X.; Kemper, B.; Kemper, J.K. Elevated MicroRNA-34a in Obesity Reduces NAD+ Levels and SIRT1 Activity by Directly Targeting NAMPT. Aging Cell 2013, 12, 1062–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tryndyak, V.P.; Latendresse, J.R.; Montgomery, B.; Ross, S.A.; Beland, F.A.; Rusyn, I.; Pogribny, I.P. Plasma MicroRNAs Are Sensitive Indicators of Inter-Strain Differences in the Severity of Liver Injury Induced in Mice by a Choline- and Folate-Deficient Diet. Toxicol. Appl. Pharmacol. 2012, 262, 52–59. [Google Scholar] [CrossRef] [Green Version]
- Trajkovski, M.; Hausser, J.; Soutschek, J.; Bhat, B.; Akin, A.; Zavolan, M.; Heim, M.H.; Stoffel, M. MicroRNAs 103 and 107 Regulate Insulin Sensitivity. Nature 2011, 474, 649–653. [Google Scholar] [CrossRef] [Green Version]
- Park, J.-H.; Ahn, J.; Kim, S.; Kwon, D.Y.; Ha, T.Y. Murine Hepatic MiRNAs Expression and Regulation of Gene Expression in Diet-Induced Obese Mice. Mol. Cells 2011, 31, 33–38. [Google Scholar] [CrossRef]
- Zhang, M.; Tang, Y.; Tang, E.; Lu, W. MicroRNA-103 Represses Hepatic de Novo Lipogenesis and Alleviates NAFLD via Targeting FASN and SCD1. Biochem. Biophys. Res. Commun. 2020, 524, 716–722. [Google Scholar] [CrossRef]
- Rottiers, V.; Najafi-Shoushtari, S.H.; Kristo, F.; Gurumurthy, S.; Zhong, L.; Li, Y.; Cohen, D.E.; Gerszten, R.E.; Bardeesy, N.; Mostoslavsky, R.; et al. MicroRNAs in Metabolism and Metabolic Diseases. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 225–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayner, K.J.; Esau, C.C.; Hussain, F.N.; McDaniel, A.L.; Marshall, S.M.; van Gils, J.M.; Ray, T.D.; Sheedy, F.J.; Goedeke, L.; Liu, X.; et al. Inhibition of MiR-33a/b in Non-Human Primates Raises Plasma HDL and Lowers VLDL Triglycerides. Nature 2011, 478, 404–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dávalos, A.; Goedeke, L.; Smibert, P.; Ramírez, C.M.; Warrier, N.P.; Andreo, U.; Cirera-Salinas, D.; Rayner, K.; Suresh, U.; Pastor-Pareja, J.C.; et al. MiR-33a/b Contribute to the Regulation of Fatty Acid Metabolism and Insulin Signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 9232–9237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, N.L.; Singh, A.K.; Rotllan, N.; Goedeke, L.; Wing, A.; Canfrán-Duque, A.; Diaz-Ruiz, A.; Araldi, E.; Baldán, Á.; Camporez, J.-P.; et al. Genetic Ablation of MiR-33 Increases Food Intake, Enhances Adipose Tissue Expansion, and Promotes Obesity and Insulin Resistance. Cell Rep. 2018, 22, 2133–2145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belleannée, C.; Calvo, É.; Caballero, J.; Sullivan, R. Epididymosomes Convey Different Repertoires of MicroRNAs throughout the Bovine Epididymis. Biol. Reprod. 2013, 89, 30. [Google Scholar] [CrossRef]
- Benatti, R.O.; Melo, A.M.; Borges, F.O.; Ignacio-Souza, L.M.; Simino, L.a.P.; Milanski, M.; Velloso, L.A.; Torsoni, M.A.; Torsoni, A.S. Maternal High-Fat Diet Consumption Modulates Hepatic Lipid Metabolism and MicroRNA-122 (MiR-122) and MicroRNA-370 (MiR-370) Expression in Offspring. Br. J. Nutr. 2014, 111, 2112–2122. [Google Scholar] [CrossRef] [Green Version]
- Cordero, P.; Campion, J.; Milagro, F.I.; Martinez, J.A. Transcriptomic and Epigenetic Changes in Early Liver Steatosis Associated to Obesity: Effect of Dietary Methyl Donor Supplementation. Mol. Genet. Metab. 2013, 110, 388–395. [Google Scholar] [CrossRef]
- Cordero, P.; Gomez-Uriz, A.M.; Campion, J.; Milagro, F.I.; Martinez, J.A. Dietary Supplementation with Methyl Donors Reduces Fatty Liver and Modifies the Fatty Acid Synthase DNA Methylation Profile in Rats Fed an Obesogenic Diet. Genes Nutr. 2013, 8, 105–113. [Google Scholar] [CrossRef] [Green Version]
- Cordero, P.; Milagro, F.I.; Campion, J.; Martinez, J.A. Supplementation with Methyl Donors during Lactation to High-Fat-Sucrose-Fed Dams Protects Offspring against Liver Fat Accumulation When Consuming an Obesogenic Diet. J. Dev. Orig. Health Dis. 2014, 5, 385–395. [Google Scholar] [CrossRef]
- Singh, B.; Saxena, A. Surrogate Markers of Insulin Resistance: A Review. World J. Diabetes 2010, 1, 36–47. [Google Scholar] [CrossRef]
- Nicolucci, A.C.; Hume, M.P.; Martínez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics Reduce Body Fat and Alter Intestinal Microbiota in Children Who Are Overweight or With Obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nettleton, J.E.; Klancic, T.; Schick, A.; Choo, A.C.; Shearer, J.; Borgland, S.L.; Chleilat, F.; Mayengbam, S.; Reimer, R.A. Low-Dose Stevia (Rebaudioside A) Consumption Perturbs Gut Microbiota and the Mesolimbic Dopamine Reward System. Nutrients 2019, 11, 1248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nettleton, J.E.; Cho, N.A.; Klancic, T.; Nicolucci, A.C.; Shearer, J.; Borgland, S.L.; Johnston, L.A.; Ramay, H.R.; Tuplin, E.N.; Chleilat, F.; et al. Maternal Low-Dose Aspartame and Stevia Consumption with an Obesogenic Diet Alters Metabolism, Gut Microbiota and Mesolimbic Reward System in Rat Dams and Their Offspring. Gut 2020, 69, 1807–1817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paul, H.A.; Collins, K.H.; Bomhof, M.R.; Vogel, H.J.; Reimer, R.A. Potential Impact of Metabolic and Gut Microbial Response to Pregnancy and Lactation in Lean and Diet-Induced Obese Rats on Offspring Obesity Risk. Mol. Nutr. Food Res. 2018, 62. [Google Scholar] [CrossRef] [PubMed]
- Paul, H.A.; Collins, K.H.; Nicolucci, A.C.; Urbanski, S.J.; Hart, D.A.; Vogel, H.J.; Reimer, R.A. Maternal Prebiotic Supplementation Reduces Fatty Liver Development in Offspring through Altered Microbial and Metabolomic Profiles in Rats. FASEB J. 2019, 33, 5153–5167. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [Green Version]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [Green Version]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic Biomarker Discovery and Explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [Green Version]




| HF/S | HF/S+M | p-Value | |
|---|---|---|---|
| Fathers | |||
| BMC (g) | 15.82 ± 0.45 | 15.39 ± 0.43 | 0.85 |
| BMD (g/cm2) | 0.17 ± 0.002 | 0.17 ± 0.002 | 0.88 |
| Fat Mass (g) | 143.82.4 ± 11.53 | 123.0 ± 11.81 | 0.86 |
| Lean+ BMC (g) | 507.76 ± 13.84 | 491.72 ± 12.81 | 0.92 |
| % Body Fat | 21.82 ± 1.22 | 19.63 ± 1.37 | 0.68 |
| Male Offspring | |||
| BMC (g) | 17.07 ± 0.18 | 16.67 ± 0.49 | 0.02 |
| BMD (g/cm2) | 0.18 ± 0.002 | 0.17 ± 0.002 | 0.34 |
| Fat Mass (g) | 135.11 ± 5.95 | 116.14 ± 1.77 | 0.005 |
| Lean+ BMC (g) | 526.17 ± 9.88 | 546.88 ± 14.48 | 0.12 |
| % Body Fat | 19.93 ± 0.73 | 16.83 ± 0.84 | 0.56 |
| Female Offspring | |||
| BMC (g) | 11.78 ± 0.53 | 11.74 ± 0.41 | 0.19 |
| BMD (g/cm2) | 0.163 ± 0.002 | 0.164 ± 0.003 | 0.96 |
| Fat Mass (g) | 100.84 ± 18.78 | 95.53 ± 5.69 | 0.005 |
| Lean+ BMC (g) | 304.36 ± 9.29 | 293.33 ± 10.26 | 0.86 |
| % Body Fat | 23.54 ± 3.40 | 24.48 ± 0.88 | 0.10 |
| HF/S | HF/S+M | |
|---|---|---|
| Males (n) | 13 | 12 |
| Males cohabited with females | 13 | 12 |
| Nights cohabited until conception | 3.00 ± 0.56 | 2.00 ± 0.33 * |
| # of successful pregnancies | 10 | 9 |
| Fertility Index (%) | 77 | 75 |
| # of pups born alive (per father) | 14.7 ± 0.6 | 13.3 ± 0.9 |
| # of stillbirth pups | 3 | 1 |
| Pup survival (%) | 97.8 | 99.9 |
| Relative abundance of male pups (%) | 51.2 ± 4.1 | 49.2 ± 1.0 |
| Relative abundance of female pups (%) | 48.8 ± 4.1 | 46.98 ± 2.0 |
| HF/S | HF/S+M | p-Value | |
|---|---|---|---|
| Fathers | |||
| miR-21 | 472.0 ± 151.2 | 701.5 ± 244.9 | 0.78 |
| miR-24 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.67 |
| miR-33 | 0.00017 ± 0.00002 | 0.0002 ± 0.00006 | 0.03 |
| miR-34a | 1.02 ± 0.17 | 0.56 ± 0.06 | 0.002 |
| miR-103 | 20.35 ± 1.91 | 21.65 ± 3.94 | 0.03 |
| miR-107 | 0.32 ± 0.01 | 0.42 ± 0.01 | 0.04 |
| miR-122a | 5966.08 ± 1122.78 | 5822.70 ± 1310.41 | 0.51 |
| miR-130a | 0.02 ± 0.01 | 0.01 ± 0.01 | 0.10 |
| miR-143 | 0.04 ± 0.01 | 0.03 ± 0.01 | 0.70 |
| miR-let-7c | 3.82 ± 0.89 | 5.05 ± 0.95 | 0.90 |
| Male Offspring | |||
| miR-21 | 1591.20 ± 722.13 | 1539.55 ± 476.72 | 0.47 |
| miR-24 | 0.12 ± 0.02 | 0.15 ± 0.04 | 0.03 |
| miR-33 | 0.00027 ± 0.00004 | 0.00034 ± 0.0009 | 0.006 |
| miR-34a | 0.66 ± 0.15 | 0.57 ± 0.10 | 0.36 |
| miR-103 | 17.78 ± 2.70 | 17.57 ± 3.29 | 0.44 |
| miR-107 | 0.34 ± 0.05 | 0.33 ± 0.03 | 0.16 |
| miR-122a | 8370.09 ± 1788.12 | 18,602.11 ± 5733.86 | 0.001 |
| miR-130a | 0.02 ± 0.004 | 0.01 ± 0.003 | 0.16 |
| miR-143 | 0.03 ± 0.01 | 0.04 ± 0.002 | 0.009 |
| miR-let-7c | 6.15 ± 2.79 | 7.00 ± 3.02 | 0.60 |
| Female Offspring | |||
| miR-21 | 910.20 ± 221.59 | 723.52 ± 468.95 | 0.14 |
| miR-24 | 0.07 ± 0.02 | 0.07 ± 0.01 | 0.42 |
| miR-33 | 0.0002 ± 0.0001 | 0.0001 ± 0.00001 | 0.0001 |
| miR-34a | 1.07 ± 0.35 | 0.99 ± 0.16 | 0.09 |
| miR-103 | 11.65 ± 1.00 | 15.47 ± 2.16 | 0.09 |
| miR-107 | 0.57 ± 0.11 | 0.58 ± 0.02 | 0.11 |
| miR-122a | 12,872.52 ± 4519.43 | 8144.62 ± 1220.41 | 0.21 |
| miR-130a | 0.007 ± 0.002 | 0.01 ± 0.003 | 0.16 |
| miR-143 | 0.02 ± 0.006 | 0.02 ± 0.002 | 0.16 |
| miR-let-7c | 12.86 ± 3.90 | 6.2 ± 0.64 | 0.06 |
| HF/S | HF/S+M | p-Value | |
|---|---|---|---|
| Paternal | |||
| Chao1 | 209.19 ± 8.37 | 265.78 ± 26.87 | 0.049 |
| Shannon | 3.87 ± 0.09 | 3.95 ± 0.13 | 0.61 |
| Simpson | 0.95 ± 0.004 | 0.95 ± 0.01 | 0.99 |
| Male Offspring | |||
| Chao1 | 182.82 ± 28.3 | 168.67 ± 7.09 | 0.63 |
| Shannon | 3.76 ± 0.08 | 3.83 ± 0.08 | 0.54 |
| Simpson | 0.95 ± 0.01 | 0.95 ± 0.01 | 0.55 |
| Female Offspring | |||
| Chao1 | 192.31 ± 11.22 | 139.44 ± 11.48 | 0.004 |
| Shannon | 3.99 ± 0.06 | 3.67 ± 0.07 | 0.002 |
| Simpson | 0.96 ± 0.003 | 0.95 ± 0.004 | 0.004 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chleilat, F.; Schick, A.; Deleemans, J.M.; Reimer, R.A. Paternal Methyl Donor Supplementation in Rats Improves Fertility, Physiological Outcomes, Gut Microbial Signatures and Epigenetic Markers Altered by High Fat/High Sucrose Diet. Int. J. Mol. Sci. 2021, 22, 689. https://doi.org/10.3390/ijms22020689
Chleilat F, Schick A, Deleemans JM, Reimer RA. Paternal Methyl Donor Supplementation in Rats Improves Fertility, Physiological Outcomes, Gut Microbial Signatures and Epigenetic Markers Altered by High Fat/High Sucrose Diet. International Journal of Molecular Sciences. 2021; 22(2):689. https://doi.org/10.3390/ijms22020689
Chicago/Turabian StyleChleilat, Faye, Alana Schick, Julie M. Deleemans, and Raylene A. Reimer. 2021. "Paternal Methyl Donor Supplementation in Rats Improves Fertility, Physiological Outcomes, Gut Microbial Signatures and Epigenetic Markers Altered by High Fat/High Sucrose Diet" International Journal of Molecular Sciences 22, no. 2: 689. https://doi.org/10.3390/ijms22020689








