Incorporation of a Nitric Oxide Donating Motif into Novel PC-PLC Inhibitors Provides Enhanced Anti-Proliferative Activity
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis of N-Nitrosylated Benzylamines
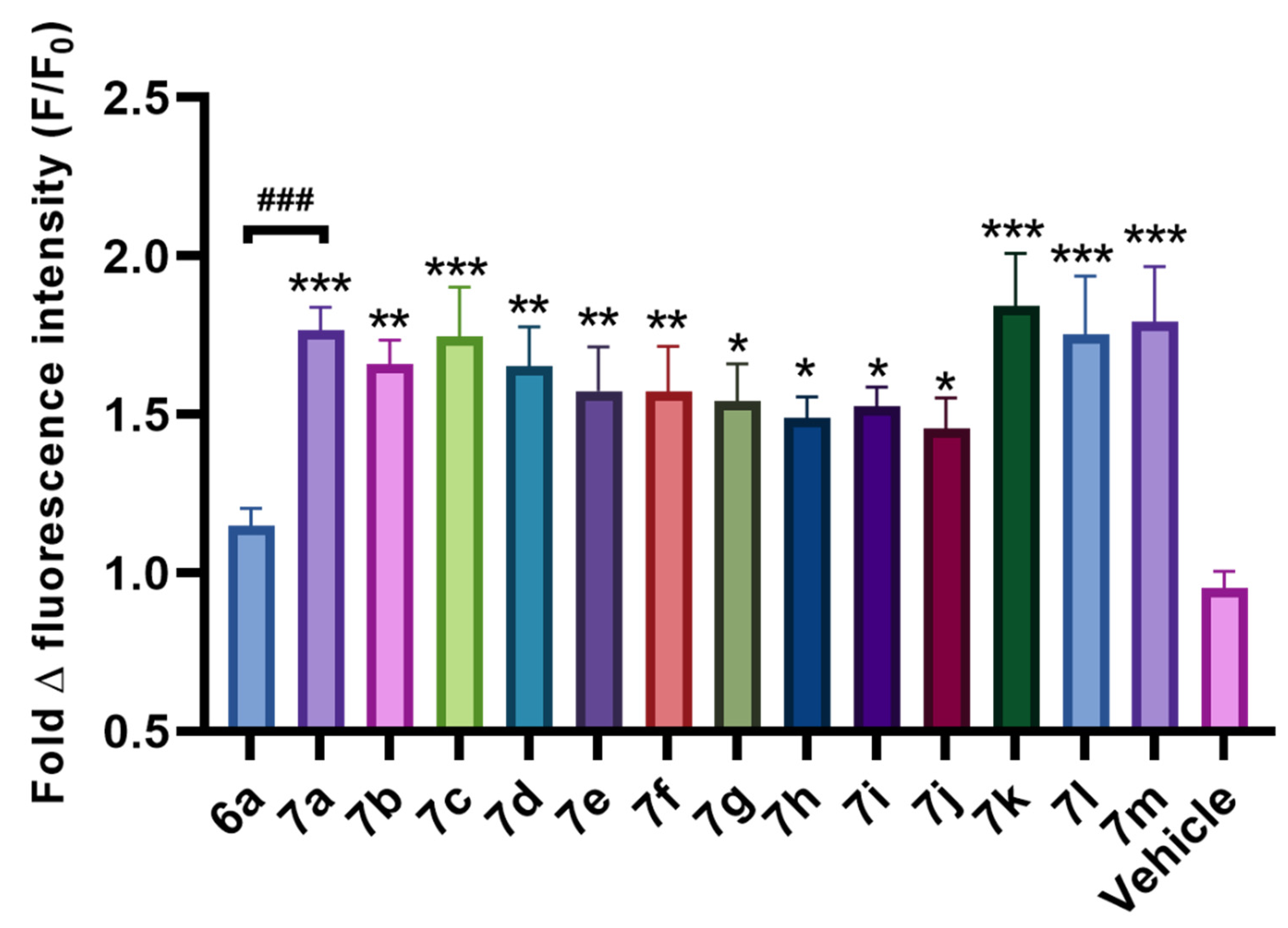
2.2. Assessment of NO Donor Activity by N-Nitrosylated Benzylamines
2.3. Anti-Proliferative Activity of N-Nitrosylated Benzylamines
3. Materials and Methods
3.1. Synthesis of N-Nitrosylated Derivatives
3.1.1. Synthesis of Compounds 2–6a–m
3.1.2. General Procedure for Synthesis of N-Nitrosylated Benzylamines 7a–m
3.2. Nitric Oxide Release Assay
3.2.1. Cell Culture
3.2.2. Intracellular NO Release and Detection
3.3. Cell Proliferation Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Z.; Yu, Y.; Fu, D.; Li, Z.; Niu, X.; Liao, M.; Lu, S. Functional Roles of PC-PLC and Cdc20 in the Cell Cycle, Proliferation, and Apoptosis. Cell Biochem. Funct. 2010, 28, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Fu, D.; Ma, Y.; Wu, W.; Zhu, X.; Jia, C.; Zhao, Q.; Zhang, C.; Wu, X.Z. Cell-Cycle-Dependent PC-PLC Regulation by APC/CCdc20-Mediated Ubiquitin-Proteasome Pathway. J. Cell. Biochem. 2009, 107, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Spadaro, F.; Ramoni, C.; Mezzanzanica, D.; Miotti, S.; Alberti, P.; Cecchetti, S.; Iorio, E.; Dolo, V.; Canevari, S.; Podo, F. Phosphatidylcholine-Specific Phospholipase C Activation in Epithelial Ovarian Cancer Cells. Cancer Res. 2008, 68, 6541–6549. [Google Scholar] [CrossRef] [PubMed]
- Abalsamo, L.; Spadaro, F.; Bozzuto, G.; Paris, L.; Cecchetti, S.; Lugini, L.; Iorio, E.; Molinari, A.; Ramoni, C.; Podo, F. Inhibition of Phosphatidylcholine-Specific Phospholipase C Results in Loss of Mesenchymal Traits in Metastatic Breast Cancer Cells. Breast Cancer Res. BCR 2012, 14, R50. [Google Scholar] [CrossRef] [PubMed]
- Bagnoli, M.; Granata, A.; Nicoletti, R.; Krishnamachary, B.; Bhujwalla, Z.M.; Canese, R.; Podo, F.; Canevari, S.; Iorio, E.; Mezzanzanica, D. Choline Metabolism Alteration: A Focus on Ovarian Cancer. Front. Oncol. 2016, 6, 153. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Bhujwalla, Z.M.; Glunde, K. Targeting Phospholipid Metabolism in Cancer. Front. Oncol. 2016, 6, 266. [Google Scholar] [CrossRef]
- Gould, C.M.; Newton, A.C. The Life and Death of Protein Kinase C. Curr. Drug Targets 2008, 9, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Kao, S.-J.; Lei, H.-C.; Kuo, C.-T.; Chang, M.-S.; Chen, B.-C.; Chang, Y.-C.; Chiu, W.-T.; Lin, C.-H. Lipoteichoic Acid Induces Nuclear Factor-KappaB Activation and Nitric Oxide Synthase Expression via Phosphatidylinositol 3-Kinase, Akt, and P38 MAPK in RAW 264.7 Macrophages. Immunology 2005, 115, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Shen, S.; Verma, I.M. NF-ΚB, an Active Player in Human Cancers. Cancer Immunol. Res. 2014, 2, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Hong, J.T. Roles of NF-ΚB in Cancer and Inflammatory Diseases and Their Therapeutic Approaches. Cells 2016, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Chiang, L.L.; Kuo, C.T.; Wang, C.H.; Chen, T.F.; Ho, Y.S.; Kuo, H.P.; Lin, C.H. Involvement of Nuclear Factor-KappaB in Lipoteichoic Acid-Induced Cyclooxygenase-2 Expression in RAW 264.7 Macrophages. J. Pharm. Pharmacol. 2003, 55, 115–123. [Google Scholar] [CrossRef]
- Schütze, S.; Potthoff, K.; Machleidt, T.; Berkovic, D.; Wiegmann, K.; Krönke, M. TNF Activates NF-Kappa B by Phosphatidylcholine-Specific Phospholipase C-Induced “Acidic” Sphingomyelin Breakdown. Cell 1992, 71, 765–776. [Google Scholar] [CrossRef]
- Lin, C.-C.; Hsiao, L.-D.; Chien, C.-S.; Lee, C.-W.; Hsieh, J.-T.; Yang, C.-M. Tumor Necrosis Factor-Alpha-Induced Cyclooxygenase-2 Expression in Human Tracheal Smooth Muscle Cells: Involvement of P42/P44 and P38 Mitogen-Activated Protein Kinases and Nuclear Factor-KappaB. Cell. Signal. 2004, 16, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Kopincová, J.; Púzserová, A.; Bernátová, I. Biochemical Aspects of Nitric Oxide Synthase Feedback Regulation by Nitric Oxide. Interdiscip. Toxicol. 2011, 4, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Vannini, F.; Kashfi, K.; Nath, N. The Dual Role of INOS in Cancer. Redox Biol. 2015, 6, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Sands, W.A.; Bulut, V.; Severn, A.; Xu, D.; Liew, F.Y. Inhibition of Nitric Oxide Synthesis by Interleukin-4 May Involve Inhibiting the Activation of Protein Kinase C Epsilon. Eur. J. Immunol. 1994, 24, 2345–2350. [Google Scholar] [CrossRef] [PubMed]
- Kengatharan, M.; De Kimpe, S.J.; Thiemermann, C. Analysis of the Signal Transduction in the Induction of Nitric Oxide Synthase by Lipoteichoic Acid in Macrophages. Br. J. Pharmacol. 1996, 117, 1163–1170. [Google Scholar] [CrossRef]
- Tschaikowsky, K.; Meisner, M.; Schönhuber, F.; Rügheimer, E. Induction of Nitric Oxide Synthase Activity in Phagocytic Cells Inhibited by Tricyclodecan-9-Yl-Xanthogenate (D609). Br. J. Pharmacol. 1994, 113, 664–668. [Google Scholar] [CrossRef][Green Version]
- Colasanti, M.; Persichini, T.; Menegazzi, M.; Mariotto, S.; Giordano, E.; Caldarera, C.M.; Sogos, V.; Lauro, G.M.; Suzuki, H. Induction of Nitric Oxide Synthase MRNA Expression: Suppression by Exogenous Nitric Oxide. J. Biol. Chem. 1995, 270, 26731–26733. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Lee, S.-J.; Cheong, I.; Billiar, T.R.; Chung, H.-T.; Han, J.-A.; Kwon, Y.-G.; Ha, K.-S.; Kim, Y.-M. Nitric Oxide Suppresses Inducible Nitric Oxide Synthase Expression by Inhibiting Post-Translational Modification of IκB. Exp. Mol. Med. 2004, 36, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.R.; Botting, C.H.; Panico, M.; Morris, H.R.; Hay, R.T. Inhibition of NF-KappaB DNA Binding by Nitric Oxide. Nucleic Acids Res. 1996, 24, 2236–2242. [Google Scholar] [CrossRef] [PubMed]
- Pilkington, L.I.; Sparrow, K.; Rees, S.W.P.; Paulin, E.K.; van Rensburg, M.; Xu, C.S.; Langley, R.J.; Leung, I.K.H.; Reynisson, J.; Leung, E.; et al. Development, Synthesis and Biological Investigation of a Novel Class of Potent PC-PLC Inhibitors. Eur. J. Med. Chem. 2020, 191, 112162. [Google Scholar] [CrossRef] [PubMed]
- Rees, S.W.P.; Leung, E.; Reynisson, J.; Barker, D.; Pilkington, L.I. Development of 2-Morpholino-N-Hydroxybenzamides as Anti-Proliferative PC-PLC Inhibitors. Bioorg. Chem. 2021, 114, 105152. [Google Scholar] [CrossRef] [PubMed]
- Borikar, S.P.; Paul, V. N-Nitrosation of Secondary Amines Using p-TSA-NaNO2 as a Novel Nitrosating Agent Under Mild Conditions. Synth. Commun. 2010, 40, 654–660. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and Computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Tjalkens, R.B.; Carbone, D.L.; Wu, G. Detection of Nitric Oxide Formation in Primary Neural Cells and Tissues. Methods Mol. Biol. 2011, 758, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Gingell, J.J.; Rees, T.A.; Hendrikse, E.R.; Siow, A.; Rennison, D.; Scotter, J.; Harris, P.W.R.; Brimble, M.A.; Walker, C.S.; Hay, D.L. Distinct Patterns of Internalization of Different Calcitonin Gene-Related Peptide Receptors. ACS Pharmacol. Transl. Sci. 2020, 3, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Pope, N.J.; Powell, S.M.; Wigle, J.C.; Denton, M.L. Wavelength- and irradiance-dependent changes in intracellular nitric oxide level. J. Biomed. Opt. 2020, 25, 085001. [Google Scholar] [CrossRef]
- Leung, E.; Kim, J.E.; Rewcastle, G.W.; Finlay, G.J.; Baguley, B.C. Comparison of the Effects of the PI3K/MTOR Inhibitors NVP-BEZ235 and GSK2126458 on Tamoxifen-Resistant Breast Cancer Cells. Cancer Biol. Ther. 2011, 11, 938–946. [Google Scholar] [CrossRef] [PubMed]



| Benzylic Substitution | Relative 3H Thymidine Incorporation (% ± s.e.m) at 10 µM in MDA-MB-231 Cells | |
|---|---|---|
| Benzylamines 6 [23] | N-Nitrosylated Benzylamines 7 | |
| H (a) | 89.1 ± 1.9 | 59.0 ± 4.4 ** |
| 2-F (b) | 105.9 ± 0.1 | 51.4 ± 1.7 *** |
| 3-F (c) | 85.9 ± 1.6 | 42.1 ± 4.2 *** |
| 4-F (d) | 81.4 ± 0.8 | 44.1 ± 3.0 *** |
| 2-Cl (e) | 77.1 ± 5.4 | 30.8 ± 0.5 ** |
| 3-Cl (f) | 79.4 ± 2.7 | 33.9 ± 0.1 *** |
| 4-Cl (g) | 77.1 ± 0.9 | 27.0 ± 0.7 *** |
| 2-Br (h) | 66.6 ± 3.4 | 4.1 ± 0.0 *** |
| 3-Br (i) | 87.4 ± 8.4 | 24.5 ± 0.7 ** |
| 4-Br (j) | 77.0 ± 2.3 | 18.8 ± 1.9 *** |
| 2-OMe (k) | 88.0 ± 5.3 | 41.6 ± 1.6 ** |
| 3-OMe (l) | 95.7 ± 11.0 | 71.7 ± 2.5 |
| 4-OMe (m) | 103.0 ± 8.0 | 25.2 ± 1.5 *** |
| D609 | 99.00 ± 2.1 | |
| Positive Control | 100 ± 0.9 | |
| Benzylic Substitution | Relative 3H Thymidine Incorporation (% ± s.e.m) at 10 µM in HCT-116 Cells | |
|---|---|---|
| Benzylamines 6 [23] | N-Nitrosylated Benzylamines 7 | |
| H (a) | 101.3 ± 0.5 | 75.5 ± 3.4 ** |
| 2-F (b) | 101.1 ± 0.3 | 75.7 ± 0.4 *** |
| 3-F (c) | 91.0 ± 4.3 | 63.2 ± 11.3 |
| 4-F (d) | 89.6 ± 5.5 | 61.0 ± 3.9 * |
| 2-Cl (e) | 86.5 ± 1.0 | 39.7 ± 1.9 *** |
| 3-Cl (f) | 92.1 ± 0.1 | 50.1 ± 2.4 *** |
| 4-Cl (g) | 83.7 ± 1.2 | 39.5 ± 2.7 *** |
| 2-Br (h) | 92.0 ± 0.4 | 24.8 ± 4.4 *** |
| 3-Br (i) | 95.1 ± 1.8 | 46.4 ± 3.5 *** |
| 4-Br (j) | 88.8 ± 0.1 | 41.3 ± 3.2 *** |
| 2-OMe (k) | 96.3 ± 0.4 | 64.9 ± 9.9 * |
| 3-OMe (l) | 102.6 ± 0.4 | 84.7 ± 5.0 * |
| 4-OMe (m) | 100.2 ± 0.3 | 50.4 ± 7.8 * |
| D609 | 98.40 ± 3.2 | |
| Positive Control | 100 ± 2.4 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rees, S.W.P.; Rees, T.A.; Leung, E.; Walker, C.S.; Barker, D.; Pilkington, L.I. Incorporation of a Nitric Oxide Donating Motif into Novel PC-PLC Inhibitors Provides Enhanced Anti-Proliferative Activity. Int. J. Mol. Sci. 2021, 22, 11518. https://doi.org/10.3390/ijms222111518
Rees SWP, Rees TA, Leung E, Walker CS, Barker D, Pilkington LI. Incorporation of a Nitric Oxide Donating Motif into Novel PC-PLC Inhibitors Provides Enhanced Anti-Proliferative Activity. International Journal of Molecular Sciences. 2021; 22(21):11518. https://doi.org/10.3390/ijms222111518
Chicago/Turabian StyleRees, Shaun W. P., Tayla A. Rees, Euphemia Leung, Christopher S. Walker, David Barker, and Lisa I. Pilkington. 2021. "Incorporation of a Nitric Oxide Donating Motif into Novel PC-PLC Inhibitors Provides Enhanced Anti-Proliferative Activity" International Journal of Molecular Sciences 22, no. 21: 11518. https://doi.org/10.3390/ijms222111518
APA StyleRees, S. W. P., Rees, T. A., Leung, E., Walker, C. S., Barker, D., & Pilkington, L. I. (2021). Incorporation of a Nitric Oxide Donating Motif into Novel PC-PLC Inhibitors Provides Enhanced Anti-Proliferative Activity. International Journal of Molecular Sciences, 22(21), 11518. https://doi.org/10.3390/ijms222111518







