Interactions in Ternary Aqueous Solutions of NMA and Osmolytes—PARAFAC Decomposition of FTIR Spectra Series
Abstract
:1. Introduction
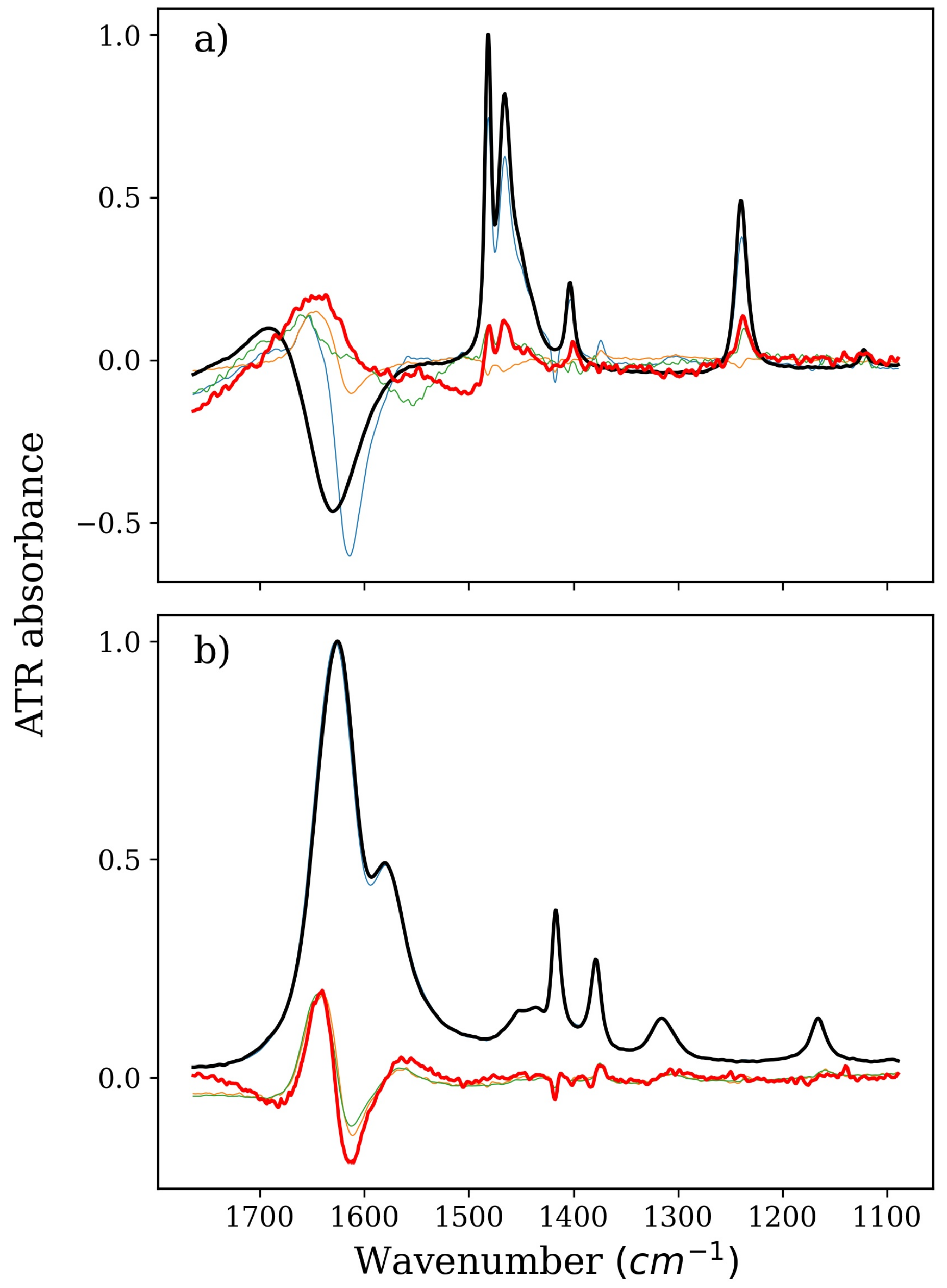
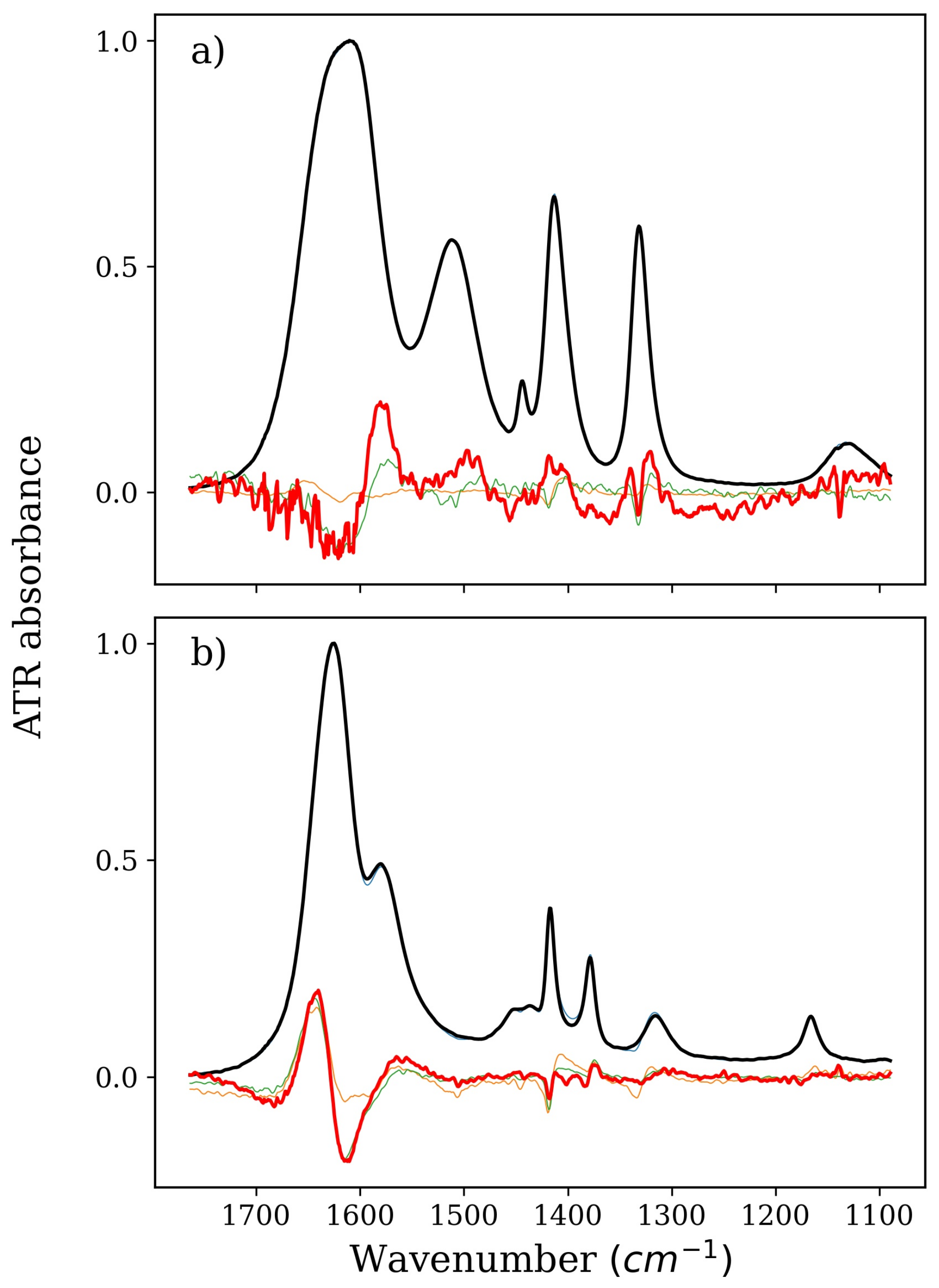
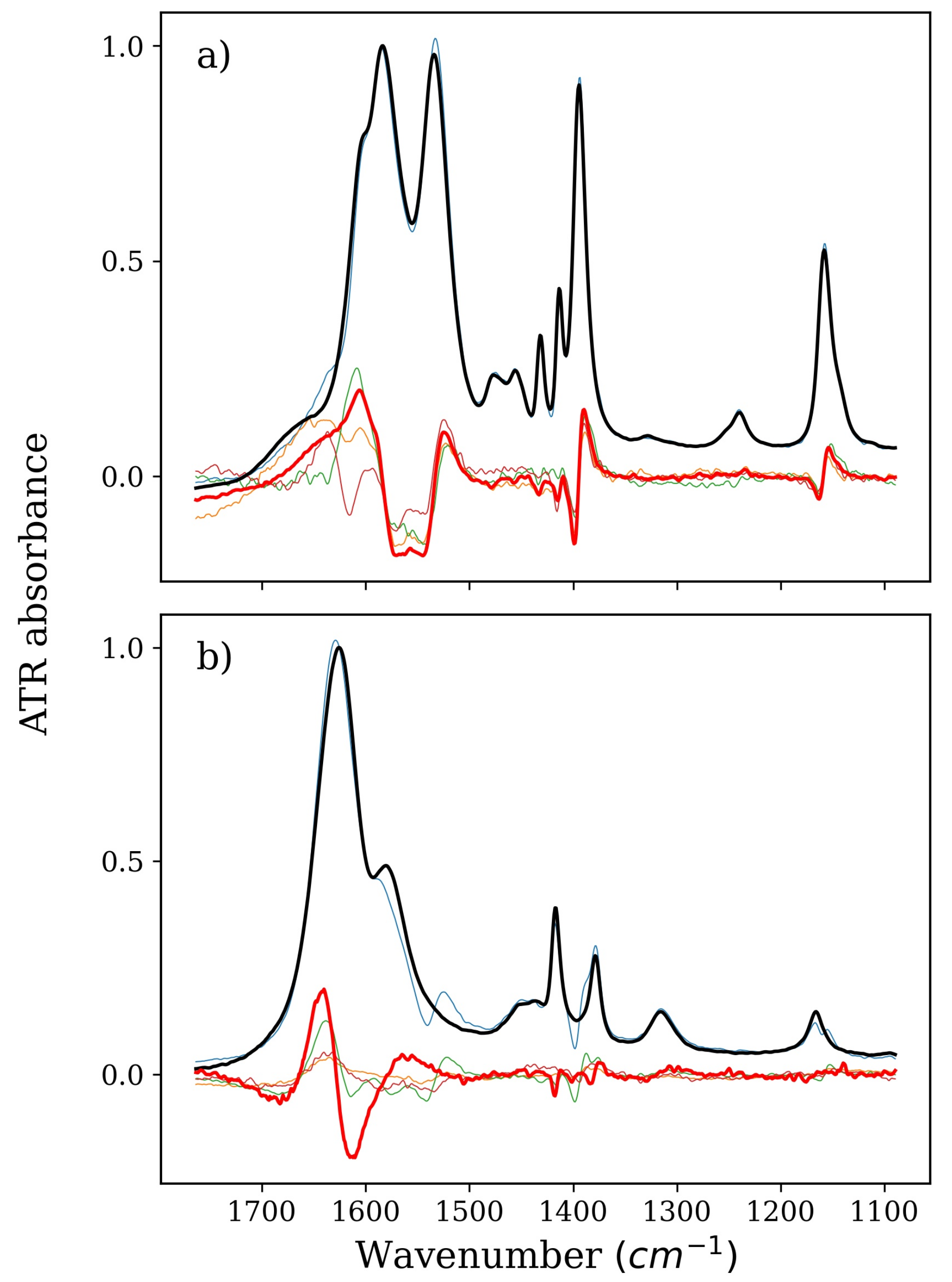
2. Results and Discussion
2.1. Stabilizers: TMAO, GLY, and BET
2.2. Denaturing Agents: TMU and NBU
3. Materials and Methods
3.1. Chemicals and Solutions
3.2. ATR-FTIR Spectroscopy
3.3. Spectra Pretreatment
3.4. Spectra Isolation with PARAFAC
3.5. Derivative Spectra of Solutes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 2D-FTIR | Two-Dimensional Infrared Spectroscopy |
| ATR | Attenuated Total Reflectance |
| BET | Glycine Betaine |
| FTIR | Fourier Transform Infrared Spectroscopy |
| GLY | Glycine |
| NBU | n-Butylurea |
| NMA | N-Methylacetamide |
| PARAFAC | Parallel Factor Analysis |
| TMAO | Trimethylamine N-Oxide |
| TMU | N,N,N’,N’-Trimethylurea |
References
- Rani, A.; Venkatesu, P. Changing relations between proteins and osmolytes: A choice of nature. Phys. Chem. Chem. Phys. 2018, 20, 20315–20333. [Google Scholar] [CrossRef] [PubMed]
- Canchi, D.R.; García, A.E. Cosolvent Effects on Protein Stability. Annu. Rev. Phys. Chem. 2013, 64, 273–293. [Google Scholar] [CrossRef]
- Khan, S.H.; Ahmad, N.; Ahmad, F.; Kumar, R. Naturally occurring organic osmolytes: From cell physiology to disease prevention. IUBMB Life 2010, 62, 891–895. [Google Scholar] [CrossRef]
- Canchi, D.R.; Jayasimha, P.; Rau, D.C.; Makhatadze, G.I.; Garcia, A.E. Molecular Mechanism for the Preferential Exclusion of TMAO from Protein Surfaces. J. Phys. Chem. B 2012, 116, 12095–12104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holthauzen, L.M.F.; Rösgen, J.; Bolen, D.W. Hydrogen Bonding Progressively Strengthens upon Transfer of the Protein Urea-Denatured State to Water and Protecting Osmolytes. Biochemistry 2010, 49, 1310–1318. [Google Scholar] [CrossRef] [PubMed]
- Felitsky, D.J.; Cannon, J.G.; Capp, M.W.; Hong, J.; Van Wynsberghe, A.W.; Anderson, C.F.; Record, M.T. The Exclusion of Glycine Betaine from Anionic Biopolymer Surface: Why Glycine Betaine Is an Effective Osmoprotectant but Also a Compatible Solute. Biochemistry 2004, 43, 14732–14743. [Google Scholar] [CrossRef] [PubMed]
- Bolen, D.W.; Rose, G.D. Structure and Energetics of the Hydrogen-Bonded Backbone in Protein Folding. Annu. Rev. Biochem. 2008, 77, 339–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Street, T.O.; Bolen, D.W.; Rose, G.D. A molecular mechanism for osmolyte-induced protein stability. Proc. Natl. Acad. Sci. USA 2006, 103, 13997–14002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panuszko, A.; Bruździak, P.; Kaczkowska, E.; Stangret, J. General Mechanism of Osmolytes’ Influence on Protein Stability Irrespective of the Type of Osmolyte Cosolvent. J. Phys. Chem. B 2016, 120, 11159–11169. [Google Scholar] [CrossRef]
- Bruździak, P.; Panuszko, A.; Kaczkowska, E.; Piotrowski, B.; Daghir, A.; Demkowicz, S.; Stangret, J. Taurine as a water structure breaker and protein stabilizer. Amino Acids 2018, 50, 125–140. [Google Scholar] [CrossRef] [Green Version]
- Bennion, B.J.; Daggett, V. Counteraction of urea-induced protein denaturation by trimethylamine N-oxide: A chemical chaperone at atomic resolution. Proc. Natl. Acad. Sci. USA 2004, 101, 6433–6438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zou, Q.; Bennion, B.J.; Daggett, V.; Murphy, K.P. The Molecular Mechanism of Stabilization of Proteins by TMAO and Its Ability to Counteract the Effects of Urea. J. Am. Chem. Soc. 2002, 124, 1192–1202. [Google Scholar] [CrossRef] [PubMed]
- Saladino, G.; Marenchino, M.; Pieraccini, S.; Campos-Olivas, R.; Sironi, M.; Gervasio, F.L. A Simple Mechanism Underlying the Effect of Protecting Osmolytes on Protein Folding. J. Chem. Theory Comput. 2011, 7, 3846–3852. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Paul, S. Exploring the Counteracting Mechanism of Trehalose on Urea Conferred Protein Denaturation: A Molecular Dynamics Simulation Study. J. Phys. Chem. B 2015, 119, 9820–9834. [Google Scholar] [CrossRef] [PubMed]
- Sarma, R.; Paul, S. Exploring the Molecular Mechanism of Trimethylamine-N-oxide’s Ability to Counteract the Protein Denaturing Effects of Urea. J. Phys. Chem. B 2013, 117, 5691–5704. [Google Scholar] [CrossRef]
- Wei, H.; Fan, Y.; Gao, Y.Q. Effects of Urea, Tetramethyl Urea, and Trimethylamine N-Oxide on Aqueous Solution Structure and Solvation of Protein Backbones: A Molecular Dynamics Simulation Study. J. Phys. Chem. B 2010, 114, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Pattanayak, S.K.; Chettiyankandy, P.; Chowdhuri, S. Effects of co-solutes on the hydrogen bonding structure and dynamics in aqueous N-methylacetamide solution: A molecular dynamics simulations study. Mol. Phys. 2014, 112, 2906–2919. [Google Scholar] [CrossRef]
- Kumar, N.; Kishore, N. Mechanistic insights into osmolyte action in protein stabilization under harsh conditions: N-methylacetamide in glycine betaine–urea mixture. Chem. Phys. 2014, 443, 133–141. [Google Scholar] [CrossRef]
- Chand, A.; Chettiyankandy, P.; Pattanayak, S.K.; Chowdhuri, S. Effects of trimethylamine-N-oxide (TMAO) on aqueous N-methylacetamide solution: A comparison of different force fields of TMAO. J. Mol. Liq. 2017, 225, 926–935. [Google Scholar] [CrossRef]
- Kaczkowska, E.; Wawer, J.; Tyczyńska, M.; Jóźwiak, M.; Boruń, A.; Krakowiak, J. The hydration properties of protein stabilizer, trimethylamine-N-oxide in aqueous solutions of N-methylacetamide – The volumetric and compressibility studies between 288.15 and 308.15 K. Thermochim. Acta 2020, 685, 1–6. [Google Scholar] [CrossRef]
- Kaczkowska, E.; Wawer, J.; Tyczyńska, M.; Jóźwiak, M.; Krakowiak, J. The interaction parameters for solutions of n-butylurea in aqueous solutions of N-methylacetamide—The volumetric and compressibility studies between 288.15 K and 308.15 K. J. Chem. Thermodyn. 2020, 149, 106143. [Google Scholar] [CrossRef]
- Kaczkowska, E.; Wawer, J.; Tyczyńska, M.; Jóźwiak, M.; Krakowiak, J. Understanding the interactions between protein stabilizers and the peptide bond through the analysis of the volumetric and compressibility properties for the model systems. J. Chem. Thermodyn. 2021, 160, 106485. [Google Scholar] [CrossRef]
- Saito, T.; Sao, H.; Ishida, K.; Aoyagi, N.; Kimura, T.; Nagasaki, S.; Tanaka, S. Application of parallel factor analysis for time-resolved laser fluorescence spectroscopy: Implication for metal speciation study. Environ. Sci. Technol. 2010, 44, 5055–5060. [Google Scholar] [CrossRef]
- Zhang, G.; Hu, X.; Fu, P. Spectroscopic studies on the interaction between carbaryl and calf thymus DNA with the use of ethidium bromide as a fluorescence probe. J. Photochem. Photobiol. B Biol. 2012, 108, 53–61. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, G.; Pan, J. Spectroscopic studies of DNA interactions with food colorant indigo carmine with the use of ethidium bromide as a fluorescence probe. J. Agric. Food Chem. 2012, 60, 10867–10875. [Google Scholar] [CrossRef]
- Murphy, K.R.; Stedmon, C.A.; Graeber, D.; Bro, R. Fluorescence spectroscopy and multi-way techniques. PARAFAC. Anal. Methods 2013, 5, 6557–6566. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhu, R.; Ni, Y.; Kokot, S. Competitive interactions of anti-carcinogens with serum albumin: A spectroscopic study of bendamustine and dexamethasone with the aid of chemometrics. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 123, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.X.; Wu, H.L.; Kang, C.; Xiang, S.X.; Yin, X.L.; Gu, H.W.; Zuo, Q.; Yu, R.Q. Quantitative investigation of the dynamic interaction of human serum albumin with procaine using a multi-way calibration method coupled with three-dimensional fluorescence spectroscopy. Anal. Methods 2015, 7, 6552–6560. [Google Scholar] [CrossRef]
- Akbarian, S.; Najafpour, M.M.; Kompany-Zareh, M. PARAFAC study of bovine serum albumin conformational changes in the interaction with nanosized manganese oxide as a biomimetic model for water-oxidizing complex. Int. J. Hydrog. Energy 2017, 42, 9733–9743. [Google Scholar] [CrossRef]
- Berto, S.; Alladio, E. Application of Chemometrics Tools to the Study of the Fe(III)–Tannic Acid Interaction. Front. Chem. 2020, 8, 614171. [Google Scholar] [CrossRef] [PubMed]
- Razavi, M.; Kompany-Zareh, M.; Khoshkam, M. PARAFAC study of L-cys@CdTe QDs interaction to BSA, cytochrome c and trypsin: An approach through electrostatic and covalent bonds. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 246, 119016. [Google Scholar] [CrossRef]
- Harshman, R.A.; Lundy, M.E. PARAFAC: Parallel factor analysis. Comput. Stat. Data Anal. 1994, 18, 39–72. [Google Scholar] [CrossRef]
- Bro, R. PARAFAC. Tutorial and applications. Chemom. Intell. Lab. Syst. 1997, 38, 149–171. [Google Scholar] [CrossRef]
- Baum, A.; Hansen, P.W.; Nørgaard, L.; Sørensen, J.; Mikkelsen, J.D. Rapid quantification of casein in skim milk using Fourier transform infrared spectroscopy, enzymatic perturbation, and multiway partial least squares regression: Monitoring chymosin at work. J. Dairy Sci. 2016, 99, 6071–6079. [Google Scholar] [CrossRef] [Green Version]
- Cui, X.; Zhang, J.; Cai, W.; Shao, X. Chemometric algorithms for analyzing high dimensional temperature dependent near infrared spectra. Chemom. Intell. Lab. Syst. 2017, 170, 109–117. [Google Scholar] [CrossRef]
- Bruździak, P.; Panuszko, A.; Stangret, J. Chemometric determination of solute-affected solvent vibrational spectra as a superior way of information extraction on solute solvation phenomena. Vib. Spectrosc. 2010, 54, 65–71. [Google Scholar] [CrossRef]
- Bruździak, P.; Rakowska, P.W.; Stangret, J. Chemometric method of spectra analysis leading to isolation of lysozyme and CtDNA spectra affected by osmolytes. Appl. Spectrosc. 2012, 66, 1302–1310. [Google Scholar] [CrossRef] [PubMed]
- Noda, I. Two-Dimensional Infrared (2D IR) Spectroscopy: Theory and Applications. Appl. Spectrosc. 1990, 44, 550–561. [Google Scholar] [CrossRef]
- Park, Y.; Jin, S.; Noda, I.; Jung, Y.M. Emerging developments in two-dimensional correlation spectroscopy (2D-COS). J. Mol. Struct. 2020, 1217, 128405. [Google Scholar] [CrossRef]
- Śmiechowski, M.; Stangret, J. Vibrational spectroscopy of semiheavy water (HDO) as a probe of solute hydration. Pure Appl. Chem. 2010, 82, 1869–1887. [Google Scholar] [CrossRef]
- Panuszko, A.; Bruździak, P.; Zielkiewicz, J.; Wyrzykowski, D.; Stangret, J. Effects of Urea and Trimethylamine-N-oxide on the Properties of Water and the Secondary Structure of Hen Egg White Lysozyme. J. Phys. Chem. B 2009, 113, 14797–14809. [Google Scholar] [CrossRef]
- Oprzeska-Zingrebe, E.A.; Smiatek, J. Aqueous Mixtures of Urea and Trimethylamine-N-oxide: Evidence for Kosmotropic or Chaotropic Behavior? J. Phys. Chem. B 2019, 123, 4415–4424. [Google Scholar] [CrossRef] [PubMed]
- Zeman, J.; Holm, C.; Smiatek, J. The Effect of Small Organic Cosolutes on Water Structure and Dynamics. J. Chem. Eng. Data 2020, 65, 1197–1210. [Google Scholar] [CrossRef]
- Bruździak, P. Vapor correction of FTIR spectra—A simple automatic least squares approach. Spectrochim. Acta—Part A Mol. Biomol. Spectrosc. 2019, 223, 117373. [Google Scholar] [CrossRef]
- Biernacki, K.A.; Kaczkowska, E.; Bruździak, P. Aqueous solutions of NMA, Na2HPO4, and NaH2PO4 as models for interaction studies in phosphate—Protein systems. J. Mol. Liq. 2018, 265, 361–371. [Google Scholar] [CrossRef]
- Kossaifi, J.; Panagakis, Y.; Anandkumar, A.; Pantic, M. TensorLy: Tensor Learning in Python. J. Mach. Learn. Res. 2019, 20, 1–6. [Google Scholar]
- Bro, R.; Kiers, H.A. A new efficient method for determining the number of components in PARAFAC models. J. Chemom. 2003, 17, 274–286. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaczkowska, E.; Panuszko, A.; Bruździak, P. Interactions in Ternary Aqueous Solutions of NMA and Osmolytes—PARAFAC Decomposition of FTIR Spectra Series. Int. J. Mol. Sci. 2021, 22, 11684. https://doi.org/10.3390/ijms222111684
Kaczkowska E, Panuszko A, Bruździak P. Interactions in Ternary Aqueous Solutions of NMA and Osmolytes—PARAFAC Decomposition of FTIR Spectra Series. International Journal of Molecular Sciences. 2021; 22(21):11684. https://doi.org/10.3390/ijms222111684
Chicago/Turabian StyleKaczkowska, Emilia, Aneta Panuszko, and Piotr Bruździak. 2021. "Interactions in Ternary Aqueous Solutions of NMA and Osmolytes—PARAFAC Decomposition of FTIR Spectra Series" International Journal of Molecular Sciences 22, no. 21: 11684. https://doi.org/10.3390/ijms222111684
APA StyleKaczkowska, E., Panuszko, A., & Bruździak, P. (2021). Interactions in Ternary Aqueous Solutions of NMA and Osmolytes—PARAFAC Decomposition of FTIR Spectra Series. International Journal of Molecular Sciences, 22(21), 11684. https://doi.org/10.3390/ijms222111684





