TRPA1-Mediated Src Family Kinases Activity Facilitates Cortical Spreading Depression Susceptibility and Trigeminovascular System Sensitization
Abstract
:1. Introduction
2. Results
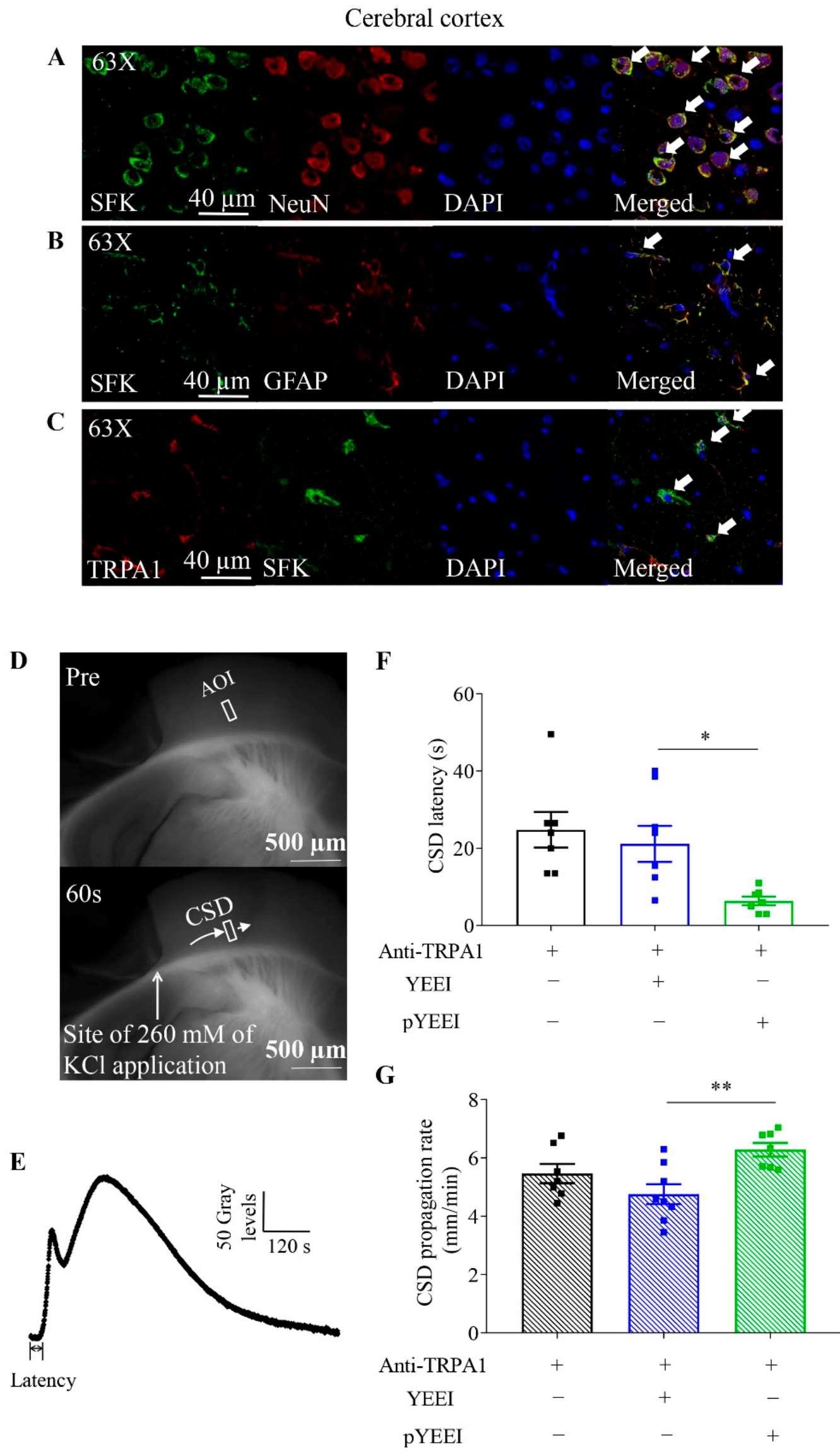
2.1. Co-Expression of TRPA1 and SFKs in Cerebral Cortices of Mice
2.2. pYEEI Restored the Anti-TRPA1 Antibody-Reduced Cortical Susceptibility to CSD in Mouse Brain Slices
2.3. The Anti-TRPA1 Antibody Reduced the Level of Phosphorylated SFKs at Y416 Induced by CSD in Cytosols of Rat Cerebral Cortices
2.4. Co-Localization of TRPA1 and SFKs in Mouse TG
2.5. Saracatinib Reversed CGRP Release and IL-1β mRNA Promoted by Umbellulone in Mouse TG
2.6. Saracatinib Reduced CGRP Release Promoted by Dibutyryl-cAMP (dbcAMP), Which Is Ineffective on IL-1β mRNA Level, in Mouse TG
2.7. PKI (14–22) Amide Reduced the Level of Phosphorylated SFKs at Y416 Promoted by Umbellulone in Mouse TG
2.8. TRPA1 Did Not Form a Complex with SFKs in Rat Cerebral Cortices after CSD
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Immunohistochemistry
4.3. CSD Induction and Recording
4.3.1. Induction and Recording of CSD by Intrinsic Optical Imaging in Mouse Brain Slices and Experimental Design
4.3.2. Induction and Recording of CSD by Electrophysiology in Rats and Experimental Design
4.4. Protein Expression Analysis
4.4.1. Protein Extraction
4.4.2. Western Blot
4.5. Mouse TG Tissue Culture and Experimental Design
4.6. ELISA
4.7. qPCR
4.8. Co-Immunoprecipitation (Co-IP)
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goadsby, P.; Holland, P.; Martins-Oliveira, M.; Hoffmann, J.; Schankin, C.; Akerman, S. Pathophysiology of Migraine: A Disorder of Sensory Processing. Physiol. Rev. 2017, 97, 553–622. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, V.; Ornello, R.; Gagliardo, A.; Torrente, A.; Illuminato, E.; Caponnetto, V.; Frattale, I.; Golini, R.; Di Felice, C.; Graziano, F.; et al. Social Distancing in Chronic Migraine during the COVID-19 Outbreak: Results from a Multicenter Observational Study. Nutrients 2021, 13, 1361. [Google Scholar] [CrossRef]
- Di Stefano, V.; Rispoli, M.G.; Pellegrino, N.; Graziosi, A.; Rotondo, E.; Napoli, C.; Pietrobon, D.; Brighina, F.; Parisi, P. Diagnostic and therapeutic aspects of hemiplegic migraine. J. Neurol. Neurosurg. Psychiatr. 2020, 91, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Spekker, E.; Laborc, K.F.; Bohár, Z.; Nagy-Grócz, G.; Fejes-Szabó, A.; Szűcs, M.; Vécsei, L.; Párdutz, Á. Effect of dural inflammatory soup application on activation and sensitization markers in the caudal trigeminal nucleus of the rat and the modulatory effects of sumatriptan and kynurenic acid. J. Headache Pain 2021, 22, 17. [Google Scholar] [CrossRef] [PubMed]
- Spekker, E.; Tanaka, M.; Szabó, Á.; Vécsei, L. Neurogenic Inflammation: One of the Participants of Migraine and the Contribution of Translational Research. Preprints 2021, 2021110064. [Google Scholar] [CrossRef]
- Ashina, M.; Hansen, J.M.; Do, T.P.; Melo-Carrillo, A.; Burstein, R.; Moskowitz, M.A. Migraine and the trigeminovascular system—40 years and counting. Lancet Neurol. 2019, 18, 795–804. [Google Scholar] [CrossRef]
- Barbanti, P.; Brighina, F.; Egeo, G.; Di Stefano, V.; Silvestro, M.; Russo, A. Migraine as a Cortical Brain Disorder. Headache J. Head Face Pain 2020, 60, 2103–2114. [Google Scholar] [CrossRef] [PubMed]
- Harriott, A.M.; Takizawa, T.; Chung, D.Y.; Chen, S.-P. Spreading depression as a preclinical model of migraine. J. Headache Pain 2019, 20, 1–12. [Google Scholar] [CrossRef]
- Noseda, R.; Burstein, R. Migraine pathophysiology: Anatomy of the trigeminovascular pathway and associated neurological symptoms, cortical spreading depression, sensitization, and modulation of pain. Pain 2013, 154 (Suppl. 1), S44–S53. [Google Scholar] [CrossRef] [Green Version]
- Messlinger, K.; Balcziak, L.K.; Russo, A.F. Cross-talk signaling in the trigeminal ganglion: Role of neuropeptides and other mediators. J. Neural Transm. 2020, 127, 431–444. [Google Scholar] [CrossRef] [Green Version]
- Negro, A.; Martelletti, P. Novel synthetic treatment options for migraine. Expert Opin Pharm. 2021, 22, 907–922. [Google Scholar] [CrossRef]
- Tanaka, M.; Török, N.; Vécsei, L. Are 5-HT(1) receptor agonists effective anti-migraine drugs? Expert Opin Pharm. 2021, 22, 1221–1225. [Google Scholar] [CrossRef]
- Moreno-Ajona, D.; Pérez-Rodríguez, A.; Goadsby, P.J. Small-molecule CGRP receptor antagonists: A new approach to the acute and preventive treatment of migraine. Med. Drug Discov. 2020, 7, 100053. [Google Scholar] [CrossRef]
- Mavridis, T.; Deligianni, C.I.; Karagiorgis, G.; Daponte, A.; Breza, M.; Mitsikostas, D.D. Monoclonal Antibodies Targeting CGRP: From Clinical Studies to Real-World Evidence-What Do We Know So Far? Pharmaceuticals 2021, 14, 700. [Google Scholar] [CrossRef]
- Nassini, R.; Materazzi, S.; Vriens, J.; Prenen, J.; Benemei, S.; De Siena, G.; la Marca, G.; Andre, E.; Preti, D.; Avonto, C.; et al. The ‘headache tree’ via umbellulone and TRPA1 activates the trigeminovascular system. Brain 2012, 135, 376–390. [Google Scholar] [CrossRef] [Green Version]
- Benemei, S.; Fusi, C.; Trevisan, G.; Geppetti, P. The TRPA1 channel in migraine mechanism and treatment. Br. J. Pharmacol. 2014, 171, 2552–2567. [Google Scholar] [CrossRef] [Green Version]
- Demartini, C.; Tassorelli, C.; Zanaboni, A.M.; Tonsi, G.; Francesconi, O.; Nativi, C.; Greco, R. The role of the transient receptor potential ankyrin type-1 (TRPA1) channel in migraine pain: Evaluation in an animal model. J. Headache Pain 2017, 18, 94. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, Y.; Xu, Y.; Ma, D.; Wang, M. The Transient Receptor Potential Ankyrin Type 1 Plays a Critical Role in Cortical Spreading Depression. Neuroscience 2018, 382, 23–34. [Google Scholar] [CrossRef]
- Jiang, L.; Ma, D.; Grubb, B.D.; Wang, M. ROS/TRPA1/CGRP signaling mediates cortical spreading depression. J. Headache Pain 2019, 20, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Kunkler, P.E.; Ballard, C.J.; Oxford, G.S.; Hurley, J.H. TRPA1 receptors mediate environmental irritant-induced meningeal vasodilatation. Pain 2011, 152, 38–44. [Google Scholar] [CrossRef] [Green Version]
- Edelmayer, R.M.; Le, L.N.; Yan, J.; Wei, X.; Nassini, R.; Materazzi, S.; Preti, D.; Appendino, G.; Geppetti, P.; Dodick, D.W.; et al. Activation of TRPA1 on dural afferents: A potential mechanism of headache pain. Pain 2012, 153, 1949–1958. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Kunkler, P.E.; Knopp, K.L.; Oxford, G.S.; Hurley, J.H. Role of intraganglionic transmission in the trigeminovascular pathway. Mol. Pain 2019, 15, 1744806919836570. [Google Scholar] [CrossRef] [Green Version]
- Marone, I.M.; De Logu, F.; Nassini, R.; Goncalves, M.D.C.; Benemei, S.; Ferreira, J.; Jain, P.; Puma, S.L.; Bunnett, N.W.; Geppetti, P.; et al. TRPA1/NOX in the soma of trigeminal ganglion neurons mediates migraine-related pain of glyceryl trinitrate in mice. Brain 2018, 141, 2312–2328. [Google Scholar] [CrossRef] [Green Version]
- Kheradpezhouh, E.; Choy, J.M.C.; Daria, V.R.; Arabzadeh, E. TRPA1 expression and its functional activation in rodent cortex. Open Biol. 2017, 7, 4. [Google Scholar] [CrossRef] [Green Version]
- Kunkler, P.E.; Zhang, L.; Johnson, P.L.; Oxford, G.S.; Hurley, J.H. Induction of chronic migraine phenotypes in a rat model after environmental irritant exposure. Pain 2018, 159, 540–549. [Google Scholar] [CrossRef]
- Kádková, A.; Synytsya, V.; Krusek, J.; Zímová, L.; Vlachová, V. Molecular basis of TRPA1 regulation in nociceptive neurons. A review. Physiol. Res. 2017, 66, 425–439. [Google Scholar] [CrossRef]
- Schmidt, M.; Dubin, A.E.; Petrus, M.J.; Earley, T.J.; Patapoutian, A. Nociceptive Signals Induce Trafficking of TRPA1 to the Plasma Membrane. Neuron 2009, 64, 498–509. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Dai, Y.; Fukuoka, T.; Yamanaka, H.; Kobayashi, K.; Obata, K.; Cui, X.; Tominaga, M.; Noguchi, K. Phospholipase C and protein kinase A mediate bradykinin sensitization of TRPA1: A molecular mechanism of inflammatory pain. Brain 2008, 131, 1241–1251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obara, Y.; Labudda, K.; Dillon, T.J.; Stork, P.J.S. PKA phosphorylation of Src mediates Rap1 activation in NGF and cAMP signaling in PC12 cells. J. Cell Sci. 2004, 117, 6085–6094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, H.B.; Yang, X.; Cao, J.; Li, S.; Liu, Y.N.; Suo, Z.W.; Cui, H.B.; Guo, Z.; Hu, X.D. cAMP-dependent protein kinase activated Fyn in spinal dorsal horn to regulate NMDA receptor function during inflammatory pain. J. Neurochem. 2011, 116, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Morgan, K.; Sadofsky, L.R.; Crow, C.; Morice, A.H. Human TRPM8 and TRPA1 pain channels, including a gene variant with increased sensitivity to agonists (TRPA1 R797T), exhibit differential regulation by SRC-tyrosine kinase inhibitor. Biosci. Rep. 2014, 34, 469–478. [Google Scholar] [CrossRef] [Green Version]
- Bu, F.; Wang, Y.; Jiang, L.; Ma, D.; Quinn, J.; Wang, M. Sarcoma family kinase activity is required for cortical spreading depression. Cephalalgia 2018, 38, 1748–1758. [Google Scholar] [CrossRef]
- Staehr, C.; Hangaard, L.; Bouzinova, E.V.; Kim, S.; Rajanathan, R.; Jessen, P.B.; Luque, N.; Xie, Z.; Lykke-Hartmann, K.; Sandow, S.L.; et al. Smooth muscle Ca2+ sensitization causes hypercontractility of middle cerebral arteries in mice bearing the familial hemiplegic migraine type 2 associated mutation. Br. J. Pharmacol. 2019, 39, 1570–1587. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Zhou, H.-R.; Wang, S.; Liu, C.-Y.; Qin, G.-C.; Fu, Q.-Q.; Zhou, J.-Y.; Chen, L.-X. NR2B-Tyr phosphorylation regulates synaptic plasticity in central sensitization in a chronic migraine rat model. J. Headache Pain 2018, 19, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Park, K.; Fehrenbacher, J.; Thompson, E.; Duarte, D.; Hingtgen, C.; Vasko, M. Signaling pathways that mediate nerve growth factor-induced increase in expression and release of calcitonin gene-related peptide from sensory neurons. Neuroscience 2010, 171, 910–923. [Google Scholar] [CrossRef] [Green Version]
- Marsakova, L.; Barvik, I.; Zima, V.; Zimova, L.; Vlachova, V. The First Extracellular Linker Is Important for Several Aspects of the Gating Mechanism of Human TRPA1 Channel. Front. Mol. Neurosci. 2017, 10, 16. [Google Scholar] [CrossRef]
- Kim, Y.S.; Son, J.Y.; Kim, T.H.; Paik, S.K.; Dai, Y.; Noguchi, K.; Ahn, D.K.; Bae, Y.C. Expression of transient receptor potential ankyrin 1 (TRPA1) in the rat trigeminal sensory afferents and spinal dorsal horn. J. Comp. Neurol. 2010, 518, 687–698. [Google Scholar] [CrossRef]
- Huang, D.; Li, S.; Dhaka, A.; Story, G.M.; Cao, Y.-Q. Expression of the Transient Receptor Potential Channels TRPV1, TRPA1 and TRPM8 in Mouse Trigeminal Primary Afferent Neurons Innervating the Dura. Mol. Pain 2012, 8, 66. [Google Scholar] [CrossRef] [Green Version]
- Jeske, N.A.; Patwardhan, A.M.; Henry, M.A.; Milam, S.B. Fibronectin stimulates TRPV1 translocation in primary sensory neurons. J. Neurochem. 2009, 108, 591–600. [Google Scholar] [CrossRef] [Green Version]
- Dux, M.; Babes, A.; Manchen, J.; Sertel-Nakajima, J.; Vogler, B.; Schramm, J.; Messlinger, K. High-dose phenylephrine increases meningeal blood flow through TRPV1 receptor activation and release of calcitonin gene-related peptide. Eur. J. Pain 2020, 24, 383–397. [Google Scholar] [CrossRef]
- Meents, J.E.; Fischer, M.J.; McNaughton, P.A. Sensitization of TRPA1 by Protein Kinase A. PLoS ONE 2017, 12, e0170097. [Google Scholar] [CrossRef]
- Parsons, S.J.; Parsons, J.T. Src family kinases, key regulators of signal transduction. Oncogene 2004, 23, 7906–7909. [Google Scholar] [CrossRef] [Green Version]
- Nie, L.; Ye, W.-R.; Chen, S.; Chirchiglia, D.; Wang, M. Src Family Kinases in the Central Nervous System: Their Emerging Role in Pathophysiology of Migraine and Neuropathic Pain. Curr. Neuropharmacol. 2021, 19, 665–678. [Google Scholar] [CrossRef]
- Messlinger, K.; Russo, A. Current understanding of trigeminal ganglion structure and function in headache. Cephalalgia 2019, 39, 1661–1674. [Google Scholar] [CrossRef]
- Mukundan, L.; Bishop, G.A.; Head, K.Z.; Zhang, L.; Wahl, L.M.; Suttles, J. TNF Receptor-Associated Factor 6 Is an Essential Mediator of CD40-Activated Proinflammatory Pathways in Monocytes and Macrophages. J. Immunol. 2005, 174, 1081–1090. [Google Scholar] [CrossRef] [Green Version]
- Panicker, N.; Saminathan, H.; Jin, H.; Neal, M.; Harischandra, D.S.; Gordon, R.; Kanthasamy, K.; Lawana, V.; Sarkar, S.; Luo, J.; et al. Fyn Kinase Regulates Microglial Neuroinflammatory Responses in Cell Culture and Animal Models of Parkinson’s Disease. J. Neurosci. 2015, 35, 10058–10077. [Google Scholar] [CrossRef]
- Thalakoti, S.; Patil, V.V.; Damodaram, S.; Vause, C.V.; Langford, L.E.; Freeman, S.E.; Durham, P.L. Neuron?Glia Signaling in Trigeminal Ganglion: Implications for Migraine Pathology. Headache J. Head Face Pain 2007, 47, 1008–1023. [Google Scholar] [CrossRef] [Green Version]
- Vause, C.V.; Durham, P.L. Calcitonin gene-related peptide differentially regulates gene and protein expression in trigeminal glia cells: Findings from array analysis. Neurosci. Lett. 2010, 473, 163–167. [Google Scholar] [CrossRef] [Green Version]
- Bowen, E.J.; Schmidt, T.W.; Firm, C.S.; Russo, A.; Durham, P.L. Tumor necrosis factor-alpha stimulation of calcitonin gene-related peptide expression and secretion from rat trigeminal ganglion neurons. J. Neurochem. 2005, 96, 65–77. [Google Scholar] [CrossRef] [Green Version]
- Capuano, A.; De Corato, A.; Lisi, L.; Tringali, G.; Navarra, P.; Russo, C.D. Proinflammatory-Activated Trigeminal Satellite Cells Promote Neuronal Sensitization: Relevance for Migraine Pathology. Mol. Pain 2009, 5, 43. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Levy, D.; Kainz, V.; Noseda, R.; Jakubowski, M.; Burstein, R. Activation of central trigeminovascular neurons by cortical spreading depression. Ann. Neurol. 2010, 69, 855–865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Levy, D.; Noseda, R.; Kainz, V.; Jakubowski, M.; Burstein, R. Activation of Meningeal Nociceptors by Cortical Spreading Depression: Implications for Migraine with Aura. J. Neurosci. 2010, 30, 8807–8814. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Wang, X. PKC and PKA, but not PKG mediate LPS-induced CGRP release and [Ca2+]i elevation in DRG neurons of neonatal rats. J. Neurosci. Res. 2001, 66, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Wang, A.; Ma, P.; Gong, S.; Tao, J.; Yu, X.; Jiang, X. Regulation of the firing activity by PKA-PKC-Src family kinases in cultured neurons of hypothalamic arcuate nucleus. J. Neurosci. Res. 2020, 98, 384–403. [Google Scholar] [CrossRef] [Green Version]
- Qiu, S.; Chen, T.; Koga, K.; Guo, Y.-Y.; Xu, H.; Song, Q.; Wang, J.-J.; Descalzi, G.; Kaang, B.-K.; Luo, J.-H.; et al. An Increase in Synaptic NMDA Receptors in the Insular Cortex Contributes to Neuropathic Pain. Sci. Signal. 2013, 6, ra34. [Google Scholar] [CrossRef]
- Cortés-Montero, E.; Rodríguez-Muñoz, M.; Ruiz-Cantero, M.D.C.; Cobos, E.J.; Sánchez-Blázquez, P.; Garzón-Niño, J. Calmodulin Supports TRPA1 Channel Association with Opioid Receptors and Glutamate NMDA Receptors in the Nervous Tissue. Int. J. Mol. Sci. 2020, 22, 229. [Google Scholar] [CrossRef]
- Batista, P.; Harris, E.; Werner, M.; Santos, A.; Story, G. Inhibition of TRPA1 and NMDA channels contributes to anti-nociception induced by (-)-linalool. J. Pain 2011, 12, P30. [Google Scholar] [CrossRef]
- Asgar, J.; Zhang, Y.; Saloman, J.; Wang, S.; Chung, M.-K.; Ro, J. The role of TRPA1 in muscle pain and mechanical hypersensitivity under inflammatory conditions in rats. Neuroscience 2015, 310, 206–215. [Google Scholar] [CrossRef] [Green Version]
- Shigetomi, E.; Jackson-Weaver, O.; Huckstepp, R.; O’Dell, T.J.; Khakh, B.S. TRPA1 Channels Are Regulators of Astrocyte Basal Calcium Levels and Long-Term Potentiation via Constitutive D-Serine Release. J. Neurosci. 2013, 33, 10143–10153. [Google Scholar] [CrossRef]
- Howe, A.K. Cross-talk between calcium and protein kinase A in the regulation of cell migration. Curr. Opin. Cell Biol. 2011, 23, 554–561. [Google Scholar] [CrossRef] [Green Version]
- Anguita, E.; Villalobo, A. Src-family tyrosine kinases and the Ca 2+ signal. Biochim. Biophys. Acta (BBA)—Bioenerg. 2017, 1864, 915–932. [Google Scholar] [CrossRef]
- Meotti, F.C.; Figueiredo, C.P.; Manjavachi, M.; Calixto, J.B. The transient receptor potential ankyrin-1 mediates mechanical hyperalgesia induced by the activation of B1 receptor in mice. Biochem. Pharmacol. 2017, 125, 75–83. [Google Scholar] [CrossRef]
- Salter, M.W.; Kalia, L.V. Src kinases: A hub for NMDA receptor regulation. Nat. Rev. Neurosci. 2004, 5, 317–328. [Google Scholar] [CrossRef]
- Iglesias, R.; Locovei, S.; Roque, A.; Alberto, A.P.; Dahl, G.; Spray, D.C.; Scemes, E. P2X7receptor-Pannexin1 complex: Pharmacology and signaling. Am. J. Physiol. Physiol. 2008, 295, C752–C760. [Google Scholar] [CrossRef] [Green Version]
- Bu, F.; Nie, L.; Quinn, J.P.; Wang, M. Sarcoma Family Kinase-Dependent Pannexin-1 Activation after Cortical Spreading Depression Is Mediated by NR2A-Containing Receptors. Int. J. Mol. Sci. 2020, 21, 1269. [Google Scholar] [CrossRef] [Green Version]
- Abdullah, M.; Qaiser, S.; Malik, A.; Chaudhry, M.; Fatima, T.; Malik, A.; Iqbal, N. Comparison Between Menstrual Migraine and Menstrual-Unrelated Migraine in Women Attending Gynecology Clinics. Cureus 2020, 12, e10976. [Google Scholar]
- Zhang, Y.; Saccani, S.; Shin, H.; Nikolajczyk, B.S. Dynamic protein associations define two phases of IL-1beta transcriptional activation. J. Immunol. 2008, 181, 503–512. [Google Scholar] [CrossRef]
- Ma, W.; Chen, X.; Cerne, R.; Syed, S.K.; Ficorilli, J.V.; Cabrera, O.; Obukhov, A.G.; Efanov, A.M. Catechol estrogens stimulate insulin secretion in pancreatic β-cells via activation of the transient receptor potential A1 (TRPA1) channel. J. Biol. Chem. 2019, 294, 2935–5880. [Google Scholar] [CrossRef] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nie, L.; Jiang, L.; Quinn, J.P.; Grubb, B.D.; Wang, M. TRPA1-Mediated Src Family Kinases Activity Facilitates Cortical Spreading Depression Susceptibility and Trigeminovascular System Sensitization. Int. J. Mol. Sci. 2021, 22, 12273. https://doi.org/10.3390/ijms222212273
Nie L, Jiang L, Quinn JP, Grubb BD, Wang M. TRPA1-Mediated Src Family Kinases Activity Facilitates Cortical Spreading Depression Susceptibility and Trigeminovascular System Sensitization. International Journal of Molecular Sciences. 2021; 22(22):12273. https://doi.org/10.3390/ijms222212273
Chicago/Turabian StyleNie, Lingdi, Liwen Jiang, John P. Quinn, Blair D. Grubb, and Minyan Wang. 2021. "TRPA1-Mediated Src Family Kinases Activity Facilitates Cortical Spreading Depression Susceptibility and Trigeminovascular System Sensitization" International Journal of Molecular Sciences 22, no. 22: 12273. https://doi.org/10.3390/ijms222212273





