Characterizing the NLRP3 Inflammasome in Mood Disorders: Overview, Technical Development, and Measures of Peripheral Activation in Adolescent Patients
Abstract
:1. Introduction
2. Results
2.1. Overview Demonstrates the Relevance of NLRP3 Inflammasome in Mood Disorders
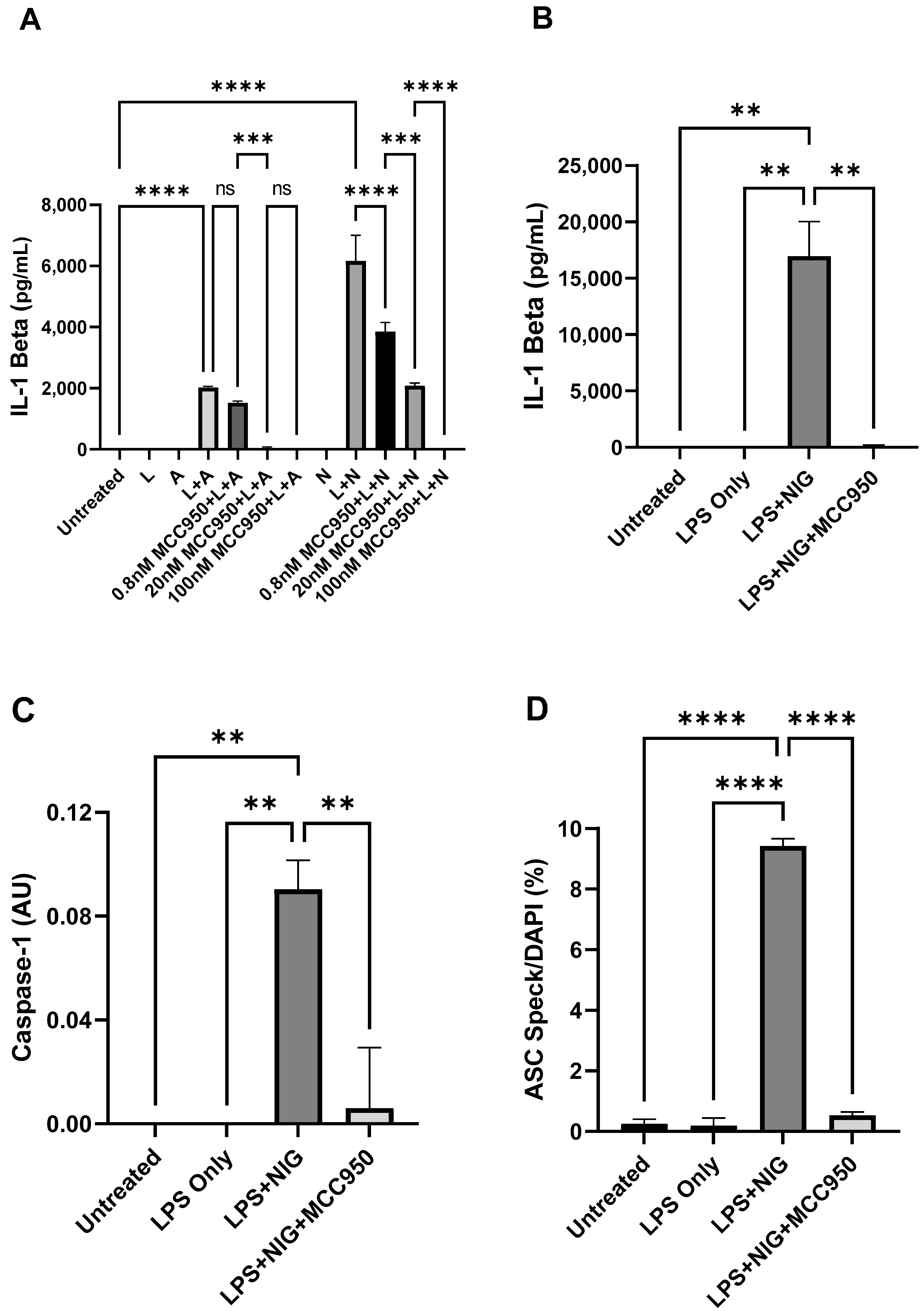
2.2. A Simple Immunofluorescence Assay to Measure ASC Speck in PBMC—A Way Forward to Characterize NLRP3 Activation in Disease
2.3. Assessment of NLRP3 Inflammasome Activation in Adolescent Patients with Mood Disorders
2.3.1. Activation of NLRP3 Inflammasome in Untreated PBMC Versus Treated PBMC
2.3.2. Comparison of NLRP3 Inflammasome in Adolescent Mood Disorder Patients over Time
3. Discussion
4. Materials and Methods
4.1. Overview of Evidence on Peripheral Activation of the NLRP3 Inflammasome
4.2. Development and Validation of a Biological Assay to Detect Activation of the NLRP3 Inflammasome
4.2.1. Cell Culture
4.2.2. Intracellular ASC Speck Formation
4.2.3. Protein Expression of Caspase-1
4.2.4. Protein Expression of IL-1 Beta
4.3. Assessment of Levels of NLRP3 Inflammasome Activation in Adolescent Patients with Mood Disorders
4.3.1. Patient Recruitment and Eligibility
4.3.2. Blood Collection and Isolation of Plasma and Peripheral Blood Mononuclear Cells
4.4. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jo, E.-K.; Kim, J.K.; Shin, D.-M.; Sasakawa, C.J.C. Molecular mechanisms regulating NLRP3 inflammasome activation. Cell. Mol. Immunol. 2016, 13, 148–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Broz, P.; Dixit, V.M. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 2016, 16, 407–420. [Google Scholar] [CrossRef]
- Paik, S.; Kim, J.K.; Silwal, P.; Sasakawa, C.; Jo, E.K. An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell. Mol. Immunol. 2021, 18, 1141–1160. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Callaway, J.B.; Ting, J.P. Inflammasomes: Mechanism of action, role in disease, and therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef] [Green Version]
- Stutz, A.; Kolbe, C.C.; Stahl, R.; Horvath, G.L.; Franklin, B.S.; van Ray, O.; Brinkschulte, R.; Geyer, M.; Meissner, F.; Latz, E. NLRP3 inflammasome assembly is regulated by phosphorylation of the pyrin domain. J. Exp. Med. 2017, 214, 1725–1736. [Google Scholar] [CrossRef]
- Martinon, F.; Mayor, A.; Tschopp, J. The inflammasomes: Guardians of the body. Annu. Rev. Immunol. 2009, 27, 229–265. [Google Scholar] [CrossRef] [Green Version]
- Subramanian, N.; Natarajan, K.; Clatworthy, M.R.; Wang, Z.; Germain, R.N. The adaptor MAVS promotes NLRP3 mitochondrial localization and inflammasome activation. Cell 2013, 153, 348–361. [Google Scholar] [CrossRef] [Green Version]
- Shimada, K.; Crother, T.R.; Karlin, J.; Dagvadorj, J.; Chiba, N.; Chen, S.; Ramanujan, V.K.; Wolf, A.J.; Vergnes, L.; Ojcius, D.M.; et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 2012, 36, 401–414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Zeng, M.Y.; Yang, D.; Motro, B.; Núñez, G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature 2016, 530, 354–357. [Google Scholar] [CrossRef] [Green Version]
- Faivre, J.M. From the Initial to the Definitive Scar; Soins Chirurgie: Paris, France, 1991; pp. 34–36. [Google Scholar]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolfes, V.; Ribeiro, L.S.; Hawwari, I.; Böttcher, L.; Rosero, N.; Maasewerd, S.; Santos, M.L.S.; Próchnicki, T.; Silva, C.M.S.; Wanderley, C.W.S.; et al. Platelets Fuel the Inflammasome Activation of Innate Immune Cells. Cell Rep. 2020, 31, 107–615. [Google Scholar] [CrossRef]
- Yang, J.; Liu, Z.; Xiao, T.S. Post-translational regulation of inflammasomes. Cell. Mol. Immunol. 2017, 14, 65–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, R.; Yazdi, A.S.; Menu, P.; Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011, 469, 221–225. [Google Scholar] [CrossRef]
- Colton, C.A.; Gilbert, D.L. Production of superoxide anions by a CNS macrophage, the microglia. FEBS Lett. 1987, 223, 284–288. [Google Scholar] [CrossRef] [Green Version]
- Gustin, A.; Kirchmeyer, M.; Koncina, E.; Felten, P.; Losciuto, S.; Heurtaux, T.; Tardivel, A.; Heuschling, P.; Dostert, C. NLRP3 inflammasome is expressed and functional in mouse brain microglia but not in astrocytes. PLoS ONE 2015, 10, e0130624. [Google Scholar]
- Koo, J.W.; Duman, R.S. IL-1β is an essential mediator of the antineurogenic and anhedonic effects of stress. Proc. Natl. Acad. Sci. USA 2008, 105, 751–756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mangan, M.S.J.; Olhava, E.J.; Roush, W.R.; Seidel, H.M.; Glick, G.D.; Latz, E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 2018, 17, 588–606. [Google Scholar] [CrossRef]
- Machado, A.K.; Pan, A.Y.; da Silva, T.M.; Duong, A.; Andreazza, A.C. Upstream pathways controlling mitochondrial function in major psychosis: A focus on bipolar disorder. Can. J. Psychiatry 2016, 61, 446–456. [Google Scholar] [CrossRef] [Green Version]
- Coll, R.C.; Robertson, A.A.; Chae, J.J.; Higgins, S.C.; Muñoz-Planillo, R.; Inserra, M.C.; Vetter, I.; Dungan, L.S.; Monks, B.G.; Stutz, A.; et al. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat. Med. 2015, 21, 248–255. [Google Scholar] [CrossRef] [Green Version]
- Zahid, A.; Li, B.; Kombe, A.J.K.; Jin, T.; Tao, J. Pharmacological Inhibitors of the NLRP3 Inflammasome. Front. Immunol. 2019, 10, 2538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutterwala, F.S.; Haasken, S.; Cassel, S.L. Mechanism of NLRP3 inflammasome activation. Ann. N. Y. Acad. Sci. 2014, 1319, 82. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Maker, G.L.; Hood, S.D.; Drummond, P.D. A review of peripheral biomarkers in major depression: The potential of inflammatory and oxidative stress biomarkers. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 48, 102–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alcocer-Gómez, E.; de Miguel, M.; Casas-Barquero, N.; Núñez-Vasco, J.; Sánchez-Alcazar, J.A.; Fernández-Rodríguez, A.; Cordero, M.D. NLRP3 inflammasome is activated in mononuclear blood cells from patients with major depressive disorder. Brain Behav. Immun. 2014, 36, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Momeni, M.; Ghorban, K.; Dadmanesh, M.; Khodadadi, H.; Bidaki, R.; Kazemi Arababadi, M.; Kennedy, D. ASC provides a potential link between depression and inflammatory disorders: A clinical study of depressed Iranian medical students. Nord. J. Psychiatry 2016, 70, 280–284. [Google Scholar] [CrossRef]
- Alcocer-Gómez, E.; Casas-Barquero, N.; Williams, M.R.; Romero-Guillena, S.L.; Cañadas-Lozano, D.; Bullón, P.; Sánchez-Alcazar, J.A.; Navarro-Pando, J.M.; Cordero, M.D. Antidepressants induce autophagy dependent-NLRP3-inflammasome inhibition in Major depressive disorder. Pharmacol. Res. 2017, 121, 114–121. [Google Scholar] [CrossRef]
- García-Álvarez, L.; Caso, J.R.; García-Portilla, M.P.; de la Fuente-Tomás, L.; González-Blanco, L.; Sáiz Martínez, P.; Leza, J.C.; Bobes, J. Regulation of inflammatory pathways in schizophrenia: A comparative study with bipolar disorder and healthy controls. Eur. Psychiatry 2018, 47, 50–59. [Google Scholar] [CrossRef]
- Scaini, G.; Barichello, T.; Fries, G.R.; Kennon, E.A.; Andrews, T.; Nix, B.R.; Zunta-Soares, G.; Valvassori, S.S.; Soares, J.C.; Quevedo, J. TSPO upregulation in bipolar disorder and concomitant downregulation of mitophagic proteins and NLRP3 inflammasome activation. Neuropsychopharmacology 2019, 44, 1291–1299. [Google Scholar] [CrossRef]
- Taene, A.; Khalili-Tanha, G.; Esmaeili, A.; Mobasheri, L.; Kooshkaki, O.; Jafari, S.; Shokouhifar, A.; Sarab, G.A. The Association of Major Depressive Disorder with Activation of NLRP3 Inflammasome, Lipid Peroxidation, and Total Antioxidant Capacity. J. Mol. Neurosci. 2020, 70, 65–70. [Google Scholar] [CrossRef]
- Li, Y.; Song, W.; Tong, Y.; Zhang, X.; Zhao, J.; Gao, X.; Yong, J.; Wang, H. Isoliquiritin ameliorates depression by suppressing NLRP3-mediated pyroptosis via miRNA-27a/SYK/NF-κB axis. J. Neuroinflamm. 2021, 18, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Brietzke, E.; Stabellini, R.; Grassi-Oliveira, R.; Lafer, B. Cytokines in bipolar disorder: Recent findings, deleterious effects but promise for future therapeutics. CNS Spectr. 2011, 16, 157–168. [Google Scholar] [CrossRef]
- Drexhage, R.C.; Knijff, E.M.; Padmos, R.C.; Heul-Nieuwenhuijzen, L.V.D.; Beumer, W.; Versnel, M.A.; Drexhage, H.A. The mononuclear phagocyte system and its cytokine inflammatory networks in schizophrenia and bipolar disorder. Expert Rev. Neurother. 2010, 10, 59–76. [Google Scholar] [CrossRef]
- Hope, S.; Dieset, I.; Agartz, I.; Steen, N.E.; Ueland, T.; Melle, I.; Aukrust, P.; Andreassen, O.A. Affective symptoms are associated with markers of inflammation and immune activation in bipolar disorders but not in schizophrenia. J. Psychiatr. Res. 2011, 45, 1608–1616. [Google Scholar] [CrossRef] [PubMed]
- Remlinger-Molenda, A.; Wojciak, P.; Michalak, M.; Karczewski, J.; Rybakowski, J.K. Selected cytokine profiles during remission in bipolar patients. Neuropsychobiology 2012, 66, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.S.; Harry, G.J.; Rapoport, S.I.; Kim, H.W. Increased excitotoxicity and neuroinflammatory markers in postmortem frontal cortex from bipolar disorder patients. Mol. Psychiatry 2010, 15, 384–392. [Google Scholar] [CrossRef]
- Söderlund, J.; Olsson, S.K.; Samuelsson, M.; Walther-Jallow, L.; Johansson, C.; Erhardt, S.; Landén, M.; Engberg, G.J. Elevation of cerebrospinal fluid interleukin-1β in bipolar disorder. J. Psychiatry Neurosci. JPN 2011, 36, 114. [Google Scholar] [CrossRef] [Green Version]
- Franklin, B.S.; Bossaller, L.; De Nardo, D.; Ratter, J.M.; Stutz, A.; Engels, G.; Brenker, C.; Nordhoff, M.; Mirandola, S.R.; Al-Amoudi, A.; et al. The adaptor ASC has extracellular and ‘prionoid’ activities that propagate inflammation. Nat. Immunol. 2014, 15, 727–737. [Google Scholar] [CrossRef] [Green Version]
- Fernandes-Alnemri, T.; Wu, J.; Yu, J.-W.; Datta, P.; Miller, B.; Jankowski, W.; Rosenberg, S.; Zhang, J.; Alnemri, E.S. Differentiation. The pyroptosome: A supramolecular assembly of ASC dimers mediating inflammatory cell death via caspase-1 activation. Cell Death Differ. 2007, 14, 1590–1604. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Oosting, M.; Deelen, P.; Ricaño-Ponce, I.; Smeekens, S.; Jaeger, M.; Matzaraki, V.; Swertz, M.A.; Xavier, R.J.; Franke, L.; et al. Inter-individual variability and genetic influences on cytokine responses to bacteria and fungi. Nat. Med. 2016, 22, 952–960. [Google Scholar] [CrossRef] [PubMed]



| Reference | Number of Patients | Sample | Assays | ||
|---|---|---|---|---|---|
| Healthy | BD/MDD | Medicated | |||
| Alcocer-Gómez et al., (2014) | 20 | 20 | 20 | PBMC, Serum | PCR, ELISA, Western Blot |
| Momeni et al., (2016) | 43 | 38 | PBMC | PCR | |
| Alcocer-Gómez et al., (2017) | 20 | 20 | 194 | PBMC, Serum | PCR, ELISA |
| García-Álvarez et al., (2018) | 80 | 102 | PBMC, Plasma | Western Blot | |
| Scaini et al., (2018) | 25 | 31 | PBMC | PCR, Western Blot | |
| Taene et al., (2020) | 20 | 20 | 20 | PBMC, Serum | PCR |
| Li et al., (2021) | 24 | 24 | Serum | PCR, ELISA | |
| Reference | NLRP3 | ASC | Caspase-1 | IL-1 beta | IL-18 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| mRNA | Protein | mRNA | Protein | mRNA | Protein | mRNA | Protein | mRNA | Protein | |
| Alcocer-Gómez et al., (2014) | Increase | Increase | Increase | Increase | Increase | |||||
| Momeni et al., (2016) | Increase | |||||||||
| Alcocer-Gómez et al., (2017) | Increase * | Increase | Increase | |||||||
| García-Álvarez et al., (2018) | Increase | |||||||||
| Scaini et al., (2018) | Increase | Increase | Increase | Increase | Increase | |||||
| Taene et al., (2020) | Increase | Increase | ||||||||
| Li et al., (2021) | Increase | Increase | Increase | Increase | Increase | |||||
| Total Mood Disorders | Bipolar Disorders | Depressive Disorders | ||||
|---|---|---|---|---|---|---|
| (n = 7) | (n = 2) | (n = 5) | ||||
| Age, M (SD) | 16.9 (1.68) | 17.5 (0.71) | 16.6 (1.95) | |||
| Sex, N (%) | ||||||
| Male | 2 (29%) | 0 (0%) | 2 (40%) | |||
| Female | 5 (71%) | 2 (100%) | 3 (60%) | |||
| Medication, N (%) | ||||||
| SSRI | 4 (57%) | 0 (0%) | 4 (80%) | |||
| SNRI | 1 (14%) | 1 (50%) | 0 (0%) | |||
| None | 2 (29%) | 1 (50%) | 1 (20%) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Fernando, S.M.; Pan, A.Y.; Laposa, R.; Cullen, K.R.; Klimes-Dougan, B.; Andreazza, A.C. Characterizing the NLRP3 Inflammasome in Mood Disorders: Overview, Technical Development, and Measures of Peripheral Activation in Adolescent Patients. Int. J. Mol. Sci. 2021, 22, 12513. https://doi.org/10.3390/ijms222212513
Zhou X, Fernando SM, Pan AY, Laposa R, Cullen KR, Klimes-Dougan B, Andreazza AC. Characterizing the NLRP3 Inflammasome in Mood Disorders: Overview, Technical Development, and Measures of Peripheral Activation in Adolescent Patients. International Journal of Molecular Sciences. 2021; 22(22):12513. https://doi.org/10.3390/ijms222212513
Chicago/Turabian StyleZhou, Xinyang, Shehan M. Fernando, Alexander Y. Pan, Rebecca Laposa, Kathryn R. Cullen, Bonnie Klimes-Dougan, and Ana C. Andreazza. 2021. "Characterizing the NLRP3 Inflammasome in Mood Disorders: Overview, Technical Development, and Measures of Peripheral Activation in Adolescent Patients" International Journal of Molecular Sciences 22, no. 22: 12513. https://doi.org/10.3390/ijms222212513






