Single-Cell Atlas of Adult Testis in Protogynous Hermaphroditic Orange-Spotted Grouper, Epinephelus coioides
Abstract
1. Introduction
2. Results
2.1. Transcriptome Profile of Adult Orange-Spotted Grouper Testis Tissue
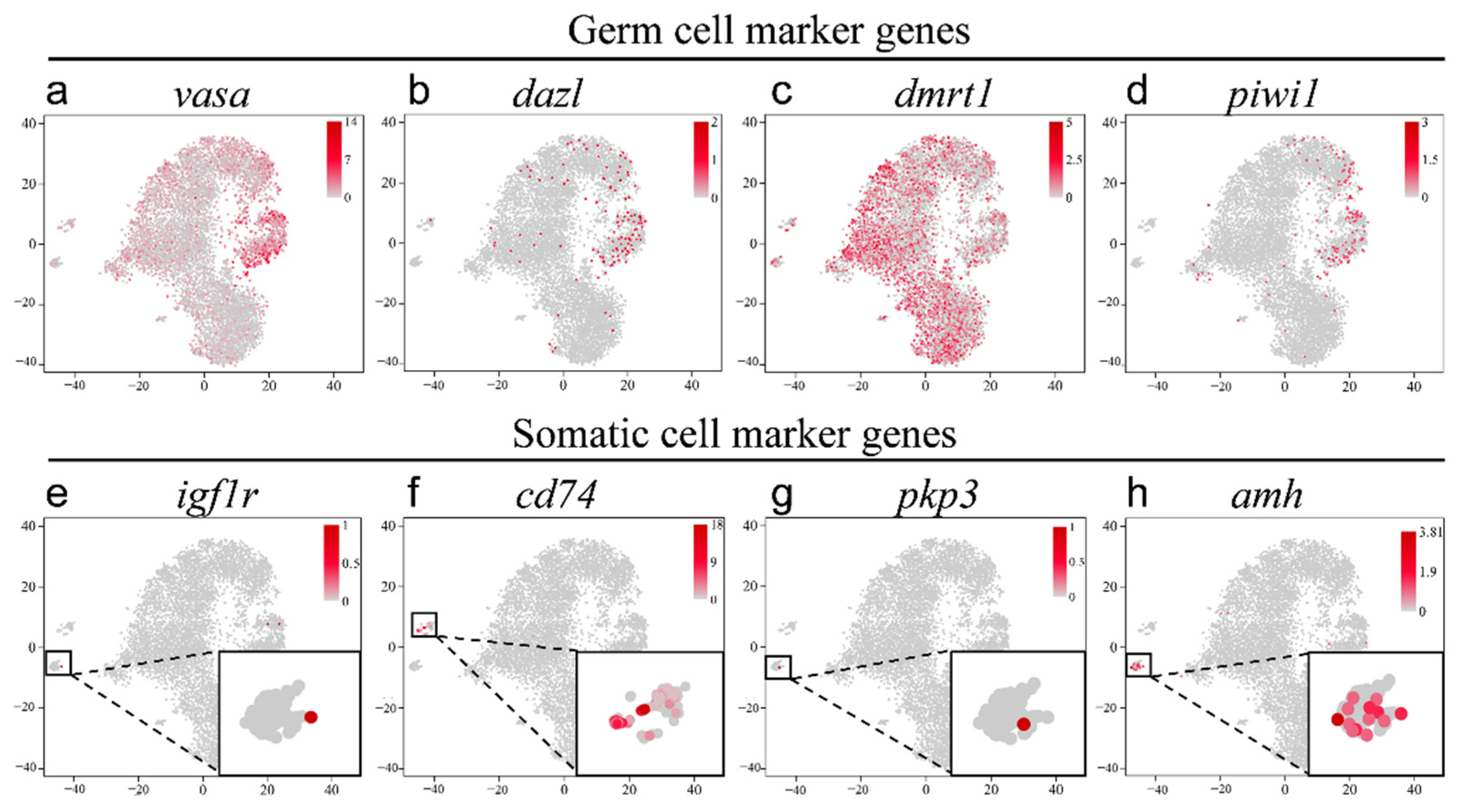
2.2. Validation of the Cell Types in scRNA-seq Data
2.3. Validation of the Known Marker Genes in Re-Clustering Clusters
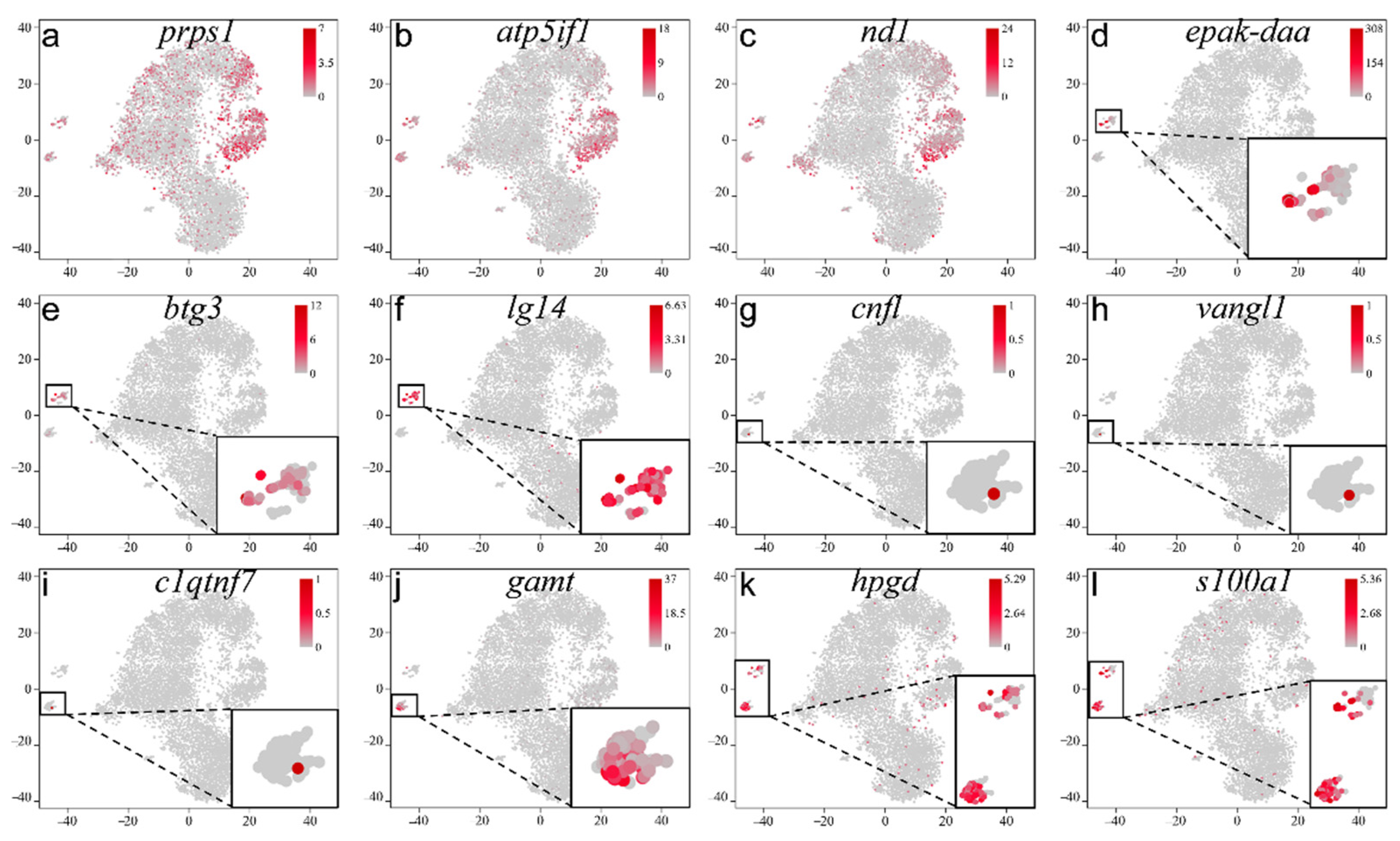
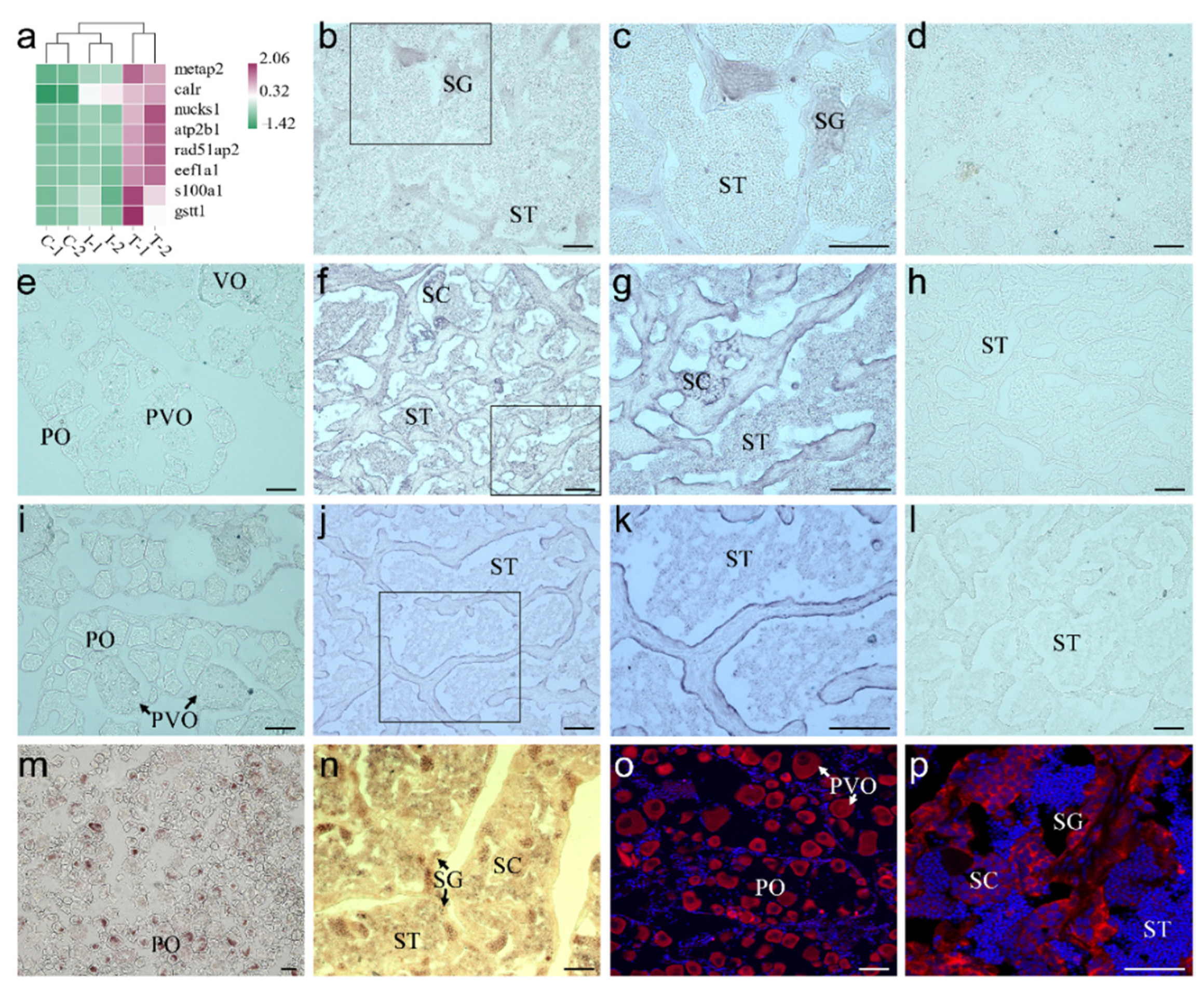
2.4. Screening and Identification of New Marker Genes
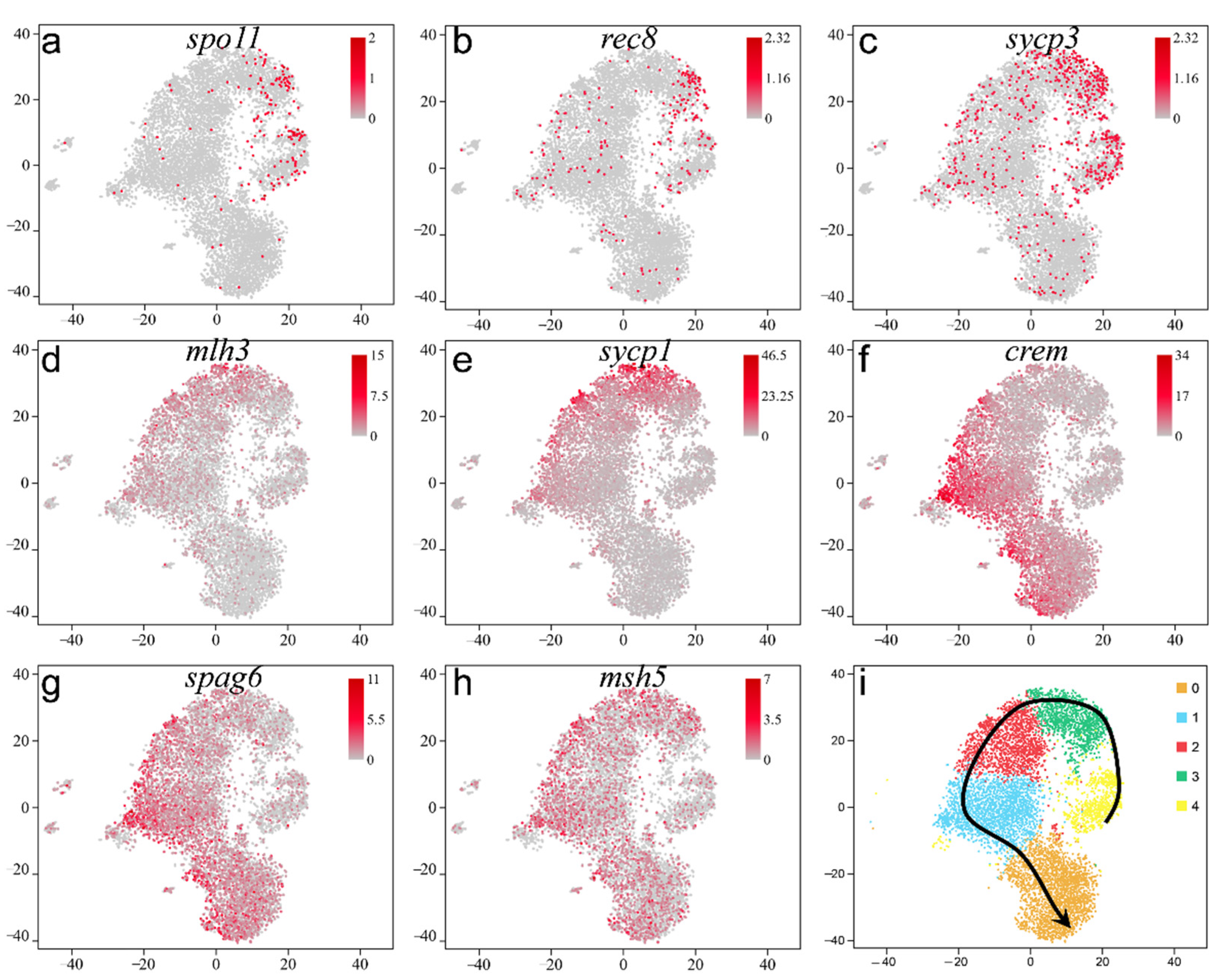
2.5. Pseudotime Trajectory of Spermatogenesis in Adult Orange-Spotted Grouper
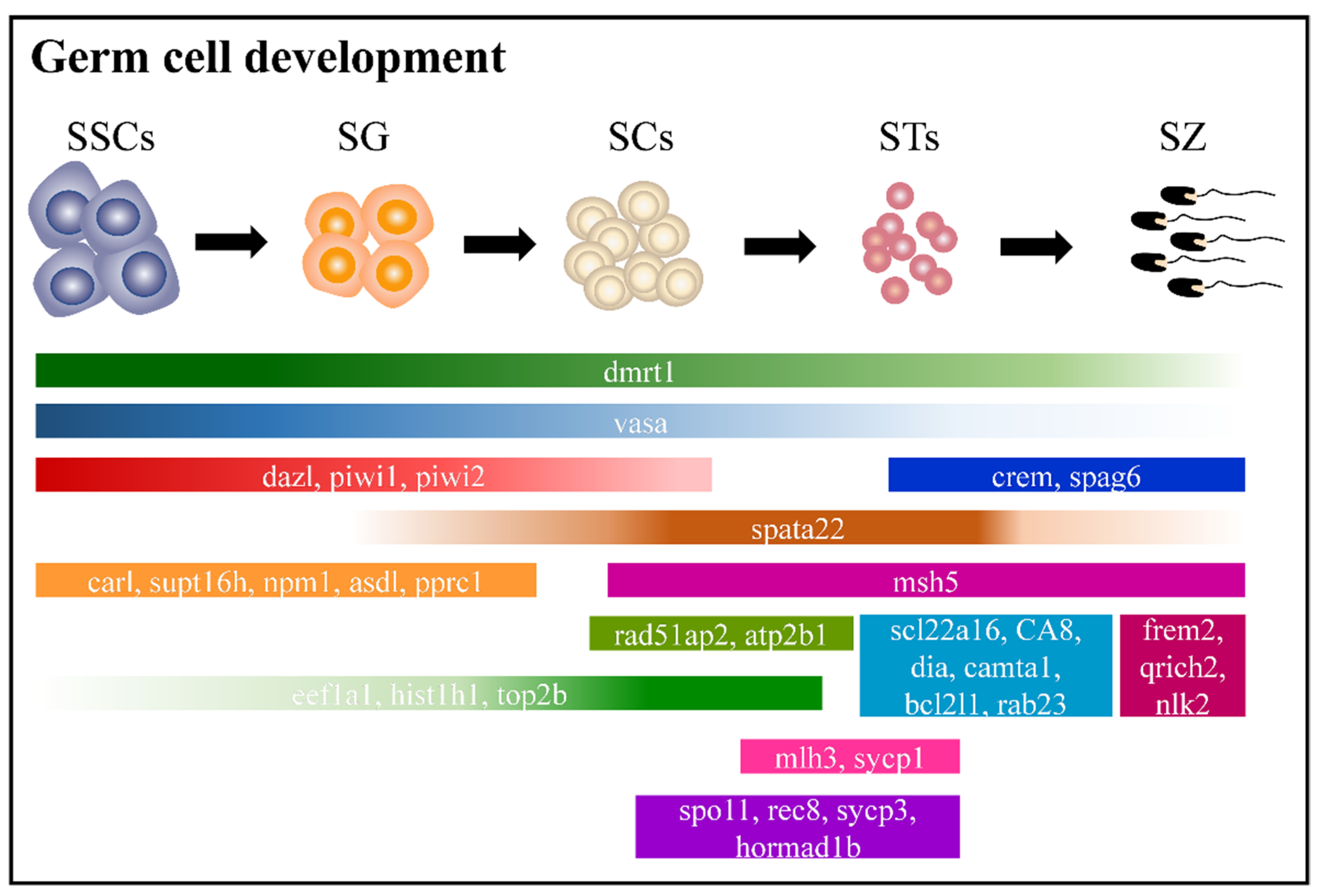
2.6. Adult Male Germ Cell Development in Orange-Spotted Grouper
3. Discussion
4. Materials and Methods
4.1. Animal
4.2. Hematoxylin-Eosin (H&E) Staining
4.3. The Preparation of Gonadal Single-Cell Suspension
4.4. Single Cell RNA-seq Library Construction and Sequencing
4.5. Process of Single Cell RNA-seq Data
4.6. Cell Clustering and Trajectory Analysis
4.7. Differential Expression Analysis
4.8. In Situ Hybridization (ISH)
4.9. Immunofluorescence (IF)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Kretser, D.M.; Loveland, K.L.; Meinhardt, A.; Simorangkir, D.; Wreford, N. Spermatogenesis. Hum. Reprod. 1998, 13 (Suppl. 1), 1–8. [Google Scholar] [CrossRef] [PubMed]
- Schulz, R.W.; de França, L.R.; Lareyre, J.-J.; Le Gac, F.; Chiarini-Garcia, H.; Nobrega, R.H.; Miura, T. Spermatogenesis in fish. Gen. Comp. Endocrinol. 2010, 165, 390–411. [Google Scholar] [CrossRef]
- Crespo, D.; Assis, L.H.C.; Furmanek, T.; Bogerd, J.; Schulz, R.W. Expression profiling identifies Sertoli and Leydig cell genes as Fsh targets in adult zebrafish testis. Mol. Cell. Endocrinol. 2016, 437, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Salva, A.; Hardy, M.P.; Wu, X.-F.; Sottas, C.M.; MacLaughlin, D.T.; Donahoe, P.K.; Lee, M.M. Müllerian-Inhibiting Substance Inhibits Rat Leydig Cell Regeneration after Ethylene Dimethanesulphonate Ablation1. Biol. Reprod. 2004, 70, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Schulz, R.W.; Menting, S.; Bogerd, J.; França, L.R.; Vilela, D.A.R.; Godinho, H.P. Sertoli Cell Proliferation in the Adult Testis—Evidence from Two Fish Species Belonging to Different Orders. Biol. Reprod. 2005, 73, 891–898. [Google Scholar] [CrossRef]
- DeFalco, T.; Potter, S.J.; Williams, A.V.; Waller, B.; Kan, M.J.; Capel, B. Macrophages Contribute to the Spermatogonial Niche in the Adult Testis. Cell Rep. 2015, 12, 1107–1119. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, S.; Meinhardt, A. The macrophages in testis function. J. Reprod. Immunol. 2017, 119, 107–112. [Google Scholar] [CrossRef]
- Lynn, A.; Soucek, R.; Börner, G.V. ZMM proteins during meiosis: Crossover artists at work. Chromosome Res. 2007, 15, 591–605. [Google Scholar] [CrossRef]
- Guo, J.; Grow, E.J.; Mlcochova, H.; Maher, G.J.; Lindskog, C.; Nie, X.; Guo, Y.; Takei, Y.; Yun, J.; Cai, L.; et al. The adult human testis transcriptional cell atlas. Cell Res. 2018, 28, 1141–1157. [Google Scholar] [CrossRef]
- Sohni, A.; Tan, K.; Song, H.W.; Burow, D.; de Rooij, D.G.; Laurent, L.; Hsieh, T.C.; Rabah, R.; Hammoud, S.S.; Vicini, E.; et al. The Neonatal and Adult Human Testis Defined at the Single-Cell Level. Cell Rep. 2019, 26, 1501–1517.e4. [Google Scholar] [CrossRef]
- Shami, A.N.; Zheng, X.; Munyoki, S.K.; Ma, Q.; Manske, G.L.; Green, C.D.; Sukhwani, M.; Orwig, K.E.; Li, J.Z.; Hammoud, S.S. Single-Cell RNA Sequencing of Human, Macaque, and Mouse Testes Uncovers Conserved and Divergent Features of Mammalian Spermatogenesis. Dev. Cell 2020, 54, 529–547.e12. [Google Scholar] [CrossRef]
- Yang, H.; Ma, J.; Wan, Z.; Wang, Q.; Wang, Z.; Zhao, J.; Wang, F.; Zhang, Y. Characterization of sheep spermatogenesis through single-cell RNA sequencing. FASEB J. 2021, 35, e21187. [Google Scholar] [CrossRef] [PubMed]
- Devlin, R.H.; Nagahama, Y. Sex determination and sex differentiation in fish an overview of genetic, physiological, and environmental influences. Aquaculture 2002, 208, 191–364. [Google Scholar] [CrossRef]
- Liu, M.; de Mitcheson, Y.S. Gonad development during sexual differentiation in hatchery-produced orange-spotted grouper (Epinephelus coioides) and humpback grouper (Cromileptes altivelis) (Pisces: Serranidae, Epinephelinae). Aquaculture 2009, 287, 191–202. [Google Scholar] [CrossRef]
- Wu, X.; Yang, Y.; Zhong, C.; Guo, Y.; Li, S.; Lin, H.; Liu, X. Transcriptome profiling of laser-captured germ cells and functional characterization of zbtb40 during 17alpha-methyltestosterone-induced spermatogenesis in orange-spotted grouper (Epinephelus coioides). BMC Genom. 2020, 21, 73. [Google Scholar] [CrossRef]
- McDavid, A.; Finak, G.; Chattopadyay, P.K.; Dominguez, M.; Lamoreaux, L.; Ma, S.S.; Roederer, M.; Gottardo, R. Data exploration, quality control and testing in single-cell qPCR-based gene expression experiments. Bioinformatics 2013, 29, 461–467. [Google Scholar] [CrossRef]
- Raz, E. Guidance of primordial germ cell migration. Curr. Opin. Cell Biol. 2004, 16, 169–173. [Google Scholar] [CrossRef]
- Aran, D.; Looney, A.P.; Liu, L.; Wu, E.; Fong, V.; Hsu, A.; Chak, S.; Naikawadi, R.P.; Wolters, P.J.; Abate, A.R.; et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat. Immunol. 2019, 20, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Wu, X.; Liu, M.; Zhong, C.; Xu, H.; Li, S.; Lin, H.; Liu, X. Identification and characterization of germ cell genes vasa and dazl in a protogynous hermaphrodite fish, orange-spotted grouper (Epinephelus coioides). Gene Expr. Patterns GEP 2020, 35, 119095. [Google Scholar] [CrossRef] [PubMed]
- Perrot, V.; Moiseeva, E.B.; Gozes, Y.; Chan, S.J.; Funkenstein, B. Insulin-Like Growth Factor Receptors and Their Ligands in Gonads of a Hermaphroditic Species, the Gilthead Seabream (Sparus aurata): Expression and Cellular Localization1. Biol. Reprod. 2000, 63, 229–241. [Google Scholar] [CrossRef][Green Version]
- Wu, G.C.; Li, H.W.; Tey, W.G.; Lin, C.J.; Chang, C.F. Expression profile of amh/Amh during bi-directional sex change in the protogynous orange-spotted grouper Epinephelus coioides. PLoS ONE 2017, 12, e0185864. [Google Scholar] [CrossRef] [PubMed]
- De Vries, S.S.; Baart, E.B.; Dekker, M.; Siezen, A.; de Rooij, D.G.; de Boer, P.; te Riele, H. Mouse MutS-like protein Msh5 is required for proper chromosome synapsis in male and female meiosis. Genes Dev. 1999, 13, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Nobrega, R.H.; Batlouni, S.R.; Franca, L.R. An overview of functional and stereological evaluation of spermatogenesis and germ cell transplantation in fish. Fish Physiol. Biochem. 2009, 35, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Diaz, V.D.; Hermann, B.P. What has single-cell RNA-seq taught us about mammalian spermatogenesis? Biol. Reprod. 2019, 101, 617–634. [Google Scholar] [CrossRef]
- Zhou, L.; Gui, J.F. Molecular mechanisms underlying sex change in hermaphroditic groupers. Fish Physiol. Biochem. 2010, 36, 181–193. [Google Scholar] [CrossRef]
- Lasko, P.F.; Ashburner, M. The product of the Drosophila gene vasa is very similar to eukaryotic initiation factor-4A. Nature 1988, 335, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Diehl-Jones, W.; Lasko, P. Localization of vasa protein to the Drosophila pole plasm is independent of its RNA-binding and helicase activities. Development 1994, 120, 1201–1211. [Google Scholar] [CrossRef]
- Braat, A.K.; van de Water, S.; Goos, H.; Bogerd, J.; Zivkovic, D. Vasa protein expression and localization in the zebrafish. Mech. Dev. 2000, 95, 271–274. [Google Scholar] [CrossRef]
- Knaut, H.; Pelegri, F.; Bohmann, K.; Schwarz, H.; Nüsslein-Volhard, C. Zebrafish vasa RNA but not its protein is a component of the germ plasm and segregates asymmetrically before germline specification. J. Cell. Biol. 2000, 149, 875–888. [Google Scholar] [CrossRef]
- Leu, D.H.; Draper, B.W. The ziwi promoter drives germline-specific gene expression in zebrafish. Dev. Dyn. 2010, 239, 2714–2721. [Google Scholar] [CrossRef]
- Houston, D.W.; King, M.L. A critical role for Xdazl, a germ plasm-localized RNA, in the differentiation of primordial germ cells in Xenopus. Development 2000, 127, 447–456. [Google Scholar] [CrossRef]
- Li, M.; Zhu, F.; Li, Z.; Hong, N.; Hong, Y. Dazl is a critical player for primordial germ cell formation in medaka. Sci. Rep. 2016, 6, 28317. [Google Scholar] [CrossRef]
- Smorag, L.; Xu, X.; Engel, W.; Pantakani, D.V. The roles of DAZL in RNA biology and development. Wiley Interdiscip. Rev. RNA 2014, 5, 527–535. [Google Scholar] [CrossRef]
- Dwarakanath, M.; Lim, M.; Xu, H.; Hong, Y. Differential expression of boule and dazl in adult germ cells of the Asian seabass. Gene 2014, 549, 237–242. [Google Scholar] [CrossRef]
- Ketting, R.F. The Many Faces of RNAi. Dev. Cell 2011, 20, 148–161. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Yang, Y.; Zhong, C.; Guo, Y.; Wei, T.; Li, S.; Lin, H.; Liu, X. Integration of ATAC-seq and RNA-seq Unravels Chromatin Accessibility during Sex Reversal in Orange-Spotted Grouper (Epinephelus coioides). Int. J. Mol. Sci. 2020, 21, 2800. [Google Scholar] [CrossRef] [PubMed]
- Gold, L.I.; Eggleton, P.; Sweetwyne, M.T.; Van Duyn, L.B.; Greives, M.R.; Naylor, S.-M.; Michalak, M.; Murphy-Ullrich, J.E. Calreticulin: Non-endoplasmic reticulum functions in physiology and disease. FASEB J. 2010, 24, 665–683. [Google Scholar] [CrossRef] [PubMed]
- Michalak, M.; Groenendyk, J.; Szabo, E.; Gold, L.I.; Opas, M. Calreticulin, a multi-process calcium-buffering chaperone of the endoplasmic reticulum. Biochem. J. 2009, 417, 651–666. [Google Scholar] [CrossRef] [PubMed]
- Gold, L.; Williams, D.; Groenendyk, J.; Michalak, M.; Eggleton, P. Unfolding the complexities of ER chaperones in health and disease: Report on the 11th international calreticulin workshop. Cell Stress Chaperones 2015, 20, 875–883. [Google Scholar] [CrossRef]
- Pandya, U.M.; Manzanares, M.A.; Tellechea, A.; Egbuta, C.; Daubriac, J.; Jimenez-Jaramillo, C.; Samra, F.; Fredston-Hermann, A.; Saadipour, K.; Gold, L.I. Calreticulin exploits TGF-β for extracellular matrix induction engineering a tissue regenerative process. FASEB J. 2020, 34, 15849–15874. [Google Scholar] [CrossRef] [PubMed]
- Borradaile, N.M.; Buhman, K.K.; Listenberger, L.L.; Magee, C.J.; Morimoto, E.T.A.; Ory, D.S.; Schaffer, J.E. A critical role for eukaryotic elongation factor 1A-1 in lipotoxic cell death. Mol. Biol. Cell 2006, 17, 770–778. [Google Scholar] [CrossRef]
- Gross, S.R.; Kinzy, T.G. Translation elongation factor 1A is essential for regulation of the actin cytoskeleton and cell morphology. Nat. Struct. Mol. Biol. 2005, 12, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Hotokezaka, Y.; Többen, U.; Hotokezaka, H.; van Leyen, K.; Beatrix, B.; Smith, D.H.; Nakamura, T.; Wiedmann, M. Interaction of the Eukaryotic Elongation Factor 1A with Newly Synthesized Polypeptides. J. Biol. Chem. 2002, 277, 18545–18551. [Google Scholar] [CrossRef]
- Mateyak, M.K.; Kinzy, T.G. eEF1A: Thinking outside the ribosome. J. Biol. Chem. 2010, 285, 21209–21213. [Google Scholar] [CrossRef]
- Diaz-Romero, J.; Nesic, D. S100A1 and S100B: Calcium Sensors at the Cross-Roads of Multiple Chondrogenic Pathways. J. Cell. Physiol. 2017, 232, 1979–1987. [Google Scholar] [CrossRef]
- Guo, Y.; Cui, L.; Jiang, S.; Wang, D.; Jiang, S.; Xie, C.; Jia, Y. S100A1 transgenic treatment of acute heart failure causes proteomic changes in rats. Mol. Med. Rep. 2016, 14, 1538–1552. [Google Scholar] [CrossRef][Green Version]
- Scott, C.E.; Kekenes-Huskey, P.M. Molecular Basis of S100A1 Activation at Saturating and Subsaturating Calcium Concentrations. Biophys. J. 2016, 110, 1052–1063. [Google Scholar] [CrossRef][Green Version]
- Geisinger, A.; Rodríguez-Casuriaga, R.; Benavente, R. Transcriptomics of Meiosis in the Male Mouse. Front. Cell Dev. Biol. 2021, 9, 626020. [Google Scholar] [CrossRef] [PubMed]
- Baudat, F.; Imai, Y.; de Massy, B. Meiotic recombination in mammals: Localization and regulation. Nat. Rev. Genet. 2013, 14, 794–806. [Google Scholar] [CrossRef]
- Hou, H.; Kyriacou, E.; Thadani, R.; Klutstein, M.; Chapman, J.H.; Cooper, J.P. Centromeres are dismantled by foundational meiotic proteins Spo11 and Rec8. Nature 2021, 591, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Pannafino, G.; Alani, E. Coordinated and Independent Roles for MLH Subunits in DNA Repair. Cells 2021, 10, 948. [Google Scholar] [CrossRef] [PubMed]
- Mhaskar, A.N.; Koornneef, L.; Zelensky, A.N.; Houtsmuller, A.B.; Baarends, W.M. High Resolution View on the Regulation of Recombinase Accumulation in Mammalian Meiosis. Front. Cell Dev. Biol. 2021, 9, 672191. [Google Scholar] [CrossRef]
- Kleene, K.C. Connecting cis-elements and trans-factors with mechanisms of developmental regulation of mRNA translation in meiotic and haploid mammalian spermatogenic cells. Reproduction 2013, 146, R1–R19. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Saito, K.; Takemoto, K.; Velilla, F.; Kawasaki, T.; Ishiguro, K.-i.; Sakai, N. Sycp1 Is Not Required for Subtelomeric DNA Double-Strand Breaks but Is Required for Homologous Alignment in Zebrafish Spermatocytes. Front. Cell Dev. Biol. 2021, 9, 664377. [Google Scholar] [CrossRef]
- Blondel, V.D.; Guillaume, J.-L.; Lambiotte, R.; Lefebvre, E. Fast unfolding of communities in large networks. J. Stat. Mech. Theory Exp. 2008, 2008, P10008. [Google Scholar] [CrossRef]
- Trozzi, F.; Wang, X.; Tao, P. UMAP as a Dimensionality Reduction Tool for Molecular Dynamics Simulations of Biomacromolecules: A Comparison Study. J. Phys. Chem. B 2021, 125, 5022–5034. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Qu, L.; Li, S.; Guo, Y.; He, J.; Liu, M.; Liu, X.; Lin, H. Molecular characterization and expression patterns of stem-loop binding protein (SLBP) genes in protogynous hermaphroditic grouper, Epinephelus coioides. Gene 2019, 700, 120–130. [Google Scholar] [CrossRef]
- Xu, H.; Gui, J.; Hong, Y. Differential expression of vasa RNA and protein during spermatogenesis and oogenesis in the gibel carp (Carassius auratus gibelio), a bisexually and gynogenetically reproducing vertebrate. Dev. Dyn. 2005, 233, 872–882. [Google Scholar] [CrossRef]



| Primers | Purpose | Sequence (from 5′ to 3′) |
|---|---|---|
| calr-F | ISH | GACGCCACCGTCTACTTCAA |
| calr-R | ISH | GTCGTCCCAGTCACTTGGTT |
| eef1a-F | ISH | AAGGGCTGGAAGATCAACCG |
| eef1a-R | ISH | TTAATCACTCCCACGGCCAC |
| s100a1-F | ISH | AAAGCCCAGAAGAACCCCAA |
| s100a1-R | ISH | CGACGAGGGGAAGAAACTCT |
| vasa-F | ISH | GAGCCTGAGACCATCATC |
| vasa-R | ISH | AGGACTCTTCACACTGTTG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.; Yang, Y.; Zhong, C.; Wang, T.; Deng, Y.; Huang, H.; Lin, H.; Meng, Z.; Liu, X. Single-Cell Atlas of Adult Testis in Protogynous Hermaphroditic Orange-Spotted Grouper, Epinephelus coioides. Int. J. Mol. Sci. 2021, 22, 12607. https://doi.org/10.3390/ijms222212607
Wu X, Yang Y, Zhong C, Wang T, Deng Y, Huang H, Lin H, Meng Z, Liu X. Single-Cell Atlas of Adult Testis in Protogynous Hermaphroditic Orange-Spotted Grouper, Epinephelus coioides. International Journal of Molecular Sciences. 2021; 22(22):12607. https://doi.org/10.3390/ijms222212607
Chicago/Turabian StyleWu, Xi, Yang Yang, Chaoyue Zhong, Tong Wang, Yanhong Deng, Hengjin Huang, Haoran Lin, Zining Meng, and Xiaochun Liu. 2021. "Single-Cell Atlas of Adult Testis in Protogynous Hermaphroditic Orange-Spotted Grouper, Epinephelus coioides" International Journal of Molecular Sciences 22, no. 22: 12607. https://doi.org/10.3390/ijms222212607
APA StyleWu, X., Yang, Y., Zhong, C., Wang, T., Deng, Y., Huang, H., Lin, H., Meng, Z., & Liu, X. (2021). Single-Cell Atlas of Adult Testis in Protogynous Hermaphroditic Orange-Spotted Grouper, Epinephelus coioides. International Journal of Molecular Sciences, 22(22), 12607. https://doi.org/10.3390/ijms222212607






