Alteration of Extracellular Matrix Components in the Anterior Pituitary Gland of Neonatal Rats Induced by a Maternal Bisphenol A Diet during Pregnancy
Abstract
:1. Introduction
2. Results
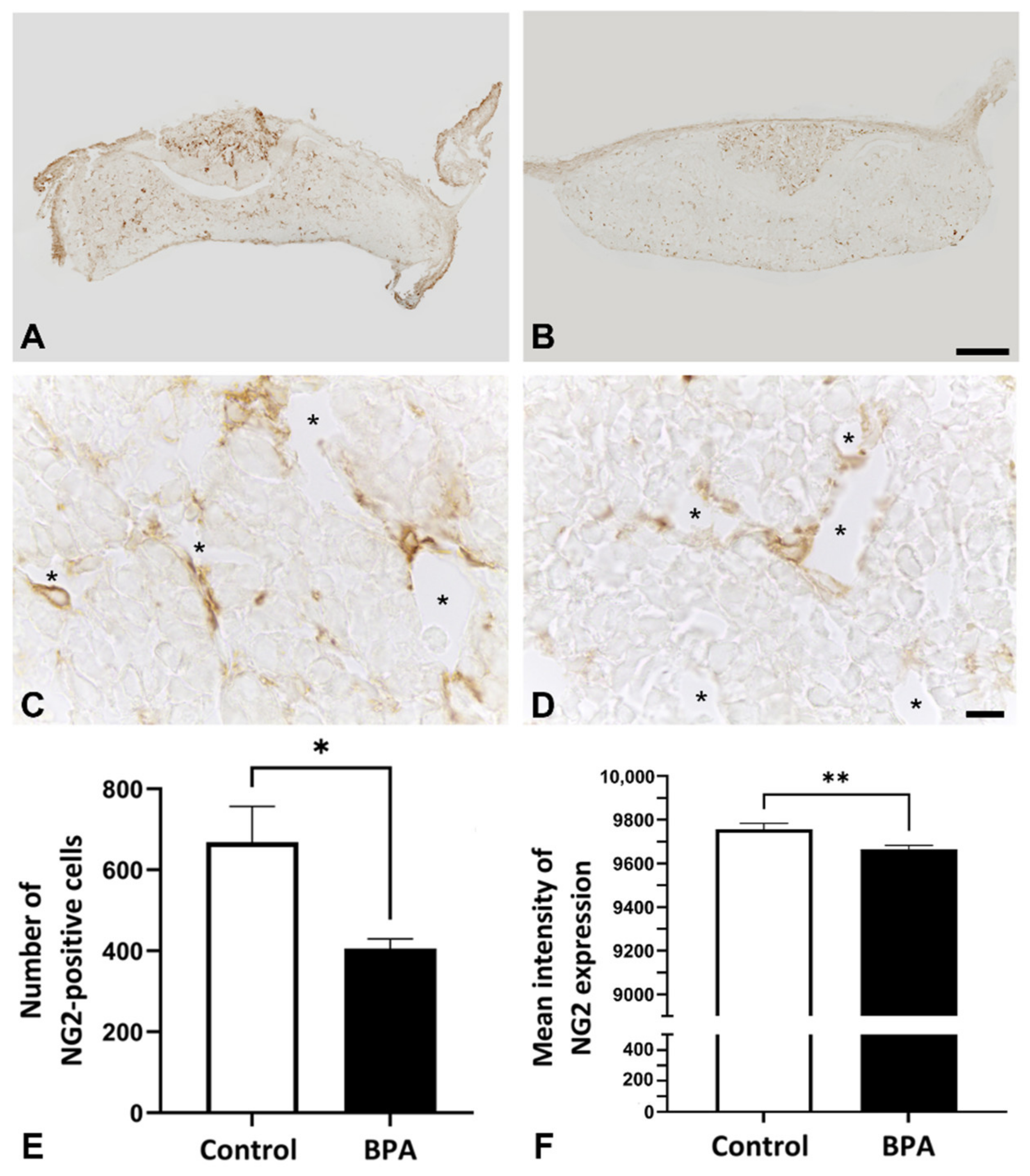
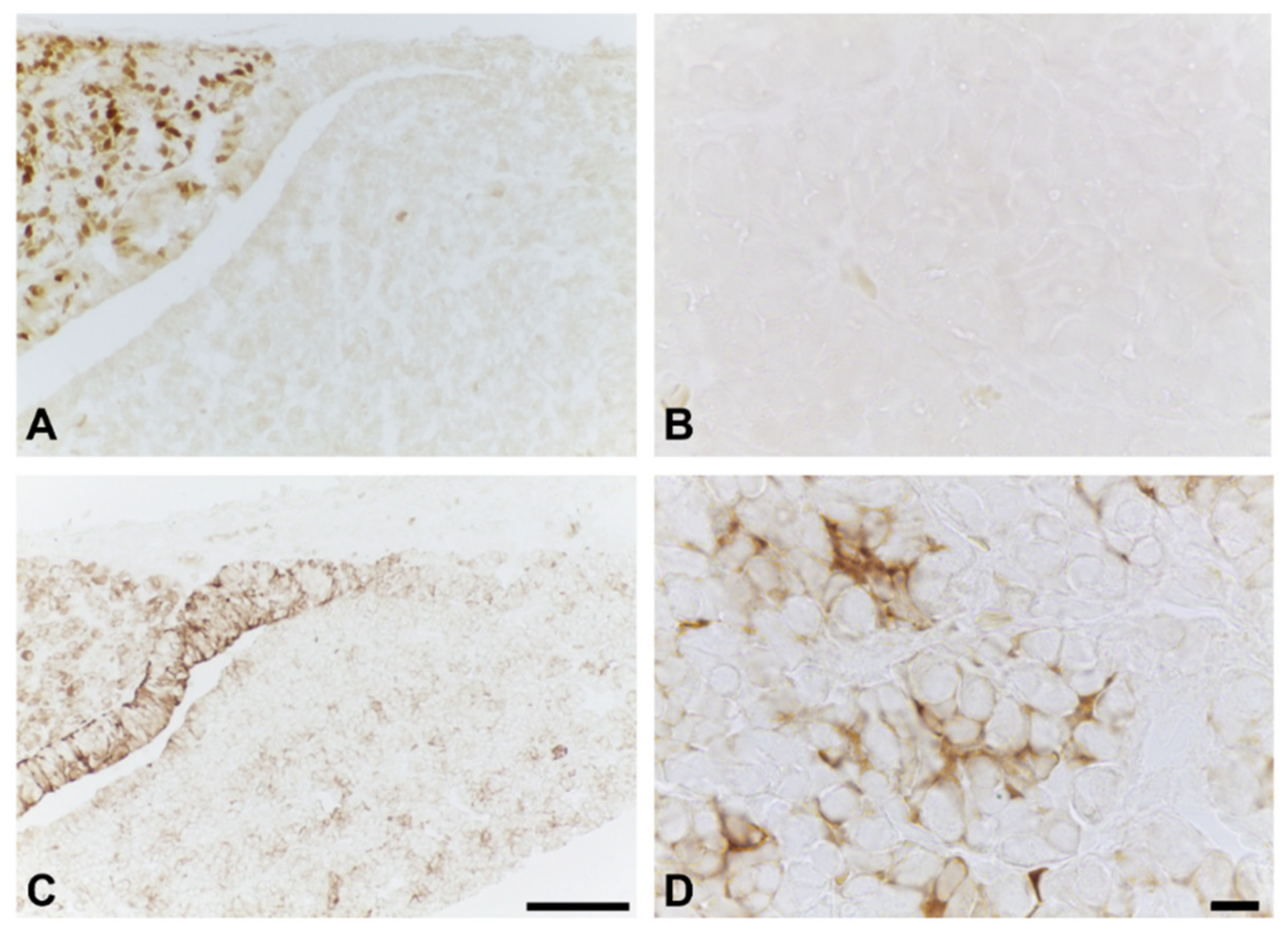
2.1. Effects of BPA at the NOAEL Level on ECM-Producing Cells
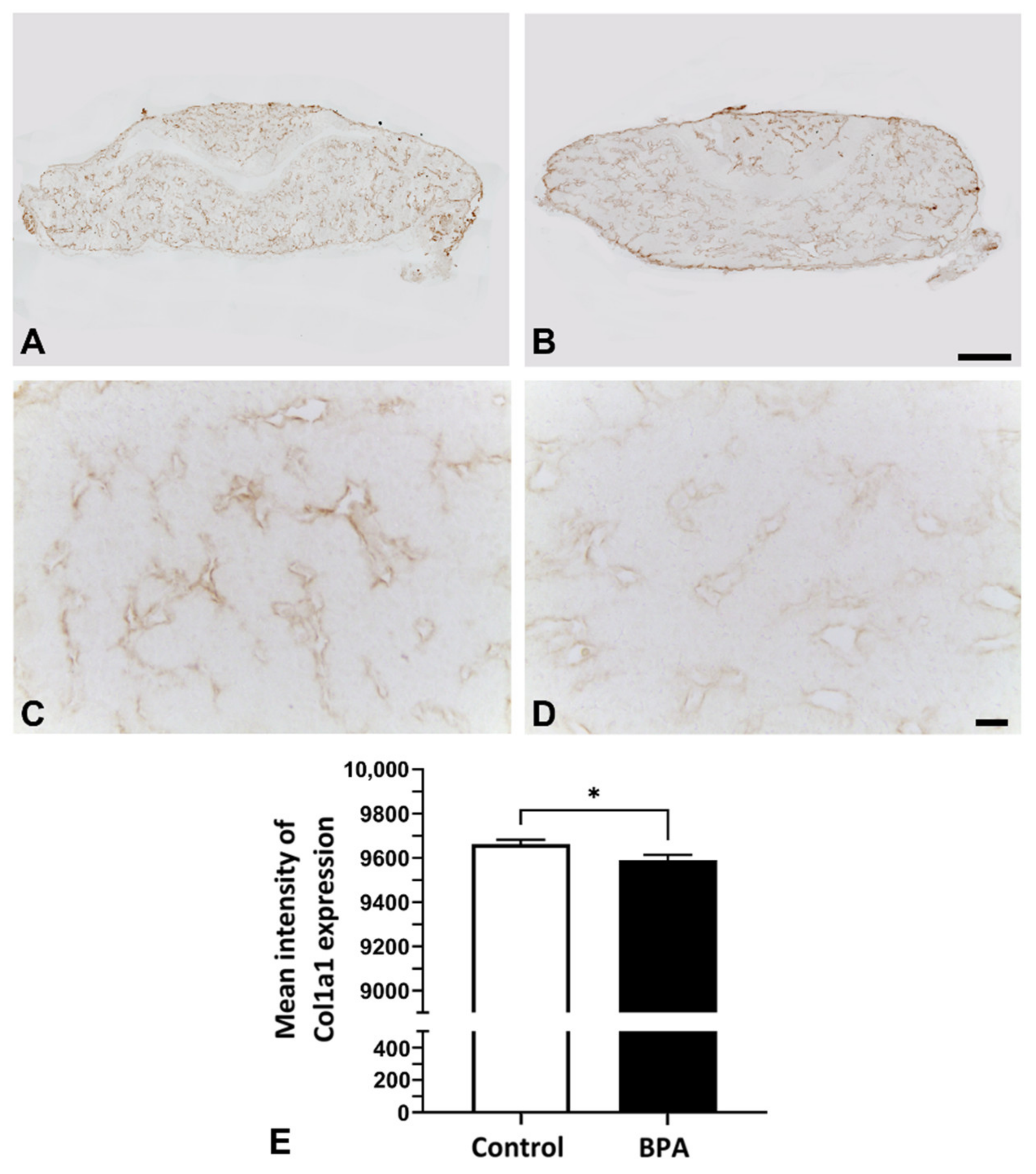
2.2. Alterations of ECM Components Focusing on Collagens
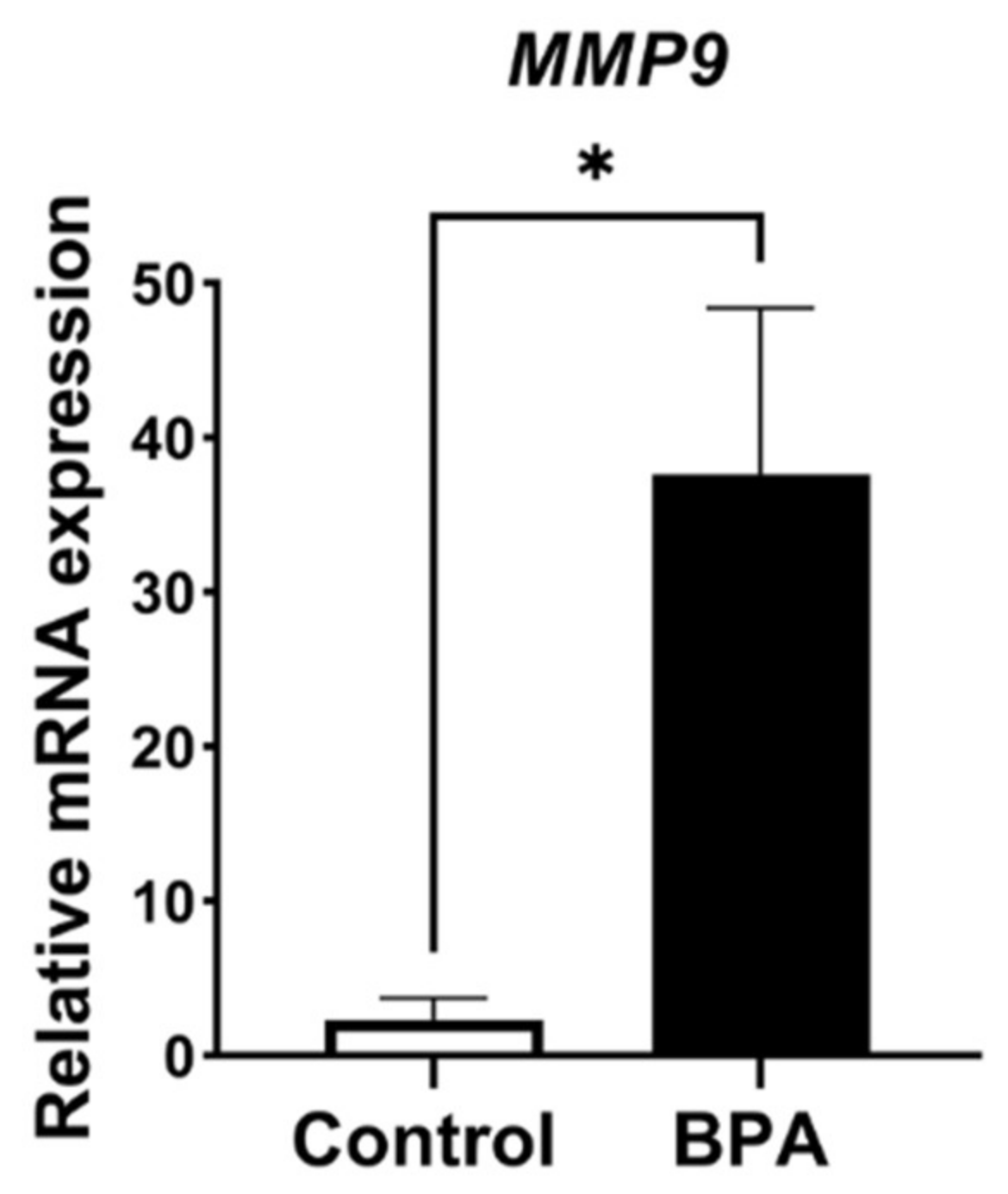
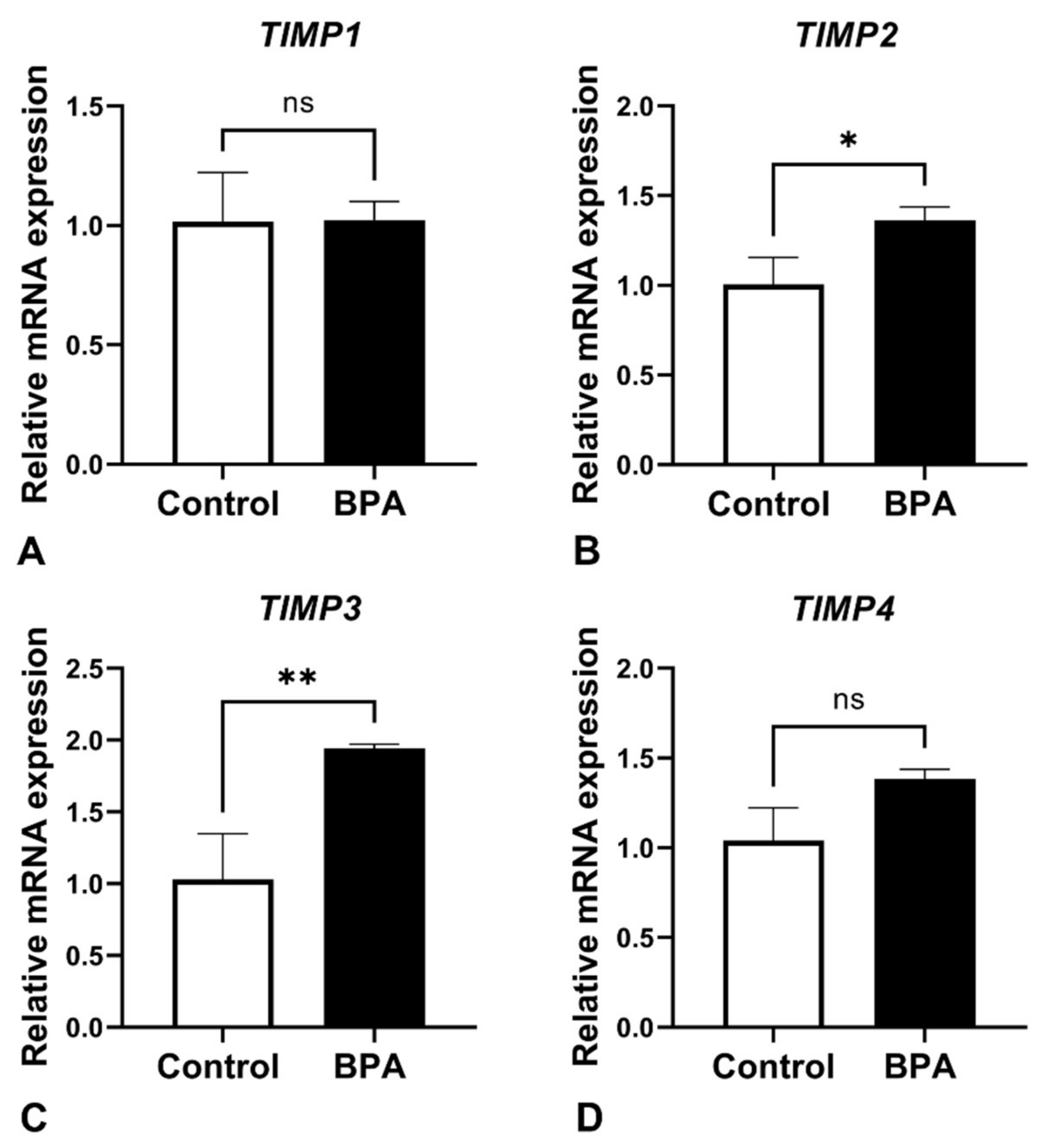
2.3. Alterations of ECM Balancing Regulators
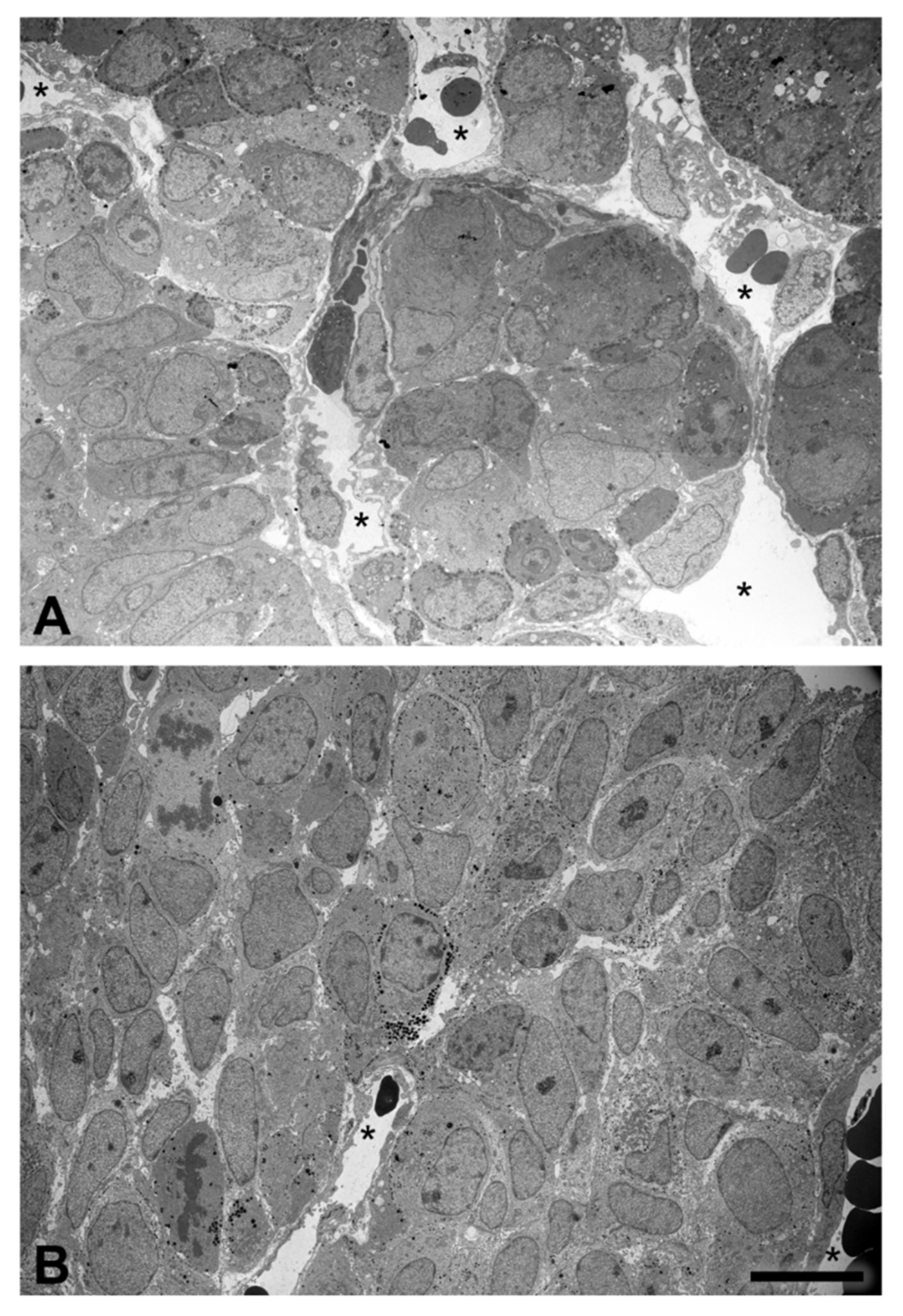
2.4. Tissue Organization Observed by Transmission Electron Microscopy
3. Discussion
4. Materials and Methods
4.1. Animal and Treatment
4.2. Tissue Collection
4.3. Immunohistochemistry
4.4. Determination of mRNA Expression by RT-qPCR
4.5. Transmission Electron Microscopic (TEM)
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gould, J.C.; Leonard, L.S.; Maness, S.C.; Wagner, B.L.; Conner, K.; Zacharewski, T.; Safe, S.; McDonnell, D.P.; Gaido, K.W. Bisphenol A interacts with the estrogen receptor alpha in a distinct manner from estradiol. Mol. Cell. Endocrinol. 1998, 142, 203–214. [Google Scholar] [CrossRef]
- Campanale, C.; Massarelli, C.; Savino, I.; Locaputo, V.; Uricchio, V.F. A detailed review study on potential effects of microplastics and additives of concern on human health. Int. J. Environ. Res. Public Health 2020, 17, 1212. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, O.; Oishi, S. Disposition of orally administered 2,2-Bis(4-hydroxyphenyl)propane (Bisphenol A) in pregnant rats and the placental transfer to fetuses. Environ. Health Perspect. 2000, 108, 931–935. [Google Scholar] [CrossRef]
- Cabaton, N.J.; Wadia, P.R.; Rubin, B.S.; Zalko, D.; Schaeberle, C.M.; Askenase, M.H.; Gadbois, J.L.; Tharp, A.P.; Whitt, G.S.; Sonnenschein, C.; et al. Perinatal exposure to environmentally relevant levels of bisphenol A decreases fertility and fecundity in CD-1 mice. Environ. Health Perspect. 2011, 119, 547–552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubin, B.S.; Murray, M.K.; Damassa, D.A.; King, J.C.; Soto, A.M. Perinatal exposure to low doses of bisphenol A affects body weight, patterns of estrous cyclicity, and plasma LH levels. Environ. Health Perspect. 2001, 109, 675–680. [Google Scholar] [CrossRef]
- Jindatip, D.; Poh, R.W.; Fujiwara, K. Insight into the Characteristics of novel desmin-immunopositive perivascular cells of the anterior pituitary gland using transmission and focused ion beam scanning electron microscopy. Int. J. Mol. Sci. 2021, 22, 8630. [Google Scholar] [CrossRef]
- Soji, T.; Herbert, D.C. Intercellular communication between rat anterior pituitary cells. Anat. Rec. 1989, 224, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, K.; Jindatip, D.; Kikuchi, M.; Yashiro, T. In situ hybridization reveals that type I and III collagens are produced by pericytes in the anterior pituitary gland of rats. Cell Tissue Res. 2010, 342, 491–495. [Google Scholar] [CrossRef]
- Tsukada, T.; Yoshida, S.; Kito, K.; Fujiwara, K.; Yako, H.; Horiguchi, K.; Isowa, Y.; Yashiro, T.; Kato, T.; Kato, Y. TGFβ signaling reinforces pericyte properties of the non-endocrine mouse pituitary cell line TtT/GF. Cell Tissue Res. 2018, 371, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Ilmiawati, C.; Horiguchi, K.; Fujiwara, K.; Yashiro, T. Matrix metalloproteinase-9 expression in folliculostellate cells of rat anterior pituitary gland. J. Endocrinol. 2012, 212, 363–370. [Google Scholar] [CrossRef]
- Azuma, M.; Tofrizal, A.; Maliza, R.; Batchuluun, K.; Ramadhani, D.; Syaidah, R.; Tsukada, T.; Fujiwara, K.; Kikuchi, M.; Horiguchi, K.; et al. Maintenance of the extracellular matrix in rat anterior pituitary gland: Identification of cells expressing tissue inhibitors of metalloproteinases. Acta Histochem. Cytochem. 2015, 48, 185–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elswefy, S.E.; Abdallah, F.R.; Atteia, H.H.; Wahba, A.S.; Hasan, R.A. Inflammation, oxidative stress and apoptosis cascade implications in bisphenol A-induced liver fibrosis in male rats. Int. J. Exp. Pathol. 2016, 97, 369–379. [Google Scholar] [CrossRef]
- Belcher, S.M.; Gear, R.B.; Kendig, E.L. Bisphenol A alters autonomic tone and extracellular matrix structure and induces sex-specific effects on cardiovascular function in male and female CD-1 mice. Endocrinology 2015, 156, 882–895. [Google Scholar] [CrossRef] [Green Version]
- Brannick, K.E.; Craig, Z.R.; Himes, A.D.; Peretz, J.R.; Wang, W.; Flaws, J.A.; Raetzman, L.T. Prenatal exposure to low doses of bisphenol A increases pituitary proliferation and gonadotroph number in female mice offspring at birth. Biol. Reprod. 2012, 87, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molina, A.; Abril, N.; Morales-Prieto, N.; Monterde, J.; Ayala, N.; Lora, A.; Moyano, R. Hypothalamic-pituitary-ovarian axis perturbation in the basis of bisphenol A (BPA) reproductive toxicity in female zebrafish (Danio rerio). Ecotoxicol. Environ. Saf. 2018, 156, 116–124. [Google Scholar] [CrossRef]
- Ahmed, R.G. Maternal bisphenol A alters fetal endocrine system: Thyroid adipokine dysfunction. Food Chem. Toxicol. 2016, 95, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Matsuda, A.; Ishigami, A.; Yonekura, S.; Ishiwata, H.; Chen, C.; Obara, Y. Suppressing effects of bisphenol A on the secretory function of ovine anterior pituitary cells. Cell Biol. Int. 2004, 28, 463–469. [Google Scholar] [CrossRef]
- Jindatip, D.; Fujiwara, K.; Horiguchi, K.; Tsukada, T.; Kouki, T.; Yashiro, T. Changes in fine structure of pericytes and novel desmin-immunopositive perivascular cells during postnatal development in rat anterior pituitary gland. Anat. Sci. Int. 2013, 88, 196–203. [Google Scholar] [CrossRef]
- Jindatip, D.; Fujiwara, K.; Sarachana, T.; Mutirangura, A.; Yashiro, T. Characteristics of pericytes in diethylstilbestrol (DES)-induced pituitary prolactinoma in rats. Med. Mol. Morphol. 2018, 51, 147–155. [Google Scholar] [CrossRef]
- Steinmetz, R.; Brown, N.G.; Allen, D.L.; Bigsby, R.M.; Ben-Jonathan, N. The environmental estrogen bisphenol A stimulates prolactin release in vitro and in vivo. Endocrinology 1997, 138, 1780–1786. [Google Scholar] [CrossRef]
- Ulbrich, S.E.; Kettler, A.; Einspanier, R. Expression and localization of estrogen receptor alpha, estrogen receptor beta and progesterone receptor in the bovine oviduct in vivo and in vitro. J. Steroid Biochem. Mol. Biol. 2003, 84, 279–289. [Google Scholar] [CrossRef]
- Hao, L.; Zhang, J.; Zhang, Y.; Hu, H.; Shao, W.; Zhang, X.; Geng, C.; Wang, Y.; Jiang, L. Effect of bisphenol a on occurrence and progression of prolactinoma and its underlying mechanisms. Am. J. Transl. Res. 2016, 8, 4195–4204. [Google Scholar] [PubMed]
- Wada, I.; Sakuma, E.; Shirasawa, N.; Wakabayashi, K.; Otsuka, T.; Hattori, K.; Yashiro, T.; Herbert, D.C.; Soji, T. Intercellular communications within the rat anterior pituitary. XVI: Postnatal changes of distribution of S-100 protein positive cells, connexin 43 and LH-RH positive sites in the pars tuberalis of the rat pituitary gland. An immunohistochemical and electron microscopic study. Tissue Cell 2014, 46, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, K.; Tsukada, T.; Horiguchi, K.; Fujiwara, Y.; Takemoto, K.; Nio-Kobayashi, J.; Ohno, N.; Inoue, K. Aldolase C is a novel molecular marker for folliculo-stellate cells in rodent pituitary. Cell Tissue Res. 2020, 381, 273–284. [Google Scholar] [CrossRef]
- Mitchner, N.A.; Garlick, C.; Ben-Jonathan, N. Cellular distribution and gene regulation of estrogen receptors alpha and beta in the rat pituitary gland. Endocrinology 1998, 139, 3976–3983. [Google Scholar] [CrossRef]
- Fauquier, T.; Rizzoti, K.; Dattani, M.; Lovell-Badge, R.; Robinson, I.C. SOX2-expressing progenitor cells generate all of the major cell types in the adult mouse pituitary gland. Proc. Natl. Acad. Sci. USA 2008, 105, 2907–2912. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, S.; Kato, T.; Yako, H.; Susa, T.; Cai, L.Y.; Osuna, M.; Inoue, K.; Kato, Y. Significant quantitative and qualitative transition in pituitary stem / progenitor cells occurs during the postnatal development of the rat anterior pituitary. J. Neuroendocrinol. 2011, 23, 933–943. [Google Scholar] [CrossRef]
- Andoniadou, C.L.; Matsushima, D.; Mousavy Gharavy, S.N.; Signore, M.; Mackintosh, A.I.; Schaeffer, M.; Gaston-Massuet, C.; Mollard, P.; Jacques, T.S.; Le Tissier, P.; et al. Sox2(+) stem/progenitor cells in the adult mouse pituitary support organ homeostasis and have tumor-inducing potential. Cell Stem Cell 2013, 13, 433–445. [Google Scholar] [CrossRef] [Green Version]
- Brokken, L.J.; Leendertse, M.; Bakker, O.; Wiersinga, W.M.; Prummel, M.F. Expression of adenohypophyseal-hormone receptors in a murine folliculo-stellate cell line. Horm. Metab. Res. 2004, 36, 538–541. [Google Scholar] [CrossRef]
- Brokken, L.J.; Bakker, O.; Wiersinga, W.M.; Prummel, M.F. Functional thyrotropin receptor expression in the pituitary folliculo-stellate cell line TtT/GF. Exp. Clin. Endocrinol. Diabetes 2005, 113, 13–20. [Google Scholar] [CrossRef]
- Chen, G.; Ge, D.; Zhu, B.; Shi, H.; Ma, Q. Upregulation of matrix metalloproteinase 9 (MMP9)/tissue inhibitor of metalloproteinase 1 (TIMP1) and MMP2/TIMP2 ratios may be involved in lipopolysaccharide-induced acute lung injury. J. Int. Med. Res. 2020, 48, 300060520919592. [Google Scholar] [CrossRef] [PubMed]
- Savabieasfahani, M.; Kannan, K.; Astapova, O.; Evans, N.P.; Padmanabhan, V. Developmental programming: Differential effects of prenatal exposure to bisphenol-A or methoxychlor on reproductive function. Endocrinology 2006, 147, 5956–5966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thongkorn, S.; Kanlayaprasit, S.; Jindatip, D.; Tencomnao, T.; Hu, V.W.; Sarachana, T. Sex Differences in the Effects of Prenatal Bisphenol A Exposure on Genes Associated with Autism Spectrum Disorder in the Hippocampus. Sci. Rep. 2019, 9, 3038. [Google Scholar] [CrossRef]
- Sukjamnong, S.; Thongkorn, S.; Kanlayaprasit, S.; Saeliw, T.; Hussem, K.; Warayanon, W.; Hu, V.W.; Tencomnao, T.; Sarachana, T. Prenatal exposure to bisphenol A alters the transcriptome-interactome profiles of genes associated with Alzheimer’s disease in the offspring hippocampus. Sci. Rep. 2020, 10, 9487. [Google Scholar] [CrossRef] [PubMed]
- Thongkorn, S.; Kanlayaprasit, S.; Panjabud, P.; Saeliw, T.; Jantheang, T.; Kasitipradit, K.; Sarobol, S.; Jindatip, D.; Hu, V.W.; Tencomnao, T.; et al. Sex differences in the effects of prenatal bisphenol A exposure on autism-related genes and their relationships with the hippocampus functions. Sci. Rep. 2021, 11, 1241. [Google Scholar] [CrossRef]



| Genes | Forward Primer (5′>-3′) | Reverse Primer (5′>-3′) |
|---|---|---|
| Collagen type 1 (Col1a1) | CCTGACGCATGGCCAAGA | CTGGGCAGAAAGGACAGCA |
| Collagen type 3 (Col3a1) | TGCCACCCTGAACTCAAGAG | CACCAGCATATGTCCACCA |
| Matrix metalloproteinase 9 (MMP9) | AGAGCGTTACTCGCTTGGA | CTGCAGGAGGTCATAGGTCA |
| Tissue inhibitor of metalloproteinase 1 (TIMP1) | CTGGCATCCTCTTGTTGCT | AGGTGGTCTCGATGATTTCTG |
| Tissue inhibitor of metalloproteinase 2 (TIMP2) | GACGTTGGAGGAAAGAAGGA | GGCTCTTCTTCTGGGTGATG |
| Tissue inhibitor of metalloproteinase 3 (TIMP3) | TGGGAAAGAAGCTGGTGAA | CACATGGGGCATCTTACTGA |
| Tissue inhibitor of metalloproteinase 4 (TIMP4) | CACGCCATTTGACTCTTCTC | CTCCCAGGGCTCAATGTAGT |
| 18S Ribosomal RNA (RN18S) | CTGGATACCGCAGCTAGGAA | GAATTTCACCTCTAGCGGCG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanannam, B.; Looprasertkul, S.; Kanlayaprasit, S.; Kitkumthorn, N.; Sarachana, T.; Jindatip, D. Alteration of Extracellular Matrix Components in the Anterior Pituitary Gland of Neonatal Rats Induced by a Maternal Bisphenol A Diet during Pregnancy. Int. J. Mol. Sci. 2021, 22, 12667. https://doi.org/10.3390/ijms222312667
Sanannam B, Looprasertkul S, Kanlayaprasit S, Kitkumthorn N, Sarachana T, Jindatip D. Alteration of Extracellular Matrix Components in the Anterior Pituitary Gland of Neonatal Rats Induced by a Maternal Bisphenol A Diet during Pregnancy. International Journal of Molecular Sciences. 2021; 22(23):12667. https://doi.org/10.3390/ijms222312667
Chicago/Turabian StyleSanannam, Bumpenporn, Sasikarn Looprasertkul, Songphon Kanlayaprasit, Nakarin Kitkumthorn, Tewarit Sarachana, and Depicha Jindatip. 2021. "Alteration of Extracellular Matrix Components in the Anterior Pituitary Gland of Neonatal Rats Induced by a Maternal Bisphenol A Diet during Pregnancy" International Journal of Molecular Sciences 22, no. 23: 12667. https://doi.org/10.3390/ijms222312667
APA StyleSanannam, B., Looprasertkul, S., Kanlayaprasit, S., Kitkumthorn, N., Sarachana, T., & Jindatip, D. (2021). Alteration of Extracellular Matrix Components in the Anterior Pituitary Gland of Neonatal Rats Induced by a Maternal Bisphenol A Diet during Pregnancy. International Journal of Molecular Sciences, 22(23), 12667. https://doi.org/10.3390/ijms222312667






