Hypothyroidism-Associated Dyslipidemia: Potential Molecular Mechanisms Leading to NAFLD
Abstract
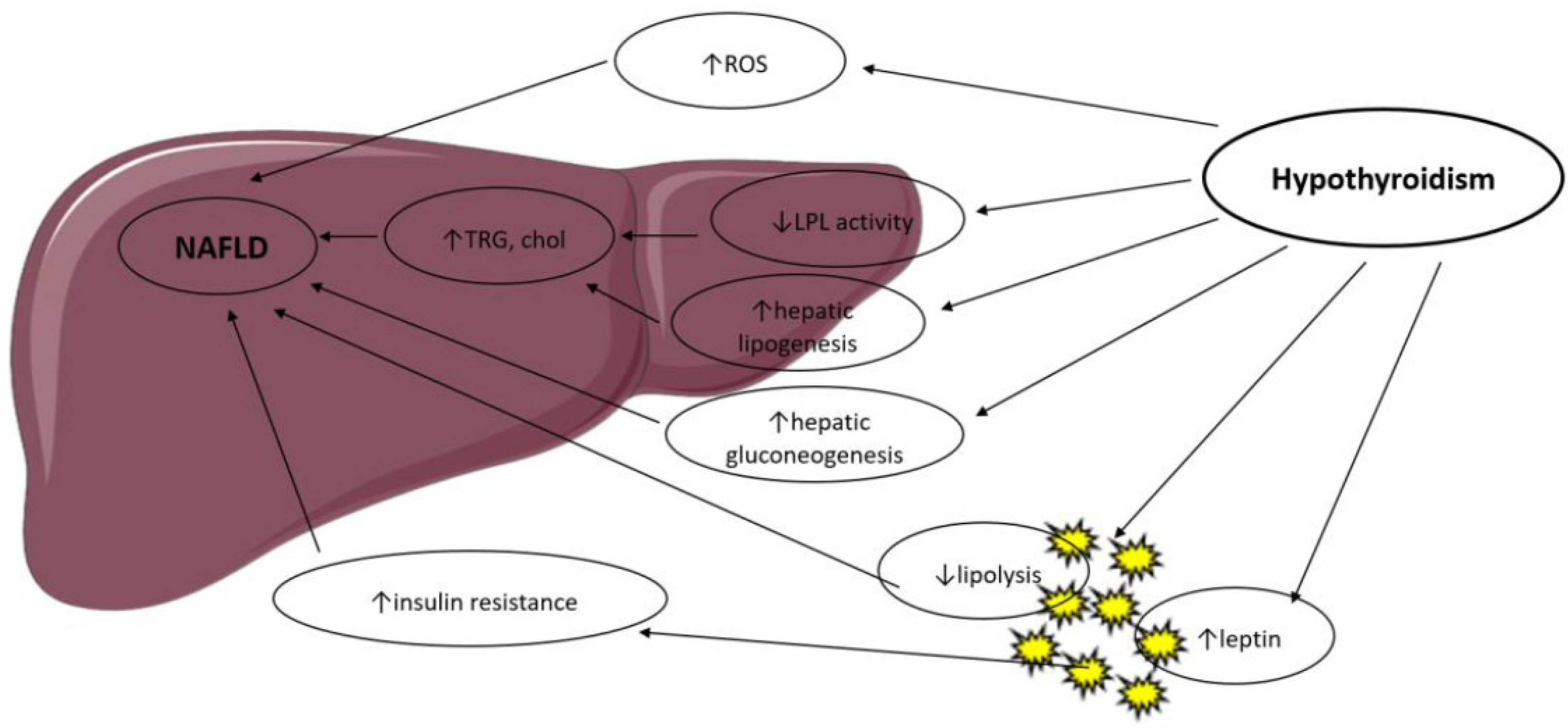
:1. Introduction
2. Thyroid Hormones and Thyroid Hormone Receptors
3. Thyroid Hormone Effects on Lipid Metabolism in the Liver and the Adipose Tissue
4. Thyroid Hormones and Dyslipidemia
5. NAFLD and Dysregulated Lipid Metabolism
6. Hypothyroidism and NAFLD: Clinical Studies
7. Thyroid Hormone Analogues for Dyslipidemia Treatment
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tanase, D.M.; Gosav, E.M.; Neculae, E.; Costea, C.F.; Ciocoiu, M.; Hurjui, L.L.; Tarniceriu, C.C.; Floria, M. Hypothyroidism-Induced Nonalcoholic Fatty Liver Disease (HIN): Mechanisms and Emerging Therapeutic Options. Int. J. Mol. Sci. 2020, 21, 5927. [Google Scholar] [CrossRef]
- Jornayvaz, F.R.; Shulman, G.I. Diacylglycerol activation of protein kinase Cepsilon and hepatic insulin resistance. Cell Metab. 2012, 15, 574–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.Y.; Gusdon, A.M.; Qu, S. Cross-talk between the thyroid and liver: A new target for nonalcoholic fatty liver disease treatment. World J. Gastroenterol. 2013, 19, 8238–8246. [Google Scholar] [CrossRef]
- Motomura, K.; Brent, G.A. Mechanisms of thyroid hormone action. Implications for the clinical manifestation of thyrotoxicosis. Endocrinol. Metab. Clin. N. Am. 1998, 27, 1–23. [Google Scholar] [CrossRef] [Green Version]
- Perra, A.; Simbula, G.; Simbula, M.; Pibiri, M.; Kowalik, M.A.; Sulas, P.; Cocco, M.T.; Ledda-Columbano, G.M.; Columbano, A. Thyroid hormone (T3) and TRbeta agonist GC-1 inhibit/reverse nonalcoholic fatty liver in rats. FASEB J. 2008, 22, 2981–2989. [Google Scholar] [CrossRef]
- Cable, E.E.; Finn, P.D.; Stebbins, J.W.; Hou, J.; Ito, B.R.; van Poelje, P.D.; Linemeyer, D.L.; Erion, M.D. Reduction of hepatic steatosis in rats and mice after treatment with a liver-targeted thyroid hormone receptor agonist. Hepatology 2009, 49, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yu, Y.; Zhao, M.; Zheng, D.; Zhang, X.; Guan, Q.; Xu, C.; Gao, L.; Zhao, J.; Zhang, H. Benefits of Levothyroxine Replacement Therapy on Nonalcoholic Fatty Liver Disease in Subclinical Hypothyroidism Patients. Int. J. Endocrinol. 2017, 2017, 5753039. [Google Scholar] [CrossRef]
- Bruinstroop, E.; Dalan, R.; Cao, Y.; Bee, Y.M.; Chandran, K.; Cho, L.W.; Soh, S.B.; Teo, E.K.; Toh, S.A.; Leow, M.K.S.; et al. Low-Dose Levothyroxine Reduces Intrahepatic Lipid Content in Patients With Type 2 Diabetes Mellitus and NAFLD. J. Clin. Endocrinol. Metab. 2018, 103, 2698–2706. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.A.; You, S.H.; Zhou, J.; Siddique, M.M.; Bay, B.H.; Zhu, X.; Privalsky, M.L.; Cheng, S.Y.; Stevens, R.D.; Summers, S.A.; et al. Thyroid hormone stimulates hepatic lipid catabolism via activation of autophagy. J. Clin. Investig. 2012, 122, 2428–2438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flores-Morales, A.; Gullberg, H.; Fernandez, L.; Stahlberg, N.; Lee, N.H.; Vennstrom, B.; Norstedt, G. Patterns of liver gene expression governed by TRbeta. Mol. Endocrinol. 2002, 16, 1257–1268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jornayvaz, F.R.; Lee, H.Y.; Jurczak, M.J.; Alves, T.C.; Guebre-Egziabher, F.; Guigni, B.A.; Zhang, D.; Samuel, V.T.; Silva, J.E.; Shulman, G.I. Thyroid hormone receptor-alpha gene knockout mice are protected from diet-induced hepatic insulin resistance. Endocrinology 2012, 153, 583–591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riis, A.L.; Gravholt, C.H.; Djurhuus, C.B.; Norrelund, H.; Jorgensen, J.O.; Weeke, J.; Moller, N. Elevated regional lipolysis in hyperthyroidism. J. Clin. Endocrinol. Metab. 2002, 87, 4747–4753. [Google Scholar] [CrossRef] [Green Version]
- Cachefo, A.; Boucher, P.; Vidon, C.; Dusserre, E.; Diraison, F.; Beylot, M. Hepatic lipogenesis and cholesterol synthesis in hyperthyroid patients. J. Clin. Endocrinol. Metab. 2001, 86, 5353–5357. [Google Scholar] [CrossRef]
- Klieverik, L.P.; Coomans, C.P.; Endert, E.; Sauerwein, H.P.; Havekes, L.M.; Voshol, P.J.; Rensen, P.C.; Romijn, J.A.; Kalsbeek, A.; Fliers, E. Thyroid hormone effects on whole-body energy homeostasis and tissue-specific fatty acid uptake in vivo. Endocrinology 2009, 150, 5639–5648. [Google Scholar] [CrossRef] [Green Version]
- Nedvidkova, J.; Haluzik, M.; Bartak, V.; Dostalova, I.; Vlcek, P.; Racek, P.; Taus, M.; Behanova, M.; Svacina, S.; Alesci, S.; et al. Changes of noradrenergic activity and lipolysis in the subcutaneous abdominal adipose tissue of hypo- and hyperthyroid patients: An in vivo microdialysis study. Ann. N. Y. Acad. Sci. 2004, 1018, 541–549. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, R.; Campi, I.; Maggioni, M.; Perbellini, R.; Giammona, E.; Stucchi, R.; Borghi, M.; Degasperi, E.; De Silvestri, A.; Persani, L.; et al. The relationship between liver histology and thyroid function tests in patients with non-alcoholic fatty liver disease (NAFLD). PLoS ONE 2021, 16, e0249614. [Google Scholar] [CrossRef] [PubMed]
- Hazlehurst, J.M.; Tomlinson, J.W. Non-alcoholic fatty liver disease in common endocrine disorders. Eur. J. Endocrinol. 2013, 169, R27–R37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, L.; Zhang, Y.; Hillgartner, F.B. Sterol regulatory element-binding protein-1 interacts with the nuclear thyroid hormone receptor to enhance acetyl-CoA carboxylase-alpha transcription in hepatocytes. J. Biol. Chem. 2002, 277, 19554–19565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dentin, R.; Girard, J.; Postic, C. Carbohydrate responsive element binding protein (ChREBP) and sterol regulatory element binding protein-1c (SREBP-1c): Two key regulators of glucose metabolism and lipid synthesis in liver. Biochimie 2005, 87, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Gariani, K.; Jornayvaz, F.R. Pathophysiology of NASH in endocrine diseases. Endocr. Connect. 2021, 10, R52–R65. [Google Scholar] [CrossRef]
- Ritter, M.J.; Amano, I.; Hollenberg, A.N. Thyroid Hormone Signaling and the Liver. Hepatology 2020, 72, 742–752. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Zhou, L.; Song, Y.; Ma, S.; Yu, C.; Zhao, J.; Xu, C.; Gao, L. Thyroid stimulating hormone increases hepatic gluconeogenesis via CRTC2. Mol. Cell. Endocrinol. 2017, 446, 70–80. [Google Scholar] [CrossRef]
- Pihlajamaki, J.; Boes, T.; Kim, E.Y.; Dearie, F.; Kim, B.W.; Schroeder, J.; Mun, E.; Nasser, I.; Park, P.J.; Bianco, A.C.; et al. Thyroid hormone-related regulation of gene expression in human fatty liver. J. Clin. Endocrinol. Metab. 2009, 94, 3521–3529. [Google Scholar] [CrossRef] [Green Version]
- Araki, O.; Ying, H.; Furuya, F.; Zhu, X.; Cheng, S.Y. Thyroid hormone receptor beta mutants: Dominant negative regulators of peroxisome proliferator-activated receptor gamma action. Proc. Natl. Acad. Sci. USA 2005, 102, 16251–16256. [Google Scholar] [CrossRef] [Green Version]
- Rautou, P.E.; Mansouri, A.; Lebrec, D.; Durand, F.; Valla, D.; Moreau, R. Autophagy in liver diseases. J. Hepatol. 2010, 53, 1123–1134. [Google Scholar] [CrossRef] [Green Version]
- Messarah, M.; Boumendjel, A.; Chouabia, A.; Klibet, F.; Abdennour, C.; Boulakoud, M.S.; Feki, A.E. Influence of thyroid dysfunction on liver lipid peroxidation and antioxidant status in experimental rats. Exp. Toxicol. Pathol. 2010, 62, 301–310. [Google Scholar] [CrossRef]
- Loria, P.; Carulli, L.; Bertolotti, M.; Lonardo, A. Endocrine and liver interaction: The role of endocrine pathways in NASH. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 236–247. [Google Scholar] [CrossRef]
- Choi, J.W.; Choi, H.S. The regulatory effects of thyroid hormone on the activity of 3-hydroxy-3-methylglutaryl coenzyme A reductase. Endocr. Res. 2000, 26, 1–21. [Google Scholar] [CrossRef]
- Galman, C.; Bonde, Y.; Matasconi, M.; Angelin, B.; Rudling, M. Dramatically increased intestinal absorption of cholesterol following hypophysectomy is normalized by thyroid hormone. Gastroenterology 2008, 134, 1127–1136. [Google Scholar] [CrossRef]
- Shin, D.J.; Osborne, T.F. Thyroid hormone regulation and cholesterol metabolism are connected through Sterol Regulatory Element-Binding Protein-2 (SREBP-2). J. Biol. Chem. 2003, 278, 34114–34118. [Google Scholar] [CrossRef] [Green Version]
- Lam, K.S.; Chan, M.K.; Yeung, R.T. High-density lipoprotein cholesterol, hepatic lipase and lipoprotein lipase activities in thyroid dysfunction--effects of treatment. Q. J. Med. 1986, 59, 513–521. [Google Scholar]
- Tan, K.C.; Shiu, S.W.; Kung, A.W. Plasma cholesteryl ester transfer protein activity in hyper- and hypothyroidism. J. Clin. Endocrinol. Metab. 1998, 83, 140–143. [Google Scholar] [CrossRef]
- Duntas, L.H. Thyroid disease and lipids. Thyroid 2002, 12, 287–293. [Google Scholar] [CrossRef]
- Pearce, E.N. Update in lipid alterations in subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 2012, 97, 326–333. [Google Scholar] [CrossRef] [Green Version]
- Vierhapper, H.; Nardi, A.; Grosser, P.; Raber, W.; Gessl, A. Low-density lipoprotein cholesterol in subclinical hypothyroidism. Thyroid 2000, 10, 981–984. [Google Scholar] [CrossRef]
- Hueston, W.J.; Pearson, W.S. Subclinical hypothyroidism and the risk of hypercholesterolemia. Ann. Fam. Med. 2004, 2, 351–355. [Google Scholar] [CrossRef] [Green Version]
- Kanaya, A.M.; Harris, F.; Volpato, S.; Perez-Stable, E.J.; Harris, T.; Bauer, D.C. Association between thyroid dysfunction and total cholesterol level in an older biracial population: The health, aging and body composition study. Arch. Intern. Med. 2002, 162, 773–779. [Google Scholar] [CrossRef] [Green Version]
- Canaris, G.J.; Manowitz, N.R.; Mayor, G.; Ridgway, E.C. The Colorado thyroid disease prevalence study. Arch. Intern. Med. 2000, 160, 526–534. [Google Scholar] [CrossRef] [Green Version]
- Parthasarathy, G.; Revelo, X.; Malhi, H. Pathogenesis of Nonalcoholic Steatohepatitis: An Overview. Hepatol. Commun. 2020, 4, 478–492. [Google Scholar] [CrossRef] [Green Version]
- Buzzetti, E.; Pinzani, M.; Tsochatzis, E.A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 2016, 65, 1038–1048. [Google Scholar] [CrossRef]
- Mansouri, A.; Gattolliat, C.H.; Asselah, T. Mitochondrial Dysfunction and Signaling in Chronic Liver Diseases. Gastroenterology 2018, 155, 629–647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guilherme, A.; Virbasius, J.V.; Puri, V.; Czech, M.P. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 367–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asrih, M.; Altirriba, J.; Rohner-Jeanrenaud, F.; Jornayvaz, F.R. Ketogenic Diet Impairs FGF21 Signaling and Promotes Differential Inflammatory Responses in the Liver and White Adipose Tissue. PLoS ONE 2015, 10, e0126364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camporez, J.P.; Asrih, M.; Zhang, D.; Kahn, M.; Samuel, V.T.; Jurczak, M.J.; Jornayvaz, F.R. Hepatic insulin resistance and increased hepatic glucose production in mice lacking Fgf21. J. Endocrinol. 2015, 226, 207–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camporez, J.P.G.; Kanda, S.; Petersen, M.C.; Jornayvaz, F.R.; Samuel, V.T.; Bhanot, S.; Petersen, K.F.; Jurczak, M.J.; Shulman, G.I. ApoA5 knockdown improves whole-body insulin sensitivity in high-fat-fed mice by reducing ectopic lipid content. J. Lipid Res. 2015, 56, 526–536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Somm, E.; Montandon, S.A.; Loizides-Mangold, U.; Gaia, N.; Lazarevic, V.; De Vito, C.; Perroud, E.; Bochaton-Piallat, M.L.; Dibner, C.; Schrenzel, J.; et al. The GLP-1R agonist liraglutide limits hepatic lipotoxicity and inflammatory response in mice fed a methionine-choline deficient diet. Transl. Res. 2021, 227, 75–88. [Google Scholar] [CrossRef]
- Montandon, S.A.; Somm, E.; Loizides-Mangold, U.; de Vito, C.; Dibner, C.; Jornayvaz, F.R. Multi-technique comparison of atherogenic and MCD NASH models highlights changes in sphingolipid metabolism. Sci. Rep. 2019, 9, 16810. [Google Scholar] [CrossRef]
- Somm, E.; Jornayvaz, F.R. Fibroblast Growth Factor 15/19: From Basic Functions to Therapeutic Perspectives. Endocr. Rev. 2018, 39, 960–989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Somm, E.; Henry, H.; Bruce, S.J.; Aeby, S.; Rosikiewicz, M.; Sykiotis, G.P.; Asrih, M.; Jornayvaz, F.R.; Denechaud, P.D.; Albrecht, U.; et al. beta-Klotho deficiency protects against obesity through a crosstalk between liver, microbiota, and brown adipose tissue. JCI Insight 2017, 2, e91809. [Google Scholar] [CrossRef] [PubMed]
- Bugianesi, E.; Moscatiello, S.; Ciaravella, M.F.; Marchesini, G. Insulin resistance in nonalcoholic fatty liver disease. Curr. Pharm. Des. 2010, 16, 1941–1951. [Google Scholar] [CrossRef]
- Jornayvaz, F.R.; Samuel, V.T.; Shulman, G.I. The role of muscle insulin resistance in the pathogenesis of atherogenic dyslipidemia and nonalcoholic fatty liver disease associated with the metabolic syndrome. Annu. Rev. Nutr. 2010, 30, 273–290. [Google Scholar] [CrossRef] [Green Version]
- Krauss, R.M.; Siri, P.W. Metabolic abnormalities: Triglyceride and low-density lipoprotein. Endocrinol. Metab. Clin. N. Am. 2004, 33, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, H.N. Insulin resistance and cardiovascular disease. J. Clin. Investig. 2000, 106, 453–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marino, L.; Jornayvaz, F.R. Endocrine causes of nonalcoholic fatty liver disease. World J. Gastroenterol. 2015, 21, 11053–11076. [Google Scholar] [CrossRef] [PubMed]
- Liangpunsakul, S.; Chalasani, N. Is hypothyroidism a risk factor for non-alcoholic steatohepatitis? J. Clin. Gastroenterol. 2003, 37, 340–343. [Google Scholar] [CrossRef]
- Reddy, A.; Dash, C.; Leerapun, A.; Mettler, T.A.; Stadheim, L.M.; Lazaridis, K.N.; Roberts, R.O.; Roberts, L.R. Hypothyroidism: A possible risk factor for liver cancer in patients with no known underlying cause of liver disease. Clin. Gastroenterol. Hepatol. 2007, 5, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Silveira, M.G.; Mendes, F.D.; Diehl, N.N.; Enders, F.T.; Lindor, K.D. Thyroid dysfunction in primary biliary cirrhosis, primary sclerosing cholangitis and non-alcoholic fatty liver disease. Liver Int. 2009, 29, 1094–1100. [Google Scholar] [CrossRef]
- Xu, C.; Xu, L.; Yu, C.; Miao, M.; Li, Y. Association between thyroid function and nonalcoholic fatty liver disease in euthyroid elderly Chinese. Clin. Endocrinol. 2011, 75, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Chung, G.E.; Kim, D.; Kim, W.; Yim, J.Y.; Park, M.J.; Kim, Y.J.; Yoon, J.H.; Lee, H.S. Non-alcoholic fatty liver disease across the spectrum of hypothyroidism. J. Hepatol. 2012, 57, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Pagadala, M.R.; Zein, C.O.; Dasarathy, S.; Yerian, L.M.; Lopez, R.; McCullough, A.J. Prevalence of hypothyroidism in nonalcoholic fatty liver disease. Dig. Dis. Sci. 2012, 57, 528–534. [Google Scholar] [CrossRef] [Green Version]
- Xu, L.; Ma, H.; Miao, M.; Li, Y. Impact of subclinical hypothyroidism on the development of non-alcoholic fatty liver disease: A prospective case-control study. J. Hepatol. 2012, 57, 1153–1154. [Google Scholar] [CrossRef] [Green Version]
- Ittermann, T.; Haring, R.; Wallaschofski, H.; Baumeister, S.E.; Nauck, M.; Dorr, M.; Lerch, M.M.; Meyer zu Schwabedissen, H.E.; Rosskopf, D.; Volzke, H. Inverse association between serum free thyroxine levels and hepatic steatosis: Results from the Study of Health in Pomerania. Thyroid 2012, 22, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Eshraghian, A.; Dabbaghmanesh, M.H.; Eshraghian, H.; Fattahi, M.R.; Omrani, G.R. Nonalcoholic fatty liver disease in a cluster of Iranian population: Thyroid status and metabolic risk factors. Arch. Iran. Med. 2013, 16, 584–589. [Google Scholar] [PubMed]
- Posadas-Romero, C.; Jorge-Galarza, E.; Posadas-Sanchez, R.; Acuna-Valerio, J.; Juarez-Rojas, J.G.; Kimura-Hayama, E.; Medina-Urrutia, A.; Cardoso-Saldana, G.C. Fatty liver largely explains associations of subclinical hypothyroidism with insulin resistance, metabolic syndrome, and subclinical coronary atherosclerosis. Eur. J. Endocrinol. 2014, 171, 319–325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.W.; Bang, K.B.; Rhee, E.J.; Kwon, H.J.; Lee, M.Y.; Cho, Y.K. Impact of hypothyroidism on the development of non-alcoholic fatty liver disease: A 4-year retrospective cohort study. Clin. Mol. Hepatol. 2015, 21, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Parikh, P.; Phadke, A.; Sawant, P. Prevalence of hypothyroidism in nonalcoholic fatty liver disease in patients attending a tertiary hospital in western India. Indian J. Gastroenterol. 2015, 34, 169–173. [Google Scholar] [CrossRef]
- Ludwig, U.; Holzner, D.; Denzer, C.; Greinert, A.; Haenle, M.M.; Oeztuerk, S.; Koenig, W.; Boehm, B.O.; Mason, R.A.; Kratzer, W.; et al. Subclinical and clinical hypothyroidism and non-alcoholic fatty liver disease: A cross-sectional study of a random population sample aged 18 to 65 years. BMC Endocr. Disord. 2015, 15, 41. [Google Scholar] [CrossRef] [Green Version]
- Bano, A.; Chaker, L.; Plompen, E.P.; Hofman, A.; Dehghan, A.; Franco, O.H.; Janssen, H.L.; Darwish Murad, S.; Peeters, R.P. Thyroid Function and the Risk of Nonalcoholic Fatty Liver Disease: The Rotterdam Study. J. Clin. Endocrinol. Metab. 2016, 101, 3204–3211. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.; Kim, W.; Joo, S.K.; Bae, J.M.; Kim, J.H.; Ahmed, A. Subclinical Hypothyroidism and Low-Normal Thyroid Function Are Associated With Nonalcoholic Steatohepatitis and Fibrosis. Clin. Gastroenterol. Hepatol. 2018, 16, 123–131.e121. [Google Scholar] [CrossRef] [PubMed]
- Martinez Escude, A.; Pera, G.; Arteaga, I.; Exposito, C.; Rodriguez, L.; Toran, P.; Caballeria, L. Relationship between hypothyroidism and non-alcoholic fatty liver disease in the Spanish population. Med. Clin. 2020, 154, 1–6. [Google Scholar] [CrossRef]
- Eshraghian, A.; Hamidian Jahromi, A. Non-alcoholic fatty liver disease and thyroid dysfunction: A systematic review. World J. Gastroenterol. 2014, 20, 8102–8109. [Google Scholar] [CrossRef] [PubMed]
- Loosen, S.H.; Demir, M.; Kostev, K.; Luedde, T.; Roderburg, C. Incidences of hypothyroidism and autoimmune thyroiditis are increased in patients with nonalcoholic fatty liver disease. Eur. J. Gastroenterol. Hepatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Jaruvongvanich, V.; Sanguankeo, A.; Upala, S. Nonalcoholic Fatty Liver Disease Is Not Associated with Thyroid Hormone Levels and Hypothyroidism: A Systematic Review and Meta-Analysis. Eur. Thyroid J. 2017, 6, 208–215. [Google Scholar] [CrossRef] [Green Version]
- Mantovani, A.; Nascimbeni, F.; Lonardo, A.; Zoppini, G.; Bonora, E.; Mantzoros, C.S.; Targher, G. Association Between Primary Hypothyroidism and Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. Thyroid 2018, 28, 1270–1284. [Google Scholar] [CrossRef]
- He, W.; An, X.; Li, L.; Shao, X.; Li, Q.; Yao, Q.; Zhang, J.A. Relationship between Hypothyroidism and Non-Alcoholic Fatty Liver Disease: A Systematic Review and Meta-analysis. Front. Endocrinol. 2017, 8, 335. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.; Yoo, E.R.; Li, A.A.; Fernandes, C.T.; Tighe, S.P.; Cholankeril, G.; Hameed, B.; Ahmed, A. Low-Normal Thyroid Function Is Associated With Advanced Fibrosis Among Adults in the United States. Clin. Gastroenterol. Hepatol. 2019, 17, 2379–2381. [Google Scholar] [CrossRef]
- Kim, D.; Vazquez-Montesino, L.M.; Escober, J.A.; Fernandes, C.T.; Cholankeril, G.; Loomba, R.; Harrison, S.A.; Younossi, Z.M.; Ahmed, A. Low Thyroid Function in Nonalcoholic Fatty Liver Disease Is an Independent Predictor of All-Cause and Cardiovascular Mortality. Am. J. Gastroenterol. 2020, 115, 1496–1504. [Google Scholar] [CrossRef]
- Sinha, R.A.; Singh, B.K.; Yen, P.M. Direct effects of thyroid hormones on hepatic lipid metabolism. Nat. Rev. Endocrinol. 2018, 14, 259–269. [Google Scholar] [CrossRef]
- Kizivat, T.; Maric, I.; Mudri, D.; Curcic, I.B.; Primorac, D.; Smolic, M. Hypothyroidism and Nonalcoholic Fatty Liver Disease: Pathophysiological Associations and Therapeutic Implications. J. Clin. Transl. Hepatol. 2020, 8, 347–353. [Google Scholar] [CrossRef]
- Redgrave, T.G.; Elsegood, C.L.; Mamo, J.C.; Callow, M.J. Effects of hypothyroidism on the metabolism of lipid emulsion models of triacylglycerol-rich lipoproteins in rats. Biochem. J. 1991, 273, 375–381. [Google Scholar] [CrossRef] [Green Version]
- Delitala, A.P.; Delitala, G.; Sioni, P.; Fanciulli, G. Thyroid hormone analogs for the treatment of dyslipidemia: Past, present, and future. Curr. Med. Res. Opin. 2017, 33, 1985–1993. [Google Scholar] [CrossRef]
- Borngraeber, S.; Budny, M.J.; Chiellini, G.; Cunha-Lima, S.T.; Togashi, M.; Webb, P.; Baxter, J.D.; Scanlan, T.S.; Fletterick, R.J. Ligand selectivity by seeking hydrophobicity in thyroid hormone receptor. Proc. Natl. Acad. Sci. USA 2003, 100, 15358–15363. [Google Scholar] [CrossRef] [Green Version]
- Trost, S.U.; Swanson, E.; Gloss, B.; Wang-Iverson, D.B.; Zhang, H.; Volodarsky, T.; Grover, G.J.; Baxter, J.D.; Chiellini, G.; Scanlan, T.S.; et al. The thyroid hormone receptor-beta-selective agonist GC-1 differentially affects plasma lipids and cardiac activity. Endocrinology 2000, 141, 3057–3064. [Google Scholar] [CrossRef]
- Grijota-Martinez, C.; Samarut, E.; Scanlan, T.S.; Morte, B.; Bernal, J. In vivo activity of the thyroid hormone receptor beta- and alpha-selective agonists GC-24 and CO23 on rat liver, heart, and brain. Endocrinology 2011, 152, 1136–1142. [Google Scholar] [CrossRef] [Green Version]
- Sjouke, B.; Langslet, G.; Ceska, R.; Nicholls, S.J.; Nissen, S.E.; Ohlander, M.; Ladenson, P.W.; Olsson, A.G.; Hovingh, G.K.; Kastelein, J.J. Eprotirome in patients with familial hypercholesterolaemia (the AKKA trial): A randomised, double-blind, placebo-controlled phase 3 study. Lancet Diabetes Endocrinol. 2014, 2, 455–463. [Google Scholar] [CrossRef]
- Ladenson, P.W.; Kristensen, J.D.; Ridgway, E.C.; Olsson, A.G.; Carlsson, B.; Klein, I.; Baxter, J.D.; Angelin, B. Use of the thyroid hormone analogue eprotirome in statin-treated dyslipidemia. N. Engl. J. Med. 2010, 362, 906–916. [Google Scholar] [CrossRef] [Green Version]
- Angelin, B.; Kristensen, J.D.; Eriksson, M.; Carlsson, B.; Klein, I.; Olsson, A.G.; Chester Ridgway, E.; Ladenson, P.W. Reductions in serum levels of LDL cholesterol, apolipoprotein B, triglycerides and lipoprotein(a) in hypercholesterolaemic patients treated with the liver-selective thyroid hormone receptor agonist eprotirome. J. Intern. Med. 2015, 277, 331–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taub, R.; Chiang, E.; Chabot-Blanchet, M.; Kelly, M.J.; Reeves, R.A.; Guertin, M.C.; Tardif, J.C. Lipid lowering in healthy volunteers treated with multiple doses of MGL-3196, a liver-targeted thyroid hormone receptor-beta agonist. Atherosclerosis 2013, 230, 373–380. [Google Scholar] [CrossRef]
- Harrison, S.A.; Bashir, M.R.; Guy, C.D.; Zhou, R.; Moylan, C.A.; Frias, J.P.; Alkhouri, N.; Bansal, M.B.; Baum, S.; Neuschwander-Tetri, B.A.; et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: A multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2019, 394, 2012–2024. [Google Scholar] [CrossRef]
- Wong, V.W.; Singal, A.K. Emerging medical therapies for non-alcoholic fatty liver disease and for alcoholic hepatitis. Transl. Gastroenterol. Hepatol. 2019, 4, 53. [Google Scholar] [CrossRef] [PubMed]
- Fujitaki, J.M.; Cable, E.E.; Ito, B.R.; Zhang, B.H.; Hou, J.; Yang, C.; Bullough, D.A.; Ferrero, J.L.; van Poelje, P.D.; Linemeyer, D.L.; et al. Preclinical pharmacokinetics of a HepDirect prodrug of a novel phosphonate-containing thyroid hormone receptor agonist. Drug Metab. Dispos. 2008, 36, 2393–2403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erion, M.D.; Cable, E.E.; Ito, B.R.; Jiang, H.; Fujitaki, J.M.; Finn, P.D.; Zhang, B.H.; Hou, J.; Boyer, S.H.; van Poelje, P.D.; et al. Targeting thyroid hormone receptor-beta agonists to the liver reduces cholesterol and triglycerides and improves the therapeutic index. Proc. Natl. Acad. Sci. USA 2007, 104, 15490–15495. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Reference | Study Design | Study Sample | Diagnosis of NAFLD | Definition of Hypothyroidism | Main Findings |
|---|---|---|---|---|---|
| Liangpunsakul et al. 2003 [55] | Cross-sectional case-control study | 174 patients with NASH and 442 controls | Biopsy (all cases had NASH) | Self-reported use of levothyroxin | Hypothyroidism was independently associated with NASH (OR 2.30, 95% CI 1.20–4.20) |
| Reddy et al. 2007 [56] | Case-control study | 54 patients with HCC of unknown etiology and 2 groups of controls (57 HCC patients with HCV and 49 HCC patients with alcoholic liver disease) | Biopsy or clinical and imaging criteria | TSH > 5 mIU/L, history of hypothyroidism | Hypothyroidism is significantly higher prevalent in subjects with HCC of unknown etiology compared to controls with viral or alcoholic HCC |
| Silveira et al. 2009 [57] | Cross-sectional study | 97 patients with NAFLD | Biopsy | TSH > 5 mIU/L or < 0.3 mIU/L Total T4 > 12.5 μg/dL or < 5 μg/dL History of hyper/hypothyroidism | The prevalence of hypothyroidism in patients with NAFLD was 20% |
| Xu et al. 2011 [58] | Cross-sectional study | 227 patients with NAFLD and 651 controls | Ultrasound | TSH > 4.5 mIU/L or < 0.5 mIU/L FT4 > 14.4 pmol/L or < 7.85 pmol/L | Patients with hypothyroidism are more likely to develop NAFLD (p < 0.001), FT4 is a risk factor for NAFLD (OR = 0.847, 95% CI: 0.743–0.966) |
| Chung et al. 2012 [59] | Cross-sectional study | 2324 patients with hypothyroidism and 2324 controls | Ultrasound | Subclinical hypothyroidism: TSH > 4.1 mIU/L and normal FT4 Overt hypothyroidism: TSH > 4.1 mIU/L and FT4 < 0.7 ng/dL | Hypothyroidism is an independent risk factor for NAFLD (OR = 1.38, 95% CI: 1.17–1.67) |
| Pagadala et al. 2012 [60] | Cross-sectional study | 233 patients with NAFLD and 430 controls | Biopsy | Clinical diagnosis and on thyroid replacement therapy | Prevalence of hypothyroidism was higher in NAFLD patients (21.1% vs. 9.5%, p < 0.001) |
| Xu et al. 2012 [61] | Prospective case-control study | 327 patients with subclinical hypothyroidism and 327 controls | Ultrasound (15% developed NAFLD after 4.9 years median follow-up) | TSH > 4.5 mIU/L and normal FT4 levels | Subclinical hypothyroidism was independently associated with risk of developing NAFLD (HR 2.21, 95% CI: 1.42–3.44) |
| Itterman et al. 2012 [62] | Population-based study | 3661 individuals without a self-reported history of thyroid or liver disease | Ultrasound (16.1% had NAFLD) | Subclinical hypothyroidism: TSH > 3 mIU/L and normal FT4 Overt hypothyroidism: TSH > 3 mIU/L and FT4 < 7 pmol/L | Hypothyroidism was not independently associated with NAFLD. FT4 levels were inversely associated with NAFLD in men (OR 0.04, 95% CI: 0.01–0.17]) and in women (OR 0.06, 95% CI:0.01–0.42) |
| Eshraghian et al. 2013 [63] | Cross-sectional study | 832 individuals | Ultrasound (15.3% had NAFLD) | Subclinical hypothyroidism: TSH > 5.2 mIU/L and normal FT4 levels Overt hypothyroidism: TSH > 5.2 mIU/L and FT4 < 11.5 pmol/L | Subclinical hypothyroidism was not associated with NAFLD (OR 1.12, 95% CI: 0.51–2.46). Overt hypothyroidism was not associated with NAFLD (OR 0.87, 95% CI: 0.33–2.28) |
| Posadas-Romero et al. 2014 [64] | Cross-sectional study | 753 adults | Computed tomography (31.1% with NAFLD) | Subclinical hypothyroidism: TSH > 4.5 mIU/L and normal FT4 | Subclinical hypothyroidism was not associated with NAFLD (OR 0.83, 95% CI: 0.55–1.25) |
| Lee et al. 2015 [65] | Retrospective cohort study | 18,544 individuals | Ultrasound | Subclinical hypothyroidism: TSH > 4.2 mIU/L, normal FT4 Overt hypothyroidism: TSH > 4.2 mIU/L, FT4 < 10.97 ng/dL | NAFLD incidence did not differ significantly with thyroid hormonal status (Subclinical hypothyroidism: HR = 0.965, 95% CI = 0.814–1.143, p = 0.67; Overt hypothyroidism group: HR = 1.255, 95% CI = 0.830–1.899, p = 0.28) |
| Parikh et al. 2015 [66] | Case-control study | 500 patients with NAFLD and 300 controls | Ultrasound | Subclinical hypothyroidism: TSH > 5.5 IU/mL and <10 IU/mL) Overt hypothyroidism: TSH > 10 IU/mL) | NAFLD was statistically significantly associated with hypothyroidism (OR: 14.94, 95% CI: 3.5–62.6) |
| Ludwig et al. 2015 [67] | Cross-sectional, population-based study | 1276 individuals | Ultrasound (24.7% with NAFLD) | Subclinical hypothyroidism: TSH > 3.4 mIU/L and normal total T4 Overt hypothyroidism: TSH > 3.4 mIU/L and total T4 < 12.8 pmol/L | Hypothyroidism was not associated with NAFLD (OR 1.19 95% CI: 0.65–2.17) |
| Bano et al. 2016 [68] | Longitudinal prospective cohort study | 9419 euthyroid adults | Ultrasound (12.9% developed incident NAFLD after 10 years of median follow-up) | Subclinical hypothyroidism: TSH > 4.0 mIU/L and normal FT4 Overt hypothyroidism: TSH > 4.0 mIU/L and FT4 < 10.9 pmol/L | Hypothyroidism was associated with a 1.24-fold higher NAFLD risk (95% CI: 1.01–1.53). NAFLD risk decreased gradually from hypothyroidism to hyperthyroidism (p for trend = 0.003). |
| Kim et al. 2018 [69] | Cross-sectional study | 425 patients with NAFLD | Biopsy | Subclinical hypothyroidism: TSH > 4.5 mIU/L and normal FT4 | Subclinical hypothyroidism was independently associated with NASH (OR 1.61, 95% CI: 1.04–2.50) and advanced fibrosis (OR 2.23 95% CI: 1.18–4.23). |
| Martinez Escude et al. 2020 [70] | Cross-sectional, retrospective population study | 10,116 adults | Ultrasound | Subclinical hypothyroidism: TSH > 4.94 UI/mL and normal T4 Overt hypothyroidism: elevated TSH and decreased T4 | Hypothyroidism is not associated with NAFLD (p = 0.631) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mavromati, M.; Jornayvaz, F.R. Hypothyroidism-Associated Dyslipidemia: Potential Molecular Mechanisms Leading to NAFLD. Int. J. Mol. Sci. 2021, 22, 12797. https://doi.org/10.3390/ijms222312797
Mavromati M, Jornayvaz FR. Hypothyroidism-Associated Dyslipidemia: Potential Molecular Mechanisms Leading to NAFLD. International Journal of Molecular Sciences. 2021; 22(23):12797. https://doi.org/10.3390/ijms222312797
Chicago/Turabian StyleMavromati, Maria, and François R. Jornayvaz. 2021. "Hypothyroidism-Associated Dyslipidemia: Potential Molecular Mechanisms Leading to NAFLD" International Journal of Molecular Sciences 22, no. 23: 12797. https://doi.org/10.3390/ijms222312797






