Influence of Incorporation of Different dn-Electron Metal Cations into Biologically Active System on Its Biological and Physicochemical Properties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
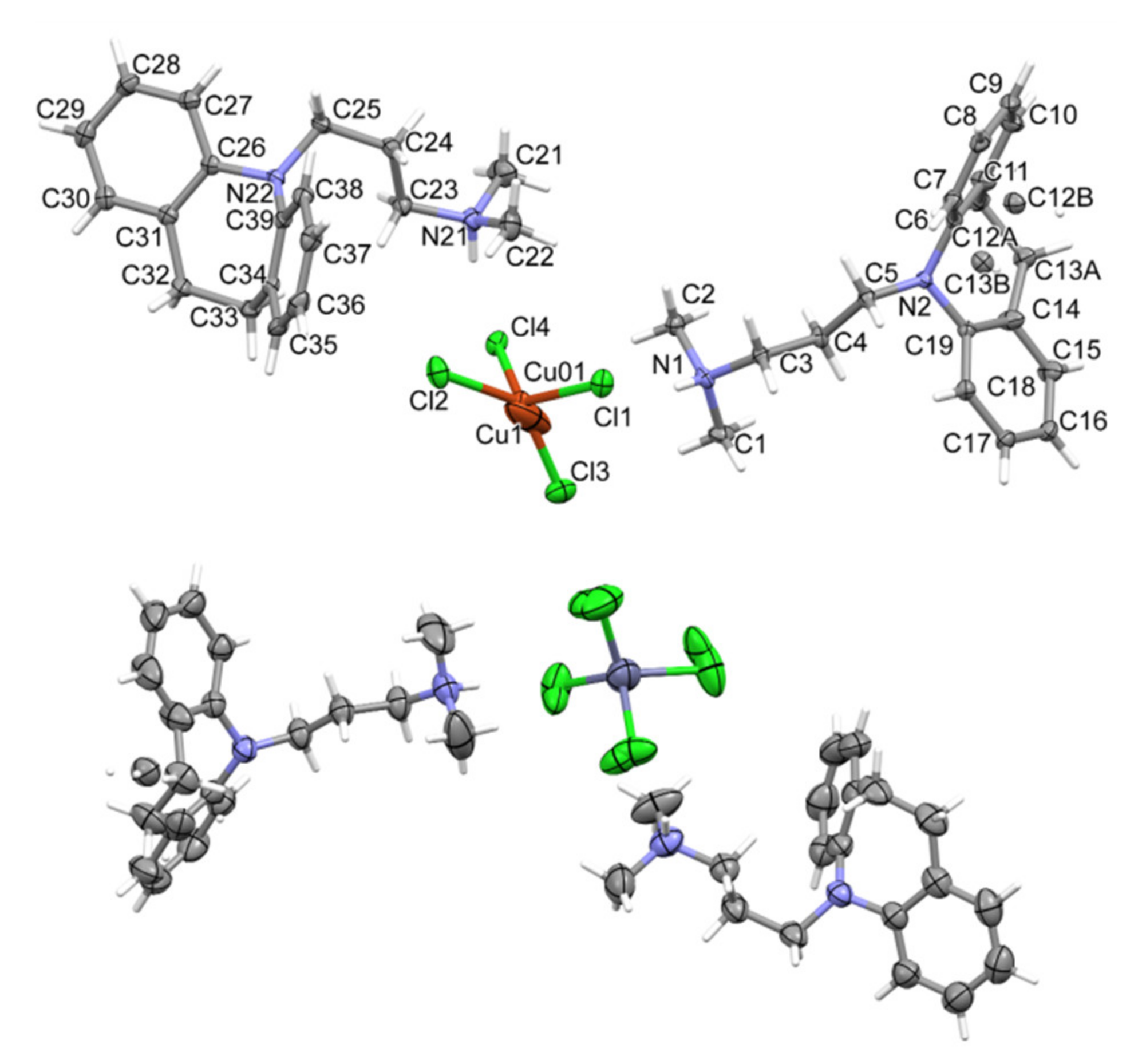
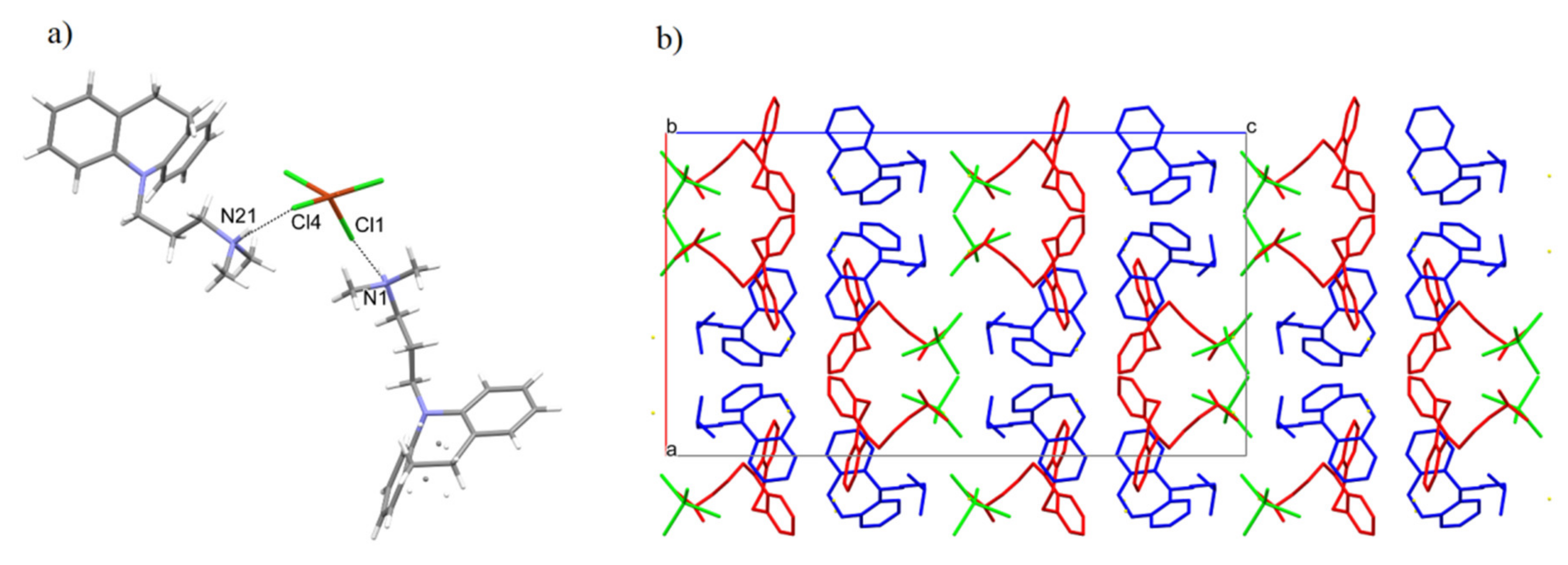
2.2. X-ray Diffraction Analysis
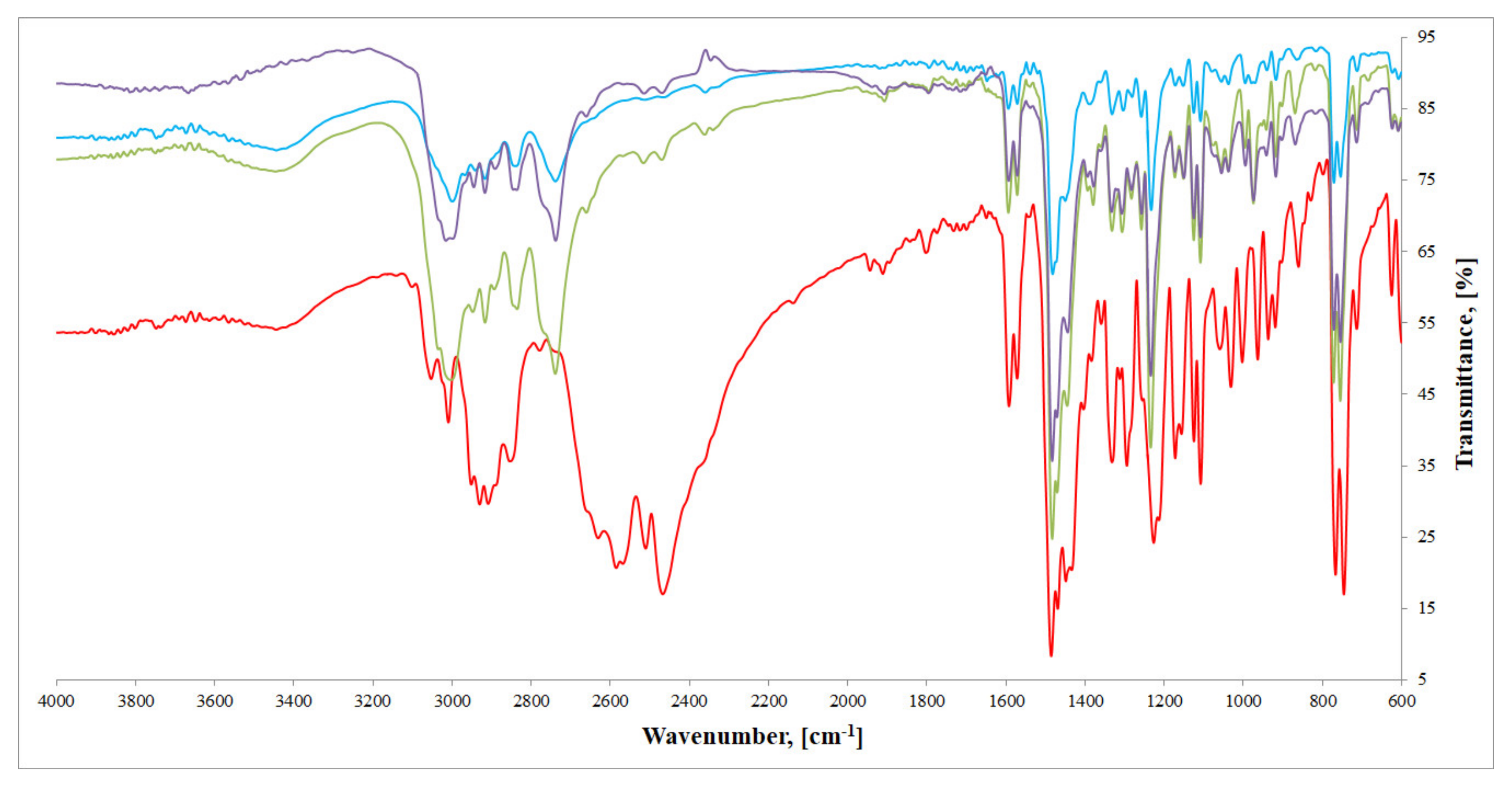
2.3. FTIR Spectra Analysis
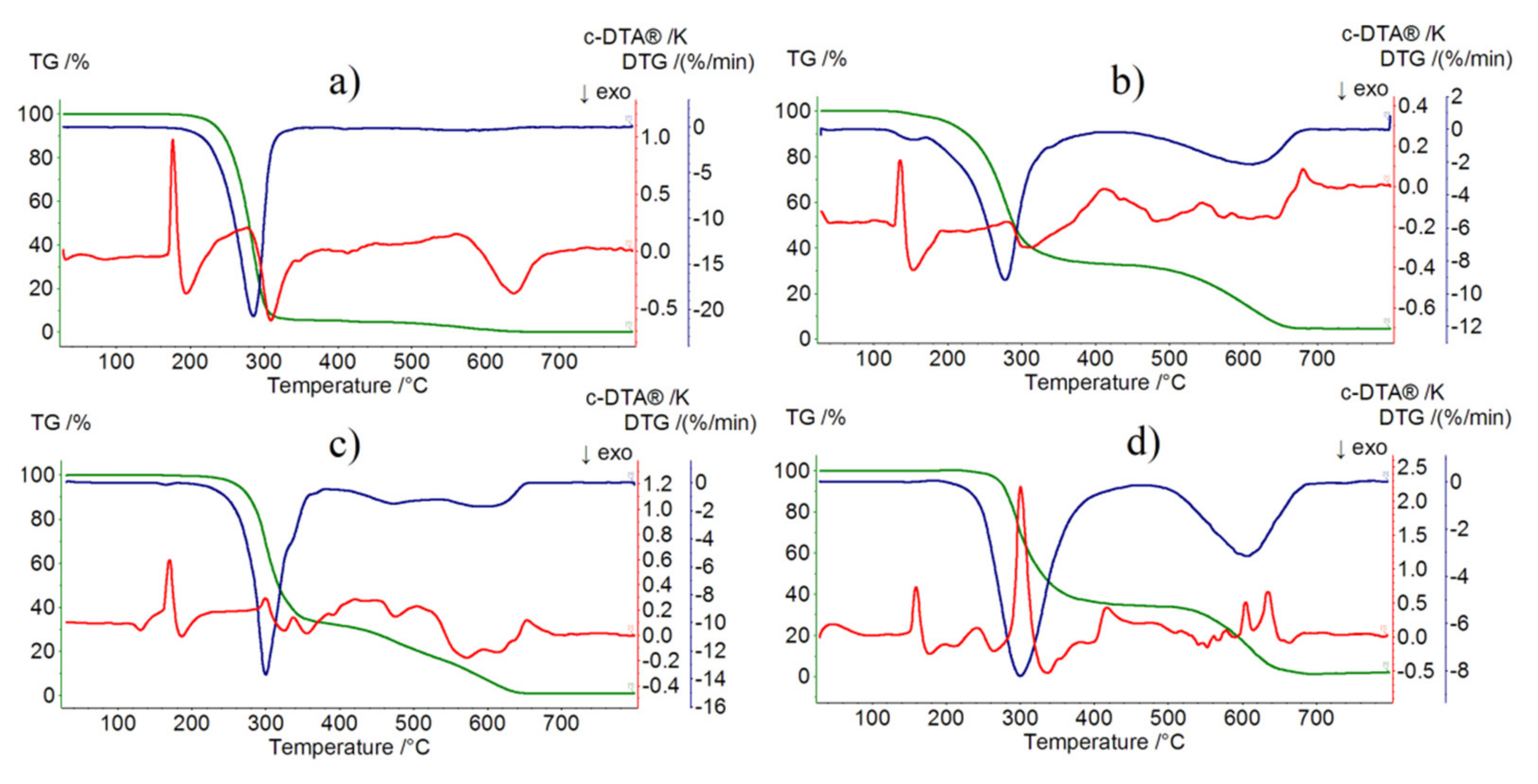
2.4. Thermogravimetric Analysis
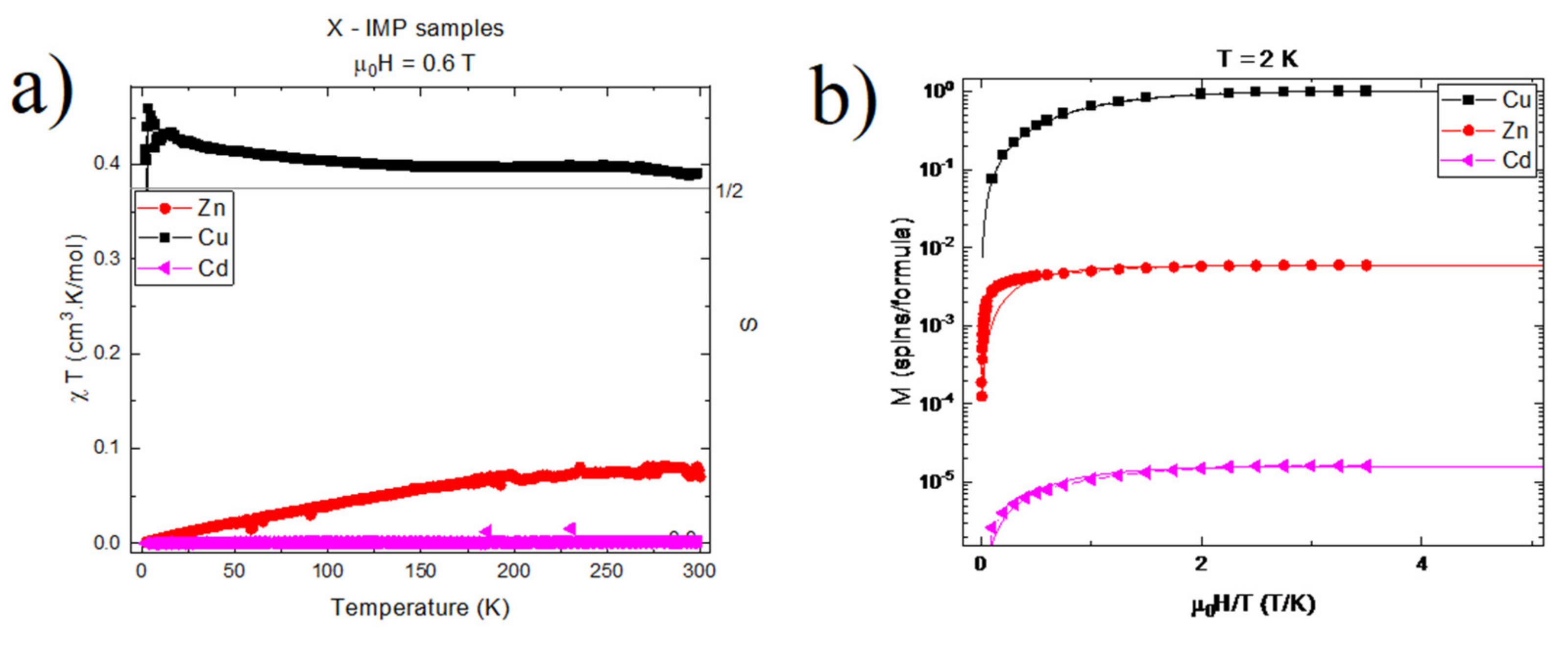
2.5. Magnetic Studies
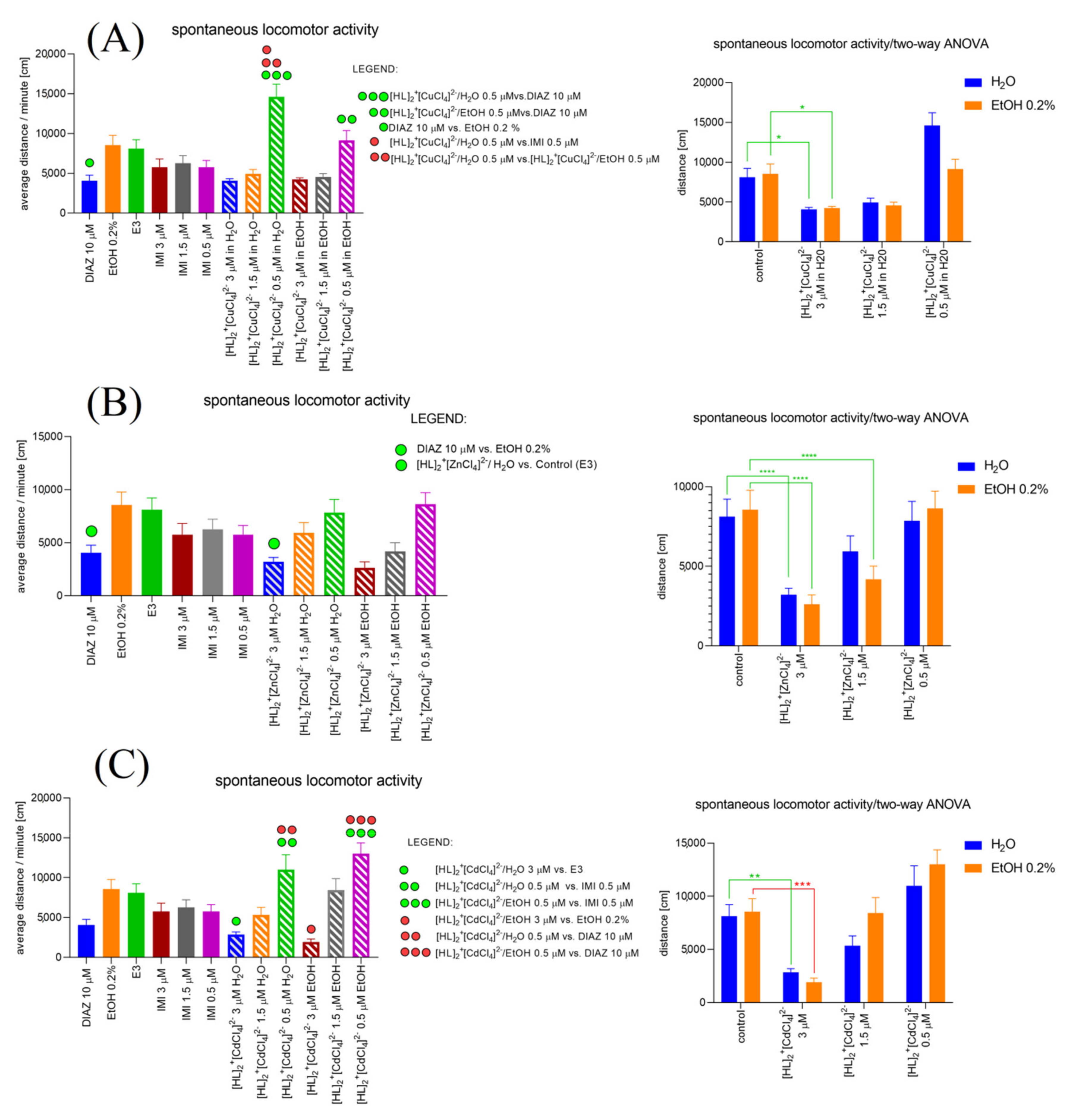
2.6. Biological Assays
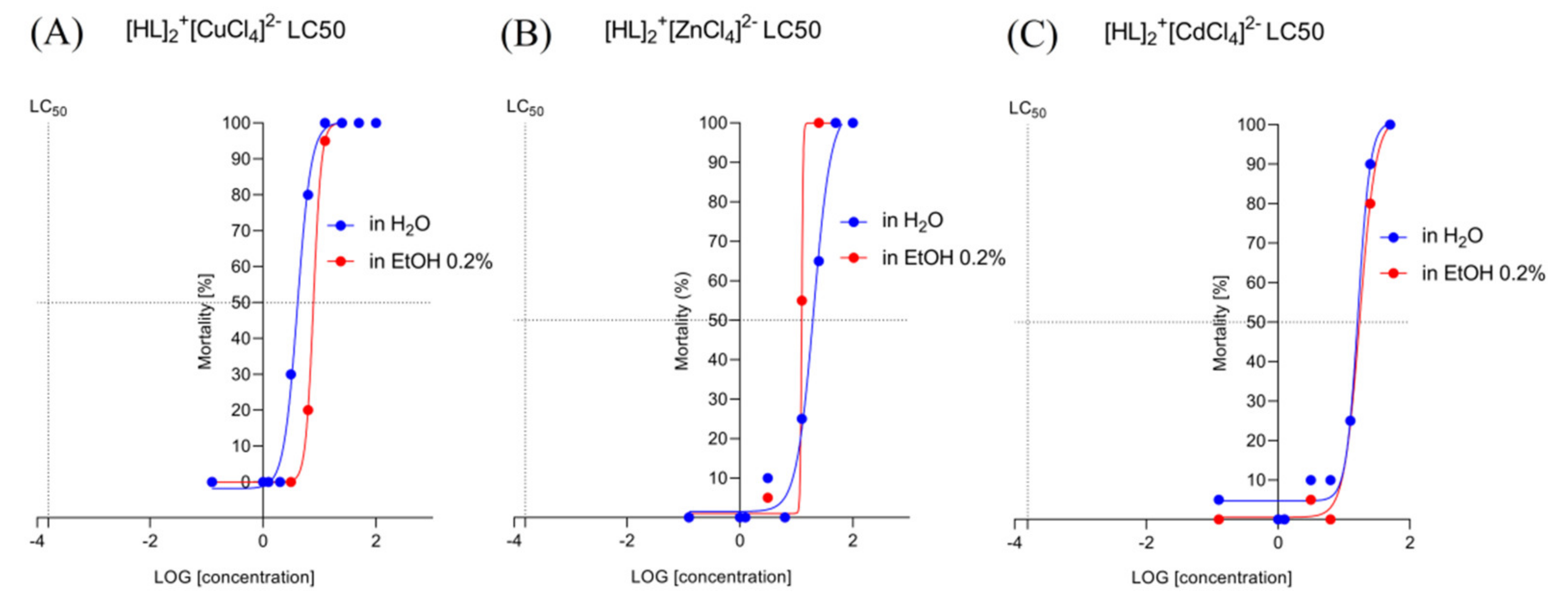
2.6.1. Determination of the Lethal Concentrations for [HL]2+[CuCl4]2− (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C) in a Solvent-Dependent Manner of Changes (Water, H2O/Ethanol, EtOH 0.2%)
2.6.2. Determination of the Heart Rate of Zebrafish Larvae in 0–5 dpf. The Solvent-Dependent Effect of [HL]2+[CuCl4]2− (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C) on the Heart Rate of Zebrafish Larvae in 48 hpf (Left) and 96 hpf (Right)
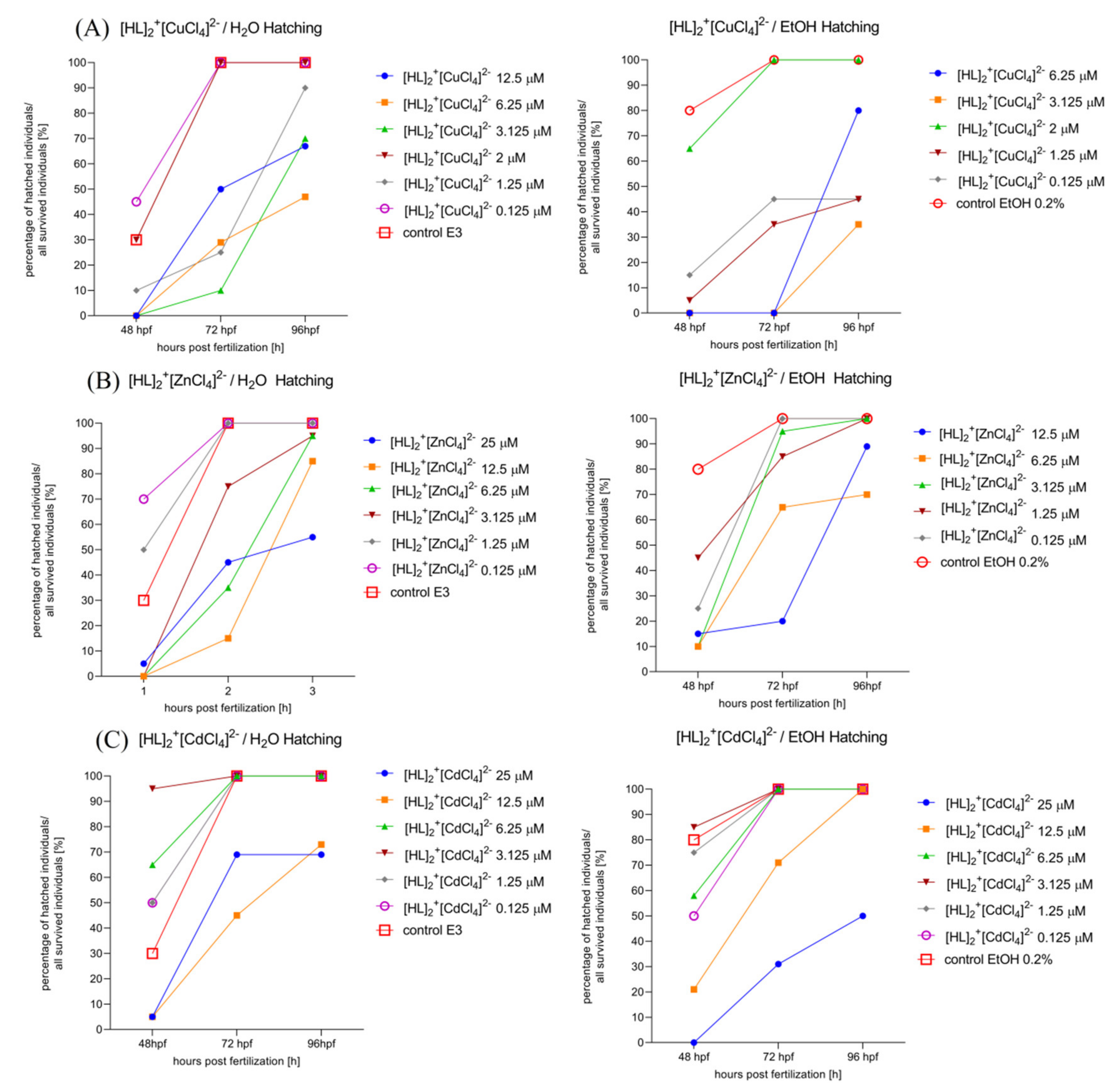
2.6.3. Determination of the Hatching Rate of Zebrafish Larvae in 0–5 dpf. Solvent-Dependent (Water (Left)/Ethanol (Right)) Effect of [HL]2+[CuCl4]2− (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C), Manner of Changes in the Hatching Rate after 48 hpf, 72 hpf, and 96 hpf of Zebrafish Larvae
2.6.4. [HL]2+[CuCl4]2− (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C) Induce Zebrafish Larvae’s Development Malformations in the FET Test (0–5 dpf)
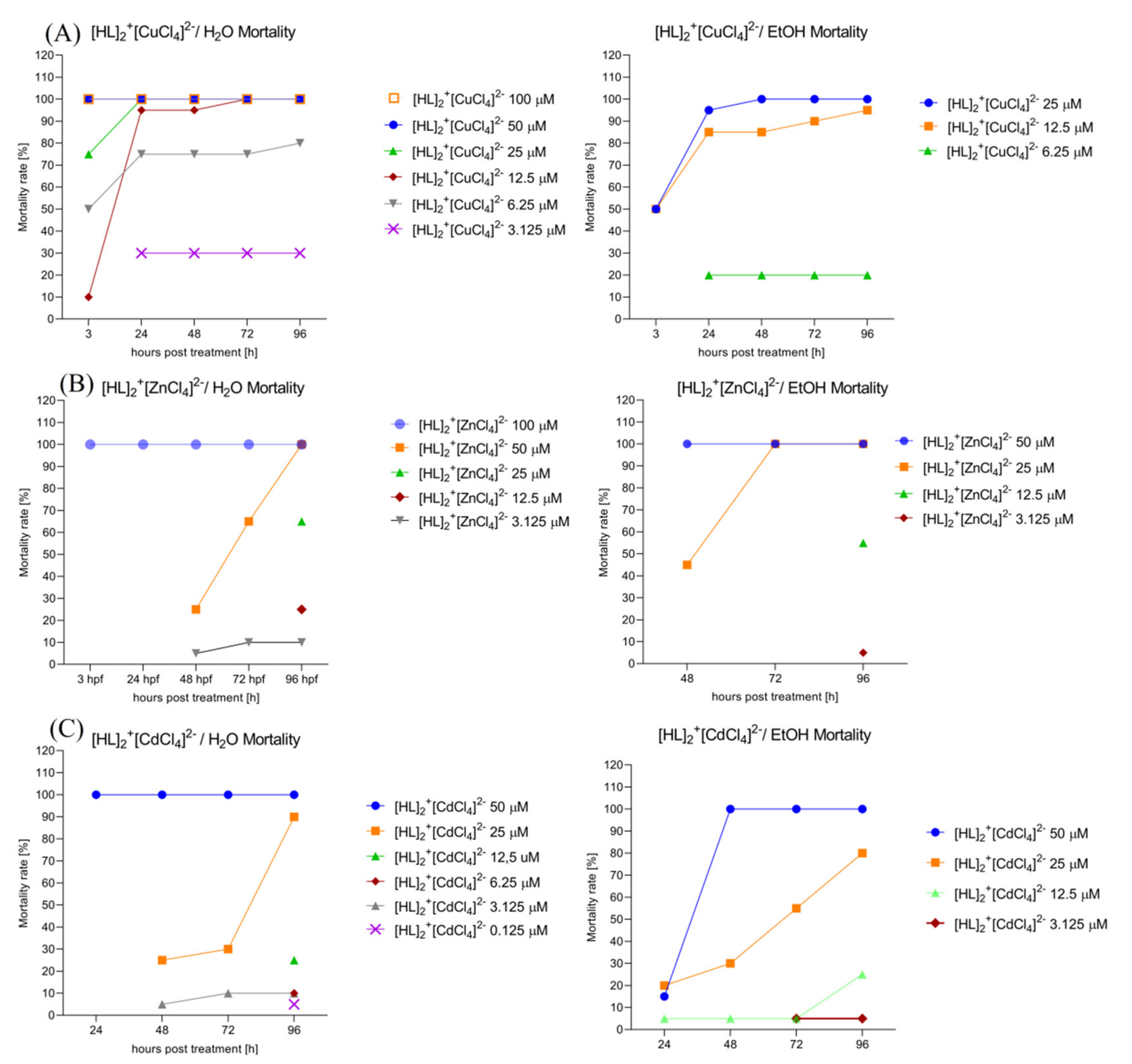
2.6.5. Assessment of the Mortality Rate within 96 h in Order to Choose the Safest Doses for Behavioral Study. [HL]2+[CuCl4]2− (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C) Solvent-Dependent (H2O, Left; EtOH 0.2%, right) Influence on the Zebrafish Larvae’s Mortality Rate
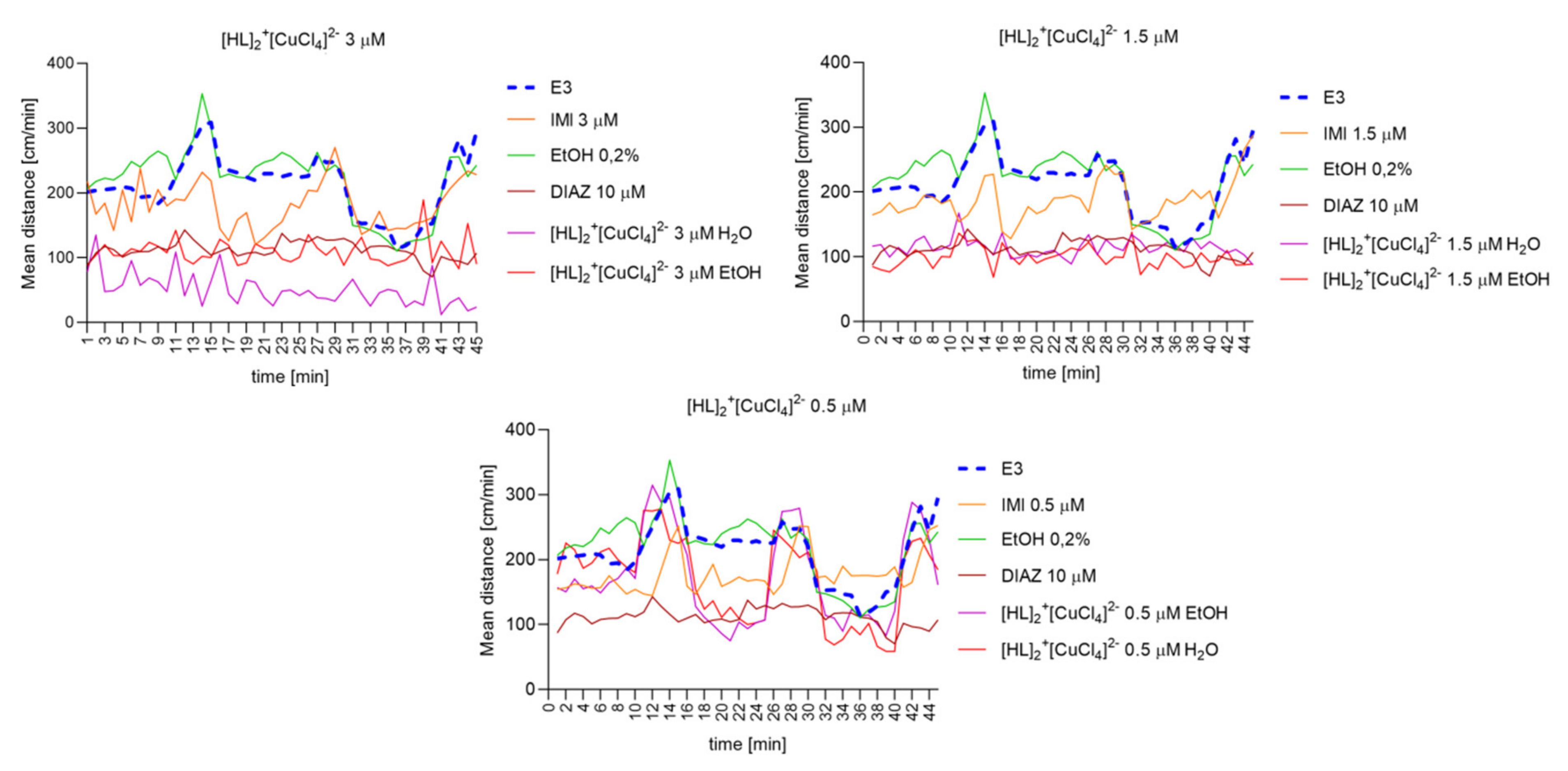
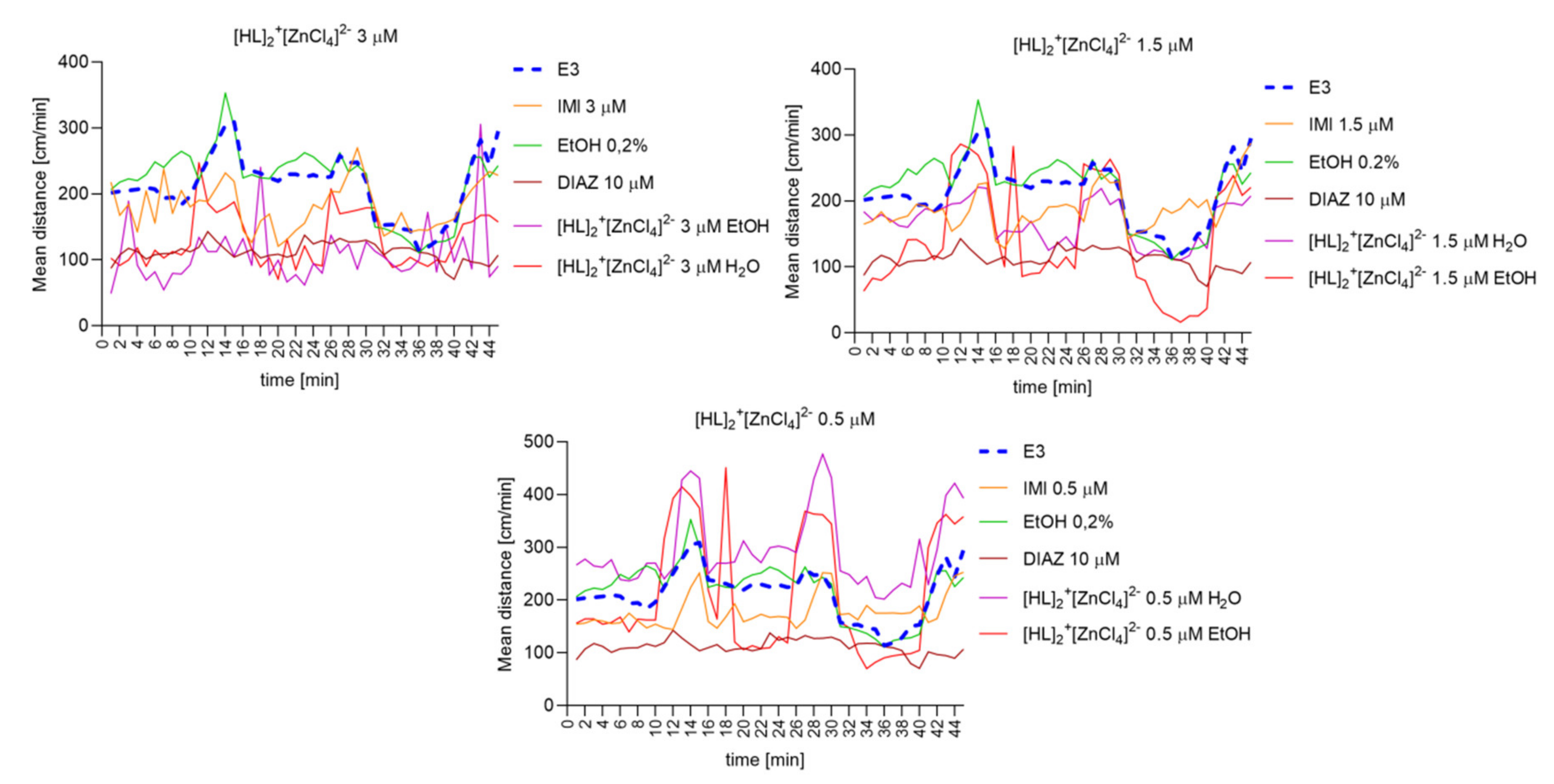
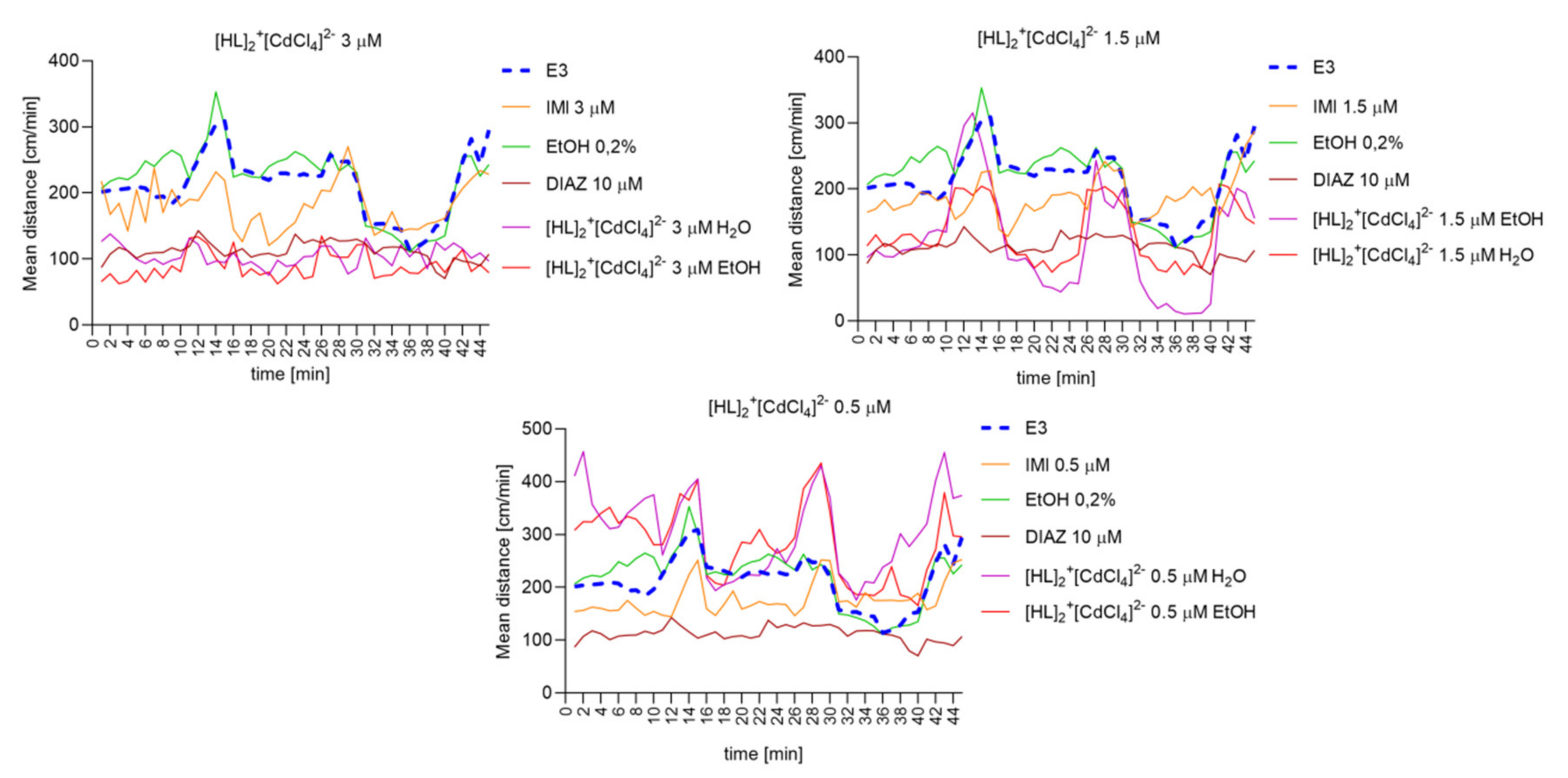
2.6.6. [HL]2+[CuCl4]2 (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C) Influence on the Spontaneous Locomotor Activity When Compared to Diazepam and Imipramine. Solvent-Dependent Effect Evaluation
2.6.7. [HL]2+[CuCl4]2− (A), [HL]2+[ZnCl4]2− (B), and [HL]2+[CdCl4]2− (C) Influences the Zebrafish Larvae’s Light–Dark Stimulated Locomotor Activity (cm) in a Solvent-Dependent Manner
2.6.8. The Changes Detected during 45 Min of the Locomotor Activity Test. Light-Dark-Dependent Activity. [HL]2+[CuCl4]2−, [HL]2+[ZnCl4]2−, and [HL]2+[CdCl4]2− Influence on The Light–Dark Dependent Locomotor Activity in a Solvent-Dependent Manner
3. Materials and Methods
3.1. Chemistry
3.2. X-ray Diffraction Analysis
3.3. Magnetic Studies
3.4. Biological Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sallee, F.R.; Pollock, B.G. Clinical pharmacokinetics of imipramine and desipramine. Clin. Pharmacokinet. 1990, 18, 346–364. [Google Scholar] [CrossRef] [PubMed]
- Vardanyan, R.S.; Hruby, V.J. Antidepressants. In Synthesis of Essential Drugs; Academic Press: Cambridge, MA, USA, 2006; pp. 103–116. [Google Scholar] [CrossRef]
- Cieslik, K.; Klenk-Majewska, B.; Danilczuk, Z.; Wrobel, A.; Lupina, T. Influence of zinc supplementation on imipramine effect in a chronic unpredictable stress (CUS) model in rats. Pharmacol. Rep. 2007, 59, 46–52. [Google Scholar] [PubMed]
- Cunha, M.P.; Machado, D.G.; Bettino, L.E.B.; Capra, J.C.; Rodrigues, A.L.S. Interaction of zinc with antidepressants in the tail suspension test. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2008, 32, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Szewczyk, B.; Branski, P.; Wieronska, J.M.; Palucha, A.; Pilc, A.; Nowak, G. Interaction of zinc with antidepressants in the forced swimming test in mice. Pol. J. Pharmacol. 2002, 54, 681–685. [Google Scholar]
- Siwek, M.; Dudek, D.; Paul, I.A.; Sowa-Kucma, M.; Zieba, A.; Popik, P.; Pilc, A.; Nowak, G. Zinc supplementation augments efficacy of imipramine in treatment resistant patients: A double blind, placebo-controlled study. J. Affect. Disord. 2009, 118, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Markin, P.A.; Brito, A.; Moskaleva, N.E.; Tagliaro, F.; La Frano, M.R.; Savitskii, M.V.; Appolonova, S.A. Short- and long-term exposures of the synthetic cannabinoid 5F-APINAC induce metabolomic alterations associated with neurotransmitter systems and embryotoxicity confirmed by teratogenicity in zebrafish. Comp. Biochem. Physiol. Toxicol. Pharmacol. CBP 2021, 243, 109000. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Stewart, A.M.; Gerlai, R. Zebrafish as an emerging model for studying complex brain disorders. Trends Pharmacol. Sci. 2014, 35, 63–75. [Google Scholar] [CrossRef] [Green Version]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, E.; McLaren, K.; Teucke, M.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.C.; Priyadarshini, M.; Panula, P. Complementary developmental expression of the two tyrosine hydroxylase transcripts in zebrafish. Histochem. Cell Biol. 2009, 132, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Maciąg, M.; Michalak, A.; Skalicka-Woźniak, K.; Zykubek, M.; Ciszewski, A.; Budzyńska, B. Zebrafish and mouse models for anxiety evaluation—A comparative study with xanthotoxin as a model compound. Brain Res. Bull. 2020, 165, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Mathuru, A.S.; Jesuthasan, S. The medial habenula as a regulator of anxiety in adult zebrafish. Front. Neural Circuits 2013, 7, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziv, L.; Muto, A.; Schoonheim, P.J.; Meijsing, S.H.; Strasser, D.; Ingraham, H.A.; Schaaf, M.J.; Yamamoto, K.R.; Baier, H. An affective disorder in zebrafish with mutation of the glucocorticoid receptor. Mol. Psychiatry 2013, 18, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, B.B.; Schoonheim, P.J.; Ziv, L.; Voelker, L.; Baier, H.; Gahtan, E. A zebrafish model of glucocorticoid resistance shows serotonergic modulation of the stress response. Front. Behav. Neurosci. 2012, 11, 68. [Google Scholar] [CrossRef] [Green Version]
- Kyzar, E.; Stewart, A.M.; Landsman, S.; Collins, C.; Gebhardt, M.; Robinson, K.; Kalueff, A.V. Behavioral effects of bidirectional modulators of brain monoamines reserpine and d-amphetamine in zebrafish. Brain Res. 2013, 21, 108–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maximino, C.; Marques de Brito, T.; Dias, C.A.; Gouveia, A.; Morato, S., Jr. Scototaxis as anxiety-like behavior in fish. Nat. Protoc. 2010, 5, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Giacomini, A.C.V.V.; Piassetta, A.S.; Genario, R.; Bonan, C.D.; Piato, A.; Barcellos, L.J.G.; de Abreu, M.S. Tryptophan alleviates neuroendocrine and behavioral responses to stress in zebrafish. Behav. Brain Res. 2020, 27, 112264. [Google Scholar] [CrossRef] [PubMed]
- Anichtchik, O.; Sallinen, V.; Peitsaro, N.; Panula, P. Distinct structure and activity of monoamine oxidase in the brain of zebrafish (Danio rerio). J. Comp. Neurol. 2006, 498, 593–610. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, L.; Aceto, S.; Agnisola, C.; De Paolo, S.; Dipineto, L.; Stilling, R.M.; Dinan, T.G.; Cryan, J.F.; Menna, L.F.; Fioretti, A. Probiotic modulation of the microbiota-gut-brain axis and behaviour in zebrafish. Sci. Rep. 2016, 6, 30046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fábián, L.; Kálmán, A. Volumetric measure of isostructurality. Acta Cryst. 1999, B55, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J.; Davis, R.E.; Shimoni, L.; Chang, N.-L. Patterns in hydrogen bonding: Functionality and graph set analysis in crystals. Angew. Chem. Int. Ed. Engl. 1995, 34, 1555–1573. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Crystallogr. Sect. B Struct. Sci. 2016, B72, 171179. [Google Scholar] [CrossRef]
- Kamel, L.T.; El Essawi, Μ.; Wartchow, R.; Berthold, H.J. Crystal structure of bis [(10, 11-dihydro-dibenzo [b, f lazepin-5-yl)-2-methyl-propyl-dimethyl-ammonium] tetrachlorocuprate (II),(C2OH27N2) 2 [CUCI4]. Ζ. Kristallogr. NCS 2001, 216, 359–360. [Google Scholar]
- Zhou, S.; Shen, S.; Zhao, D.; Zhang, Z.; Yan, S. Evaporation and decomposition of eutectics of cupric chloride and sodium chloride. J. Therm. Anal. Calorim. 2017, 129, 1445–1452. [Google Scholar] [CrossRef]
- CrysAlisPro 1. 171.39.33c. Rigaku Oxford Diffraction; Agilent Technologies UK Ltd.: Yarnton, UK, 2017. [Google Scholar]
- Sheldrick, G.M. SHELXT-Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; Mccabe, P.; Pidcock, E.; Rodriguez–Monge, L.; Taylor, R.; Van De Streek, J.; Wood, P.A. Mercury: Visualization and analysis of crystal structures. J. Appl. Cryst. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Westrip, S.P. publCIF: Software for editing, validating and formatting crystallographic information files. J. Appl. Cryst. 2010, 43, 920–925. [Google Scholar] [CrossRef] [Green Version]
- Tran, S.; Gerlai, R. Time-course of behavioural changes induced by ethanol in zebrafish (Danio rerio). Behav. Brain Res. 2013, 252, 204–213. [Google Scholar] [CrossRef] [Green Version]
- Gerlai, R.; Lahav, M.; Guo, S.; Rosenthal, A. Drinks like a fish: Zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol. Biochem. Behav. 2000, 67, 773–782. [Google Scholar] [CrossRef]


| [HL]2+[CuCl4]2− | [HL]2+[ZnCl4]2− | |
|---|---|---|
| Crystal data | ||
| Chemical formula | Cl4Cu·2(C19H25N2) | Cl4Zn·2(C19H25N2) |
| Mr | 768.16 | 769.99 |
| Crystal system, space group | Orthorhombic, Pbca | Orthorhombic, Pbca |
| Temperature (K) | 100 | 294 |
| a, b, c (Å) | 18.8810 (3), 11.6982 (2), 33.9528 (5) | 19.2344 (5), 11.6465 (3), 34.6682 (8) |
| V (Å3) | 7499.3 (2) | 7766.1 (3) |
| Z | 8 | 8 |
| Radiation type | Mo Kα | Mo Kα |
| No. of measured, independent and observed [I > 2σ(I)] reflections | 37,197, 37,197, 29,899 | 124,476, 12,164, 7813 |
| R(F2 > 2σ(F2)), wR(F2), S | 0.048, 0.121, 1.04 | 0.044, 0.108, 1.01 |
| No. of reflections | 37,197 | 12,164 |
| No. of parameters | 465 | 500 |
| Δ〉max, Δ〉min (e Å−3) | 0.77, −0.72 | 0.25, −0.34 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rogalewicz, B.; Szczesio, M.; Poleszak, E.; Kowalczyk, J.; Szewczyk, B.; Camargo, B.C.; Szczytko, J.; Witkowski, M.; Fruziński, A.; Raducka, A.; et al. Influence of Incorporation of Different dn-Electron Metal Cations into Biologically Active System on Its Biological and Physicochemical Properties. Int. J. Mol. Sci. 2021, 22, 12909. https://doi.org/10.3390/ijms222312909
Rogalewicz B, Szczesio M, Poleszak E, Kowalczyk J, Szewczyk B, Camargo BC, Szczytko J, Witkowski M, Fruziński A, Raducka A, et al. Influence of Incorporation of Different dn-Electron Metal Cations into Biologically Active System on Its Biological and Physicochemical Properties. International Journal of Molecular Sciences. 2021; 22(23):12909. https://doi.org/10.3390/ijms222312909
Chicago/Turabian StyleRogalewicz, Bartłomiej, Małgorzata Szczesio, Ewa Poleszak, Joanna Kowalczyk, Bernadeta Szewczyk, Bruno Cury Camargo, Jacek Szczytko, Marcin Witkowski, Andrzej Fruziński, Anita Raducka, and et al. 2021. "Influence of Incorporation of Different dn-Electron Metal Cations into Biologically Active System on Its Biological and Physicochemical Properties" International Journal of Molecular Sciences 22, no. 23: 12909. https://doi.org/10.3390/ijms222312909











