Kinetic Study on the Reactivity of Azanone (HNO) toward Cyclic C-Nucleophiles
Abstract
:1. Introduction
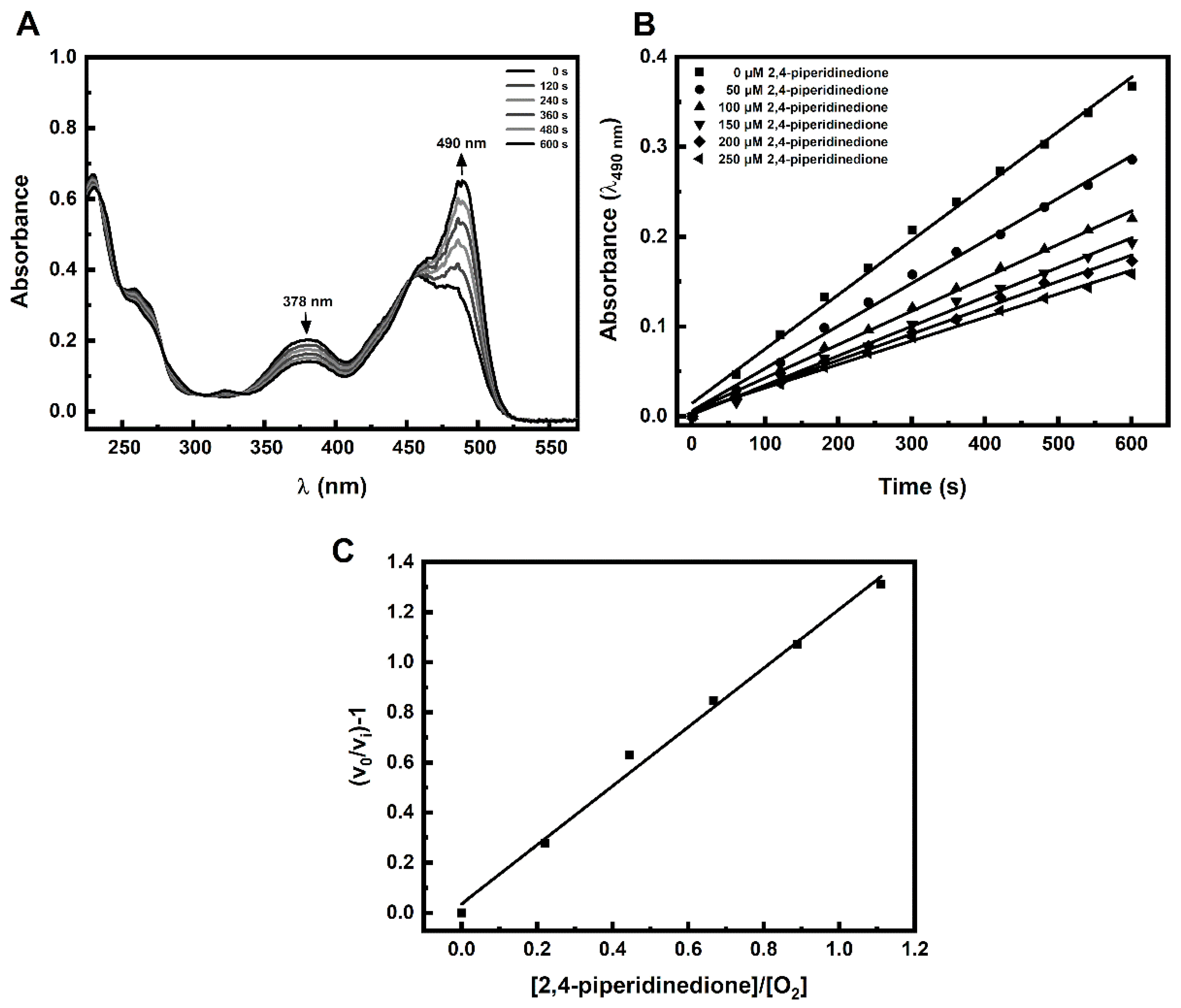
2. Results and Discussion
3. Materials and Methods
3.1. Equipment
3.2. Chemicals
3.3. Kinetic Experiments
3.4. Computational Details
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ignarro, L.J.; Byrns, R.E.; Buga, G.M.; Wood, K.S. Endothelium-derived relaxing factor from pulmonary artery and vein possesses pharmacologic and chemical properties identical to those of nitric oxide radical. Circ. Res. 1987, 61, 866–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerwin, J.F., Jr.; Lancaster, J.R., Jr.; Feldman, P.L. Nitric oxide: A new paradigm for second messengers. J. Med. Chem. 1995, 38, 4343–4362. [Google Scholar] [CrossRef]
- Bianco, C.L.; Toscano, J.P.; Bartberger, M.D.; Fukuto, J.M. The chemical biology of HNO signaling. Arch. Biochem. Biophys. 2017, 617, 129–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R. Shared signaling pathways among gasotransmitters. Proc. Natl. Acad. Sci. USA 2012, 109, 8801–8802. [Google Scholar] [CrossRef] [Green Version]
- Fukuto, J.M. A recent history of nitroxyl chemistry, pharmacology and therapeutic potential. Br. J. Pharmacol. 2019, 176, 135–146. [Google Scholar] [CrossRef] [Green Version]
- Kemp-Harper, B.K.; Horowitz, J.D.; Ritchie, R.H. Therapeutic Potential of Nitroxyl (HNO) Donors in the Management of Acute Decompensated Heart Failure. Drugs 2016, 76, 1337–1348. [Google Scholar] [CrossRef]
- Kemp-Harper, B.K.; Velagic, A.; Paolocci, N.; Horowitz, J.D.; Ritchie, R.H. Cardiovascular Therapeutic Potential of the Redox Siblings, Nitric Oxide (NO•) and Nitroxyl (HNO), in the Setting of Reactive Oxygen Species Dysregulation. Handb. Exp. Pharmacol. 2021, 264, 311–337. [Google Scholar] [CrossRef] [PubMed]
- Felker, G.M.; Borentain, M.; Cleland, J.G.; DeSouza, M.M.; Kessler, P.D.; O’Connor, C.M.; Seiffert, D.; Teerlink, J.R.; Voors, A.A.; McMurray, J.J.V. Rationale and design for the development of a novel nitroxyl donor in patients with acute heart failure. Eur. J. Heart Fail. 2019, 21, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Shafirovich, V.; Lymar, S.V. Nitroxyl and its anion in aqueous solutions: Spin states, protic equilibria, and reactivities toward oxygen and nitric oxide. Proc. Natl. Acad. Sci. USA 2002, 99, 7340–7345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miao, Z.; King, S.B. Recent advances in the chemical biology of nitroxyl (HNO) detection and generation. Nitric Oxide Biol. Chem. 2016, 57, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angeli, A. Sopra la nitroidrossilammina. Gazz. Chim. Ital. 1896, 26, 17–25. [Google Scholar]
- Piloty, O. Ueber eine oxydation des hydroxylamins durch benzolsulfochlorid. Ber. Dtsch. Chem. Ges. 1896, 29, 1559–1567. [Google Scholar] [CrossRef] [Green Version]
- Aizawa, K.; Nakagawa, H.; Matsuo, K.; Kawai, K.; Ieda, N.; Suzuki, T.; Miyata, N. Piloty’s acid derivative with improved nitroxyl-releasing characteristics. Bioorganic Med. Chem. Lett. 2013, 23, 2340–2343. [Google Scholar] [CrossRef]
- Cline, M.R.; Tu, C.; Silverman, D.N.; Toscano, J.P. Detection of nitroxyl (HNO) by membrane inlet mass spectrometry. Free Radic. Biol. Med. 2011, 50, 1274–1279. [Google Scholar] [CrossRef] [PubMed]
- Sirsalmath, K.; Suárez, S.A.; Bikiel, D.E.; Doctorovich, F. The pH of HNO donation is modulated by ring substituents in Piloty’s acid derivatives: Azanone donors at biological pH. J. Inorg. Biochem. 2013, 118, 134–139. [Google Scholar] [CrossRef]
- Smulik-Izydorczyk, R.; Rostkowski, M.; Gerbich, A.; Jarmoc, D.; Adamus, J.; Leszczyńska, A.; Michalski, R.; Marcinek, A.; Kramkowski, K.; Sikora, A. Decomposition of Piloty’s acid derivatives—Toward the understanding of factors controlling HNO release. Arch. Biochem. Biophys. 2019, 661, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Salmon, D.J.; Torres de Holding, C.L.; Thomas, L.; Peterson, K.V.; Goodman, G.P.; Saavedra, J.E.; Srinivasan, A.; Davies, K.M.; Keefer, L.K.; Miranda, K.M. HNO and NO release from a primary amine-based diazeniumdiolate as a function of pH. Inorg. Chem. 2011, 50, 3262–3270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miranda, K.M.; Katori, T.; Torres de Holding, C.L.; Thomas, L.; Ridnour, L.A.; McLendon, W.J.; Cologna, S.M.; Dutton, A.S.; Champion, H.C.; Mancardi, D.; et al. Comparison of the NO and HNO donating properties of diazeniumdiolates: Primary amine adducts release HNO in Vivo. J. Med. Chem. 2005, 48, 8220–8228. [Google Scholar] [CrossRef]
- Bharadwaj, G.; Benini, P.G.; Basudhar, D.; Ramos-Colon, C.N.; Johnson, G.M.; Larriva, M.M.; Keefer, L.K.; Andrei, D.; Miranda, K.M. Analysis of the HNO and NO donating properties of alicyclic amine diazeniumdiolates. Nitric Oxide Biol. Chem. 2014, 42, 70–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, X.; Chen, K.; Shi, G.; Lin, W.; Bai, H.; Li, H.; Tang, G.; Wang, C. Design and tuning of ionic liquid-based HNO donor through intramolecular hydrogen bond for efficient inhibition of tumor growth. Sci. Adv. 2020, 6, eabb7788. [Google Scholar] [CrossRef] [PubMed]
- Sha, X.; Isbell, T.S.; Patel, R.P.; Day, C.S.; King, S.B. Hydrolysis of acyloxy nitroso compounds yields nitroxyl (HNO). J. Am. Chem. Soc. 2006, 128, 9687–9692. [Google Scholar] [CrossRef] [PubMed]
- Shoman, M.E.; DuMond, J.F.; Isbell, T.S.; Crawford, J.H.; Brandon, A.; Honovar, J.; Vitturi, D.A.; White, C.R.; Patel, R.P.; King, S.B. Acyloxy nitroso compounds as nitroxyl (HNO) donors: Kinetics, reactions with thiols, and vasodilation properties. J. Med. Chem. 2011, 54, 1059–1070. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, H.A.; Abdel-Aziz, M.; Abuo-Rahma Gel, D.; King, S.B. New acyloxy nitroso compounds with improved water solubility and nitroxyl (HNO) release kinetics and inhibitors of platelet aggregation. Bioorganic Med. Chem. 2015, 23, 6069–6077. [Google Scholar] [CrossRef] [Green Version]
- Guthrie, D.A.; Ho, A.; Takahashi, C.G.; Collins, A.; Morris, M.; Toscano, J.P. “Catch-and-release” of HNO with pyrazolones. J. Org. Chem. 2015, 80, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, D.A.; Nourian, S.; Takahashi, C.G.; Toscano, J.P. Curtailing the hydroxylaminobarbituric acid-hydantoin rearrangement to favor HNO generation. J. Org. Chem. 2015, 80, 1349–1356. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, D.A.; Kim, N.Y.; Siegler, M.A.; Moore, C.D.; Toscano, J.P. Development of N-substituted hydroxylamines as efficient nitroxyl (HNO) donors. J. Am. Chem. Soc. 2012, 134, 1962–1965. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, D.; Nourian, S.; Toscano, J. Hydroxylamines with Organic-Based Leaving Groups as HNO Donors; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Smulik, R.; Dębski, D.; Zielonka, J.; Michałowski, B.; Adamus, J.; Marcinek, A.; Kalyanaraman, B.; Sikora, A. Nitroxyl (HNO) reacts with molecular oxygen and forms peroxynitrite at physiological pH. Biological Implications. J. Biol. Chem. 2014, 289, 35570–35581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smulik-Izydorczyk, R.; Dębowska, K.; Rostkowski, M.; Adamus, J.; Michalski, R.; Sikora, A. Kinetics of Azanone (HNO) Reactions with Thiols: Effect of pH. Cell Biochem. Biophys. 2021, 79, 845–856. [Google Scholar] [CrossRef]
- Smulik-Izydorczyk, R.; Mesjasz, A.; Gerbich, A.; Adamus, J.; Michalski, R.; Sikora, A. A kinetic study on the reactivity of azanone (HNO) toward its selected scavengers: Insight into its chemistry and detection. Nitric Oxide Biol. Chem. 2017, 69, 61–68. [Google Scholar] [CrossRef]
- Prolo, C.; Rios, N.; Piacenza, L.; Álvarez, M.N.; Radi, R. Fluorescence and chemiluminescence approaches for peroxynitrite detection. Free Radic. Biol. Med. 2018, 128, 59–68. [Google Scholar] [CrossRef]
- Rios, N.; Piacenza, L.; Trujillo, M.; Martínez, A.; Demicheli, V.; Prolo, C.; Álvarez, M.N.; López, G.V.; Radi, R. Sensitive detection and estimation of cell-derived peroxynitrite fluxes using fluorescein-boronate. Free Radic. Biol. Med. 2016, 101, 284–295. [Google Scholar] [CrossRef] [PubMed]
- Sieracki, N.A.; Gantner, B.N.; Mao, M.; Horner, J.H.; Ye, R.D.; Malik, A.B.; Newcomb, M.E.; Bonini, M.G. Bioluminescent detection of peroxynitrite with a boronic acid-caged luciferin. Free Radic. Biol. Med. 2013, 61, 40–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sikora, A.; Zielonka, J.; Dębowska, K.; Michalski, R.; Smulik-Izydorczyk, R.; Pięta, J.; Podsiadły, R.; Artelska, A.; Pierzchała, K.; Kalyanaraman, B. Boronate-Based Probes for Biological Oxidants: A Novel Class of Molecular Tools for Redox Biology. Front. Chem. 2020, 8, 580899. [Google Scholar] [CrossRef] [PubMed]
- Sikora, A.; Zielonka, J.; Lopez, M.; Joseph, J.; Kalyanaraman, B. Direct oxidation of boronates by peroxynitrite: Mechanism and implications in fluorescence imaging of peroxynitrite. Free Radic. Biol. Med. 2009, 47, 1401–1407. [Google Scholar] [CrossRef] [Green Version]
- Dickinson, B.C.; Huynh, C.; Chang, C.J. A palette of fluorescent probes with varying emission colors for imaging hydrogen peroxide signaling in living cells. J. Am. Chem. Soc. 2010, 132, 5906–5915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, V.; Carroll, K.S. Profiling the Reactivity of Cyclic C-Nucleophiles towards Electrophilic Sulfur in Cysteine Sulfenic Acid. Chem. Sci. 2016, 7, 400–415. [Google Scholar] [CrossRef] [Green Version]
- Hughes, M.N.; Cammack, R. Synthesis, chemistry, and applications of nitroxyl ion releasers sodium trioxodinitrate or Angeli’s salt and Piloty’s acid. Methods Enzymol. 1999, 301, 279–287. [Google Scholar] [CrossRef]
- Beckman, J.S.; Koppenol, W.H. Nitric oxide, superoxide, and peroxynitrite: The good, the bad, and ugly. Am. J. Physiol. 1996, 271, C1424–C1437. [Google Scholar] [CrossRef] [Green Version]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, Revision E.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Cramer, C.J.; Truhlar, D.G. Implicit Solvation Models: Equilibria, Structure, Spectra, and Dynamics. Chem. Rev. 1999, 99, 2161–2200. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [Green Version]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef]
- Goerigk, L.; Grimme, S. Efficient and Accurate Double-Hybrid-Meta-GGA Density Functionals—Evaluation with the Extended GMTKN30 Database for General Main Group Thermochemistry, Kinetics, and Noncovalent Interactions. J. Chem. Theory Comput. 2011, 7, 291–309. [Google Scholar] [CrossRef] [PubMed]
- Wiberg, K.B. Ab Initio Molecular Orbital Theory by Hehre, W.J., Radom, L., Schleyer, P.v.R. and Pople, J.A., John Wiley, New York, 548p. Price: $79.95 (1986). J. Comput. Chem. 1986, 7, 379. [Google Scholar] [CrossRef]
- Goerigk, L.; Hansen, A.; Bauer, C.; Ehrlich, S.; Najibi, A.; Grimme, S. A look at the density functional theory zoo with the advanced GMTKN55 database for general main group thermochemistry, kinetics and noncovalent interactions. Phys. Chem. Chem. Phys. 2017, 19, 32184–32215. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Scavenger | kS/kOxygen | kS (M−1s−1) | ΔG‡(kJ/mol) |
|---|---|---|---|
| 1,3-Cyclopentanedione (1) | 0.016 ± 0.001 | (2.8 ± 0.6) × 102 | 72.53 |
| 2-Methyl-1,3-cyclopentanedione (2) | 0.18 ± 0.02 | (3.2 ± 0.9) × 103 | 57.92 |
| 2-Acetyl-1,3-cyclopentanedione (3) | (1.8 ± 0.2) × 10−3 [a] | (3.3 ± 0.8) × 101 [a] | 90.93 |
| 1,3-Cyclohexanedione (4) | 0.12 ± 0.01 | (2.2 ± 0.4) × 103 | 59.28 |
| 2-Methyl-1,3-cyclohexanedione (5) | 0.61 ± 0.04 | (1.1 ± 0.2) × 104 | 50.03 |
| Dimedone (6) | 0.14 ± 0.01 | (2.5 ± 0.6) × 103 | 58.46 |
| 1,3-Cycloheptanedione (7) | 0.38 ± 0.02 | (6.8 ± 1.5) × 103 | 57.52 |
| 2,4-Piperidinedione (8) | 1.13 ± 0.03 | (2.0 ± 0.4) × 104 | 45.65 |
| 1-(4-Methoxybenzyl)-2,4-piperidinedione (9) | 0.80 ± 0.03 | (1.4 ± 0.3) × 104 | 41.34 |
| 1,3-Dimethylbarbituric acid (10) | 0.72 ± 0.03 | (1.3 ± 0.3) × 104 | 50.93 |
| Barbituric acid (11) | 0.20 ± 0.01 | (3.5 ± 0.8) × 103 | 64.65 |
| 2-Thiobarbituric acid (12) | 0.046 ± 0.003 | (8.2 ± 1.9) × 102 | 76.28 |
| Meldrum’s acid (13) | 0.048 ± 0.002 | (8.7 ± 1.8) × 102 | 70.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Artelska, A.; Rola, M.; Rostkowski, M.; Pięta, M.; Pięta, J.; Michalski, R.; Sikora, A.B. Kinetic Study on the Reactivity of Azanone (HNO) toward Cyclic C-Nucleophiles. Int. J. Mol. Sci. 2021, 22, 12982. https://doi.org/10.3390/ijms222312982
Artelska A, Rola M, Rostkowski M, Pięta M, Pięta J, Michalski R, Sikora AB. Kinetic Study on the Reactivity of Azanone (HNO) toward Cyclic C-Nucleophiles. International Journal of Molecular Sciences. 2021; 22(23):12982. https://doi.org/10.3390/ijms222312982
Chicago/Turabian StyleArtelska, Angelika, Monika Rola, Michał Rostkowski, Marlena Pięta, Jakub Pięta, Radosław Michalski, and Adam Bartłomiej Sikora. 2021. "Kinetic Study on the Reactivity of Azanone (HNO) toward Cyclic C-Nucleophiles" International Journal of Molecular Sciences 22, no. 23: 12982. https://doi.org/10.3390/ijms222312982
APA StyleArtelska, A., Rola, M., Rostkowski, M., Pięta, M., Pięta, J., Michalski, R., & Sikora, A. B. (2021). Kinetic Study on the Reactivity of Azanone (HNO) toward Cyclic C-Nucleophiles. International Journal of Molecular Sciences, 22(23), 12982. https://doi.org/10.3390/ijms222312982






