Gene Expression Data Mining Reveals the Involvement of GPR55 and Its Endogenous Ligands in Immune Response, Cancer, and Differentiation
Abstract
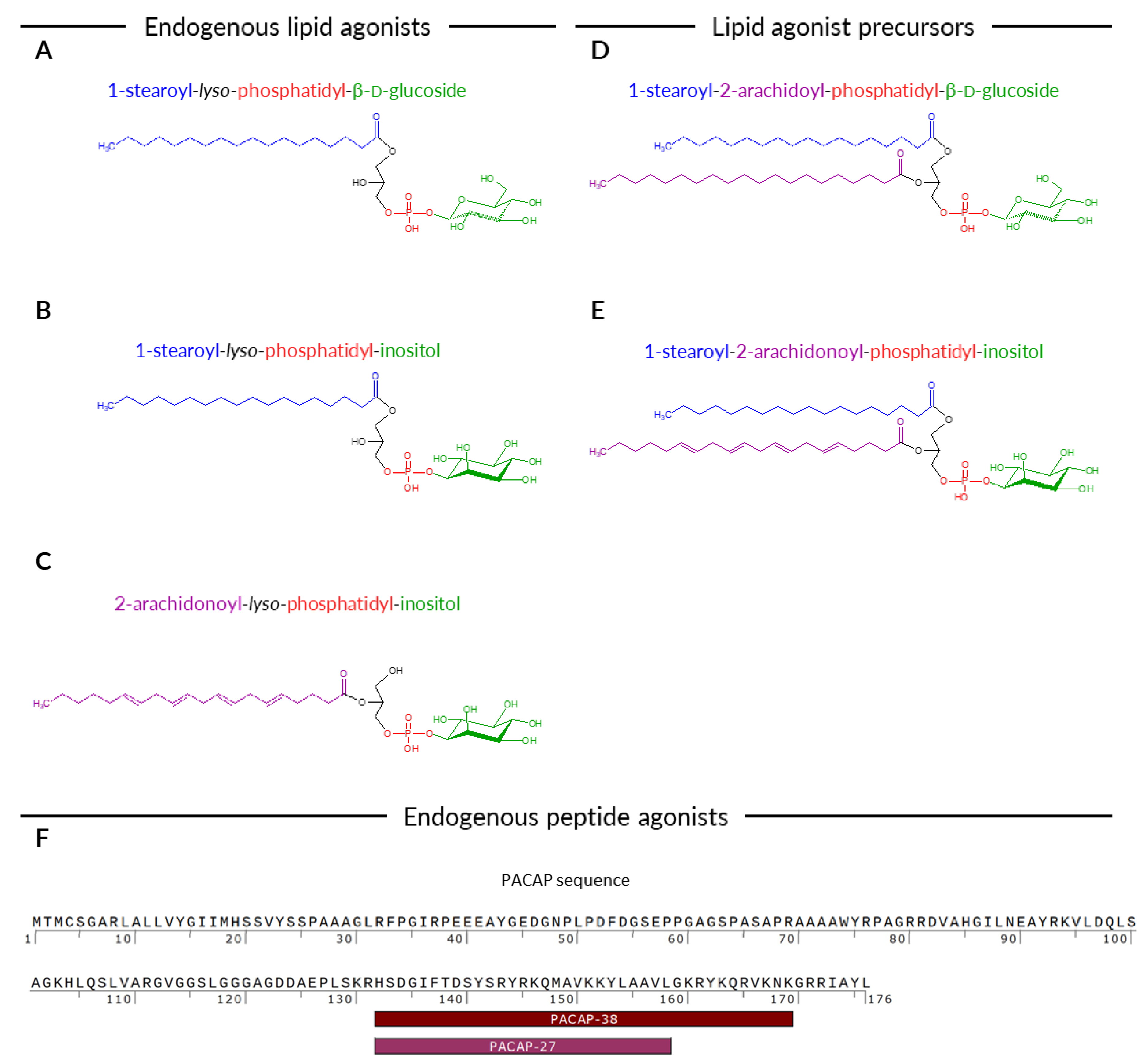
:1. Introduction
2. Results
2.1. GPR55 Upregulation Occurs Predominantly during Activation of Immune Cells
2.2. Conditions Promoting LPI Accumulation Exist in Tumors and LPI Suppression Is Preferred during Differentiation
2.3. PACAP27 and PACAP38 Expression
3. Discussion
4. Materials and Methods
4.1. Software
4.2. Perturbations
4.3. Assessment of LPI Production Capacity
- The LPI-score was increased by one point (+1) whenever a gene positively affecting LPI level was upregulated or a gene negatively affecting LPI level was downregulated;
- Conversely, the LPI-score was decreased by on point (−1) whenever a gene positively affecting LPI level was downregulated or a gene negatively affecting LPI level was upregulated.
4.4. Assessment of PACAP27 and PACAP38 Production Capacity
- The PC-score was increased by one point (+1) whenever a convertase-coding gene (PCSK1, PCSK2, or PCSK4) was upregulated;
- The PC-score was decreased by one point (−1) whenever a convertase-coding gene (PCSK1, PCSK2, or PCSK4) was downregulated.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brown, A.J.; Wise, A. Glaxosmithkline. Identification of Modulators of Gpr55 Activity. International Patent WO2001086305A2, 4 May 2001. [Google Scholar]
- Drmota, T.; Greasley, P.; Groblewski, T. Screening assays for cannabinoid-ligand-type modulators of gpr55. European Patent EP1595146A1, 23 April 2008. [Google Scholar]
- Lauckner, J.E.; Jensen, J.B.; Chen, H.Y.; Lu, H.C.; Hille, B.; Mackie, K. Gpr55 is a cannabinoid receptor that increases intracellular calcium and inhibits m current. Proc. Natl. Acad. Sci. USA 2008, 105, 2699–2704. [Google Scholar] [CrossRef] [Green Version]
- Ryberg, E.; Larsson, N.; Sjogren, S.; Hjorth, S.; Hermansson, N.O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor gpr55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef]
- Whyte, L.S.; Ryberg, E.; Sims, N.A.; Ridge, S.A.; Mackie, K.; Greasley, P.J.; Ross, R.A.; Rogers, M.J. The putative cannabinoid receptor gpr55 affects osteoclast function in vitro and bone mass in vivo. Proc. Natl. Acad. Sci. USA 2009, 106, 16511–16516. [Google Scholar] [CrossRef] [Green Version]
- Heynen-Genel, S.; Dahl, R.; Shi, S.; Milan, L.; Hariharan, S.; Sergienko, E.; Hedrick, M.; Dad, S.; Stonich, D.; Su, Y.; et al. Screening for selective ligands for gpr55—Antagonists. In Probe Reports from the Nih Molecular Libraries Program; NCBI: Bethesda, MD, USA, 2010. [Google Scholar]
- Heynen-Genel, S.; Dahl, R.; Shi, S.; Milan, L.; Hariharan, S.; Bravo, Y.; Sergienko, E.; Hedrick, M.; Dad, S.; Stonich, D.; et al. Screening for selective ligands for gpr55—Agonists. In Probe Reports from the Nih Molecular Libraries Program; NCBI: Bethesda, MD, USA, 2010. [Google Scholar]
- Oka, S.; Toshida, T.; Maruyama, K.; Nakajima, K.; Yamashita, A.; Sugiura, T. 2-arachidonoyl-sn-glycero-3-phosphoinositol: A possible natural ligand for gpr55. J. Biochem. 2009, 145, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Guy, A.T.; Nagatsuka, Y.; Ooashi, N.; Inoue, M.; Nakata, A.; Greimel, P.; Inoue, A.; Nabetani, T.; Murayama, A.; Ohta, K.; et al. Neuronal development. Glycerophospholipid regulation of modality-specific sensory axon guidance in the spinal cord. Science 2015, 349, 974–977. [Google Scholar] [CrossRef] [Green Version]
- Foster, S.R.; Hauser, A.S.; Vedel, L.; Strachan, R.T.; Huang, X.P.; Gavin, A.C.; Shah, S.D.; Nayak, A.P.; Haugaard-Kedstrom, L.M.; Penn, R.B.; et al. Discovery of human signaling systems: Pairing peptides to g protein-coupled receptors. Cell 2019, 179, 895–908.e821. [Google Scholar] [CrossRef] [Green Version]
- Oka, S.; Nakajima, K.; Yamashita, A.; Kishimoto, S.; Sugiura, T. Identification of gpr55 as a lysophosphatidylinositol receptor. Biochem. Biophys. Res. Commun. 2007, 362, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Guy, A.T.; Kano, K.; Ohyama, J.; Kamiguchi, H.; Hirabayashi, Y.; Ito, Y.; Matsuo, I.; Greimel, P. Preference for glucose over inositol headgroup during lysolipid activation of g protein-coupled receptor 55. ACS Chem. Neurosci. 2019, 10, 716–727. [Google Scholar] [CrossRef]
- Hirabayashi, Y. A world of sphingolipids and glycolipids in the brain--novel functions of simple lipids modified with glucose. Proc. Jpn. Academy. Ser. B Phys. Biol. Sci. 2012, 88, 129–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alhouayek, M.; Masquelier, J.; Muccioli, G.G. Lysophosphatidylinositols, from cell membrane constituents to gpr55 ligands. Trends Pharmacol. Sci. 2018, 39, 586–604. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, A.; Oka, S.; Tanikawa, T.; Hayashi, Y.; Nemoto-Sasaki, Y.; Sugiura, T. The actions and metabolism of lysophosphatidylinositol, an endogenous agonist for gpr55. Prostaglandins Other Lipid Mediat. 2013, 107, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, A.; Kumazawa, T.; Koga, H.; Suzuki, N.; Oka, S.; Sugiura, T. Generation of lysophosphatidylinositol by ddhd domain containing 1 (ddhd1): Possible involvement of phospholipase d/phosphatidic acid in the activation of ddhd1. Biochim. Biophys. Acta 2010, 1801, 711–720. [Google Scholar] [CrossRef]
- Mariggio, S.; Sebastia, J.; Filippi, B.M.; Iurisci, C.; Volonte, C.; Amadio, S.; De Falco, V.; Santoro, M.; Corda, D. A novel pathway of cell growth regulation mediated by a pla2alpha-derived phosphoinositide metabolite. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2006, 20, 2567–2569. [Google Scholar]
- Pineiro, R.; Maffucci, T.; Falasca, M. The putative cannabinoid receptor gpr55 defines a novel autocrine loop in cancer cell proliferation. Oncogene 2011, 30, 142–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruban, E.L.; Ferro, R.; Arifin, S.A.; Falasca, M. Lysophosphatidylinositol: A novel link between abc transporters and g-protein-coupled receptors. Biochem. Soc. Trans. 2014, 42, 1372–1377. [Google Scholar] [CrossRef]
- Thomas, G.; Betters, J.L.; Lord, C.C.; Brown, A.L.; Marshall, S.; Ferguson, D.; Sawyer, J.; Davis, M.A.; Melchior, J.T.; Blume, L.C.; et al. The serine hydrolase abhd6 is a critical regulator of the metabolic syndrome. Cell Rep. 2013, 5, 508–520. [Google Scholar] [CrossRef] [Green Version]
- Tokumura, A.; Majima, E.; Kariya, Y.; Tominaga, K.; Kogure, K.; Yasuda, K.; Fukuzawa, K. Identification of human plasma lysophospholipase d, a lysophosphatidic acid-producing enzyme, as autotaxin, a multifunctional phosphodiesterase. J. Biol. Chem. 2002, 277, 39436–39442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Umezu-Goto, M.; Kishi, Y.; Taira, A.; Hama, K.; Dohmae, N.; Takio, K.; Yamori, T.; Mills, G.B.; Inoue, K.; Aoki, J.; et al. Autotaxin has lysophospholipase d activity leading to tumor cell growth and motility by lysophosphatidic acid production. J. Cell Biol. 2002, 158, 227–233. [Google Scholar] [CrossRef]
- Briand-Mesange, F.; Pons, V.; Allart, S.; Masquelier, J.; Chicanne, G.; Beton, N.; Payrastre, B.; Muccioli, G.G.; Ausseil, J.; Davignon, J.L.; et al. Glycerophosphodiesterase 3 (gde3) is a lysophosphatidylinositol-specific ecto-phospholipase c acting as an endocannabinoid signaling switch. J. Biol. Chem. 2020, 295, 15767–15781. [Google Scholar] [CrossRef] [PubMed]
- Christophe, J. Type i receptors for pacap (a neuropeptide even more important than vip?). Biochim. Biophys. Acta 1993, 1154, 183–199. [Google Scholar]
- Smith, J.S.; Lefkowitz, R.J.; Rajagopal, S. Biased signalling: From simple switches to allosteric microprocessors. Nat. Rev. Drug Discov. 2018, 17, 243–260. [Google Scholar] [CrossRef]
- Ogi, K.; Miyamoto, Y.; Masuda, Y.; Habata, Y.; Hosoya, M.; Ohtaki, T.; Masuo, Y.; Onda, H.; Fujino, M. Molecular cloning and functional expression of a cdna encoding a human pituitary adenylate cyclase activating polypeptide receptor. Biochem. Biophys. Res. Commun. 1993, 196, 1511–1521. [Google Scholar] [CrossRef]
- Vaudry, D.; Falluel-Morel, A.; Bourgault, S.; Basille, M.; Burel, D.; Wurtz, O.; Fournier, A.; Chow, B.K.; Hashimoto, H.; Galas, L.; et al. Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol. Rev. 2009, 61, 283–357. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Shuto, Y.; Somogyvari-Vigh, A.; Arimura, A. Prohormone convertases 1 and 2 process propacap and generate matured, bioactive pacap38 and pacap27 in transfected rat pituitary gh4c1 cells. Neuroendocrinology 1999, 69, 217–226. [Google Scholar] [CrossRef]
- Li, M.; Mbikay, M.; Arimura, A. Pituitary adenylate cyclase-activating polypeptide precursor is processed solely by prohormone convertase 4 in the gonads. Endocrinology 2000, 141, 3723–3730. [Google Scholar] [CrossRef] [PubMed]
- Green, B.D.; Irwin, N.; Flatt, P.R. Pituitary adenylate cyclase-activating peptide (pacap): Assessment of dipeptidyl peptidase iv degradation, insulin-releasing activity and antidiabetic potential. Peptides 2006, 27, 1349–1358. [Google Scholar] [CrossRef]
- Durinx, C.; Lambeir, A.M.; Bosmans, E.; Falmagne, J.B.; Berghmans, R.; Haemers, A.; Scharpe, S.; De Meester, I. Molecular characterization of dipeptidyl peptidase activity in serum: Soluble cd26/dipeptidyl peptidase iv is responsible for the release of x-pro dipeptides. Eur. J. Biochem. 2000, 267, 5608–5613. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Tamvakopoulos, C.; Xie, D.; Dragovic, J.; Shen, X.; Fenyk-Melody, J.E.; Schmidt, K.; Bagchi, A.; Griffin, P.R.; Thornberry, N.A.; et al. The role of dipeptidyl peptidase iv in the cleavage of glucagon family peptides: In vivo metabolism of pituitary adenylate cyclase activating polypeptide-(1–38). J. Biol. Chem. 2003, 278, 22418–22423. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Maderdrut, J.L.; Lertora, J.J.; Batuman, V. Intravenous infusion of pituitary adenylate cyclase-activating polypeptide (pacap) in a patient with multiple myeloma and myeloma kidney: A case study. Peptides 2007, 28, 1891–1895. [Google Scholar] [CrossRef]
- Ferro, R.; Adamska, A.; Lattanzio, R.; Mavrommati, I.; Edling, C.E.; Arifin, S.A.; Fyffe, C.A.; Sala, G.; Sacchetto, L.; Chiorino, G.; et al. Gpr55 signalling promotes proliferation of pancreatic cancer cells and tumour growth in mice, and its inhibition increases effects of gemcitabine. Oncogene 2018, 37, 6368–6382. [Google Scholar] [CrossRef]
- McCloskey, A.G.; Miskelly, M.G.; Moore, C.B.T.; Nesbit, M.A.; Christie, K.A.; Owolabi, A.I.; Flatt, P.R.; McKillop, A.M. Crispr/cas9 gene editing demonstrates metabolic importance of gpr55 in the modulation of gip release and pancreatic beta cell function. Peptides 2020, 125, 170251. [Google Scholar] [CrossRef] [PubMed]
- McKillop, A.M.; Moran, B.M.; Abdel-Wahab, Y.H.; Flatt, P.R. Evaluation of the insulin releasing and antihyperglycaemic activities of gpr55 lipid agonists using clonal beta-cells, isolated pancreatic islets and mice. Br. J. Pharmacol. 2013, 170, 978–990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saliba, S.W.; Jauch, H.; Gargouri, B.; Keil, A.; Hurrle, T.; Volz, N.; Mohr, F.; van der Stelt, M.; Brase, S.; Fiebich, B.L. Anti-neuroinflammatory effects of gpr55 antagonists in lps-activated primary microglial cells. J. Neuroinflammation 2018, 15, 322. [Google Scholar] [CrossRef] [PubMed]
- Sousa, I.G.; Simi, K.C.R.; do Almo, M.M.; Bezerra, M.A.G.; Doose, G.; Raiol, T.; Stadler, P.F.; Hoffmann, S.; Maranhao, A.Q.; Brigido, M.M. Gene expression profile of human t cells following a single stimulation of peripheral blood mononuclear cells with anti-cd3 antibodies. BMC Genom. 2019, 20, 593. [Google Scholar] [CrossRef] [Green Version]
- Chemin, K.; Ramskold, D.; Diaz-Gallo, L.M.; Herrath, J.; Houtman, M.; Tandre, K.; Ronnblom, L.; Catrina, A.; Malmstrom, V. Eomes-positive cd4(+) t cells are increased in ptpn22 (1858t) risk allele carriers. Eur. J. Immunol. 2018, 48, 655–669. [Google Scholar] [CrossRef] [Green Version]
- Hirahara, K.; Onodera, A.; Villarino, A.V.; Bonelli, M.; Sciume, G.; Laurence, A.; Sun, H.W.; Brooks, S.R.; Vahedi, G.; Shih, H.Y.; et al. Asymmetric action of stat transcription factors drives transcriptional outputs and cytokine specificity. Immunity 2015, 42, 877–889. [Google Scholar] [CrossRef] [Green Version]
- Kanduri, K.; Tripathi, S.; Larjo, A.; Mannerstrom, H.; Ullah, U.; Lund, R.; Hawkins, R.D.; Ren, B.; Lahdesmaki, H.; Lahesmaa, R. Identification of global regulators of t-helper cell lineage specification. Genome Med. 2015, 7, 122. [Google Scholar] [CrossRef] [Green Version]
- Couch, D.G.; Tasker, C.; Theophilidou, E.; Lund, J.N.; O’Sullivan, S.E. Cannabidiol and palmitoylethanolamide are anti-inflammatory in the acutely inflamed human colon. Clin. Sci. 2017, 131, 2611–2626. [Google Scholar] [CrossRef]
- Wlodarczyk, M.; Sobolewska-Wlodarczyk, A.; Cygankiewicz, A.I.; Jacenik, D.; Krajewska, W.M.; Stec-Michalska, K.; Piechota-Polanczyk, A.; Wisniewska-Jarosinska, M.; Fichna, J. G protein-coupled receptor 55 (gpr55) expresses differently in patients with crohn’s disease and ulcerative colitis. Scand. J. Gastroenterol. 2017, 52, 711–715. [Google Scholar] [CrossRef]
- Li, X.; Hanafusa, K.; Kage, M.; Yokoyama, N.; Nakayama, H.; Hotta, T.; Oshima, E.; Kano, K.; Matsuo, I.; Nagatsuka, Y.; et al. Lysophosphatidylglucoside is a gpr55 -mediated chemotactic molecule for human monocytes and macrophages. Biochem. Biophys. Res. Commun. 2021, 569, 86–92. [Google Scholar] [CrossRef]
- Luther, S.A.; Cyster, J.G. Chemokines as regulators of t cell differentiation. Nat. Immunol. 2001, 2, 102–107. [Google Scholar] [CrossRef]
- Chiurchiu, V.; Lanuti, M.; De Bardi, M.; Battistini, L.; Maccarrone, M. The differential characterization of gpr55 receptor in human peripheral blood reveals a distinctive expression in monocytes and nk cells and a proinflammatory role in these innate cells. Int. Immunol. 2015, 27, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Price, A.M.; Tourigny, J.P.; Forte, E.; Salinas, R.E.; Dave, S.S.; Luftig, M.A. Analysis of epstein-barr virus-regulated host gene expression changes through primary b-cell outgrowth reveals delayed kinetics of latent membrane protein 1-mediated nf-kappab activation. J. Virol. 2012, 86, 11096–11106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Birkenbach, M.; Josefsen, K.; Yalamanchili, R.; Lenoir, G.; Kieff, E. Epstein-barr virus-induced genes: First lymphocyte-specific g protein-coupled peptide receptors. J. Virol. 1993, 67, 2209–2220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arfelt, K.N.; Fares, S.; Rosenkilde, M.M. Ebv, the human host, and the 7tm receptors: Defense or offense? Prog. Mol. Biol. Transl. Sci. 2015, 129, 395–427. [Google Scholar] [PubMed]
- Mihret, A. The role of dendritic cells in mycobacterium tuberculosis infection. Virulence 2012, 3, 654–659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, M.P.; Graham, C.M.; McNab, F.W.; Xu, Z.; Bloch, S.A.; Oni, T.; Wilkinson, K.A.; Banchereau, R.; Skinner, J.; Wilkinson, R.J.; et al. An interferon-inducible neutrophil-driven blood transcriptional signature in human tuberculosis. Nature 2010, 466, 973–977. [Google Scholar] [CrossRef] [Green Version]
- Scriba, T.J.; Penn-Nicholson, A.; Shankar, S.; Hraha, T.; Thompson, E.G.; Sterling, D.; Nemes, E.; Darboe, F.; Suliman, S.; Amon, L.M.; et al. Sequential inflammatory processes define human progression from m. Tuberculosis infection to tuberculosis disease. PLoS Pathog. 2017, 13, e1006687. [Google Scholar] [CrossRef] [Green Version]
- Ko, I.K.; Kim, B.G.; Awadallah, A.; Mikulan, J.; Lin, P.; Letterio, J.J.; Dennis, J.E. Targeting improves msc treatment of inflammatory bowel disease. Mol. Ther. J. Am. Soc. Gene Ther. 2010, 18, 1365–1372. [Google Scholar] [CrossRef]
- Schicho, R.; Bashashati, M.; Bawa, M.; McHugh, D.; Saur, D.; Hu, H.M.; Zimmer, A.; Lutz, B.; Mackie, K.; Bradshaw, H.B.; et al. The atypical cannabinoid o-1602 protects against experimental colitis and inhibits neutrophil recruitment. Inflamm. Bowel Dis. 2011, 17, 1651–1664. [Google Scholar] [CrossRef] [Green Version]
- Nemetz, N.; Abad, C.; Lawson, G.; Nobuta, H.; Chhith, S.; Duong, L.; Tse, G.; Braun, J.; Waschek, J.A. Induction of colitis and rapid development of colorectal tumors in mice deficient in the neuropeptide pacap. Int. J. Cancer 2008, 122, 1803–1809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horvath, G.; Illes, A.; Heimesaat, M.M.; Bardosi, A.; Bardosi, S.; Tamas, A.; Fulop, B.D.; Opper, B.; Nemeth, J.; Ferencz, A.; et al. Protective intestinal effects of pituitary adenylate cyclase activating polypeptide. In Pituitary Adenylate Cyclase Activating Polypeptide—Pacap; Reglodi, D., Tamas, A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 271–288. [Google Scholar]
- Andradas, C.; Caffarel, M.M.; Perez-Gomez, E.; Salazar, M.; Lorente, M.; Velasco, G.; Guzman, M.; Sanchez, C. The orphan g protein-coupled receptor gpr55 promotes cancer cell proliferation via erk. Oncogene 2011, 30, 245–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, X.L.; Guo, X.; Song, Y.P.; Zhu, C.Y.; Zou, W. The lpi/gpr55 axis enhances human breast cancer cell migration via hbxip and p-mlc signaling. Acta Pharmacol. Sin. 2018, 39, 459–471. [Google Scholar] [CrossRef] [Green Version]
- Ford, L.A.; Roelofs, A.J.; Anavi-Goffer, S.; Mowat, L.; Simpson, D.G.; Irving, A.J.; Rogers, M.J.; Rajnicek, A.M.; Ross, R.A. A role for l-alpha-lysophosphatidylinositol and gpr55 in the modulation of migration, orientation and polarization of human breast cancer cells. Br. J. Pharmacol. 2010, 160, 762–771. [Google Scholar] [CrossRef] [Green Version]
- Lelievre, V.; Seksenyan, A.; Nobuta, H.; Yong, W.H.; Chhith, S.; Niewiadomski, P.; Cohen, J.R.; Dong, H.; Flores, A.; Liau, L.M.; et al. Disruption of the pacap gene promotes medulloblastoma in ptc1 mutant mice. Dev. Biol. 2008, 313, 359–370. [Google Scholar] [CrossRef] [Green Version]
- Moody, T.W.; Osefo, N.; Nuche-Berenguer, B.; Ridnour, L.; Wink, D.; Jensen, R.T. Pituitary adenylate cyclase-activating polypeptide causes tyrosine phosphorylation of the epidermal growth factor receptor in lung cancer cells. J. Pharmacol. Exp. Ther. 2012, 341, 873–881. [Google Scholar] [CrossRef] [Green Version]
- Moody, T.W.; Di Florio, A.; Jensen, R.T. Pyk-2 is tyrosine phosphorylated after activation of pituitary adenylate cyclase activating polypeptide receptors in lung cancer cells. J. Mol. Neurosci. 2012, 48, 660–666. [Google Scholar] [CrossRef] [Green Version]
- Moody, T.W.; Lee, L.; Jensen, R.T. The g protein-coupled receptor pac1 regulates transactivation of the receptor tyrosine kinase her3. J. Mol. Neurosci. 2020, 71, 1589–1597. [Google Scholar] [CrossRef]
- Ushiyama, M.; Ikeda, R.; Sugawara, H.; Yoshida, M.; Mori, K.; Kangawa, K.; Inoue, K.; Yamada, K.; Miyata, A. Differential intracellular signaling through pac1 isoforms as a result of alternative splicing in the first extracellular domain and the third intracellular loop. Mol. Pharmacol. 2007, 72, 103–111. [Google Scholar] [CrossRef] [Green Version]
- Hruz, T.; Laule, O.; Szabo, G.; Wessendorp, F.; Bleuler, S.; Oertle, L.; Widmayer, P.; Gruissem, W.; Zimmermann, P. Genevestigator v3: A reference expression database for the meta-analysis of transcriptomes. Adv. Bioinform. 2008, 2008, 420747. [Google Scholar] [CrossRef] [PubMed]




| Gene Symbol | Full Name (Gene; Protein) | Effect on LPI | Mode of Action | Ref. |
|---|---|---|---|---|
| DDHD1 | DDHD domain containing 1; phospholipase DDHD1 | positive | involved in the formation of 2-arachidonoyl-LPI (endogenous agonist) | [15,16] |
| PLA2G4A | phospholipase A2 group IVA; cytosolic phospholipase A2 | positive | involved in the formation of 2-stearotyl-LPI (endogenous agonist) | [15,18,19] |
| ABCC1 (MRP1) | ATP binding cassette subfamily C member 1; multidrug resistance-associated protein 1 | positive | pumps LPI out of the cell enabling autocrine and paracrine activation of GPR55 | [18,19] |
| ABHD6 | abhydrolase domain containing 6, acylglycerol lipase; monoacylglycerol lipase ABHD6 | negative | has lysophospholipase A activity; degrades LPI into GPI and a fatty acid, depleting agonist pool | [20] |
| ENPP2 (ATX) | ectonucleotide pyrophosphatase/phosphodiesterase 2; autotaxin | negative | has lysophospholipase D activity; degrades LPI into LPA | [21,22] |
| GDPD2 (GDE3) | glycerophosphodiester phosphodiesterase domain containing 2; glycerophosphoinositol inositolphosphodiesterase GDPD2, glycerophosphodiesterase 3 | negative | has lysophospholipase C activity; degrades LPI into 2-AG; generates CB2 agonist from LPI | [23] |
| Gene Symbol | Full Name (Gene; Protein) | Effect on PACAP27/38 | Mode of Action | Ref. |
|---|---|---|---|---|
| ADCYAP1 (PACAP) | adenylate cyclase activating polypeptide 1; pituitary adenylate cyclase-activating polypeptide | positive | undergoes cleavage that generates PACAP-27 peptide (endogenous agonist of GPR55) | [10] |
| PCSK1 (PC1) | proprotein convertase subtilisin/kexin type 1; neuroendocrine convertase 1; proprotein convertase 1 | positive | cleaves ADCYAP1-encoded polypeptide into shorter PACAP-27 | [27,28] |
| PCSK2 (PC2) | proprotein convertase subtilisin/kexin type 2; neuroendocrine convertase 2; proprotein convertase 2 | positive | cleaves ADCYAP1-encoded polypeptide into shorter PACAP-27 | [27,28] |
| PCSK4 (PC4) | proprotein convertase subtilisin/kexin type 4; proprotein convertase 4 | positive | cleaves ADCYAP1-encoded polypeptide into shorter PACAP-27 | [27,29] |
| DPP4 (CD26, ADCP2) | dipeptidyl peptidase 4; cluster of differentiation 26, adenosine deaminase complexing protein 2 | negative | exopeptidase with a dipeptidyl peptidase activity; degrades PACAP-27 | [27] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wnorowski, A.; Wójcik, J.; Maj, M. Gene Expression Data Mining Reveals the Involvement of GPR55 and Its Endogenous Ligands in Immune Response, Cancer, and Differentiation. Int. J. Mol. Sci. 2021, 22, 13328. https://doi.org/10.3390/ijms222413328
Wnorowski A, Wójcik J, Maj M. Gene Expression Data Mining Reveals the Involvement of GPR55 and Its Endogenous Ligands in Immune Response, Cancer, and Differentiation. International Journal of Molecular Sciences. 2021; 22(24):13328. https://doi.org/10.3390/ijms222413328
Chicago/Turabian StyleWnorowski, Artur, Jakub Wójcik, and Maciej Maj. 2021. "Gene Expression Data Mining Reveals the Involvement of GPR55 and Its Endogenous Ligands in Immune Response, Cancer, and Differentiation" International Journal of Molecular Sciences 22, no. 24: 13328. https://doi.org/10.3390/ijms222413328







