Augmented KCa2.3 Channel Feedback Regulation of Oxytocin Stimulated Uterine Strips from Nonpregnant Mice
Abstract
:1. Introduction
2. Results
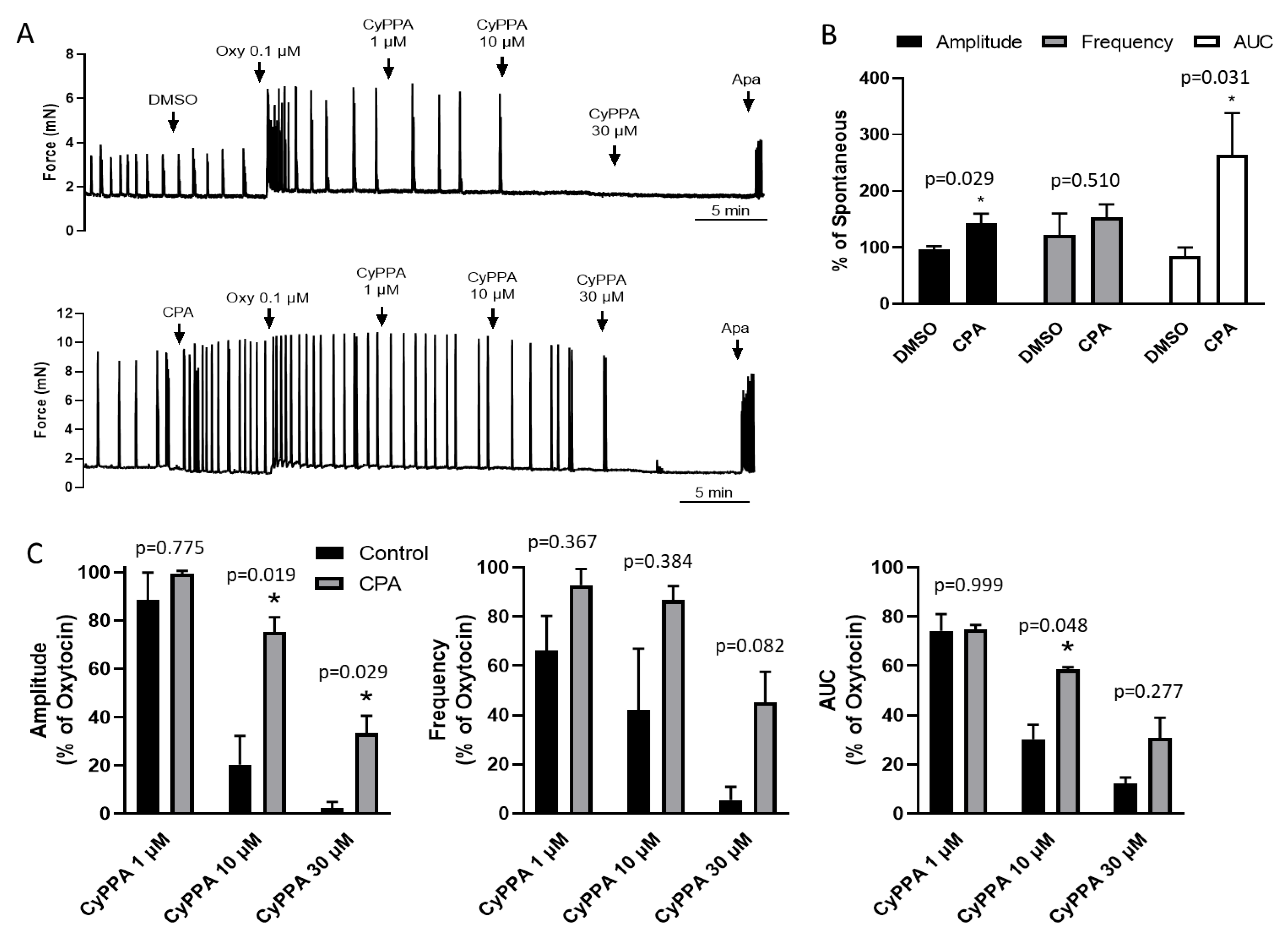
2.1. CyPPA Prevents Oxytocin Potentiation of Uterine Contractions
2.2. Inhibition of SERCA Impairs CyPPA Relaxation
2.3. Inhibition of NSCCs Does Not Alter CyPPA Relaxation
2.4. KCa2.3 Channels and IP3Rs Exhibit Overlapping Punctate Expression Patterns in the Myometrium
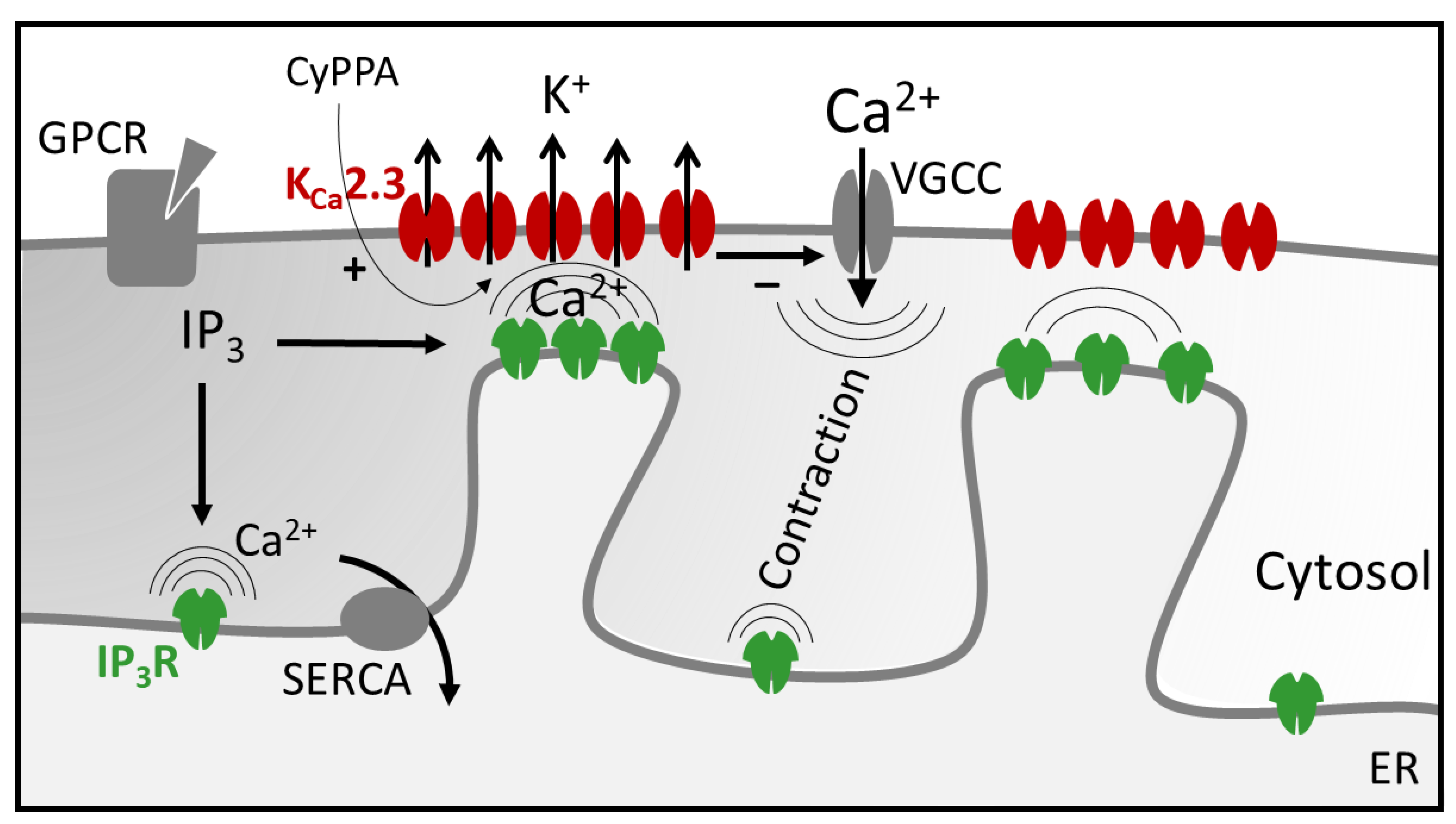
3. Discussion
4. Materials and Methods
4.1. Tissue Preparation
4.2. Isometric Force Myography
4.3. Immunofluorescence
4.4. Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frey, H.A.; Klebanoff, M.A. The Epidemiology, Etiology, and Costs of Preterm Birth. Semin. Fetal. Neonatal Med. 2016, 21, 68–73. [Google Scholar] [CrossRef]
- Romero, R.; Dey, S.K.; Fisher, S.J. Preterm Labor: One Syndrome, Many Causes. Science 2014, 345, 760–765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matthew, A.; Kupittayanant, S.; Burdyga, T.; Wray, S. Characterization of Contractile Activity and Intracellular Ca2+ Signalling in Mouse Myometrium. J. Soc. Gynecol. Investig. 2004, 11, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Ludmir, J. Drugs for the Treatment and Prevention of Preterm Labor. Clin. Perinatol. 2019, 46, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Blumenfeld, Y.J.; Lyell, D.J. Prematurity Prevention: The Role of Acute Tocolysis. Curr. Opin. Obstet. Gynecol. 2009, 21, 136–141. [Google Scholar] [CrossRef]
- Abramovici, A.; Cantu, J.; Jenkins, S.M. Tocolytic Therapy for Acute Preterm Labor. Obstet. Gynecol. Clin. N. Am. 2012, 39, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Dolphin, A.C. Voltage-Gated Calcium Channels and Their Auxiliary Subunits: Physiology and Pathophysiology and Pharmacology: Voltage-Gated Calcium Channels. J. Physiol. 2016, 594, 5369–5390. [Google Scholar] [CrossRef] [Green Version]
- Brown, A.; Cornwell, T.; Korniyenko, I.; Solodushko, V.; Bond, C.T.; Adelman, J.P.; Taylor, M.S. Myometrial Expression of Small Conductance Ca2+-Activated K+ Channels Depresses Phasic Uterine Contraction. Am. J. Physiol. Cell Physiol. 2007, 292, C832–C840. [Google Scholar] [CrossRef]
- Skarra, D.V.; Cornwell, T.; Solodushko, V.; Brown, A.; Taylor, M.S. CyPPA, a Positive Modulator of Small-Conductance Ca(2+)-Activated K(+) Channels, Inhibits Phasic Uterine Contractions and Delays Preterm Birth in Mice. Am. J. Physiol. Cell Physiol. 2011, 301, C1027–C1035. [Google Scholar] [CrossRef] [Green Version]
- Pierce, S.L.; Kresowik, J.D.K.; Lamping, K.G.; England, S.K. Overexpression of SK3 Channels Dampens Uterine Contractility to Prevent Preterm Labor in Mice. Biol. Reprod. 2008, 78, 1058–1063. [Google Scholar] [CrossRef] [Green Version]
- Rosenbaum, S.T.; Svalo, J.; Nielsen, K.; Larsen, T.; Jorgensen, J.C.; Bouchelouche, P. Immunolocalization and Expression of Small-Conductance Calcium-Activated Potassium Channels in Human Myometrium. J. Cell. Mol. Med. 2012, 16, 3001–3008. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.M.; Fakler, B.; Rivard, A.; Wayman, G.; Johnson-Pais, T.; Keen, J.E.; Ishii, T.; Hirschberg, B.; Bond, C.T.; Lutsenko, S.; et al. Mechanism of Calcium Gating in Small-Conductance Calcium-Activated Potassium Channels. Nature 1998, 395, 503–507. [Google Scholar] [CrossRef]
- Bond, C.T.; Sprengel, R.; Bissonnette, J.M.; Kaufmann, W.A.; Pribnow, D.; Neelands, T.; Storck, T.; Baetscher, M.; Jerecic, J.; Maylie, J.; et al. Respiration and Parturition Affected by Conditional Overexpression of the Ca2+-Activated K+ Channel Subunit, SK3. Science 2000, 289, 1942–1946. [Google Scholar] [CrossRef]
- Hougaard, C.; Fraser, M.O.; Chien, C.; Bookout, A.; Katofiasc, M.; Jensen, B.S.; Rode, F.; Bitsch-Norhave, J.; Teuber, L.; Thor, K.B.; et al. A Positive Modulator of K Ca 2 and K Ca 3 Channels, 4,5-Dichloro-1,3-Diethyl-1,3-Dihydro-Benzoimidazol-2-One (NS4591), Inhibits Bladder Afferent Firing in Vitro and Bladder Overactivity in Vivo. J. Pharmacol. Exp. Ther. 2009, 328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenbaum, S.T.; Svalo, J.; Larsen, T.; Joergensen, J.; Bouchelouche, P. Relaxant Effect of Cyppa In Vitro and Expression Analysis of SK3 Splice Variants in the Myometrium of Pregnant and Non-Pregnant Women. Gynecol. Obstet. 2013, 3, 171. [Google Scholar] [CrossRef] [Green Version]
- Vrachnis, N.; Malamas, F.M.; Sifakis, S.; Deligeoroglou, E.; Iliodromiti, Z. The Oxytocin-Oxytocin Receptor System and Its Antagonists as Tocolytic Agents. Int. J. Endocrinol. 2011, 2011, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Jaggar, J.H.; Porter, V.A.; Lederer, W.J.; Nelson, M.T. Calcium Sparks in Smooth Muscle. Am. J. Physiol. 2000, 278, C235–C256. [Google Scholar] [CrossRef]
- ZhuGe, R.; Bao, R.; Fogarty, K.E.; Lifshitz, L.M. Ca2+ Sparks Act as Potent Regulators of Excitation-Contraction Coupling in Airway Smooth Muscle. J. Biol. Chem. 2010, 285, 2203–2210. [Google Scholar] [CrossRef] [Green Version]
- Ledoux, J.; Taylor, M.S.; Bonev, A.D.; Hannah, R.M.; Solodushko, V.; Shui, B.; Tallini, Y.; Kotlikoff, M.I.; Nelson, M.T. Functional Architecture of Inositol 1,4,5-Trisphosphate Signaling in Restricted Spaces of Myoendothelial Projections. Proc. Natl. Acad. Sci. USA 2008, 105, 9627–9632. [Google Scholar] [CrossRef] [Green Version]
- Wellman, G.C.; Nathan, D.J.; Saundry, C.M.; Perez, G.; Bonev, A.D.; Penar, P.L.; Tranmer, B.I.; Nelson, M.T. Ca2+ Sparks and Their Function in Human Cerebral Arteries. Stroke 2002, 33, 802–808. [Google Scholar] [CrossRef] [Green Version]
- Beech, D.J.; Muraki, K.; Flemming, R. Non-Selective Cationic Channels of Smooth Muscle and the Mammalian Homologues of Drosophila TRP: Smooth Muscle TRPs and Cationic Channels. J. Physiol. 2004, 559, 685–706. [Google Scholar] [CrossRef]
- Wray, S.; Burdyga, T.; Noble, K. Calcium Signalling in Smooth Muscle. Cell Calcium 2005, 38. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, A. Molecular Identification and Localization of Trp Homologues, Putative Calcium Channels, in Pregnant Human Uterus. Mol. Hum. Reprod. 2002, 8, 946–951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ying, L.; Becard, M.; Lyell, D.; Han, X.; Shortliffe, L.; Husted, C.I.; Alvira, C.M.; Cornfield, D.N. The Transient Receptor Potential Vanilloid 4 Channel Modulates Uterine Tone during Pregnancy. Sci. Transl. Med. 2015, 7, 319ra204. [Google Scholar] [CrossRef] [Green Version]
- Qian, X.; Francis, M.; Kohler, R.; Solodushko, V.; Lin, M.; Taylor, M.S. Positive Feedback Regulation of Agonist-Stimulated Endothelial Ca2+ Dynamics by KCa3.1 Channels in Mouse Mesenteric Arteries. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 127–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.T.; Jian, M.-Y.; Taylor, M.S.; Cioffi, D.L.; Yap, F.C.; Liedtke, W.; Townsley, M.I. Functional Coupling of TRPV4, IK, and SK Channels Contributes to Ca 2+ -Dependent Endothelial Injury in Rodent Lung. Pulm. Circ. 2015, 5, 279–290. [Google Scholar] [CrossRef] [Green Version]
- Feetham, C.H.; Nunn, N.; Lewis, R.; Dart, C.; Barrett-Jolley, R. TRPV4 and K Ca Ion Channels Functionally Couple as Osmosensors in the Paraventricular Nucleus: TRPV4 and KCa as Osmosensors in the PVN. Br. J. Pharmacol. 2015, 172, 1753–1768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villegas, D.; Giard, O.; Brochu-Gaudreau, K.; Rousseau, É. Activation of TRPV4 Channels Leads to a Consistent Tocolytic Effect on Human Myometrial Tissues. Eur. J. Obstet. Gynecol. Reprod. Biol. X 2021, 10, 100124. [Google Scholar] [CrossRef] [PubMed]
- Hill-Eubanks, D.C.; Werner, M.E.; Heppner, T.J.; Nelson, M.T. Calcium Signaling in Smooth Muscle. Cold Spring Harb. Perspect. Biol. 2011, 3, a004549. [Google Scholar] [CrossRef]
- Michelangeli, F.; Mezna, M.; Tovey, S.; Sayers, L.G. Pharmacological Modulators of the Inositol 1,4,Strisphosphate Receptor. Neuropharmacology 1995, 34, 1111–1122. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zak, M.; Kestler, B.; Cornwell, T.; Taylor, M.S. Augmented KCa2.3 Channel Feedback Regulation of Oxytocin Stimulated Uterine Strips from Nonpregnant Mice. Int. J. Mol. Sci. 2021, 22, 13585. https://doi.org/10.3390/ijms222413585
Zak M, Kestler B, Cornwell T, Taylor MS. Augmented KCa2.3 Channel Feedback Regulation of Oxytocin Stimulated Uterine Strips from Nonpregnant Mice. International Journal of Molecular Sciences. 2021; 22(24):13585. https://doi.org/10.3390/ijms222413585
Chicago/Turabian StyleZak, Megan, Bri Kestler, Trudy Cornwell, and Mark S. Taylor. 2021. "Augmented KCa2.3 Channel Feedback Regulation of Oxytocin Stimulated Uterine Strips from Nonpregnant Mice" International Journal of Molecular Sciences 22, no. 24: 13585. https://doi.org/10.3390/ijms222413585
APA StyleZak, M., Kestler, B., Cornwell, T., & Taylor, M. S. (2021). Augmented KCa2.3 Channel Feedback Regulation of Oxytocin Stimulated Uterine Strips from Nonpregnant Mice. International Journal of Molecular Sciences, 22(24), 13585. https://doi.org/10.3390/ijms222413585




