Nanotechnology Therapy for Alzheimer′s Disease Memory Impairment Attenuation
Abstract
:1. Nanotechnology for Alzheimer’s Disease Therapy
2. Alzheimer′s Disease and Memory Impairment
2.1. Short-Term and Long-Term Memory
2.2. Memory Impairment
2.3. Alzheimer′s Disease, Dementia and Memory Impairment
3. Potential Animal Models for Alzheimer′s Disease Memory Impairment
3.1. Transgenic Rodents
3.1.1. Transgenic Mice Expressing Human APP (Amyloid Precursor Protein) and PSEN1 (Presenilin 1) with efAD Mutations
3.1.2. Transgenic Mice Expressing Tau
3.1.3. Transgenic Mice with Both Plaques and Tangles
3.1.4. Knock-in Mouse Models
3.1.5. Transgenic Rat Models
3.2. Rat Brain Injury Models of Alzheimer’s Disease
3.3. Animal Models of Nanoparticle-Based Therapy That Attenuate the Effect of Alzheimer’s Disease on Memory
4. Mouse and Rat Models Where NPs Attenuated Alzheimer’s Disease Memory Impairment
4.1. Animal Models of Alzheimer’s Disease Where NPs Attenuated Memory Loss
4.2. Molecular and Cellular Effects of NPs-Based Treatments in Mouse and Rat Models Where Alzheimer′s Disease Memory Impairment Was Attenuated
4.3. Memory Assessment Tests of NPs-Based Treatments in Mouse and Rat Models Where Alzheimer′s Disease Memory Impairment Was Attenuated
5. Discussion
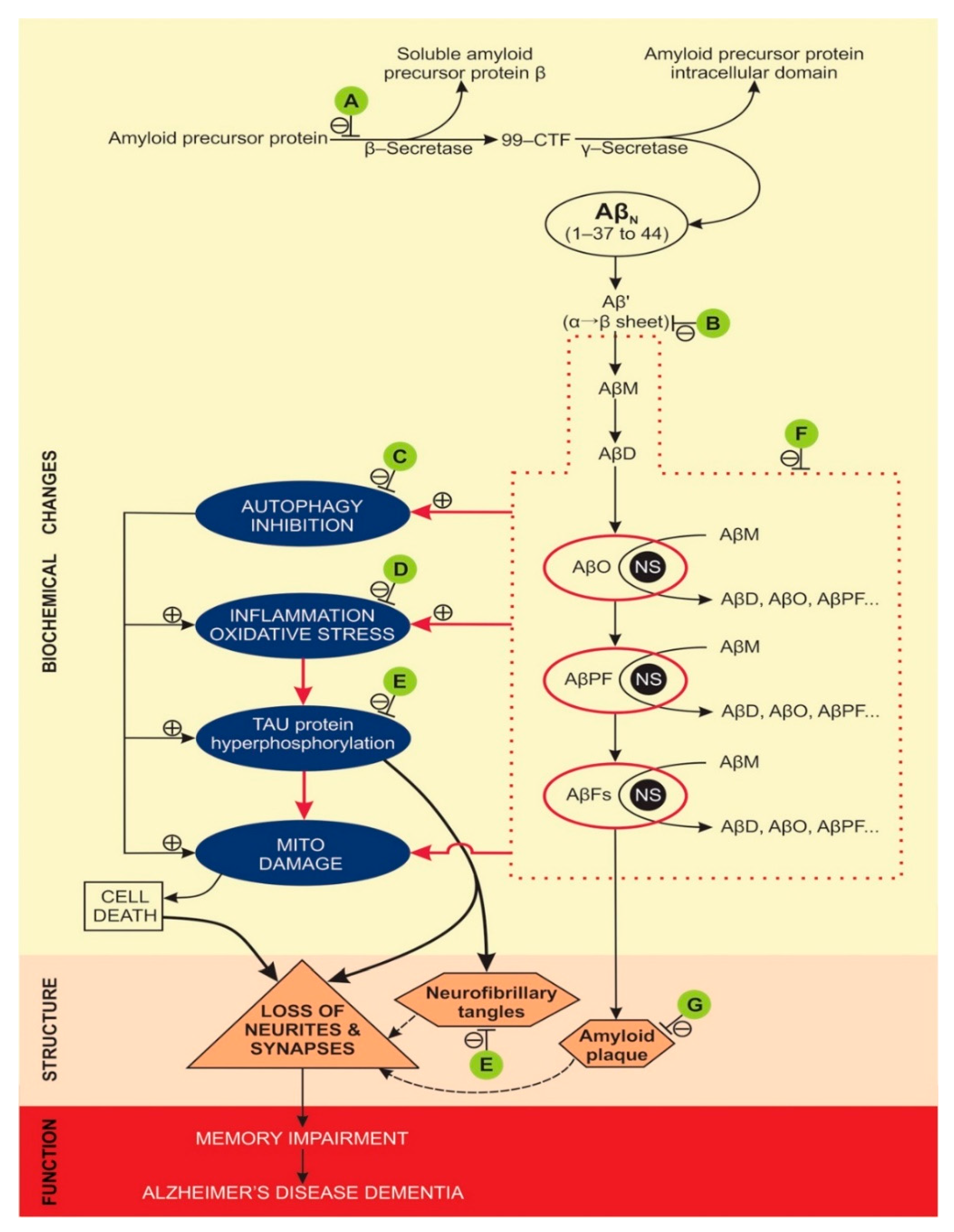
5.1. Development of Alzheimer’s Disease over Time
5.2. Molecular Mechanisms of Alzheimer’s Disease
5.3. Animal Models of NPs Based Therapy for Alzheimer’s Disease Associated Memory Dysfunction
5.4. Current Challenges for Nanotechnology Therapy in Animal Models of Alzheimer’s Disease
5.4.1. The Alzheimer’s Disease Aetiology Challenge
5.4.2. Dosing and Target Selectivity
5.4.3. Shortcomings of Alzheimer’s Disease Animal Models
5.4.4. Time Scale of NPs Based Therapies in Animal Models of Alzheimer’s Disease
5.4.5. Pharmacodynamic and Pharmacodynamic Drug-Drug Interactions
5.5. Specifications for the Ideal NPs Preparations to Treat the Alzheimer’s Disease Memory Dysfunction in Human
- (a)
- What are the key molecular changes that first initiate, and later sustain the progression of AD-related brain pathology in human?
- (b)
- How can NPs regulate tau protein and Aβ1-40/-42 peptides levels without interfering with their normal functions in brain (e.g., when adjusting synaptic plasticity in response to changes in nerve activity or BBB permeability? and
- (c)
- What will be the clinical effect of NPs treatment on memory decline in human? Will they have a cognitive reserve “enhancing effect” with a delayed start of dementia, followed by an accelerated dementia progression, or will NPs treatments also slow the rate of dementia progression?
- (a)
- multifunctional NPs stimulate autophagy and simultaneously attenuate several molecular pathways involved in AD pathogenesis, e.g., tau hyperphosphorylation, AβOs formation and neuroinflammation;
- (b)
- NPs preparations are biodegradable, metabolised by the human body, or exert their effect without having to cross the BBB (e.g., via the sink-effect);
- (c)
- long-term use of NPs does not lead to interactions with the bodies enzymes that lead to toxic modifications of NPs in the human body;
- (d)
- NPs function as physiological buffers, preventing pathological changes in tau proteins and Aβ peptides without interfering with their physiological functions in the human body; and
- (e)
- NPs do not interact with other NPs-treatment preparations, nor with conventional drugs used for treatment of acute or chronic diseases.
5.6. Suggestions for Further Work on the Animal Models of NPs Based Therapies for Attenuation of the Alzheimer’s Disease Associated Memory Dysfunction
- (a)
- development of animal models for the late-onset, sporadic form of AD;
- (b)
- development of animal models that evaluate dietary and lifestyle contributions to AD pathogenesis;
- (c)
- use of a standardised memory tests battery (e.g., Morris water maze, Novel object recognition and Passive avoidance test) to better compare the effects of different NPs on memory dysfunction treatment;
- (d)
- comparison of therapeutic efficacies for different routes of NPs′ based therapies administration (e.g., nose-to-brain versus BBB, versus sink mechanism or cleansing extra corporal systems);
- (e)
- comparison of pharmacokinetic and pharmacodynamic profiles of different NPs based preparations.
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ⊕ | promotes/accelerates |
| ↓/⊥ | attenuates/inhibits |
| γ-S | γ-secretase |
| β-S (BACE1) | β-secretase |
| 5XFAD | 5XFAD mice express human APP and PSEN1 transgenes with a total of five AD-linked mutations: the Swedish (K670N/M671L), Florida (I716V), and London (V717I) mutations in APP, and the M146L and L286V mutations in PSEN1 |
| 99-CTF | 99-amino acid membrane bound C-terminal fragment |
| Aβ-PL | amyloid beta plaque |
| Aβ’ | misfolded Aβ peptide with first α-sheet, then β-sheet structure |
| Aβ1-40/-42 | amyloid β peptides with 40 or 42 amino acid residues |
| AβD | amyloid β dimer |
| AβFs | amyloid β fibrils |
| AβM | amyloid β monomer |
| Aβn | native Aβ peptide with α-helix structure |
| AβO (s) | toxic, soluble amyloid β oligomer (s) |
| Aβp | Aβ peptides with 40 or 42 amino-acid residues |
| AβPF | amyloid β protofibril |
| AD | Alzheimer’s disease |
| AlCl3 | aluminium chloride |
| Apo- | apolipoprotein |
| APP | encodes amyloid precursor protein |
| APP23 | overexpresses mutant human APP with the Swedish mutation |
| B6; SJL-Tg(APPSWE)2576Kha | Taconic Europe transgenic mice (express a transgene coding for 695-amino acid isoform of human Alzheimer amyloid-β (Aβ) precursor protein carrying the Swedish mutation |
| BBB | blood-brain barrier |
| Co | Conditioned avoidance response test |
| Cas9 | CRISPR associated protein 9 |
| CR | cognitive reserve |
| CRISPR–Cas9 | RNA-guided Cas9 nuclease from the microbial clustered regularly interspaced short palindromic repeats adaptive immune system |
| CSF | cerebrospinal fluid |
| efAD | familial early-onset forms of AD |
| FAD | mutation of APP, is associated with a loss of its synaptotrophic activity in human |
| GSH | glutathione |
| HDL | high-density lipoprotein cholesterol |
| i.c.v.i. | intra-cerebro-ventricular injection |
| i.h.i. | intra-hippocampal injection |
| i.n. | intra-nasal administration |
| i.v.i. | intra-venous injection |
| L-DOPA | L-3,4-dihydroxy-phenylalanine |
| LTM | long-term memory |
| M | Morris water maze test |
| MAPT | encodes microtubule associated protein tau |
| MCI | mild cognitive impairment |
| MDA | malondialdehyde, a lipid peroxidation marker |
| N | Novel object recognition test |
| NFTs | neurofibrillary tangles |
| NP(s) | nanoparticle(s) |
| NS | nucleation site |
| NT | nanotechnology |
| O | Open field test |
| P | Passive avoidance response test |
| p.o. | oral administration |
| PAW | plasmon-activated water |
| PEG | polyethylene glycol |
| PLA | poly(lactic acid) |
| PLGA | poly(lactic-co-glycolic acid) |
| PPaRγ | peroxisome proliferator-activated receptor gamma |
| PSEN1 | presenilin 1 protein encoded by the PSEN1 gene in humans |
| R | Rectangular-maze test |
| sAD | late-onset, sporadic form of AD |
| siRNA | silencing RNA |
| STM | short-term memory |
| STZ | streptozotocin |
| ● | observed in vivo effect (in Table 3) or used memory test (in Table 5) |
| ❍ | observed in vitro effect (in Table 3) |
Appendix A
| Nanoparticles (NPs), Animal Model, and Treatment Regime | NPs Cross BBB | LTM Assessment | In Vivo Data Supporting NPs Elicited Reduced Memory Loss | Refs. |
|---|---|---|---|---|
| Mouse animal models | ||||
| 01-curcumin lipid-core NPs 10 or 1 mg/kg of NPs, p.o. for 14 days, after a single i.c.v.i. of Aβ1-42 (400 pmol/animal) to aged Swiss Albino mice. | Yes | M | Decreased levels of inflammatory cytokines in prefrontal cortex, hippocampus and serum of NPs treated mice, compared to untreated controls. | [36] |
| 02-Donepezil apolipoprotein A1 reconstituted HDL NPs for Aβ-targeting clearance and acetylcholinesterase inhibition tested in AD model of adult nude mice with one bilateral i.h.i. of Aβ1-42, (5μL of 82μM solution), followed by an i.v.i. daily of NPs with a donepezil dose of 1mg/kg, for 4 weeks. | Yes | M | NPs treated AD model rats had less neuronal damage, attenuated acetylcholinesterase activity and less amyloid β plaques deposition in mice brains, compared to untreated controls. | [35] |
| Transgenic mice animal models | ||||
| 03-β-sheet breaker (H102)-loaded PEG-PLA NPs modified with TGN peptides (as the BBB ligand) and QSH peptides for enhanced Aβ1-42-binding APP/PS1 double transgenic mice treated with an i.v.i. 250 μg/kg per day, for 19-consecutive days. | Yes | M | Decreased amyloid β plaques size and number, decreased tau protein phosphorylation and reduced synaptic loss in hippocampus of NPs treated mice. | [42] |
| 04-Biodegradable, PEGylated NPs surface functionalized with an antibody directed against Aβ1-42 B6; SJL-Tg(APPSWE)2576Kha, Taconic Europe transgenic mice treated once every other day, an i.v.i. with 100 μL (40 mg/kg of polymer) of fluorescently-labelled anti-Aβ1-42-NPs (0.8 mg/kg of antibody) for 3-weeks. | No | N | NPs promoted the sink effect: reduction of soluble Aβ1-42 and Aβ oligomer concentrations in mice brains with a concomitant increase in Aβ1-42 plasma levels. | [50] |
| 05-Biodegradable, PLGA NPs loaded with Vitamin D-binding protein 5XFAD transgenic mice treated with an i.v.i. of NPs 2.5 mg/kg, for 4-weeks. | Yes | N | Cortical changes in mice were: reduced Aβ1-42 peptide accumulation, neuroinflammation and neuronal death. | [56] |
| 06-CRISPR–Cas9 amphiphilic NPs 5XFAD transgenic mice treated with a single injection of 10 μL of Cas9 nanocomplex into the CA3 hippocampal region. | No | M | Hippocampus of NPs treated mice had: (a) less inflammation, reactive microglia, apoptosis; (b) a significantly decreased Bace1 expression and concomitantly reduced production of APP β-cleavage products; and (3) a reduced amyloid β plaques formation. | [57] |
| 07-Curcumin-loaded self-nano micellizing solid dispersion system NPs APP/PS1 double transgenic mice treated p.o. in drinking water, dose of NPs equivalent to a curcumin dose of 47 mg/kg, for 3-months. | Yes | M, N | The NPs better protected cultured neuroblastoma cells against copper metal ion, H2O2, and Aβ1-42 oligomers cytotoxicity then curcumin only. NPs treated mice had memory performance comparable to younger transgenic mice when the AD-like behavioural deficit has not yet developed. | [46] |
| 08-D-glutathione stabilised gold NPs APP/PS1 double transgenic mice treated with an i.v.i. of NPs, 25 mg/kg, every week for 4 weeks. | Yes | M | Compared to untreated mice, NPs treated animals had a reduced amyloid β plaques deposition in hippocampus. | [45] |
| 09-dual function self-destructive NPs from peptide-polymers that capture Aβ and promote its degradation by stimulating autophagy APP/PS1 double transgenic mice treated with an i.v.i. of cyclosporine (10 μM), followed by an i.v.i. of a single dose of 200 μg·mL−1 nano sweeper for 8-consecutive days. | Yes | M | Decreased soluble and insoluble Aβ1-42 levels in mice brain homogenates due to an upregulated autophagy. | [48] |
| 10-Epigallocatechin-3-gallate formulated as dual-drug loaded PEGylated PLGA NPs APP/PS1 double transgenic mice treated with single daily p.o. 40 mg/kg, for 3-months. | Yes | M, N | NPs treated mice had enhanced SYN staining in CA3 region of hippocampus (indicating increased synaptic expression) and untreated mice had a reduced SYN staining in the same brain region. Compared to untreated controls, NPs treated mice had reduced neuroinflammation and amyloid β plaques accumulation in hippocampus and reduced accumulation of soluble and insoluble Aβ1-42 in cortical samples. In summary, cortical changes in mice are: reduced soluble and insoluble Aβ1-42 concentration; reduced inflammation; and increased synapse density. | [39] |
| 11-Magnetite/Ceria NPs Assemblies (MCNA-Aβ1-42 antibodies conjugated to NPs) Extra-corporeal circulation, in an anesthetised 5XFAD transgenic mouse, from femoral vein blood to jugular vein with a 150 μL min−1 flow rate, established in an. Sequestration of Aβ1-42s performed by injecting an MCNA solution (1.8 × 10−3 m [Fe]) at starting point of extracorporeal circuit and removing the antibody bound Aβ1-42 by external magnet at the end point of extra corporal circuit. | No | N | The extra corporal Aβ1-42 blood cleansing system reduced Aβ peptide concentrations in blood and brain tissue of mice. | [12] |
| 12-Memantine loaded PLGA PEGylated NPs APP/PS1 and C57BL/6 transgenic mice treated p.o. with NPs, with a memantine therapeutic dose of 30 mg/kg on alternate days, for 2-months. | Yes | M | Reduced number of amyloid β plaques and inflammation markers in mouse brain histology samples. | [37] |
| 13-PEGylated dendrigraft poly-L-lysine loaded NPs with BACE1 mRNA silencing gene and D-peptide to inhibit p-tau-associated fibril formation APP/PS1 double transgenic mice treated with i.v., once a week (350 μg) for 5-weeks. | Yes | M | Cortical changes in mice: reduced extracellular formation of amyloid β plaques and reduced intracellular formation of tau-fibrils. | [47] |
| 14-Poly(propylene imine) dendrimers with histidine-maltose APP/PS1 double transgenic mice treated with NP i.n., 10 mg/kg, for 3 months. | Yes | N | Mouse brain: no significant changes in size or number of amyloid β plaques, nor in oxidative stress markers or ratio and levels of soluble Aβ42 to Aβ40. Attenuated Aβ1-42 aggregation and Aβ protein toxicity in cultured neuroblastoma cells. | [38] |
| 15-PPaRγ agonist-loaded PLGA-PEG NPs APP/PS1 double transgenic mice treated with 10 mg/kg administered p.o., once a day, 5 days per week, for 4-weeks. | Yes | N | amyloid β plaques deposition was reduced in mouse cortex. | [40] |
| 16-Resonantly illuminated gold NPs generating negatively charged plasmon-activated water (PAW) APP/PS1 double transgenic mice treated p.o. ad libitum, for 9 months with PAW generated with NPs that reduce the hydrogen- bonded structure of water, giving PAW anti-oxidative and anti-inflammatory effects. | No | N | Compared to untreated transgenic mice, PAW treated mice had a reduced amyloid β plaques and p-tau burden in the hippocampus. PAW treatment reduced the levels of pro-inflammatory cytokines IL-1β and IL-6 in animals’ brains, however, the reduction was not statistically significant. | [43] |
| 17-Resveratrol loaded mesoporous nano-selenium release delivery system based on borneol target, β-cyclodextrin nano valves APP/PS1 double transgenic mice treated with an i.v.i. of 1 mg/kg per day, repeated for 14-days. | Yes | M | Brain histology of NPs treated mice showed reduced amyloid β plaques formation, tau hyperphosphorylation and loss of neurons (i.e., an increased number of Nissl bodies). | [41] |
| 18-Sphere-like sulphur nanoparticles RVG@Met@SNPs APP/PS1 double transgenic mice treated with an i.v.i. of 5.0 mg/kg, 2-times per week (Monday and Thursday), 8 injections in 4-weeks. | Yes | M | In a cell model, NPs significantly reduced Aβ1-42 self-aggregation and, by absorption of Cu 2+, aggregation of Aβ petide−Cu2+ complex. The brain location of NPs in vivo was confirmed with mouse real-time imaging combined with X-ray location. | [11] |
| 19-Zinc loaded NPs APP23 transgenic mice received 2 daily i.p. of NPs (total daily amount of 392 μg Zn) for 14-consecutive days. | Yes | Co | Brain histology of NPs treated mice showed amyloid β plaques with a significantly decreased size. Brain expression levels of proinflammatory interleukins were significantly decreased and anti-inflammatory interleukin expression levels were comparable to normal mice controls. However, no significant change in brain Aβ fibrillary or oligomer levels in NPs treated mice. | [49] |
| Rat animal models | ||||
| 20-Berberine-loaded multiwalled carbon nanotubes with polysorbate and phospholipid coating AD model induced in adult Wistar rats by a single β amyloid i.c.v.i. followed by NPs treatment with an i.v.i. of NPs equivalent to 10 mg/kg of berberine. | Yes | M | Greater changes, consistent with β amyloid peptides toxic effect, in brain homogenates of NPs untreated animals (more increased malondialdehyde and nitrite levels, and more reduced glutathione levels) compared to NPs treated rats. | [32] |
| 21-Diphtheria toxoid NPs AD model in adult Wistar rats with a single i.c.v.i. of Aβ oligomers (10 μL of 1 μg/μL), 14 days after a single i.n. NPs treatment of 15 Lf diphtheria vaccine in 40μL volume. | Yes | M | Compared to untreated AD model controls, NPs inhibited XBP-1 mRNA gene splicing (an early marker for ER stress, elicited by the presence of Aβ oligomers that elicit neuroinflammation, mitochondrial dysfunction, oxidative stress, and apoptosis. | [53] |
| 22-pigallocatechin-gallate loaded NPs, AD model in adult Wistar rats after chronic administration of AlCl3 (100 mg/kg p.o. for 60 days), followed by NPs treatment 10 mg/kg per day, p.o., for 30 days. | Yes | M, N | The quantity of brain markers for AlCl3 brain lesions, amyloid β plaques and neurofibrillary tangles, was significantly reduced in NPs treated rats. | [34] |
| 23-Erythropoietin solid lipid NPs AD model in adult Wistar rats with one bilateral i.h.i. of Aβ1-42 (concentration 0.5 μg/μL, 2 μL of suspension per site) on day 1, followed by NPs treatment with a single i.p. dose of 1250 IU/kg or 2500 IU/kg on days 2, 4, 6, 8, 10 and 12. | Yes | M | NPs treated animals, at both daily doses, reduced (a) oxidative stress, (b) ADP/ATP ration, and amyloid β plaques deposition in rat′s hippocampus, compared to untreated controls. | [51] |
| 24-Gold NPs AD model in adult Wistar rats with one bilateral i.h.i. of Aβ1-42 (1 μL of 1 μg/μL to each side) on day one, followed by NPs treatment, i.p. 200 μg/mL/kg on days 4, 8, 12 and 16. | Yes | M | Hippocampal changes in NPs treated AD model rats are (a) an improved nerve survival, measured by increased BDNF, CREB, STIM1 and 2 expression; and (b) a reduced size and number of amyloid β plaques. | [52] |
| 25-Metformin phosphatidylserine NPs NPs treatment of adult Wistar rats with 50 mg/kg, i.p., for 16 days, and a single i.c.v.i. of STZ (3 mg/kg) on day 2. | Yes | M | NPs attenuated the STZ elicited (a) the increase in cytokine levels IL1-β, TNF-α, and TGF-β and (b) nerve cell death and degeneration in NPs, compared to control. | [55] |
| 26-Nano-hesperetin STZ induced model of AD in adult Wistar rats with a single i.c.v.i. of STZ (3 mg/kg) and NP treatment with 20 mg/kg, p.o., daily, for 3-weeks. | Yes | N, P | Rat hippocampal area: increased activity of antioxidant enzymes superoxide dismutase, glutathione peroxidase, glutathione reductase and catalase. | [54] |
| 27-Nicotinamide loaded solid lipid NPs functionalized with phosphatidylserine STZ induced model of AD in adult Sprague-Dawley rats with a bilateral i.c.v.i. of 3 mg/kg repeated after 48 h and followed by NP treatment. i.p. 200 mg/kg per day, every other day, 4 injections in total. | Yes | M | Brain histology of NPs treated rats showed a reduced tau hyperphosphorylation, and a reduced number of apoptotic of neurons. | [31] |
| 28-Silica/iron oxide and ceria/tau binding peptide NPs, loaded with a tau aggregation inhibitor methylene blue Okadaic acid induced AD model in adult Sprague-Dawley rats with a single, unilateral i.h.i. (300 ng in 1.5 μL of saline), followed 5 days later by NPs treatment with a single unilateral, i.h.i. of 10 μL silica/iron oxide and ceria/tau binding peptide NPs | Yes | M | NPs treatment attenuated brain neuroinflammation and tau hyperphosphorylation, compared to untreated control. | [13] |
| 29-Quercetin NPs AD model in adult Wistar rats with a single injection of scopolamine followed by NPs treatment 30 mg/kg p.o., for 8-consecutive days. | Yes | Co, R | NPs treatment of rats: (a) attenuated the rise in scopolamine-associated biochemical (malondialdehyde lipid peroxidation and AChE levels) and morphological (gliosis) brain damage markers and (b) enabled near to normal levels of catalase and glutathione. | [33] |
| 30-Tetrahedral DNA NPs AD model in adult Sprague-Dawley rats with one bilateral i.h.i. of Aβ1-40 (10 μL of 1 μg/μL), followed by NPs treatment i.v. 100 μL daily for 21 days. | Yes | M | Compared to untreated AD model controls, the hippocampus of NPs treated rats had (a) a higher number of neurons, (b) a reduced amyloid β plaques deposition and (c) reduced expression levels of pro-apoptotic signalling molecules caspase-3 and Bax and a normalised expression level of the anti-apoptotic Bcl-2. | [44] |
References
- Athira, S.; Nadukkandy, P.; Mohanan, P. Interaction of nanoparticles with central nervous system and its consequences. Am. J. Res. Med. Sci. 2018, 4, 12. [Google Scholar] [CrossRef]
- Teleanu, D.M.; Chircov, C.; Grumezescu, A.M.; Volceanov, A.; Teleanu, R.I. Impact of nanoparticles on brain health: An up to date overview. J. Clin. Med. 2018, 7, 490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saraiva, C.; Praca, C.; Ferreira, R.; Santos, T.; Ferreira, L.; Bernardino, L. Nanoparticle-mediated brain drug delivery: Overcoming blood-brain barrier to treat neurodegenerative diseases. J. Control. Release 2016, 235, 34–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajipour, M.J.; Santoso, M.R.; Rezaee, F.; Aghaverdi, H.; Mahmoudi, M.; Perry, G. Advances in alzheimer’s diagnosis and therapy: The implications of nanotechnology. Trends Biotechnol. 2017, 35, 937–953. [Google Scholar] [CrossRef] [PubMed]
- Pires, P.C.; Santos, A.O. Nanosystems in nose-to-brain drug delivery: A review of non-clinical brain targeting studies. J. Control. Release 2018, 270, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Gupta, J.; Fatima, M.T.; Islam, Z.; Khan, R.H.; Uversky, V.N.; Salahuddin, P. Nanoparticle formulations in the diagnosis and therapy of alzheimer’s disease. Int. J. Biol. Macromol. 2019, 130, 515–526. [Google Scholar] [CrossRef]
- Fonseca-Santos, B.; Gremiao, M.P.; Chorilli, M. Nanotechnology-based drug delivery systems for the treatment of alzheimer’s disease. Int. J. Nanomed. 2015, 10, 4981–5003. [Google Scholar] [CrossRef] [Green Version]
- de la Torre, C.; Cena, V. The delivery challenge in neurodegenerative disorders: The nanoparticles role in alzheimer’s disease therapeutics and diagnostics. Pharmaceutics 2018, 10, 190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, K.H.; Riaz, M.K.; Xie, Y.N.; Zhang, X.; Liu, Q.; Chen, H.J.; Bian, Z.X.; Chen, X.Y.; Lu, A.P.; Yang, Z.J. Review of current strategies for delivering alzheimer’s disease drugs across the blood-brain barrier. Int. J. Mol. Sci. 2019, 20, 381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greish, K.; Alqahtani, A.A.; Alotaibi, A.F.; Abdulla, A.M.; Bukelly, A.T.; Alsobyani, F.M.; Alharbi, G.H.; Alkiyumi, I.S.; Aldawish, M.M.; Alshahrani, T.F.; et al. The effect of silver nanoparticles on learning, memory and social interaction in balb/c mice. Int. J. Env. Res. Public Health 2019, 16, 148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.; Xie, W.; Zhu, X.; Xu, M.; Liu, J. Sulfur nanoparticles with novel morphologies coupled with brain-targeting peptides rvg as a new type of inhibitor against metal-induced abeta aggregation. ACS Chem. Neurosci. 2018, 9, 749–761. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kwon, H.J.; Hyeon, T. Magnetite/ceria nanoparticle assemblies for extracorporeal cleansing of amyloid-beta in alzheimer’s disease. Adv. Mater. 2019, 31, e1807965. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Du, Y.; Zhang, K.; Liang, Z.; Li, J.; Yu, H.; Ren, R.; Feng, J.; Jin, Z.; Li, F.; et al. Tau-targeted multifunctional nanocomposite for combinational therapy of alzheimer’s disease. ACS Nano 2018, 12, 1321–1338. [Google Scholar] [CrossRef] [PubMed]
- Chenthamara, D.; Subramaniam, S.; Ramakrishnan, S.G.; Krishnaswamy, S.; Essa, M.M.; Lin, F.H.; Qoronfleh, M.W. Therapeutic efficacy of nanoparticles and routes of administration. Biomater. Res. 2019, 23, 20. [Google Scholar] [CrossRef] [PubMed]
- Pulgar, V.M. Transcytosis to cross the blood brain barrier, new advancements and challenges. Front. Neurosci. 2018, 12, 1019. [Google Scholar] [CrossRef] [PubMed]
- Moura, R.P.; Martins, C.; Pinto, S.; Sousa, F.; Sarmento, B. Blood-brain barrier receptors and transporters: An insight on their function and how to exploit them through nanotechnology. Expert. Opin. Drug. Deliv. 2019, 16, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Bisaz, R.; Travaglia, A.; Alberini, C.M. The neurobiological bases of memory formation: From physiological conditions to psychopathology. Psychopathology 2014, 47, 347–356. [Google Scholar] [CrossRef] [Green Version]
- Mayford, M.; Siegelbaum, S.A.; Kandel, E.R. Synapses and memory storage. Cold. Spring. Harb. Perspect. Biol. 2012, 4, a005751. [Google Scholar] [CrossRef]
- Brothers, H.M.; Gosztyla, M.L.; Robinson, S.R. The physiological roles of amyloid-beta peptide hint at new ways to treat alzheimer’s disease. Front. Aging Neurosci. 2018, 10, 118. [Google Scholar] [CrossRef]
- Matthews, B.R. Memory dysfunction. Continuum 2015, 21, 613–626. [Google Scholar] [CrossRef] [Green Version]
- Sperling, R.A.; Dickerson, B.C.; Pihlajamaki, M.; Vannini, P.; LaViolette, P.S.; Vitolo, O.V.; Hedden, T.; Becker, J.A.; Rentz, D.M.; Selkoe, D.J.; et al. Functional alterations in memory networks in early alzheimer’s disease. Neuromol. Med. 2010, 12, 27–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Ipolyi, A.R.; Rankin, K.P.; Mucke, L.; Miller, B.L.; Gorno-Tempini, M.L. Spatial cognition and the human navigation network in ad and mci. Neurology 2007, 69, 986–997. [Google Scholar] [CrossRef] [PubMed]
- Drummond, E.; Wisniewski, T. Alzheimer’s disease: Experimental models and reality. Acta Neuropathol. 2017, 133, 155–175. [Google Scholar] [CrossRef] [PubMed]
- Wimo, A.; Jonsson, L.; Winblad, B. An estimate of the worldwide prevalence and direct costs of dementia in 2003. Dement. Geriatr. Cogn. Disord. 2006, 21, 175–181. [Google Scholar] [CrossRef]
- Wingo, T.S.; Cutler, D.J.; Wingo, A.P.; Le, N.A.; Rabinovici, G.D.; Miller, B.L.; Lah, J.J.; Levey, A.I. Association of early-onset alzheimer disease with elevated low-density lipoprotein cholesterol levels and rare genetic coding variants of apob. JAMA Neurol. 2019, 76, 809–817. [Google Scholar] [CrossRef]
- Mayeux, R. Gene-environment interaction in late-onset alzheimer disease: The role of apolipoprotein-epsilon4. Alzheimer Dis. Assoc. Disord. 1998, 12 (Suppl. 3), S10–S15. Available online: https://www.ncbi.nlm.nih.gov/pubmed/9876937 (accessed on 21 January 2021).
- Bufill, E.; Carbonell, E. Apolipoprotein e polymorphism and neuronal plasticity. Am. J. Hum. Biol. 2006, 18, 556–558. [Google Scholar] [CrossRef]
- Holger, J. Memory loss in alzheimer’s disease. Clin. Res. 2013, 15, 445–454. [Google Scholar]
- Terry, R.D.; Katzman, R. Life span and synapses: Will there be a primary senile dementia? Neurobiol. Aging 2001, 22, 347–348. [Google Scholar] [CrossRef]
- van Loenhoud, A.C.; Wink, A.M.; Groot, C.; Verfaillie, S.C.J.; Twisk, J.; Barkhof, F.; van Berckel, B.; Scheltens, P.; van der Flier, W.M.; Ossenkoppele, R. A neuroimaging approach to capture cognitive reserve: Application to alzheimer’s disease. Hum. Brain Mapp. 2017, 38, 4703–4715. [Google Scholar] [CrossRef] [Green Version]
- Vakilinezhad, M.A.; Amini, A.; Akbari Javar, H.; Baha’addini Beigi Zarandi, B.F.; Montaseri, H.; Dinarvand, R. Nicotinamide loaded functionalized solid lipid nanoparticles improves cognition in alzheimer’s disease animal model by reducing tau hyperphosphorylation. DARU 2018, 26, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Lohan, S.; Raza, K.; Mehta, S.K.; Bhatti, G.K.; Saini, S.; Singh, B. Anti-alzheimer’s potential of berberine using surface decorated multi-walled carbon nanotubes: A preclinical evidence. Int. J. Pharm. 2017, 530, 263–278. [Google Scholar] [CrossRef] [PubMed]
- Palle, S.; Neerati, P. Quercetin nanoparticles attenuates scopolamine induced spatial memory deficits and pathological damages in rats. Bull. Fac. Pharm. Cairo Univ. Cairo Univ. 2017, 55, 101–106. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.A.; Bhardwaj, V.; Ravi, C.; Ramesh, N.; Mandal, A.K.A.; Khan, Z.A. Egcg nanoparticles attenuate aluminum chloride induced neurobehavioral deficits, beta amyloid and tau pathology in a rat model of alzheimer’s disease. Front. Aging Neurosci. 2018, 10, 244. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhao, Y.; Yu, M.; Zhao, Z.; Liu, P.; Cheng, H.; Ji, Y.; Jin, Y.; Sun, B.; Zhou, J.; et al. Reassembly of native components with donepezil to execute dual-missions in alzheimer’s disease therapy. J. Control Release 2019, 296, 14–28. [Google Scholar] [CrossRef] [PubMed]
- Giacomeli, R.; Izoton, J.C.; Dos Santos, R.B.; Boeira, S.P.; Jesse, C.R.; Haas, S.E. Neuroprotective effects of curcumin lipid-core nanocapsules in a model alzheimer’s disease induced by beta-amyloid 1-42 peptide in aged female mice. Brain Res. 2019, 1721, 146325. [Google Scholar] [CrossRef]
- Sanchez-Lopez, E.; Ettcheto, M.; Egea, M.A.; Espina, M.; Cano, A.; Calpena, A.C.; Camins, A.; Carmona, N.; Silva, A.M.; Souto, E.B.; et al. Memantine loaded plga pegylated nanoparticles for alzheimer’s disease: In vitro and in vivo characterization. J. Nanobiotechnol. 2018, 16, 32. [Google Scholar] [CrossRef]
- Aso, E.; Martinsson, I.; Appelhans, D.; Effenberg, C.; Benseny-Cases, N.; Cladera, J.; Gouras, G.; Ferrer, I.; Klementieva, O. Poly(propylene imine) dendrimers with histidine-maltose shell as novel type of nanoparticles for synapse and memory protection. Nanomedicine 2019, 17, 198–209. [Google Scholar] [CrossRef]
- Cano, A.; Ettcheto, M.; Chang, J.H.; Barroso, E.; Espina, M.; Kuhne, B.A.; Barenys, M.; Auladell, C.; Folch, J.; Souto, E.B.; et al. Dual-drug loaded nanoparticles of epigallocatechin-3-gallate (egcg)/ascorbic acid enhance therapeutic efficacy of egcg in a appswe/ps1de9 alzheimer’s disease mice model. J. Control Release 2019, 301, 62–75. [Google Scholar] [CrossRef]
- Silva-Abreu, M.; Calpena, A.C.; Andres-Benito, P.; Aso, E.; Romero, I.A.; Roig-Carles, D.; Gromnicova, R.; Espina, M.; Ferrer, I.; Garcia, M.L.; et al. Ppargamma agonist-loaded plga-peg nanocarriers as a potential treatment for alzheimer’s disease: In vitro and in vivo studies. Int. J. Nanomed. 2018, 13, 5577–5590. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Wei, C.; Liu, Y.; Xie, W.; Xu, M.; Zhou, H.; Liu, J. Progressive release of mesoporous nano-selenium delivery system for the multi-channel synergistic treatment of alzheimer’s disease. Biomaterials 2019, 197, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Zhang, C.; Guo, Q.; Wan, X.; Shao, X.; Liu, Q.; Zhang, Q. Dual-functional nanoparticles for precise drug delivery to alzheimer’s disease lesions: Targeting mechanisms, pharmacodynamics and safety. Int. J. Pharm. 2017, 525, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.H.; Lin, K.J.; Hong, C.T.; Wu, D.; Chang, H.M.; Liu, C.H.; Hsiao, I.T.; Yang, C.P.; Liu, Y.C.; Hu, C.J. Plasmon-activated water reduces amyloid burden and improves memory in animals with alzheimer’s disease. Sci. Rep. 2019, 9, 13252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, X.; Cui, W.; Xie, X.; Ma, W.; Zhan, Y.; Lin, Y. Treatment of alzheimer’s disease with framework nucleic acids. Cell Prolif. 2020, 53, e12787. [Google Scholar] [CrossRef]
- Hou, K.; Zhao, J.; Wang, H.; Li, B.; Li, K.; Shi, X.; Wan, K.; Ai, J.; Lv, J.; Wang, D.; et al. Chiral gold nanoparticles enantioselectively rescue memory deficits in a mouse model of alzheimer’s disease. Nat. Commun. 2020, 11, 4790. [Google Scholar] [CrossRef]
- Parikh, A.; Kathawala, K.; Li, J.; Chen, C.; Shan, Z.; Cao, X.; Zhou, X.F.; Garg, S. Curcumin-loaded self-nanomicellizing solid dispersion system: Part ii: In vivo safety and efficacy assessment against behavior deficit in alzheimer disease. Drug Deliv. Transl. Res. 2018, 8, 1406–1420. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, R.Q.; Han, L.; Ke, W.L.; Shao, K.; Ye, L.Y.; Lou, J.N.; Jiang, C. Brain-targeting gene delivery and cellular internalization mechanisms for modified rabies virus glycoprotein rvg29 nanoparticles. Biomaterials 2009, 30, 4195–4202. [Google Scholar] [CrossRef]
- Luo, Q.; Lin, Y.X.; Yang, P.P.; Wang, Y.; Qi, G.B.; Qiao, Z.Y.; Li, B.N.; Zhang, K.; Zhang, J.P.; Wang, L.; et al. A self-destructive nanosweeper that captures and clears amyloid beta-peptides. Nat. Commun. 2018, 9, 1802. [Google Scholar] [CrossRef]
- Vilella, A.; Belletti, D.; Sauer, A.K.; Hagmeyer, S.; Sarowar, T.; Masoni, M.; Stasiak, N.; Mulvihill, J.J.E.; Ruozi, B.; Forni, F.; et al. Reduced plaque size and inflammation in the app23 mouse model for alzheimer’s disease after chronic application of polymeric nanoparticles for cns targeted zinc delivery. J. Trace. Elem. Med. Biol. 2018, 49, 210–221. [Google Scholar] [CrossRef]
- Carradori, D.; Balducci, C.; Re, F.; Brambilla, D.; Le Droumaguet, B.; Flores, O.; Gaudin, A.; Mura, S.; Forloni, G.; Ordonez-Gutierrez, L.; et al. Antibody-functionalized polymer nanoparticle leading to memory recovery in alzheimer’s disease-like transgenic mouse model. Nanomedicine 2018, 14, 609–618. [Google Scholar] [CrossRef]
- Dara, T.; Vatanara, A.; Sharifzadeh, M.; Khani, S.; Vakilinezhad, M.A.; Vakhshiteh, F.; Nabi Meybodi, M.; Sadegh Malvajerd, S.; Hassani, S.; Mosaddegh, M.H. Improvement of memory deficits in the rat model of alzheimer’s disease by erythropoietin-loaded solid lipid nanoparticles. Neurobiol. Learn. Mem. 2019, 166, 107082. [Google Scholar] [CrossRef] [PubMed]
- Sanati, M.; Khodagholi, F.; Aminyavari, S.; Ghasemi, F.; Gholami, M.; Kebriaeezadeh, A.; Sabzevari, O.; Hajipour, M.J.; Imani, M.; Mahmoudi, M.; et al. Impact of gold nanoparticles on amyloid beta-induced alzheimer’s disease in a rat animal model: Involvement of stim proteins. ACS Chem. Neurosci. 2019, 10, 2299–2309. [Google Scholar] [CrossRef] [PubMed]
- Heydari, S.; Hedayati Ch, M.; Saadat, F.; Abedinzade, M.; Nikokar, I.; Aboutaleb, E.; Khafri, A.; Mokarram, A.R. Diphtheria toxoid nanoparticles improve learning and memory impairment in animal model of alzheimer’s disease. Pharmacol. Rep. 2020, 72, 814–826. [Google Scholar] [CrossRef] [PubMed]
- Kheradmand, E.; Hajizadeh Moghaddam, A.; Zare, M. Neuroprotective effect of hesperetin and nano-hesperetin on recognition memory impairment and the elevated oxygen stress in rat model of alzheimer’s disease. Biomed. Pharmacother. 2018, 97, 1096–1101. [Google Scholar] [CrossRef]
- Saffari, P.M.; Alijanpour, S.; Takzaree, N.; Sahebgharani, M.; Etemad-Moghadam, S.; Noorbakhsh, F.; Partoazar, A. Metformin loaded phosphatidylserine nanoliposomes improve memory deficit and reduce neuroinflammation in streptozotocin-induced alzheimer’s disease model. Life Sci. 2020, 255, 117861. [Google Scholar] [CrossRef]
- Jeon, S.G.; Cha, M.Y.; Kim, J.I.; Hwang, T.W.; Kim, K.A.; Kim, T.H.; Song, K.C.; Kim, J.J.; Moon, M. Vitamin d-binding protein-loaded plga nanoparticles suppress alzheimer’s disease-related pathology in 5xfad mice. Nanomedicine 2019, 17, 297–307. [Google Scholar] [CrossRef]
- Park, H.; Oh, J.; Shim, G.; Cho, B.; Chang, Y.; Kim, S.; Baek, S.; Kim, H.; Shin, J.; Choi, H.; et al. In vivo neuronal gene editing via crispr-cas9 amphiphilic nanocomplexes alleviates deficits in mouse models of alzheimer’s disease. Nat. Neurosci. 2019, 22, 524–528. [Google Scholar] [CrossRef]
- Norton, S.; Matthews, F.E.; Barnes, D.E.; Yaffe, K.; Brayne, C. Potential for primary prevention of alzheimer’s disease: An analysis of population-based data. Lancet Neurol. 2014, 13, 788–794. [Google Scholar] [CrossRef] [Green Version]
- Zanetti, O.; Solerte, S.B.; Cantoni, F. Life expectancy in alzheimer’s disease (ad). Arch. Gerontol. Geriatr. 2009, 49 (Suppl. 1), 237–243. [Google Scholar] [CrossRef]
- Bateman, R.J.; Xiong, C.; Benzinger, T.L.; Fagan, A.M.; Goate, A.; Fox, N.C.; Marcus, D.S.; Cairns, N.J.; Xie, X.; Blazey, T.M.; et al. Clinical and biomarker changes in dominantly inherited alzheimer’s disease. N. Engl. J. Med. 2012, 367, 795–804. [Google Scholar] [CrossRef] [Green Version]
- Lesne, S.E.; Sherman, M.A.; Grant, M.; Kuskowski, M.; Schneider, J.A.; Bennett, D.A.; Ashe, K.H. Brain amyloid-beta oligomers in ageing and alzheimer’s disease. Brain 2013, 136, 1383–1398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadjichrysanthou, C.; Evans, S.; Bajaj, S.; Siakallis, L.C.; McRae-McKee, K.; de Wolf, F.; Anderson, R.M.; Alzheimer’s Disease Neuroimaging Initiative. The dynamics of biomarkers across the clinical spectrum of alzheimer’s disease. Alzheimer’s Res. Ther. 2020, 12, 74. [Google Scholar] [CrossRef] [PubMed]
- Bilgel, M.; Jedynak, B.M. Predicting time to dementia using a quantitative template of disease progression. Alzheimer’s Dement. 2019, 11, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Venkatraghavan, V.; Bron, E.E.; Niessen, W.J.; Klein, S.; Alzheimer’s Disease Neuroimaging Initiative. Disease progression timeline estimation for alzheimer’s disease using discriminative event based modeling. Neuroimage 2019, 186, 518–532. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.R.; Liu, R.T. The toxicity and polymorphism of beta-amyloid oligomers. Int. J. Mol. Sci. 2020, 21, 4477. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.F.; Xu, T.H.; Yan, Y.; Zhou, Y.R.; Jiang, Y.; Melcher, K.; Xu, H.E. Amyloid beta: Structure, biology and structure-based therapeutic development. Acta Pharmacol. Sin. 2017, 38, 1205–1235. [Google Scholar] [CrossRef]
- Kametani, F.; Hasegawa, M. Reconsideration of amyloid hypothesis and tau hypothesis in alzheimer’s disease. Front. Neurosci-Switz. 2018, 12, 25. [Google Scholar] [CrossRef] [Green Version]
- Spires-Jones, T.L.; Kopeikina, K.J.; Koffie, R.M.; de Calignon, A.; Hyman, B.T. Are tangles as toxic as they look? J. Mol. Neurosci. 2011, 45, 438–444. [Google Scholar] [CrossRef] [Green Version]
- Haass, C.; Selkoe, D.J. Soluble protein oligomers in neurodegeneration: Lessons from the alzheimer’s amyloid beta-peptide. Nat. Rev. Mol. Cell Biol. 2007, 8, 101–112. [Google Scholar] [CrossRef]
- Abbott, A.; Dolgin, E. Failed alzheimer’s trial does not kill leading theory of disease. Nature 2016, 540, 15–16. [Google Scholar] [CrossRef]
- Imbimbo, B.P. Why did tarenflurbil fail in alzheimer’s disease? J. Alzheimer’s Dis. 2009, 17, 757–760. [Google Scholar] [CrossRef] [PubMed]
- Reardon, S. Alzheimer antibody drugs show questionable potential. Nat. Rev. Drug Discov. 2015, 14, 591–592. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L.; Morstorf, T.; Zhong, K. Alzheimer’s disease drug-development pipeline: Few candidates, frequent failures. Alzheimer’s Res. Ther. 2014, 6, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doody, R.S.; Thomas, R.G.; Farlow, M.; Iwatsubo, T.; Vellas, B.; Joffe, S.; Kieburtz, K.; Raman, R.; Sun, X.; Aisen, P.S.; et al. Phase 3 trials of solanezumab for mild-to-moderate alzheimer’s disease. N. Engl. J. Med. 2014, 370, 311–321. [Google Scholar] [CrossRef]
- Salloway, S.; Sperling, R.; Fox, N.C.; Blennow, K.; Klunk, W.; Raskind, M.; Sabbagh, M.; Honig, L.S.; Porsteinsson, A.P.; Ferris, S.; et al. Two phase 3 trials of bapineuzumab in mild-to-moderate alzheimer’s disease. N. Engl. J. Med. 2014, 370, 322–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiman, E.M.; Quiroz, Y.T.; Fleisher, A.S.; Chen, K.; Velez-Pardo, C.; Jimenez-Del-Rio, M.; Fagan, A.M.; Shah, A.R.; Alvarez, S.; Arbelaez, A.; et al. Brain imaging and fluid biomarker analysis in young adults at genetic risk for autosomal dominant alzheimer’s disease in the presenilin 1 e280a kindred: A case-control study. Lancet Neurol. 2012, 11, 1048–1056. [Google Scholar] [CrossRef] [Green Version]
- Corbett, A.; Pickett, J.; Burns, A.; Corcoran, J.; Dunnett, S.B.; Edison, P.; Hagan, J.J.; Holmes, C.; Jones, E.; Katona, C.; et al. Drug repositioning for alzheimer’s disease. Nat. Rev. Drug Discov. 2012, 11, 833–846. [Google Scholar] [CrossRef]
- Mapstone, M.; Cheema, A.K.; Fiandaca, M.S.; Zhong, X.; Mhyre, T.R.; MacArthur, L.H.; Hall, W.J.; Fisher, S.G.; Peterson, D.R.; Haley, J.M.; et al. Plasma phospholipids identify antecedent memory impairment in older adults. Nat. Med. 2014, 20, 415–418. [Google Scholar] [CrossRef]
- Hye, A.; Lynham, S.; Thambisetty, M.; Causevic, M.; Campbell, J.; Byers, H.L.; Hooper, C.; Rijsdijk, F.; Tabrizi, S.J.; Banner, S.; et al. Proteome-based plasma biomarkers for alzheimer’s disease. Brain 2006, 129, 3042–3050. [Google Scholar] [CrossRef] [Green Version]
- ManafiRad, A.; Farzadfar, F.; Habibi, L.; Azhdarzadeh, M.; Aghaverdi, H.; Tehrani, K.H.; Lotfi, M.; Kehoe, P.G.; Sheidaei, A.; Ghasemian, A.; et al. Is amyloid-beta an innocent bystander and marker in alzheimer’s disease? Is the liability of multivalent cation homeostasis and its influence on amyloid-beta function the real mechanism? J. Alzheimer’s Dis. 2014, 42, 69–85. [Google Scholar] [CrossRef]
- Pratico, D. Oxidative stress hypothesis in alzheimer’s disease: A reappraisal. Trends Pharmacol. Sci. 2008, 29, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Rammal, H.; Soulimani, R. Oxidative stress and anxiety: Relationship and cellular pathways. Oxid. Med. Cell. Longev. 2009, 2, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Drummond, E.; Goni, F.; Liu, S.; Prelli, F.; Scholtzova, H.; Wisniewski, T. Potential novel approaches to understand the pathogenesis and treat alzheimer’s disease. J. Alzheimer’s. Dis. 2018, 64, S299–S312. [Google Scholar] [CrossRef] [PubMed]
- Milisav, I.; Suput, D.; Ribaric, S. Unfolded protein response and macroautophagy in alzheimer’s, parkinson’s and prion diseases. Molecules 2015, 20, 22718–22756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Green, K.N.; Steffan, J.S.; Martinez-Coria, H.; Sun, X.; Schreiber, S.S.; Thompson, L.M.; LaFerla, F.M. Nicotinamide restores cognition in alzheimer’s disease transgenic mice via a mechanism involving sirtuin inhibition and selective reduction of thr231-phosphotau. J. Neurosci. 2008, 28, 11500–11510. [Google Scholar] [CrossRef] [PubMed]
- Doody, R.S.; Raman, R.; Farlow, M.; Iwatsubo, T.; Vellas, B.; Joffe, S.; Kieburtz, K.; He, F.; Sun, X.; Thomas, R.G.; et al. A phase 3 trial of semagacestat for treatment of alzheimer’s disease. N. Engl. J. Med. 2013, 369, 341–350. [Google Scholar] [CrossRef]
- De Strooper, B. Lessons from a failed gamma-secretase alzheimer trial. Cell 2014, 159, 721–726. [Google Scholar] [CrossRef] [Green Version]
- King, A. The search for better animal models of alzheimer’s disease. Nature 2018, 559, S13–S15. [Google Scholar] [CrossRef] [Green Version]
- Niu, J.; Straubinger, R.M.; Mager, D.E. Pharmacodynamic drug-drug interactions. Clin. Pharmacol. Ther. 2019, 105, 1395–1406. [Google Scholar] [CrossRef]
- Kang, H.; Mintri, S.; Menon, A.V.; Lee, H.Y.; Choi, H.S.; Kim, J. Pharmacokinetics, pharmacodynamics and toxicology of theranostic nanoparticles. Nanoscale 2015, 7, 18848–18862. [Google Scholar] [CrossRef] [Green Version]
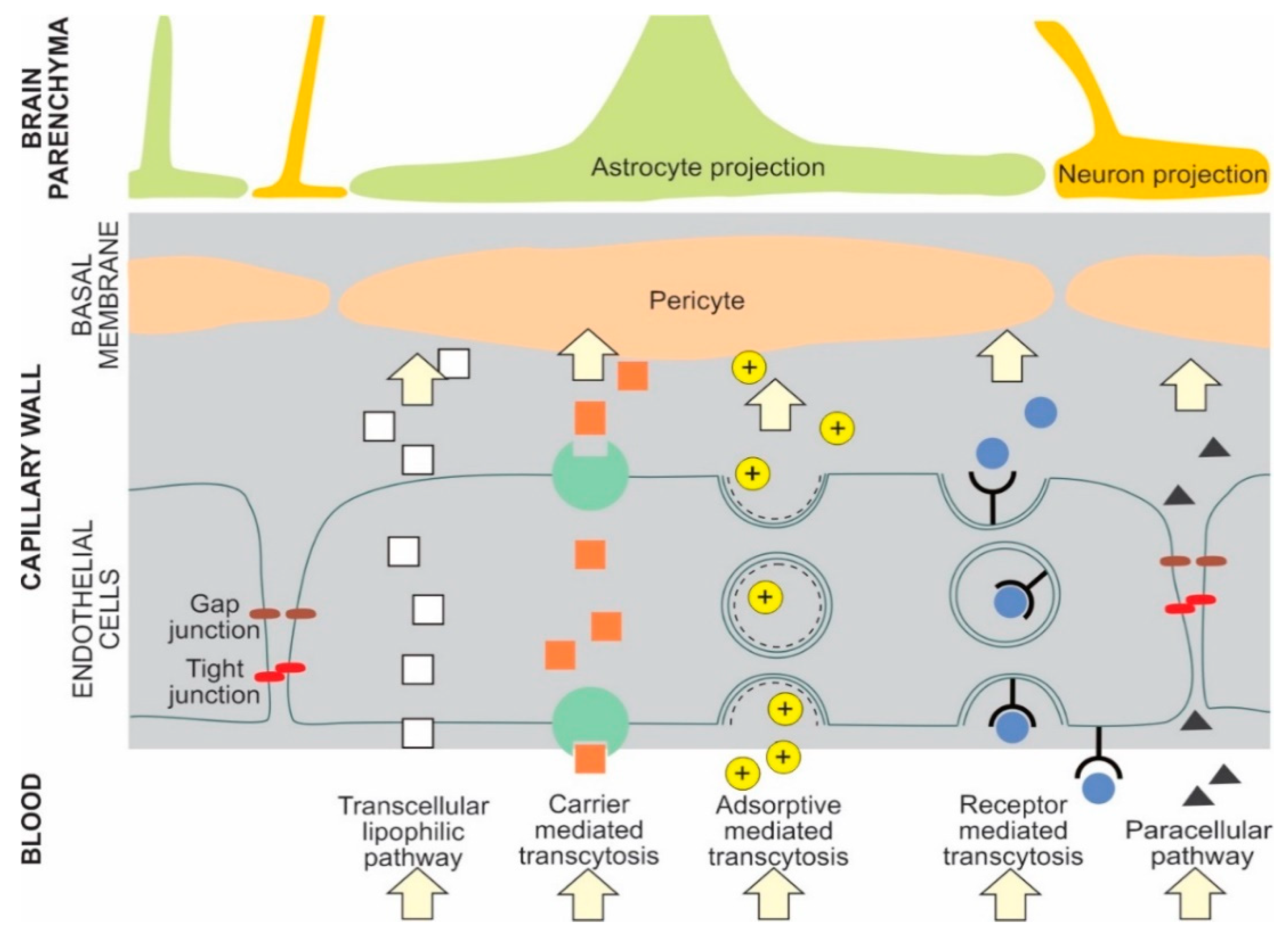
| Nanoparticle Types | Core Structures | Surface Modifications | Cargo |
|---|---|---|---|
| 3-dimensional DNA nanostructures | tetrahedral DNA nanostructures | ||
| Carbon nanotubes | single-walled carbon nanotubes; multi-walled carbon nanotubes | anti-Tau antibody; gold & antibody-binding protein & Aβ antibody; polysorbate or phospholipid coating | acetylcholine; berberine |
| Carbon quantum dots | polymerised o-phenylenediamine quasispheroidal carbon based nanomaterial of quantum size; | ||
| Dendrimers | gallic acid-triethylene glycol; cationic phosphorous dendrimers;poly-amidoamine; poly-propylene-imine | helical β-peptide foldamers; maltose; morpholine groups; tetra-maleimidopropionyl | |
| Gold | gold nanoparticles; gold nanorods | carboxyl groups conjugated to nanoparticles; N-terminal cysteine peptide conjugated to gold nanorods; | |
| Lipid nanoparticles | solid lipid nanoparticle; nanostructured lipid carrier | monoclonal antibodies to transferrin receptors on BBB; pluronic acid; polyethylene glycol and lactoferrin; polysorbate 80 | BACE1 siRNA; curcumin; donepezil; galantamine; resveratrol |
| Liposomes | cholesterol and phosphatidyl-choline; cholesterol and 1,2-distearoyl-sn-glycero-3-phosphocholine; cholesterol and sphingomyelin | cell penetration peptides and polyethylene glycol; phosphatidic acid and Apo-E; phosphatidic acid; polyethylene glycol | curcumin; galantamine; rivastigmine |
| Magnetic nanoparticles | gadopentetic acid; iron oxide (Fe3O4; Fe3O3); magnetite/ceria nanoparticles; polysiloxane matrix with gadolinium chelates; | chitosan and IgG-anti-amyloid antibodies; curcumin and polyethylene glycol and polyvinylpyrrolidone; Aβ oligomers monoclonal antibodies and polyethylene glycol and nitro-L-DOPA; Aβ-antibodies and polyethylene glycol | cyclophos-phamide |
| Polymeric nanoparticles | Poly (lactic-co-glycolic acid); Poly (lactic acid); chitosan; amino-group-modified mesoporous silica nanoparticles; selenium-(poly-lactide-co-glycolide) nanospheres | polyethylene glycol; polysorbate 80; polyethylene glycol and Aβ-binding peptide and targeting peptide to overcome blood brain barrier; tau-binding peptide and iron oxide and ceria nanocrystals | curcumin; galantamine; rivastigmine; tacrine; methylene blue |
| Silver | silver nanoparticles | / | / |
| Sulphur | volute, tadpole or sphere-like nanoparticles | / | / |
| AD Animal Experimental Model | References |
|---|---|
| Mice | |
| Aβ1-42 induced AD model in adult nude mice | [35] |
| Aβ1-42 induced AD model in aged Swiss Albino mice | [36] |
| Transgenic mice | |
| 5XFAD transgenic mice | [12,37,38] |
| APP/PS1 and C57BL/6 transgenic mice | [37] |
| APP/PS1 double transgenic mice | [11,38,39,40,41,42,43,44,45,46,47,48] |
| APP23 transgenic mice | [49] |
| B6; SJL-Tg (APPSWE) 2576Kha, Taconic Europe transgenic mice | [50] |
| Rats | |
| Aβ induced AD model in adult Wistar rats | [32] |
| AlCl3 induced AD model in adult Wistar rats | [34] |
| Aβ1-40 induced AD model in adult Sprague-Dawley rats | [44] |
| Aβ1-42 induced AD model in adult Wistar rats | [51,52] |
| Aβ1-42 induced AD model in adult Wistar rats | [53] |
| okadaic acid induced AD model in adult Sprague-Dawley rats | [13] |
| scopolamine induced AD model in adult Wistar rats | [33] |
| streptozotocin induced AD in adult Wistar rats | [54,55] |
| streptozotocin induced AD model in adult Sprague-Dawley rats | [31] |
| Nanoparticle’s Number, Name & Reference | ↓ Neuro-Inflammation | ↓ AβP Aggregation, ↓Amyloid Plaque Formation | ↓ Tau Hyper Phos-phorylation | ↓ Nerve Cell Death |
|---|---|---|---|---|
| 01-curcumin lipid-core NPs [36] | ● | |||
| 02-donepezil apolipoprotein A1 reconstituted HDL NPs [35] | ● | ● | ||
| 03-β-sheet breaker (H102) PEG-PLA NPs [42] | ● | ● | ||
| 04-PEGylated NPs, with Aβ1-42 Ab [50] | ● | |||
| 05-PLGA NPs with Vitamin D-binding protein [56] | ● | ● | ● | |
| 06-CRISPR–Cas9 amphiphilic NPs [57] | ● | |||
| 07-curcumin NPs [46] | ❍ | ❍ | ||
| 08-D-glutathione stabilised gold NPs [45] | ● | |||
| 09-dual function self-destructive nano sweeper from peptide-polymers [48] | ● | |||
| 10-epigallocatechin-3-gallate/ascorbic acid PEGylated PLGA NPs [39] | ● | ● | ||
| 11-Magnetite/Ceria NPs [12] | ❍ | |||
| 12-memantine PLGA PEGylated NPs [37] | ● | ● | ||
| 13-NPs with BACE1 mRNA silencing gene & D-peptide [47] | ● | ● | ||
| 14-poly (propylene imine) dendrimers with histidine-maltose [38] | ❍ | ❍ | ||
| 15-PPaRγ agonist-loaded PLGA-PEG NPs [40] | ● | |||
| 16-resonantly illuminated gold NPs generating PAW [43] | ● | ● | ||
| 17-resveratrol selenium delivery system NPs [41] | ● | ● | ● | ● |
| 18-sphere-like sulphur NPs [11] | ❍ | ❍ | ||
| 19-zinc NPs [49] | ● | ● | ||
| 20-berberine polysorbate-phospholipid NPs [32] | ● | |||
| 21-Diphtheria toxoid NPs [53] | ● | |||
| 22-epigallocatechin-gallate NPs [34] | ● | ● | ||
| 23-erythropoietin solid lipid NPs [51] | ● | ● | ||
| 24-gold NPs [52] | ● | ● | ||
| 25-metformin phosphatidylserine NPs [55] | ● | ● | ||
| 26-hesperetin NPs [54] | ● | |||
| 27-nicotinamide phosphatidylserine NPs [31] | ● | ● | ||
| 28-silica/tau-binding peptide/iron oxide and ceria/MB NPs [13] | ● | ● | ● | |
| 29-quercetin NPs [33] | ● | ● | ||
| 30-tetrahedral DNA NPs [44] | ● | ● |
| Nanoparticle′s Number, Name & Reference | Morris Water Maze | Novel Object Recognition | Other |
|---|---|---|---|
| 01-curcumin lipid-core NPs [36] | ● | ||
| 02-donepezil apolipoprotein A1 reconstituted HDL NPs [35] | ● | ||
| 03-β-sheet breaker (H102) PEG-PLA NPs [42] | ● | ||
| 04-PEGylated NPs, with Aβ1-42 Ab [50] | ● | ||
| 05-PLGA NPs with Vitamin D-binding protein [56] | ● | ||
| 06-CRISPR–Cas9 amphiphilic NPs [57] | ● | ||
| 07-curcumin NPs [46] | ● | ● | |
| 08-D-glutathione stabilised gold NPs [45] | ● | ||
| 09-dual function self-destructive NP from peptide-polymers [48] | ● | ||
| 10-epigallocatechin-3-gallate/ascorbic acid PEGylated PLGA NPs [39] | ● | ● | |
| 11-Magnetite/Ceria NPs [12] | ● | ||
| 12-memantine PLGA PEGylated NPs [37] | ● | ||
| 13-NPs with BACE1 mRNA silencing gene & D-peptide [47] | ● | ||
| 14-poly (propylene imine) dendrimers with histidine-maltose [38] | ● | ||
| 15-PPaRγ agonist-loaded PLGA-PEG NPs [40] | ● | ||
| 16-resonantly illuminated gold NPs generating PAW [43] | ● | ||
| 17-resveratrol selenium delivery system NPs [41] | ● | ||
| 18-sphere-like sulphur NPs [11] | ● | ||
| 19-zinc NPs [49] | Co | ||
| 20-berberine polysorbate-phospholipid NPs [32] | ● | ||
| 21-Diphtheria toxoid NPs [53] | ● | ||
| 22-epigallocatechin-gallate NPs [34] | ● | ● | |
| 23-erythropoietin solid lipid NPs [51] | ● | ||
| 24-gold NPs [52] | ● | ||
| 25-metformin phosphatidylserine NPs [55] | ● | ||
| 26-hesperetin NPs [54] | ● | P | |
| 27-nicotinamide phosphatidylserine NPs [31] | ● | ||
| 28-silica/tau-binding peptide/iron oxide and ceria/MB NPs [13] | ● | ||
| 29-quercetin NPs [33] | Co, R | ||
| 30-tetrahedral DNA NPs [44] | ● |
| NP’s Number, Reference | i-Core Structure, ii-Surface Modifications and iii-Cargo | Mechanisms of Nanoparticle’s Core Structure, Surface Modifications or Cargo That Attenuate Memory Loss |
|---|---|---|
| 01-, [36] | i- lipid core (sorbitan monostearate dispersion in medium-chain triglycerides core, polymer wall from poly(epsilon-caprolactone), and polysorbate 80 micelles stabilizers) iii- curcumin. | Curcumin attenuates Aβ1-42 elicited neuroinflammation by inhibition of nuclear factor kappa B signalling pathway and reduction of proinflammatory cytokines (e.g., interleukins 1β, 6, and tumour necrosis factor α). |
| 02-, [35] | i- apolipoprotein-A1 reconstituted high-density lipoprotein with antibody-like high Aβ1-42 binding affinity is made from phospholipid vesicles and lipid free apoA-I; iii-donepezil. | The apolipoprotein A1 reconstituted high-density lipoprotein: (a) captures Aβ1-42 and facilitates its degradation in microglial cells by the endo/lysosomal pathway and (b) releases donepezil that inhibits acetylcholinesterase activity and prolongs acetylcholine activity in brain synapses. |
| 03-, [42] | i- poly(ethylene glycol)-poly(lactic acid); ii- TGN peptides to enable crossing the blood-brain barrier and QSH peptides that bind to Aβ1-42. | QSH peptides bind to Aβ1-42 and prevent their oligomerisation, fibrillization and plaque formation by blocking the α→β sheet conversion in Aβ1-42. Also, the nanoparticle attenuated the Aβ1-42 aggregation stimulated tau hyperphosphorylation. |
| 04-, [50] | i- poly(ethylene glycol); ii- antibody against Aβ1-42. | The anti-Aβ1-42-functionalized NPs bound with Aβ1-42 in the blood and reduced the levels of soluble Aβ1-42 peptide and Aβ1-42 oligomers in the brain through the “sink effect”. |
| 05-, [56] | i- D,L-lactic acid-co-glycolic acid; ii- methylpenta(oxyethyl) heptadecanoate; iii- vitamin D-binding protein. | Vitamin D-binding protein binds to Aβ peptides thus preventing (a) their oligomerisation, fibrillization and plaque formation, (b) neuro-inflammation, and (c) cell death. |
| 06-, [57] | i- Cas9, single-guide RNAs targeting BACE1 gene, and amphiphilic R7L10 peptide formed an amphiphilic nanocomplex. | Inhibition of BACE1 gene expression attenuated Aβ1-42 secretion and accumulation of Aβ plaques. |
| 07-, [46] | i- soluplus polymer; iii- curcumin | The improved oral bioavailability enabled sufficient brain concentrations of curcumin to attenuate neuronal cytotoxicity induced by hydrogen peroxide, copper metal ions and Aβ1-42. |
| 08-, [45] | i-gold; ii- antioxidant tripeptides L- and D-glutathione | L- and D-glutathione, conjugated with gold NPs, attenuated aggregation of Aβ1-42 in the brain by their antioxidant effect on reactive oxygen species. |
| 09-, [48] | i- cationic chitosan; ii- GKLVFF peptide (recognises and co-assembles with Aβ1-42 through hydrogen-bonding interactions) and Beclin-1 (stimulates autophagy) attached to polyethylene glycol. | The nanosweeper binds Aβ1-42 in the extracellular brain space, enters the cell and upregulates autophagy, enhances self and Aβ1-42 digestion, and brain parenchyma clearance of soluble and insoluble Aβ1-42 forms. |
| 10-, [39] | i- polyethylene glycol and poly (lactic-co-glycolic acid); iii- epigallocatechin-3-gallate and ascorbic acid (to prevent epigallocate-chin-3-gallate′s oxidation). | Epigallocatechin-3-gallate attenuates accumulation of soluble and insoluble Aβ1-42 forms, inhibits expression of inflammatory interleukins, protects synapses, and increases synaptogenesis. Ascorbic acid contributes to the anti-inflammatory effects of epigallocatechin-3-gallate. |
| 11-, [12] | i- magnetite particles core (enable magnetic isolation of captured Aβ peptides with an external magnetic field) and ceria particles shell (scavenges reactive oxygen species triggered by the experimental animal’s immune response); ii- Aβ1-42-antibodies and polyethylene glycol conjugated to the ceria shell. | The extra corporal Aβ1-42 blood cleansing system reduced Aβ peptide concentrations in the blood and brain tissue of experimental animals. The animal′s immune response, to the Aβ1-42-antibodies, was attenuated by scavenging the reactive oxygen species with the ceria particles. |
| 12-, [37] | i- poly (lactic-co-glycolic acid); ii- polyethylene glycol surface coating; iii- memantine (binds to the N-methyl-D-aspartate receptor-operated cation channels with a low-to-moderate affinity that preserves normal receptor function in response to a physiological release of glutamate at the synapses). | Memantine binds to N-methyl-D-aspartate channels and attenuates excessive glutamate cell stimulation (by inhibiting the prolonged influx of calcium ions) and consequent neuronal death thus improving memory. The memantine loaded NPs also reduce Aβ plaques formation and neuroinflammation. |
| 13-, [47] | i- dendrigraft poly-L-lysine; ii- polyethylene glycol and peptide RVG29 (to enable binding to n-acetylcholine receptors in the blood-brain barrier and brain parenchyma cells); iii- D-peptide (inhibitor of tau fibril formation) and a plasmid DNA encoding β-secretase-antisense shRNA (inhibits expression of β-secretase) | After crossing the blood-brain barrier, the NPs enter the brain cells where D-peptide and β-secretase-antisense shRNA are released from the NPs to decrease intracellular tau fibrils formation and production of Aβ soluble and insoluble forms (i.e., Aβ plaques). |
| 14-, [38] | i- poly(propylene imine) core with a maltose-histidine shell that improves the NPs ability to cross the blood-brain barrier. | The maltose-histidine shell does not inhibit formation of Aβ1-40 fibrils; it stimulates their clumping. Formation of Aβ1-40 oligomers and neuronal death is inhibited in the presence of maltose-histidine. |
| 15-, [40] | i- poly (lactic-co-glycolic acid) and polyethylene glycol; ii-, iii- peroxisome proliferator-activated receptor agonist, pioglitazone (to facilitate crossing of the blood-brain barrier and for anti-inflammatory effects in the brain); | Pioglitazone binds with the peroxisome proliferator-activated receptor to modulate the inflammatory response and reduce Aβ plaques formation in AD model animals. |
| 16- [43] | i- resonantly illuminated gold NPs. | The resonantly illuminated gold NPs reduce the hydrogen bonded structure of water, creating negatively charged plasmon-activated water. This water has anti-oxidative and anti-inflammatory effects that are assumed to attenuate formation of Aβ plaques, tau hyperphosphorylation, and neuroinflammation. |
| 17-, [41] | i- mesoporous nano-selenium; ii- borneol target, β-cyclodextrin and ferrocene nanovalves; iii-resveratrol. | The interactions with blood or intracellular esterases release borneol, enabling the passage of NPs across the blood-brain barrier. Increased concentrations of hydrogen peroxide in the brain parenchyma trigger oxidation of ferrocene. Ferrocene oxidation leads to β-cyclodextrin dissociation and release of resveratrol. Resveratrol inhibits aggregation of Aβ peptides and thus attenuates oxidative stress, and tau hyperphosphorylation in nerve cells. Resveratrol also decreases brain pro-inflammatory cytokines interleukin 6 and tumour necrosis factor α, and increases anti-inflammatory cytokines interleukins 4 and 10. |
| 18-, [11] | i- methionine modified morphology of sphere-like sulphur NPs. | The sphere-like sulphur NPs reduced aggregation of the copper ion-Aβ peptide complexes by; (a) attenuating the interaction between Aβ monomers and copper ions; and (b) interfering with the formation of hydrogen bonds. The NPs also decreased the intracellular reactive oxygen species and attenuated the copper ion-Aβ peptide complexes mediated cell cytotoxicity. |
| 19-, [49] | i- poly(lactic-co-glycolic acid); ii- 7 amino acid glycopeptide conjugated with poly(lactic-co-glycolic acid to enable the crossing of the blood-brain barrier; iii-zinc | The NPs reduce the size of Aβ plaques and the levels of pro-inflammatory interleukins 6 and 18, and increase the levels of anti-inflammatory interleukin 10. |
| 20-, [32] | i- multiple-walled carbon nanotubes; ii- carboxylated polysorbate or carboxylated phospholipid coating; iii-berberine | The NPs′ attenuated memory loss effect was attributed to berberine inhibition of brain oxidative damage induced by Aβ1-42. |
| 21-, [53] | i- chitosan capsule; iii- diphtheria toxoid (to supress the AβOs elicited unfolded protein response, a sing of endoplasmic reticulum stress). In human, the exposure to diphtheria toxoid in adulthood is associated with the significantly reduced risk of AD. | AβOs elicit the cell′s unfolded protein response due to the accumulation of undigested, abnormal protein aggregates of AβOs, mitochondrial dysfunction, oxidative stress, and disruption of calcium homeostasis. Pre-treatment with NPs containing the diphtheria toxoid is assumed to activate an alternative pathway that attenuates the toxic effects of AβOs with a concomitant down regulation of the unfolded protein response. |
| 22-, [34] | i- polyethylene glycol and poly (D, L-lactic acid) shell; iii-epigallocatechin-gallate (has antioxidant and metal chelation properties, promotes formation of less toxic amorphous Aβ1-42 aggregates over toxic, insoluble Aβ1-42 fibrils, and inhibits formation of neurofibrillary tangles by activating the phosphoinositide 3-kinase pathway). | The epigallocatechin-gallate loaded NPs attenuated (a) formation of Aβ plaques and neurofibrillary tangles and (b) reduced oxidative stress markers (NO and reactive oxygen species) and Aβ1-42 levels in the brain. |
| 23-, [51] | i- glycerine monostearate solid lipid NP;iii- erythropoietin (promotes neuronal survival and neurogenesis by (a) nuclear factor kappa-light-chain-enhancer activity of activated B cells stimulation, inhibition of apoptotic proteins, and (b) inhibition of lipid peroxidation, and restoration of the antioxidant enzymes cytosolic catalase and glutathione peroxidase activity). | The erythropoietin loaded NPs reduced the oxidative stress and Aβ plaques deposition in the brain due to inhibition of lipid peroxidation and restoration of the antioxidant enzymes. |
| 24-, [52] | i- gold; ii- citrate conjugated gold NPs. | The NPs; (a) improved neuronal survival by promoting the expression of brain-derived neurotrophic factor, cyclic adenosine monophosphate response element binding protein, and stromal interaction molecules; and (b) inhibited Aβ1-42 aggregation into toxic, soluble Aβ oligomers and fibrils. |
| 25-, [55] | i- phosphatidylserine liposome shell; iii- metformin (reduces interleukin 1β and tumour necrosis factor α elicited neuroinflammation, and oxidative stress). | Metformin loaded liposomes; (a) decreased levels of pro-inflammatory cytokines interleukin 1β, tumour necrosis factor α and transforming growth factor β; and (b) reduced neuroinflammation, and neural cell death in the brain. |
| 26-, [54] | i- hesperetin (has a neuroprotective effect due to scavenging of hydrogen peroxide hydroxyl radicals, and due to attenuation of calcium ions level and caspase-3 activity). | Hesperetin NPs increase the brain’s antioxidant enzymes (catalase, glutathione peroxidase, glutathione reductase and superoxide dismutase), and decrease malondialdehyde (a marker for lipid peroxidation during oxidative stress). |
| 27-, [31] | i- solid lipid from phosphatidylserine; iii- nicotinamide. | Previous research reported that nicotinamide restores cognition in AD model animals by sirtuin inhibition, and selective reduction of phosphorylated tau [85]. Nicotinamide loaded NPs are assumed to attenuate memory loss by the aforementioned mechanisms. |
| 28-, [13] | i- amino-group-modified mesoporous silica NPs; ii- tau-binding peptide, iron oxide and ceria nanocrystals; iii- methylene blue (a tau aggregation inhibitor). | The NPs attenuated memory loss by reducing mitochondrial oxidative stress, neuroinflammation, tau hyperphosphorylation and tau aggregation, and neuronal death in the brain. |
| 29-, [33] | i- quercetin (an antioxidant that attenuates inflammation). | The NPs’ antioxidant effect; (a) increased brain antioxidant enzymes catalase, glutathione peroxidase and glutathione reductase; (b) reduced brain malondialdehyde; and (c) reduced oxidative stress related neuronal death and neuroinflammation in the brain. |
| 30-, [44] | i- tetrahedral DNA nanostructures. | The NPs treatment inhibited Aβ1-40 aggregation into plaques, and mitochondria triggered apoptosis in the brain. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribarič, S. Nanotechnology Therapy for Alzheimer′s Disease Memory Impairment Attenuation. Int. J. Mol. Sci. 2021, 22, 1102. https://doi.org/10.3390/ijms22031102
Ribarič S. Nanotechnology Therapy for Alzheimer′s Disease Memory Impairment Attenuation. International Journal of Molecular Sciences. 2021; 22(3):1102. https://doi.org/10.3390/ijms22031102
Chicago/Turabian StyleRibarič, Samo. 2021. "Nanotechnology Therapy for Alzheimer′s Disease Memory Impairment Attenuation" International Journal of Molecular Sciences 22, no. 3: 1102. https://doi.org/10.3390/ijms22031102





