Post-Translational Modifications of the Energy Guardian AMP-Activated Protein Kinase
Abstract
:1. Introduction
2. Regulation by Phosphorylation
2.1. α-Subunit
2.1.1. Activation Loop
2.1.2. α-Linker
2.1.3. β-SID and ST Loop
2.2. β-Subunit
2.2.1. NH2-Terminal Domain
2.2.2. CBM
2.2.3. β-Linker
2.3. γ-Subunit
3. Other AMPK Modifications (Non-Phosphorylation)
3.1. Ubiquitination
3.2. Sumoylation
3.3. Acetylation
3.4. Methylation
3.5. Oxidative Modifications
4. Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| α-RIM | α-regulatory subunit interacting motif |
| AICAR | 5-aminoimadazole-4-carboxamide-1-β-D-ribofuranoside |
| ACC | acetyl-CoA carboxylase |
| ALD | adrenoleukodystrophy |
| AMP | 5′-adenosine monophosphate |
| AMPK | AMP-activated protein kinase |
| ADaM | allosteric drug and metabolite |
| ADP | 5′-adenosine diphosphate |
| ATP | 5′-adenosine triphosphate |
| α-γ-SBS | α-γ-subunit binding sequence |
| BMMC | bone marrow–derived mast cells |
| BR | BRAFi-resistant melanoma |
| β-SID | β-subunit interacting domain |
| CBM | carbohydrate binding module |
| CaMKK | calcium/calmodulin-dependent kinase kinase |
| CDK4 | cyclin-dependent kinase 4 |
| CIDEA | cell death-inducing DFF45-like effector A |
| CoA | coenzyme A |
| CRBN | cereblon |
| CRL4A | cullin-RING 4A |
| DNA-PK | DNA-dependent protein kinase |
| GID | glucose-induced degradation |
| GSK3 | glycogen synthase kinase 3 |
| HCC | hepatocellular carcinoma |
| HPAEC | human pulmonary arterial endothelial cell |
| HSL | hormone sensitive lipase |
| HTP | high-throughput |
| LKB1 | liver kinase B1 |
| MAGE | melanoma antigens |
| MEF | mouse embryonic fibroblast |
| MKRN1 | makorin ring finger protein 1 |
| MLK3 | mixed-linkage kinase 3 |
| mTORC1 | mammalian (mechanistic) target of rapamycin complex 1 |
| NTD | NH2-terminal domain |
| PEDFR | pigment epithelium derived factor receptor |
| PIAS4/γ | protein inhibitor of activated STAT protein 4/g |
| PKA | protein kinase A |
| PKC | protein kinase C |
| PKD1 | protein kinase D1 |
| PPARγ | peroxisome proliferator activated receptor γ |
| PP2A | protein phosphatase 2A |
| PP2C | protein phosphatase 2C |
| PPM1E | Mg2+/Mn2+-dependent protein phosphatase 1E |
| PTM | post-translational modification |
| RNF44 | RING finger 44 |
| ROS | reactive oxygen species |
| Sip2 | SNF1-interacting protein 2 |
| SIK | salt-inducible kinase |
| Snf1 | sucrose non-fermenting protein 1 |
| STIM1 | stromal interaction molecule 1 |
| SUMO | Small Ubiquitin-like Modifier |
| TAK1 | transforming growth factor-β-activated kinase 1 |
| TNFα | tumour necrosis factor α |
| TRIM27 | tripartite motif-containing protein 27 |
| TSC2 | tuberous sclerosis complex 2 |
| Ub | ubiquitin |
| UBE2O | E2/E3 hybrid ubiquitin-protein ligase |
| ULK1 | Unc-51-like kinase 1 |
| USP10 | ubiquitin-specific-processing protease 10 |
| UPS | ubiquitin-proteasome system |
| VRK1 | vaccinia-related kinase 1 |
| WWP1 | WW domain-containing protein 1 |
References
- Kemp, G.J.; Meyerspeer, M.; Moser, E. Absolute quantification of phosphorus metabolite concentrations in human muscle in vivo by 31P MRS: A quantitative review. NMR Biomed. 2007, 20, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Barclay, C.J. Energy demand and supply in human skeletal muscle. J. Muscle Res. Cell Motil. 2017, 38, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.S.; McCormick, M.C.; Robergs, R.A. Interaction among Skeletal Muscle Metabolic Energy Systems during Intense Exercise. J. Nutr. Metab. 2010, 2010, 905612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karlsson, J.; Nordesjo, L.O.; Jorfeldt, L.; Saltin, B. Muscle lactate, ATP, and CP levels during exercise after physical training in man. J. Appl. Physiol. 1972, 33, 199–203. [Google Scholar] [CrossRef]
- Nicotera, P.; Leist, M.; Ferrando-May, E. Intracellular ATP, a switch in the decision between apoptosis and necrosis. Toxicol. Lett. 1998, 102–103, 139–142. [Google Scholar] [CrossRef] [Green Version]
- Perry, C.G.; Lally, J.; Holloway, G.P.; Heigenhauser, G.J.; Bonen, A.; Spriet, L.L. Repeated transient mRNA bursts precede increases in transcriptional and mitochondrial proteins during training in human skeletal muscle. J. Physiol. 2010, 588, 4795–4810. [Google Scholar] [CrossRef]
- Hingst, J.R.; Bruhn, L.; Hansen, M.B.; Rosschou, M.F.; Birk, J.B.; Fentz, J.; Foretz, M.; Viollet, B.; Sakamoto, K.; Faergeman, N.J.; et al. Exercise-induced molecular mechanisms promoting glycogen supercompensation in human skeletal muscle. Mol. Metab. 2018, 16, 24–34. [Google Scholar] [CrossRef]
- Bergstrom, J.; Hultman, E. Muscle glycogen synthesis after exercise: An enhancing factor localized to the muscle cells in man. Nature 1966, 210, 309–310. [Google Scholar] [CrossRef]
- Oakhill, J.S.; Scott, J.W.; Kemp, B.E. AMPK functions as an adenylate charge-regulated protein kinase. Trends Endocrinol. Metab. 2012, 23, 125–132. [Google Scholar] [CrossRef]
- Nelson, M.E.; Parker, B.L.; Burchfield, J.G.; Hoffman, N.J.; Needham, E.J.; Cooke, K.C.; Naim, T.; Sylow, L.; Ling, N.X.; Francis, D.; et al. Phosphoproteomics reveals conserved exercise-stimulated signaling and AMPK regulation of store-operated calcium entry. EMBO J. 2019, 38, e102578. [Google Scholar] [CrossRef]
- Vara-Ciruelos, D.; Russell, F.M.; Hardie, D.G. The strange case of AMPK and cancer: Dr Jekyll or Mr Hyde? (dagger). Open Biol. 2019, 9, 190099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.J.; Moon, J.; Chung, I.; Chung, J.H.; Park, C.; Lee, J.O.; Han, J.A.; Kang, M.J.; Yoo, E.H.; Kwak, S.Y.; et al. ATP synthase inhibitory factor 1 (IF1), a novel myokine, regulates glucose metabolism by AMPK and Akt dual pathways. FASEB J. 2019, 33, 14825–14840. [Google Scholar] [CrossRef] [Green Version]
- Xiao, B.; Sanders, M.J.; Carmena, D.; Bright, N.J.; Haire, L.F.; Underwood, E.; Patel, B.R.; Heath, R.B.; Walker, P.A.; Hallen, S.; et al. Structural basis of AMPK regulation by small molecule activators. Nat. Commun. 2013, 4, 3017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steinberg, G.R.; Carling, D. AMP-activated protein kinase: The current landscape for drug development. Nat. Rev. Drug Discov. 2019, 18, 527–551. [Google Scholar] [CrossRef] [PubMed]
- Mahlapuu, M.; Johansson, C.; Lindgren, K.; Hjalm, G.; Barnes, B.R.; Krook, A.; Zierath, J.R.; Andersson, L.; Marklund, S. Expression profiling of the gamma-subunit isoforms of AMP-activated protein kinase suggests a major role for gamma3 in white skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E194–E200. [Google Scholar] [CrossRef] [PubMed]
- Barnes, B.R.; Marklund, S.; Steiler, T.L.; Walter, M.; Hjalm, G.; Amarger, V.; Mahlapuu, M.; Leng, Y.; Johansson, C.; Galuska, D.; et al. The 5′-AMP-activated protein kinase gamma3 isoform has a key role in carbohydrate and lipid metabolism in glycolytic skeletal muscle. J. Biol. Chem. 2004, 279, 38441–38447. [Google Scholar] [CrossRef] [Green Version]
- Birk, J.B.; Wojtaszewski, J.F. Predominant alpha2/beta2/gamma3 AMPK activation during exercise in human skeletal muscle. J. Physiol. 2006, 577, 1021–1032. [Google Scholar] [CrossRef]
- Kjobsted, R.; Hingst, J.R.; Fentz, J.; Foretz, M.; Sanz, M.N.; Pehmoller, C.; Shum, M.; Marette, A.; Mounier, R.; Treebak, J.T.; et al. AMPK in skeletal muscle function and metabolism. FASEB J. 2018, 32, 1741–1777. [Google Scholar] [CrossRef] [Green Version]
- Kjobsted, R.; Roll, J.L.W.; Jorgensen, N.O.; Birk, J.B.; Foretz, M.; Viollet, B.; Chadt, A.; Al-Hasani, H.; Wojtaszewski, J.F.P. AMPK and TBC1D1 Regulate Muscle Glucose Uptake After, but Not During, Exercise and Contraction. Diabetes 2019, 68, 1427–1440. [Google Scholar] [CrossRef] [Green Version]
- Hingst, J.R.; Kjobsted, R.; Birk, J.B.; Jorgensen, N.O.; Larsen, M.R.; Kido, K.; Larsen, J.K.; Kjeldsen, S.A.S.; Fentz, J.; Frosig, C.; et al. Inducible deletion of skeletal muscle AMPKalpha reveals that AMPK is required for nucleotide balance but dispensable for muscle glucose uptake and fat oxidation during exercise. Mol. Metab. 2020, 40, 101028. [Google Scholar] [CrossRef]
- Myers, R.W.; Guan, H.P.; Ehrhart, J.; Petrov, A.; Prahalada, S.; Tozzo, E.; Yang, X.; Kurtz, M.M.; Trujillo, M.; Gonzalez Trotter, D.; et al. Systemic pan-AMPK activator MK-8722 improves glucose homeostasis but induces cardiac hypertrophy. Science 2017, 357, 507–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blair, E.; Redwood, C.; Ashrafian, H.; Oliveira, M.; Broxholme, J.; Kerr, B.; Salmon, A.; Ostman-Smith, I.; Watkins, H. Mutations in the gamma(2) subunit of AMP-activated protein kinase cause familial hypertrophic cardiomyopathy: Evidence for the central role of energy compromise in disease pathogenesis. Hum. Mol. Genet. 2001, 10, 1215–1220. [Google Scholar] [CrossRef]
- Gollob, M.H.; Green, M.S.; Tang, A.S.; Gollob, T.; Karibe, A.; Ali Hassan, A.S.; Ahmad, F.; Lozado, R.; Shah, G.; Fananapazir, L.; et al. Identification of a gene responsible for familial Wolff-Parkinson-White syndrome. N. Engl. J. Med. 2001, 344, 1823–1831. [Google Scholar] [CrossRef] [PubMed]
- Yavari, A.; Bellahcene, M.; Bucchi, A.; Sirenko, S.; Pinter, K.; Herring, N.; Jung, J.J.; Tarasov, K.V.; Sharpe, E.J.; Wolfien, M.; et al. Mammalian gamma2 AMPK regulates intrinsic heart rate. Nat. Commun. 2017, 8, 1258. [Google Scholar] [CrossRef] [PubMed]
- Cokorinos, E.C.; Delmore, J.; Reyes, A.R.; Albuquerque, B.; Kjobsted, R.; Jorgensen, N.O.; Tran, J.L.; Jatkar, A.; Cialdea, K.; Esquejo, R.M.; et al. Activation of Skeletal Muscle AMPK Promotes Glucose Disposal and Glucose Lowering in Non-human Primates and Mice. Cell Metab. 2017, 25, 1147–1159.e10. [Google Scholar] [CrossRef] [PubMed]
- Stephenne, X.; Foretz, M.; Taleux, N.; van der Zon, G.C.; Sokal, E.; Hue, L.; Viollet, B.; Guigas, B. Metformin activates AMP-activated protein kinase in primary human hepatocytes by decreasing cellular energy status. Diabetologia 2011, 54, 3101–3110. [Google Scholar] [CrossRef] [Green Version]
- Katwan, O.J.; Alghamdi, F.; Almabrouk, T.A.; Mancini, S.J.; Kennedy, S.; Oakhill, J.S.; Scott, J.W.; Salt, I.P. AMP-activated protein kinase complexes containing the beta2 regulatory subunit are up-regulated during and contribute to adipogenesis. Biochem. J. 2019, 476, 1725–1740. [Google Scholar] [CrossRef] [Green Version]
- Ngoei, K.R.W.; Langendorf, C.G.; Ling, N.X.Y.; Hoque, A.; Varghese, S.; Camerino, M.A.; Walker, S.R.; Bozikis, Y.E.; Dite, T.A.; Ovens, A.J.; et al. Structural Determinants for Small-Molecule Activation of Skeletal Muscle AMPK alpha2beta2gamma1 by the Glucose Importagog SC4. Cell Chem. Biol. 2018, 25, 728–737.e9. [Google Scholar] [CrossRef] [Green Version]
- Hornbeck, P.V.; Zhang, B.; Murray, B.; Kornhauser, J.M.; Latham, V.; Skrzypek, E. PhosphoSitePlus, 2014: Mutations, PTMs and recalibrations. Nucleic Acids Res. 2015, 43, D512–D520. [Google Scholar] [CrossRef] [Green Version]
- Hawley, S.A.; Davison, M.; Woods, A.; Davies, S.P.; Beri, R.K.; Carling, D.; Hardie, D.G. Characterization of the AMP-activated protein kinase kinase from rat liver and identification of threonine 172 as the major site at which it phosphorylates AMP-activated protein kinase. J. Biol. Chem. 1996, 271, 27879–27887. [Google Scholar] [CrossRef] [Green Version]
- Hawley, S.A.; Boudeau, J.; Reid, J.L.; Mustard, K.J.; Udd, L.; Makela, T.P.; Alessi, D.R.; Hardie, D.G. Complexes between the LKB1 tumor suppressor, STRAD alpha/beta and MO25 alpha/beta are upstream kinases in the AMP-activated protein kinase cascade. J. Biol. 2003, 2, 28. [Google Scholar] [CrossRef] [Green Version]
- Shaw, R.J.; Kosmatka, M.; Bardeesy, N.; Hurley, R.L.; Witters, L.A.; DePinho, R.A.; Cantley, L.C. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc. Natl. Acad. Sci. USA 2004, 101, 3329–3335. [Google Scholar] [CrossRef] [Green Version]
- Hawley, S.A.; Pan, D.A.; Mustard, K.J.; Ross, L.; Bain, J.; Edelman, A.M.; Frenguelli, B.G.; Hardie, D.G. Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for AMP-activated protein kinase. Cell Metab. 2005, 2, 9–19. [Google Scholar] [CrossRef] [Green Version]
- Hurley, R.L.; Anderson, K.A.; Franzone, J.M.; Kemp, B.E.; Means, A.R.; Witters, L.A. The Ca2+/calmodulin-dependent protein kinase kinases are AMP-activated protein kinase kinases. J. Biol. Chem. 2005, 280, 29060–29066. [Google Scholar] [CrossRef] [Green Version]
- Momcilovic, M.; Hong, S.P.; Carlson, M. Mammalian TAK1 activates Snf1 protein kinase in yeast and phosphorylates AMP-activated protein kinase in vitro. J. Biol. Chem. 2006, 281, 25336–25343. [Google Scholar] [CrossRef] [Green Version]
- Scholz, R.; Sidler, C.L.; Thali, R.F.; Winssinger, N.; Cheung, P.C.; Neumann, D. Autoactivation of transforming growth factor beta-activated kinase 1 is a sequential bimolecular process. J. Biol. Chem. 2010, 285, 25753–25766. [Google Scholar] [CrossRef] [Green Version]
- Luo, L.; Jiang, S.; Huang, D.; Lu, N.; Luo, Z. MLK3 phophorylates AMPK independently of LKB1. PLoS ONE 2015, 10, e0123927. [Google Scholar] [CrossRef]
- Park, S.; Artan, M.; Han, S.H.; Park, H.H.; Jung, Y.; Hwang, A.B.; Shin, W.S.; Kim, K.T.; Lee, S.V. VRK-1 extends life span by activation of AMPK via phosphorylation. Sci. Adv. 2020, 6. [Google Scholar] [CrossRef]
- Djouder, N.; Tuerk, R.D.; Suter, M.; Salvioni, P.; Thali, R.F.; Scholz, R.; Vaahtomeri, K.; Auchli, Y.; Rechsteiner, H.; Brunisholz, R.A.; et al. PKA phosphorylates and inactivates AMPKalpha to promote efficient lipolysis. EMBO J. 2010, 29, 469–481. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Mejia, I.C.; Lagarrigue, S.; Giralt, A.; Martinez-Carreres, L.; Zanou, N.; Denechaud, P.D.; Castillo-Armengol, J.; Chavey, C.; Orpinell, M.; Delacuisine, B.; et al. CDK4 Phosphorylates AMPKalpha2 to Inhibit Its Activity and Repress Fatty Acid Oxidation. Mol. Cell 2017, 68, 336–349.e6. [Google Scholar] [CrossRef] [Green Version]
- Ling, N.X.Y.; Kaczmarek, A.; Hoque, A.; Davie, E.; Ngoei, K.R.W.; Morrison, K.R.; Smiles, W.J.; Forte, G.M.; Wang, T.; Lie, S.; et al. mTORC1 directly inhibits AMPK to promote cell proliferation under nutrient stress. Nat. Metab. 2020, 2, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Yamada, E.; Okada, S.; Bastie, C.C.; Vatish, M.; Nakajima, Y.; Shibusawa, R.; Ozawa, A.; Pessin, J.E.; Yamada, M. Fyn phosphorylates AMPK to inhibit AMPK activity and AMP-dependent activation of autophagy. Oncotarget 2016, 7, 74612–74629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, T.; Bridges, D.; Nakada, D.; Skiniotis, G.; Morrison, S.J.; Lin, J.D.; Saltiel, A.R.; Inoki, K. Inhibition of AMPK catabolic action by GSK3. Mol. Cell 2013, 50, 407–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woods, A.; Vertommen, D.; Neumann, D.; Turk, R.; Bayliss, J.; Schlattner, U.; Wallimann, T.; Carling, D.; Rider, M.H. Identification of phosphorylation sites in AMP-activated protein kinase (AMPK) for upstream AMPK kinases and study of their roles by site-directed mutagenesis. J. Biol. Chem. 2003, 278, 28434–28442. [Google Scholar] [CrossRef] [Green Version]
- Horman, S.; Vertommen, D.; Heath, R.; Neumann, D.; Mouton, V.; Woods, A.; Schlattner, U.; Wallimann, T.; Carling, D.; Hue, L.; et al. Insulin antagonizes ischemia-induced Thr172 phosphorylation of AMP-activated protein kinase alpha-subunits in heart via hierarchical phosphorylation of Ser485/491. J. Biol. Chem. 2006, 281, 5335–5340. [Google Scholar] [CrossRef] [Green Version]
- Kovacic, S.; Soltys, C.L.; Barr, A.J.; Shiojima, I.; Walsh, K.; Dyck, J.R. Akt activity negatively regulates phosphorylation of AMP-activated protein kinase in the heart. J. Biol. Chem. 2003, 278, 39422–39427. [Google Scholar] [CrossRef] [Green Version]
- Hawley, S.A.; Ross, F.A.; Gowans, G.J.; Tibarewal, P.; Leslie, N.R.; Hardie, D.G. Phosphorylation by Akt within the ST loop of AMPK-alpha1 down-regulates its activation in tumour cells. Biochem. J. 2014, 459, 275–287. [Google Scholar] [CrossRef] [Green Version]
- Hurley, R.L.; Barre, L.K.; Wood, S.D.; Anderson, K.A.; Kemp, B.E.; Means, A.R.; Witters, L.A. Regulation of AMP-activated protein kinase by multisite phosphorylation in response to agents that elevate cellular cAMP. J. Biol. Chem. 2006, 281, 36662–36672. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Cotarelo, P.; Escribano-Diaz, C.; Gonzalez-Bethencourt, I.L.; Gomez-Moreira, C.; Deguiz, M.L.; Torres-Bacete, J.; Gomez-Cabanas, L.; Fernandez-Barrera, J.; Delgado-Martin, C.; Mellado, M.; et al. A novel MEK-ERK-AMPK signaling axis controls chemokine receptor CCR7-dependent survival in human mature dendritic cells. J. Biol. Chem. 2015, 290, 827–840. [Google Scholar] [CrossRef] [Green Version]
- Park, D.W.; Jiang, S.; Liu, Y.; Siegal, G.P.; Inoki, K.; Abraham, E.; Zmijewski, J.W. GSK3beta-dependent inhibition of AMPK potentiates activation of neutrophils and macrophages and enhances severity of acute lung injury. Am. J. Physiol. Lung Cell Mol. Physiol. 2014, 307, L735–L745. [Google Scholar] [CrossRef] [Green Version]
- Dagon, Y.; Hur, E.; Zheng, B.; Wellenstein, K.; Cantley, L.C.; Kahn, B.B. p70S6 kinase phosphorylates AMPK on serine 491 to mediate leptin’s effect on food intake. Cell Metab. 2012, 16, 104–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coughlan, K.A.; Valentine, R.J.; Sudit, B.S.; Allen, K.; Dagon, Y.; Kahn, B.B.; Ruderman, N.B.; Saha, A.K. PKD1 Inhibits AMPKalpha2 through Phosphorylation of Serine 491 and Impairs Insulin Signaling in Skeletal Muscle Cells. J. Biol. Chem. 2016, 291, 5664–5675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchelhill, K.I.; Michell, B.J.; House, C.M.; Stapleton, D.; Dyck, J.; Gamble, J.; Ullrich, C.; Witters, L.A.; Kemp, B.E. Posttranslational modifications of the 5′-AMP-activated protein kinase beta1 subunit. J. Biol. Chem. 1997, 272, 24475–24479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warden, S.M.; Richardson, C.; O’Donnell, J., Jr.; Stapleton, D.; Kemp, B.E.; Witters, L.A. Post-translational modifications of the beta-1 subunit of AMP-activated protein kinase affect enzyme activity and cellular localization. Biochem. J. 2001, 354, 275–283. [Google Scholar] [CrossRef]
- Sanders, M.J.; Ali, Z.S.; Hegarty, B.D.; Heath, R.; Snowden, M.A.; Carling, D. Defining the mechanism of activation of AMP-activated protein kinase by the small molecule A-769662, a member of the thienopyridone family. J. Biol. Chem. 2007, 282, 32539–32548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, J.W.; Ling, N.; Issa, S.M.; Dite, T.A.; O’Brien, M.T.; Chen, Z.P.; Galic, S.; Langendorf, C.G.; Steinberg, G.R.; Kemp, B.E.; et al. Small molecule drug A-769662 and AMP synergistically activate naive AMPK independent of upstream kinase signaling. Chem. Biol. 2014, 21, 619–627. [Google Scholar] [CrossRef] [Green Version]
- Dite, T.A.; Ling, N.X.Y.; Scott, J.W.; Hoque, A.; Galic, S.; Parker, B.L.; Ngoei, K.R.W.; Langendorf, C.G.; O’Brien, M.T.; Kundu, M.; et al. The autophagy initiator ULK1 sensitizes AMPK to allosteric drugs. Nat. Commun. 2017, 8, 571. [Google Scholar] [CrossRef]
- Oligschlaeger, Y.; Miglianico, M.; Chanda, D.; Scholz, R.; Thali, R.F.; Tuerk, R.; Stapleton, D.I.; Gooley, P.R.; Neumann, D. The recruitment of AMP-activated protein kinase to glycogen is regulated by autophosphorylation. J. Biol. Chem. 2015, 290, 11715–11728. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Heierhorst, J.; Mann, R.J.; Mitchelhill, K.I.; Michell, B.J.; Witters, L.A.; Lynch, G.S.; Kemp, B.E.; Stapleton, D. Expression of the AMP-activated protein kinase beta1 and beta2 subunits in skeletal muscle. FEBS Lett. 1999, 460, 343–348. [Google Scholar] [CrossRef] [Green Version]
- Puustinen, P.; Keldsbo, A.; Corcelle-Termeau, E.; Ngoei, K.; Sonder, S.L.; Farkas, T.; Kaae Andersen, K.; Oakhill, J.S.; Jaattela, M. DNA-dependent protein kinase regulates lysosomal AMP-dependent protein kinase activation and autophagy. Autophagy 2020, 1–18. [Google Scholar] [CrossRef]
- Loffler, A.S.; Alers, S.; Dieterle, A.M.; Keppeler, H.; Franz-Wachtel, M.; Kundu, M.; Campbell, D.G.; Wesselborg, S.; Alessi, D.R.; Stork, B. Ulk1-mediated phosphorylation of AMPK constitutes a negative regulatory feedback loop. Autophagy 2011, 7, 696–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuerk, R.D.; Auchli, Y.; Thali, R.F.; Scholz, R.; Wallimann, T.; Brunisholz, R.A.; Neumann, D. Tracking and quantification of 32P-labeled phosphopeptides in liquid chromatography matrix-assisted laser desorption/ionization mass spectrometry. Anal. Biochem. 2009, 390, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Kornev, A.P.; Taylor, S.S. Defining the conserved internal architecture of a protein kinase. Biochim. Biophys. Acta 2010, 1804, 440–444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davies, S.P.; Helps, N.R.; Cohen, P.T.; Hardie, D.G. 5′-AMP inhibits dephosphorylation, as well as promoting phosphorylation, of the AMP-activated protein kinase. Studies using bacterially expressed human protein phosphatase-2C alpha and native bovine protein phosphatase-2AC. FEBS Lett. 1995, 377, 421–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oakhill, J.S.; Steel, R.; Chen, Z.P.; Scott, J.W.; Ling, N.; Tam, S.; Kemp, B.E. AMPK is a direct adenylate charge-regulated protein kinase. Science 2011, 332, 1433–1435. [Google Scholar] [CrossRef] [PubMed]
- Oakhill, J.S.; Chen, Z.P.; Scott, J.W.; Steel, R.; Castelli, L.A.; Ling, N.; Macaulay, S.L.; Kemp, B.E. beta-Subunit myristoylation is the gatekeeper for initiating metabolic stress sensing by AMP-activated protein kinase (AMPK). Proc. Natl. Acad. Sci. USA 2010, 107, 19237–19241. [Google Scholar] [CrossRef] [Green Version]
- Woods, A.; Johnstone, S.R.; Dickerson, K.; Leiper, F.C.; Fryer, L.G.; Neumann, D.; Schlattner, U.; Wallimann, T.; Carlson, M.; Carling, D. LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr. Biol. 2003, 13, 2004–2008. [Google Scholar] [CrossRef] [Green Version]
- Zoncu, R.; Bar-Peled, L.; Efeyan, A.; Wang, S.; Sancak, Y.; Sabatini, D.M. mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H(+)-ATPase. Science 2011, 334, 678–683. [Google Scholar] [CrossRef] [Green Version]
- Menon, S.; Dibble, C.C.; Talbott, G.; Hoxhaj, G.; Valvezan, A.J.; Takahashi, H.; Cantley, L.C.; Manning, B.D. Spatial control of the TSC complex integrates insulin and nutrient regulation of mTORC1 at the lysosome. Cell 2014, 156, 771–785. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.S.; Jiang, B.; Li, M.; Zhu, M.; Peng, Y.; Zhang, Y.L.; Wu, Y.Q.; Li, T.Y.; Liang, Y.; Lu, Z.; et al. The lysosomal v-ATPase-Ragulator complex is a common activator for AMPK and mTORC1, acting as a switch between catabolism and anabolism. Cell Metab. 2014, 20, 526–540. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.S.; Hawley, S.A.; Zong, Y.; Li, M.; Wang, Z.; Gray, A.; Ma, T.; Cui, J.; Feng, J.W.; Zhu, M.; et al. Fructose-1,6-bisphosphate and aldolase mediate glucose sensing by AMPK. Nature 2017, 548, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Woods, A.; Dickerson, K.; Heath, R.; Hong, S.P.; Momcilovic, M.; Johnstone, S.R.; Carlson, M.; Carling, D. Ca2+/calmodulin-dependent protein kinase kinase-beta acts upstream of AMP-activated protein kinase in mammalian cells. Cell Metab. 2005, 2, 21–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neumann, D. Is TAK1 a Direct Upstream Kinase of AMPK? Int. J. Mol. Sci. 2018, 19, 2412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, J.; Bissa, B.; Brecht, L.; Allers, L.; Choi, S.W.; Gu, Y.; Zbinden, M.; Burge, M.R.; Timmins, G.; Hallows, K.; et al. AMPK, a Regulator of Metabolism and Autophagy, Is Activated by Lysosomal Damage via a Novel Galectin-Directed Ubiquitin Signal Transduction System. Mol. Cell 2020, 77, 951–969.e9. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Jiang, Y.; Sun, J.; Geng, S.; Pan, Z.; Prinz, R.A.; Wang, C.; Sun, J.; Jiao, X.; Xu, X. Activation of TGF-beta-activated kinase 1 (TAK1) restricts Salmonella Typhimurium growth by inducing AMPK activation and autophagy. Cell Death Dis. 2018, 9, 570. [Google Scholar] [CrossRef]
- Corrado, M.; Mariotti, F.R.; Trapani, L.; Taraborrelli, L.; Nazio, F.; Cianfanelli, V.; Soriano, M.E.; Schrepfer, E.; Cecconi, F.; Scorrano, L.; et al. Macroautophagy inhibition maintains fragmented mitochondria to foster T cell receptor-dependent apoptosis. EMBO J. 2016, 35, 1793–1809. [Google Scholar] [CrossRef] [Green Version]
- Aw, D.K.; Sinha, R.A.; Xie, S.Y.; Yen, P.M. Differential AMPK phosphorylation by glucagon and metformin regulates insulin signaling in human hepatic cells. Biochem. Biophys. Res. Commun. 2014, 447, 569–573. [Google Scholar] [CrossRef]
- Voss, M.; Paterson, J.; Kelsall, I.R.; Martin-Granados, C.; Hastie, C.J.; Peggie, M.W.; Cohen, P.T. Ppm1E is an in cellulo AMP-activated protein kinase phosphatase. Cell Signal. 2011, 23, 114–124. [Google Scholar] [CrossRef]
- Willows, R.; Sanders, M.J.; Xiao, B.; Patel, B.R.; Martin, S.R.; Read, J.; Wilson, J.R.; Hubbard, J.; Gamblin, S.J.; Carling, D. Phosphorylation of AMPK by upstream kinases is required for activity in mammalian cells. Biochem. J. 2017, 474, 3059–3073. [Google Scholar] [CrossRef] [Green Version]
- Langendorf, C.G.; Ngoei, K.R.W.; Scott, J.W.; Ling, N.X.Y.; Issa, S.M.A.; Gorman, M.A.; Parker, M.W.; Sakamoto, K.; Oakhill, J.S.; Kemp, B.E. Structural basis of allosteric and synergistic activation of AMPK by furan-2-phosphonic derivative C2 binding. Nat. Commun. 2016, 7, 10912. [Google Scholar] [CrossRef]
- Dite, T.A.; Langendorf, C.G.; Hoque, A.; Galic, S.; Rebello, R.J.; Ovens, A.J.; Lindqvist, L.M.; Ngoei, K.R.W.; Ling, N.X.Y.; Furic, L.; et al. AMP-activated protein kinase selectively inhibited by the type II inhibitor SBI-0206965. J. Biol. Chem. 2018, 293, 8874–8885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vahsen, B.F.; Ribas, V.T.; Sundermeyer, J.; Boecker, A.; Dambeck, V.; Lenz, C.; Shomroni, O.; Caldi Gomes, L.; Tatenhorst, L.; Barski, E.; et al. Inhibition of the autophagic protein ULK1 attenuates axonal degeneration in vitro and in vivo, enhances translation, and modulates splicing. Cell Death Differ. 2020, 27, 2810–2827. [Google Scholar] [CrossRef] [PubMed]
- Pang, T.; Xiong, B.; Li, J.Y.; Qiu, B.Y.; Jin, G.Z.; Shen, J.K.; Li, J. Conserved alpha-helix acts as autoinhibitory sequence in AMP-activated protein kinase alpha subunits. J. Biol. Chem. 2007, 282, 495–506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Xin, F.J.; Wang, J.; Hu, J.; Zhang, Y.Y.; Wan, S.; Cao, L.S.; Lu, C.; Li, P.; Yan, S.F.; et al. Conserved regulatory elements in AMPK. Nature 2013, 498, E8–E10. [Google Scholar] [CrossRef]
- Xin, F.J.; Wang, J.; Zhao, R.Q.; Wang, Z.X.; Wu, J.W. Coordinated regulation of AMPK activity by multiple elements in the alpha-subunit. Cell Res. 2013, 23, 1237–1240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, A.; Krug, K.; Pengelley, S.; Macek, B.; Hauf, S. Mitotic substrates of the kinase aurora with roles in chromatin regulation identified through quantitative phosphoproteomics of fission yeast. Sci. Signal. 2011, 4, rs6. [Google Scholar] [CrossRef]
- Wilson-Grady, J.T.; Villen, J.; Gygi, S.P. Phosphoproteome analysis of fission yeast. J. Proteome Res. 2008, 7, 1088–1097. [Google Scholar] [CrossRef]
- Wissing, J.; Jansch, L.; Nimtz, M.; Dieterich, G.; Hornberger, R.; Keri, G.; Wehland, J.; Daub, H. Proteomics analysis of protein kinases by target class-selective prefractionation and tandem mass spectrometry. Mol. Cell Proteom. 2007, 6, 537–547. [Google Scholar] [CrossRef] [Green Version]
- Gwinn, D.M.; Shackelford, D.B.; Egan, D.F.; Mihaylova, M.M.; Mery, A.; Vasquez, D.S.; Turk, B.E.; Shaw, R.J. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol. Cell 2008, 30, 214–226. [Google Scholar] [CrossRef] [Green Version]
- Inoki, K.; Zhu, T.; Guan, K.L. TSC2 mediates cellular energy response to control cell growth and survival. Cell 2003, 115, 577–590. [Google Scholar] [CrossRef] [Green Version]
- Dreyer, H.C.; Fujita, S.; Cadenas, J.G.; Chinkes, D.L.; Volpi, E.; Rasmussen, B.B. Resistance exercise increases AMPK activity and reduces 4E-BP1 phosphorylation and protein synthesis in human skeletal muscle. J. Physiol. 2006, 576, 613–624. [Google Scholar] [CrossRef] [PubMed]
- Apro, W.; Wang, L.; Ponten, M.; Blomstrand, E.; Sahlin, K. Resistance exercise induced mTORC1 signaling is not impaired by subsequent endurance exercise in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E22–E32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drummond, M.J.; Fry, C.S.; Glynn, E.L.; Dreyer, H.C.; Dhanani, S.; Timmerman, K.L.; Volpi, E.; Rasmussen, B.B. Rapamycin administration in humans blocks the contraction-induced increase in skeletal muscle protein synthesis. J. Physiol. 2009, 587, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, R.; Fujita, S.; Hornberger, T.A.; Kitaoka, Y.; Makanae, Y.; Nakazato, K.; Naokata, I. The role of mTOR signalling in the regulation of skeletal muscle mass in a rodent model of resistance exercise. Sci. Rep. 2016, 6, 31142. [Google Scholar] [CrossRef]
- Knudsen, J.R.; Li, Z.; Persson, K.W.; Li, J.; Henriquez-Olguin, C.; Jensen, T.E. Contraction-regulated mTORC1 and protein synthesis: Influence of AMPK and glycogen. J. Physiol. 2020, 598, 2637–2649. [Google Scholar] [CrossRef]
- Zong, Y.; Zhang, C.S.; Li, M.; Wang, W.; Wang, Z.; Hawley, S.A.; Ma, T.; Feng, J.W.; Tian, X.; Qi, Q.; et al. Hierarchical activation of compartmentalized pools of AMPK depends on severity of nutrient or energy stress. Cell Res. 2019, 29, 460–473. [Google Scholar] [CrossRef] [Green Version]
- Yoon, I.; Nam, M.; Kim, H.K.; Moon, H.S.; Kim, S.; Jang, J.; Song, J.A.; Jeong, S.J.; Kim, S.B.; Cho, S.; et al. Glucose-dependent control of leucine metabolism by leucyl-tRNA synthetase 1. Science 2020, 367, 205–210. [Google Scholar] [CrossRef]
- Mounier, R.; Lantier, L.; Leclerc, J.; Sotiropoulos, A.; Pende, M.; Daegelen, D.; Sakamoto, K.; Foretz, M.; Viollet, B. Important role for AMPKalpha1 in limiting skeletal muscle cell hypertrophy. FASEB J. 2009, 23, 2264–2273. [Google Scholar] [CrossRef]
- Oakhill, J.S.; Scott, J.W.; Kemp, B.E. Structure and function of AMP-activated protein kinase. Acta Physiol. 2009, 196, 3–14. [Google Scholar] [CrossRef]
- Cohen, P.; Frame, S. The renaissance of GSK3. Nat. Rev. Mol. Cell Biol. 2001, 2, 769–776. [Google Scholar] [CrossRef]
- Frame, S.; Cohen, P.; Biondi, R.M. A common phosphate binding site explains the unique substrate specificity of GSK3 and its inactivation by phosphorylation. Mol. Cell 2001, 7, 1321–1327. [Google Scholar] [CrossRef]
- Soltys, C.L.; Kovacic, S.; Dyck, J.R. Activation of cardiac AMP-activated protein kinase by LKB1 expression or chemical hypoxia is blunted by increased Akt activity. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H2472–H2479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heathcote, H.R.; Mancini, S.J.; Strembitska, A.; Jamal, K.; Reihill, J.A.; Palmer, T.M.; Gould, G.W.; Salt, I.P. Protein kinase C phosphorylates AMP-activated protein kinase alpha1 Ser487. Biochem. J. 2016, 473, 4681–4697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, J.W.; Galic, S.; Graham, K.L.; Foitzik, R.; Ling, N.X.; Dite, T.A.; Issa, S.M.; Langendorf, C.G.; Weng, Q.P.; Thomas, H.E.; et al. Inhibition of AMP-Activated Protein Kinase at the Allosteric Drug-Binding Site Promotes Islet Insulin Release. Chem. Biol. 2015, 22, 705–711. [Google Scholar] [CrossRef] [Green Version]
- Langendorf, C.G.; Kemp, B.E. Choreography of AMPK activation. Cell Res. 2015, 25, 5–6. [Google Scholar] [CrossRef]
- Pinkosky, S.L.; Scott, J.W.; Desjardins, E.M.; Smith, B.K.; Day, E.A.; Ford, R.J.; Langendorf, C.G.; Ling, N.X.Y.; Nero, T.L.; Loh, K.; et al. Long-chain fatty acyl-CoA esters regulate metabolism via allosteric control of AMPK beta1 isoforms. Nat. Metab. 2020, 2, 873–881. [Google Scholar] [CrossRef]
- Scott, J.W.; van Denderen, B.J.; Jorgensen, S.B.; Honeyman, J.E.; Steinberg, G.R.; Oakhill, J.S.; Iseli, T.J.; Koay, A.; Gooley, P.R.; Stapleton, D.; et al. Thienopyridone drugs are selective activators of AMP-activated protein kinase beta1-containing complexes. Chem. Biol. 2008, 15, 1220–1230. [Google Scholar] [CrossRef] [Green Version]
- Hoffman, N.J.; Whitfield, J.; Janzen, N.R.; Belhaj, M.R.; Galic, S.; Murray-Segal, L.; Smiles, W.J.; Ling, N.X.Y.; Dite, T.A.; Scott, J.W.; et al. Genetic loss of AMPK-glycogen binding destabilises AMPK and disrupts metabolism. Mol. Metab. 2020, 41, 101048. [Google Scholar] [CrossRef]
- Iseli, T.J.; Oakhill, J.S.; Bailey, M.F.; Wee, S.; Walter, M.; van Denderen, B.J.; Castelli, L.A.; Katsis, F.; Witters, L.A.; Stapleton, D.; et al. AMP-activated protein kinase subunit interactions: Beta1:gamma1 association requires beta1 Thr-263 and Tyr-267. J. Biol. Chem. 2008, 283, 4799–4807. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.S.; Han, H.J.; Han, S.Y.; Kim, I.Y.; Chae, S.; Lee, C.S.; Kim, S.E.; Yoon, S.G.; Park, J.W.; Kim, J.H.; et al. Loss of the E3 ubiquitin ligase MKRN1 represses diet-induced metabolic syndrome through AMPK activation. Nat. Commun. 2018, 9, 3404. [Google Scholar] [CrossRef]
- Deng, M.; Yang, X.; Qin, B.; Liu, T.; Zhang, H.; Guo, W.; Lee, S.B.; Kim, J.J.; Yuan, J.; Pei, H.; et al. Deubiquitination and Activation of AMPK by USP10. Mol. Cell 2016, 61, 614–624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, E.; Li, X.; Deng, Y.; Chang, H.W.; Kim, D.Y. AMPK is down-regulated by the CRL4A-CRBN axis through the polyubiquitination of AMPKalpha isoforms. FASEB J. 2019, 33, 6539–6550. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ding, J.; Kohnlein, K.; Urban, N.; Ori, A.; Villavicencio-Lorini, P.; Walentek, P.; Klotz, L.O.; Hollemann, T.; Pfirrmann, T. The GID ubiquitin ligase complex is a regulator of AMPK activity and organismal lifespan. Autophagy 2019, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, F.; Zhang, H.; Yuan, Y.; Liu, Z.; Huang, B.; Miao, H.; Liu, X.; Zhao, Q.; Zhang, H.; Dong, H.; et al. A decrease of ATP production steered by PEDF in cardiomyocytes with oxygen-glucose deprivation is associated with an AMPK-dependent degradation pathway. Int. J. Cardiol. 2018, 257, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Giri, S. Loss of AMP-activated protein kinase in X-linked adrenoleukodystrophy patient-derived fibroblasts and lymphocytes. Biochem. Biophys. Res. Commun. 2014, 445, 126–131. [Google Scholar] [CrossRef]
- Pineda, C.T.; Ramanathan, S.; Fon Tacer, K.; Weon, J.L.; Potts, M.B.; Ou, Y.H.; White, M.A.; Potts, P.R. Degradation of AMPK by a cancer-specific ubiquitin ligase. Cell 2015, 160, 715–728. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.Y.; Wu, C.; Shah, S.S.; Chen, S.M.; Wangpaichitr, M.; Kuo, M.T.; Feun, L.G.; Han, X.; Suarez, M.; Prince, J.; et al. Degradation of AMPK-alpha1 sensitizes BRAF inhibitor-resistant melanoma cells to arginine deprivation. Mol. Oncol. 2017, 11, 1806–1825. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Huang, Z.; Zhu, L.; Yu, X.; Gao, T.; Feng, J.; Hong, H.; Yin, H.; Zhou, T.; Qi, W.; et al. The contrary intracellular and extracellular functions of PEDF in HCC development. Cell Death Dis. 2019, 10, 742. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Zhang, R.Y.; Song, J.; Guan, Y.F.; Xu, T.Y.; Du, H.; Viollet, B.; Miao, C.Y. Loss of AMP-activated protein kinase-alpha2 impairs the insulin-sensitizing effect of calorie restriction in skeletal muscle. Diabetes 2012, 61, 1051–1061. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.O.; Lee, S.K.; Kim, N.; Kim, J.H.; You, G.Y.; Moon, J.W.; Jie, S.; Kim, S.J.; Lee, Y.W.; Kang, H.J.; et al. E3 ubiquitin ligase, WWP1, interacts with AMPKalpha2 and down-regulates its expression in skeletal muscle C2C12 cells. J. Biol. Chem. 2013, 288, 4673–4680. [Google Scholar] [CrossRef] [Green Version]
- Vila, I.K.; Yao, Y.; Kim, G.; Xia, W.; Kim, H.; Kim, S.J.; Park, M.K.; Hwang, J.P.; Gonzalez-Billalabeitia, E.; Hung, M.C.; et al. A UBE2O-AMPKalpha2 Axis that Promotes Tumor Initiation and Progression Offers Opportunities for Therapy. Cancer Cell 2017, 31, 208–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, J.; Gong, J.; Zhao, T.; Zhao, J.; Lam, P.; Ye, J.; Li, J.Z.; Wu, J.; Zhou, H.M.; Li, P. Downregulation of AMP-activated protein kinase by Cidea-mediated ubiquitination and degradation in brown adipose tissue. EMBO J. 2008, 27, 1537–1548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, M.; Zhang, D.; Zhao, Z.; Sit, J.; Saint-Sume, M.; Shabandri, O.; Zhang, K.; Yin, L.; Tong, X. Hepatic E4BP4 induction promotes lipid accumulation by suppressing AMPK signaling in response to chemical or diet-induced ER stress. FASEB J. 2020. [Google Scholar] [CrossRef]
- Moreno, D.; Towler, M.C.; Hardie, D.G.; Knecht, E.; Sanz, P. The laforin-malin complex, involved in Lafora disease, promotes the incorporation of K63-linked ubiquitin chains into AMP-activated protein kinase beta subunits. Mol. Biol. Cell 2010, 21, 2578–2588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.M.; Jo, S.; Kim, H.; Lee, J.; Park, C.S. Functional modulation of AMP-activated protein kinase by cereblon. Biochim. Biophys. Acta 2011, 1813, 448–455. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.M.; Yang, S.J.; Kim, Y.D.; Choi, Y.D.; Nam, J.H.; Choi, C.S.; Choi, H.S.; Park, C.S. Disruption of the cereblon gene enhances hepatic AMPK activity and prevents high-fat diet-induced obesity and insulin resistance in mice. Diabetes 2013, 62, 1855–1864. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.J.; Jeon, S.J.; Van Nguyen, T.; Deshaies, R.J.; Park, C.S.; Lee, K.M. Ubiquitin-dependent proteasomal degradation of AMPK gamma subunit by Cereblon inhibits AMPK activity. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118729. [Google Scholar] [CrossRef]
- Yan, Y.; Ollila, S.; Wong, I.P.L.; Vallenius, T.; Palvimo, J.J.; Vaahtomeri, K.; Makela, T.P. SUMOylation of AMPKalpha1 by PIAS4 specifically regulates mTORC1 signalling. Nat. Commun. 2015, 6, 8979. [Google Scholar] [CrossRef]
- Rubio, T.; Vernia, S.; Sanz, P. Sumoylation of AMPKbeta2 subunit enhances AMP-activated protein kinase activity. Mol. Biol. Cell 2013, 24, 1801–1811. [Google Scholar] [CrossRef] [Green Version]
- Choudhary, C.; Kumar, C.; Gnad, F.; Nielsen, M.L.; Rehman, M.; Walther, T.C.; Olsen, J.V.; Mann, M. Lysine acetylation targets protein complexes and co-regulates major cellular functions. Science 2009, 325, 834–840. [Google Scholar] [CrossRef] [Green Version]
- Larsen, S.C.; Sylvestersen, K.B.; Mund, A.; Lyon, D.; Mullari, M.; Madsen, M.V.; Daniel, J.A.; Jensen, L.J.; Nielsen, M.L. Proteome-wide analysis of arginine monomethylation reveals widespread occurrence in human cells. Sci. Signal. 2016, 9, rs9. [Google Scholar] [CrossRef]
- Olsen, J.B.; Cao, X.J.; Han, B.; Chen, L.H.; Horvath, A.; Richardson, T.I.; Campbell, R.M.; Garcia, B.A.; Nguyen, H. Quantitative Profiling of the Activity of Protein Lysine Methyltransferase SMYD2 Using SILAC-Based Proteomics. Mol. Cell Proteom. 2016, 15, 892–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, C.I.; Uhrigshardt, H.; O’Meally, R.N.; Cole, R.N.; Van Eyk, J.E. Identification and quantification of S-nitrosylation by cysteine reactive tandem mass tag switch assay. Mol. Cell Proteom. 2012, 11, M111.013441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, H.S.; Murray, C.I.; Venkatraman, V.; Crowgey, E.L.; Rainer, P.P.; Cole, R.N.; Bomgarden, R.D.; Rogers, J.C.; Balkan, W.; Hare, J.M.; et al. Dual Labeling Biotin Switch Assay to Reduce Bias Derived From Different Cysteine Subpopulations: A Method to Maximize S-Nitrosylation Detection. Circ. Res. 2015, 117, 846–857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zmijewski, J.W.; Banerjee, S.; Bae, H.; Friggeri, A.; Lazarowski, E.R.; Abraham, E. Exposure to hydrogen peroxide induces oxidation and activation of AMP-activated protein kinase. J. Biol. Chem. 2010, 285, 33154–33164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, D.; Oka, S.; Liu, T.; Zhai, P.; Ago, T.; Sciarretta, S.; Li, H.; Sadoshima, J. A redox-dependent mechanism for regulation of AMPK activation by Thioredoxin1 during energy starvation. Cell Metab. 2014, 19, 232–245. [Google Scholar] [CrossRef] [Green Version]
- Rennie, M.L.; Chaugule, V.K.; Walden, H. Modes of allosteric regulation of the ubiquitination machinery. Curr. Opin. Struct. Biol. 2020, 62, 189–196. [Google Scholar] [CrossRef]
- Al-Hakim, A.K.; Zagorska, A.; Chapman, L.; Deak, M.; Peggie, M.; Alessi, D.R. Control of AMPK-related kinases by USP9X and atypical Lys(29)/Lys(33)-linked polyubiquitin chains. Biochem. J. 2008, 411, 249–260. [Google Scholar] [CrossRef] [Green Version]
- Wilson, M.A.; Koutelou, E.; Hirsch, C.; Akdemir, K.; Schibler, A.; Barton, M.C.; Dent, S.Y. Ubp8 and SAGA regulate Snf1 AMP kinase activity. Mol. Cell Biol. 2011, 31, 3126–3135. [Google Scholar] [CrossRef] [Green Version]
- Onodera, W.; Asahi, T.; Sawamura, N. Positive selection of cereblon modified function including its E3 ubiquitin ligase activity and binding efficiency with AMPK. Mol. Phylogenet. Evol. 2019, 135, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Sawamura, N.; Yamada, M.; Fujiwara, M.; Yamada, H.; Hayashi, H.; Takagi, N.; Asahi, T. The Neuroprotective Effect of Thalidomide against Ischemia through the Cereblon-mediated Repression of AMPK Activity. Sci. Rep. 2018, 8, 2459. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, K.M.; Park, C.S.; Park, W.J. Ablation of cereblon attenuates myocardial ischemia-reperfusion injury. Biochem. Biophys. Res. Commun. 2014, 447, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.R.; Seo, D.Y.; Park, S.H.; Kwak, H.B.; Kim, M.; Ko, K.S.; Rhee, B.D.; Han, J. Aerobic exercise training decreases cereblon and increases AMPK signaling in the skeletal muscle of STZ-induced diabetic rats. Biochem. Biophys. Res. Commun. 2018, 501, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Bavley, C.C.; Rice, R.C.; Fischer, D.K.; Fakira, A.K.; Byrne, M.; Kosovsky, M.; Rizzo, B.K.; Del Prete, D.; Alaedini, A.; Moron, J.A.; et al. Rescue of Learning and Memory Deficits in the Human Nonsyndromic Intellectual Disability Cereblon Knock-Out Mouse Model by Targeting the AMP-Activated Protein Kinase-mTORC1 Translational Pathway. J. Neurosci. 2018, 38, 2780–2795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vila, I.K.; Park, M.K.; Setijono, S.R.; Yao, Y.; Kim, H.; Badin, P.M.; Choi, S.; Narkar, V.; Choi, S.W.; Chung, J.; et al. A muscle-specific UBE2O/AMPKalpha2 axis promotes insulin resistance and metabolic syndrome in obesity. JCI Insight 2019, 4. [Google Scholar] [CrossRef]
- Zhou, Z.; Yon Toh, S.; Chen, Z.; Guo, K.; Ng, C.P.; Ponniah, S.; Lin, S.C.; Hong, W.; Li, P. Cidea-deficient mice have lean phenotype and are resistant to obesity. Nat. Genet. 2003, 35, 49–56. [Google Scholar] [CrossRef]
- Simpson-Lavy, K.J.; Johnston, M. SUMOylation regulates the SNF1 protein kinase. Proc. Natl. Acad. Sci. USA 2013, 110, 17432–17437. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.Y.; Lin, Y.Y.; Sheu, J.C.; Wu, J.T.; Lee, F.J.; Chen, Y.; Lin, M.I.; Chiang, F.T.; Tai, T.Y.; Berger, S.L.; et al. Acetylation of yeast AMPK controls intrinsic aging independently of caloric restriction. Cell 2011, 146, 969–979. [Google Scholar] [CrossRef] [Green Version]
- Kusuyama, J.; Alves-Wagner, A.B.; Makarewicz, N.S.; Goodyear, L.J. Effects of maternal and paternal exercise on offspring metabolism. Nat. Metab. 2020, 2, 858–872. [Google Scholar] [CrossRef]
- McGee, S.L.; Walder, K.R. Exercise and the Skeletal Muscle Epigenome. Cold Spring Harb. Perspect. Med. 2017, 7. [Google Scholar] [CrossRef] [Green Version]
- Ren, Y.; Shen, H.M. Critical role of AMPK in redox regulation under glucose starvation. Redox Biol. 2019, 25, 101154. [Google Scholar] [CrossRef] [PubMed]
- Byrne, D.P.; Shrestha, S.; Galler, M.; Cao, M.; Daly, L.A.; Campbell, A.E.; Eyers, C.E.; Veal, E.A.; Kannan, N.; Eyers, P.A. Aurora A regulation by reversible cysteine oxidation reveals evolutionarily conserved redox control of Ser/Thr protein kinase activity. Sci. Signal. 2020, 13. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Lee, M.; Jung, S.N.; Lee, H.J.; Kang, I.; Kim, S.S.; Ha, J. AMP-activated protein kinase activity is required for vanadate-induced hypoxia-inducible factor 1alpha expression in DU145 cells. Carcinogenesis 2004, 25, 2497–2507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hinchy, E.C.; Gruszczyk, A.V.; Willows, R.; Navaratnam, N.; Hall, A.R.; Bates, G.; Bright, T.P.; Krieg, T.; Carling, D.; Murphy, M.P. Mitochondria-derived ROS activate AMP-activated protein kinase (AMPK) indirectly. J. Biol. Chem. 2018, 293, 17208–17217. [Google Scholar] [CrossRef] [Green Version]
- Emerling, B.M.; Weinberg, F.; Snyder, C.; Burgess, Z.; Mutlu, G.M.; Viollet, B.; Budinger, G.R.; Chandel, N.S. Hypoxic activation of AMPK is dependent on mitochondrial ROS but independent of an increase in AMP/ATP ratio. Free Radic. Biol. Med. 2009, 46, 1386–1391. [Google Scholar] [CrossRef] [Green Version]
- Mungai, P.T.; Waypa, G.B.; Jairaman, A.; Prakriya, M.; Dokic, D.; Ball, M.K.; Schumacker, P.T. Hypoxia triggers AMPK activation through reactive oxygen species-mediated activation of calcium release-activated calcium channels. Mol. Cell Biol. 2011, 31, 3531–3545. [Google Scholar] [CrossRef] [Green Version]
- Altarejos, J.Y.; Taniguchi, M.; Clanachan, A.S.; Lopaschuk, G.D. Myocardial ischemia differentially regulates LKB1 and an alternate 5′-AMP-activated protein kinase kinase. J. Biol. Chem. 2005, 280, 183–190. [Google Scholar] [CrossRef] [Green Version]
- Toyoda, T.; Hayashi, T.; Miyamoto, L.; Yonemitsu, S.; Nakano, M.; Tanaka, S.; Ebihara, K.; Masuzaki, H.; Hosoda, K.; Inoue, G.; et al. Possible involvement of the alpha1 isoform of 5′-AMP-activated protein kinase in oxidative stress-stimulated glucose transport in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E166–E173. [Google Scholar] [CrossRef] [Green Version]
- Quintero, M.; Colombo, S.L.; Godfrey, A.; Moncada, S. Mitochondria as signaling organelles in the vascular endothelium. Proc. Natl. Acad. Sci. USA 2006, 103, 5379–5384. [Google Scholar] [CrossRef] [Green Version]
- Evans, A.M.; Mustard, K.J.; Wyatt, C.N.; Peers, C.; Dipp, M.; Kumar, P.; Kinnear, N.P.; Hardie, D.G. Does AMP-activated protein kinase couple inhibition of mitochondrial oxidative phosphorylation by hypoxia to calcium signaling in O2-sensing cells? J. Biol. Chem. 2005, 280, 41504–41511. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.L.; Kim, S.J.; Lee, K.T.; Kim, J.; Mu, J.; Birnbaum, M.J.; Soo Kim, S.; Ha, J. The regulation of AMP-activated protein kinase by H(2)O(2). Biochem. Biophys. Res. Commun. 2001, 287, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Auciello, F.R.; Ross, F.A.; Ikematsu, N.; Hardie, D.G. Oxidative stress activates AMPK in cultured cells primarily by increasing cellular AMP and/or ADP. FEBS Lett. 2014, 588, 3361–3366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hawley, S.A.; Ross, F.A.; Chevtzoff, C.; Green, K.A.; Evans, A.; Fogarty, S.; Towler, M.C.; Brown, L.J.; Ogunbayo, O.A.; Evans, A.M.; et al. Use of cells expressing gamma subunit variants to identify diverse mechanisms of AMPK activation. Cell Metab. 2010, 11, 554–565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, H.; Jedrychowski, M.P.; Schweppe, D.K.; Huttlin, E.L.; Yu, Q.; Heppner, D.E.; Li, J.; Long, J.; Mills, E.L.; Szpyt, J.; et al. A Quantitative Tissue-Specific Landscape of Protein Redox Regulation during Aging. Cell 2020, 180, 968–983.e24. [Google Scholar] [CrossRef] [PubMed]
- Humphries, K.M.; Deal, M.S.; Taylor, S.S. Enhanced dephosphorylation of cAMP-dependent protein kinase by oxidation and thiol modification. J. Biol. Chem. 2005, 280, 2750–2758. [Google Scholar] [CrossRef] [Green Version]
- Chegary, M.; Brinke, H.; Ruiter, J.P.; Wijburg, F.A.; Stoll, M.S.; Minkler, P.E.; van Weeghel, M.; Schulz, H.; Hoppel, C.L.; Wanders, R.J.; et al. Mitochondrial long chain fatty acid beta-oxidation in man and mouse. Biochim. Biophys. Acta 2009, 1791, 806–815. [Google Scholar] [CrossRef] [Green Version]
- Ross, F.A.; Jensen, T.E.; Hardie, D.G. Differential regulation by AMP and ADP of AMPK complexes containing different gamma subunit isoforms. Biochem. J. 2016, 473, 189–199. [Google Scholar] [CrossRef] [Green Version]
- Roskoski, R., Jr. Targeting ERK1/2 protein-serine/threonine kinases in human cancers. Pharmacol. Res. 2019, 142, 151–168. [Google Scholar] [CrossRef] [PubMed]
- Pelech, S.L. Networking with proline-directed protein kinases implicated in tau phosphorylation. Neurobiol. Aging 1995, 16, 247–256, discussion 257–261. [Google Scholar] [CrossRef]
- Cao, Y.; Bojjireddy, N.; Kim, M.; Li, T.; Zhai, P.; Nagarajan, N.; Sadoshima, J.; Palmiter, R.D.; Tian, R. Activation of gamma2-AMPK Suppresses Ribosome Biogenesis and Protects Against Myocardial Ischemia/Reperfusion Injury. Circ. Res. 2017, 121, 1182–1191. [Google Scholar] [CrossRef]
- Mayer, C.; Zhao, J.; Yuan, X.; Grummt, I. mTOR-dependent activation of the transcription factor TIF-IA links rRNA synthesis to nutrient availability. Genes Dev. 2004, 18, 423–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
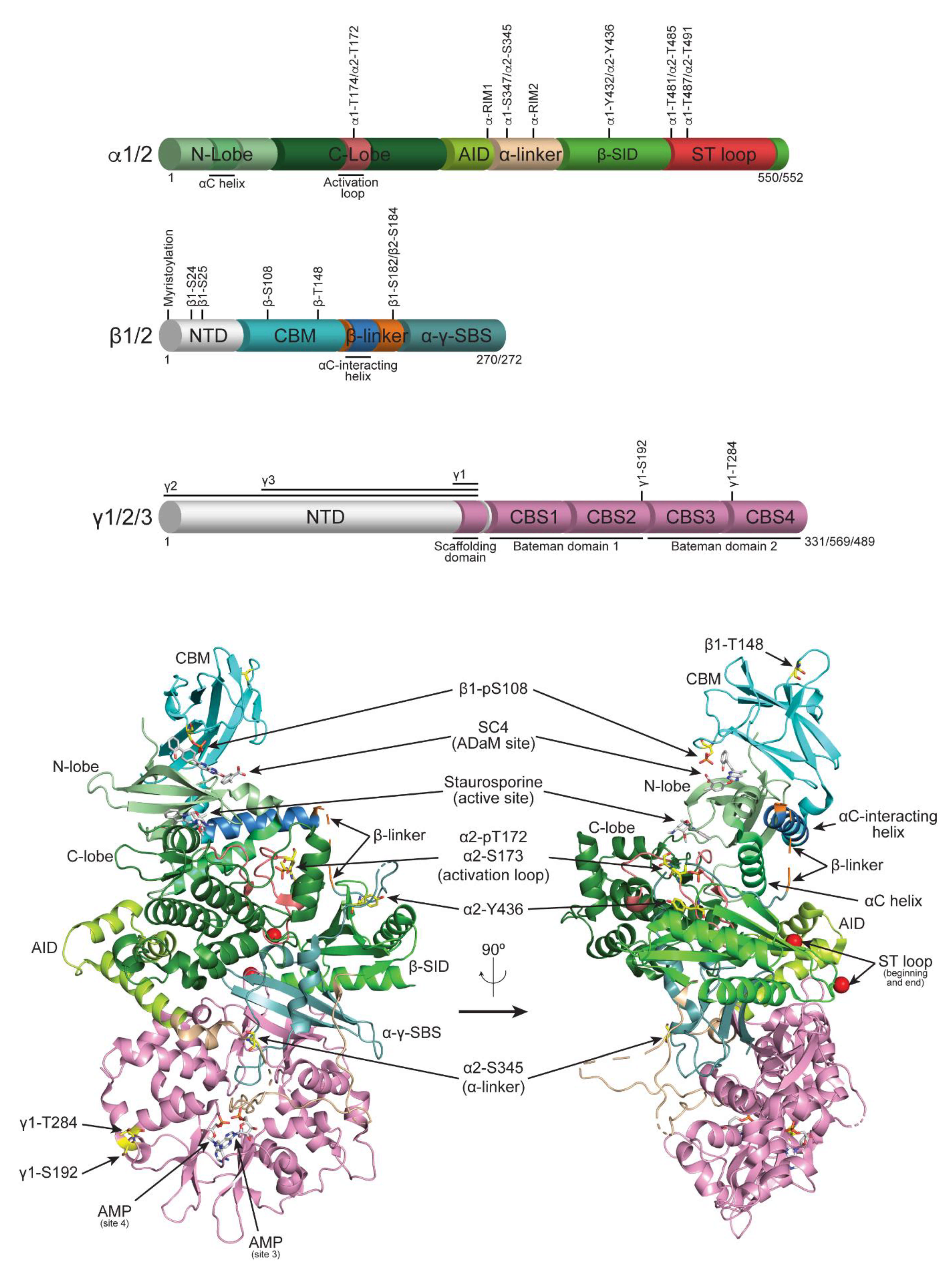
| Isoform/Residue | Location | Regulatory Function | Notes | Kinase | Primary Ref. |
|---|---|---|---|---|---|
| α-subunit Uniprot IDs α1: Q13131-1 (numbering starts from initiation Met10, most commonly used in the field); α2: P54646-1 | |||||
| α1-T174/α2-T172 [30] | Activation loop | Activatory | Fully validated upstream kinases | LKB1 | [31,32] |
| CaMKK2 | [33] [34] | ||||
| Less well validated upstream kinases | TAK1 | [35,36] | |||
| MLK3 | [37] | ||||
| CaMKK1 | [34] | ||||
| VRK1 | [38] | ||||
| α1-S175/α2-S173 | Activation loop | Inhibitory | Suppresses T172 phosphorylation | PKA | [39] |
| α1-S347/α2-S345 | α-linker | Inhibitory | Suppresses T172 phosphorylation | CDK4 | [40] |
| mTORC1 | [41] | ||||
| α1-Y432/α2-Y436 | β-SID | Inhibitory | Fyn | [42] | |
| α1-T481/α2-T485 | ST loop | Inhibitory | Promotes T172 dephosphorylation | GSK3α/β | [43] |
| CDK4 (only α2-T485?) | [40] | ||||
| α1-S487/α2-S491 [44] | ST loop | Inhibitory | Suppresses T172 phosphorylation | autophos. (α2-S491 more than α1-S487) | [45] |
| Akt/PKB (α1-S487 more than α2-S491) | [46] [45] [47] | ||||
| PKA (only α1-S487) | [39,48] | ||||
| PKC (only α1-S487?) | [44] | ||||
| ERK1/2 | [49] | ||||
| IKKβ | [50] | ||||
| p70S6K | [51] | ||||
| PKD1 (only α2-S491?) | [52] | ||||
| β-subunit Uniprot IDs β1: Q9Y478-1; β2: O43741-1 | |||||
| β1-S24/25 [53] | NH2-terminus | Localisation | S24/25A promotes nuclear transport [54] | Akt/PKB | [39] |
| β1/2-S108 [53] | CBM | Activatory | Stabilises ADaM site and sensitises AMPK to ligands [13,55,56]; β1-S108A reduced basal activity [54] | cis-autophos. | [53] |
| ULK1 (β1-S108 only) | [57] | ||||
| β1/2-T148 | CBM | Localisation | T148D destabilises glycogen complex | autophos. | [58] |
| β1-S182/ β2-S184 [53] | β-linker | Localisation | β1-S182A promotes nuclear transport; skeletal muscle: β1-pS182 stoichiometric; β2-pS184 substoichiometric, 41 HTPs | [53,54,59] | |
| γ-subunit Uniprot ID γ1: P54619-1 | |||||
| γ1-S192 | CBS2-3 linker | Activatory | γ1-S192A (with γ1-T284A) attenuates lysosome transport | DNA-PK | [60] |
| γ1-T284 | CBS4 | Activatory | γ1-T284A (with γ1-S192A) attenuates lysosome transport | DNA-PK | [60] |
| Isoform/Residue | Location | Notes | Kinase | Primary Ref. |
|---|---|---|---|---|
| α-subunit Uniprot IDs α1: Q13131-1 (numbering starts from initiation Met10, most commonly used in the field); α2: P54646-1 | ||||
| α1-S178/α2-S176 | Activation loop | 18 HTPs | [29] | |
| α1-T260 | Kinase domain C-lobe | In vitro phosphorylation | autophos. | [44] |
| α1-S351/T359 | α-linker | In vitro phosphorylation | ULK1 | [61] |
| α1-T373 | α-linker | 30 HTPs | [29] | |
| α2-S377 | α-linker | 28 HTPs; in vitro phosphorylation | CDK4 | [29,40] |
| α1-T379 | α-linker | In vitro phosphorylation | autophos. | [62] |
| α1-S388 | α-linker | In vitro phosphorylation | ULK1 | [61] |
| α1-S473 | ST loop | In vitro phosphorylation | GSK3α/β | [43] |
| α1-S477 | ST loop | In vitro phosphorylation | GSK3α/β | [43] |
| ULK1 | [61] | |||
| α1-T479 | ST loop | In vitro phosphorylation | ULK1 | [61] |
| α1-S497 | ST loop | In vitro phosphorylation | PKA | [39] |
| α1-S499 | ST loop | 27 HTPs ; in vitro phosphorylation | PKA | [29,39] |
| α2-S529 | ST loop | In vitro phosphorylation | CDK4 | [40] |
| β-subunit Uniprot IDs β1: Q9Y478-1; β2: O43741-1 | ||||
| β2-S39 | NH2-terminus | In vitro phosphorylation; 22 HTPs | ULK1 | [61] |
| β2-T40 | NH2-terminus | In vitro phosphorylation | ULK1 | [61] |
| β1-S49 | NH2-terminus | In vitro phosphorylation | ULK1 | [57] |
| β2-S69 | CBM | In vitro phosphorylation | ULK1 | [61] |
| β1-T80 | CBM | In vitro phosphorylation | autophos. | [62] |
| β1-S96 | CBM | In vitro phosphorylation | autophos. | [44] |
| ULK1 | [57] | |||
| β1-S101 | CBM | In vitro phosphorylation | autophos. | [44] |
| β1-T158 | CBM | In vitro phosphorylation | autophos. | [62] |
| β1-S170 | αC-interacting helix | In vitro phosphorylation | ULK1 | [57] |
| β1/2-S174 | αC-interacting helix | In vitro phosphorylation | autophos. (β1-S174) | [62] |
| ULK1 (β2-S174) | [61] | |||
| β1-S177 | β-linker | In vitro phosphorylation | autophos. | [62] |
| γ-subunit Uniprot IDs γ1: P54619-1; γ2: Q9UGJ0-1 | ||||
| γ1-S261/T263 | CBS3 | In vitro phosphorylation | ULK1 | [57,61] |
| γ1-S270 | CBS3-4 linker | In vitro phosphorylation | ULK1 | [57,61] |
| γ1-Y272 | CBS3-4 linker | 65 HTPs | [29] | |
| γ2-S65 | NH2-terminus | 33 HTPs | [29] | |
| γ2-S71 | NH2-terminus | 25 HTPs | [29] | |
| γ2-S122 | NH2-terminus | 16 HTPs | [29] | |
| γ2-S196 | NH2-terminus | 25 HTPs | [29] | |
| Isoform/Residue | Location | Notes | Modifying Enzyme | Primary Ref. |
|---|---|---|---|---|
| Ubiquitination | ||||
| α1/α2 | Systemic metabolism | MKRN1 | [110] | |
| α1-K62/276/388/476 α2-K60/379/391/470 | K63-linked deubiquitination promotes AMPK activity | USP10 | [111] | |
| α1/α2 | Increased allergic mediator secretion from BMMCs | CRL4A, enhanced by CRBN | [112] | |
| α1/α2 | Suppression of autophagy during prolonged starvation | GID ubiquitin ligase complex | [113] | |
| α1/α2 | Mediated by PEDF/PEDFR/PPARγ axis | [114] | ||
| α1 | Reduced AMPK expression and bioenergetic profile in ALD fibroblasts and lymphocytes | [115] | ||
| α1 | Cancer-specific | MAGE-A3/6-TRIM28 | [116] | |
| α1 | Promotes α1 degradation in BRAFi-resistant melanoma cells | RNF44 | [117] | |
| α1 | PEDF promotes α1 degradation to disturb lipid metabolism in HCC cells | [118] | ||
| α2 | USP9X-mediated α2 ubiquitination in C2C12 cells suppressed by calorie restriction serum | [119] | ||
| α2 | Downregulates α2 in C2C12 cells | WWP1 | [120] | |
| α1-K387/α2-K391 | α-linker | 4 HTPs | [29] | |
| α1-K476 | ST loop | 5 HTPs | [29] | |
| α2-K470 | β-SID | Cancer progression | UBE2O | [121] |
| β1 | Promotes β1 degradation in brown adipose tissue and adipocytes to suppress fat oxidation | Promoted by Cidea | [122] | |
| β1 | Induced by E4BP4, promoting degradation | [123] | ||
| β2 | K63-linked Ub chains; accumulation, possibly associated with localisation | Laforin-malin complex | [124] | |
| γ1 | Promotes γ1 degradation through interaction with α, exacerbating high fat diet-induced metabolic defects | CRL4ACRBN | [125,126,127] | |
| γ1-K234 | CBS3 | 6 HTPs | [29] | |
| γ1-K264 | CBS3 | 7 HTPs | [29] | |
| γ1-K329 | CBS4 | 6 HTPs | [29] | |
| Sumoylation | ||||
| α1-K109/α2 | Kinase domain C-lobe | Down regulates AMPK activity towards mTORC1 signalling | PIAS4 | [128] |
| β2 | Increases AMPK activity; competes with ubiquitination | PIAS4 | [129] | |
| Acetylation | ||||
| α1-K31 | Kinase domain N-lobe (ADaM site) | K31 stabilizes ADaM site | [130] | |
| α1-K33 | Kinase domain N-lobe (ADaM site) | K33 stabilizes ADaM site | [130] | |
| α1-K71/α2-K69 | Kinase domain C-helix | 4 HTPs | [29] | |
| γ1 | NH2-terminus | Deduced from rat liver prep ToF-MS | NatB complex? | [53] |
| γ1-K264 | CBS3 | 3 HTPs | [29] | |
| γ2-K9/K10 | NH2-terminus | 1 HTP | [130] | |
| Methylation | ||||
| α2-R227 | Kinase domain C-lobe | 1 HTPs; monomethylation | [29,131] | |
| γ1-K34 | NH2-terminus | 1 HTP | [29,132] | |
| γ2-K62 | NH2-terminus | 1 HTP | [29] | |
| Oxidation | ||||
| α1-C176 | Activation loop | s-nitrosylation; in vitro using HPAEC and HEK lysates | [133,134] | |
| α1-C299/304 | AID | Activating; C304 may be a specific target for s-glutathionylation | [135] | |
| α1-C416 | β-SID | s-nitrosylation; in vitro using HEK lysates | [134] | |
| α2-C130/174 | Kinase domain C-lobe | Inhibitory; interferes with AMPK kinase interactions. Suppressed by Trx1 | [136] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ovens, A.J.; Scott, J.W.; Langendorf, C.G.; Kemp, B.E.; Oakhill, J.S.; Smiles, W.J. Post-Translational Modifications of the Energy Guardian AMP-Activated Protein Kinase. Int. J. Mol. Sci. 2021, 22, 1229. https://doi.org/10.3390/ijms22031229
Ovens AJ, Scott JW, Langendorf CG, Kemp BE, Oakhill JS, Smiles WJ. Post-Translational Modifications of the Energy Guardian AMP-Activated Protein Kinase. International Journal of Molecular Sciences. 2021; 22(3):1229. https://doi.org/10.3390/ijms22031229
Chicago/Turabian StyleOvens, Ashley J., John W. Scott, Christopher G. Langendorf, Bruce E. Kemp, Jonathan S. Oakhill, and William J. Smiles. 2021. "Post-Translational Modifications of the Energy Guardian AMP-Activated Protein Kinase" International Journal of Molecular Sciences 22, no. 3: 1229. https://doi.org/10.3390/ijms22031229








