Gelatin Methacryloyl–Riboflavin (GelMA–RF) Hydrogels for Bone Regeneration
Abstract
:1. Introduction
2. Results
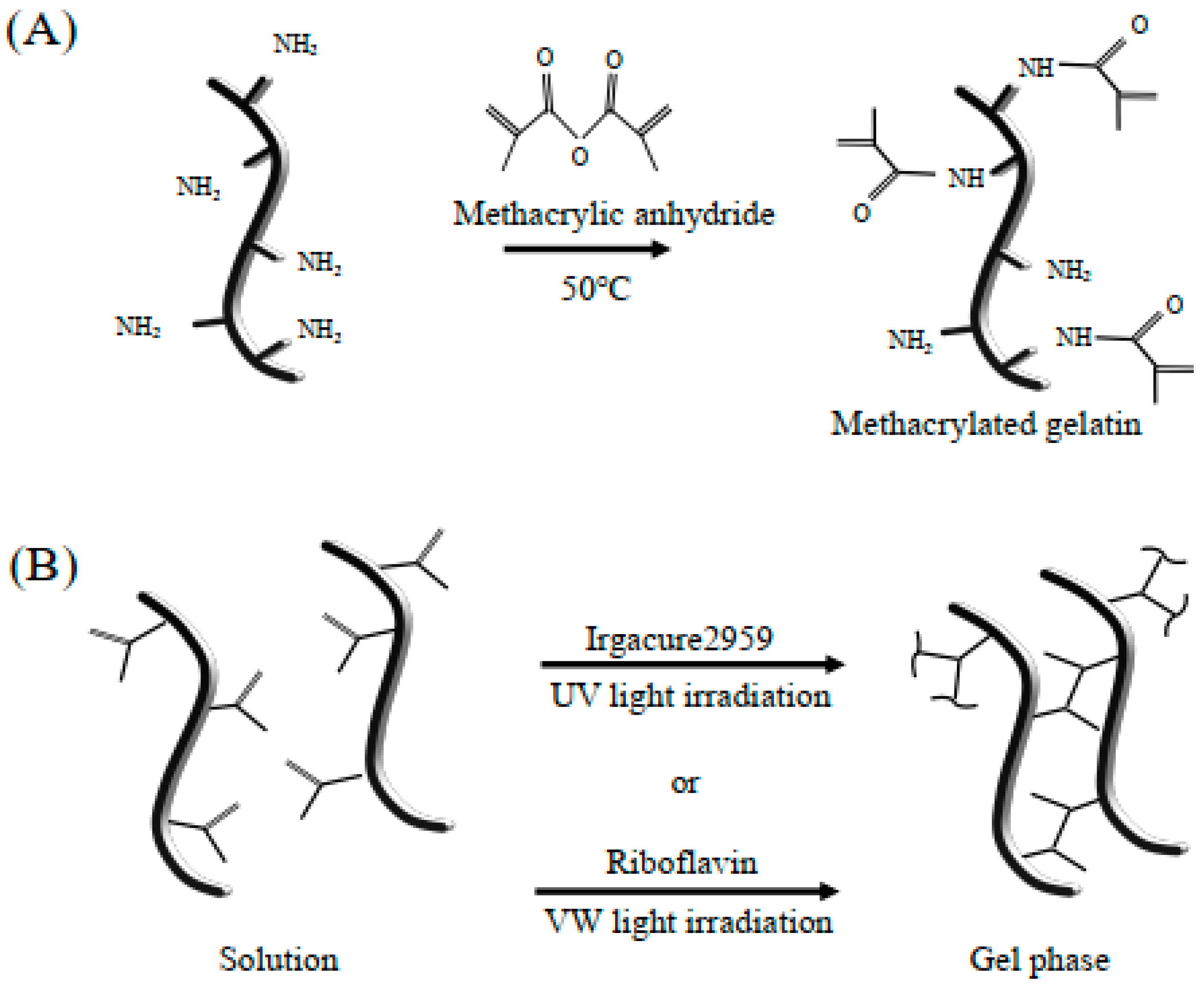
2.1. Physical and Mechanical Properties of GelMA–RF Hydrogels
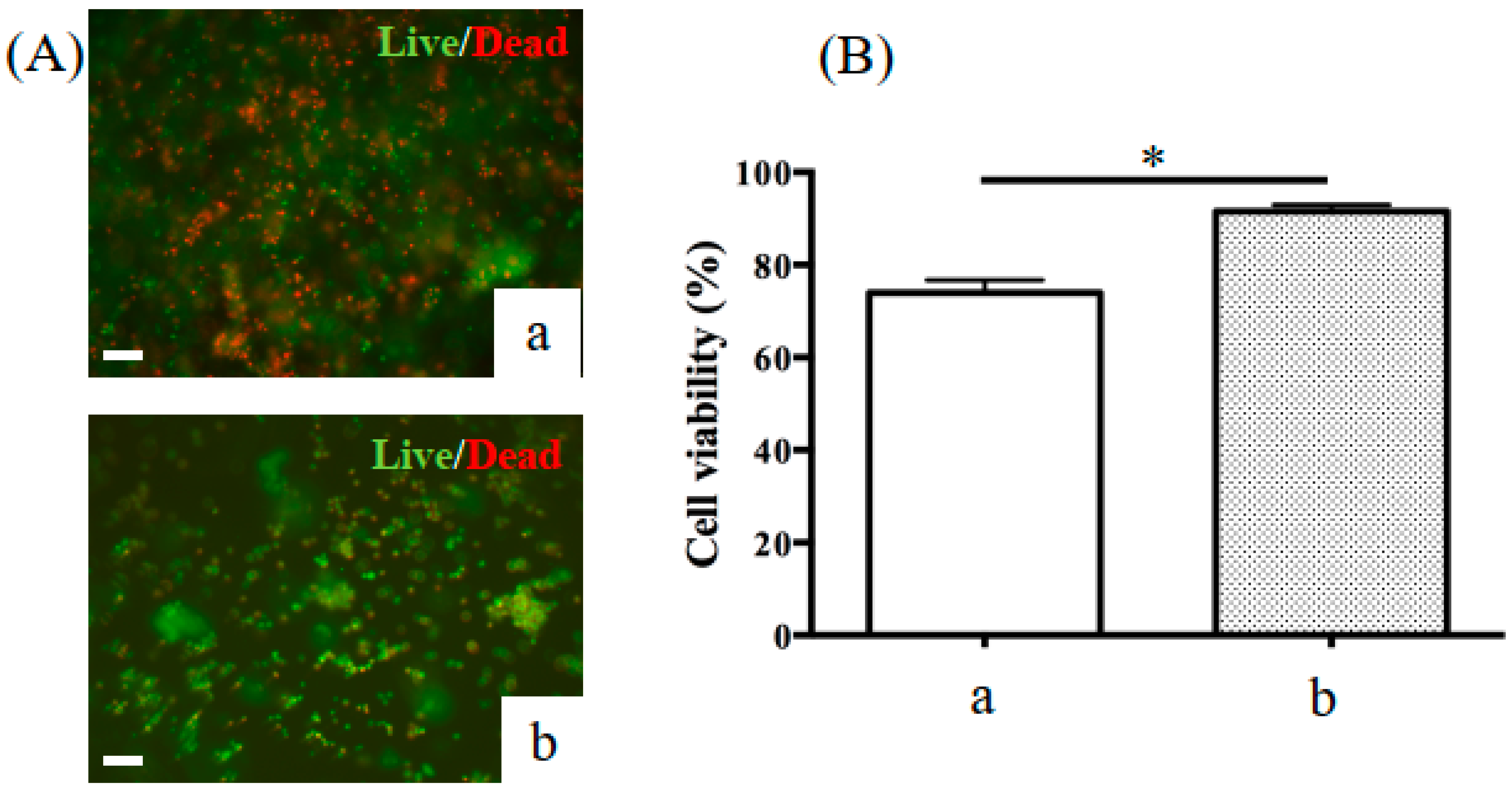
2.2. Cell Viability Following VW and UV Light Photopolymerization
2.3. Osteoblastic Differentiation of KUSA-A1 Cells in a 2D Environment
2.4. Expression of Osteogenesis-Related Genes in Osteogenic Induced KUSA-A1 Cells Encapsulated in GelMA
3. Discussion
4. Materials and Methods
4.1. GelMA Hydrogel Preparation
4.2. Determination of the Polymerization Conditions of GelMA Hydrogels
4.3. Mechanical Stability
4.4. Cell Culture and Osteo-Induction in a Two-Dimensional (2D) Environment
4.5. Cell Encapsulation and Cell Viability in a 3D Environment
4.6. Cell Encapsulation and Osteo-Induction in a 3D Environment
4.7. qPCR Analysis
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| GelMA | Gelatin methacryloyl |
| UV | Ultraviolet |
| VW | Visible wavelength |
| RF | Riboflavin |
| MA | Methacrylic anhydride |
| IR | Irgacure2959 |
| Runx2 | Runt-Related Transcription Factor 2 |
| Osx | Osterix |
| Bsp | Bone Sialoprotein |
| Ocn | Osteocalcin |
Appendix A

References
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [Green Version]
- Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef]
- Nichol, J.W.; Koshy, S.T.; Bae, H.; Hwang, C.M.; Yamanlar, S.; Khademhosseini, A. Cell-laden microengineered gelatin methacrylate hydrogels. Biomaterials 2010, 31, 5536–5544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noshadi, I.; Hong, S.; Sullivan, K.E.; Shirzaei Sani, E.; Portillo-Lara, R.; Tamayol, A.; Shin, S.R.; Gao, A.E.; Stoppel, W.L.; Black, L.D., III; et al. In vitro and in vivo analysis of visible light crosslinkable gelatin methacryloyl (GelMA) hydrogels. Biomater. Sci. 2017, 5, 2093–2105. [Google Scholar] [CrossRef]
- Paul, A.; Manoharan, V.; Krafft, D.; Assmann, A.; Uquillas, J.A.; Shin, S.R.; Hasan, A.; Hussain, M.A.; Memic, A.; Gaharwar, A.K.; et al. Nanoengineered biomimetic hydrogels for guiding human stem cell osteogenesis in three dimensional microenvironments. J. Mater. Chem. 2016, 4, 3544–3554. [Google Scholar] [CrossRef] [Green Version]
- McBeth, C.; Lauer, J.; Ottersbach, M.; Campbell, J.; Sharon, A.; Sauer-Budge, A. 3D bioprinting of GelMA scaffolds triggers mineral deposition by primary human osteoblasts. Biofabrication 2016. [Google Scholar] [CrossRef]
- Yue, K.; Trujillo-de Santiago, G.; Alvarez, M.M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015, 73, 254–271. [Google Scholar] [CrossRef] [Green Version]
- Selimovic, S.; Oh, J.; Bae, H.; Dokmeci, M.; Khademhosseini, A. Microscale Strategies for Generating Cell-Encapsulating Hydrogels. Polymers 2012, 4, 1554–1579. [Google Scholar] [CrossRef] [Green Version]
- Dua, R.; Ramaswamy, S. Relative survivability of human osteoblasts is enhanced by 39 degrees C and ascorbic acid after exposure to photopolymerization ingredients. Cytotechnology 2013, 65, 587–596. [Google Scholar] [CrossRef] [Green Version]
- Arakawa, C.; Ng, R.; Tan, S.; Kim, S.; Wu, B.; Lee, M. Photopolymerizable chitosan-collagen hydrogels for bone tissue engineering. J. Tissue Eng. Regen. Med. 2017, 11, 164–174. [Google Scholar] [CrossRef]
- Covre, J.L.; Cristovam, P.C.; Loureiro, R.R.; Hazarbassanov, R.M.; Campos, M.; Sato, E.H.; Gomes, J.A. The effects of riboflavin and ultraviolet light on keratocytes cultured in vitro. Arq. Bras. Oftalmol. 2016, 79, 180–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Husain, E.; Naseem, I. Riboflavin-mediated cellular photoinhibition of cisplatin-induced oxidative DNA breakage in mice epidermal keratinocytes. Photodermatol. Photoimmunol. Photomed. 2008, 24, 301–307. [Google Scholar] [CrossRef]
- Hardwick, C.C.; Herivel, T.R.; Hernandez, S.C.; Ruane, P.H.; Goodrich, R.P. Separation, identification and quantification of riboflavin and its photoproducts in blood products using high-performance liquid chromatography with fluorescence detection: A method to support pathogen reduction technology. Photochem. Photobiol. 2004, 80, 609–615. [Google Scholar] [CrossRef]
- Knappe, S.; Stachs, O.; Zhivov, A.; Hovakimyan, M.; Guthoff, R. Results of confocal microscopy examinations after collagen cross-linking with riboflavin and UVA light in patients with progressive keratoconus. Ophthalmologica 2011, 225, 95–104. [Google Scholar] [CrossRef]
- Orlowska, M.; Koutchma, T.; Grapperhaus, M.; Gallagher, J.; Schaefer, R.; Defelice, C. Continuous and Pulsed Ultraviolet Light for Nonthermal Treatment of Liquid Foods. Part 1: Effects on Quality of Fructose Solution, Apple Juice, and Milk. Food Bioprocess Technol. 2012, 6, 1580–1592. [Google Scholar] [CrossRef]
- Bahney, C.S.; Lujan, T.J.; Hsu, C.W.; Bottlang, M.; West, J.L.; Johnstone, B. Visible light photoinitiation of mesenchymal stem cell-laden bioresponsive hydrogels. Eur. Cells Mater. 2011, 22, 43–55. [Google Scholar] [CrossRef]
- Monteiro, N.; Thrivikraman, G.; Athirasala, A.; Tahayeri, A.; Franca, C.M.; Ferracane, J.L.; Bertassoni, L.E. Photopolymerization of cell-laden gelatin methacryloyl hydrogels using a dental curing light for regenerative dentistry. Dent. Mater. 2018, 34, 389–399. [Google Scholar] [CrossRef]
- Eguchi, T.; Watanabe, K.; Hara, E.S.; Ono, M.; Kuboki, T.; Calderwood, S.K. OstemiR: A novel panel of microRNA biomarkers in osteoblastic and osteocytic differentiation from mesencymal stem cells. PLoS ONE 2013, 8, e58796. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Chen, X.; Chen, H.; Xu, D.; Lin, C.; Peng, B. Rho/Rho-associated protein kinase signaling pathway-mediated downregulation of runt-related transcription factor 2 expression promotes the differentiation of dental pulp stem cells into odontoblasts. Exp. Ther. Med. 2018, 15, 4457–4464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yousefi, A.M.; James, P.F.; Akbarzadeh, R.; Subramanian, A.; Flavin, C.; Oudadesse, H. Prospect of Stem Cells in Bone Tissue Engineering: A Review. Stem Cells Int. 2016, 2016, 6180487. [Google Scholar] [CrossRef] [Green Version]
- Sheng, G. The developmental basis of mesenchymal stem/stromal cells (MSCs). BMC Dev. Biol. 2015, 15, 44. [Google Scholar] [CrossRef] [Green Version]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [Green Version]
- Huebsch, N.; Arany, P.R.; Mao, A.S.; Shvartsman, D.; Ali, O.A.; Bencherif, S.A.; Rivera-Feliciano, J.; Mooney, D.J. Harnessing traction-mediated manipulation of the cell/matrix interface to control stem-cell fate. Nat. Mater. 2010, 9, 518–526. [Google Scholar] [CrossRef] [Green Version]
- Khan, S.N.; Cammisa, F.P., Jr.; Sandhu, H.S.; Diwan, A.D.; Girardi, F.P.; Lane, J.M. The biology of bone grafting. J. Am. Acad. Orthop. Surg. 2005, 13, 77–86. [Google Scholar] [CrossRef]
- Janssen, N.G.; Weijs, W.L.; Koole, R.; Rosenberg, A.J.; Meijer, G.J. Tissue engineering strategies for alveolar cleft reconstruction: A systematic review of the literature. Clin. Oral Investig. 2014, 18, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Khademhosseini, A.; Langer, R. Microengineered hydrogels for tissue engineering. Biomaterials 2007, 28, 5087–5092. [Google Scholar] [CrossRef] [PubMed]
- Murtuza, B.; Nichol, J.W.; Khademhosseini, A. Micro- and nanoscale control of the cardiac stem cell niche for tissue fabrication. Tissue Eng. 2009, 15, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Aubin, H.; Nichol, J.W.; Hutson, C.B.; Bae, H.; Sieminski, A.L.; Cropek, D.M.; Akhyari, P.; Khademhosseini, A. Directed 3D cell alignment and elongation in microengineered hydrogels. Biomaterials 2010, 31, 6941–6951. [Google Scholar] [CrossRef] [Green Version]
- Bertassoni, L.E.; Cecconi, M.; Manoharan, V.; Nikkhah, M.; Hjortnaes, J.; Cristino, A.L.; Barabaschi, G.; Demarchi, D.; Dokmeci, M.R.; Yang, Y.; et al. Hydrogel bioprinted microchannel networks for vascularization of tissue engineering constructs. Lab Chip 2014, 14, 2202–2211. [Google Scholar] [CrossRef] [Green Version]
- Kirner, S.V.; Guldi, D.M.; Megiatto, J.D., Jr.; Schuster, D.I. Synthesis and photophysical properties of new catenated electron donor-acceptor materials with magnesium and free base porphyrins as donors and C60 as the acceptor. Nanoscale 2015, 7, 1145–1160. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, A.K.; Gittard, S.D.; Koroleva, A.; Schlie, S.; Gaidukeviciute, A.; Chichkov, B.N.; Narayan, R.J. Two-photon polymerization of polyethylene glycol diacrylate scaffolds with riboflavin and triethanolamine used as a water-soluble photoinitiator. Regen. Med. 2013, 8, 725–738. [Google Scholar] [CrossRef] [PubMed]
- Fairbanks, B.D.; Schwartz, M.P.; Bowman, C.N.; Anseth, K.S. Photoinitiated polymerization of PEG-diacrylate with lithium phenyl-2,4,6-trimethylbenzoylphosphinate: Polymerization rate and cytocompatibility. Biomaterials 2009, 30, 6702–6707. [Google Scholar] [CrossRef] [Green Version]
- Matsubara, T.; Kida, K.; Yamaguchi, A.; Hata, K.; Ichida, F.; Meguro, H.; Aburatani, H.; Nishimura, R.; Yoneda, T. BMP2 regulates Osterix through Msx2 and Runx2 during osteoblast differentiation. J. Biol. Chem. 2008, 283, 29119–29125. [Google Scholar] [CrossRef] [Green Version]
- Nishida, E.; Sasaki, T.; Ishikawa, S.K.; Kosaka, K.; Aino, M.; Noguchi, T.; Teranaka, T.; Shimizu, N.; Saito, M. Transcriptome database KK-Periome for periodontal ligament development: Expression profiles of the extracellular matrix genes. Gene 2007, 404, 70–79. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goto, R.; Nishida, E.; Kobayashi, S.; Aino, M.; Ohno, T.; Iwamura, Y.; Kikuchi, T.; Hayashi, J.-i.; Yamamoto, G.; Asakura, M.; et al. Gelatin Methacryloyl–Riboflavin (GelMA–RF) Hydrogels for Bone Regeneration. Int. J. Mol. Sci. 2021, 22, 1635. https://doi.org/10.3390/ijms22041635
Goto R, Nishida E, Kobayashi S, Aino M, Ohno T, Iwamura Y, Kikuchi T, Hayashi J-i, Yamamoto G, Asakura M, et al. Gelatin Methacryloyl–Riboflavin (GelMA–RF) Hydrogels for Bone Regeneration. International Journal of Molecular Sciences. 2021; 22(4):1635. https://doi.org/10.3390/ijms22041635
Chicago/Turabian StyleGoto, Ryoma, Eisaku Nishida, Shuichiro Kobayashi, Makoto Aino, Tasuku Ohno, Yuki Iwamura, Takeshi Kikuchi, Jun-ichiro Hayashi, Genta Yamamoto, Masaki Asakura, and et al. 2021. "Gelatin Methacryloyl–Riboflavin (GelMA–RF) Hydrogels for Bone Regeneration" International Journal of Molecular Sciences 22, no. 4: 1635. https://doi.org/10.3390/ijms22041635





