Anticonvulsant Effects of Topiramate and Lacosamide on Pilocarpine-Induced Status Epilepticus in Rats: A Role of Reactive Oxygen Species and Inflammation
Abstract
:1. Introduction
2. Results
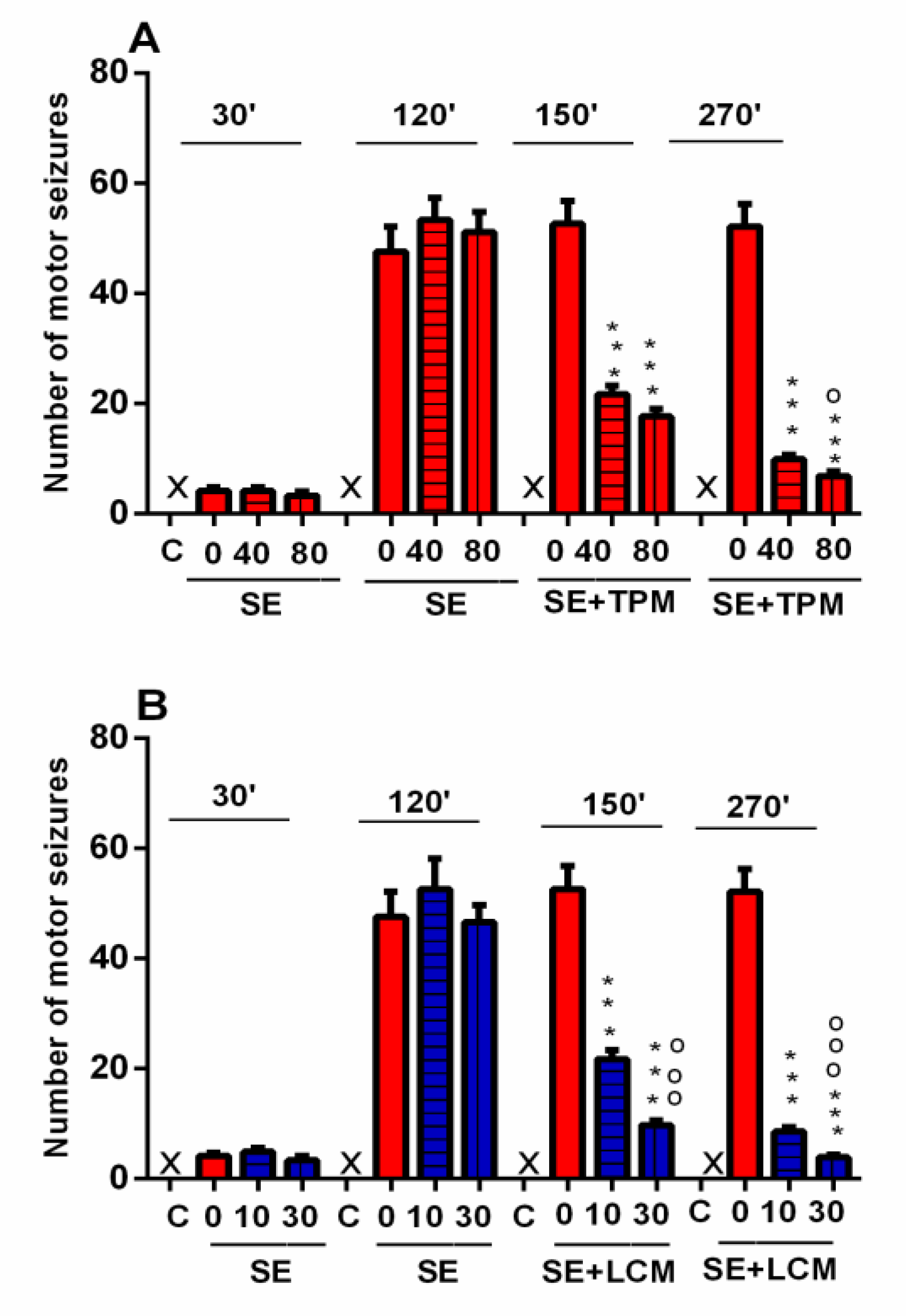
2.1. Number of Motor Seizures during SE
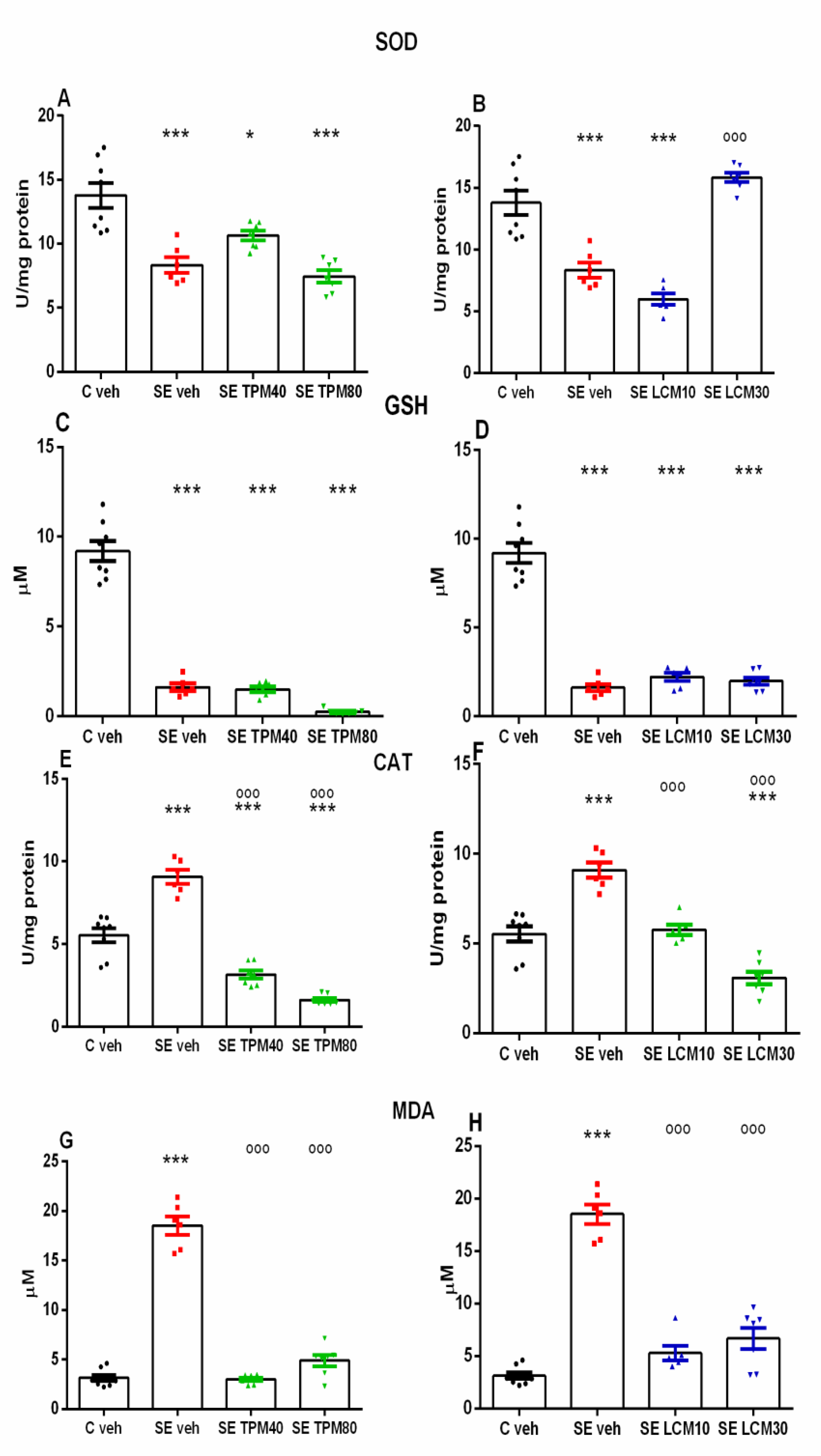
2.2. Biomarkers of Oxidative Stress in the Hippocampus
2.3. IL-1β and TNF-α Levels in the Hippocampus
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Animals
4.3. Experimental Design and Induction of Status Epilepticus
4.4. Biomarkers of Oxidative Stress in the Hippocampus
4.4.1. Preparation of Tissue Homogenate
4.4.2. SOD Activity Assay
4.4.3. CAT Activity Assay
4.4.4. GSH Assay
4.4.5. MDA Assay
4.4.6. Protein Content
4.5. Measurement of IL-1β and TNF-α
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| AEDs | antiepileptic drugs |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| ANOVA | analysis of variance |
| CAT | catalase |
| DTNB | 5,5′-dithio-bis-(2-nitrobenzoic acid |
| GABA | γ-aminobutyric acid |
| GPx | glutathione peroxidase |
| GR | glutathione reductase |
| GSH | reduced glutathione |
| IL-1β | interleukin-1β |
| IL-6 | interleukin-6 |
| KA | kainic acid |
| LCM | lacosamide |
| MDA | malondialdehyde |
| NBT | nitroblue tetrazolium |
| NMDA | N-methyl-d-aspartate |
| Pilo | pilocarpine |
| PMS | phenazine methosulfate |
| ROS | reactive oxygen species |
| SE | status epilepticus |
| SOD | superoxide dismutase |
| TBA | thiobarbituric acid |
| TCA | trichloroacetic acid |
| TNF-α | tumor necrosis factor-α |
| TPM | topiramate |
References
- Trinka, E.; Cock, H.; Hesdorffer, D.; Rossetti, A.O.; Scheffer, I.E.; Shinnar, S.; Shorvon, S.; Lowenstein, D.H. A definition and classification of status epilepticus–report of the ILAE task force on classification of status epilepticus. Epilepsia 2015, 56, 1515–1523. [Google Scholar] [CrossRef]
- Schmoll, H.; Badan, I.; Grecksch, G.; Walker, L.; Kessler, C.; Popa-Wagner, A. Kindling status in sprague-dawley rats induced by pentylenetetrazole: Involvement of a critical development period. Am. J. Patho. 2003, 162, 1027–1034. [Google Scholar] [CrossRef]
- Buga, A.-M.; Vintilescu, R.; Balseanu, A.T.; Pop, O.T.; Streba, C.; Toescu, E.; Popa-Wagner, A. Repeated PTZ treatment at 25-day intervals leads to a highly efficient accumulation of doublecortin in the dorsal hippocampus of rats. PLoS ONE 2012, 7, e39302. [Google Scholar] [CrossRef]
- Terrone, G.; Frigerio, F.; Balosso, S.; Ravizza, T.; Vezzani, A. Inflammation and reactive oxygen species in status epilepticus: Biomarkers and implications for therapy. Epilepsy Behav. 2019, 101 Pt B, 106275. [Google Scholar] [CrossRef]
- Yu, H.M.; Liu, W.H.; He, X.H.; Peng, B.W. IL-1β: An important cytokine associated with febrile seizures? Neurosci. Bull. 2012, 28, 301–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González-Reyes, S.; Santillán-Cigales, J.J.; Jiménez-Osorio, A.S.; Pedraza-Chaverri, J.; Guevara-Guzmán, R. Glycyrrhizin ameliorates oxidative stress and inflammation in hippocampus and olfactory bulb in lithium/pilocarpine-induced status epilepticus in rats. Epilepsy Res. 2016, 126, 126–133. [Google Scholar] [CrossRef]
- Shin, E.-J.; Jeong, J.H.; Chungc, Y.H.; Kimdm, W.-K.; Ko, K.-H.; Bach, J.-H.; Hong, J.-S.; Yoneda, Y.; Kim, H.-C. Role of oxidative stress in epileptic seizures. Neurochem. Int. 2011, 59, 122–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méndez-Armenta, M.; Nava-Ruíz, C.; Juárez-Rebollar, D.; Rodríguez-Martínez, E.; Gómez, P.Y. Oxidative stress associated with neuronal apoptosis in experimental models of epilepsy. Oxid. Med. Cell. Longev. 2014, 293689. [Google Scholar] [CrossRef] [PubMed]
- Swissa, E.; Serlin, Y.; Vazana, U.; Prager, O.; Friedman, A. Blood-brain barrier dysfunction in status epileptics: Mechanisms and role in epileptogenesis. Epilepsy Behav. 2019, 101 Pt B, 106285. [Google Scholar] [CrossRef]
- Zhang, R.; Yamada, J.; Hayashi, Y.; Wu, Z.; Koyama, S.; Nakanishi, H. Inhibition of NMDA-induced outward currents by interleukin-1beta in hippocampal neurons. Biochem. Biophys. Res. Commun. 2008, 372, 816–820. [Google Scholar] [CrossRef]
- Vezzani, A.; Viviani, B. Neuromodulatory properties of inflammatory cytokines and their impact on neuronal excitability. Neuropharmacology 2015, 96 Pt A, 70–82. [Google Scholar] [CrossRef]
- Li, G.; Bauer, S.; Nowak, M.; Norwood, B.; Tackenberg, B.; Rosenow, F.; Knake, S.; Oertel, W.H.; Hamer, H.M. Cytokines and epilepsy. Seizure 2011, 20, 249–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Z.S.; Huang, W.L.; Gong, S.J. Effect of adenovirus-mediated overexpression of PTEN on brain oxidative damage and neuroinflammation in a rat kindling model of epilepsy. Chin. Med. J. 2019, 132, 2628–2635. [Google Scholar] [CrossRef]
- Chen, J.; Quan, Q.Y.; Yang, F.; Wang, Y.; Wang, J.C.; Zhao, G.; Jiang, W. Effects of lamotrigine and topiramate on hippocampal neurogenesis in experimental temporal-lobe epilepsy. Brain Res. 2010, 1313, 270–282. [Google Scholar] [CrossRef]
- Pauletti, A.; Terrone, G.; Shekh-Ahmad, T.; Salamone, A.; Ravizza, T.; Rizzi, M.; Pastore, A.; Pascente, R.; Liang, L.-P.; Villa, B.R.; et al. Targeting Oxidative Stress Improves Disease Outcomes in a Rat Model of Acquired Epilepsy. Brain 2019, 142, e39. [Google Scholar] [CrossRef]
- Nakhutina, L.; Kunnakkat, S.D.; Coleman, M.; Lushbough, C.; Arnedo, V.; Soni, N.; Grant, A.C. Effects of adjunctive lacosamide on mood and quality of life in patients with epilepsy. Epilepsy Behav. 2017, 73, 90–94. [Google Scholar] [CrossRef]
- Racine, R.J. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr. Clin. Neurophysiol. 1972, 32, 281–294. [Google Scholar] [CrossRef]
- Madžar, D.; Kuramatsu, J.B.; Gerner, S.T.; Huttner, H.B. Assessing the value of topiramate in refractory status epilepticus. Seizure 2016, 38, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Davidson, K.E.; Newell, J.; Alsherbini, K.; Krushinski, J.; Jones, G.M. Safety and Efficiency of Intravenous Push Lacosamide Administration. Neurocrit. Care 2018, 29, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Mnatsakanyan, L.; Chung, J.M.; Tsimerinov, E.I.; Eliashiv, D.S. Intravenous lacosamide in refractory nonconvulsive status epilepticus. Seizure 2012, 21, 198–201. [Google Scholar] [CrossRef] [Green Version]
- Hofler, J.; Unterberger, I.; Dobesberger, J.; Kuchukhidze, G.; Walser, G.; Trinka, E. Intravenous lacosamide in status epilepticus and seizure clusters. Epilepsia 2011, 52, e148–e152. [Google Scholar] [CrossRef]
- Hofler, J.; Trinka, E. Lacosamide as a new treatment option in status epilepticus. Epilepsia 2013, 54, 393–404. [Google Scholar] [CrossRef]
- Strzelczyk, A.; Zöllner, J.P.; Willems, L.M.; Jost, J.; Paule, E.; Schubert-Bast, S.; Rosenow, F.; Bauer, S. Lacosamide in Status Epilepticus: Systematic Review of Current Evidence. Epilepsia 2017, 58, 933–950. [Google Scholar] [CrossRef] [Green Version]
- Tchekalarova, J.; Atanasova, D.; Kortenska, L.; Lazarov, N.; Shishmanova-Doseva, M.; Galchev, T.; Marinov, P. Agomelatine alleviates neuronal loss through BDNF signaling in the post-status epilepticus model induced by kainic acid in rat. Brain Res. Bull. 2019, 147, 22–35. [Google Scholar] [CrossRef]
- Nirwan, N.; Siraj, F.; Vohora, D. Inverted-U response of lacosamide on pilocarpine-induced status epilepticus and oxidative stress in C57BL/6 mice is independent of hippocampal collapsin response mediator protein-2. Epilepsy Res. 2018, 145, 93–101. [Google Scholar] [CrossRef]
- Behr, C.; Lévesque, M.; Ragsdale, D.; Avoli, M. Lacosamide Modulates Interictal Spiking and High-Frequency Oscillations in a Model of Mesial Temporal Lobe Epilepsy. Epilepsy Res. 2015, 115, 8–16. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Yu, Y.; Ma, R.; Shao, N.; Meng, H. Lacosamide modulates collapsin response mediator protein 2 and inhibits mossy fiber sprouting after kainic acid-induced status epilepticus. Neuroreport 2018, 29, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Patel, M. Targeting Oxidative Stress in Central Nervous System Disorders. Trends Pharmacol. Sci. 2016, 37, 768–778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araújo, I.M.; Ambrósio, A.F.; Leal, E.C.; Verdasca, M.J.; Malva, J.O.; Soares-da-Silva, P.; Carvalho, A.P.; Carvalho, C.M. Neurotoxicity induced by antiepileptic drugs in cultured hippocampal neurons: A comparative study between carbamazepine, oxcarbazepine, and two new putative antiepileptic drugs, BIA 2-024 and BIA 2-093. Epilepsia 2004, 45, 1498–1505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazhar, F.; Malhi, S.M.; Simjee, S.U. Comparative Studies on the Effects of Clinically Used Anticonvulsants on the Oxidative Stress Biomarkers in Pentylenetetrazole-Induced Kindling Model of Epileptogenesis in Mice. J. Basic Clin. Physiol. Pharmacol. 2017, 28, 31–42. [Google Scholar] [CrossRef]
- Valdovinos-Flores, C.; Gonsebatt, M.E. The Role of Amino Acid Transporters in GSH Synthesis in the Blood-Brain Barrier and Central Nervous System. Neurochem. Int. 2012, 61, 405–414. [Google Scholar] [CrossRef]
- Aguiar, C.C.; Almeida, A.B.; Araújo, P.V.; de Abreu, R.N.; Chaves, E.M.; do Vale, O.C.; Macêdo, D.S.; Woods, D.J.; Fonteles, M.M.; Vasconcelos, S.M. Oxidative stress and epilepsy: Literature review. Oxid. Med. Cell. Longev. 2012, 795259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abraham, M.; Mohamed-Faisal, K.P.; Biju, C.R.; Babu, G. Neuroprotective effect of lacosamide and pregabalin on strychnine induced seizure models in rat. World J. Pharm. Pharmac. Sci. 2014, 3, 1324–1329. [Google Scholar]
- Attia, G.M.; Elmansy, R.A.; Elsaed, W.M. Neuroprotective effect of nilotinib on pentylenetetrazol-induced epilepsy in adult rat hippocampus: Involvement of oxidative stress, autophagy, inflammation, and apoptosis. Folia Neuropathol. 2019, 57, 146–160. [Google Scholar] [CrossRef] [PubMed]
- Freitas, R.M.; Nascimento, V.S.; Vasconcelos, S.M.; Sousa, F.C.; Viana, G.S.; Fonteles, M.M. Catalase activity in cerebellum, hippocampus, frontal cortex and striatum after status epilepticus induced by pilocarpine in Wistar rats. Neurosci. Lett. 2004, 365, 102–105. [Google Scholar] [CrossRef]
- Freitas, R.M.; Vasconcelos, S.M.; Souza, F.C.; Viana, G.S.; Fonteles, M.M. Oxidative stress in the hippocampus after pilocarpine-induced status epilepticus in Wistar rats. FEBS J. 2005, 272, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.F.; Freitas, R.L.; Xavier, S.M.; Saldanha, G.B.; Freitas, R.M. Neuroprotective actions of vitamin C related to decreased lipid peroxidation and increased catalase activity in adult rats after pilocarpine-induced seizures. Pharmacol. Biochem. Behav. 2018, 89, 1–5. [Google Scholar] [CrossRef]
- Agarwal, N.B.; Agarwal, N.K.; Mediratta, P.K.; Sharma, K.K. Effect of lamotrigine, oxcarbazepine and topiramate on cognitive functions and oxidative stress in PTZ-kindled mice. Seizure 2011, 20, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Sarangi, S.C.; Kakkar, A.K.; Kumar, R.; Gupta, Y.K. Effect of Lamotrigine, Levetiracetam & Topiramate on Neurobehavioural Parameters & Oxidative Stress in Comparison With Valproate in Rats. Indian J. Med. Res. 2016, 144, 104–111. [Google Scholar] [CrossRef]
- Kimura, A.; Namekata, K.; Guo, X.; Noro, T.; Harada, C.; Harada, T. Targeting Oxidative Stress for Treatment of Glaucoma and Optic Neuritis. Oxid. Med. Cell. Longev. 2017, 2817252. [Google Scholar] [CrossRef]
- Aycicek, A.; Iscan, A. The Effects of Carbamazepine, Valproic Acid and Phenobarbital on the Oxidative and Antioxidative Balance in Epileptic Children. Eur Neurol. 2006, 57, 65–69. [Google Scholar] [CrossRef]
- Verrotti, A.; Scardapane, A.; Franzoni, E.; Manco, R.; Chiarelli, F. Increased Oxidative Stress in Epileptic Children Treated with Valproic Acid. Epilepsy Res. 2008, 78, 171–177. [Google Scholar] [CrossRef]
- Löscher, W.; Schmidt, D. Experimental and Clinical Evidence for Loss of Effect (Tolerance) During Prolonged Treatment with Antiepileptic Drugs. Epilepsia 2006, 47, 1253–1284. [Google Scholar] [CrossRef]
- Mohd Sairazi, N.S.; Sirajudeen, K.N.S.; Asari, M.A.; Mummedy, S.; Muzaimi, M.; Sulaiman, S.A. Effect of tualang honey against KA-induced oxidative stress and neurodegeneration in the cortex of rats. BMC Complement. Altern. Med. 2017, 17, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demiroz, S.; Ur, K.; Ulucan, A.; Bengu, A.S.; Ur, F.D.; Gergin, O.O.; Erdem, S. Neuroprotective Effects of Lacosamide in Experimental Traumatic Spinal Cord Injury in Rats. Turk. Neurosurg. 2019, 29, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Patterson, K.P.; Brennan, G.P.; Curran, M.; Kinney-Lang, E.; Dubé, C.; Rashid, F.; Ly, C.; Obenaus, A.; Baram, T.Z. Rapid, Coordinate Inflammatory Responses After Experimental Febrile Status Epilepticus: Implications for Epileptogenesis. eNeuro 2015, 2. [Google Scholar] [CrossRef] [Green Version]
- Vezzani, A.; Moneta, D.; Richichi, C.; Aliprandi, M.; Burrows, S.J.; Ravizza, T.; Perego, C.; De Simoni, M.G. Functional role of inflammatory cytokines and antiinflammatory molecules in seizures and epileptogenesis. Epilepsia 2002, 43, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Mi, X.; Gao, B.; Gu, J.; Wang, W.; Zhang, Y.; Wang, X. Minocycline inhibits brain inflammation and attenuates spontaneous recurrent seizures following pilocarpine-induced status epilepticus. Neuroscience 2015, 26, 144–156. [Google Scholar] [CrossRef]
- Auvin, S.; Shin, D.; Mazarati, A.; Sankar, R. Inflammation induced by LPS enhances epileptogenesis in immature rat and may be partially reversed by IL1RA. Epilepsia 2010, 51, 34–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rao, R.S.; Medhi, B.; Saikia, U.N.; Arora, S.K.; Toor, J.S.; Khanduja, K.L.; Pandhi, P. Experimentally induced various inflammatory models and seizure: Understanding the role of cytokine in rat. Eur. Neuropsychopharmacol. 2008, 18, 760–767. [Google Scholar] [CrossRef]
- Bauer, S.; Cepok, S.; Todorova-Rudolph, A.; Nowak, M.; Köller, M.; Lorenz, R.; Oertel, W.H.; Rosenow, F.; Hemmer, B.; Hamer, H.M. Etiology and site of temporal lobe epilepsy influence postictal cytokine release. Epilepsy Res. 2009, 86, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Andrzejczak, D.; Woldan-Tambor, A.; Bednarska, K.; Zawilska, J.B. The effects of topiramate on lipopolysaccharide (LPS)-induced proinflammatory cytokine release from primary rat microglial cell cultures. Epilepsy Res. 2016, 127, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Savran, M.; Ozmen, O.; Erzurumlu, Y.; Savas, H.B.; Asci, S.; Kaynak, M. The Impact of Prophylactic Lacosamide on LPS-Induced Neuroinflammation in Aged Rats. Inflammation 2019, 42, 1913–1924. [Google Scholar] [CrossRef]
- Tchekalarova, J.; Ivanova, N.; Nenchovska, Z.; Tzoneva, R.; Stoyanova, T.; Uzunova, V.; Surcheva, S.; Tzonev, A.; Angelova, V.; Andreeva-Gateva, P. Evaluation of neurobiological and antioxidant effects of novel melatonin analogs in mice. Saudi Pharm. J. 2020, 28, 1566–1579. [Google Scholar] [CrossRef] [PubMed]
- Kakkar, P.; Das, B.; Viswanathan, P.N. A modified spectrophotometric assay of superoxide dismutase. Indian J. Biochem. Biophys. 1984, 21, 130–132. [Google Scholar] [PubMed]
- Khan, R.A.; Khan, M.R.; Sahreen, S. Brain antioxidant markers, cognitive performance and acetylcholinesterase activity of rats: Efficiency of Sonchus asper. Behav. Brain Funct. 2012, 16, 8–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shishmanova-Doseva, M.; Peychev, L.; Yoanidu, L.; Uzunova, Y.; Atanasova, M.; Georgieva, K.; Tchekalarova, J. Anticonvulsant Effects of Topiramate and Lacosamide on Pilocarpine-Induced Status Epilepticus in Rats: A Role of Reactive Oxygen Species and Inflammation. Int. J. Mol. Sci. 2021, 22, 2264. https://doi.org/10.3390/ijms22052264
Shishmanova-Doseva M, Peychev L, Yoanidu L, Uzunova Y, Atanasova M, Georgieva K, Tchekalarova J. Anticonvulsant Effects of Topiramate and Lacosamide on Pilocarpine-Induced Status Epilepticus in Rats: A Role of Reactive Oxygen Species and Inflammation. International Journal of Molecular Sciences. 2021; 22(5):2264. https://doi.org/10.3390/ijms22052264
Chicago/Turabian StyleShishmanova-Doseva, Michaela, Lyudmil Peychev, Lyubka Yoanidu, Yordanka Uzunova, Milena Atanasova, Katerina Georgieva, and Jana Tchekalarova. 2021. "Anticonvulsant Effects of Topiramate and Lacosamide on Pilocarpine-Induced Status Epilepticus in Rats: A Role of Reactive Oxygen Species and Inflammation" International Journal of Molecular Sciences 22, no. 5: 2264. https://doi.org/10.3390/ijms22052264







