piRNAs as Modulators of Disease Pathogenesis
Abstract
:1. Introduction
2. Structure and Function of piRNAs
2.1. piRNA Structure and Biogenesis
2.2. Transposon Silencing
2.3. Epigenetic Regulation
2.4. Post-transcriptional and Translational Control
3. piRNAs vs other sncRNAs
4. The Role of piRNAs in Cardiomyopathies
5. Influence of piRNAs in Cancer Pathogenesis
5.1. Breast Cancer
5.2. Lung Cancer
5.3. Colorectal Cancer
5.4. Gastric Cancer
5.5. Bladder Cancer
5.6. Liver Cancer
5.7. Melanoma
5.8. Glioblastoma
5.9. Pancreatic Cancer
6. piRNAs and Infectious Diseases
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Farazi, T.A.; Juranek, S.A.; Tuschl, T. The growing catalog of small RNAs and their association with distinct Argonaute/Piwi family members. Development 2008, 135, 1201–1214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leung, Y.Y.; Kuksa, P.P.; Amlie-Wolf, A.; Valladares, O.; Ungar, L.H.; Kannan, S.; Gregory, B.D.; Wang, L.-S. DASHR: Database of small human noncoding RNAs. Nucleic Acids Res. 2015, 44, D216–D222. [Google Scholar] [CrossRef] [Green Version]
- Fang, Z.; Du, R.; Edwards, A.; Flemington, E.K.; Zhang, K. The Sequence Structures of Human MicroRNA Molecules and Their Implications. PLoS ONE 2013, 8, e54215. [Google Scholar] [CrossRef] [PubMed]
- Brennecke, J.; Aravin, A.A.; Stark, A.; Dus, M.; Kellis, M.; Sachidanandam, R.; Hannon, G.J. Discrete Small RNA-Generating Loci as Master Regulators of Transposon Activity in Drosophila. Cell 2007, 128, 1089–1103. [Google Scholar] [CrossRef] [Green Version]
- Murchison, E.P.; Kheradpour, P.; Sachidanandam, R.; Smith, C.; Hodges, E.; Xuan, Z.; Kellis, M.; Grutzner, F.; Stärk, A.; Hannon, G.J. Conservation of small RNA pathways in platypus. Genome Res. 2008, 18, 995–1004. [Google Scholar] [CrossRef] [Green Version]
- Juliano, C.E.; Reich, A.; Liu, N.; Götzfried, J.; Zhong, M.; Uman, S.; Reenan, R.A.; Wessel, G.M.; Steele, R.E.; Lin, H. PIWI proteins and PIWI-interacting RNAs function in Hydra somatic stem cells. Proc. Natl. Acad. Sci. USA 2013, 111, 337–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vagin, V.V.; Sigova, A.A.; Li, C.; Seitz, H.; Gvozdev, V.; Zamore, P.D. A Distinct Small RNA Pathway Silences Selfish Genetic Elements in the Germline. Science 2006, 313, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Aravin, A.A.; Sachidanandam, R.; Girard, A.; Fejes-Toth, K.; Hannon, G.J. Developmentally Regulated piRNA Clusters Implicate MILI in Transposon Control. Science 2007, 316, 744–747. [Google Scholar] [CrossRef] [Green Version]
- Aravin, A.A.; Sachidanandam, R.; Bourc’His, D.; Schaefer, C.; Pezic, D.; Toth, K.F.; Bestor, T.; Hannon, G.J. A piRNA Pathway Primed by Individual Transposons Is Linked to De Novo DNA Methylation in Mice. Mol. Cell 2008, 31, 785–799. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roovers, E.F.; Rosenkranz, D.; Mahdipour, M.; Han, C.-T.; He, N.; Lopes, S.M.C.D.S.; Van der Westerlaken, L.A.; Zischler, H.; Butter, F.; Roelen, B.A.; et al. Piwi Proteins and piRNAs in Mammalian Oocytes and Early Embryos. Cell Rep. 2015, 10, 2069–2082. [Google Scholar] [CrossRef] [Green Version]
- Praher, D.; Zimmermann, B.; Genikhovich, G.; Columbus-Shenkar, Y.; Modepalli, V.; Aharoni, R.; Moran, Y.; Technau, U. Characterization of the piRNA pathway during development of the sea anemone Nematostella vectensis. RNA Biol. 2017, 14, 1727–1741. [Google Scholar] [CrossRef] [Green Version]
- Gainetdinov, I.; Skvortsova, Y.; Kondratieva, S.; Funikov, S.; Azhikina, T. Two modes of targeting transposable elements by piRNA pathway in human testis. RNA 2017, 23, 1614–1625. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.C.; Lin, H. Beyond transposons: The epigenetic and somatic functions of the Piwi-piRNA mechanism. Curr. Opin. Cell Biol. 2013, 25, 190–194. [Google Scholar] [CrossRef] [Green Version]
- Kozomara, A.; Griffiths-Jones, S. miRBase: Annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res. 2014, 42, D68–D73. [Google Scholar] [CrossRef] [Green Version]
- Song, J.-J.; Liu, J.; Tolia, N.H.; Schneiderman, J.; Smith, S.K.; Martienssen, R.A.; Hannon, G.J.; Joshua-Tor, L. The crystal structure of the Argonaute2 PAZ domain reveals an RNA binding motif in RNAi effector complexes. Nat. Struct. Mol. Biol. 2003, 10, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.S.; Roe, S.M.; Barford, D. Crystal structure of a PIWI protein suggests mechanisms for siRNA recognition and slicer activity. EMBO J. 2004, 23, 4727–4737. [Google Scholar] [CrossRef] [Green Version]
- Cheng, E.-C.; Kang, D.; Wang, Z.; Lin, H. PIWI Proteins Are Dispensable for Mouse Somatic Development and Reprogramming of Fibroblasts into Pluripotent Stem Cells. PLoS ONE 2014, 9, e97821. [Google Scholar] [CrossRef] [Green Version]
- Ross, R.J.; Weiner, M.M.; Lin, H. PIWI proteins and PIWI-interacting RNAs in the soma. Nat. Cell Biol. 2014, 505, 353–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, D.; Zeeman, A.-M.; Deng, W.; Looijenga, L.H.J.; Lin, H. Molecular characterization of hiwi, a human member of the piwi gene family whose overexpression is correlated to seminomas. Oncogene 2002, 21, 3988–3999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuramochi-Miyagawa, S.; Kimura, T.; Yomogida, K.; Kuroiwa, A.; Tadokoro, Y.; Fujita, Y.; Sato, M.; Matsuda, Y.; Nakano, T. Two mouse piwi-related genes: Miwi and mili. Mech. Dev. 2001, 108, 121–133. [Google Scholar] [CrossRef]
- Mani, S.R.; Juliano, C.E. Untangling the web: The diverse functions of the PIWI/piRNA pathway. Mol. Reprod. Dev. 2013, 80, 632–664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, X.-Q.; Zhang, Y.-H.; Liu, F.; Ponnusamy, M.; Zhao, X.-M.; Zhou, L.-Y.; Zhai, M.; Liu, C.-Y.; Li, X.-M.; Wang, M.; et al. The piRNA CHAPIR regulates cardiac hypertrophy by controlling METTL3-dependent N6-methyladenosine methylation of Parp10 mRNA. Nat. Cell Biol. 2020, 22, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Xue, F.-T.; Li, Y.-Y.; Liu, W.; Zhang, S. Exosomal piRNA sequencing reveals differences between heart failure and healthy patients. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7952–7961. [Google Scholar] [PubMed]
- Rajan, K.S.; Velmurugan, G.; Gopal, P.; Ramprasath, T.; Babu, D.V.; Krithika, S.; Jenifer, Y.C.; Freddy, A.; William, G.; Kalpana, K.; et al. Abundant and Altered Expression of PIWI-Interacting RNAs during Cardiac Hypertrophy. Heart Lung Circ. 2016, 25, 1013–1020. [Google Scholar] [CrossRef]
- Peng, L.; Song, L.; Liu, C.; Lv, X.; Li, X.; Jie, J.; Zhao, D.; Li, D. piR-55490 inhibits the growth of lung carcinoma by suppressing mTOR signaling. Tumor Biol. 2015, 37, 2749–2756. [Google Scholar] [CrossRef] [PubMed]
- Chong, Z.Z.; Shang, Y.C.; Maiese, K. Cardiovascular Disease and mTOR Signaling. Trends Cardiovasc. Med. 2011, 21, 151–155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, G.; Hu, H.; Xue, X.; Shen, S.; Gao, E.; Guo, G.; Shen, X.; Zhang, X. Altered expression of piRNAs and their relation with clinicopathologic features of breast cancer. Clin. Transl. Oncol. 2012, 15, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Fu, A.; Jacobs, D.I.; Hoffman, A.E.; Zheng, T.; Zhu, Y. PIWI-interacting RNA 021285 is involved in breast tumorigenesis possibly by remodeling the cancer epigenome. Carcinogenesis 2015, 36, 1094–1102. [Google Scholar] [CrossRef] [Green Version]
- Mei, Y.; Wanh, Y.; Kumari, P.; Shetty, A.C.; Clark, D.; Gable, T.; MacKerell, A.D.; Ma, M.Z.; Weber, D.J.; Yang, A.J.; et al. A piRNA-like small RNA interacts with and modulates p-ERM proteins in human somatic cells. Nat. Commun. 2015, 6, 7316. [Google Scholar] [CrossRef] [Green Version]
- Yao, J.; Wang, Y.W.; Fang, B.B.; Zhang, S.J.; Cheng, B.L. piR-651 and its function in 95-D lung cancer cells. Biomed. Rep. 2016, 4, 546–550. [Google Scholar] [CrossRef] [Green Version]
- Weng, W.; Liu, N.; Toiyama, Y.; Kusunoki, M.; Nagasaka, T.; Fujiwara, T.; Wei, Q.; Qin, H.; Lin, H.; Ma, Y.; et al. Novel evidence for a PIWI-interacting RNA (piRNA) as an oncogenic mediator of disease progression, and a potential prognostic biomarker in colorectal cancer. Mol. Cancer 2018, 17, 1–12. [Google Scholar] [CrossRef]
- Yan, H.; Wu, Q.-L.; Sun, C.-Y.; Ai, L.-S.; Deng, J.; Zhang, L.; Chen, L.; Chu, Z.-B.; Tang, B.; Wang, K.; et al. piRNA-823 contributes to tumorigenesis by regulating de novo DNA methylation and angiogenesis in multiple myeloma. Leukemia 2014, 29, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Hui, G.; Yuan, L.; Shi, D.; Wang, Y.; Du, M.; Zhong, D.; Ma, L.; Tong, N.; Qin, C.; et al. Identification of novel piRNAs in bladder cancer. Cancer Lett. 2015, 356, 561–567. [Google Scholar] [CrossRef]
- Rizzo, F.; Rinaldi, A.; Marchese, G.; Coviello, E.; Sellitto, A.; Cordella, A.; Giurato, G.; Nassa, G.; Ravo, M.; Tarallo, R.; et al. Specific patterns of PIWI-interacting small noncoding RNA expression in dysplastic liver nodules and hepatocellular carcinoma. Oncotarget 2016, 7, 54650–54661. [Google Scholar] [CrossRef] [PubMed]
- Law, P.T.-Y.; Qin, H.; Ching, A.K.-K.; Lai, K.P.; Na Co, N.; He, M.; Lung, R.W.-M.; Chan, A.W.-H.; Chan, T.-F.; Wong, N. Deep sequencing of small RNA transcriptome reveals novel non-coding RNAs in hepatocellular carcinoma. J. Hepatol. 2013, 58, 1165–1173. [Google Scholar] [CrossRef]
- Müller, S.; Raulefs, S.; Bruns, P.; Afonso-Grunz, F.; Plötner, A.; Thermann, R.; Jäger, C.; Schlitter, A.M.; Kong, B.; Regel, I.; et al. Next-generation sequencing reveals novel differentially regulated mRNAs, lncRNAs, miRNAs, sdRNAs and a piRNA in pancreatic cancer. Mol. Cancer 2015, 14, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Rayford, K.J.; Cooley, A.; Arun, A.; Rachakonda, G.; Kleschenko, Y.; Villalta, F.; Pratap, S.; Lima, M.F.; Nde, P.N. Trypanosoma cruzi Modulates PIWI-Interacting RNA Expression in Primary Human Cardiac Myocytes during the Early Phase of Infection. Int. J. Mol. Sci. 2020, 21, 9439. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Dong, Y.; Xiao-Guang, C.; Puthiyakunnon, S.; Wu, Y.; Chen, X.-G. Developmental piRNA profiles of the invasive vector mosquito Aedes albopictus. Parasites Vectors 2016, 9, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Kirino, Y.; Mourelatos, Z. Mouse Piwi-interacting RNAs are 2’-O-methylated at their 3’ termini. Nat. Struct. Mol. Biol. 2007, 14, 347–348. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Sakaguchi, Y.; Suzuki, T.; Siomi, H.; Siomi, M.C. Pimet, the Drosophila homolog of HEN1, mediates 2’-O-methylation of Piwi-interacting RNAs at their 3’ ends. Genes Dev. 2007, 21, 1603–1608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, S.L.; Qu, Z.P.; Kortschak, D.R.; Lawrence, D.M.; Geoghegan, J.; Hempfling, A.L.; Bergmann, M.; Goodnow, C.C.; Ormandy, C.J.; Wong, L.; et al. HENMT1 and piRNA Stability Are Required for Adult Male Germ Cell Transposon Repression and to Define the Spermatogenic Program in the Mouse. PLoS Genet. 2015, 11, e1005620. [Google Scholar]
- Weick, E.-M.; Miska, E.A. piRNAs: From biogenesis to function. Development 2014, 141, 3458–3471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Welker, N.C.; Pavelec, D.M.; Nix, D.A.; Duchaine, T.F.; Kennedy, S.; Bass, B.L. Dicer’s helicase domain is required for accumulation of some, but not all, C. elegans endogenous siRNAs. RNA 2010, 16, 893–903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saito, K.; Nishida, K.M.; Mori, T.; Kawamura, Y.; Miyoshi, K.; Nagami, T.; Siomi, H.; Siomi, M.C. Specific association of Piwi with rasiRNAs derived from retrotransposon and heterochromatic regions in the Drosophila genome. Genes Dev. 2006, 20, 2214–2222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aravin, A.; Gaidatzis, D.; Pfeffer, S.; Lagos-Quintana, M.; Landgraf, P.; Iovino, N.; Morris, P.; Brownstein, M.J.; Kuramochi-Miyagawa, S.; Nakano, T.; et al. A novel class of small RNAs bind to MILI protein in mouse testes. Nat. Cell Biol. 2006, 442, 203–207. [Google Scholar] [CrossRef]
- Girard, A.; Sachidanandam, R.; Hannon, G.J.; Carmell, M.A. A germline-specific class of small RNAs binds mammalian Piwi proteins. Nat. Cell Biol. 2006, 442, 199–202. [Google Scholar] [CrossRef]
- Grivna, S.T.; Beyret, E.; Wang, Z.; Lin, H. A novel class of small RNAs in mouse spermatogenic cells. Genes Dev. 2006, 20, 1709–1714. [Google Scholar] [CrossRef] [Green Version]
- Gu, W.; Lee, H.-C.; Chaves, D.A.; Youngman, E.M.; Pazour, G.J.; Conte, D.; Mello, C.C. CapSeq and CIP-TAP Identify Pol II Start Sites and Reveal Capped Small RNAs as C. elegans piRNA Precursors. Cell 2012, 151, 1488–1500. [Google Scholar] [CrossRef] [Green Version]
- Hirakata, S.; Ishizu, H.; Fujita, A.; Tomoe, Y.; Siomi, M.C. Requirements for multivalent Yb body assembly in transposon silencing in Drosophila. EMBO Rep. 2019, 20, e47708. [Google Scholar] [CrossRef]
- Qi, H.; Watanabe, T.; Ku, H.-Y.; Liu, N.; Zhong, M.; Lin, H. The Yb Body, a Major Site for Piwi-associated RNA Biogenesis and a Gateway for Piwi Expression and Transport to the Nucleus in Somatic Cells. J. Biol. Chem. 2011, 286, 3789–3797. [Google Scholar] [CrossRef] [Green Version]
- Saito, K.; Ishizu, H.; Komai, M.; Kotani, H.; Kawamura, Y.; Nishida, K.M.; Siomi, H.; Siomi, M.C. Roles for the Yb body components Armitage and Yb in primary piRNA biogenesis in Drosophila. Genes Dev. 2010, 24, 2493–2498. [Google Scholar] [CrossRef] [Green Version]
- Haase, A.D.; Fenoglio, S.; Muerdter, F.; Guzzardo, P.M.; Czech, B.; Pappin, D.J.; Chen, C.; Gordon, A.; Hannon, G.J. Probing the initiation and effector phases of the somatic piRNA pathway in Drosophila. Genes Dev. 2010, 24, 2499–2504. [Google Scholar] [CrossRef] [Green Version]
- Olivieri, D.; Sykora, M.M.; Sachidanandam, R.; Mechtler, K.; Brennecke, J. An in vivo RNAi assay identifies major genetic and cellular requirements for primary piRNA biogenesis in Drosophila. EMBO J. 2010, 29, 3301–3317. [Google Scholar] [CrossRef] [Green Version]
- Zamparini, A.L.; Davis, M.Y.; Malone, C.D.; Vieira, E.; Zavadil, J.; Sachidanandam, R.; Hannon, G.J.; Lehmann, R. Vreteno, a gonad-specific protein, is essential for germline development and primary piRNA biogenesis in Drosophila. Development 2011, 138, 4039–4050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handler, D.; Olivieri, D.; Novatchkova, M.; Gruber, F.S.; Meixner, K.; Mechtler, K.; Stark, A.; Sachidanandam, R.; Brennecke, J. A systematic analysis of DrosophilaTUDOR domain-containing proteins identifies Vreteno and the Tdrd12 family as essential primary piRNA pathway factors. EMBO J. 2011, 30, 3977–3993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pane, A.; Wehr, K.; Schüpbach, T. zucchini and squash Encode Two Putative Nucleases Required for rasiRNA Production in the Drosophila Germline. Dev. Cell 2007, 12, 851–862. [Google Scholar] [CrossRef] [Green Version]
- Olivieri, D.; Senti, K.-A.; Subramanian, S.L.; Sachidanandam, R.; Brennecke, J. The Cochaperone Shutdown Defines a Group of Biogenesis Factors Essential for All piRNA Populations in Drosophila. Mol. Cell 2012, 47, 954–969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Preall, J.B.; Czech, B.; Guzzardo, P.M.; Muerdter, F.; Hannon, G.J. shutdown is a component of the Drosophila piRNA biogenesis machinery. RNA 2012, 18, 1446–1457. [Google Scholar] [CrossRef] [Green Version]
- Horwich, M.D.; Li, C.; Matranga, C.; Vagin, V.; Farley, G.; Wang, P.; Zamore, P.D. The Drosophila RNA Methyltransferase, DmHen1, Modifies Germline piRNAs and Single-Stranded siRNAs in RISC. Curr. Biol. 2007, 17, 1265–1272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malone, C.D.; Brennecke, J.; Dus, M.; Stark, A.; McCombie, W.R.; Sachidanandam, R.; Hannon, G.J. Specialized piRNA Pathways Act in Germline and Somatic Tissues of the Drosophila Ovary. Cell 2009, 137, 522–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gunawardane, L.S.; Saito, K.; Nishida, K.M.; Miyoshi, K.; Kawamura, Y.; Nagami, T.; Siomi, H.; Siomi, M.C. A Slicer-Mediated Mechanism for Repeat-Associated siRNA 5’ End Formation in Drosophila. Science 2007, 315, 1587–1590. [Google Scholar] [CrossRef] [Green Version]
- Lim, A.K.; Kai, T. Unique germ-line organelle, nuage, functions to repress selfish genetic elements in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2007, 104, 6714–6719. [Google Scholar] [CrossRef] [Green Version]
- Patil, V.S.; Kai, T. Repression of Retroelements in Drosophila Germline via piRNA Pathway by the Tudor Domain Protein Tejas. Curr. Biol. 2010, 20, 724–730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cook, H.A.; Koppetsch, B.S.; Wu, J.; Theurkauf, W.E. The Drosophila SDE3 Homolog armitage Is Required for oskar mRNA Silencing and Embryonic Axis Specification. Cell 2004, 116, 817–829. [Google Scholar] [CrossRef] [Green Version]
- Han, B.W.; Zamore, P.D. PiRNAs. Curr. Biol. 2014, 24, R730–R733. [Google Scholar] [CrossRef] [Green Version]
- Calcagno, D.Q.; Mota, E.R.D.S.; Moreira, F.; De Sousa, S.B.M.; Burbano, R.R.; Assumpção, P.P.; Patel, V.; Preedy, V. Role of PIWI-Interacting RNA (piRNA) as Epigenetic Regulation. In Handbook of Nutrition, Diet, and Epigenetics; Springer: Berlin/Heidelberg, Germany, 2017; pp. 1–23. [Google Scholar]
- Gainetdinov, I.; Colpan, C.; Arif, A.; Cecchini, K.; Zamore, P.D. A Single Mechanism of Biogenesis, Initiated and Directed by PIWI Proteins, Explains piRNA Production in Most Animals. Mol. Cell 2018, 71, 775–790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayarpadikannan, S.; Kim, H.-S. The Impact of Transposable Elements in Genome Evolution and Genetic Instability and Their Implications in Various Diseases. Genom. Inform. 2014, 12, 98–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ng, K.W.; Anderson, C.S.; Marshall, E.A.; Minatel, B.C.; Enfield, K.S.S.; Saprunoff, H.L.; Lam, W.L.; Martinez, V.D. Piwi-interacting RNAs in cancer: Emerging functions and clinical utility. Mol. Cancer 2016, 15, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Pane, A.; Schüpbach, T. Cutoff and aubergine Mutations Result in Retrotransposon Upregulation and Checkpoint Activation in Drosophila. Curr. Biol. 2007, 17, 637–642. [Google Scholar] [CrossRef] [Green Version]
- Sarot, E.; Payen-Groschêne, G.; Bucheton, A.; Pélisson, A. Evidence for a piwi-Dependent RNA Silencing of the gypsy Endogenous Retrovirus by the Drosophila melanogaster flamenco Gene. Genetics 2004, 166, 1313–1321. [Google Scholar] [CrossRef] [Green Version]
- Girard, A.; Hannon, G.J. Conserved themes in small-RNA-mediated transposon control. Trends Cell Biol. 2008, 18, 136–148. [Google Scholar] [CrossRef] [Green Version]
- Biryukova, I.; Ye, T. Endogenous siRNAs and piRNAs derived from transposable elements and genes in the malaria vector mosquito Anopheles gambiae. BMC Genom. 2015, 16, 278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vagin, V.V.; Klenov, M.S.; Kalmykova, A.; Stolyarenko, A.D.; Kotelnikov, R.N.; Gvozdev, V.A. The RNA interference proteins and vasa locus are involved in the silencing of retrotransposons in the female germline of Drosophila melanogaster. RNA Biol. 2004, 1, 53–57. [Google Scholar] [CrossRef] [Green Version]
- Savitsky, M.; Kwon, D.; Georgiev, P.; Kalmykova, A.; Gvozdev, V. Telomere elongation is under the control of the RNAi-based mechanism in the Drosophila germline. Genes Dev. 2006, 20, 345–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pélisson, A.; Sarot, E.; Payen-Groscheêne, G.; Bucheton, A. A Novel Repeat-Associated Small Interfering RNA-Mediated Silencing Pathway Downregulates Complementary Sense Gypsy Transcripts in Somatic Cells of the Drosophila Ovary. J. Virol. 2006, 81, 1951–1960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shpiz, S.; Kwon, D.; Uneva, A.; Kim, M.; Klenov, M.; Rozovsky, Y.; Georgiev, P.; Savitsky, M.; Kalmykova, A. Characterization of Drosophila Telomeric Retroelement TAHRE: Transcription, Transpositions, and RNAi-based Regulation of Expression. Mol. Biol. Evol. 2007, 24, 2535–2545. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Vagin, V.V.; Lee, S.; Xu, J.; Ma, S.; Xi, H.; Seitz, H.; Horwich, M.D.; Syrzycka, M.; Honda, B.M.; et al. Collapse of Germline piRNAs in the Absence of Argonaute3 Reveals Somatic piRNAs in Flies. Cell 2009, 137, 509–521. [Google Scholar] [CrossRef] [Green Version]
- Carmell, M.A.; Girard, A.; Van De Kant, H.J.G.; Bourc’His, D.; Bestor, T.H.; De Rooij, D.G.; Hannon, G.J. MIWI2 Is Essential for Spermatogenesis and Repression of Transposons in the Mouse Male Germline. Dev. Cell 2007, 12, 503–514. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.A.; Yin, H.; Sweeney, S.; Raha, D.; Snyder, M.; Lin, H. A Major Epigenetic Programming Mechanism Guided by piRNAs. Dev. Cell 2013, 24, 502–516. [Google Scholar] [CrossRef] [Green Version]
- Yin, H.; Lin, H. An epigenetic activation role of Piwi and a Piwi-associated piRNA in Drosophila melanogaster. Nat. Cell Biol. 2007, 450, 304–308. [Google Scholar] [CrossRef]
- Watanabe, T.; Cui, X.; Yuan, Z.; Qi, H.; Lin, H. MIWI2 targets RNAs transcribed from piRNA-dependent regions to drive DNA methylation in mouse prospermatogonia. EMBO J. 2018, 37, e95329. [Google Scholar] [CrossRef] [PubMed]
- Pal-Bhadra, M.; Leibovtch, B.A.; Gandhi, S.G.; Rao, M.; Bhadra, U.; Birchler, J.A.; Elgin, S.C.R. Heterochromatic Silencing and HP1 Localization in Drosophila are Dependent on the RNAi Machinery. Science 2004, 303, 669–672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pal-Bhadra, M.; Bhadra, U.; Birchler, J.A. RNAi Related Mechanisms Affect Both Transcriptional and Posttranscriptional Transgene Silencing in Drosophila. Mol. Cell 2002, 9, 315–327. [Google Scholar] [CrossRef]
- Gomes, A.Q.; Nolasco, S.; Soares, H. Non-Coding RNAs: Multi-Tasking Molecules in the Cell. Int. J. Mol. Sci. 2013, 14, 16010–16039. [Google Scholar] [CrossRef]
- Rajasethupathy, P.; Antonov, I.; Sheridan, R.; Frey, S.; Sander, C.; Tuschl, T.; Kandel, E.R. A Role for Neuronal piRNAs in the Epigenetic Control of Memory-Related Synaptic Plasticity. Cell 2012, 149, 693–707. [Google Scholar] [CrossRef] [Green Version]
- Kuramochi-Miyagawa, S.; Watanabe, T.; Gotoh, K.; Totoki, Y.; Toyoda, A.; Ikawa, M.; Asada, N.; Kojima, K.; Yamaguchi, Y.; Ijiri, T.W.; et al. DNA methylation of retrotransposon genes is regulated by Piwi family members MILI and MIWI2 in murine fetal testes. Genes Dev. 2008, 22, 908–917. [Google Scholar] [CrossRef] [Green Version]
- Fu, A.; Jacobs, D.I.; Zhu, Y. Epigenome-wide analysis of piRNAs in gene-specific DNA methylation. RNA Biol. 2014, 11, 1301–1312. [Google Scholar] [CrossRef] [Green Version]
- Zoch, A.; Auchynnikava, T.; Berrens, R.V.; Kabayama, Y.; Schopp, T.; Heep, M.; Vasiliauskaite, L.; Pere-Rico, Y.A.; Cook, A.G.; Shkumatava, A.; et al. SPOCD1 is an essential executor of piRNA-directed de novo DNA methylation. Nature 2020, 584, 635–639. [Google Scholar] [CrossRef]
- Schöpp, T.; Zoch, A.; Berrens, R.V.; Auchynnikava, T.; Kabayama, Y.; Vasiliauskaitė, L.; Rappsilber, J.; Allshire, R.C.; O’Carroll, D. TEX15 is an essential executor of MIWI2-directed transposon DNA methylation and silencing. Nat. Commun. 2020, 11, 1–8. [Google Scholar] [CrossRef]
- Mohn, F.; Handler, D.; Brennecke, J. piRNA-guided slicing specifies transcripts for Zucchini-dependent, phased piRNA biogenesis. Science 2015, 348, 812–817. [Google Scholar] [CrossRef] [Green Version]
- Robine, N.; Lau, N.C.; Balla, S.; Jin, Z.; Okamura, K.; Kuramochi-Miyagawa, S.; Blower, M.D.; Lai, E.C. A Broadly Conserved Pathway Generates 3′UTR-Directed Primary piRNAs. Curr. Biol. 2009, 19, 2066–2076. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishida, K.M.; Saito, K.; Mori, T.; Kawamura, Y.; Nagami-Okada, T.; Inagaki, S.; Siomi, H.; Siomi, M.C. Gene silencing mechanisms mediated by Aubergine piRNA complexes in Drosophila male gonad. RNA 2007, 13, 1911–1922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vourekas, A.; Zheng, Q.; Alexiou, P.; Maragkakis, M.; Kirino, Y.; Gregory, B.D.; Mourelatos, Z. Mili and Miwi target RNA repertoire reveals piRNA biogenesis and function of Miwi in spermiogenesis. Nat. Struct. Mol. Biol. 2012, 19, 773–781. [Google Scholar] [CrossRef] [Green Version]
- Lee, E.J.; Banerjee, S.; Zhou, H.; Jammalamadaka, A.; Arcila, M.; Manjunath, B.S.; Kosik, K.S. Identification of piRNAs in the central nervous system. RNA 2011, 17, 1090–1099. [Google Scholar] [CrossRef] [Green Version]
- Esposito, T.; Magliocca, S.; Formicola, D.; Gianfrancesco, F. piR_015520 Belongs to Piwi-Associated RNAs Regulates Expression of the Human Melatonin Receptor 1A Gene. PLoS ONE 2011, 6, e22727. [Google Scholar] [CrossRef]
- Temme, C.; Simonelig, M.; Wahle, E. Deadenylation of mRNA by the CCR4—NOT complex in Drosophila: Molecular and developmental aspects. Front. Genet. 2014, 5, 143. [Google Scholar] [CrossRef] [Green Version]
- Filipowicz, W.; Bhattacharyya, S.N.; Sonenberg, N. Mechanisms of post-transcriptional regulation by microRNAs: Are the answers in sight? Nat. Rev. Genet. 2008, 9, 102–114. [Google Scholar] [CrossRef]
- Grivna, S.T.; Pyhtila, B.; Lin, H. MIWI associates with translational machinery and PIWI-interacting RNAs (piRNAs) in regulating spermatogenesis. Proc. Natl. Acad. Sci. USA 2006, 103, 13415–13420. [Google Scholar] [CrossRef] [Green Version]
- Ramat, A.; Garcia-Silva, M.-R.; Jahan, C.; Naït-Saïdi, R.; Dufourt, J.; Garret, C.; Chartier, A.; Cremaschi, J.; Patel, V.; Decourcelle, M.; et al. The PIWI protein Aubergine recruits eIF3 to activate translation in the germ plasm. Cell Res. 2020, 30, 421–435. [Google Scholar] [CrossRef] [Green Version]
- Unhavaithaya, Y.; Hao, Y.; Beyret, E.; Yin, H.; Kuramochi-Miyagawa, S.; Nakano, T.; Lin, H. MILI, a PIWI-interacting RNA-binding Protein, Is Required for Germ Line Stem Cell Self-renewal and Appears to Positively Regulate Translation. J. Biol. Chem. 2009, 284, 6507–6519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aravin, A.A.; Lagos-Quintana, M.; Yalcin, A.; Zavolan, M.; Marks, D.; Snyder, B.; Gaasterland, T.; Meyer, J.; Tuschl, T. The Small RNA Profile during Drosophila melanogaster Development. Dev. Cell 2003, 5, 337–350. [Google Scholar] [CrossRef] [Green Version]
- Lau, N.C.; Seto, A.G.; Kim, J.; Kuramochi-Miyagawa, S.; Nakano, T.; Bartel, D.P.; Kingston, R.E. Characterization of the piRNA Complex from Rat Testes. Science 2006, 313, 363–367. [Google Scholar] [CrossRef] [Green Version]
- Meister, G.; Tuschl, T. Mechanisms of gene silencing by double-stranded RNA. Nat. Cell Biol. 2004, 431, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Doudna, J.A. A three-dimensional view of the molecular machinery of RNA interference. Nat. Cell Biol. 2008, 457, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Kim, V.N.; Nam, J.-W. Genomics of microRNA. Trends Genet. 2006, 22, 165–173. [Google Scholar] [CrossRef]
- Liu, B.; Li, J.; Cairns, M.J. Identifying miRNAs, targets and functions. Brief. Bioinform. 2014, 15, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Dana, H.; Chalbatani, G.M.; Mahmoodzadeh, H.; Karimloo, R.; Rezaiean, O.; Moradzadeh, A.; Mehmandoost, N.; Moazzen, F.; Mazraeh, A.; Marmari, V.; et al. Molecular Mechanisms and Biological Functions of siRNA. Int. J. Biomed. Sci. IJBS 2017, 13, 48–57. [Google Scholar]
- Lam, J.K.W.; Chow, M.Y.T.; Zhang, Y.; Leung, S.W.S. siRNA Versus miRNA as Therapeutics for Gene Silencing. Mol. Ther. Nucleic Acids 2015, 4, e252. [Google Scholar] [CrossRef] [Green Version]
- Bartel, D.P. MicroRNAs: Target Recognition and Regulatory Functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [Green Version]
- Martinez, N.J.; Walhout, A.J.M. The interplay between transcription factors and microRNAs in genome-scale regulatory networks. Bioessays 2009, 31, 435–445. [Google Scholar] [CrossRef] [Green Version]
- Place, R.F.; Li, L.-C.; Pookot, D.; Noonan, E.J.; Dahiya, R. MicroRNA-373 induces expression of genes with complementary promoter sequences. Proc. Natl. Acad. Sci. USA 2008, 105, 1608–1613. [Google Scholar] [CrossRef] [Green Version]
- Ni, W.J.; Leng, X.M. Dynamic miRNA-mRNA paradigms: New faces of miRNAs. Biochem. Biophys. Rep. 2015, 4, 337–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Catalanotto, C.; Cogoni, C.; Zardo, G. MicroRNA in Control of Gene Expression: An Overview of Nuclear Functions. Int. J. Mol. Sci. 2016, 17, 1712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellison, M.; Mittal, M.; Chaudhuri, M.; Chaudhuri, G.; Misra, S. The role of the redox/miR-6855-3p/PRDX5A axis in reversing SLUG-mediated BRCA2 silencing in breast cancer cells. Cell Commun. Signal. 2020, 18, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Lei, C.; He, Q.; Pan, Z.; Xiao, D.; Tao, Y. Nuclear functions of mammalian MicroRNAs in gene regulation, immunity and cancer. Mol. Cancer 2018, 17, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Aravin, A.A.; Hannon, G.J.; Brennecke, J. The Piwi-piRNA pathway provides an adaptive defense in the transposon arms race. Science 2007, 318, 761–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gou, L.-T.; Dai, P.; Yang, J.-H.; Xue, Y.; Hu, Y.-P.; Zhou, Y.; Kang, J.-Y.; Wang, X.; Li, H.; Hua, M.-M.; et al. Pachytene piRNAs instruct massive mRNA elimination during late spermiogenesis. Cell Res. 2014, 24, 680–700. [Google Scholar] [CrossRef]
- Rouget, C.; Papin, C.; Boureux, A.; Meunier, A.-C.; Franco, B.; Robine, N.; Lai, E.C.; Pelisson, A.; Simonelig, M. Maternal mRNA deadenylation and decay by the piRNA pathway in the early Drosophila embryo. Nature 2010, 467, 1128–1132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Kolb, F.A.; Brondani, V.; Billy, E.; Filipowicz, W. Human Dicer preferentially cleaves dsRNAs at their termini without a requirement for ATP. EMBO J. 2002, 21, 5875–5885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rana, T.M. Illuminating the silence: Understanding the structure and function of small RNAs. Nat. Rev. Mol. Cell Biol. 2007, 8, 23–36. [Google Scholar] [CrossRef]
- Orban, T.I.; Izaurralde, E. Decay of mRNAs targeted by RISC requires XRN1, the Ski complex, and the exosome. RNA 2005, 11, 459–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La Greca, A.; Scarafía, M.A.; Cañás, M.C.H.; Pérez, N.; Castañeda, S.; Colli, C.; Möbbs, A.M.; Velazque, N.L.S.; Neiman, G.; Garate, X.; et al. PIWI-interacting RNAs are differentially expressed during cardiac differentiation of human pluripotent stem cells. PLoS ONE 2020, 15, e0232715. [Google Scholar] [CrossRef] [PubMed]
- Rajan, K.S.; Ramasamy, S. Retrotransposons and piRNA: The missing link in central nervous system. Neurochem. Int. 2014, 77, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Vella, S.; Gallo, A.; Nigro, A.L.; Galvagno, D.; Raffa, G.M.; Pilato, M.; Conaldi, P.G. PIWI-interacting RNA (piRNA) signatures in human cardiac progenitor cells. Int. J. Biochem. Cell Biol. 2016, 76, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Di Giacomo, M.; Comazzetto, S.; Saini, H.; De Fazio, S.; Carrieri, C.; Morgan, M.; Vasiliauskaite, L.; Benes, V.; Enright, A.J.; O’Carroll, D. Multiple Epigenetic Mechanisms and the piRNA Pathway Enforce LINE1 Silencing during Adult Spermatogenesis. Mol. Cell 2013, 50, 601–608. [Google Scholar] [CrossRef] [Green Version]
- Lucchinetti, E.; Feng, J.; Da Silva, R.; Tolstonog, G.V.; Schaub, M.C.; Schumann, G.G.; Zaugg, M. Inhibition of LINE-1 expression in the heart decreases ischemic damage by activation of Akt/PKB signaling. Physiol. Genom. 2006, 25, 314–324. [Google Scholar] [CrossRef] [Green Version]
- Sugden, P.H.; Fuller, S.J.; Weiss, S.C.; Clerk, A. Glycogen synthase kinase 3 (GSK3) in the heart: A point of integration in hypertrophic signalling and a therapeutic target? A critical analysis. Br. J. Pharmacol. 2008, 153, S137–S153. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, R.; Honda, S.; Kirino, Y. PIWI Expression and Function in Cancer. Front. Genet. 2012, 3, 204. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Sun, Y.; Guo, J.; Ma, H.; Li, J.; Dong, B.; Jin, G.; Zhang, J.; Wu, J.; Meng, L.; et al. Expression of hiwi gene in human gastric cancer was associated with proliferation of cancer cells. Int. J. Cancer 2006, 118, 1922–1929. [Google Scholar] [CrossRef]
- Lee, T.I.; Jenner, R.G.; Boyer, L.A.; Guenther, M.G.; Levine, S.S.; Kumar, R.M.; Chevalier, B.; Johnstone, S.E.; Cole, M.F.; Isono, K.-I.; et al. Control of Developmental Regulators by Polycomb in Human Embryonic Stem Cells. Cell 2006, 125, 301–313. [Google Scholar] [CrossRef] [Green Version]
- Tan, L.; Mai, D.; Zhang, B.; Jiang, X.; Zhang, J.; Bai, R.; Ye, Y.; Li, M.; Pan, L.; Su, J.; et al. PIWI-interacting RNA-36712 restrains breast cancer progression and chemoresistance by interaction with SEPW1 pseudogene SEPW1P RNA. Mol. Cancer 2019, 18, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Hashim, A.; Rizzo, F.; Marchese, G.; Ravo, M.; Tarallo, R.; Nassa, G.; Giurato, G.; Santamaria, G.; Cordella, A.; Cantarella, C.; et al. RNA sequencing identifies specific PIWI-interacting small non-coding RNA expression patterns in breast cancer. Oncotarget 2014, 5, 9901–9910. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Luo, Y.; Gao, Y.; Yang, Y.; Wang, Y.; Xu, Y.; Tan, S.; Zhang, Y.; Duan, J. piR-651 promotes tumor formation in non-small cell lung carcinoma through the upregulation of cyclin D1 and CDK4. Int. J. Mol. Med. 2016, 38, 927–936. [Google Scholar] [CrossRef] [Green Version]
- Mendillo, M.L.; Santagata, S.; Koeva, M.; Bell, G.W.; Hu, R.; Tamimi, R.M.; Fraenkel, E.; Ince, T.A.; Whitesell, L.; Lindquist, S. HSF1 Drives a Transcriptional Program Distinct from Heat Shock to Support Highly Malignant Human Cancers. Cell 2012, 150, 549–562. [Google Scholar] [CrossRef] [Green Version]
- Yin, J.; Jiang, X.; Qi, W.; Ji, C.; Xie, X.; Zhang, D.; Cui, Z.; Wang, C.; Bai, Y.; Wang, J.; et al. piR-823 contributes to colorectal tumorigenesis by enhancing the transcriptional activity of HSF 1. Cancer Sci. 2017, 108, 1746–1756. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Tomizawa, S.-I.; Mitsuya, K.; Totoki, Y.; Yamamoto, Y.; Kuramochi-Miyagawa, S.; Iida, N.; Hoki, Y.; Murphy, P.J.; Toyoda, A.; et al. Role for piRNAs and Noncoding RNA in de Novo DNA Methylation of the Imprinted Mouse Rasgrf1 Locus. Science 2011, 332, 848–852. [Google Scholar] [CrossRef] [Green Version]
- Shi, S.; Yang, Z.Z.; Liu, S.; Yang, F.; Lin, H. PIWIL1 promotes gastric cancer via a piRNA-independent mechanism. Proc. Natl. Acad. Sci. USA 2020, 117, 22390–22401. [Google Scholar] [CrossRef]
- Gambichler, T.; Kohsik, C.; Höh, A.-K.; Lang, K.; Käfferlein, H.U.; Brüning, T.; Stockfleth, E.; Stücker, M.; Dreißigacker, M.; Sand, M. Expression of PIWIL3 in primary and metastatic melanoma. J. Cancer Res. Clin. Oncol. 2017, 143, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Leng, X.; Ma, J.; Liu, Y.; Shen, S.; Yu, H.; Zheng, J.; Liu, X.; Liu, L.; Chen, J.; Zhao, L.; et al. Mechanism of piR-DQ590027/MIR17HG regulating the permeability of glioma conditioned normal BBB. J. Exp. Clin. Cancer Res. 2018, 37, 246. [Google Scholar] [CrossRef]
- Jacobs, D.I.; Qin, Q.; Fu, A.; Chen, Z.; Zhou, J.; Zhu, Y. piRNA-8041 is downregulated in human glioblastoma and suppresses tumor growth in vitro and in vivo. Oncotarget 2018, 9, 37616–37626. [Google Scholar] [CrossRef] [Green Version]
- Shen, S.; Yu, H.; Liu, X.; Liu, Y.; Zheng, J.; Wang, P.; Gong, W.; Chen, J.; Zhao, L.; Xue, Y. PIWIL1/piRNA-DQ593109 Regulates the Permeability of the Blood-Tumor Barrier via the MEG3/miR-330-5p/RUNX3 Axis. Mol. Ther. Nucleic Acids 2018, 10, 412–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, M.; Viginier, B.; Saint-Michel, E.; Arnaud, F.; Ratinier, M.; Fablet, M. Viral infection impacts transposable element transcript amounts inDrosophila. Proc. Natl. Acad. Sci. USA 2020, 117, 12249–12257. [Google Scholar] [CrossRef] [PubMed]
- Belicard, T.; Jareosettasin, P.; Sarkies, P. The piRNA pathway responds to environmental signals to establish intergenerational adaptation to stress. BMC Biol. 2018, 16, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-H.; Aliyari, R.; Li, W.-X.; Li, H.-W.; Kim, K.; Carthew, R.; Atkinson, P.; Ding, S.-W. RNA Interference Directs Innate Immunity Against Viruses in Adult Drosophila. Science 2006, 312, 452–454. [Google Scholar] [CrossRef] [Green Version]
- Vogel, E.; Santos, D.; Mingels, L.; Verdonckt, T.-W.; Broeck, J.V. RNA Interference in Insects: Protecting Beneficials and Controlling Pests. Front. Physiol. 2019, 9, 1912. [Google Scholar] [CrossRef] [Green Version]
- Morazzani, E.M.; Wiley, M.R.; Murreddu, M.G.; Adelman, Z.N.; Myles, K.M. Production of virus-derived ping-pong-dependent piRNA-like small RNAs in the mosquito soma. PLoS Pathog. 2012, 8, e1002470. [Google Scholar] [CrossRef]
- Ter Horst, A.M.; Nigg, J.C.; Dekker, F.M.; Falk, B.W. Endogenous Viral Elements Are Widespread in Arthropod Genomes and Commonly Give Rise to PIWI-Interacting RNAs. J. Virol. 2018, 93. [Google Scholar] [CrossRef] [Green Version]
- Yu, T.; Koppetsch, B.; Chappell, K.; Pagliarani, S.; Johnston, S.; Silverstein, N.J.; Luban, J.; Weng, Z.; Thauerkauf, W.E. The PiRNA Response to Retroviral Invasion of the Koala Genome. Cell 2019, 179, 632–643. [Google Scholar] [CrossRef]
- Pinto, P.; Da Silva, M.B.; Moreira, F.C.; Bouth, R.C.; Gobbo, A.R.; Sandoval, T.V.; Ribeiro-Dos-Santos, A.M.; Vidal, A.F.; Barreto, J.G.; Santos, S.; et al. Leprosy piRnome: Exploring new possibilities for an old disease. Sci. Rep. 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Arun, A.; Rayford, K.J.; Cooley, A.; Rachakonda, G.; Villalta, F.; Pratap, S.; Lima, M.F.; Sheibani, N.; Nde, P.N. Thrombospondin-1 Plays an Essential Role in Yes-Associated Protein Nuclear Translocation during the Early Phase of Trypanosoma cruzi Infection in Heart Endothelial Cells. Int. J. Mol. Sci. 2020, 21, 4912. [Google Scholar] [CrossRef]
- Suman, S.; Rachakonda, G.; Mandape, S.; Sakhare, S.S.; Villalta, F.; Pratap, S.; Lima, M.F.; Nde, P.N. Phospho-proteomic analysis of primary human colon epithelial cells during the early Trypanosoma cruzi infection phase. PLoS Negl. Trop. Dis. 2018, 12, e0006792. [Google Scholar] [CrossRef]
- Udoko, A.N.; Johnson, C.A.; Dykan, A.; Rachakonda, G.; Villalta, F.; Mandape, S.; Lima, M.F.; Pratap, S.; Nde, P.N. Early Regulation of Profibrotic Genes in Primary Human Cardiac Myocytes by Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2016, 10, e0003747. [Google Scholar] [CrossRef] [Green Version]
- Ferrao, P.M.; D’Avila-Levy, C.M.; Araujo-Jorge, T.C.; Degrave, W.M.; Da Silva, A.G.; Garzoni, L.R.; Lima, A.P.; Feige, J.J.; Baily, S.; Mendonca-Lima, L.; et al. Cruzipain Activates Latent TGF-beta from Host Cells during T. cruzi Invasion. PLoS ONE 2015, 10, e0124832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrao, P.M.; De Oliveira, F.L.; Degrave, W.M.; Araujo-Jorge, T.C.; Mendonca-Lima, L.; Waghabi, M.C. A phosphoproteomic approach towards the understanding of the role of TGF-beta in Trypanosoma cruzi biology. PLoS ONE 2012, 7, e38736. [Google Scholar] [CrossRef] [Green Version]
- Araujo-Jorge, T.C.; Waghabi, M.C.; Soeiro, M.D.N.C.; Keramidas, M.; Bailly, S.; Feige, J.J. Pivtal role for TGF-beta in infectious heart disease: The case of Trypanosoma cruzi infection and consequent Chagasic myocardiopathy. Cytokine Growth Fact. Rev. 2008, 19, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Bourajjaj, M.; Armand, A.-S.; Martins, P.A.D.C.; Weijts, B.; Van Der Nagel, R.; Heeneman, S.; Wehrens, X.H.; De Windt, L.J. NFATc2 Is a Necessary Mediator of Calcineurin-dependent Cardiac Hypertrophy and Heart Failure. J. Biol. Chem. 2008, 283, 22295–22303. [Google Scholar] [CrossRef] [Green Version]
- Sarikhani, M.; Maity, S.; Mishra, S.; Jain, A.; Tamta, A.K.; Ravi, V.; Kondapalli, M.S.; Desingu, P.A.; Khan, D.; Kumar, S.; et al. SIRT2 deacetylase represses NFAT transcription factor to maintain cardiac homeostasis. J. Biol. Chem. 2018, 293, 5281–5294. [Google Scholar] [CrossRef] [Green Version]
- Molkentin, J.D. Calcineurin? NFAT signaling regulates the cardiac hypertrophic response in coordination with the MAPKs. Cardiovasc. Res. 2004, 63, 467–475. [Google Scholar] [CrossRef]

| Disease/ Pathology | piRNA | Upregulated/ Downregulated | Gene Target | Reference |
|---|---|---|---|---|
| Cardiac Hypertrophy/Cardiac Failure | piR-141981/cardiac-hypertrophy-associated piRNA (CHAPIR) | Upregulated in mice that underwent transverse aortic construction surgery | Myh7, Anp, Bnp, and PARP10 mRNA | [22] |
| Cardiac Hypertrophy/Cardiac Failure | piR-13375 and piR-106654 | Downregulated in mice that underwent transverse aortic construction surgery | Not identified | [22] |
| Cardiac Failure | has-piR-006426 and has-piR-0200009 | Downregulated in the serum exosomes of patients experiencing heart failure | Not identified | [23] |
| Myocardial Infarction | piR_2106027 | Upregulated in the serum of patients with Troponin-I negative myocardial infarction | Troponin-I | [24] |
| Cardiac Tissue Regeneration/ Angiogenesis | piR-55490 | Not specified | mTOR | [25,26] |
| Breast Cancer | piR-4987 | Upregulation promotes lymph node metastasis | Not identified | [27] |
| Breast Cancer | piR-021285 | Not specified | Induces methylation several genes related to breast cancer in vitro | [28] |
| Lung Cancer | piR-L-163 | Upregulated upon cisplatin treatment in vitro and in vivo | Regulates ezrin-radixin-moesin (ERM) proteins | [29] |
| Lung Cancer | piR-651 | Upregulated | Promotes the upregulation of cyclins and CDKs | [30] |
| Colorectal Cancer | piR-1245 | Upregulation promotes colorectal cancer tumor growth |
Suppresses multiple tumor suppressors such as ATF3, DUSP1, and SESN2 | [31] |
| Gastric Cancer | PiRNA-823 | Upregulated in patients with multiple myeloma | Regulates de novo DNA methylases DNMT3A and 3B and angiogenesis | [32] |
| Bladder Cancer | piRNA-DQ594040 | Upregulated in tissue from patients with bladder cancer | May regulate Tumor Necrosis Factor Superfamily Member 4 (TNFSF4) | [33] |
| Liver Cancer | piR-Hep1 | Upregulated in hepatocellular carcinoma samples | Upregulates PI3K/AKT | [34,35] |
| Pancreatic Cancer | piR-017061 | Downregulated in pancreatic ductal adenocarcinoma tissue | Not identified | [36] |
| Pancreatic Cancer | piR-016658 and piR-001311 | Have been detected in the extracellular RNA of pancreatic cancer patients | Not identified | [36] |
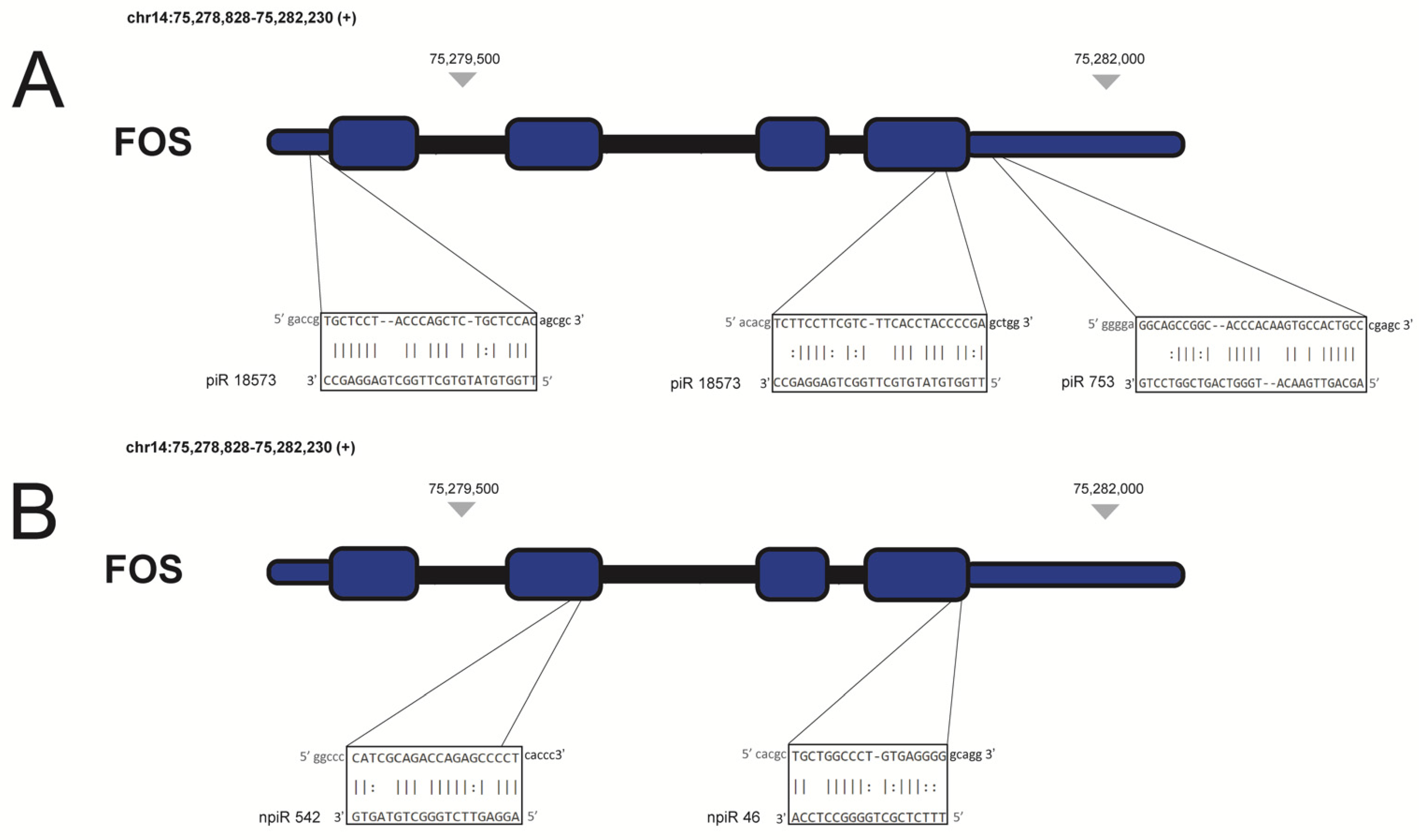
| Trypanosoma cruzi infection in cardiac myocytes | piR-753 and piR-18573 | Upregulate d during early phase of infection | FOS | [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rayford, K.J.; Cooley, A.; Rumph, J.T.; Arun, A.; Rachakonda, G.; Villalta, F.; Lima, M.F.; Pratap, S.; Misra, S.; Nde, P.N. piRNAs as Modulators of Disease Pathogenesis. Int. J. Mol. Sci. 2021, 22, 2373. https://doi.org/10.3390/ijms22052373
Rayford KJ, Cooley A, Rumph JT, Arun A, Rachakonda G, Villalta F, Lima MF, Pratap S, Misra S, Nde PN. piRNAs as Modulators of Disease Pathogenesis. International Journal of Molecular Sciences. 2021; 22(5):2373. https://doi.org/10.3390/ijms22052373
Chicago/Turabian StyleRayford, Kayla J., Ayorinde Cooley, Jelonia T. Rumph, Ashutosh Arun, Girish Rachakonda, Fernando Villalta, Maria F. Lima, Siddharth Pratap, Smita Misra, and Pius N. Nde. 2021. "piRNAs as Modulators of Disease Pathogenesis" International Journal of Molecular Sciences 22, no. 5: 2373. https://doi.org/10.3390/ijms22052373







