A New Family of Benzo[h]Chromene Based Azo Dye: Synthesis, In-Silico and DFT Studies with In Vitro Antimicrobial and Antiproliferative Assessment
Abstract
:1. Introduction
2. Results and Discussion
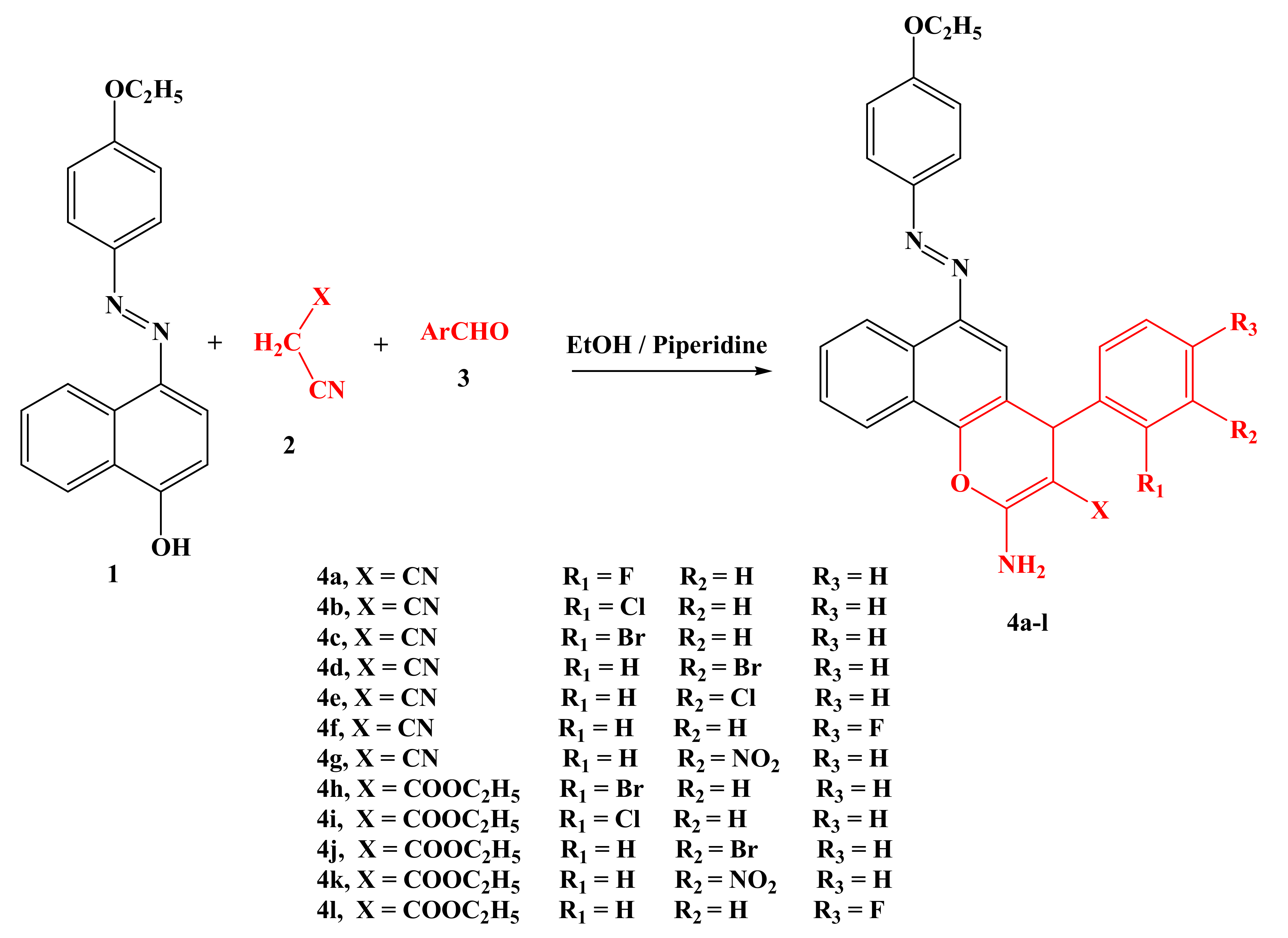
2.1. Synthesis and Characterization
2.2. UV–Visible and Halochromism Studies
2.3. Calculated Frontier Molecular Orbitals (FMOs)
2.4. Biological Screening
2.4.1. Antimicrobial Screening
2.4.2. Cytotoxic Screening
2.5. Computational Studies and SAR Analysis
2.5.1. Docking Studies
2.5.2. Molecular Descriptors-Based SAR (Structure–Activity Relationship) Analysis
3. Materials and Methods
3.1. Materials and Instrumentation
3.2. Biological Studies
3.2.1. Antimicrobial Screening
3.2.2. Cytotoxic Screening
3.3. Molecular Modeling
3.4. Computational Methods and Calculations
3.5. Synthesis
3.5.1. General Procedure for the Synthesis of 2-Amino-6-(4-Ethoxyphenylazo)-4(-Phenyl)-4H-Benzo[h]Chromene Derivatives
3.5.2. 2-Amino-6-(4-Ethoxyphenylazo)-4-(2-Fluoro-Phenyl)-4H-Benzo[h]Chromene-3-Carbonitrile (4a)
3.5.3. 2-Amino-6-(4-Ethoxyphenylazo)-4-(2-Chloro-Phenyl)-4H-Benzo[h]Chromene-3-Carbonitrile (4b)
3.5.4. 2-Amino-6-(4-Ethoxyphenylazo)-4-(2-Bromo-Phenyl)-4H-Benzo[h]Chromene-3-Carbonitrile (4c)
3.5.5. 2-Amino-6-(4-Ethoxyphenylazo)-4-(3-Bromo-Phenyl)-4H-Benzo[h]Chromene-3-Carbonitrile (4d)
3.5.6. 2-Amino-6-(4-Ethoxyphenylazo)-4-(3-Chloro-Phenyl)-4H-Benzo[h]Chromene-3-Carbonitrile (4e)
3.5.7. 2-Amino-6-(4-Ethoxyphenylazo)-4-(4-Fluoro-Phenyl)-4H-Benzo [h] Chromene-3-Carbonitrile (4f)
3.5.8. 2-Amino-6-(4-Ethoxyphenylazo)-4-(3-Nitro-Phenyl)-4H-Benzo[h]Chromene-3-Carbonitrile (4g)
3.5.9. Ethyl-2-Amino-6-(4-Ethoxyphenylazo)-4-(2-Bromo-Phenyl)-4H-Benzo[h]Chromene -3-Carboxylate (4h)
3.5.10. Ethyl-2-Amino-6-(4-Ethoxyphenylazo)-4-(2-Chloro-Phenyl)-4H-Benzo[h]Chromene -3-Carboxylate (4i)
3.5.11. Ethyl-2-Amino-6-(4-Ethoxyphenylazo)-4-(3-Bromo-Phenyl)-4H-Benzo[h]Chromene-3-Carboxylate (4j)
3.5.12. Ethyl-2-Amino-6-(4-Ethoxyphenylazo)-4-(3-Nitro-Phenyl)-4H-Benzo[h]Chromene-3-Carboxylate (4k)
3.5.13. Ethyl-2-Amino-6-(4-Ethoxyphenylazo)-4-(4-Fluoro-Phenyl)-4H-Benzo[h]Chromene-3-Carboxylate (4l)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gupta, S.M.; Mania-Pramanik, J. RETRACTED ARTICLE: Molecular mechanisms in progression of HPV-associated cervical carcinogenesis. J. Biomed. Sci. 2019, 26, 1–19. [Google Scholar] [CrossRef]
- Lee, J.; Pyo, H.K.; Park, M.-O.; Kim, H.; Yoon, S.Y.; Han, J.; Jung, N.; Yun, S.; Kim, J.; Chae, Y.S. Top publications by reads. J. Stroke Cerebrovasc. Dis. 2018, 27. [Google Scholar]
- Gourdeau, H.; Leblond, L.; Hamelin, B.; Desputeau, C.; Dong, K.; Kianicka, I.; Custeau, D.; Boudreau, C.; Geerts, L.; Cai, S.-X. Antivascular and antitumor evaluation of 2-amino-4-(3-bromo-4, 5-dimethoxy-phenyl)-3-cyano-4H-chromenes, a novel series of anticancer agents. Mol. Cancer Ther. 2004, 3, 1375–1384. [Google Scholar]
- Sangani, C.B.; Shah, N.M.; Patel, M.P.; Patel, R.G. Microwave assisted synthesis of novel 4H-chromene derivatives bearing phenoxypyrazole and their antimicrobial activity assess. J. Serb. Chem. Soc. 2012, 77, 1165–1174. [Google Scholar] [CrossRef]
- Thareja, S.; Verma, A.; Kalra, A.; Gosain, S.; Rewatkar, P.V.; Kokil, G.R. Novel chromeneimidazole derivatives as antifungal compounds: Synthesis and in vitro evaluation. Acta Pol. Pharm. 2010, 67, 423–427. [Google Scholar]
- Mori, J.; Iwashima, M.; Takeuchi, M.; Saito, H. A synthetic study on antiviral and antioxidative chromene derivative. Chem. Pharm. Bull. 2006, 54, 391–396. [Google Scholar] [CrossRef] [Green Version]
- Kamdar, N.R.; Haveliwala, D.D.; Mistry, P.T.; Patel, S.K. Synthesis and evaluation of in vitro antitubercular activity and antimicrobial activity of some novel 4H-chromeno [2, 3-d] pyrimidine via 2-amino-4-phenyl-4H-chromene-3-carbonitriles. Med. Chem. Res. 2011, 20, 854–864. [Google Scholar] [CrossRef]
- Gupta, S.; Kumar, N.; Kumar, S.; Dudhe, R.; Sharma, P. 3-Hydroxy-2-(substituted phenyl)-4H-chromen-4-one derivatives-synthesis, spectral characterization and pharmacological screening. IJT A 2012, 7, 1–8. [Google Scholar]
- Bhat, M.A.; Siddiqui, N.; Khan, S.A. Synthesis of novel 3-(4-acetyl-5H/methyl-5-substituted phenyl-4, 5-dihydro-1, 3, 4-oxadiazol-2-yl)-2H-chromen-2-ones as potential anticonvulsant agents. Acta Pol. Pharm. 2008, 65, 235–239. [Google Scholar]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. BioMed Res. Int. 2013, 2013, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Mccarroll, J.; Parker, A.; Kavallaris, M. Microtubules and their role in cellular stress in cancer. Front. Oncol. 2014, 4, 3389. [Google Scholar]
- Ren, Q.; Siau, W.Y.; Du, Z.; Zhang, K.; Wang, J. Expeditious assembly of a 2-amino-4H-chromene skeleton by using an enantioselective mannich intramolecular ring cyclization–tautomerization cascade sequence. Chem. A Eur. J. 2011, 17, 7781–7785. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Xu, J.; Kanojia, R.M.; Du, F.; Jian-Zhong, G.; Pacia, E.; Lai, M.-T.; Musto, A.; Allan, G.; Reuman, M. Identification and Structure–Activity Relationships of Chromene-Derived Selective Estrogen Receptor Modulators for Treatment of Postmenopausal Symptoms. J. Med. Chem. 2009, 52, 7544–7569. [Google Scholar] [CrossRef] [PubMed]
- Alblewi, F.F.; Okasha, R.M.; Eskandrani, A.A.; Afifi, T.H.; Mohamed, H.M.; Halawa, A.H.; Fouda, A.M.; Al-Dies, A.-A.M.; Mora, A.; El-Agrody, A.M. Design and synthesis of novel heterocyclic-based 4H-benzo [h] chromene moieties: Targeting antitumor caspase 3/7 activities and cell cycle analysis. Molecules 2019, 24, 1060. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, H.E.; El-Nassag, M.A.; Hassan, A.H.; Mohamed, H.M.; Halawa, A.H.; Okasha, R.M.; Ihmaid, S.; Abd El-Gilil, S.M.; Khattab, E.S.; Fouda, A.M. Developing lipophilic aromatic halogenated fused systems with specific ring orientations, leading to potent anticancer analogs and targeting the c-Src Kinase enzyme. J. Mol. Struct. 2019, 1186, 212–223. [Google Scholar] [CrossRef]
- Alblewi, F.F.; Okasha, R.M.; Hritani, Z.M.; Mohamed, H.M.; El-Nassag, M.A.; Halawa, A.H.; Mora, A.; Fouda, A.M.; Assiri, M.A.; Al-Dies, A.-A.M. Antiproliferative effect, cell cycle arrest and apoptosis generation of novel synthesized anticancer heterocyclic derivatives based 4H-benzo [h] chromene. Bioorg. Chem. 2019, 87, 560–571. [Google Scholar] [CrossRef]
- Okasha, R.M.; Alblewi, F.F.; Afifi, T.H.; Naqvi, A.; Fouda, A.M.; Al-Dies, A.-A.M.; El-Agrody, A.M. Design of new benzo [h] chromene derivatives: Antitumor activities and structure-activity relationships of the 2, 3-positions and fused rings at the 2, 3-positions. Molecules 2017, 22, 479. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Aziz, A.; El-Ghezlani, E.; Elaasser, M.; Afifi, T.; Okasha, R. First Example of Cationic Cyclopentadienyliron Based Chromene Complexes and Polymers: Synthesis, Characterization, and Biological Applications. J. Inorg. Organomet. Polym. Mater. 2020, 30, 131–146. [Google Scholar] [CrossRef]
- Khafagy, M.M.; Abd El-Wahab, A.H.; Eid, F.A.; El-Agrody, A.M. Synthesis of halogen derivatives of benzo [h] chromene and benzo [a] anthracene with promising antimicrobial activities. Il Farmaco 2002, 57, 715–722. [Google Scholar] [CrossRef]
- Smith, P.W.; Sollis, S.L.; Howes, P.D.; Cherry, P.C.; Starkey, I.D.; Cobley, K.N.; Weston, H.; Scicinski, J.; Merritt, A.; Whittington, A. Dihydropyrancarboxamides related to zanamivir: A new series of inhibitors of influenza virus sialidases. 1. Discovery, synthesis, biological activity, and structure− activity relationships of 4-guanidino-and 4-amino-4 H-pyran-6-carboxamides. J. Med. Chem. 1998, 41, 787–797. [Google Scholar] [CrossRef]
- Naseer, M.A.; Husain, A. Studies on Chromene based 2, 6-disubstituted-Thiazolo [3, 2-B][1, 2, 4] Triazole derivatives: Synthesis and Biological Evaluation. J. Drug Deliv. Ther. 2019, 9, 236–242. [Google Scholar]
- Safari, F.; Hosseini, H.; Bayat, M.; Ranjbar, A. Synthesis and evaluation of antimicrobial activity, cytotoxic and pro-apoptotic effects of novel spiro-4 H-pyran derivatives. RSC Adv. 2019, 9, 24843–24851. [Google Scholar] [CrossRef] [Green Version]
- El-Agrody, A.M.; Al-Dies, A.-A.M.; Fouda, A.M. Microwave assisted synthesis of 2-amino-6-methoxy-4H-benzo [h] chromene derivatives. Eur. J. Chem. 2014, 5, 133–137. [Google Scholar] [CrossRef] [Green Version]
- Sabry, N.M.; Mohamed, H.M.; Khattab, E.S.A.; Motlaq, S.S.; El-Agrody, A.M. Synthesis of 4H-chromene, coumarin, 12H-chromeno [2, 3-d] pyrimidine derivatives and some of their antimicrobial and cytotoxicity activities. Eur. J. Med. Chem. 2011, 46, 765–772. [Google Scholar] [CrossRef]
- Kushwaha, R.K.; Singh, K.; Kumar, P.; Chandra, D. Review on Chromen derivatives and their Pharmacological Activities. Res. J. Pharm. Technol. 2019, 12, 5566–5574. [Google Scholar] [CrossRef]
- Halawa, A.H.; Elaasser, M.M.; El Kerdawy, A.M.; Abd El-Hady, A.M.; Emam, H.A.; El-Agrody, A.M. Anticancer activities, molecular docking and structure–activity relationship of novel synthesized 4H-chromene, and 5H-chromeno [2, 3-d] pyrimidine candidates. Med. Chem. Res. 2017, 26, 2624–2638. [Google Scholar] [CrossRef]
- Anderson, R.; Nickless, G. Heterocyclic azo dyestuffs in analytical chemistry. A review. Analyst 1967, 92, 207–238. [Google Scholar] [CrossRef]
- Chung, K.-T. Azo dyes and human health: A review. J. Environ. Sci. Health Part C 2016, 34, 233–261. [Google Scholar] [CrossRef]
- Rafii, F.; Hall, J.; Cerniglia, C. Mutagenicity of azo dyes used in foods, drugs and cosmetics before and after reduction by Clostridium species from the human intestinal tract. Food Chem. Toxicol. 1997, 35, 897–901. [Google Scholar] [CrossRef]
- Alhaddad, O.A.; Abu Al-Ola, K.A.; Hagar, M.; Ahmed, H.A. Chair-and V-Shaped of H-bonded Supramolecular Complexes of Azophenyl Nicotinate Derivatives; Mesomorphic and DFT Molecular Geometry Aspects. Molecules 2020, 25, 1510. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, N.H.; Saad, G.R.; Ahmed, H.A.; Hagar, M. New wide-stability four-ring azo/ester/Schiff base liquid crystals: Synthesis, mesomorphic, photophysical, and DFT approaches. RSC Adv. 2020, 10, 9643–9656. [Google Scholar] [CrossRef] [Green Version]
- Alhaddad, O.A.; Ahmed, H.A.; Hagar, M.; Saad, G.R.; Abu Al-Ola, K.A.; Naoum, M.M. Thermal and photophysical studies of binary mixtures of liquid crystal with different geometrical mesogens. Crystals 2020, 10, 223. [Google Scholar] [CrossRef] [Green Version]
- Al-Mutabagani, L.A.; Abdullah Alshabanah, L.; Ahmed, H.A.; Abu Al-Ola, K.A.; Hagar, M. New Rod-like H-bonded Assembly Systems: Mesomorphic and Geometrical Aspects. Crystals 2020, 10, 795. [Google Scholar] [CrossRef]
- Weglarz-Tomczak, E.; Gorecki, L. Azo dyes–Biological activity and synthetic strategy. Chemik 2012, 66, 1298–1307. [Google Scholar]
- Thaokar, S.F.; Patel, D.M.; Patel, M.P.; Patel, R.G. Synthesis and antibacterial activity of novel pyrazolo [3, 4-b] quinoline based heterocyclic azo compounds and their dyeing performance. Saudi Pharm. J. 2007, 15, 48–54. [Google Scholar]
- Parekh, N.; Maheria, K.; Patel, P.; Rathod, M. Study on antibacterial activity for multidrug resistance stain by using phenyl pyrazolones substituted 3-amino 1H-pyrazolon (3, 4-b) quinoline derivative in vitro condition. Int. J. Pharm. Tech. Res. 2011, 3, 540–548. [Google Scholar]
- Khedr, A.M.; Gaber, M.; Abd El-Zaher, E.H. Synthesis, structural characterization, and antimicrobial activities of Mn (II), Co (II), Ni (II), Cu (II) and Zn (II) complexes of triazole-based azodyes. Chin. J. Chem. 2011, 29, 1124–1132. [Google Scholar] [CrossRef]
- Swati, G.; Romila, K.; Sharma, I.; Verma, P. Synthesis, characterization and antimicrobial screening of some azo compounds. Int. J. Appl. Biol. Pharm. Tech. 2011, 2, 332–338. [Google Scholar]
- Kantar, C.; Akal, H.; Kaya, B.; Islamoğlu, F.; Türk, M.; Şaşmaz, S. Novel phthalocyanines containing resorcinol azo dyes; synthesis, determination of pKa values, antioxidant, antibacterial and anticancer activity. J. Organomet. Chem. 2015, 783, 28–39. [Google Scholar] [CrossRef]
- Abadi, A.H.; Eissa, A.A.H.; Hassan, G.S. Synthesis of novel 1, 3, 4-trisubstituted pyrazole derivatives and their evaluation as antitumor and antiangiogenic agents. Chem. Pharm. Bull. 2003, 51, 838–844. [Google Scholar] [CrossRef] [Green Version]
- Afifi, T.H.; Okasha, R.M.; Alsherif, H.; Ahmed, H.E.A.; Abd-El-Aziz, A.S. Design, synthesis, and docking studies of 4H-chromene and chromene based azo chromophores: A novel series of potent antimicrobial and anticancer agents. Curr. Org. Synth. 2017, 14, 1036–1051. [Google Scholar] [CrossRef]
- Afifi, T.H.; Okasha, R.M.; Ahmed, H.E.; Ilaš, J.; Saleh, T.; Abd-El-Aziz, A.S. Structure-activity relationships and molecular docking studies of chromene and chromene based azo chromophores: A novel series of potent antimicrobial and anticancer agents. Excli J. 2017, 16, 868–902. [Google Scholar] [PubMed]
- Abd-El-Aziz, A.S.; Alsaggaf, A.T.; Okasha, R.M.; Ahmed, H.E.; Bissessur, R.; Abdelghani, A.A.; Afifi, T.H. Antimicrobial and Antitumor Screening of Fluorescent 5, 7-Dihydroxy-4-Propyl-2H-Chromen-2-One Derivatives with Docking Studies. ChemistrySelect 2016, 1, 5025–5033. [Google Scholar] [CrossRef]
- Ali, A.A.S.; Khan, D.; Naqvi, A.; Al-Blewi, F.F.; Rezki, N.; Aouad, M.R.; Hagar, M. Design, Synthesis, Molecular Modeling, Anticancer Studies, and Density Functional Theory Calculations of 4-(1, 2, 4-Triazol-3-ylsulfanylmethyl)-1, 2, 3-triazole Derivatives. ACS Omega 2021, 6, 301. [Google Scholar]
- Mohammed, F.F.; Hagar, M.; Parveen, S.; Alnoman, R.B.; Ahmed, H.A.; Ashry, E.S.H.E.; Rasheed, H.A. 2-(Alkylthio)-3-(Naphthalen-1-yl) Quinazolin-4 (3 H)-Ones: Ultrasonic Synthesis, DFT and Molecular Docking Aspects. Polycycl. Aromat. Compd. 2021, 1–15. [Google Scholar] [CrossRef]
- Parveen, S.; Hagar, M.B.; Alnoman, R.; Ahmed, H.A.; El Ashry, E.S.H.; Zakaria, M.A. Synthesis, Docking and Density Functional Theory Approaches on 1, 3-Bis-3-(4-Chlorophenyl)-2, 3-Dihydroquinazolin-4 (1H)-on-2-Thioxopropane toward the Discovery of Dual Kinase Inhibitor. Polycycl. Aromat. Compd. 2021, 1–12. [Google Scholar] [CrossRef]
- Voskressensky, L.G.; Festa, A.A.; Varlamov, A.V. Domino reactions based on Knoevenagel condensation in the synthesis of heterocyclic compounds. Recent advances. Tetrahedron 2014, 3, 551–572. [Google Scholar] [CrossRef]
- Griffiths, J. Recent developments in the color and constitution of organic dyes. Rev. Prog. Coloration Relat. Top. 1981, 11, 37–57. [Google Scholar] [CrossRef]
- EUCAST. European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID): Terminology relating to methods for the determination of susceptibility of bacteria to antimicrobial agents. CMI 2000, 6, 503–538. [Google Scholar]
- Dabur, R.; Chhillar, A.; Yadav, V.; Kamal, P.K.; Gupta, J.; Sharma, G. In vitro antifungal activity of 2-(3, 4-dimethyl-2, 5-dihydro-1H-pyrrol-2-yl)-1-methylethyl pentanoate, a dihydropyrrole derivative. J. Med. Microbiol. 2005, 54, 549–552. [Google Scholar] [CrossRef]
- Choudhary, M.I.; Thomsen, W.J. Bioassay Techniques for Drug Development; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar]
- Alaa, A.-M.; Asiri, Y.A.; Al-Agamy, M.H. Design, synthesis and antibacterial activity of fluoroquinolones containing bulky arenesulfonyl fragment: 2D-QSAR and docking study. Eur. J. Med. Chem. 2011, 46, 5487–5497. [Google Scholar]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, H.E.; El-Nassag, M.A.; Hassan, A.H.; Okasha, R.M.; Ihmaid, S.; Fouda, A.M.; Afifi, T.H.; Aljuhani, A.; El-Agrody, A.M. Introducing novel potent anticancer agents of 1H-benzo [f] chromene scaffolds, targeting c-Src kinase enzyme with MDA-MB-231 cell line anti-invasion effect. J. Enzym. Inhib. Med. Chem. 2018, 33, 1074–1088. [Google Scholar] [CrossRef] [Green Version]
- Hagar, M.; Ahmed, H.A.; Aljohani, G.; Alhaddad, O.A. Investigation of Some Antiviral N-Heterocycles as COVID 19 Drug: Molecular Docking and DFT Calculations. Int. J. Mol. Sci. 2020, 21, 3922. [Google Scholar] [CrossRef]
- Al-Otaibi, J.S.; Mary, Y.S.; Mary, Y.S.; Panicker, C.Y.; Thomas, R. Cocrystals of pyrazinamide with p-toluenesulfonic and ferulic acids: DFT investigations and molecular docking studies. J. Mol. Struct. 2019, 1175, 916–926. [Google Scholar] [CrossRef]
- Mohapatra, R.K.; El-ajaily, M.M.; Alassbaly, F.S.; Sarangi, A.K.; Das, D.; Maihub, A.A.; Ben-Gweirif, S.F.; Mahal, A.; Suleiman, M.; Perekhoda, L. DFT, anticancer, antioxidant and molecular docking investigations of some ternary Ni (II) complexes with 2-[(E)-[4-(dimethylamino) phenyl] methyleneamino] phenol. Chem. Pap. 2020, 1–15. [Google Scholar] [CrossRef]
- Joshi, R.; Pandey, N.; Yadav, S.K.; Tilak, R.; Mishra, H.; Pokharia, S. Synthesis, spectroscopic characterization, DFT studies and antifungal activity of (E)-4-amino-5-[N’-(2-nitro-benzylidene)-hydrazino]-2, 4-dihydro-[1, 2, 4] triazole-3-thione. J. Mol. Struct. 2018, 1164, 386–403. [Google Scholar] [CrossRef]
- Joshi, R.; Kumari, A.; Singh, K.; Mishra, H.; Pokharia, S. Triorganotin (IV) complexes of Schiff base derived from 1, 2, 4-triazole moiety: Synthesis, spectroscopic investigation, DFT studies, antifungal activity and molecular docking studies. J. Mol. Struct. 2020, 1206, 127639–127653. [Google Scholar] [CrossRef]
- Khodair, A.I.; Awad, M.K.; Gesson, J.-P.; Elshaier, Y.A. New N-ribosides and N-mannosides of rhodanine derivatives with anticancer activity on leukemia cell line: Design, synthesis, DFT and molecular modelling studies. Carbohydr. Res. 2020, 487, 107894–107907. [Google Scholar] [CrossRef]
- Kumar, S.S.; Athimoolam, S.; Sridhar, B. Structural, spectral, theoretical and anticancer studies on new co-crystal of the drug 5-fluorouracil. J. Mol. Struct. 2018, 1173, 951–958. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; El-Sayed, T.; Alnoman, R. Mesophase behavior and DFT conformational analysis of new symmetrical diester chalcone liquid crystals. J. Mol. Liq. 2019, 285, 96–105. [Google Scholar] [CrossRef]
- Grover, M.; Singh, B.; Bakshi, M.; Singh, S. Quantitative structure–property relationships in pharmaceutical research–Part 1. Pharm. Sci. Technol. Today 2000, 3, 28–35. [Google Scholar] [CrossRef]
- Malhotra, R.; Ravesh, A.; Singh, V. Synthesis, characterization, antimicrobial activities, and QSAR studies of organotin (IV) complexes. Phosphorus Sulfur Silicon Relat. Elem. 2017, 192, 1–31. [Google Scholar] [CrossRef]
- Kumer, A.; Sarker, M.N.; Paul, S. The Simulating Study of HOMO, LUMO, thermo physical and Quantitative Structure of Activity Relationship (QSAR) of Some Anticancer Active Ionic Liquids. Eurasian J. Environ. Res. 2019, 3, 1–10. [Google Scholar]
- Hagar, M.; Chaieb, K.; Parveen, S.; Ahmed, H.; Alnoman, R. N-alkyl 2-pyridone versus O-alkyl 2-pyridol: Ultrasonic synthesis, DFT, docking studies and their antimicrobial evaluation. J. Mol. Struct. 2020, 1199, 126926. [Google Scholar] [CrossRef]
- Ali, M.S.; Farah, M.A.; Al-Lohedan, H.A.; Al-Anazi, K.M. Comprehensive exploration of the anticancer activities of procaine and its binding with calf thymus DNA: A multi spectroscopic and molecular modelling study. RSC Adv. 2018, 8, 9083–9093. [Google Scholar] [CrossRef] [Green Version]
- Rachedi, K.O.; Ouk, T.-S.; Bahadi, R.; Bouzina, A.; Djouad, S.-E.; Bechlem, K.; Zerrouki, R.; Hadda, T.B.; Almalki, F.; Berredjem, M. Synthesis, DFT and POM analyses of cytotoxicity activity of α-amidophosphonates derivatives: Identification of potential antiviral O, O-pharmacophore site. J. Mol. Struct. 2019, 1197, 196–203. [Google Scholar] [CrossRef]
- Da Costa, R.M.; Bastos, J.K.; Costa, M.C.; Ferreira, M.M.; Mizuno, C.S.; Caramori, G.F.; Nagurniak, G.R.; Simão, M.R.; Dos Santos, R.A.; Veneziani, R.C. In vitro cytotoxicity and structure-activity relationship approaches of ent-kaurenoic acid derivatives against human breast carcinoma cell line. Phytochemistry 2018, 156, 214–223. [Google Scholar] [CrossRef]
- Lewis, D.F. Quantitative structure–activity relationships (QSARs) within the cytochrome P450 system: QSARs describing substrate binding, inhibition and induction of P450s. Inflammopharmacology 2003, 11, 43–73. [Google Scholar] [CrossRef] [PubMed]
- Almehmadi, M.A.; Aljuhani, A.; Alraqa, S.Y.; Ali, I.; Rezki, N.; Aouad, M.R.; Hagar, M. Design, synthesis, DNA binding, modeling, anticancer studies and DFT calculations of Schiff bases tethering benzothiazole-1, 2, 3-triazole conjugates. J. Mol. Struct. 2021, 1225, 129148–129163. [Google Scholar] [CrossRef]
- Bouachrine, M.; Hamidi, M.; BOUZZINEA, S.; Taoufik, H. Theoretical study on the structure and electronic properties of new materials based on thiophene and oxadiazole. J. Appl. Chem. Res. 2009, 10, 29–37. [Google Scholar]
- Yang, L.; Feng, J.-K.; Ren, A.-M. Theoretical studies on the electronic and optical properties of two thiophene–fluorene based π-conjugated copolymers. Polymer 2005, 46, 10970–10981. [Google Scholar] [CrossRef]
- Alnoman, R.B.; Parveen, S.; Hagar, M.; Ahmed, H.A.; Knight, J.G. A new chiral boron-dipyrromethene (BODIPY)-based fluorescent probe: Molecular docking, DFT, antibacterial and antioxidant approaches. J. Biomol. Struct. Dyn. 2019, 1–14. [Google Scholar] [CrossRef]
- Alnoman, R.B.; Hagar, M.; Parveen, S.; Ahmed, H.A.; Knight, J.G. Computational and molecular docking approaches of a New axially chiral BODIPY fluorescent dye. J. Photochem. Photobiol. A Chem. 2020, 395, 112508–112522. [Google Scholar] [CrossRef]
- Kouza, M.; Banerji, A.; Kolinski, A.; Buhimschi, I.; Kloczkowski, A. Role of Resultant Dipole Moment in Mechanical Dissociation of Biological Complexes. Molecules 2018, 23, 1995. [Google Scholar] [CrossRef] [Green Version]
- Shawon, J.; Khan, A.M.; Rahman, A.; Hoque, M.M.; Khan, M.A.K.; Sarwar, M.G.; Halim, M.A. Molecular Recognition of Azelaic Acid and Related Molecules with DNA Polymerase I Investigated by Molecular Modeling Calculations. Interdiscip. Sci. Comput. Life Sci. 2018, 10, 525–537. [Google Scholar] [CrossRef]
- Uzzaman, M.; Hoque, M.J. Physiochemical, molecular docking, and pharmacokinetic studies of Naproxen and its modified derivatives based on DFT. Int. J. Sci. Res. Manag. 2018, 6. [Google Scholar] [CrossRef]
- Ortega, J.T.; Serrano, M.L.; Pujol, F.H.; Rangel, H.R. Unrevealing sequence and structural features of novel coronavirus using in silico approaches: The main protease as molecular target. EXCLI J. 2020, 19, 400–409. [Google Scholar]
- Gangadevi, V.; Muthumary, J. Isolation of Colletotrichum gloeosporioides, a novel endophytic taxol-producing fungus from the leaves of a medicinal plant, Justicia gendarussa. Mycol. Balc. 2008, 5, 1–4. [Google Scholar]
- Gangadevi, V.; Muthumary, J. Preliminary studies on cytotoxic effect of fungal taxol on cancer cell lines. Afr. J. Biotechnol. 2007, 6, 1382–1386. [Google Scholar]
- Chemical Computing Group Inc. Molecular Operating Environment (MOE); Chemical Computing Group Inc.: Montreal, QC, Canada, 2016. [Google Scholar]




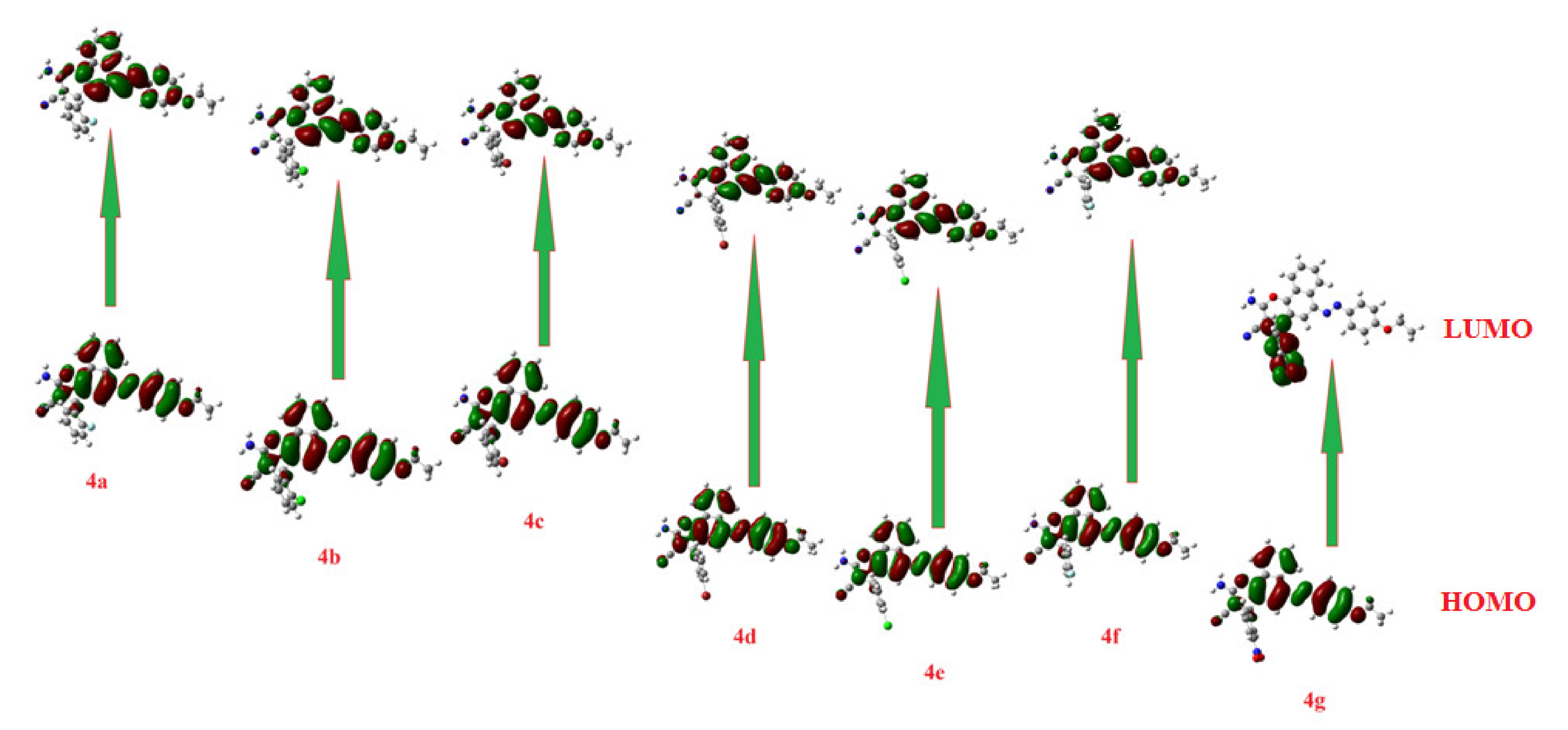










| Compound | HOMO | LUMO | ΔELUMO-HOMO | λmax (Theo) |
|---|---|---|---|---|
| 4a | −5.76 | −2.51 | 3.25 | 381.95 |
| 4b | −5.78 | −2.54 | 3.24 | 382.65 |
| 4c | −5.77 | −2.53 | 3.24 | 382.97 |
| 4d | −5.83 | −2.60 | 3.23 | 383.45 |
| 4e | −5.84 | −2.61 | 3.23 | 383.42 |
| 4f | −5.83 | −2.60 | 3.23 | 383.74 |
| 4g | −5.93 | −3.01 | 2.92 | 424.23 |
| 4h | −5.65 | −2.41 | 3.25 | 381.64 |
| 4i | −5.66 | −2.41 | 3.25 | 381.54 |
| 4j | −5.73 | −2.48 | 3.24 | 382.21 |
| 4k | −5.84 | −2.90 | 2.94 | 421.93 |
| 4l | −5.60 | −2.38 | 3.22 | 384.67 |
| Inhibition Zone Diameter (mm) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Compounds | Gram Positive | Gram Negative | Fungi | |||||
| S.P. | B.S. | P.A. | E.C. | A.F. | S.R. | G.C. | C.A. | |
| 4a | 20.5 ± 0.44 | 22.4 ± 0.58 | NA | 19.4 ± 0.19 | 18.7 ± 0.25 | 15.6 ± 0.25 | 19.2 ± 0.58 | NA |
| 4b | 18.4 ± 0.34 | 21.5 ± 0.25 | NA | 20.4 ± 0.58 | 20.9 ± 0.63 | 19.2 ± 0.27 | 22.8 ± 0.35 | NA |
| 4c | 17.4 ± 0.34 | 20.2 ± 0.25 | NA | 17.7 ± 0.58 | 16.2 ± 0.63 | 17.1 ± 0.27 | 17.1 ± 0.35 | NA |
| 4d | 19.6 ± 0.55 | 20.5 ± 0.52 | NA | 15.9 ± 0.58 | 18.6 ± 0.36 | 17.3 ± 0.35 | 18.9 ± 0.44 | NA |
| 4e | 20.3 ± 0.44 | 20.9 ± 0.25 | NA | 15.4 ± 0.39 | 18.3 ± 0.55 | 20.1 ± 0.27 | 20.9 ± 0.58 | NA |
| 4f | 20.3 ± 0.28 | 21.4 ± 0.37 | NA | 16.9 ± 0.19 | 17.6 ± 0.11 | 18.4 ± 0.23 | 20.9 ± 0.27 | NA |
| 4g | 19.3 ± 0.16 | 20.3 ± 0.19 | NA | 18.9 ± 0.27 | 16.2 ± 0.20 | 20.8 ± 0.15 | 21.8 ± 0.18 | NA |
| 4h | 19.6 ± 0.52 | 21.4 ± 0.63 | NA | 19.3 ± 0.62 | 19.3 ± 0.62 | 20.4 ± 0.34 | 20.8 ± 0.64 | NA |
| 4i | 21.4 ± 0.25 | 23.4 ± 0.58 | NA | 21.3 ± 0.16 | 23.7 ± 0.26 | 21.4 ± 0.35 | 25.4 ± 0.18 | NA |
| 4j | 16.1 ± 0.55 | 18.6 ± 0.25 | NA | 13.4 ± 0.19 | 10.6 ± 0.44 | 18.7 ± 0.25 | 23.4 ± 0.58 | NA |
| 4k | 18.3 ± 0.19 | 20.3 ± 0.24 | NA | 17.7 ± 0.25 | NA | 15.4 ± 0.24 | 18.3 ± 0.15 | NA |
| 4l | 20.8 ± 0.34 | 22.6 ± 0.25 | NA | 18.2 ± 0.58 | 18.7 ± 0.63 | 16.3 ± 0.27 | 19.4 ± 0.35 | NA |
| Ampicillin Gentamicin Amphotericin B | 21.9 ± 0.2 | 25.4 ± 0.3 | – | – | – | – | – | – |
| – | – | 17.3 ± 0.1 | 19.9 ± 0.3 | – | – | – | – | |
| – | – | – | – | 23.9 ± 0.1 | 19.7 ± 0.2 | 17.9 ± 0.2 | 19.8 ± 0.1 | |
| Minimal Inhibitory Concentration (MIC, µg/mL) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Compounds | Gram Positive | Gram Negative | Fungi | |||||
| S.P. | B.S. | P.A. | E.C. | A.F. | S.R. | G.C. | C.A. | |
| 4a | 1.95 | 0.98 | NA | 62.5 | 15.63 | 31.25 | 7.81 | NA |
| 4b | 7.81 | 1.95 | NA | 3.9 | 1.95 | 3.9 | 0.98 | NA |
| 4c | 3.9 | 1.95 | NA | 31.25 | 7.81 | 15.63 | 0.98 | NA |
| 4d | 3.9 | 15.63 | NA | 3.9 | 125 | 125 | 62.5 | NA |
| 4e | 62.5 | 31.25 | NA | 15.63 | 125 | 125 | 62.5 | NA |
| 4f | 7.81 | 7.81 | NA | 3.9 | 7.81 | 62.5 | 0.49 | NA |
| 4g | 3.9 | 3.9 | NA | 3.9 | 31.25 | 3.9 | 1.95 | NA |
| 4h | 0.98 | 1.95 | NA | 3.9 | 3.9 | 3.9 | 15.63 | NA |
| 4i | 1.95 | 0.98 | NA | 1.95 | 0.98 | 1.95 | 0.49 | NA |
| 4j | 7.81 | 7.81 | NA | 3.9 | 15.63 | 31.25 | 7.81 | NA |
| 4k | 1.95 | 0.49 | NA | 15.63 | NA | 62.5 | 7.81 | NA |
| 4l | 3.9 | 0.98 | NA | 7.81 | 7.81 | 31.25 | 3.9 | NA |
| Ampicillin Gentamicin Amphotericin B | 0.49 | 0.06 | – | – | – | – | – | – |
| – | – | 3.9 | 7.81 | – | – | – | – | |
| – | – | – | – | 0.12 | 0.49 | 7.81 | 1.95 | |
| Compounds | IC50 (µg/mL) | ||
|---|---|---|---|
| HCT-116 | MCF-7 | HepG-2 | |
| 4a | 42.1 | 21.9 | 11.6 |
| 4b | 19.2 | 16.2 | 11.3 |
| 4c | 12.2 | 21.1 | 7.28 |
| 4d | 15.3 | 9.16 | 7.64 |
| 4e | 5.54 | 19.7 | 9.99 |
| 4f | 42.6 | 22.5 | 14.3 |
| 4g | 9.01 | 7.14 | 12.0 |
| 4h | 40.9 | 5.5 | 9.19 |
| 4i | 30.0 | 11.5 | 8.76 |
| 4j | 4.35 | 4.72 | 11.6 |
| 4k | 11.2 | 9.5 | 10.5 |
| 4l | 2.51 | 3.91 | 5.41 |
| Doxorubicin | 0.88 | 1.02 | 1.19 |
| Vinblastine | 2.6 | 6.1 | 4.6 |
| Colchicine | 42.8 | 17.7 | 10.6 |
| Compound No. | 4b | 4h | Fat Brown B | 1-(4-Ethoxyphenyl)-2-(4-Phenyl-4H-Benzo[h]Chromen-6-yl)Diazene | |
|---|---|---|---|---|---|
| Amino acid (Distance Å) | Asp 73 | -NH2 (2.3, 2.4) | – | --OH (2.4) | – |
| Thr 165 | -CN (3.2) | – | – | – | |
| Asn 46 | – | --NH2 (2.0) | – | – | |
| Asp 49 | – | --Br (3.2) | – | – | |
| Ile 78 | – | -phenyl | – | – | |
| Interaction type | H-bonding | H-bonding (aromatic) | H-bonding | H-bonding | |
| ∆G (kcal/mol) (MOE) | −13.61 | −12.23 | −11.24 | – | |
| ∆G (kcal/mol) (Pyrx) | −9.0 | −8.6 | −7.7 | −7.9 | |
| Compounds | A = −ELUMO | I = −EHOMO | µ | ||||
|---|---|---|---|---|---|---|---|
| 4a | 4.14 | 1.63 | 0.62 | 5.26 | 2.51 | 5.76 | 6.4 |
| 4b | 4.16 | 1.62 | 0.62 | 5.34 | 2.54 | 5.78 | 6.9 |
| 4c | 4.15 | 1.62 | 0.62 | 5.32 | 2.53 | 5.77 | 6.7 |
| 4d | 4.22 | 1.62 | 0.62 | 5.50 | 2.60 | 5.83 | 7.7 |
| 4e | 4.23 | 1.62 | 0.62 | 5.53 | 2.61 | 5.84 | 8.0 |
| 4f | 4.22 | 1.62 | 0.62 | 5.50 | 2.60 | 5.83 | 7.2 |
| 4g | 4.47 | 1.46 | 0.68 | 6.84 | 3.01 | 5.93 | 10.3 |
| 4h | 4.03 | 1.62 | 0.62 | 5.01 | 2.41 | 5.65 | 2.9 |
| 4i | 4.04 | 1.63 | 0.62 | 5.01 | 2.41 | 5.66 | 3.1 |
| 4j | 4.11 | 1.63 | 0.62 | 5.18 | 2.48 | 5.73 | 4.2 |
| 4k | 4.37 | 1.47 | 0.68 | 6.50 | 2.90 | 5.84 | 7.4 |
| 4l | 3.99 | 1.61 | 0.62 | 4.94 | 2.38 | 5.60 | 2.5 |
| Compound | Lipophilicity | Polar Surface Area | H-Bond Acceptor | H-Bond Donor | Violations | Volume |
|---|---|---|---|---|---|---|
| 4a | 6.57 | 93.01 | 6 | 2 | 1 | 407.77 |
| 4b | 7.08 | 93.01 | 6 | 2 | 1 | 416.38 |
| 4c | 7.21 | 93.01 | 6 | 2 | 2 | 420.73 |
| 4d | 7.24 | 93.01 | 6 | 2 | 2 | 420.73 |
| 4e | 7.11 | 93.01 | 6 | 2 | 1 | 416.38 |
| 4f | 6.62 | 93.01 | 6 | 2 | 1 | 407.77 |
| 4g | 6.62 | 93.01 | 6 | 2 | 1 | 407.77 |
| 4h | 6.39 | 138.84 | 9 | 2 | 1 | 426.17 |
| 4i | 7.67 | 95.52 | 7 | 2 | 2 | 465.20 |
| 4j | 7.54 | 95.52 | 7 | 2 | 2 | 460.85 |
| 4k | 7.69 | 95.52 | 7 | 2 | 2 | 465.20 |
| 4l | 6.84 | 141.35 | 10 | 2 | 2 | 470.64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd-El-Aziz, A.S.; Alsaggaf, A.; Assirey, E.; Naqvi, A.; Okasha, R.M.; Afifi, T.H.; Hagar, M. A New Family of Benzo[h]Chromene Based Azo Dye: Synthesis, In-Silico and DFT Studies with In Vitro Antimicrobial and Antiproliferative Assessment. Int. J. Mol. Sci. 2021, 22, 2807. https://doi.org/10.3390/ijms22062807
Abd-El-Aziz AS, Alsaggaf A, Assirey E, Naqvi A, Okasha RM, Afifi TH, Hagar M. A New Family of Benzo[h]Chromene Based Azo Dye: Synthesis, In-Silico and DFT Studies with In Vitro Antimicrobial and Antiproliferative Assessment. International Journal of Molecular Sciences. 2021; 22(6):2807. https://doi.org/10.3390/ijms22062807
Chicago/Turabian StyleAbd-El-Aziz, Alaa S., Azhaar Alsaggaf, Eman Assirey, Arshi Naqvi, Rawda M. Okasha, Tarek H. Afifi, and Mohamed Hagar. 2021. "A New Family of Benzo[h]Chromene Based Azo Dye: Synthesis, In-Silico and DFT Studies with In Vitro Antimicrobial and Antiproliferative Assessment" International Journal of Molecular Sciences 22, no. 6: 2807. https://doi.org/10.3390/ijms22062807








