Potential Enhancement of Metformin Hydrochloride in Lipid Vesicles Targeting Therapeutic Efficacy in Diabetic Treatment
Abstract
:1. Introduction
2. Results
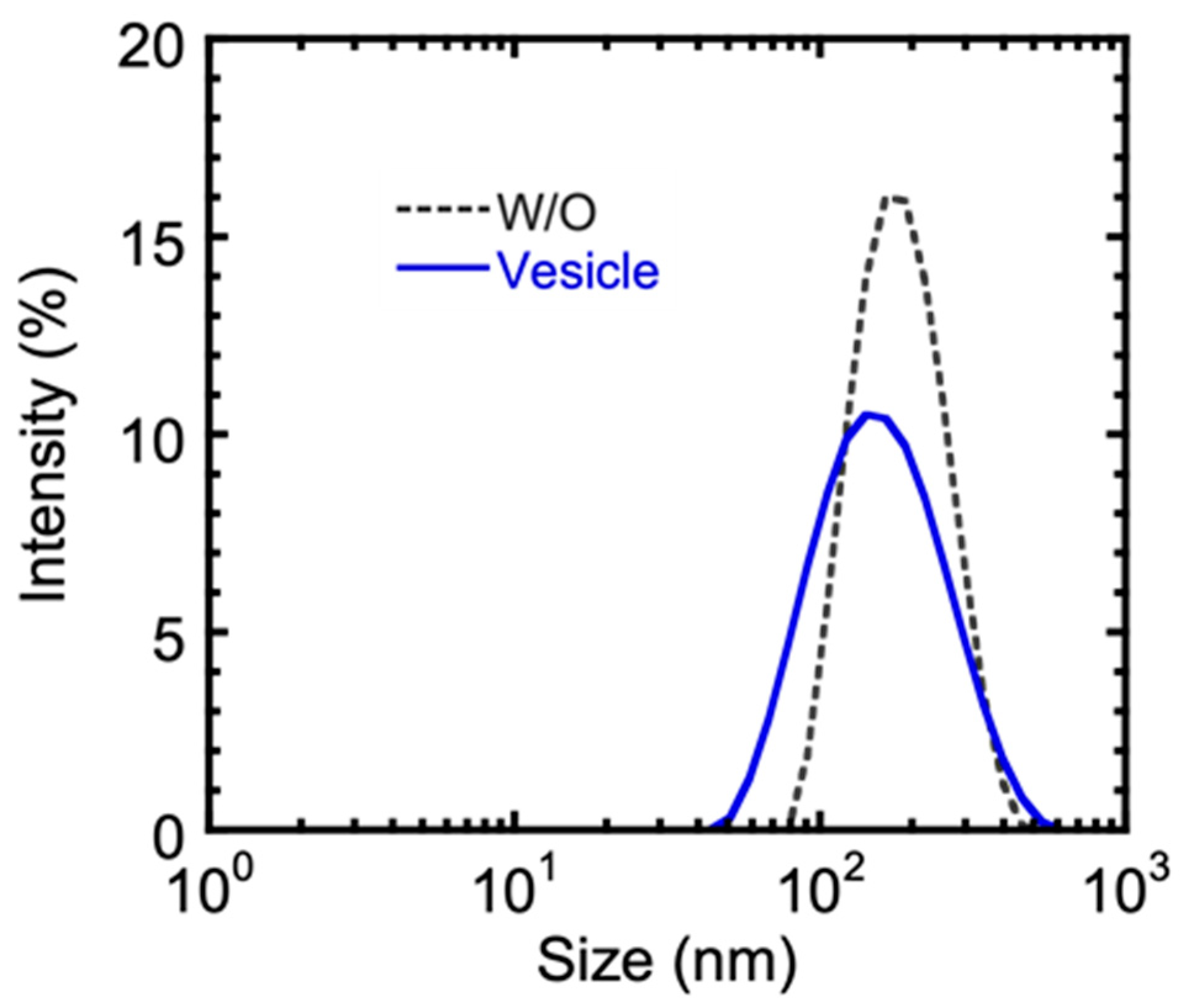
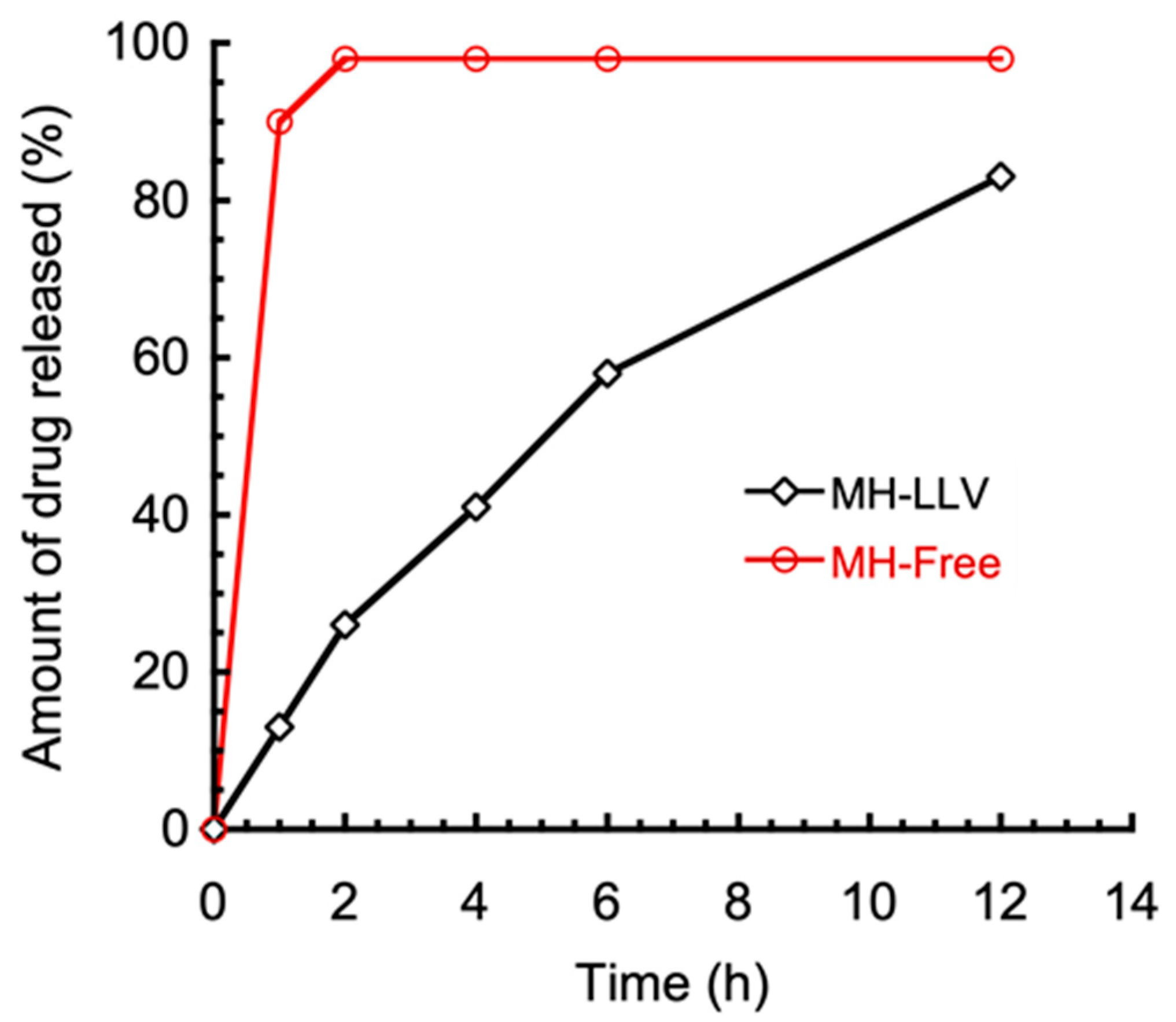
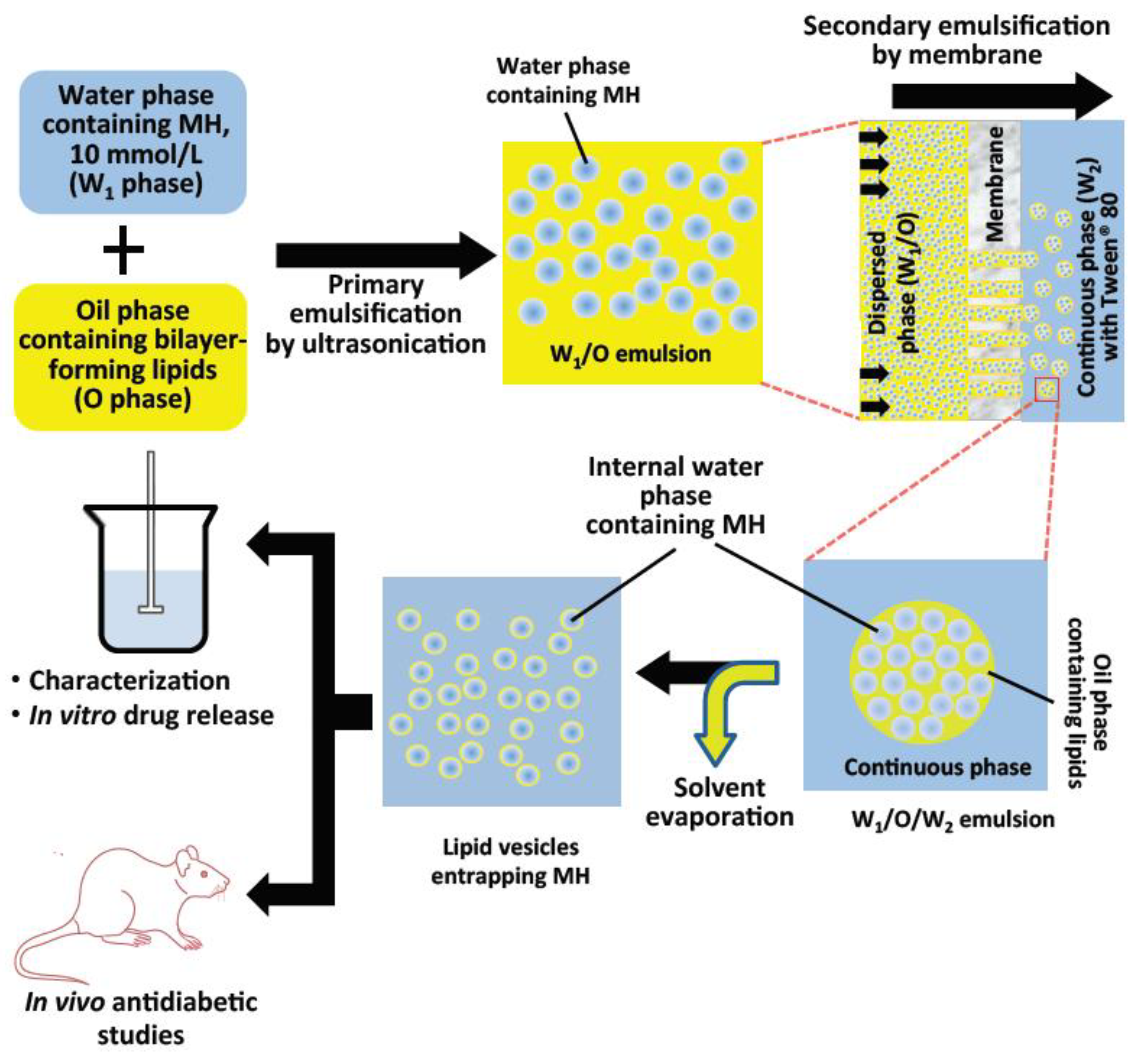
2.1. Preparation and Characterization of Metformin Hydrochloride-Loaded Lipid Vesicles
2.2. Biochemical Studies
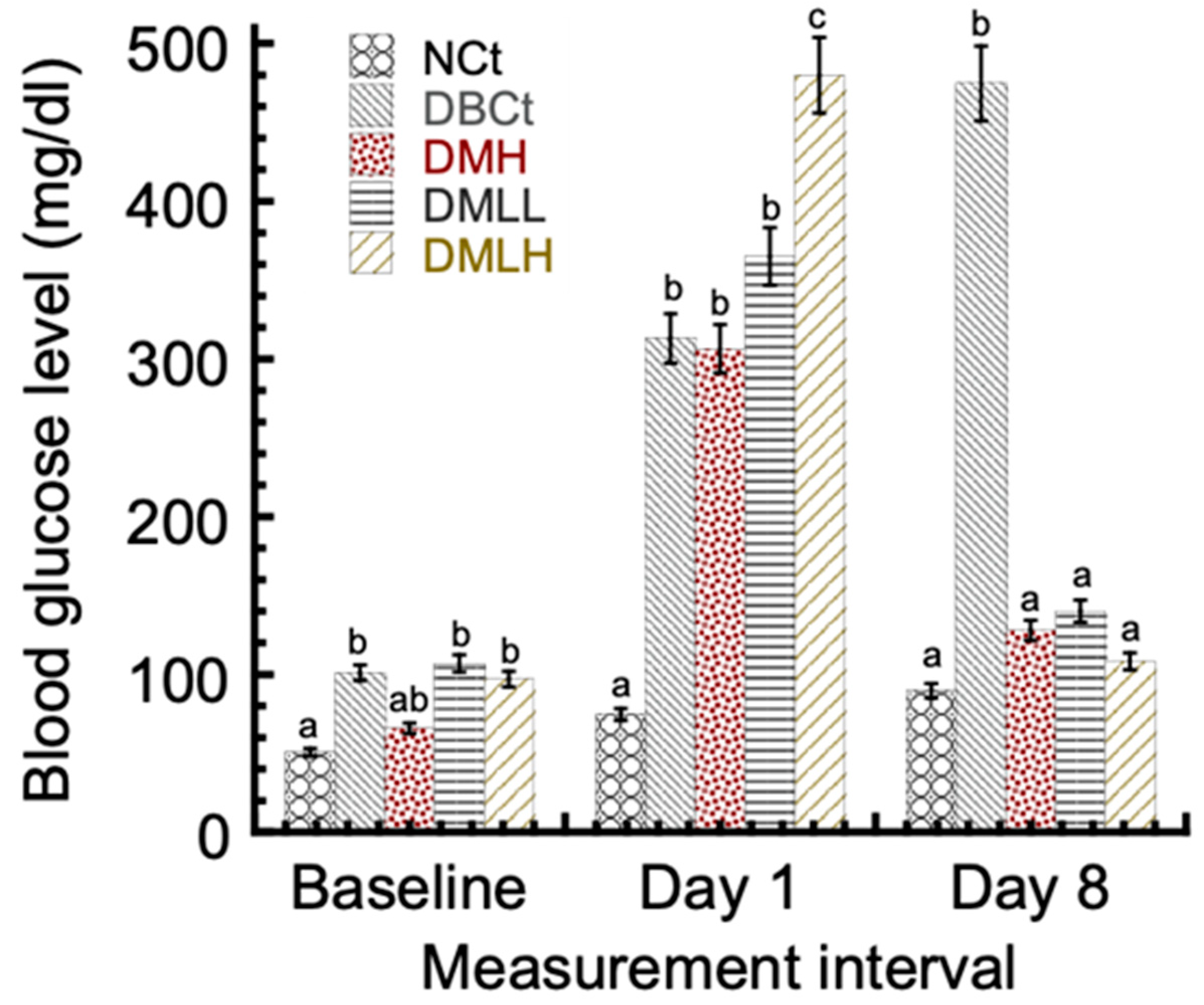
2.2.1. The Effects of Metformin Hydrochloride-Loaded Lipid Vesicles on the Body Weight and Serum Glucose Level of Alloxan-Induced Diabetic Rats
2.2.2. The Effect of Metformin Hydrochloride-Loaded Lipid Vesicles on the Serum Lipid Profile and Liver Enzymes of Alloxan-Induced Diabetic Rats
2.2.3. The Effect of Metformin Hydrochloride-Loaded Lipid Vesicles on the Levels of Oxidative Stress in Alloxan-Induced Diabetic Rats
2.2.4. The Effect of Metformin Hydrochloride-Loaded Lipid Vesicles on the Kidney Profile and Serum Electrolyte Levels in Alloxan-Induced Diabetic Rats
3. Discussion
4. Materials and Methods
4.1. Schematic Overview of Experimental Program
4.2. Chemicals and Reagents
4.3. Methods
4.3.1. Preparation of Metformin Hydrochloride-Loaded Lipid Vesicles
4.3.2. Characterization of the Lipid Vesicles
4.3.3. Metformin Hydrochloride In Vitro Drug Release Study
4.3.4. In Vivo Antidiabetic Studies
Animals, Feeding, and Ethical Guidelines
Induction of Diabetes
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Viollet, B.; Guigas, B.; Garcia, N.S.; Leclerc, J.; Foretz, M.; Andreelli, F. Cellular and molecular mechanisms of metformin: An overview. Clin. Sci. 2011, 122, 253–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, S.K.; Chan, A.; Parvathaneni, V.; Gupta, V. Metformin-loaded chitosomes for treatment of malignant pleural mesothelioma—A rare thoracic cancer. Int. J. Biol. Macromol. 2020, 160, 128–141. [Google Scholar] [CrossRef]
- Rao, C.V. Biguanides. In Encyclopedia of Toxicology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 1, pp. 452–455. [Google Scholar] [CrossRef]
- Rojas, L.B.A.; Gomes, M.B. Metformin: An old but still the best treatment for type 2 diabetes. Diabetol. Metab. Syndr. 2013, 5, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Çetin, M.; Atila, A.; Sahin, S.; Vural, I. Preparation and characterization of metformin hydrochloride loaded-Eudragit®RSPO and Eudragit®RSPO/PLGA nanoparticles. Pharm. Dev. Technol. 2011, 18, 570–576. [Google Scholar] [CrossRef]
- Moffat, A.C.; Osselton, M.D.; Widdop, B. Clarke’s Analysis of Drug and Poison, 4th ed.; Pharmaceutical Press: London, UK, 2011; ISBN 978-0-85369-711-4. [Google Scholar]
- Warren, O.H.; Joel, D.B.; Kelly, R.; Youling, Y. Chitosan-Coated Calcium Sulfate Based Medicament Delivery System. U.S. Patent No. 20080081060A1, 3 April 2008. [Google Scholar]
- Brayfield, A. Martindale: The Complete Drug Reference; Pharmaceutical Press: London, UK, 2014; p. 468. [Google Scholar]
- Corti, G.; Cirri, M.; Maestrelli, F.; Mennini, N.; Mura, P. Sustained-release matrix tablets of metformin hydrochloride in combination with triacetyl-β-cyclodextrin. Eur. J. Pharm. Biopharm. 2008, 68, 303–309. [Google Scholar] [CrossRef]
- Basavaraj, S.; Betageri, G.V. Can formulation and drug delivery reduce attrition during drug discovery and development—Review of feasibility, benefits and challenges. Acta Pharm. Sin. B 2014, 4, 3–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navneet, G.; Akanksha, G.; Neetesh, J. Formulation design and in-vitro evaluation of metformin microspheres using ionotropic gelation technique. J. Pharm. Res. 2011, 4, 2103–2106. [Google Scholar]
- Patel, F.M.; Patel, A.N.; Rathore, K.S. Release of metformin hydrochloride from ispaghula sodium alginate beads adhered cock intestinal mucosa. Int. J. Curr. Pharm. Res. 2011, 3, 52–55. [Google Scholar]
- Kumar, A.; Balakrishna, T.; Rajiv, J. Formulation and evaluation of mucoadhesive microcapsules of metformin HCl with gum karaya. Int. J. Pharm. Pharm. Sci. 2011, 3, 150–155. [Google Scholar]
- Timmins, P.; Donahue, S.; Meeker, J.; Marathe, P. Steady-State Pharmacokinetics of a Novel Extended-Release Metformin Formulation. Clin. Pharmacokinet. 2005, 44, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.X.; Jan, S.; Chou, J.; Chen, C. Controlled Release Metformin Formulations. U.S. Patent No. 008475841B2, 2 July 2013. [Google Scholar]
- U.S. Food & Drug Administration. Dapagliflozin and Metformin Hydrochloride Extended-Release Tablets. ANDA Tentative Approval; ANDA 211491; U.S. Food & Drug Administration: Silver Spring, MD, USA, 2018; pp. 1–6. [Google Scholar]
- Li, M.; Shen, Q.; Lu, W.; Chen, J.; Yu, L.; Liu, S.; Nie, X.; Shao, L.; Liu, Y.; Gao, S.; et al. Development and evaluation of controlled release of metformin hydrochloride for improving the oral bioavailability based on a novel enteric osmotic pump capsule. J. Drug Deliv. Sci. Technol. 2020, 60, 102054. [Google Scholar] [CrossRef]
- Schwartz, S.L.; Gordi, T.; Hou, E.; Cramer, M.; Heritier, M.; E Cowles, V. Clinical development of metformin extended-release tablets for type 2 diabetes: An overview. Expert Opin. Drug Metab. Toxicol. 2008, 4, 1235–1243. [Google Scholar] [CrossRef] [PubMed]
- DeFronzo, R.A.; Buse, J.B.; Kim, T.; Burns, C.; Skare, S.; Baron, A.; Fineman, M. Once-daily delayed-release metformin lowers plasma glucose and enhances fasting and postprandial GLP-1 and PYY: Results from two randomised trials. Diabetologia 2016, 59, 1645–1654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasan, A.A.; Madkor, H.; Wageh, S. Formulation and evaluation of metformin hydrochloride-loaded niosomes as controlled release drug delivery system. Drug Deliv. 2013, 20, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Rai, V.K.; Mishra, N.; Agrawal, A.K.; Jain, S.; Yadav, N.P. Novel drug delivery system: An immense hope for diabetics. Drug Deliv. 2016, 23, 2371–2390. [Google Scholar] [CrossRef] [PubMed]
- Kuroiwa, T.; Horikoshi, K.; Suzuki, A.; Neves, M.A.; Kobayashi, I.; Uemura, K.; Nakajima, M.; Kanazawa, A.; Ichikawa, S. Efficient Encapsulation of a Water-Soluble Molecule into Lipid Vesicles Using W/O/W Multiple Emulsions via Solvent Evaporation. J. Am. Oil Chem. Soc. 2016, 93, 421–430. [Google Scholar] [CrossRef]
- Ossai, E.C.; Kuroiwa, T.; Horikoshi, K.; Otsuka, Y.; Terasawa, J.; Kanazawa, A.; Sato, S.; Ichikawa, S. Lipid Vesicle Preparation Using W/O/W Multiple Emulsions Via Solvent Evaporation: The Effect of Emulsifiers on the Entrapment Yield of Hydrophilic Materials. J. Am. Oil Chem. Soc. 2019, 96, 1405–1416. [Google Scholar] [CrossRef]
- Senior, J.H. Fate and behavior of liposomes in vivo: A review of controlling factors. Crit. Rev. Ther. Drug Carr. Syst. 1987, 3, 123–135. [Google Scholar]
- Wang, T.; Deng, Y.; Geng, Y.; Gao, Z.; Zou, J.; Wang, Z. Preparation of submicron unilamellar liposomes by freeze-drying double emulsions. Biochim. Biophys. Acta Biomembr. 2006, 1758, 222–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garti, N. Progress in Stabilization and Transport Phenomena of Double Emulsions in Food Applications. LWT Food Sci. Technol. 1997, 30, 222–235. [Google Scholar] [CrossRef]
- Sunil, C.; Irudayaraj, S.S.; Duraipandiyan, V.; Alrashood, S.T.; Alharbi, S.A.; Ignacimuthu, S. Friedelin exhibits antidiabetic effect in diabetic rats via modulation of glucose metabolism in liver and muscle. J. Ethnopharmacol. 2021, 268, 113659. [Google Scholar] [CrossRef] [PubMed]
- Desai, P.; Patlolla, R.R.; Singh, M. Interaction of nanoparticles and cell-penetrating peptides with skin for transdermal drug delivery. Mol. Membr. Biol. 2010, 27, 247–259. [Google Scholar] [CrossRef] [Green Version]
- Longe, A.O.; Momoh, J.; Adepoju, P.A. Effects of cinnamon aqueous extract on blood level liver biomarker enzymes, hematological and lipid profile parameters in alloxan- induced diabetic male albino rats. Eur. Sci. J. 2015, 1, 418–426. [Google Scholar]
- Cox, R.A.; García-Palmieri, M.R. Cholesterol, Triglycerides, and Associated Lipoproteins, 3rd ed.; Walker, H.K., Hall, W.D., Hurst, J.W., Eds.; Butterworths: Boston, MA, USA, 1990; ISBN 040990077X. [Google Scholar]
- Drotman, R.B.; Lawhorn, G.T. Serum Enzymes as Indicators of Chemically Induced Liver Damage. Drug Chem. Toxicol. 1978, 1, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Effiong, G.S.; Akpan, H.D. The effect of Nauclea latifolia leaf extract on some biochemical parameters in streptozotocin diabetic rat models. Int. Res. J. Med. Med. Sci. 2015, 6, 47–52. [Google Scholar] [CrossRef]
- Rath, D.; Kar, D.M.; Panigrahi, S.K.; Maharana, L. Antidiabetic effects of Cuscuta reflexa Roxb. in streptozotocin induced diabetic rats. J. Ethnopharmacol. 2016, 192, 442–449. [Google Scholar] [CrossRef]
- Nasri, H.; Rafieian-Kopaei, M. Metformin: Current knowledge. J. Res. Med. Sci. 2014, 19, 658–664. [Google Scholar] [PubMed]
- Nna, V.U.; Abu Bakar, A.B.; Lazin, R.M.L.M.; Mohamed, M. Antioxidant, anti-inflammatory and synergistic anti-hyperglycemic effects of Malaysian propolis and metformin in streptozotocin—Induced diabetic rats. Food Chem. Toxicol. 2018, 120, 305–320. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, U.; Das, D.; Banerjee, R.K. Reactive oxygen species: Oxidative damage and pathogenesis. Curr. Sci. 1999, 77, 658–666. [Google Scholar]
- Nandini, H.S.; Naik, P.R. Antidiabetic, antihyperlipidemic and antioxidant effect of Vincamine, in streptozotocin-induced diabetic rats. Eur. J. Pharmacol. 2019, 843, 233–239. [Google Scholar] [CrossRef]
- Atangwho, I.J.; Ebong, P.E.; Eteng, M.U.; Eyong, E.U.; Obi, A.U. NEffect of Vernonia amygdalina del leaf on kidney function of diabetic rats. Int. J. Pharmacol. 2007, 3, 143–148. [Google Scholar] [CrossRef] [Green Version]
- Harita, N.; Hayashi, T.; Sata, K.K.; Nakamura, Y.; Yoneda, T.; Endo, G.; Kambe, H. Lower serum creatinine is a new risk factor of type 2 diabetes: The kansai healthcare study. Diabetes Care 2009, 32, 424–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanko, Y.; Daniel, P.A.; Mohammed, K.A.; Jimoh, A.; Yerima, M.; Mohammed, A. Effect of ethanolic extract of Caralluma daizielii on serum electrolytes levels on fructose induced diabetes in wistar rats. Sch. Res. 2013, 4, 162–166. [Google Scholar]
- Onunogbo, C.; Ohaeri, O.; Eleazu, C. Effect of Mistletoe (Viscum album) Extract on the Blood Glucose, Liver Enzymes and Electrolyte Balance in Alloxan Induced Diabetic Rats. Am. J. Biochem. Mol. Biol. 2012, 3, 143–150. [Google Scholar] [CrossRef]
- Association American Diabetes. Diagnosis and classification of diabetes mellitus. Diabetes Care 2010, S62–S69. [Google Scholar] [CrossRef] [Green Version]
- Ishiguro, H.; Yamamoto, A.; Nakakuki, M.; Yi, L.; Ishiguro, M.; Yamaguchi, M.; Kondo, S.; Mochimaru, Y. Physiology and pathophysiology of bicarbonate secretion by pancreatic duct epithelium. Nagoya J. Med. Sci. 2012, 74, 1–18. [Google Scholar] [CrossRef]
- European Community. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the Protection of Animals Used for Scientific Purposes. 2010, pp. 33–79. Available online: https://eur-lex.europa.eu/eli/dir/2010/63/oj (accessed on 28 January 2021).

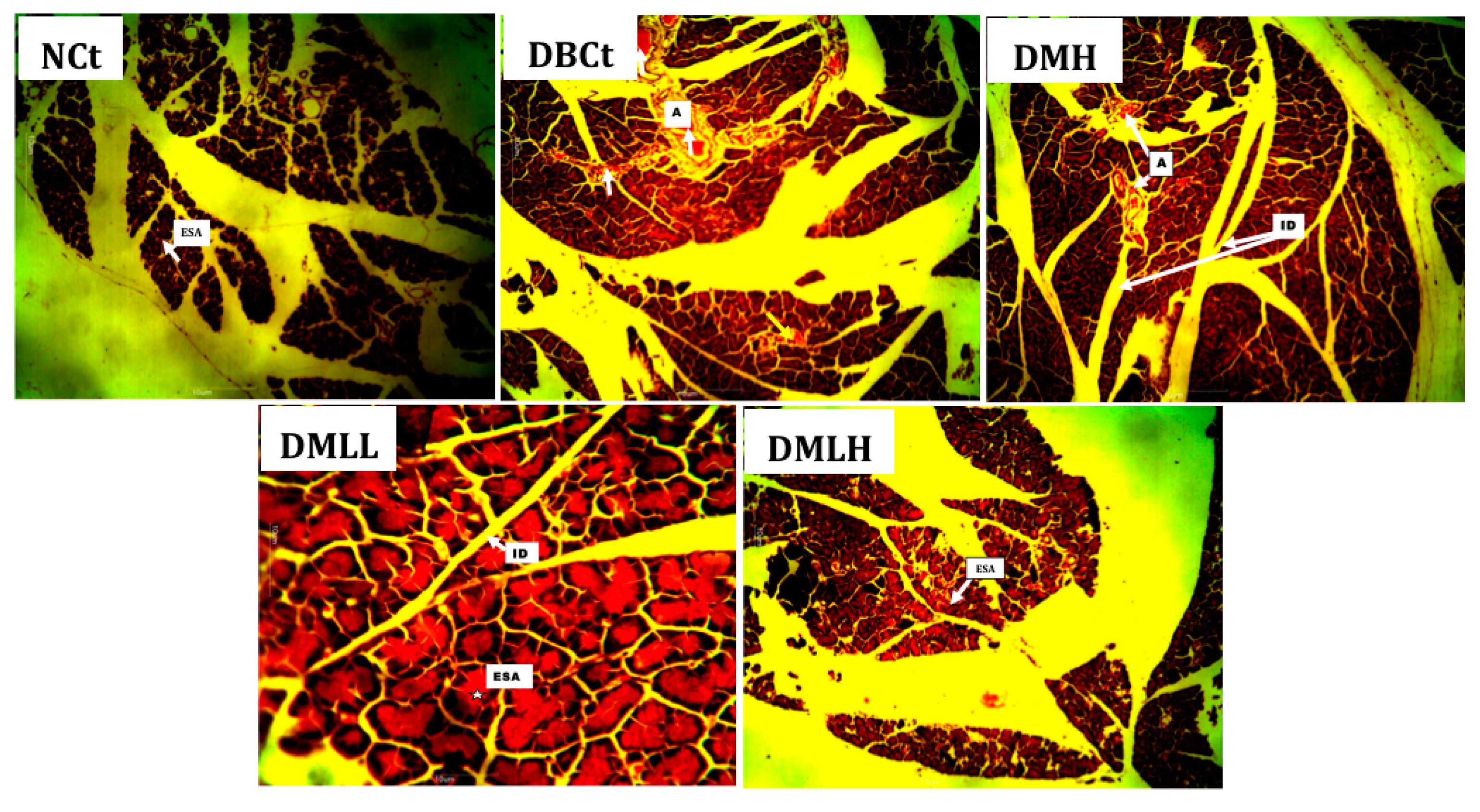
| Parameters/Groups | NCt | DBCt | DMH | DMLL | DMLH |
|---|---|---|---|---|---|
| Total cholesterol (mmol/L) | 2.75 ± 0.64 a | 4.70 ± 0.13 c | 3.91 ± 0.35 b | 2.17 ± 0.17 a | 2.46 ± 0.46 a |
| Triacylglycerols (mmol/L) | 1.43 ± 0.29 a | 2.47 ± 0.03 b | 1.69 ± 0.03 a | 1.64 ± 0.64 a | 1.58 ± 0.58 a |
| HDL-chol. (mmol/L) | 1.15 ± 0.54 b | 0.75 ± 0.05 a | 1.10 ± 0.03 ab | 1.06 ± 0.06 ab | 1.18 ± 0.18 ab |
| LDL-chol. (mmol/L) | 1.26 ± 0.69 a | 2.89 ± 0.78 b | 1.72 ± 0.73 a | 0.76 ± 0.05 a | 0.96 ± 0.06 a |
| AST (U/min/mL) | 13.11 ± 1.07 a | 19.89 ± 0.84 d | 16.66 ± 0.66 c | 15.33 ± 0.33 b | 13.54 ± 0.46 a |
| ALT (U/min/mL) | 10.21 ± 0.51 a | 16.67 ± 0.01 d | 15.29 ± 0.61 cd | 14.66 ± 0.66 c | 12.17 ± 1.49 b |
| ALP (U/min/mL) | 14.90 ± 0.46 a | 19.28 ± 0.22 d | 15.53 ± 0.52 ab | 18.00 ± 1.00 c | 16.65 ± 0.65 b |
| Total bilirubin (mg/dL) | 0.51 ± 0.03 b | 0.65 ± 0.04 c | 0.48 ± 0.03 b | 0.39 ± 0.01 a | 0.45 ± 0.05 b |
| Parameters/Groups | NCt | DBCt | DMH | DMLL | DMLH |
|---|---|---|---|---|---|
| GSH (mg/dL) | 3.10 ± 0.19 a | 2.46 ± 0.44 a | 3.51 ± 0.10 a | 2.48 ± 1.49 a | 4.03 ± 0.08 b |
| SOD (U/min/mL) | 1.12 ± 0.02 b | 1.02 ± 0.08 ab | 1.12 ± 0.01 b | 0.91 ± 0.08 a | 1.12 ± 0.09 b |
| CAT (U/min/mL) | 1.77 ± 0.05 b | 1.07 ± 0.10 a | 1.48 ± 0.04 ab | 1.39 ± 0.46 ab | 1.49 ± 0.18 ab |
| GPx (U/min/mL) | 6.38 ± 0.44 c | 4.64 ± 0.35 a | 5.50 ± 0.44 abc | 5.04 ± 0.88 ab | 6.03 ± 0.86 bc |
| MDA (mg/dL) | 2.49 ± 0.28 b | 3.32 ± 0.19 c | 3.16 ± 0.03 c | 2.21 ± 0.37 ab | 2.01 ± 0.19 a |
| Parameters/Groups | NCt | DBCt | DMH | DMLL | DMLH |
|---|---|---|---|---|---|
| Creatinine (mg/dL) | 0.92 ± 0.08 ab | 0.68 ± 0.11 a | 0.88 ± 0.01 ab | 1.00 ± 0.10 b | 1.07 ± 0.07 c |
| Urea (mg/dL) | 21.03 ±1.12 ab | 26.22 ± 3.40 b | 24.22 ± 0.52 ab | 24.29 ± 0.29 ab | 19.40 ± 5.39 a |
| Sodium (mmol/L) | 113.67 ± 0.88 b | 100.33 ± 7.97 a | 115.00 ± 0.58 b | 120.00 ± 2.89 b | 119.00 ± 0.58 b |
| Potassium (mmol/L) | 3.28 ± 0.40 b | 2.83 ± 0.09 a | 3.28 ± 0.02 b | 3.28 ± 0.16 b | 3.33 ± 0.19 b |
| Chloride (mmol/L) | 78.90 ± 1.23 b | 64.10 ± 9.17 a | 81.15 ± 0.11 b | 79.44 ± 0.28 b | 81.74 ± 0.43 b |
| Bicarbonate (mmol/L) | 24.05 ± 0.23 b | 19.83 ± 1.95 a | 24.31 ± 0.20 b | 23.03 ± 0.17 b | 24.55 ± 0.32 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ossai, E.C.; Madueke, A.C.; Amadi, B.E.; Ogugofor, M.O.; Momoh, A.M.; Okpala, C.O.R.; Anosike, C.A.; Njoku, O.U. Potential Enhancement of Metformin Hydrochloride in Lipid Vesicles Targeting Therapeutic Efficacy in Diabetic Treatment. Int. J. Mol. Sci. 2021, 22, 2852. https://doi.org/10.3390/ijms22062852
Ossai EC, Madueke AC, Amadi BE, Ogugofor MO, Momoh AM, Okpala COR, Anosike CA, Njoku OU. Potential Enhancement of Metformin Hydrochloride in Lipid Vesicles Targeting Therapeutic Efficacy in Diabetic Treatment. International Journal of Molecular Sciences. 2021; 22(6):2852. https://doi.org/10.3390/ijms22062852
Chicago/Turabian StyleOssai, Emmanuel Chekwube, Augustine Chidi Madueke, Benjamin Emenike Amadi, Martins Obinna Ogugofor, Audu Mumuni Momoh, Charles Odilichukwu R. Okpala, Chioma Assumpta Anosike, and Obioma Uzoma Njoku. 2021. "Potential Enhancement of Metformin Hydrochloride in Lipid Vesicles Targeting Therapeutic Efficacy in Diabetic Treatment" International Journal of Molecular Sciences 22, no. 6: 2852. https://doi.org/10.3390/ijms22062852








