Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis
Abstract
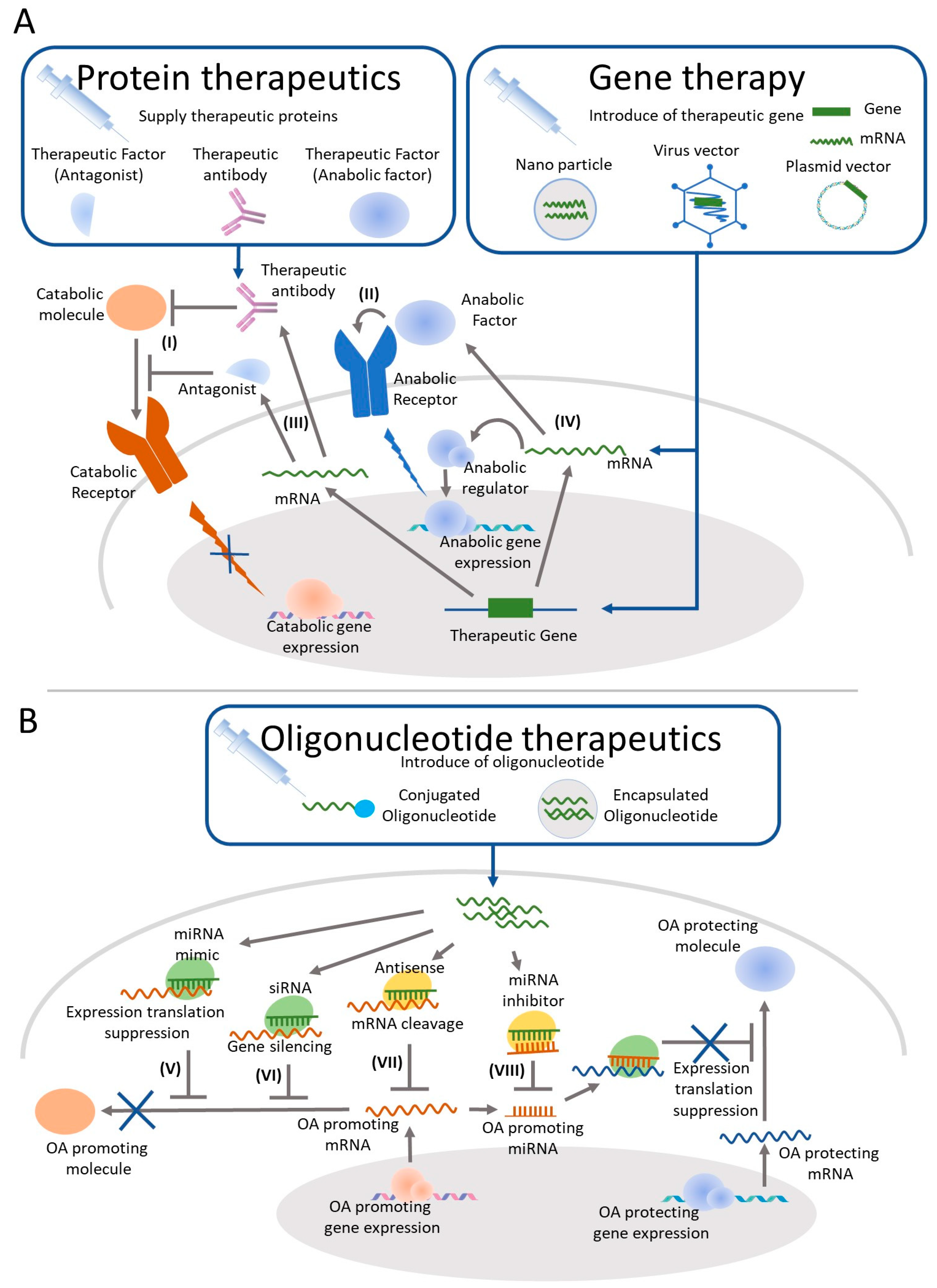
:1. Introduction
2. Target Biological Pathways for DMOADs
3. The Advantage of IA Delivery in OA Treatment
4. OA Treatment by IA Injection of Protein Therapeutics
| Study Name (ClinicalTrials.gov 1) | Mode of Action Biologicals | Study Phase Outcome Measures | Study Identifier Current Status 1 (Completion Year) |
|---|---|---|---|
| Biologic Therapy to Prevent Osteoarthritis After ACL Injury | Inhibit IL-1β IL-1 Receptor antagonist r-metHuIL-1ra (Anakinra) | Early Phase 1 cytokine level/Knee pain and function/marker level | NCT03968913 [22] Not yet recruiting |
| Study of Safety, Tolerability, Preliminary Efficacy of Intra-articular LNA043 Injections in Patients with Articular Cartilage Lesions and Knee Osteoarthritis | Assist cartilage repair. A modified human angiopoietin-like 3 protein | Phase 2 MRI/AEs/protein level/antibodies/Others | NCT03275064 [23] 2017~Recruiting |
| A Study to Investigate the Safety and Effectiveness of Different Doses of Sprifermin in Participants with Osteoarthritis of the Knee (FORWARD) | Assist cartilage repair Fibroblast growth factor 18 (Sprifermin) | Phase 2 MRI/WOMAC/PGA/mJSW/Protein level | NCT01919164 [24] Completed (2019) has results |
| Dose Finding Study of Bone Morphogenetic Protein 7 (BMP-7) in Subjects with Osteoarthritis (OA) of the Knee | Assist cartilage repair Bone Morphogenetic Protein 7 (BMP-7/OP-1) | Phase 2 WOMAC | NCT01111045 [25] Completed (2011) |
| To Determine the Safety, Tolerability, Pharmacokinetics and Effect on Pain of a Single Intra-articular Administration of Canakinumab in Patients with Osteoarthritis in the Knee | Inhibit IL-1β humanized monoclonal antibody to interleukin-1β (Canakinumab) | Phase 2 AEs/VAS/WOMAC/Others | NCT01160822 [26] Completed (2010) has results |
| Treatment of Knee Osteoarthritis with Intra-Articular Infliximab | Inhibit TNFα chimeric monoclonal antibody to TNF-α (Infliximab) | Phase 4 Cellular infiltrates/Effusion/WOMAC/Others | NCT01144143 [27] Completed (2011) has results |
| Study of Intra-articular DLX105 Applied to Patients with Severely Painful Osteoarthritis of the Knee | Inhibit TNFα a single-chain (scFv) antibody fragment against TNF-α (DLX105) | Phase 1/2 AEs/VAS/WOMAC | NCT00819572 [28] Completed (2010) |
5. Gene Therapy for OA Treatment by IA
| Study Name (ClinicalTrials.gov 1) | Mode of Action Biologicals | Study Phase Outcome Measures | Study Identifier Current Status 1 (Completion Year) |
|---|---|---|---|
| Safety of Intra-Articular Sc-rAAV2.5IL-1Ra in Subjects with Moderate Knee OA (AAVIL-1Ra) | Inhibit IL-1β sc-rAAV2.5IL-1Ra | Phase 1 AEs | NCT02790723 [61] 2019~Recruiting |
| Study to Evaluate the Safety and Tolerability of FX201 in Patients with Osteoarthritis of the Knee | Inhibit IL-1β humantakinogene hadenovec IL-1Ra (FX201) | Phase 1 AEs/biodistribution | NCT04119687 [62] 2020~Recruiting |
| Efficacy and Safety of XT-150 in Osteoarthritis of the Knee | Supply IL-10 plasmid DNA with a variant of human IL-10 transgene (XT-150) | Phase 2 KOOS/WOMAC /Others | NCT04124042 [63] 2020~Recruiting |
| A Single Dose Clinical Trial to Study the Safety of ART-I02 in Patients with Arthritis | Supply IFN-β Recombinant AAV type 2/5 containing a hIFN-b gene (ART-I02) | Phase 1 AEs/clinical scores distribution/immune response /Others | NCT02727764 [64] Active (2022) |
6. Oligonucleotide Therapeutics as a Candidate for OA Treatment by IA Injection
7. Discussion
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dillon, C.F.; Rasch, E.K.; Gu, Q.; Hirsch, R. Prevalence of Knee Osteoarthritis in the United States: Arthritis Data from the Third National Health and Nutrition Examination Survey 1991-94. J. Rheumatol. 2006, 33, 2271–2279. [Google Scholar]
- Yoshimura, N.; Muraki, S.; Oka, H.; Mabuchi, A.; En-Yo, Y.; Yoshida, M.; Saika, A.; Yoshida, H.; Suzuki, T.; Yamamoto, S.; et al. Prevalence of Knee Osteoarthritis, Lumbar Spondylosis, and Osteoporosis in Japanese Men and Women: The Research on Osteoarthritis/Osteoporosis against Disability Study. J. Bone Miner. Metab. 2009, 27, 620–628. [Google Scholar] [CrossRef]
- Silverwood, V.; Blagojevic-Bucknall, M.; Jinks, C.; Jordan, J.L.; Protheroe, J.; Jordan, K.P. Current Evidence on Risk Factors for Knee Osteoarthritis in Older Adults: A Systematic Review and Meta-Analysis. Osteoarthr. Cartil. 2015, 23, 507–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vina, E.R.; Kwoh, C.K. Epidemiology of Osteoarthritis: Literature Update. Curr. Opin. Rheumatol. 2018, 30, 160–167. [Google Scholar] [CrossRef]
- Bannuru, R.R.; Osani, M.C.; Vaysbrot, E.E.; Arden, N.K.; Bennell, K.; Bierma-Zeinstra, S.M.A.; Kraus, V.B.; Lohmander, L.S.; Abbott, J.H.; Bhandari, M.; et al. OARSI Guidelines for the Non-Surgical Management of Knee, Hip, and Polyarticular Osteoarthritis. Osteoarthr. Cartil. 2019, 27, 1578–1589. [Google Scholar] [CrossRef] [Green Version]
- Mathiessen, A.; Conaghan, P.G. Synovitis in Osteoarthritis: Current Understanding with Therapeutic Implications. Arthritis Res. Ther. 2017, 19, 18. [Google Scholar] [CrossRef] [Green Version]
- Rai, M.F.; Pham, C.T. Intra-Articular Drug Delivery Systems for Joint Diseases. Curr. Opin. Pharmacol. 2018, 40, 67–73. [Google Scholar] [CrossRef]
- Sellam, J.; Berenbaum, F. The Role of Synovitis in Pathophysiology and Clinical Symptoms of Osteoarthritis. Nat. Rev. Rheumatol. 2010, 6, 625–635. [Google Scholar] [CrossRef]
- Loeser, R.F.; Goldring, S.R.; Scanzello, C.R.; Goldring, M.B. Osteoarthritis: A Disease of the Joint as an Organ. Arthritis Rheumatol. 2012, 64, 1697–1707. [Google Scholar] [CrossRef] [Green Version]
- Sokolove, J.; Lepus, C.M. Role of Inflammation in the Pathogenesis of Osteoarthritis: Latest Findings and Interpretations. Ther. Adv. Musculoskelet. Dis. 2013, 5, 77–94. [Google Scholar] [CrossRef]
- Robinson, W.H.; Lepus, C.M.; Wang, Q.; Raghu, H.; Mao, R.; Lindstrom, T.M.; Sokolove, J. Low-Grade Inflammation as a Key Mediator of the Pathogenesis of Osteoarthritis. Nat. Rev. Rheumatol. 2016, 12, 580–592. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Shen, J.; Zhao, W.; Wang, T.; Han, L.; Hamilton, J.L.; Im, H.-J. Osteoarthritis: Toward a Comprehensive Understanding of Pathological Mechanism. Bone Res. 2017, 5, 1–13. [Google Scholar] [CrossRef]
- de Lange-Brokaar, B.J.E.; Ioan-Facsinay, A.; van Osch, G.J.V.M.; Zuurmond, A.-M.; Schoones, J.; Toes, R.E.M.; Huizinga, T.W.J.; Kloppenburg, M. Synovial Inflammation, Immune Cells and Their Cytokines in Osteoarthritis: A Review. Osteoarthr. Cartil. 2012, 20, 1484–1499. [Google Scholar] [CrossRef] [Green Version]
- Sohn, D.H.; Sokolove, J.; Sharpe, O.; Erhart, J.C.; Chandra, P.E.; Lahey, L.J.; Lindstrom, T.M.; Hwang, I.; Boyer, K.A.; Andriacchi, T.P.; et al. Plasma Proteins Present in Osteoarthritic Synovial Fluid Can Stimulate Cytokine Production via Toll-like Receptor 4. Arthritis Res. Ther. 2012, 14, R7. [Google Scholar] [CrossRef] [Green Version]
- Wojdasiewicz, P.; Poniatowski, Ł.A.; Szukiewicz, D. The Role of Inflammatory and Anti-Inflammatory Cytokines in the Pathogenesis of Osteoarthritis. Mediat. Inflamm. 2014, 2014, 561459. [Google Scholar] [CrossRef] [Green Version]
- Goldring, M.B. Chondrogenesis, Chondrocyte Differentiation, and Articular Cartilage Metabolism in Health and Osteoarthritis. Ther. Adv. Musculoskelet. Dis. 2012, 4, 269–285. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Lester, C.; Drapp, R.; Hu, D.Z.; Glimcher, L.H.; Jones, D. Tetraspanin CD9 and Ectonucleotidase CD73 Identify an Osteochondroprogenitor Population with Elevated Osteogenic Properties. Development 2015, 142, 438–443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minozzi, S.; Bonovas, S.; Lytras, T.; Pecoraro, V.; González-Lorenzo, M.; Bastiampillai, A.J.; Gabrielli, E.M.; Lonati, A.C.; Moja, L.; Cinquini, M.; et al. Risk of Infections Using Anti-TNF Agents in Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis: A Systematic Review and Meta-Analysis. Expert Opin. Drug Saf. 2016, 15, 11–34. [Google Scholar] [CrossRef]
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT00110916 (accessed on 31 January 2021).
- Chevalier, X.; Goupille, P.; Beaulieu, A.D.; Burch, F.X.; Bensen, W.G.; Conrozier, T.; Loeuille, D.; Kivitz, A.J.; Silver, D.; Appleton, B.E. Intraarticular Injection of Anakinra in Osteoarthritis of the Knee: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Study. Arthritis Rheumatol. 2009, 61, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Martel-Pelletier, J.; Pelletier, J.-P. A Single Injection of Anakinra for Treating Knee OA? Nat. Rev. Rheumatol. 2009, 5, 363–364. [Google Scholar] [CrossRef]
- Clinicaltrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03968913 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03275064 (accessed on 25 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01919164 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01111045 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01160822 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01144143 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT00819572 (accessed on 31 January 2021).
- Quartier, P.; Alexeeva, E.; Constantin, T.; Chasnyk, V.; Wulffraat, N.; Palmblad, K.; Wouters, C.; I Brunner, H.; Marzan, K.; Schneider, R.; et al. Tapering Canakinumab Monotherapy in Patients with Systemic Juvenile Idiopathic Arthritis in Clinical Remission: Results from a Phase IIIb/IV Open-Label, Randomized Study. Arthritis Rheumatol. 2021, 73, 336–346. [Google Scholar] [CrossRef]
- Schieker, M.; Conaghan, P.G.; Mindeholm, L.; Praestgaard, J.; Solomon, D.H.; Scotti, C.; Gram, H.; Thuren, T.; Roubenoff, R.; Ridker, P.M. Effects of Interleukin-1β Inhibition on Incident Hip and Knee Replacement: Exploratory Analyses From a Randomized, Double-Blind, Placebo-Controlled Trial. Ann. Intern. Med. 2020, 173, 509–515. [Google Scholar] [CrossRef]
- Caron, M.M.J.; Emans, P.J.; Cremers, A.; Surtel, D.A.M.; Coolsen, M.M.E.; van Rhijn, L.W.; Welting, T.J.M. Hypertrophic Differentiation during Chondrogenic Differentiation of Progenitor Cells Is Stimulated by BMP-2 but Suppressed by BMP-7. Osteoarthr. Cartil. 2013, 21, 604–613. [Google Scholar] [CrossRef] [Green Version]
- Hunter, D.J.; Pike, M.C.; Jonas, B.L.; Kissin, E.; Krop, J.; McAlindon, T. Phase 1 Safety and Tolerability Study of BMP-7 in Symptomatic Knee Osteoarthritis. BMC Musculoskelet. Disord. 2010, 11, 232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sreekumar, V.; Aspera-Werz, R.H.; Tendulkar, G.; Reumann, M.K.; Freude, T.; Breitkopf-Heinlein, K.; Dooley, S.; Pscherer, S.; Ochs, B.G.; Flesch, I.; et al. BMP9 a Possible Alternative Drug for the Recently Withdrawn BMP7? New Perspectives for (Re-)Implementation by Personalized Medicine. Arch. Toxicol. 2017, 91, 1353–1366. [Google Scholar] [CrossRef]
- Ellsworth, J.L.; Berry, J.; Bukowski, T.; Claus, J.; Feldhaus, A.; Holderman, S.; Holdren, M.S.; Lum, K.D.; Moore, E.E.; Raymond, F.; et al. Fibroblast Growth Factor-18 Is a Trophic Factor for Mature Chondrocytes and Their Progenitors. Osteoarthr. Cartil. 2002, 10, 308–320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mori, Y.; Saito, T.; Chang, S.H.; Kobayashi, H.; Ladel, C.H.; Guehring, H.; Chung, U.; Kawaguchi, H. Identification of Fibroblast Growth Factor-18 as a Molecule to Protect Adult Articular Cartilage by Gene Expression Profiling. J. Biol. Chem. 2014, 289, 10192–10200. [Google Scholar] [CrossRef] [Green Version]
- Dahlberg, L.E.; Aydemir, A.; Muurahainen, N.; Gühring, H.; Fredberg Edebo, H.; Krarup-Jensen, N.; Ladel, C.H.; Jurvelin, J.S. A First-in-Human, Double-Blind, Randomised, Placebo-Controlled, Dose Ascending Study of Intra-Articular RhFGF18 (Sprifermin) in Patients with Advanced Knee Osteoarthritis. Clin. Exp. Rheumatol. 2016, 34, 445–450. [Google Scholar]
- Hochberg, M.C.; Guermazi, A.; Guehring, H.; Aydemir, A.; Wax, S.; Fleuranceau-Morel, P.; Reinstrup Bihlet, A.; Byrjalsen, I.; Ragnar Andersen, J.; Eckstein, F. Effect of Intra-Articular Sprifermin vs Placebo on Femorotibial Joint Cartilage Thickness in Patients with Osteoarthritis: The FORWARD Randomized Clinical Trial. JAMA 2019, 322, 1360–1370. [Google Scholar] [CrossRef]
- Brett, A.; Bowes, M.A.; Conaghan, P.G.; Ladel, C.; Guehring, H.; Moreau, F.; Eckstein, F. Automated MRI Assessment Confirms Cartilage Thickness Modification in Patients with Knee Osteoarthritis: Post-Hoc Analysis from a Phase II Sprifermin Study. Osteoarthr. Cartil. 2020, 28, 1432–1436. [Google Scholar] [CrossRef]
- Eckstein, F.; Kraines, J.L.; Aydemir, A.; Wirth, W.; Maschek, S.; Hochberg, M.C. Intra-Articular Sprifermin Reduces Cartilage Loss in Addition to Increasing Cartilage Gain Independent of Location in the Femorotibial Joint: Post-Hoc Analysis of a Randomised, Placebo-Controlled Phase II Clinical Trial. Ann. Rheum. Dis. 2020, 79, 525–528. [Google Scholar] [CrossRef]
- Roemer, F.W.; Kraines, J.; Aydemir, A.; Wax, S.; Hochberg, M.C.; Crema, M.D.; Guermazi, A. Evaluating the Structural Effects of Intra-Articular Sprifermin on Cartilage and Non-Cartilaginous Tissue Alterations, Based on SqMRI Assessment over 2 Years. Osteoarthr. Cartil. 2020, 28, 1229–1234. [Google Scholar] [CrossRef]
- Scotti, C.; Gimbel, J.; Laurent, D.; Madar, A.; Peters, T.; Zhang, Y.; Polus, F.; Beste, M.; Vostiar, I.; Choudhury, S.; et al. LNA043, A Novel Cartilage Regenerative Treatment for Osteoarthritis. Arthritis Rheumatol. 2020, 72 (Suppl. 19), 1485. [Google Scholar]
- Candela, M.E.; Yasuhara, R.; Iwamoto, M.; Enomoto-Iwamoto, M. Resident Mesenchymal Progenitors of Articular Cartilage. Matrix Biol. 2014, 39, 44–49. [Google Scholar] [CrossRef]
- De Bari, C.; Dell’Accio, F.; Tylzanowski, P.; Luyten, F.P. Multipotent Mesenchymal Stem Cells from Adult Human Synovial Membrane. Arthritis Rheumatol. 2001, 44, 1928–1942. [Google Scholar] [CrossRef]
- Ok, S.M.; Kim, J.H.; Kim, J.S.; Jeong, E.G.; Park, Y.M.; Jeon, H.M.; Heo, J.Y.; Ahn, Y.W.; Yu, S.N.; Park, H.R.; et al. Local Injection of Growth Hormone for Temporomandibular Joint Osteoarthritis. Yonsei Med. J. 2020, 61, 331–340. [Google Scholar] [CrossRef]
- Lubis, A.M.T.; Wonggokusuma, E.; Marsetio, A.F. Intra-Articular Recombinant Human Growth Hormone In-jection Compared with Hyaluronic Acid and Placebo for an Osteoarthritis Model of New Zealand Rabbits. Knee Surg. Relat. Res. 2019, 31, 44–53. [Google Scholar] [CrossRef] [Green Version]
- Loffredo, F.S.; Pancoast, J.R.; Cai, L.; Vannelli, T.; Dong, J.Z.; Lee, R.T.; Patwari, P. Targeted Delivery to Carti-lage Is Critical for in Vivo Efficacy of Insulin-like Growth Factor 1 in a Rat Model of Osteoarthritis. Arthritis Rheumatol. 2014, 66, 1247–1255. [Google Scholar] [CrossRef]
- Ding, Z.; Lu, W.; Dai, C.; Huang, W.; Liu, F.; Shan, W.; Cheng, C.; Xu, J.; Yin, Z.; He, W. The CRD of Frizzled 7 Exhibits Chondroprotective Effects in Osteoarthritis via Inhibition of the Canonical Wnt3a/β-Catenin Signal-ing Pathway. Int. Immunopharmacol. 2020, 82, 106367. [Google Scholar] [CrossRef]
- Xu, C.; Zhu, S.; Wu, M.; Zhao, Y.; Han, W.; Yu, Y. The Therapeutic Effect of RhMK on Osteoarthritis in Mice, Induced by Destabilization of the Medial Meniscus. Biol. Pharm. Bull. 2014, 37, 1803–1810. [Google Scholar] [CrossRef] [Green Version]
- van Helvoort, E.M.; Popov-Celeketic, J.; Eijkelkamp, N.; Coeleveld, K.; Tryfonidou, M.A.; Wijne, C.D.; Hack, C.E.; Lafeber, F.P.J.G.; Mastbergen, S.C. Canine IL4-10 Fusion Protein Provides Disease Modifying Activity in a Canine Model of OA; an Exploratory Study. PLoS ONE 2019, 14, e0219587. [Google Scholar] [CrossRef] [Green Version]
- Steen-Louws, C.; Popov-Celeketic, J.; Mastbergen, S.C.; Coeleveld, K.; Hack, C.E.; Eijkelkamp, N.; Tryfonidou, M.; Spruijt, S.; van Roon, J.A.G.; Lafeber, F.P.J.G. IL4-10 Fusion Protein Has Chondroprotective, An-ti-Inflammatory and Potentially Analgesic Effects in the Treatment of Osteoarthritis. Osteoarthr. Cartil. 2018, 26, 1127–1135. [Google Scholar] [CrossRef] [Green Version]
- Chang, J.C.; Christiansen, B.A.; Murugesh, D.K.; Sebastian, A.; Hum, N.R.; Collette, N.M.; Hatsell, S.; Econo-mides, A.N.; Blanchette, C.D.; Loots, G.G. SOST/Sclerostin Improves Posttraumatic Osteoarthritis and Inhibits MMP2/3 Expression After Injury. J. Bone Miner. Res. 2018, 33, 1105–1113. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.-L.; Fu, W.; Ding, Y.-J.; Hettinghouse, A.; Lendhey, M.; Schwarzkopf, R.; Kennedy, O.D.; Liu, C.-J. Progranulin Derivative Atsttrin Protects against Early Osteoarthritis in Mouse and Rat Models. Arthritis Res. Ther. 2017, 19, 280. [Google Scholar] [CrossRef] [Green Version]
- Parrish, W.R.; Byers, B.A.; Su, D.; Geesin, J.; Herzberg, U.; Wadsworth, S.; Bendele, A.; Story, B. Intra-Articular Therapy with Recombinant Human GDF5 Arrests Disease Progression and Stimulates Cartilage Repair in the Rat Medial Meniscus Transection (MMT) Model of Osteoarthritis. Osteoarthr. Cartil. 2017, 25, 554–560. [Google Scholar] [CrossRef] [Green Version]
- Waller, K.A.; Chin, K.E.; Jay, G.D.; Zhang, L.X.; Teeple, E.; McAllister, S.; Badger, G.J.; Schmidt, T.A.; Fleming, B.C. Intra-Articular Recombinant Human Proteoglycan 4 Mitigates Cartilage Damage After Destabilization of the Medial Meniscus in the Yucatan Minipig. Am. J. Sports Med. 2017, 45, 1512–1521. [Google Scholar] [CrossRef]
- Jay, G.D.; Fleming, B.C.; Watkins, B.A.; McHugh, K.A.; Anderson, S.C.; Zhang, L.X.; Teeple, E.; Waller, K.A.; Elsaid, K.A. Prevention of Cartilage Degeneration and Restoration of Chondroprotection by Lubricin Tribo-supplementation in the Rat Following Anterior Cruciate Ligament Transection. Arthritis Rheumatol. 2010, 62, 2382–2391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiusaroli, R.; Visentini, M.; Galimberti, C.; Casseler, C.; Mennuni, L.; Covaceuszach, S.; Lanza, M.; Ugolini, G.; Caselli, G.; Rovati, L.C.; et al. Targeting of ADAMTS5′s Ancillary Domain with the Recombinant MAb CRB0017 Ameliorates Disease Progression in a Spontaneous Murine Model of Osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1807–1810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.-H.; Chang, C.-C.; Shieh, M.-J.; Wang, J.-P.; Chen, Y.-T.; Young, T.-H.; Hung, S.-C. Hypoxia Enhances Chondrogenesis and Prevents Terminal Differentiation through PI3K/Akt/FoxO Dependent Anti-Apoptotic Effect. Sci. Rep. 2013, 3, 2683. [Google Scholar] [CrossRef] [Green Version]
- Ulrich-Vinther, M.; Duch, M.R.; Søballe, K.; O’Keefe, R.J.; Schwarz, E.M.; Pedersen, F.S. In Vivo Gene Delivery to Articular Chondrocytes Mediated by an Adeno-Associated Virus Vector. J. Orthop. Res. 2004, 22, 726–734. [Google Scholar] [CrossRef]
- Watson, R.S.; Broome, T.A.; Levings, P.P.; Rice, B.L.; Kay, J.D.; Smith, A.D.; Gouze, E.; Gouze, J.-N.; Dacanay, E.A.; Hauswirth, W.W.; et al. ScAAV-Mediated Gene Transfer of Interleukin-1-Receptor Antagonist to Synovium and Articular Cartilage in Large Mammalian Joints. Gene Ther. 2013, 20, 670–677. [Google Scholar] [CrossRef] [Green Version]
- Evans, C.H.; Ghivizzani, S.C.; Robbins, P.D. Gene Delivery to Joints by Intra-Articular Injection. Hum. Gene Ther. 2018, 29, 2–14. [Google Scholar] [CrossRef]
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02790723 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://www.clinicaltrials.gov/ct2/show/NCT04119687 (accessed on 21 February 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04124042 (accessed on 31 January 2021).
- Clinicaltraials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02727764 (accessed on 31 January 2021).
- Nixon, A.J.; Grol, M.W.; Lang, H.M.; Ruan, M.Z.C.; Stone, A.; Begum, L.; Chen, Y.; Dawson, B.; Gannon, F.; Plutizki, S.; et al. Disease-Modifying Osteoarthritis Treatment with Interleukin-1 Receptor Antagonist Gene Therapy in Small and Large Animal Models. Arthritis Rheumatol. 2018, 70, 1757–1768. [Google Scholar] [CrossRef] [Green Version]
- Stone, A.; Grol, M.W.; Ruan, M.Z.C.; Dawson, B.; Chen, Y.; Jiang, M.-M.; Song, I.-W.; Jayaram, P.; Cela, R.; Gannon, F.; et al. Combinatorial Prg4 and Il-1ra Gene Therapy Protects Against Hyperalgesia and Cartilage Degeneration in Post-Traumatic Osteoarthritis. Hum. Gene Ther. 2019, 30, 225–235. [Google Scholar] [CrossRef]
- Nathwani, A.C.; Gray, J.T.; Ng, C.Y.C.; Zhou, J.; Spence, Y.; Waddington, S.N.; Tuddenham, E.G.D.; Kemball-Cook, G.; McIntosh, J.; Boon-Spijker, M.; et al. Self-Complementary Adeno-Associated Virus Vectors Containing a Novel Liver-Specific Human Factor IX Expression Cassette Enable Highly Efficient Transduction of Murine and Nonhuman Primate Liver. Blood 2006, 107, 2653–2661. [Google Scholar] [CrossRef]
- Wang, G.; Evans, C.H.; Benson, J.M.; Hutt, J.A.; Seagrave, J.; Wilder, J.A.; Grieger, J.C.; Samulski, R.J.; Terse, P.S. Safety and Biodistribution Assessment of Sc-RAAV2.5IL-1Ra Administered via Intra-Articular Injection in a Mono-Iodoacetate-Induced Osteoarthritis Rat Model. Mol. Ther. Methods Clin. Dev. 2016, 3, 15052. [Google Scholar] [CrossRef]
- Watson Levings, R.S.; Broome, T.A.; Smith, A.D.; Rice, B.L.; Gibbs, E.P.; Myara, D.A.; Hyddmark, E.V.; Nasri, E.; Zarezadeh, A.; Levings, P.P.; et al. Gene Therapy for Osteoarthritis: Pharmacokinetics of Intra-Articular Self-Complementary Adeno-Associated Virus Interleukin-1 Receptor Antagonist Delivery in an Equine Model. Hum. Gene Ther. Clin. Dev. 2018, 29, 90–100. [Google Scholar] [CrossRef]
- Katsikis, P.D.; Chu, C.Q.; Brennan, F.M.; Maini, R.N.; Feldmann, M. Immunoregulatory Role of Interleukin 10 in Rheumatoid Arthritis. J. Exp. Med. 1994, 179, 1517–1527. [Google Scholar] [CrossRef] [Green Version]
- Moroguchi, A.; Ishimura, K.; Okano, K.; Wakabayashi, H.; Maeba, T.; Maeta, H. Interleukin-10 Suppresses Proliferation and Remodeling of Extracellular Matrix of Cultured Human Skin Fibroblasts. Eur. Surg. Res. 2004, 36, 39–44. [Google Scholar] [CrossRef]
- Broeren, M.G.A.; de Vries, M.; Bennink, M.B.; Arntz, O.J.; van Lent, P.L.E.M.; van der Kraan, P.M.; van den Berg, W.B.; van den Hoogen, F.H.J.; Koenders, M.I.; van de Loo, F.A.J. Suppression of the Inflammatory Response by Disease-Inducible Interleukin-10 Gene Therapy in a Three-Dimensional Micromass Model of the Human Synovial Membrane. Arthritis Res. Ther. 2016, 18. [Google Scholar] [CrossRef] [Green Version]
- Watkins, L.R.; Chavez, R.A.; Landry, R.; Fry, M.; Green-Fulgham, S.M.; Coulson, J.D.; Collins, S.D.; Glover, D.K.; Rieger, J.; Forsayeth, J.R. Targeted Interleukin-10 Plasmid DNA Therapy in the Treatment of Osteoarthritis: Toxicology and Pain Efficacy Assessments. Brain Behav. Immun. 2020, 90, 155–166. [Google Scholar] [CrossRef]
- Aalbers, C.J.; Bevaart, L.; Loiler, S.; de Cortie, K.; Wright, J.F.; Mingozzi, F.; Tak, P.P.; Vervoordeldonk, M.J. Preclinical Potency and Biodistribution Studies of an AAV 5 Vector Expressing Human Interferon-β (ART-I02) for Local Treatment of Patients with Rheumatoid Arthritis. PLoS ONE 2015, 10, e0130612. [Google Scholar] [CrossRef] [Green Version]
- Bevaart, L.; Aalbers, C.J.; Vierboom, M.P.M.; Broekstra, N.; Kondova, I.; Breedveld, E.; Hauck, B.; Wright, J.F.; Tak, P.P.; Vervoordeldonk, M.J. Safety, Biodistribution, and Efficacy of an AAV-5 Vector Encoding Human Interferon-Beta (ART-I02) Delivered via Intra-Articular Injection in Rhesus Monkeys with Collagen-Induced Arthritis. Hum. Gene Ther. Clin. Dev. 2015, 26, 103–112. [Google Scholar] [CrossRef]
- Ruan, M.Z.; Cerullo, V.; Cela, R.; Clarke, C.; Lundgren-Akerlund, E.; Barry, M.A.; Lee, B.H. Treatment of Osteo-arthritis Using a Helper-Dependent Adenoviral Vector Retargeted to Chondrocytes. Mol. Ther. Methods Clin. Dev. 2016, 3, 16008. [Google Scholar] [CrossRef] [Green Version]
- Ortved, K.; Wagner, B.; Calcedo, R.; Wilson, J.; Schaefer, D.; Nixon, A. Humoral and Cell-Mediated Immune Response, and Growth Factor Synthesis after Direct Intraarticular Injection of RAAV2-IGF-I and RAAV5-IGF-I in the Equine Middle Carpal Joint. Hum. Gene Ther. 2015, 26, 161–171. [Google Scholar] [CrossRef] [Green Version]
- Setten, R.L.; Rossi, J.J.; Han, S.-P. The Current State and Future Directions of RNAi-Based Therapeutics. Nat. Rev. Drug Discov. 2019, 18, 421–446. [Google Scholar] [CrossRef]
- Sahin, U.; Karikó, K.; Türeci, Ö. MRNA-Based Therapeutics--Developing a New Class of Drugs. Nat. Rev. Drug Discov. 2014, 13, 759–780. [Google Scholar] [CrossRef]
- Bennett, C.F.; Krainer, A.R.; Cleveland, D.W. Antisense Oligonucleotide Therapies for Neurodegenerative Diseases. Annu. Rev. Neurosci. 2019, 42, 385–406. [Google Scholar] [CrossRef]
- Heo, Y.-A. Golodirsen: First Approval. Drugs 2020, 80, 329–333. [Google Scholar] [CrossRef]
- Ashizawa, A.T.; Cortes, J. Liposomal Delivery of Nucleic Acid-Based Anticancer Therapeutics: BP-100-1.01. Expert Opin. Drug Deliv. 2015, 12, 1107–1120. [Google Scholar] [CrossRef]
- Falzarano, M.S.; Passarelli, C.; Ferlini, A. Nanoparticle Delivery of Antisense Oligonucleotides and Their Application in the Exon Skipping Strategy for Duchenne Muscular Dystrophy. Nucleic Acid. Ther. 2014, 24, 87–100. [Google Scholar] [CrossRef] [Green Version]
- Mäe, M.; Andaloussi, S.E.; Lehto, T.; Langel, U. Chemically Modified Cell-Penetrating Peptides for the Delivery of Nucleic Acids. Expert Opin. Drug Deliv. 2009, 6, 1195–1205. [Google Scholar] [CrossRef]
- Vester, B.; Wengel, J. LNA (Locked Nucleic Acid): High-Affinity Targeting of Complementary RNA and DNA. Biochemistry 2004, 43, 13233–13241. [Google Scholar] [CrossRef]
- Dhillon, S. Viltolarsen: First Approval. Drugs 2020, 80, 1027–1031. [Google Scholar] [CrossRef]
- Hair, P.; Cameron, F.; McKeage, K. Mipomersen Sodium: First Global Approval. Drugs 2013, 73, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Hoy, S.M. Nusinersen: First Global Approval. Drugs 2017, 77, 473–479. [Google Scholar] [CrossRef]
- Hoy, S.M. Patisiran: First Global Approval. Drugs 2018, 78, 1625–1631. [Google Scholar] [CrossRef]
- Keam, S.J. Inotersen: First Global Approval. Drugs 2018, 78, 1371–1376. [Google Scholar] [CrossRef]
- Paik, J.; Duggan, S. Volanesorsen: First Global Approval. Drugs 2019, 79, 1349–1354. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.M.; Balfour, J.A. Fomivirsen. Drugs 1999, 57, 375–380. [Google Scholar] [CrossRef]
- Scott, L.J. Givosiran: First Approval. Drugs 2020, 80, 335–339. [Google Scholar] [CrossRef]
- Scott, L.J.; Keam, S.J. Lumasiran: First Approval. Drugs 2021. [Google Scholar] [CrossRef]
- Syed, Y.Y. Eteplirsen: First Global Approval. Drugs 2016, 76, 1699–1704. [Google Scholar] [CrossRef]
- Steinberg, J.; Ritchie, G.R.S.; Roumeliotis, T.I.; Jayasuriya, R.L.; Clark, M.J.; Brooks, R.A.; Binch, A.L.A.; Shah, K.M.; Coyle, R.; Pardo, M.; et al. Integrative Epigenomics, Transcriptomics and Proteomics of Patient Chon-drocytes Reveal Genes and Pathways Involved in Osteoarthritis. Sci. Rep. 2017, 7, 8935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Ning, Y.; Zhou, B.; Yang, L.; Wang, Y.; Guo, X. Integrated Bioinformatics Analysis of the Osteoarthri-tis-associated MicroRNA Expression Signature. Mol. Med. Rep. 2018, 17, 1833–1838. [Google Scholar] [CrossRef] [Green Version]
- Peffers, M.; Liu, X.; Clegg, P. Transcriptomic Signatures in Cartilage Ageing. Arthritis Res. Ther. 2013, 15, R98. [Google Scholar] [CrossRef] [Green Version]
- Malemud, C.J. MicroRNAs and Osteoarthritis. Cells 2018, 7, 72. [Google Scholar] [CrossRef]
- Sondag, G.R.; Haqqi, T.M. The Role of MicroRNAs and Their Targets in Osteoarthritis. Curr. Rheumatol. Rep. 2016, 18, 56. [Google Scholar] [CrossRef]
- Wu, C.; Tian, B.; Qu, X.; Liu, F.; Tang, T.; Qin, A.; Zhu, Z.; Dai, K. MicroRNAs Play a Role in Chondrogenesis and Osteoarthritis (Review). Int. J. Mol. Med. 2014, 34, 13–23. [Google Scholar] [CrossRef] [Green Version]
- Ji, M.-L.; Jiang, H.; Wu, F.; Geng, R.; Ya, L.K.; Lin, Y.C.; Xu, J.H.; Wu, X.T.; Lu, J. Precise Targeting of MiR-141/200c Cluster in Chondrocytes Attenuates Osteoarthritis Development. Ann. Rheum. Dis. 2020. [Google Scholar] [CrossRef]
- Tian, L.; Su, Z.; Ma, X.; Wang, F.; Guo, Y. Inhibition of MiR-203 Ameliorates Osteoarthritis Cartilage Degrada-tion in the Postmenopausal Rat Model: Involvement of Estrogen Receptor α. Hum. Gene Ther. Clin. Dev. 2019, 30, 160–168. [Google Scholar] [CrossRef]
- Wang, X.-B.; Zhao, F.-C.; Yi, L.-H.; Tang, J.-L.; Zhu, Z.-Y.; Pang, Y.; Chen, Y.-S.; Li, D.-Y.; Guo, K.-J.; Zheng, X. MicroRNA-21-5p as a Novel Therapeutic Target for Osteoarthritis. Rheumatology 2019. [Google Scholar] [CrossRef]
- Endisha, H.; Datta, P.; Sharma, A.; Nakamura, S.; Rossomacha, E.; Younan, C.; Ali, S.A.; Tavallaee, G.; Lively, S.; Potla, P.; et al. MicroRNA-34a-5p Promotes Joint Destruction During Osteoarthritis. Arthritis Rheumatol. 2020. [Google Scholar] [CrossRef]
- Liu, X.; Liu, L.; Zhang, H.; Shao, Y.; Chen, Z.; Feng, X.; Fang, H.; Zhao, C.; Pan, J.; Zhang, H.; et al. MiR-146b Accelerates Osteoarthritis Progression by Targeting Alpha-2-Macroglobulin. Aging 2019, 11, 6014–6028. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Rampersaud, Y.R.; Nakamura, S.; Sharma, A.; Zeng, F.; Rossomacha, E.; Ali, S.A.; Krawetz, R.; Haroon, N.; Perruccio, A.V.; et al. MicroRNA-181a-5p Antisense Oligonucleotides Attenuate Osteoarthritis in Facet and Knee Joints. Ann. Rheum. Dis. 2019, 78, 111–121. [Google Scholar] [CrossRef]
- Lian, W.-S.; Ko, J.-Y.; Wu, R.-W.; Sun, Y.-C.; Chen, Y.-S.; Wu, S.-L.; Weng, L.-H.; Jahr, H.; Wang, F.-S. Mi-croRNA-128a Represses Chondrocyte Autophagy and Exacerbates Knee Osteoarthritis by Disrupting Atg12. Cell Death Dis. 2018, 9, 919. [Google Scholar] [CrossRef]
- Baek, D.; Lee, K.-M.; Park, K.W.; Suh, J.W.; Choi, S.M.; Park, K.H.; Lee, J.W.; Kim, S.-H. Inhibition of MiR-449a Promotes Cartilage Regeneration and Prevents Progression of Osteoarthritis in In Vivo Rat Models. Mol. Ther. Nucleic Acids 2018, 13, 322–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pi, Y.; Zhang, X.; Shao, Z.; Zhao, F.; Hu, X.; Ao, Y. Intra-Articular Delivery of Anti-Hif-2α SiRNA by Chondro-cyte-Homing Nanoparticles to Prevent Cartilage Degeneration in Arthritic Mice. Gene Ther. 2015, 22, 439–448. [Google Scholar] [CrossRef]
- Hoshi, H.; Akagi, R.; Yamaguchi, S.; Muramatsu, Y.; Akatsu, Y.; Yamamoto, Y.; Sasaki, T.; Takahashi, K.; Sasho, T. Effect of Inhibiting MMP13 and ADAMTS5 by Intra-Articular Injection of Small Interfering RNA in a Sur-gically Induced Osteoarthritis Model of Mice. Cell Tissue Res. 2017, 368, 379–387. [Google Scholar] [CrossRef]
- Nakagawa, R.; Akagi, R.; Yamaguchi, S.; Enomoto, T.; Sato, Y.; Kimura, S.; Ogawa, Y.; Sadamasu, A.; Ohtori, S.; Sasho, T. Single vs. Repeated Matrix Metalloproteinase-13 Knockdown with Intra-Articular Short Interfering RNA Administration in a Murine Osteoarthritis Model. Connect. Tissue Res. 2019, 60, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Li, S.-J.; Liu, R.; Zhan, J.-F.; Tan, C.; Fang, Y.-F.; Chen, Y.; Yu, B. Inhibition of YAP with SiRNA Pre-vents Cartilage Degradation and Ameliorates Osteoarthritis Development. J. Mol. Med. 2019, 97, 103–114. [Google Scholar] [CrossRef]
- Li, L.; Li, M.; Pang, Y.; Wang, J.; Wan, Y.; Zhu, C.; Yin, Z. Abnormal Thyroid Hormone Receptor Signaling in Osteoarthritic Osteoblasts Regulates Microangiogenesis in Subchondral Bone. Life Sci. 2019, 239, 116975. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, Y.; Zhang, H.; Gao, W.; Lu, M.; Liu, W.; Li, Y.; Yin, Z. Forkhead Box C1 Promotes the Pathology of Osteoarthritis by Upregulating β-Catenin in Synovial Fibroblasts. FEBS J. 2020, 287, 3065–3087. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, Y.; Nakasa, T.; Shoji, T.; Hamanishi, M.; Shimizu, R.; Kamei, N.; Usman, M.A.; Ochi, M. In-tra-Articular Injection of Synthetic MicroRNA-210 Accelerates Avascular Meniscal Healing in Rat Medial Meniscal Injured Model. Arthritis Res. Ther. 2014, 16. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Wang, Z.; Pan, Y.; Ma, J.; Miao, X.; Qi, X.; Zhou, H.; Jia, L. MiR-26a and MiR-26b Mediate Osteoarthritis Progression by Targeting FUT4 via NF-ΚB Signaling Pathway. Int. J. Biochem. Cell Biol. 2018, 94, 79–88. [Google Scholar] [CrossRef]
- Hu, G.; Zhao, X.; Wang, C.; Geng, Y.; Zhao, J.; Xu, J.; Zuo, B.; Zhao, C.; Wang, C.; Zhang, X. MicroRNA-145 At-tenuates TNF-α-Driven Cartilage Matrix Degradation in Osteoarthritis via Direct Suppression of MKK4. Cell Death Dis. 2017, 8, e3140. [Google Scholar] [CrossRef]
- Si, H.-B.; Zeng, Y.; Liu, S.-Y.; Zhou, Z.-K.; Chen, Y.-N.; Cheng, J.-Q.; Lu, Y.-R.; Shen, B. Intra-Articular Injection of MicroRNA-140 (MiRNA-140) Alleviates Osteoarthritis (OA) Progression by Modulating Extracellular Ma-trix (ECM) Homeostasis in Rats. Osteoarthr. Cartil. 2017, 25, 1698–1707. [Google Scholar] [CrossRef] [Green Version]
- Brown, S.; Kumar, S.; Sharma, B. Intra-Articular Targeting of Nanomaterials for the Treatment of Osteoarthri-tis. Acta Biomater. 2019, 93, 239–257. [Google Scholar] [CrossRef] [PubMed]
- Almagro, J.C.; Daniels-Wells, T.R.; Perez-Tapia, S.M.; Penichet, M.L. Progress and Challenges in the Design and Clinical Development of Antibodies for Cancer Therapy. Front. Immunol. 2017, 8, 1751. [Google Scholar] [CrossRef] [Green Version]
- Veronese, F.M.; Mero, A. The Impact of PEGylation on Biological Therapies. BioDrugs 2008, 22, 315–329. [Google Scholar] [CrossRef]
- Liang, X.; Chen, Y.; Wu, L.; Maharjan, A.; Regmi, B.; Zhang, J.; Gui, S. In Situ Hexagonal Liquid Crystal for In-tra-Articular Delivery of Sinomenine Hydrochloride. Biomed. Pharmacother. 2019, 117, 108993. [Google Scholar] [CrossRef]
- Roberts, T.C.; Langer, R.; Wood, M.J.A. Advances in Oligonucleotide Drug Delivery. Nat. Rev. Drug Discov. 2020, 19, 673–694. [Google Scholar] [CrossRef]
- Geary, R.S.; Norris, D.; Yu, R.; Bennett, C.F. Pharmacokinetics, Biodistribution and Cell Uptake of Antisense Oligonucleotides. Adv. Drug Deliv. Rev. 2015, 87, 46–51. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Prasadam, I.; Fang, W.; Crawford, R.; Xiao, Y. Chondromodulin-1 Ameliorates Osteoarthritis Pro-gression by Inhibiting HIF-2α Activity. Osteoarthr. Cartil. 2016, 24, 1970–1980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, X.; You, H.; Yuan, X.; Zhao, W.; Li, W.; Guo, X. Protective Effect of Lentivirus-Mediated SiRNA Targeting ADAMTS-5 on Cartilage Degradation in a Rat Model of Osteoarthritis. Int. J. Mol. Med. 2013, 31, 1222–1228. [Google Scholar] [CrossRef] [Green Version]
- Ji, Q.; Xu, X.; Kang, L.; Xu, Y.; Xiao, J.; Goodman, S.B.; Zhu, X.; Li, W.; Liu, J.; Gao, X.; et al. Hematopoietic PBX-Interacting Protein Mediates Cartilage Degeneration during the Pathogenesis of Osteoarthritis. Nat. Commun. 2019, 10, 313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Crawford, R.; Xiao, Y. Inhibition of Vascular Endothelial Growth Factor with ShRNA in Chondro-cytes Ameliorates Osteoarthritis. J. Mol. Med. (Berl.) 2016, 94, 787–798. [Google Scholar] [CrossRef]
- Peng, J.-S.; Chen, S.-Y.; Wu, C.-L.; Chong, H.-E.; Ding, Y.-C.; Shiau, A.-L.; Wang, C.-R. Amelioration of Experi-mental Autoimmune Arthritis Through Targeting of Synovial Fibroblasts by Intraarticular Delivery of Mi-croRNAs 140-3p and 140-5p. Arthritis Rheumatol. 2016, 68, 370–381. [Google Scholar] [CrossRef] [Green Version]
- Song, J.; Kim, D.; Chun, C.-H.; Jin, E.-J. MiR-370 and MiR-373 Regulate the Pathogenesis of Osteoarthritis by Modulating One-Carbon Metabolism via SHMT-2 and MECP-2, Respectively. Aging Cell 2015, 14, 826–837. [Google Scholar] [CrossRef]
- Yan, S.; Wang, M.; Zhao, J.; Zhang, H.; Zhou, C.; Jin, L.; Zhang, Y.; Qiu, X.; Ma, B.; Fan, Q. MicroRNA-34a Af-fects Chondrocyte Apoptosis and Proliferation by Targeting the SIRT1/P53 Signaling Pathway during the Pathogenesis of Osteoarthritis. Int. J. Mol. Med. 2016, 38, 201–209. [Google Scholar] [CrossRef] [Green Version]
- Anguela, X.M.; High, K.A. Entering the Modern Era of Gene Therapy. Annu. Rev. Med. 2019, 70, 273–288. [Google Scholar] [CrossRef] [Green Version]
- Brown, T.D.; Johnston, R.C.; Saltzman, C.L.; Marsh, J.L.; Buckwalter, J.A. Posttraumatic Osteoarthritis: A First Estimate of Incidence, Prevalence, and Burden of Disease. J. Orthop. Trauma 2006, 20, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Kuyinu, E.L.; Narayanan, G.; Nair, L.S.; Laurencin, C.T. Animal Models of Osteoarthritis: Classification, Up-date, and Measurement of Outcomes. J. Orthop. Surg. Res. 2016, 11. [Google Scholar] [CrossRef] [Green Version]
- Bijlsma, J.W.J.; Berenbaum, F.; Lafeber, F.P.J.G. Osteoarthritis: An Update with Relevance for Clinical Practice. Lancet 2011, 377, 2115–2126. [Google Scholar] [CrossRef]
- Glyn-Jones, S.; Palmer, A.J.R.; Agricola, R.; Price, A.J.; Vincent, T.L.; Weinans, H.; Carr, A.J. Osteoarthritis. Lancet 2015, 386, 376–387. [Google Scholar] [CrossRef]
- Juras, V.; Chang, G.; Regatte, R.R. Current Status of Functional MRI of Osteoarthritis for Diagnosis and Prog-nosis. Curr. Opin. Rheumatol. 2020, 32, 102–109. [Google Scholar] [CrossRef]
| Protein | OA Animal Models | References | |
|---|---|---|---|
| GH | Growth hormone | Rat TMJ-MIA model | [44] |
| Rabbit collagenase injection model | [45] | ||
| HB-IGF-1 | Humanized insulin like growth factor-1 fusion protein with a heparin-binding domain for targeting to cartilage | Rat MMx model | [46] |
| FzD7 CRD | Recombinant-Frizzled 7-cysteine-rich domain designed to inhibit Wnt3a/β-catenin signaling | Mouse DMM model | [47] |
| rhMidkine | rhMidkine | Mouse DMM model | [48] |
| IL4-10 FP | A fusion protein, the biological activity of IL-4 and IL-10 are preserved | Canine groove model | [49,50] |
| Sclerostin | Mouse tibial compression OA injury model | [51] | |
| Atsttrin | An engineered protein composed of three tumor necrosis factor receptor (TNFR)-binding fragments of progranulin (PGRN) | Rat noninvasive ACL rupture model mouse ACLT model | [52] |
| rhGDF5 | rh growth differentiation factor-5 | Rat MMx model | [53] |
| rhPRG4 | rh lubricin | Yucatan minipigs DMM model | [54] |
| Rat ACLT model | [55] | ||
| CRB0017 | recombinant monoclonal antibodies directed against ADAMTS5 | STR/ort | [56] |
| Mode of Action | Oligonucleotide | Target Gene(s) | Outcomes | References |
|---|---|---|---|---|
| miRNA inhibitor | miRNA inhibitors antisense oligonucleotide | miR-141/200c | Recover SIRT1/modify IL-6/STAT3 pathway/prevent OA in mouse DMM model | [102] |
| miR-203 | Recover Erα/decrease cartilage degradation in postmenopausal OA rats | [103] | ||
| miR-21-5p | Recover FGF18/attenuate the severity of OA in the mouse DMM model | [104] | ||
| miR-34-5a | Protect cartilage in the DMM and high-fat diet/DMM mice | [105] | ||
| miR-146b | Recover α2-macroglobulin/ prevent OA in mouse DMM model | [106] | ||
| miR-181a-5p | Attenuate cartilage destruction, hypertrophic, apoptotic/cell death, and type II collagen breakdown markers in mouse DMM model | [107] | ||
| miR-128a | Recover ATG12/slow articular tissue destruction in rat ACLT model | [108] | ||
| miR-449a | Recover SIRT 1/Prevent cartilage degradation in rat DMM model | [109] | ||
| mRNA inhibition | siRNA for target genes | Hif-2α | Prevent cartilage degeneration in ACLT/DMM mice | [110] |
| Mmp13 | Delay cartilage degradation in mouse DMM model | [111,112] | ||
| Yap | Ameliorate OA development and reduce subchondral bone formation in ACLT mice | [113] | ||
| Thr | Reduce angiogenic activities in subchondral bone ameliorated cartilage degradation in mouse DMM model | [114] | ||
| FoxC1 | Decrease β-catenin, ADAMTS-5, fibronectin, MMP3, and MMP13/decrease cartilage destruction in mouse DMM model | [115] | ||
| miRNA supplement | miR-210 mimic | Not mentioned | Upregulate Col2a1 expression in the meniscus cells and VEGF and FGF2 expression in the synovial cell/enhance repair of the meniscus and prevent cartilage degeneration in rat DMM model | [116] |
| miR-26a/26b mimic | Fut4 | Promote chondrocytes proliferation and inhibit apoptosis/attenuate OA progression in rat ACLT-MMx model | [117] | |
| miR-145 mimic | Mkk4 | Suppress the expression of MMP-3 and MMP-13, as well as p-MKK4, p-c-Jun, and p-ATF2/reduce cartilage destruction in rat MCLT-DMM model | [118] | |
| miR-140 mimic | Not mentioned | Reduce pathological scores and MMP-13 and ADAMTS-5 expression in rat ACLT-MMx model | [119] |
| Protein Therapeutics | Gene Therapy | Oligonucleotide Therapeutics | |
|---|---|---|---|
| Mechanism of action | Supply the required protein | Transduce target gene in cells Gene expression and translation is needed | Transfer to target cells Modulate the function or the fate of target mRNA |
| Application range of targets | For proteins that act extracellularly | Limitation in size of gene | Oligonucleotide sequence can be designed without off target effect |
| Delivery and distribution | Distribute to whole joint by IA | AAV vector provide cell type specific gene transfer | Drug delivery system for target need to be established |
| Retention time | Short Rapidly excreted from the joint | Vector: Continuous expression can be expected when transfected cells and vector retained mRNA: transient protein production | Effect will continue as long as oligonucleotide remain in cytosol |
| Control dosage and time | Possible | Amount of protein depend on transfection efficiency, host cell activity and etc. Promotor design provide regulated induction of protein | Possible |
| Relative manufacturing cost | High Biological manufacturing | High Biological manufacturing | Low Chemical synthesis” |
| Technical establishment and Safety concerns | Established Predictable | Approved mainly on life threatening disease Remain unknown risks? | Approved mainly in specific genetical disorder Remain unknown risks? |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toyoda, E.; Maehara, M.; Watanabe, M.; Sato, M. Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis. Int. J. Mol. Sci. 2021, 22, 3594. https://doi.org/10.3390/ijms22073594
Toyoda E, Maehara M, Watanabe M, Sato M. Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis. International Journal of Molecular Sciences. 2021; 22(7):3594. https://doi.org/10.3390/ijms22073594
Chicago/Turabian StyleToyoda, Eriko, Miki Maehara, Masahiko Watanabe, and Masato Sato. 2021. "Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis" International Journal of Molecular Sciences 22, no. 7: 3594. https://doi.org/10.3390/ijms22073594







