Lipofilling in Breast Oncological Surgery: A Safe Opportunity or Risk for Cancer Recurrence?
Abstract
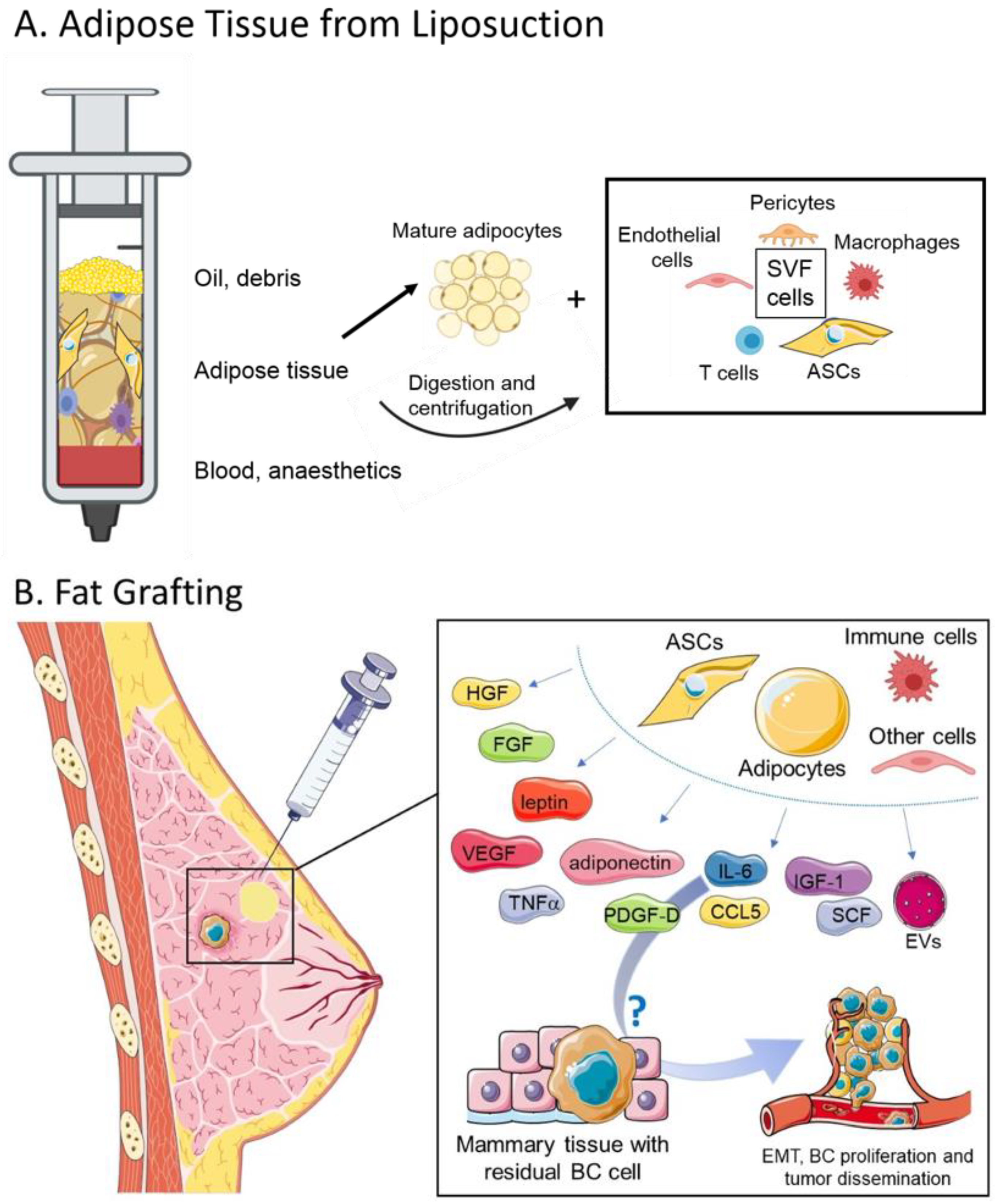
:1. Introduction
2. Lipofilling of the Breast
2.1. History and Surgical Practice
2.2. Cosmetic Success after Breast Surgery
2.3. Impact on Cancer Recurrence in BC Patients
3. Adipose Tissue: Composition and Functions
3.1. Composition of the Adipose Tissue
3.2. ASCs and the Regenerative Property of Fat Grafting
4. Adipose Tissue and BC Crosstalk
5. Oncologic Safety of Breast Lipofilling: A Still Debated Issue
5.1. Effect of Lipoaspirate on BC Cells In Vitro
5.2. In Vivo Studies on Animal Models of BC
6. Open Questions
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bircoll, M. Cosmetic breast augmentation utilizing autologous fat and liposuction techniques. Plast. Reconstr. Surg. 1987, 79, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Sekigami, Y.; Schwartz, T.; Losken, A.; Margenthaler, J.; Chatterjee, A. Lipofilling after breast conserving surgery: A comprehensive literature review investigating its oncologic safety. Gland Surg. 2019, 8, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Cohn, A.B.; Restrepo, E.R.D. Fat transfer in breast reconstruction: A vital reconstructive tool and the importance of postoperative multidisciplinary collaboration. Breast J. 2020, 26, 845–846. [Google Scholar] [CrossRef]
- Granoff, M.D.; Guo, L.; Singhal, D. Lipofilling after breast conserving surgery: A plastic surgery perspective. Gland Surg. 2020, 9, 617–619. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Daquinag, A.; Traktuev, D.O.; Amaya-Manzanares, F.; Simmons, P.J.; March, K.L.; Pasqualini, R.; Arap, W.; Kolonin, M.G. White adipose tissue cells are recruited by experimental tumors and promote cancer progression in mouse models. Cancer Res. 2009, 69, 5259–5266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vona-Davis, L.; Rose, D.P. Adipokines as endocrine, paracrine, and autocrine factors in breast cancer risk and progression. Endocr. Relat. Cancer 2007, 14, 189–206. [Google Scholar] [CrossRef]
- Lamszus, K.; Jin, L.; Fuchs, A.; Shi, E.; Chowdhury, S.; Yao, Y.; Polverini, P.J.; Laterra, J.; Goldberg, I.D.; Rosen, E.M. Scatter factor stimulates tumor growth and tumor angiogenesis in human breast cancers in the mammary fat pads of nude mice. Lab. Investig. J. Tech. Methods Pathol. 1997, 76, 339–353. [Google Scholar]
- Manabe, Y.; Toda, S.; Miyazaki, K.; Sugihara, H. Mature adipocytes, but not preadipocytes, promote the growth of breast carcinoma cells in collagen gel matrix culture through cancer-stromal cell interactions. J. Pathol. 2003, 201, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Schäffler, A.; Schölmerich, J.; Buechler, C. Mechanisms of disease: Adipokines and breast cancer—Endocrine and paracrine mechanisms that connect adiposity and breast cancer. Nat. Clin. Pract. Endocrinol. Metab. 2007, 3, 345–354. [Google Scholar] [CrossRef]
- Iyengar, P.; Espina, V.; Williams, T.W.; Lin, Y.; Berry, D.; Jelicks, L.A.; Lee, H.; Temple, K.; Graves, R.; Pollard, J.; et al. Adipocyte-derived collagen VI affects early mammary tumor progression in vivo, demonstrating a critical interaction in the tumor/stroma microenvironment. J. Clin. Investig. 2005, 115, 1163–1176. [Google Scholar] [CrossRef] [Green Version]
- Motrescu, E.R.; Rio, M.-C. Cancer cells, adipocytes and matrix metalloproteinase 11: A vicious tumor progression cycle. Biol. Chem. 2008, 389, 1037–1041. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, N.; Tremblay, E.; McAdam, L.; Roberts, A.; Elliott, B. Autocrine secretion of TGF-beta 1 and TGF-beta 2 by pre-adipocytes and adipocytes: A potent negative regulator of adipocyte differentiation and proliferation of mammary carcinoma cells. In Vitro Cell. Dev. Biol. Anim. 1998, 34, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Tayeh, S.; Muktar, S.; Wazir, U.; Carmichael, A.R.; Al-Fardan, Z.; Kasem, A.; Hamdi, M.; Mokbel, K. Is Autologous Fat Grafting an Oncologically Safe Procedure following Breast Conserving Surgery for Breast Cancer? A Comprehensive Review. J. Investig. Surg. Off. J. Acad. Surg. Res. 2020, 1–10. [Google Scholar] [CrossRef]
- Holländer, E. Report on a case of continuously disappearing fatty tissue and its cosmetic substitution through human fat. Münch. Med. Wochenschr. 1910, 57, 1794–1795. [Google Scholar]
- Coleman, S.R.; Saboeiro, A.P. Fat grafting to the breast revisited: Safety and efficacy. Plast. Reconstr. Surg. 2007, 119, 775–785. [Google Scholar] [CrossRef] [Green Version]
- Delay, E.; Garson, S.; Tousson, G.; Sinna, R. Fat injection to the breast: Technique, results, and indications based on 880 procedures over 10 years. Aesthet. Surg. J. 2009, 29, 360–376. [Google Scholar] [CrossRef] [PubMed]
- Rigotti, G.; Marchi, A.; Galiè, M.; Baroni, G.; Benati, D.; Krampera, M.; Pasini, A.; Sbarbati, A. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: A healing process mediated by adipose-derived adult stem cells. Plast. Reconstr. Surg. 2007, 119, 1409–1422. [Google Scholar] [CrossRef]
- Rietjens, M.; De Lorenzi, F.; Rossetto, F.; Brenelli, F.; Manconi, A.; Martella, S.; Intra, M.; Venturino, M.; Lohsiriwat, V.; Ahmed, Y.; et al. Safety of fat grafting in secondary breast reconstruction after cancer. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2011, 64, 477–483. [Google Scholar] [CrossRef]
- Kasem, A.; Wazir, U.; Headon, H.; Mokbel, K. Breast lipofilling: A review of current practice. Arch. Plast. Surg. 2015, 42, 126–130. [Google Scholar] [CrossRef] [Green Version]
- Hamza, A.; Lohsiriwat, V.; Rietjens, M. Lipofilling in breast cancer surgery. Gland Surg. 2013, 2, 7–14. [Google Scholar] [CrossRef]
- Klein, J.A. Tumescent technique for local anesthesia improves safety in large-volume liposuction. Plast. Reconstr. Surg. 1993, 92, 1085–1098; discussion 1099–1100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agostini, T.; Lazzeri, D.; Pini, A.; Marino, G.; Li Quattrini, A.; Bani, D.; Dini, M. Wet and dry techniques for structural fat graft harvesting: Histomorphometric and cell viability assessments of lipoaspirated samples. Plast. Reconstr. Surg. 2012, 130, 331e–339e. [Google Scholar] [CrossRef] [PubMed]
- Coleman, S.R. Structural fat grafting: More than a permanent filler. Plast. Reconstr. Surg. 2006, 118 (Suppl. 3), 108S–120S. [Google Scholar] [CrossRef] [Green Version]
- Erdim, M.; Tezel, E.; Numanoglu, A.; Sav, A. The effects of the size of liposuction cannula on adipocyte survival and the optimum temperature for fat graft storage: An experimental study. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2009, 62, 1210–1214. [Google Scholar] [CrossRef]
- Keck, M.; Kober, J.; Riedl, O.; Kitzinger, H.B.; Wolf, S.; Stulnig, T.M.; Zeyda, M.; Gugerell, A. Power assisted liposuction to obtain adipose-derived stem cells: Impact on viability and differentiation to adipocytes in comparison to manual aspiration. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2014, 67, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Shridharani, S.M.; Broyles, J.M.; Matarasso, A. Liposuction devices: Technology update. Med. Devices Auckl. N. Z. 2014, 7, 241–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagy, M.W.; Vanek, P.F. A multicenter, prospective, randomized, single-blind, controlled clinical trial comparing VASER-assisted Lipoplasty and suction-assisted Lipoplasty. Plast. Reconstr. Surg. 2012, 129, 681e–689e. [Google Scholar] [CrossRef] [PubMed]
- Cimino, W.W. The physics of soft tissue fragmentation using ultrasonic frequency vibration of metal probes. Clin. Plast. Surg. 1999, 26, 447–461. [Google Scholar] [CrossRef]
- Chung, M.T.; Zimmermann, A.S.; Paik, K.J.; Morrison, S.D.; Hyun, J.S.; Lo, D.D.; McArdle, A.; Montoro, D.T.; Walmsley, G.G.; Senarath-Yapa, K.; et al. Isolation of human adipose-derived stromal cells using laser-assisted liposuction and their therapeutic potential in regenerative medicine. Stem Cells Transl. Med. 2013, 2, 808–817. [Google Scholar] [CrossRef]
- Coleman, S.R. Facial augmentation with structural fat grafting. Clin. Plast. Surg. 2006, 33, 567–577. [Google Scholar] [CrossRef]
- Fournier, P.F. Fat grafting: My technique. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. Al 2000, 26, 1117–1128. [Google Scholar] [CrossRef]
- Quoc, C.H.; Foyatier, J.-L.; Meruta, A.; Piat, J.-M.; Michel, G.; Delay, E. Lipofilling, an efficient solution for breast sequelae after cardiothoracic surgery. Ann. Chir. Plast. Esthet. 2015, 60, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Sarfati, I.; Ihrai, T.; Kaufman, G.; Nos, C.; Clough, K.B. Adipose-tissue grafting to the post-mastectomy irradiated chest wall: Preparing the ground for implant reconstruction. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2011, 64, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Salgarello, M.; Visconti, G.; Barone-Adesi, L. Fat grafting and breast reconstruction with implant: Another option for irradiated breast cancer patients. Plast. Reconstr. Surg. 2012, 129, 317–329. [Google Scholar] [CrossRef]
- Andrews, G.; Ahsan, M.D.; Tabrizi, A.R.; Razzouk, K.; Fitoussi, A. Long term surgical outcome of lipofilling based Breast reconstruction after mastectomy and radiotherapy: Clinical retrospective Descriptive study. Eur. J. Surg. Oncol. 2021, 47, E44–E45. [Google Scholar] [CrossRef]
- Ribuffo, D.; Atzeni, M.; Guerra, M.; Bucher, S.; Politi, C.; Deidda, M.; Atzori, F.; Dessi, M.; Madeddu, C.; Lay, G. Treatment of irradiated expanders: Protective lipofilling allows immediate prosthetic breast reconstruction in the setting of postoperative radiotherapy. Aesthetic Plast. Surg. 2013, 37, 1146–1152. [Google Scholar] [CrossRef]
- Panettiere, P.; Marchetti, L.; Accorsi, D. The serial free fat transfer in irradiated prosthetic breast reconstructions. Aesthetic Plast. Surg. 2009, 33, 695–700. [Google Scholar] [CrossRef]
- Serra-Renom, J.M.; Muñoz-Olmo, J.L.; Serra-Mestre, J.M. Fat grafting in postmastectomy breast reconstruction with expanders and prostheses in patients who have received radiotherapy: Formation of new subcutaneous tissue. Plast. Reconstr. Surg. 2010, 125, 12–18. [Google Scholar] [CrossRef]
- Sommeling, C.E.; Van Landuyt, K.; Depypere, H.; Van den Broecke, R.; Monstrey, S.; Blondeel, P.N.; Morrison, W.A.; Stillaert, F.B. Composite breast reconstruction: Implant-based breast reconstruction with adjunctive lipofilling. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2017, 70, 1051–1058. [Google Scholar] [CrossRef]
- Stillaert, F.B.J.L.; Sommeling, C.; D’Arpa, S.; Creytens, D.; Van Landuyt, K.; Depypere, H.; Van den Broecke, R.; Monstrey, S.; Blondeel, P.N.; Morrison, W.A. Intratissular expansion-mediated, serial fat grafting: A step-by-step working algorithm to achieve 3D biological harmony in autologous breast reconstruction. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2016, 69, 1579–1587. [Google Scholar] [CrossRef]
- Fabiocchi, L.; Semprini, G.; Cattin, F.; Dellachiesa, L.; Fogacci, T.; Frisoni, G.; Samorani, D. “Reverse expansion”: A new technique of breast reconstruction with autologous tissue. J. Plast. Reconstr. Aesthetic Surg. JPRAS 2017, 70, 1537–1542. [Google Scholar] [CrossRef] [Green Version]
- Nava, M.B.; Catanuto, G.; Rocco, N. Hybrid breast reconstruction. Minerva Chir. 2018, 73, 329–333. [Google Scholar] [CrossRef]
- Calabrese, S.; Zingaretti, N.; De Francesco, F.; Riccio, M.; De Biasio, F.; Massarut, S.; Almesberger, D.; Parodi, P.C. Long-term impact of lipofilling in hybrid breast reconstruction: Retrospective analysis of two cohorts. Eur. J. Plast. Surg. 2020, 43, 257–268. [Google Scholar] [CrossRef] [Green Version]
- Simonacci, F.; Bertozzi, N.; Grieco, M.P.; Grignaffini, E.; Raposio, E. Procedure, applications, and outcomes of autologous fat grafting. Ann. Med. Surg. 2017, 20, 49–60. [Google Scholar] [CrossRef]
- Gutowski, K.A.; ASPS Fat Graft Task Force. Current applications and safety of autologous fat grafts: A report of the ASPS fat graft task force. Plast. Reconstr. Surg. 2009, 124, 272–280. [Google Scholar] [CrossRef]
- Pulagam, S.R.; Poulton, T.; Mamounas, E.P. Long-term clinical and radiologic results with autologous fat transplantation for breast augmentation: Case reports and review of the literature. Breast J. 2006, 12, 63–65. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.Y.; Lee, S.H.; Park, H.-L.; Kim, J.-Y.; Kim, S.E.; Kim, E.-K. Sonographic findings in complications of cosmetic breast augmentation with autologous fat obtained by liposuction. J. Clin. Ultrasound JCU 2004, 32, 299–301. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.Y.; Lohsiriwat, V.; Clough, K.B.; Sarfati, I.; Ihrai, T.; Rietjens, M.; Veronesi, P.; Rossetto, F.; Scevola, A.; Delay, E. The oncologic outcome and immediate surgical complications of lipofilling in breast cancer patients: A multicenter study--Milan-Paris-Lyon experience of 646 lipofilling procedures. Plast. Reconstr. Surg. 2011, 128, 341–346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abboud, M.; Vadoud-Seyedi, J.; de Mey, A.; Cukierfajn, M.; Lejour, M. Incidence of calcifications in the breast after surgical reduction and liposuction. Plast. Reconstr. Surg. 1995, 96, 620–626. [Google Scholar] [CrossRef]
- Chala, L.F.; de Barros, N.; de Camargo Moraes, P.; Endo, E.; Kim, S.J.; Pincerato, K.M.; Carvalho, F.M.; Cerri, G.G. Fat necrosis of the breast: Mammographic, sonographic, computed tomography, and magnetic resonance imaging findings. Curr. Probl. Diagn. Radiol. 2004, 33, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Pierrefeu-Lagrange, A.C.; Delay, E.; Guerin, N.; Chekaroua, K.; Delaporte, T. Radiological evaluation of breasts reconstructed with lipomodeling. Ann. Chir. Plast. Esthet. 2006, 51, 18–28. [Google Scholar] [CrossRef]
- Costantini, M.; Cipriani, A.; Belli, P.; Bufi, E.; Fubelli, R.; Visconti, G.; Salgarello, M.; Bonomo, L. Radiological findings in mammary autologous fat injections: A multi-technique evaluation. Clin Radiol. 2013, 68, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, B.S.; Werb, Z. Stromal effects on mammary gland development and breast cancer. Science 2002, 296, 1046–1049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petit, J.Y.; Botteri, E.; Lohsiriwat, V.; Rietjens, M.; De Lorenzi, F.; Garusi, C.; Rossetto, F.; Martella, S.; Manconi, A.; Bertolini, F.; et al. Locoregional recurrence risk after lipofilling in breast cancer patients. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2012, 23, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Wazir, U.; Chehade, H.e.; Headon, H.; Oteifa, M.; Kasem, A.; Mokbel, K. Oncological Safety of Lipofilling in Patients with Breast Cancer: A Meta-analysis and Update on Clinical Practice. Anticancer Res. 2016, 36, 4521–4528. [Google Scholar] [CrossRef] [Green Version]
- Kronowitz, S.J.; Mandujano, C.C.; Liu, J.; Kuerer, H.M.; Smith, B.; Garvey, P.; Jagsi, R.; Hsu, L.; Hanson, S.; Valero, V. Lipofilling of the Breast Does Not Increase the Risk of Recurrence of Breast Cancer: A Matched Controlled Study. Plast. Reconstr. Surg. 2016, 137, 385–393. [Google Scholar] [CrossRef] [Green Version]
- Aroldi, F.; Prochilo, T.; Huscher, A.; Mutti, S.; Stringhini, P.; Marchina, E.; Padolecchia, E.; Soregaroli, A.; Santus, D.; Zaniboni, A. Safety of Lipofilling in BRCA Mutated Breast Cancer Patients. J. Breast Cancer Res. Adv. 2018, 1. [Google Scholar] [CrossRef]
- Bellini, E.; Grieco, M.P.; Raposio, E. The science behind autologous fat grafting. Ann. Med. Surg. 2017, 24, 65–73. [Google Scholar] [CrossRef]
- Scheja, L.; Heeren, J. The endocrine function of adipose tissues in health and cardiometabolic disease. Nat. Rev. Endocrinol. 2019. [Google Scholar] [CrossRef]
- Lenz, M.; Arts, I.C.W.; Peeters, R.L.M.; de Kok, T.M.; Ertaylan, G. Adipose tissue in health and disease through the lens of its building blocks. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Bjørndal, B.; Burri, L.; Staalesen, V.; Skorve, J.; Berge, R.K. Different Adipose Depots: Their Role in the Development of Metabolic Syndrome and Mitochondrial Response to Hypolipidemic Agents. J. Obes. 2011, 2011. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, M.M. Subcutaneous and visceral adipose tissue: Structural and functional differences. Obes. Rev. 2010. [Google Scholar] [CrossRef]
- Hernandez, T.L.; Kittelson, J.M.; Law, C.K.; Ketch, L.L.; Stob, N.R.; Lindstrom, R.C.; Scherzinger, A.; Stamm, E.R.; Eckel, R.H. Fat Redistribution Following Suction Lipectomy: Defense of Body Fat and Patterns of Restoration. Obesity 2011, 19, 1388–1395. [Google Scholar] [CrossRef] [PubMed]
- Geer, E.B.; Shen, W. Gender differences in insulin resistance, body composition, and energy balance. Gend. Med. 2009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fried, S.K.; Lee, M.J.; Karastergiou, K. Shaping fat distribution: New insights into the molecular determinants of depot- and sex-dependent adipose biology. Obesity 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karastergiou, K.; Smith, S.R.; Greenberg, A.S.; Fried, S.K. Sex differences in human adipose tissues—The biology of pear shape. Biol. Sex Diff. 2012, 3, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogel, M.A.A.; Jocken, J.; Sell, H.; Hoebers, N.; Essers, Y.; Rouschop, K.; Cajlakovic, M.; Blaak, E.E.; Goossens, G.H. Differences in upper and lower body adipose tissue oxygen tension contribute to the adipose tissue phenotype in humans. J. Clin. Endocrinol. Metab. 2018. [Google Scholar] [CrossRef] [Green Version]
- Vogel, M.A.A.; Wang, P.; Bouwman, F.G.; Hoebers, N.; Blaak, E.E.; Renes, J.; Mariman, E.C.; Goossens, G.H. A comparison between the abdominal and femoral adipose tissue proteome of overweight and obese women. Sci. Rep. 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, J.T.; McLeod, K.; Kim, S.; Conway, S.J.; Hamilton, D.W. Periostin as a multifunctional modulator of the wound healing response. Cell Tissue Res. 2016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seguin, L.; Desgrosellier, J.S.; Weis, S.M.; Cheresh, D.A. Integrins and cancer: Regulators of cancer stemness, metastasis, and drug resistance. Trends Cell Biol. 2015. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Liu, J.; Wang, Z.; Huang, Y.; Liu, W.; Zhu, X.; Cai, Y.; Fang, X.; Lin, S.; Yuan, L.; et al. Periostin Contributes to the Acquisition of Multipotent Stem Cell-Like Properties in Human Mammary Epithelial Cells and Breast Cancer Cells. PLoS ONE 2013. [Google Scholar] [CrossRef] [Green Version]
- Christodoulides, N.; Kelly, G.A.; O’Sullivan, S.T. Breast lipofilling as a treatment for breast Hypoplasia in Becker naevus syndrome: A. case report. J. Surg. Case Rep. 2021. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International So. Cytotherapy 2013. [Google Scholar] [CrossRef] [Green Version]
- Rodeheffer, M.S.; Birsoy, K.; Friedman, J.M. Identification of white adipocyte progenitor cells in vivo. Cell 2008, 135, 240–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodbell, M. Metabolism of isolated fat cells. II. The similar effects of phospholipase C (Clostridium perfringens alpha toxin) and of insulin on glucose and amino acid metabolism. J. Biol. Chem. 1966, 241, 130–139. [Google Scholar] [CrossRef]
- Si, Z.; Wang, X.; Sun, C.; Kang, Y.; Xu, J.; Wang, X.; Hui, Y. Biomedicine & Pharmacotherapy Adipose-derived stem cells: Sources, potency, and implications for regenerative therapies. Biomed. Pharmacother. 2019, 114, 108765. [Google Scholar] [CrossRef] [PubMed]
- Suga, H.; Matsumoto, D.; Eto, H.; Inoue, K.; Aoi, N.; Kato, H.; Araki, J.; Yoshimura, K. Functional Implications of CD34 Expression in Human. Stem Cells Dev. 2009, 18, 1201–1210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Liu, Y.; Chen, Y.; Yuan, L.; Liu, H.; Wang, J.; Liu, Q.; Zhang, Y. Review Article Adipose-Derived Stem Cells: Current Applications and Future Directions in the Regeneration of Multiple Tissues. Stem Cells Int. 2020. [Google Scholar] [CrossRef]
- Bora, P.; Majumdar, A.S. Adipose tissue-derived stromal vascular fraction in regenerative medicine: A brief review on biology and translation. Stem Cell Res Ther. 2017, 1–10. [Google Scholar] [CrossRef]
- Guneta, V.; Tan, N.S.; Sugii, S.; Lim, T.C.; Wong, T.C.M.; Choong, C. Comparative study of adipose-derived stem cells from abdomen and breast. Ann. Plast. Surg. 2016. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Liu, H.; Chen, M.; Ren, S.; Cheng, P.; Zhang, H. miR-301b~miR-130b-PPARγ axis underlies the adipogenic capacity of mesenchymal stem cells with different tissue origins. Sci. Rep. 2017, 7, 1160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharath, S.S.; Ramu, J.; Nair, S.V.; Iyer, S.; Mony, U.; Rangasamy, J. Human Adipose Tissue Derivatives as a Potent Native Biomaterial for Tissue Regenerative Therapies. Tissue Eng. Regen. Med. 2020, 17, 123–140. [Google Scholar] [CrossRef]
- Almalki, S.G.; Agrawal, D.K. Key transcription factors in the differentiation of mesenchymal stem cells. Diff. Res. Biol. Divers. 2016, 92, 41–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertozzi, N.; Simonacci, F.; Grieco, M.P.; Grignaffini, E.; Raposio, E. The biological and clinical basis for the use of adipose-derived stem cells in the field of wound healing. Ann. Med. 2017, S2012, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004, 109, 1292–1298. [Google Scholar] [CrossRef]
- Cui, L.; Yin, S.; Liu, W.; Li, N.; Zhang, W.; Cao, Y. Expanded adipose-derived stem cells suppress mixed lymphocyte reaction by secretion of prostaglandin E2. Tissue Eng. 2007, 13, 1185–1195. [Google Scholar] [CrossRef]
- Wankhade, U.D.; Shen, M.; Kolhe, R.; Fulzele, S. Advances in Adipose-Derived Stem Cells Isolation, Characterization, and Application in Regenerative Tissue Engineering. Stem Cells Int. 2016, 2016, 3206807. [Google Scholar] [CrossRef] [Green Version]
- Yañez, R.; Lamana, M.L.; García-Castro, J.; Colmenero, I.; Ramírez, M.; Bueren, J.A. Adipose tissue-derived mesenchymal stem cells have in vivo immunosuppressive properties applicable for the control of the graft-versus-host disease. Stem Cells Dayt. Ohio 2006, 24, 2582–2591. [Google Scholar] [CrossRef]
- Doornaert, M.; Colle, J.; de Maere, E.; Declercq, H.; Blondeel, P. Autologous fat grafting: Latest insights. Ann. Med. Surg. 2019, 37, 47–53. [Google Scholar] [CrossRef]
- Frye, C.A.; Wu, X.; Patrick, C.W. Microvascular endothelial cells sustain preadipocyte viability under hypoxic conditions. In Vitro Cell. Dev. Biol. Anim. 2005, 41, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Zhou, Z.; Chen, Y.; Schreiber, R.; Ransom, J.T.; Fraser, J.K.; Hedrick, M.H.; Pinkernell, K.; Kuo, H.C. Supplementation of fat grafts with adipose-derived regenerative cells improves long-term graft retention. Ann. Plast. Surg. 2010, 64, 222–228. [Google Scholar] [CrossRef]
- Pérez-Cano, R.; Vranckx, J.J.; Lasso, J.M.; Calabrese, C.; Merck, B.; Milstein, A.M.; Sassoon, E.; Delay, E.; Weiler-Mithoff, E.M. Prospective trial of adipose-derived regenerative cell (ADRC)-enriched fat grafting for partial mastectomy defects: The RESTORE-2 trial. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2012, 38, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Bashir, M.M.; Sohail, M.; Ahmad, F.J.; Choudhery, M.S. Preenrichment with Adipose Tissue-Derived Stem Cells Improves Fat Graft Retention in Patients with Contour Deformities of the Face. Stem Cells Int. 2019, 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kølle, S.F.T.; Dusher, D.; Taudorf, M.; Fisher-Nielsen, A.; Svalgaard, J.D.; Munthe-Fog, L.; Jønsson, B.; Selvig, P.B.; Mamsen, F.P.; Katz, A.J. Ex vivo-expanded autologous adipose tissue-derived stromal cells ensure enhanced fat graft retention in breast augmentation: A randomized controlled clinical trial. Stem Cells Transl. Med. 2020, 9, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Estevez, L.; Moreno-Bueno, G. Updating the role of obesity and cholesterol in breast cancer. Breast Cancer Res. 2019. [Google Scholar] [CrossRef] [Green Version]
- Kothari, C.; Diorio, C.; Durocher, F. The importance of breast adipose tissue in breast cancer. Int. J. Mol. Sci. 2020, 21, 5760. [Google Scholar] [CrossRef] [PubMed]
- Scherer, P.E. The many secret lives of adipocytes: Implications for diabetes. Diabetolo 2019. [Google Scholar] [CrossRef] [Green Version]
- Corsa, C.A.S.; MacDougald, O.A. Cyclical Dedifferentiation and Redifferentiation of Mammary Adipocytes. Cell Metab. 2018, 28, 187–189. [Google Scholar] [CrossRef] [Green Version]
- Zwick, R.K.; Rudolph, M.C.; Shook, B.A.; Holtrup, B.; Roth, E.; Lei, V.; Van Keymeulen, A.; Seewaldt, V.; Kwei, S.; Wysolmerski, J.; et al. Adipocyte hypertrophy and lipid dynamics underlie mammary gland remodeling after lactation. Nat. Commun. 2018. [Google Scholar] [CrossRef] [Green Version]
- Pascual, G.; Avgustinova, A.; Mejetta, S.; Martín, M.; Castellanos, A.; Attolini, C.S.; Berenguer, A.; Prats, N.; Toll, A.; Hueto, J.A.; et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 2017. [Google Scholar] [CrossRef]
- D’Esposito, V.; Ambrosio, M.R.; Giuliano, M.; Cabaro, S.; Miele, C.; Beguinot, F.; Formisano, P. Mammary Adipose Tissue Control of Breast Cancer Progression: Impact of Obesity and Diabetes. Front. Oncol. 2020, 10, 1–8. [Google Scholar] [CrossRef]
- Kusminski, C.M.; Holland, W.L.; Sun, K.; Park, J.; Spurgin, S.B.; Lin, Y.; Askew, G.R.; Simcox, J.A.; McClain, D.A.; Li, C.; et al. MitoNEET-driven alterations in adipocyte mitochondrial activity reveal a crucial adaptive process that preserves insulin sensitivity in obesity. Nat. Med. 2012. [Google Scholar] [CrossRef] [Green Version]
- Rybinska, I.; Agresti, R.; Trapani, A.; Tagliabue, E.; Triulzi, T. Adipocytes in Breast Cancer, the Thick and the Thin. Cells 2020, 9, 560. [Google Scholar] [CrossRef] [Green Version]
- Picon-Ruiz, M.; Morata-Tarifa, C.; Valle-Goffin, J.J.; Friedman, E.R.; Slingerland, J.M. Obesity and adverse breast cancer risk and outcome: Mechanistic insights and strategies for intervention. CA Cancer J. Clin. 2017. [Google Scholar] [CrossRef]
- Borugian, M.J.; Sheps, S.B.; Kim-Sing, C.; Olivotto, I.A.; Van Patten, C.; Dunn, B.P.; Coldman, A.J.; Potter, J.D.; Gallagher, R.P.; Hislop, T.G. Waist-to-Hip Ratio and Breast Cancer Mortality. Am. J. Epidemiol. 2003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faria, S.S.; Corrêa, L.H.; Heyn, G.S.; de Sant’Ana, L.P.; Almeida, R.D.N.; Magalhães, K.G. Obesity and breast cancer: The role of crown-like structures in breast adipose tissue in tumor progression, prognosis, and therapy. J. Breast Can. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Andrade, I.; Moraes, J.; Brandão-Costa, R.M.; Vargas da Silva, S.; de Souza, A.; da Silva, C.; Renovato-Martins, M.; Barja-Fidalgo, C. Obese adipose tissue extracellular vesicles raise breast cancer cell malignancy. Endocr. Relat. Cancer 2020, 27, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.J.; Choi, M.S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kita, S.; Maeda, N.; Shimomura, I. Interorgan communication by exosomes, adipose tissue, and adiponectin in metabolic syndrome. J. Clin. Investig. 2019, 129, 4041–4049. [Google Scholar] [CrossRef] [Green Version]
- Dirat, B.; Bochet, L.; Dabek, M.; Daviaud, D.; Dauvillier, S.; Majed, B.; Wang, Y.Y.; Meulle, A.; Salles, B.; Le Gonidec, S.; et al. Cancer-associated adipocytes exhibit an activated phenotype and contribute to breast cancer invasion. Cancer Res. 2011, 71, 2455–2465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, J.; Cha, Y.J.; Koo, J.S. Adipocyte biology in breast cancer: From silent bystander to active facilitator. Prog. Lipid Res. 2018, 69, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Mao, A.S.; Seo, B.R.; Zhao, X.; Gupta, S.K.; Chen, M.; Han, Y.L.; Shih, T.Y.; Mooney, D.J.; Guo, M. Compression-induced dedifferentiation of adipocytes promotes tumor progression. Sci. Adv. 2020, 6, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Wu, Q.; Li, B.; Li, Z.; Li, J.; Sun, S.; Sun, S. Cancer-associated adipocytes: Key players in breast cancer progression. J. Hematol. Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Berg, A.H.; Combs, T.P.; Du, X.; Brownlee, M.; Scherer, P.E. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat. Med. 2001. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Shao, J. Adiponectin and lipid metabolism in skeletal muscle. Acta Pharm. Sin. B 2012. [Google Scholar] [CrossRef] [Green Version]
- Scherer, P.E.; Williams, S.; Fogliano, M.; Baldini, G.; Lodish, H.F. A novel serum protein similar to C1q, produced exclusively in adipocytes. J. Biol. Chem. 1995. [Google Scholar] [CrossRef] [Green Version]
- Naimo, G.D.; Gelsomino, L.; Catalano, S.; Mauro, L.; Andò, S. Interfering Role of ERα on Adiponectin Action in Breast Cancer. Front. Endocrinol. 2020. [Google Scholar] [CrossRef] [Green Version]
- Gelsomino, L.; Naimo, G.D.; Catalano, S.; Mauro, L.; Andò, S. The emerging role of adiponectin in female malignancies. Int. J. Mol. Sci. 2019, 20, 2127. [Google Scholar] [CrossRef] [Green Version]
- Macis, D.; Guerrieri-Gonzaga, A.; Gandini, S. Circulating adiponectin and breast cancer risk: A systematic review and meta-analysis. Int. J. Epidemiol. 2014. [Google Scholar] [CrossRef] [Green Version]
- Guven, H.E.; Dogan, L.; Gulçelik, M.A.; Gulcelik, N.E. Adiponectin: A Predictor for Breast Cancer Survival? Eur. J. Breast Health 2019, 15, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Jardé, T.; Caldefie-Chézet, F.; Damez, M.; Mishellany, F.; Perrone, D.; Penault-Llorca, F.; Guillot, J.; Vasson, M.P. Adiponectin and leptin expression in primary ductal breast cancer and in adjacent healthy epithelial and myoepithelial tissue. Histopathology 2008. [Google Scholar] [CrossRef] [PubMed]
- Takahata, C.; Miyoshi, Y.; Irahara, N.; Taguchi, T.; Tamaki, Y.; Noguchi, S. Demonstration of Adiponectin Receptors 1 and 2 mRNA expression in human breast cancer cells. Cancer Lett. 2007. [Google Scholar] [CrossRef] [PubMed]
- Panno, M.L.; Naimo, G.D.; Spina, E.; Andò, S.; Mauro, L. Different molecular signaling sustaining adiponectin action in breast cancer. Curr. Opin. Pharmacol. 2016. [Google Scholar] [CrossRef]
- Mauro, L.; Naimo, G.D.; Gelsomino, L.; Malivindi, R.; Bruno, L.; Pellegrino, M.; Tarallo, R.; Memoli, D.; Weisz, A.; Panno, M.L.; et al. Uncoupling effects of estrogen receptor a on LKB1/AMPK interaction upon adiponectin exposure in breast cancer. FASEB J. 2018, 32, 4343–4355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994. [Google Scholar] [CrossRef]
- Linares, R.L.; Benítez, J.G.S.; Reynoso, M.O.; Romero, C.G.; Sandoval-Cabrera, A. Modulation of the leptin receptors expression in breast cancer cell lines exposed to leptin and tamoxifen. Sci. Rep. 2019. [Google Scholar] [CrossRef]
- Zhao, C.; Wu, M.; Zeng, N.; Xiong, M.; Hu, W.; Lv, W.; Yi, Y.; Zhang, Q.; Wu, Y. Cancer-associated adipocytes: Emerging supporters in breast cancer. J. Exp. Clin. Cancer Res. 2020, 39, 1–17. [Google Scholar] [CrossRef]
- Zheng, Q.; Banaszak, L.; Fracci, S.; Basali, D.; Dunlap, S.M.; Hursting, S.D.; Rich, J.N.; Hjlemeland, A.B.; Vasanji, A.; Berger, N.A.; et al. Leptin receptor maintains cancer stem-like properties in triple negative breast cancer cells. Endocr. Relat. Cancer 2013. [Google Scholar] [CrossRef]
- Crean-Tate, K.K.; Reizes, O. Leptin regulation of cancer stem cells in breast and gynecologic cancer. Endocrinology 2018. [Google Scholar] [CrossRef]
- Raffaghello, L.; Dazzi, F. Classification and biology of tumour associated stromal cells. Immunol. Lett. 2015. [Google Scholar] [CrossRef] [PubMed]
- Plava, J.; Cihova, M.; Burikova, M.; Bohac, M.; Adamkov, M.; Drahosova, S.; Rusnakova, D.; Pindak, D.; Karaba, M.; Simo, J.; et al. Permanent Pro-Tumorigenic Shift in Adipose Tissue-Derived Mesenchymal Stromal Cells Induced by Breast Malignancy. Cells 2020, 9, 480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 2007. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.W.; So, C.; Yau, K.L.; Chiu, K.C.; Wang, X.; Chan, F.L.; Tsang, S.Y. Adipose-derived stem cells and cancer cells fuse to generate cancer stem cell-like cells with increased tumorigenicity. J. Cell. Physiol. 2020, 235, 6794–6807. [Google Scholar] [CrossRef] [PubMed]
- Almarzouqi, F.; Rennekampff, H.-O.; Stromps, J.-P.; Alharbi, Z.; Pallua, N. The effect of lipoaspirates vs. dissected abdominal fat on breast cancer cells in vitro. Eur. J. Med. Res. 2017, 22, 10. [Google Scholar] [CrossRef] [Green Version]
- Massa, M.; Gasparini, S.; Baldelli, I.; Scarabelli, L.; Santi, P.; Quarto, R.; Repaci, E. Interaction Between Breast Cancer Cells and Adipose Tissue Cells Derived from Fat Grafting. Aesthet. Surg. J. 2016, 36, 358–363. [Google Scholar] [CrossRef] [Green Version]
- Rowan, B.G.; Gimble, J.M.; Sheng, M.; Anbalagan, M.; Jones, R.K.; Frazier, T.P.; Asher, M.; Lacayo, E.A.; Friedlander, P.L.; Kutner, R.; et al. Human adipose tissue-derived stromal/stem cells promote migration and early metastasis of triple negative breast cancer xenografts. PLoS ONE 2014, 9, e89595. [Google Scholar] [CrossRef] [Green Version]
- Kucerova, L.; Skolekova, S.; Matuskova, M.; Bohac, M.; Kozovska, Z. Altered features and increased chemosensitivity of human breast cancer cells mediated by adipose tissue-derived mesenchymal stromal cells. BMC Cancer 2013, 13, 535. [Google Scholar] [CrossRef] [Green Version]
- Devarajan, E.; Song, Y.-H.; Krishnappa, S.; Alt, E. Epithelial-mesenchymal transition in breast cancer lines is mediated through PDGF-D released by tissue-resident stem cells. Int. J. Cancer 2012, 131, 1023–1031. [Google Scholar] [CrossRef]
- Lin, R.; Wang, S.; Zhao, R.C. Exosomes from human adipose-derived mesenchymal stem cells promote migration through Wnt signaling pathway in a breast cancer cell model. Mol. Cell. Biochem. 2013, 383, 13–20. [Google Scholar] [CrossRef]
- Eterno, V.; Zambelli, A.; Pavesi, L.; Villani, L.; Zanini, V.; Petrolo, G.; Manera, S.; Tuscano, A.; Amato, A. Adipose-derived Mesenchymal Stem Cells (ASCs) may favour breast cancer recurrence via HGF/c-Met signaling. Oncotarget 2014, 5, 613–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.-C.; Wang, W.T.; Huang, L.J.; Cheng, R.Y.; Kuo, Y.R.; Hou, M.F.; Lai, C.S.; Yu, J. Differential Response of Non-cancerous and Malignant Breast Cancer Cells to Conditioned Medium of Adipose tissue-derived Stromal Cells (ASCs). Int. J. Med. Sci. 2019, 16, 893–901. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orecchioni, S.; Gregato, G.; Martin-Padura, I.; Reggiani, F.; Braidotti, P.; Mancuso, P.; Calleri, A.; Quarna, J.; Marighetti, P.; Aldeni, C.; et al. Complementary populations of human adipose CD34+ progenitor cells promote growth, angiogenesis, and metastasis of breast cancer. Cancer Res. 2013, 73, 5880–5891. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsuji, W.; Valentin, J.E.; Marra, K.G.; Donnenberg, A.D.; Donnenberg, V.S.; Rubin, J.P. An Animal Model of Local Breast Cancer Recurrence in the Setting of Autologous Fat Grafting for Breast Reconstruction. Stem Cells Transl. Med. 2018, 7, 125–134. [Google Scholar] [CrossRef]
- Silva, M.M.A.; Kokai, L.E.; Donnenberg, V.S.; Fine, J.L.; Marra, K.G.; Donnenberg, A.D.; Neto, M.S.; Rubin, J.P. Oncologic Safety of Fat Grafting for Autologous Breast Reconstruction in an Animal Model of Residual Breast Cancer. Plast. Reconstr. Surg. 2019, 143, 103–112. [Google Scholar] [CrossRef]
- Sabol, R.A.; Bowles, A.C.; Côté, A.; Wise, R.; O’Donnell, B.; Matossian, M.D.; Hossain, F.M.; Burks, H.E.; Del Valle, L.; Miele, L.; et al. Leptin produced by obesity-altered adipose stem cells promotes metastasis but not tumorigenesis of triple-negative breast cancer in orthotopic xenograft and patient-derived xenograft models. Breast Cancer Res. 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabol, R.A.; Beighley, A.; Giacomelli, P.; Wise, R.M.; Harrison, M.; O’Donnnell, B.A.; Sullivan, B.N.; Lampenfeld, J.D.; Matossian, M.D.; Bratton, M.R.; et al. Obesity-Altered Adipose Stem Cells Promote ER⁺ Breast Cancer Metastasis through Estrogen Independent Pathways. Int. J. Mol. Sci. 2019, 20, 1419. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Li, W.; Luo, S.; Yuan, J.; Hao, L. Adipose derived stem cells promote tumor metastasis in breast Cancer cells by stem cell factor inhibition of miR20b. Cell. Signal. 2019, 62, 109350. [Google Scholar] [CrossRef]
- Stylianopoulos, T. The Solid Mechanics of Cancer and Strategies for Improved Therapy. J. Biomech. Eng. 2017, 139, 2. [Google Scholar] [CrossRef]
- Waterman, R.S.; Henkle, S.L.; Betancourt, A.M. Mesenchymal stem cell 1 (MSC1)-based therapy attenuates tumor growth whereas MSC2-treatment promotes tumor growth and metastasis. PLoS ONE 2012, 7, e45590. [Google Scholar] [CrossRef] [Green Version]
- Cordeiro, P.G. Discussion: The oncologic outcome and immediate surgical complications of lipofilling in breast cancer patients: A multicenter study--Milan-Paris-Lyon experience of 646 lipofilling procedures. Plast. Reconstr. Surg. 2011, 128, 347–348. [Google Scholar] [CrossRef]
- Fajka-Boja, R.; Szebeni, G.J.; Hunyadi-Gulyás, É.; Puskás, L.G.; Katona, R.L. Polyploid Adipose Stem Cells Shift the Balance of IGF1/IGFBP2 to Promote the Growth of Breast Cancer. Front. Oncol. 2020, 10, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fajka-Boja, R.; Marton, A.; Tóth, A.; Blazsó, P.; Tubak, V.; Bálint, B.; Nagy, I.; Hegedűs, Z.; Vizler, C.; Katona, R.L. Increased insulin-like growth factor 1 production by polyploid adipose stem cells promotes growth of breast cancer cells. BMC Cancer 2018, 18, 872. [Google Scholar] [CrossRef] [PubMed]
- Fragomeni, S.M.; Sciallis, A.; Jeruss, J.S. Molecular Subtypes and Local-Regional Control of Breast Cancer. Surg. Oncol. Clin. N. Am. 2018, 27, 95–120. [Google Scholar] [CrossRef]
- Sorrentino, L.; Regolo, L.; Scoccia, E.; Petrolo, G.; Bossi, D.; Albasini, S.; Caruso, A.; Vanna, R.; Morasso, C.; Mazzucchelli, S.; et al. Autologous fat transfer after breast cancer surgery: An exact-matching study on the long-term oncological safety. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2019, 45, 1827–1834. [Google Scholar] [CrossRef] [PubMed]
- Kamdje, A.H.N.; Etet, P.F.S.; Tagne, R.S.; Vecchio, L.; Lukong, K.E.; Krampera, M. Tumor Microenvironment Uses a Reversible Reprogramming of Mesenchymal Stromal Cells to Mediate Pro-tumorigenic Effects. Front. Cell Dev. Biol. 2020, 8, 545126. [Google Scholar] [CrossRef] [PubMed]
| Reference | Study Setting | AT Component | BC Model | Results |
|---|---|---|---|---|
| Almarzouqi et al. [135] | In vitro | Whole AT | MCF-7 cells | Increased BC proliferation rate; triggered expression of MMP1, integrin α2 |
| Massa et al. [136] | In vitro | Whole AT; in vitro differentiated adipocytes | MDA-MB-231, MCF-7, ZR-75 cells | Increased BC proliferation |
| Rowan et al. [137] | In vitro, in vivo | ASCs | MDA-MB-231 cells | Stimulated BC migration; stimulated tumor growth by ASCs from BMI 18.3 patient but not BMI 25.0 patient |
| Kucerova et al. [138] | In vitro | ASCs | SKBR3 cells | Increased BC migration, mammosphere formation, EMT, chemosensitivity |
| Devarajan et al. [139] | In vitro | ASCs | T47D, MCF-7, BT-474 cells | Induced EMT; promoted PDGF-D mediated colony formation |
| Lin et al. [140] | In vitro | ASCs | MCF7 cells | Promoted BC migration through ASCs-EVs |
| Eterno et al. [141] | In vitro, in vivo | Autologous ASCs | BC cells from human donors; MDA-MB-231, MCF-7 cells | Enhanced aggressiveness in MDA-MB-231 and in 50% of patient-derived BC |
| Wu et al. [142] | In vitro | ASCs | MDA-MB-231, MDA-MB-468, MCF-7 cells | Reduced BC viability |
| Orecchioni et al. [143] | In vitro, in vivo | Whole AT, ASCs, endothelial progenitors | MDA-MB-436, HCC1937 cells | Induced EMT; increased tumor volume and lung metastases |
| Tsuji et al. [144] | In vivo | lipograft | MDA-MB-231, BT-474 xenografts | Reduced engraftment; decreased BC proliferation |
| Silva et al. [145] | In vivo | Whole AT | MCF-7 xenograft | Reduced tumor volume and Ki67 |
| Sabol et al. [146] | In vitro, in vivo | obASCs | TNBC cell lines; TNBC patient-derived xenograft | Promoted metastasis through leptin signaling |
| Sabol et al. [147] | In vitro, in vivo | obASCs | MCF7-Y537S cells; PDX models WHIM20 (Y537S mutation) and WHIM43 (D538G mutation) | Promoted metastasis in BC with mutant ERα; in vitro obASCs promoted proliferation and migration of ER WT and ER MUT cells |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piccotti, F.; Rybinska, I.; Scoccia, E.; Morasso, C.; Ricciardi, A.; Signati, L.; Triulzi, T.; Corsi, F.; Truffi, M. Lipofilling in Breast Oncological Surgery: A Safe Opportunity or Risk for Cancer Recurrence? Int. J. Mol. Sci. 2021, 22, 3737. https://doi.org/10.3390/ijms22073737
Piccotti F, Rybinska I, Scoccia E, Morasso C, Ricciardi A, Signati L, Triulzi T, Corsi F, Truffi M. Lipofilling in Breast Oncological Surgery: A Safe Opportunity or Risk for Cancer Recurrence? International Journal of Molecular Sciences. 2021; 22(7):3737. https://doi.org/10.3390/ijms22073737
Chicago/Turabian StylePiccotti, Francesca, Ilona Rybinska, Elisabetta Scoccia, Carlo Morasso, Alessandra Ricciardi, Lorena Signati, Tiziana Triulzi, Fabio Corsi, and Marta Truffi. 2021. "Lipofilling in Breast Oncological Surgery: A Safe Opportunity or Risk for Cancer Recurrence?" International Journal of Molecular Sciences 22, no. 7: 3737. https://doi.org/10.3390/ijms22073737









