NFAT5 Is Involved in GRP-Enhanced Secretion of GLP-1 by Sodium
Abstract
1. Introduction
2. Results
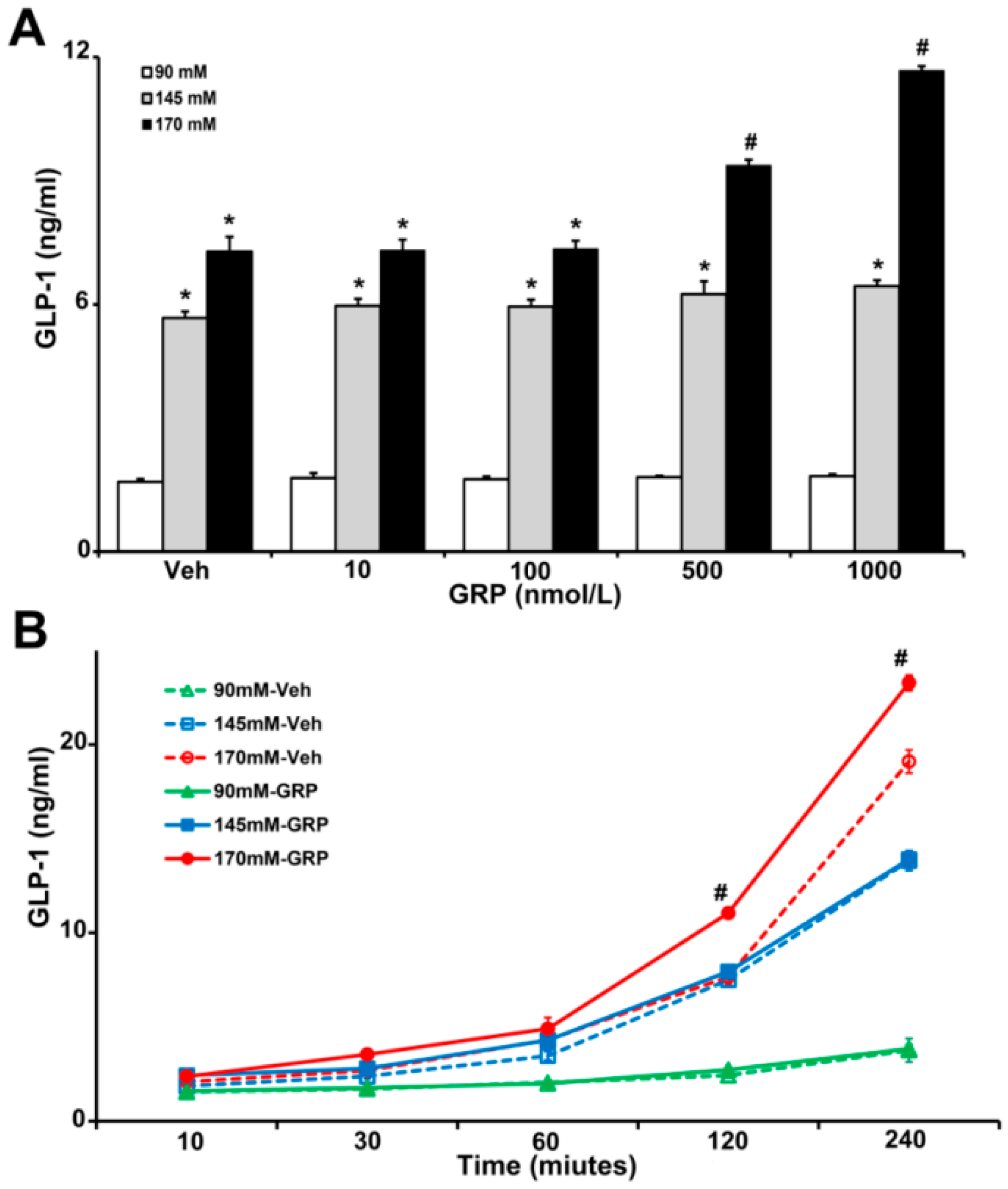
2.1. Effect of Increasing Sodium Content in the Food on GLP-1 Secretion In Vivo
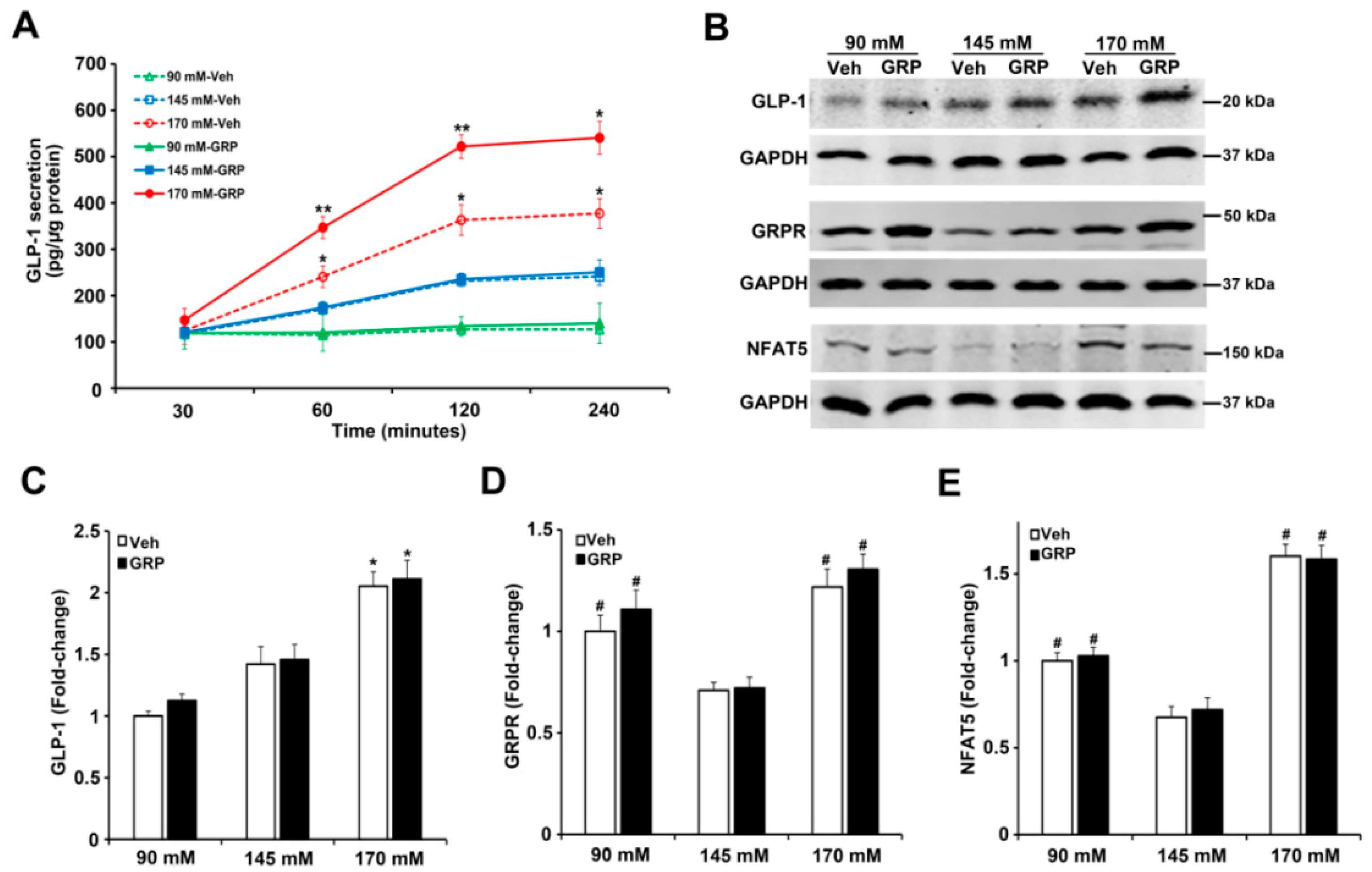
2.2. Gastrin Releasing Peptide (GRP) Enhances Sodium-Induced Increase in GLP-1 Secretion by Human L-Cells
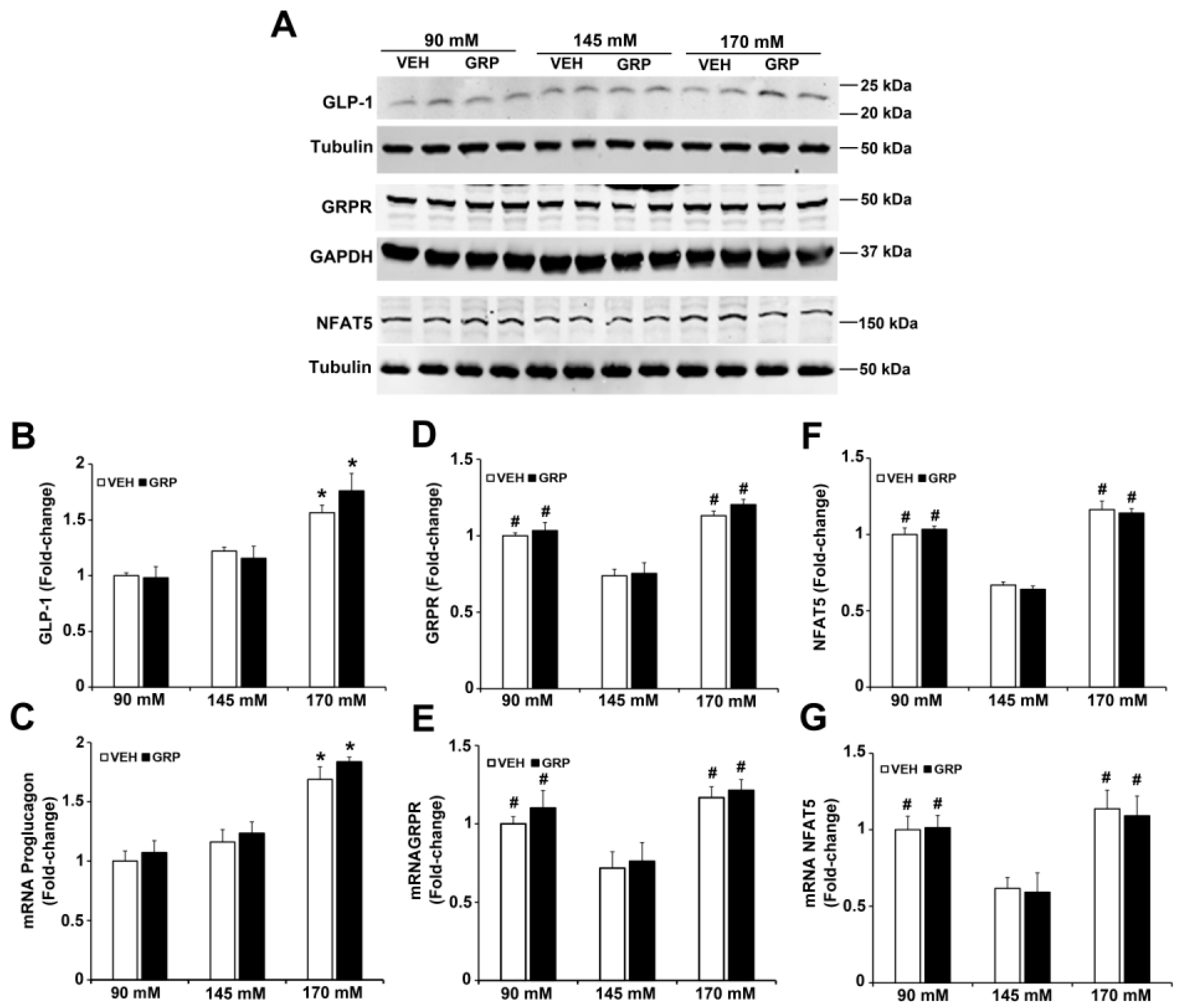
2.3. The Effect of Sodium and GRP on GLP-1 Secretion and Expressions of GRPR and NFAT5 in Mouse Ileum
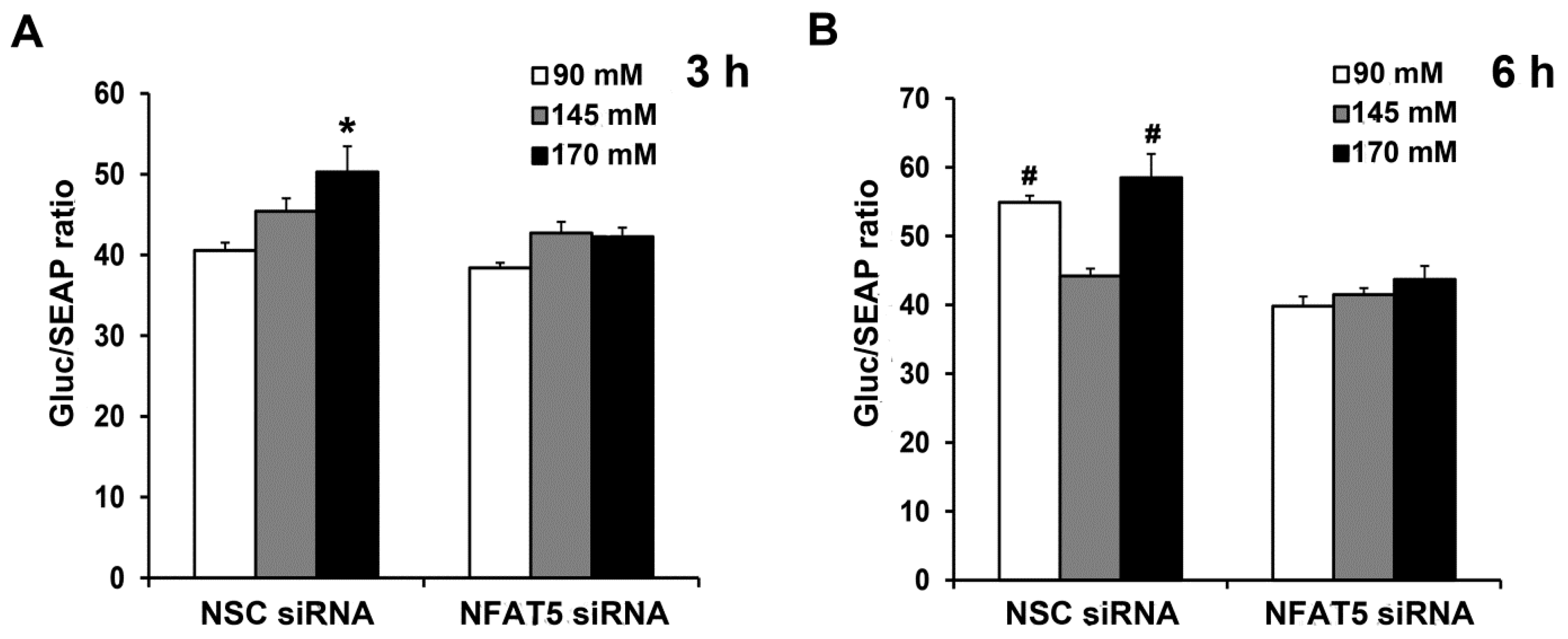
2.4. NFAT5 Is Involved in the High NaCl-Induced GLP-1 Secretion Caused by GRP
2.5. Sodium-Induced Binding of NFAT5 to GRPR Gene Promoter Increases Its Transcription and Subsequent Translation
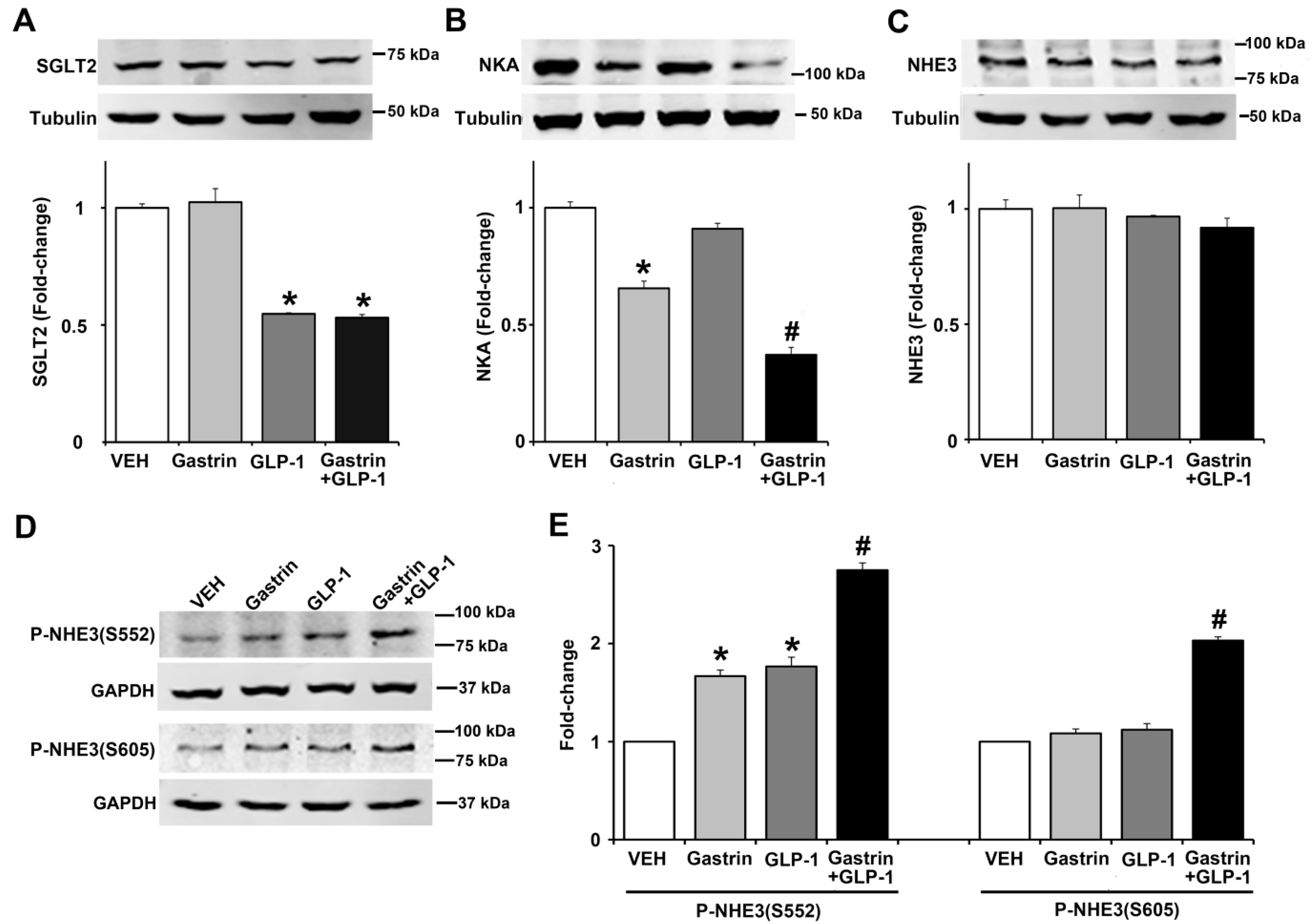
2.6. Gastrin, GLP-1, or Gastrin plus GLP-1 Negatively Regulates the Sodium Transporter/Exchanger/Pump in Human Renal Proximal Tubule Cells (hRPTCs)
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Reagents
4.2. GLP-1 Secretion
4.2.1. In Vivo Studies
4.2.2. In Vitro Studies
4.2.3. Ex Vivo Studies
4.3. Immunoblotting
4.4. RNA Extraction and cDNA Preparation
4.5. Real-Time Quantitative Polymerase Chain Reaction
4.6. siRNA Transfections and Assays
4.7. Dual Luminescence Assay (GRPR Promoter Reporter Analysis)
4.8. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kotchen, T.A.; Cowley, A.W.; Frohlich, E.D. Salt in health and disease a delicate balance. N. Engl. J. Med. 2013, 368, 1229–1237. [Google Scholar] [CrossRef]
- Kawarazaki, W.; Fujita, T. Kidney and epigenetic mechanisms of salt-sensitive hypertension. Nat. Rev. Nephrol. 2021. [Google Scholar] [CrossRef]
- Yang, J.; Jose, P.A.; Zeng, C. Gastrointestinal-renal axis: Role in the regulation of blood pressure. J. Am. Heart Assoc. 2017, 6, e00553. [Google Scholar] [CrossRef]
- Chen, Y.; Asico, L.D.; Zheng, S.; Villar, V.A.; He, D.; Zhou, L.; Zeng, C.; Jose, P.A. Gastrin and D1 dopamine receptor interact to induce natriuresis and diuresis. Hypertension 2013, 62, 927–933. [Google Scholar] [CrossRef]
- Michell, A.R.; Debnam, E.S.; Unwin, R.J. Regulation of renal function by the gastrointestinal tract: Potential role of gut-derived peptides and hormones. Annu. Rev. Physiol. 2008, 70, 379–403. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Moreno, C.; Hoagland, K.M.; Dahly, A.; Ditter, K.; Mistry, M.; Roman, R.J. Antihypertensive effect of glucagon-like peptide 1 in Dahl salt-sensitive rats. J. Hypertens. 2003, 21, 1125–1135. [Google Scholar] [CrossRef]
- Asmar, A.; Cramon, P.K.; Asmar, M.; Simonsen, L.; Sorensen, C.M.; Madsbad, S.; Moro, C.; Hartmann, B.; Rehfeld, J.F.; Holst, J.J.; et al. Increased oral sodium chloride intake in humans amplifies selectively postprandial GLP-1 but not GIP, CCK, and gastrin in plasma. Physiol. Rep. 2020, 8, e14519. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Jin, T. The incretin hormone GLP-1 and mechanisms underlying its secretion. J. Diabetes 2016, 8, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Roberge, J.N.; Gronau, K.A.; Brubaker, P.L. Gastrin-releasing peptide is a novel mediator of proximal nutrient-induced proglucagon-derived peptide secretion from the distal gut. Endocrinology 1996, 137, 2383–2388. [Google Scholar] [CrossRef]
- Persson, K.; Gingerich, R.L.; Nayak, S.; Wada, K.; Wada, E.; Ahrén, B. Reduced GLP-1 and insulin responses and glucose intolerance after gastric glucose in GRP receptor-deleted mice. Am. J. Physiol. Endocrinol. Metab. 2000, 279, E956–E962. [Google Scholar] [CrossRef]
- Natochin, Y.V.; Kutina, A.V.; Marina, A.S.; Shakhmatova, E.I. Stimulus for glucagon-like peptide-1 secretion in rats. Dokl. Biol. Sci. 2018, 479, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, S.A.; Schneider, J.A.; Lopez-Rivas, A.; Sinnett-Smith, J.W.; Rozengurt, E. Early events elicited by bombesin and structurally related peptides in quiescent Swiss 3T3 cells. II. Changes in Na+ and Ca2+ fluxes, Na+/K+ pump activity, and intracellular pH. J. Cell Biol. 1986, 102, 2223–2233. [Google Scholar] [CrossRef] [PubMed]
- Gorboulev, V.; Schürmann, A.; Vallon, V.; Kipp, H.; Jaschke, A.; Klessen, D.; Friedrich, A.; Scherneck, S.; Rieg, T.; Cunard, R.; et al. Na(+)-d-glucose cotransporter SGLT1 is pivotal for intestinal glucose absorption and glucose-dependent incretin secretion. Diabetes 2012, 61, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.L.; Chu, C.; Lv, Y.B.; Wang, Y.; Hu, J.W.; Ma, Q.; Yan, Y.; Cao, Y.M.; Dang, X.L.; Wang, K.K.; et al. Effect of salt intake on serum glucagon-like peptide-1 levels in normotensive salt-sensitive subjects. Kidney Blood Press Res. 2017, 4, 728–737. [Google Scholar] [CrossRef]
- Zhou, X.M. How do kinases contribute to tonicity-dependent regulation of the transcription factor NFAT5? World J. Nephrol. 2016, 5, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Halterman, J.A.; Kwon, H.M.; Wamhoff, B.R. Tonicity-independent regulation of the osmosensitive transcription factor TonEBP (NFAT5). Am. J. Physiol. Cell Physiol. 2012, 302, C1–C8. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, S.; Suazo, C.; Santapau, D.; Konkalmatt, P.; Cuevas, S.; Wang, D.B.; Tran, H.T.; Jose, P.A.; Felder, R.A. NFAT5 is activated by hypoxia: Role in ischemia and reperfusion in the rat kidney. PLoS ONE 2012, 7, e39665. [Google Scholar] [CrossRef]
- Xu, P.; Gildea, J.J.; Zhang, C.; Konkalmatt, P.; Cuevas, S.; Wang, D.B.; Tran, H.T.; Jose, P.A.; Felder, R.A. Stomach gastrin is regulated by sodium via PPAR-and dopamine D1 receptor. J. Mol. Endocrinol. 2020, 64, 53–65. [Google Scholar] [CrossRef]
- Reimer, R.A.; Darimont, C.; Gremlich, S.; Nicolas-Métral, V.; Rüegg, U.T.; Macé, K. A human cellular model for studying the regulation of glucagon-like peptide-1 secretion. Endocrinology 2001, 142, 4522–4528. [Google Scholar] [CrossRef]
- Qu, X.; Wang, H.; Liu, R. Recent insights into biological functions of mammalian bombesin-like peptides and their receptors. Curr. Opin. Endocrinol. Diabetes Obes. 2018, 25, 36–41. [Google Scholar] [CrossRef]
- Hampton, L.L.; Ladenheim, E.E.; Akeson, M.; Way, J.M.; Weber, H.C.; Sutliff, V.E.; Jensen, R.T.; Wine, L.J.; Arnheiter, H.; Battey, J.F. Loss of bombesin-induced feeding suppression in gastrin-releasing peptide receptor-deficient mice. Proc. Natl. Acad. Sci. USA 1998, 95, 3188–3192. [Google Scholar] [CrossRef]
- Woo, S.K.; Dahl, S.C.; Hander, J.S.; Kwon, H.M. Bidirectional regulation of tonicity-responsive enhancer binding protein in response to changes in tonicity. Am. J. Physiol. Renal. Physiol. 2000, 278, F1006–F1012. [Google Scholar] [CrossRef]
- Farah, L.X.; Valentini, V.; Pessoa, T.D.; Malnic, G.; McDonough, A.A.; Girardi, A.C. The physiological role of glucagon-like peptide-1 in the regulation of renal function. Am. J. Physiol. Renal. Physiol. 2016, 310, F123–F127. [Google Scholar] [CrossRef]
- Liu, T.; Konkalmatt, P.R.; Yang, Y.; Jose, P.A. Gastrin decreases Na, K-ATPase activity via a PI3-kinase and PKC-dependent pathway in human renal proximal tubule cells. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E565–E571. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Jose, P.A. Gastrin induces sodium-hydrogen exchanger3 phosphorylation and mTOR activation via a phosphoinositide 3-Kinase-/Protein Kinase C dependent but AKT-independent pathway in renal proximal tubule cells derived from a normotensive male human. Endocrinology 2013, 154, 865–875. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, Y.; Yang, Y.; Yang, J.; Asico, L.D.; Chen, W.; Felder, R.A.; Armando, I.; Jose, P.A.; Yang, Z. Gastrin stimulates renal dopamine production by increasing the renal tubular uptake of l-DOPA. Am. J. Physiol. Endocrinol. Metab. 2017, 312, E1–E10. [Google Scholar] [CrossRef] [PubMed]
- Puzziferri, N.; Roshek, T.B., III; Mayo, H.G.; Gallagher, R.; Belle, S.H.; Livingston, E.H. Long-term follow-up after bariatric surgery: A systematic review. JAMA 2014, 312, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Inge, T.H.; Courcoulas, A.P.; Jenkins, T.M.; Michalsky, M.P.; Helmrath, M.A.; Brandt, M.L.; Harmon, C.M.; Zeller, M.H.; Chen, M.K.; Xanthakos, S.A.; et al. Weight loss and health status 3 years after bariatric surgery in adolescents. N. Engl. J. Med. 2016, 374, 113–123. [Google Scholar] [CrossRef]
- Bueter, M.; Ashrafian, H.; Frankel, A.H.; Tam, F.W.; Unwin, R.J.; le Roux, C.W. Sodium and water handling after gastric bypass surgery in a rat model. Surg. Obes. Relat. Dis. 2011, 7, 68–73. [Google Scholar] [CrossRef]
- Grong, E.; Arbo, I.B.; Thu, O.K.; Kuhry, E.; Kulseng, B.; Mårvik, R. The effect of duodenojejunostomy and sleeve gastrectomy on type 2 diabetes mellitus and gastrin secretion in Goto-Kakizaki rats. Surg. Endosc. 2015, 29, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Meek, C.L.; Lewis, H.B.; Reimann, F.; Gribble, F.M.; Park, A.J. The effect of bariatric surgery on gastrointestinal and pancreatic peptide hormones. Peptides 2016, 77, 28–37. [Google Scholar] [CrossRef]
- Yousseif, A.; Emmanuel, J.; Karra, E.; Millet, Q.; Elkalaawy, M.; Jenkinson, A.D.; Hashemi, M.; Adamo, M.; Finer, N.; Fiennes, A.G.; et al. Differential effects of laparoscopic sleeve gastrectomy and laparoscopic gastric bypass on appetite, circulating acyl-ghrelin, peptide YY3-36 and active GLP-1 levels in non-diabetic humans. Obes. Surg. 2014, 24, 241–252. [Google Scholar] [CrossRef]
- Wheeler, S.E.; Stacey, H.M.; Nahaei, Y.; Hale, S.J.; Hardy, A.B.; Reimann, F.; Gribble, F.M.; Larraufie, P.; Gaisano, H.Y.; Brubaker, P.L. The SNARE Protein Syntaxin-1a Plays an Essential Role in Biphasic Exocytosis of the Incretin Hormone Glucagon-Like Peptide 1. Diabetes 2017, 66, 2327–2338. [Google Scholar] [CrossRef]
- Baggio, L.; Drucker, D.J. Biology of incretins: GLP-1 and GIP. Gastroenterology 2007, 132, 2131–2157. [Google Scholar] [CrossRef] [PubMed]
- Seino, Y.; Ogata, H.; Maekawa, R.; Izumoto, T.; Iida, A.; Harada, N.; Miki, T.; Seino, S.; Inagaki, N.; Tsunekawa, S.; et al. Fructose induces glucose-dependent insulinotropic polypeptide, glucagon-like peptide-1 and insulin secretion: Role of adenosine triphosphate-sensitive K(+) channels. J. Diabetes Investig. 2015, 6, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.J.; Flatt, P.R.; Deacon, C.F.; Shaw, C.; Conlon, J.M. Substance P, neurokinin A, vasoactive intestinal polypeptide and gastrin releasing peptide in the intestine and pancreas of spontaneously obese-diabetic mice. Regul. Pept. 1986, 16, 339–348. [Google Scholar] [CrossRef]
- Hernanz, A. Characterization and distribution of bombesin-like peptides in the rat brain and gastrointestinal tract. Biochem. Cell Biol. 1990, 68, 1142–1145. [Google Scholar] [CrossRef]
- Cuevas, S.; Asico, L.D.; Jose, P.A.; Konkalmatt, P. Renal hydrogen peroxide production prevents salt-sensitive hypertension. J. Am. Heart Assoc. 2020, 9, e013818. [Google Scholar] [CrossRef]
- Choi, S.Y.; Lee-Kwon, W.; Kwon, H.M. The evolving role of TonEBP as an immunometabolic stress protein. Nat. Rev. Nephrol. 2020, 16, 352–364. [Google Scholar] [CrossRef]
- Li, L.; Wu, L.; Chen, J.; Lin, X.H.; Wan, C.; Li, Q.N. Effects of sodium on rat osteoblast and the role of epithelial sodium channel. Nan Fang Yi Ke Da Xue Xue Bao 2011, 31, 1871–1874. [Google Scholar]
- von Websky, K.; Reichetzeder, C.; Hocher, B. Physiology and pathophysiology of incretins in the kidney. Curr. Opin. Nephrol. Hypertens. 2014, 23, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Skov, J.; Dejgaard, A.; Frøkiær, J.; Holst, J.J.; Jonassen, T.; Rittig, S.; Christiansen, J.S. Glucagon-like peptide-1 (GLP-1): Effect on kidney hemodynamics and renin-angiotensin-aldosterone system in healthy men. J. Clin. Endocrinol. Metab. 2013, 98, E664–E671. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, E.; Bell, D.S.H. Combination treatment of SGLT2 inhibitors and GLP-1 receptor agonists: Symbiotic effects on metabolism and cardiorenal risk. Diabetes Ther. 2018, 9, 919–926. [Google Scholar] [CrossRef]
- Sancar-Bas, S.; Gezginci-Oktayoglu, S.; Bolkent, S. Exendin-4 attenuates renal tubular injury by decreasing oxidative stress and inflammation in streptozotocin-induced diabetic mice. Growth Factors 2015, 33, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Tonneijck, L.; Marcel, H.A.; Muskiet, M.H.A.; Blijdorp, C.J.; Smits, M.M.; Twisk, J.W.; Kramer, M.H.; Danser, A.J.; Diamant, M.; Joles, J.A.; et al. Renal tubular effects of prolonged therapy with the GLP-1 receptor agonist lixisenatide in patients with type 2 diabetes mellitus. Am. J. Physiol. Renal Physiol. 2019, 316, F231–F240. [Google Scholar] [CrossRef]
- Deacon, C. What do we know about the secretion and degradation of incretin hormones? Regul. Pept. 2005, 128, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Felder, R.A.; Sanada, H.; Xu, J.; Yu, P.Y.; Wang, Z.; Watanabe, H.; Asico, L.D.; Wang, W.; Zheng, S.; Yamaguchi, I.; et al. G protein-coupled receptor kinase 4 gene variants in human essential hypertension. Proc. Natl. Acad. Sci. USA 2002, 99, 3872–3877. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.E.; Egan, J.M. Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharmacol. Ther. 2007, 113, 546–593. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, C.; Asico, L.D.; Villar, V.A.M.; Hunt, J.; Cuevas, S.; Armando, I.; Jose, P.A.; Konkalmatt, P.R. NFAT5 Is Involved in GRP-Enhanced Secretion of GLP-1 by Sodium. Int. J. Mol. Sci. 2021, 22, 3951. https://doi.org/10.3390/ijms22083951
Fan C, Asico LD, Villar VAM, Hunt J, Cuevas S, Armando I, Jose PA, Konkalmatt PR. NFAT5 Is Involved in GRP-Enhanced Secretion of GLP-1 by Sodium. International Journal of Molecular Sciences. 2021; 22(8):3951. https://doi.org/10.3390/ijms22083951
Chicago/Turabian StyleFan, Caini, Laureano D. Asico, Van Anthony M. Villar, Jessica Hunt, Santiago Cuevas, Ines Armando, Pedro A. Jose, and Prasad R. Konkalmatt. 2021. "NFAT5 Is Involved in GRP-Enhanced Secretion of GLP-1 by Sodium" International Journal of Molecular Sciences 22, no. 8: 3951. https://doi.org/10.3390/ijms22083951
APA StyleFan, C., Asico, L. D., Villar, V. A. M., Hunt, J., Cuevas, S., Armando, I., Jose, P. A., & Konkalmatt, P. R. (2021). NFAT5 Is Involved in GRP-Enhanced Secretion of GLP-1 by Sodium. International Journal of Molecular Sciences, 22(8), 3951. https://doi.org/10.3390/ijms22083951





